Carbon-Filled Organic Phase-Change Materials for Thermal Energy Storage: A Review
Abstract
1. Introduction
2. PCMs
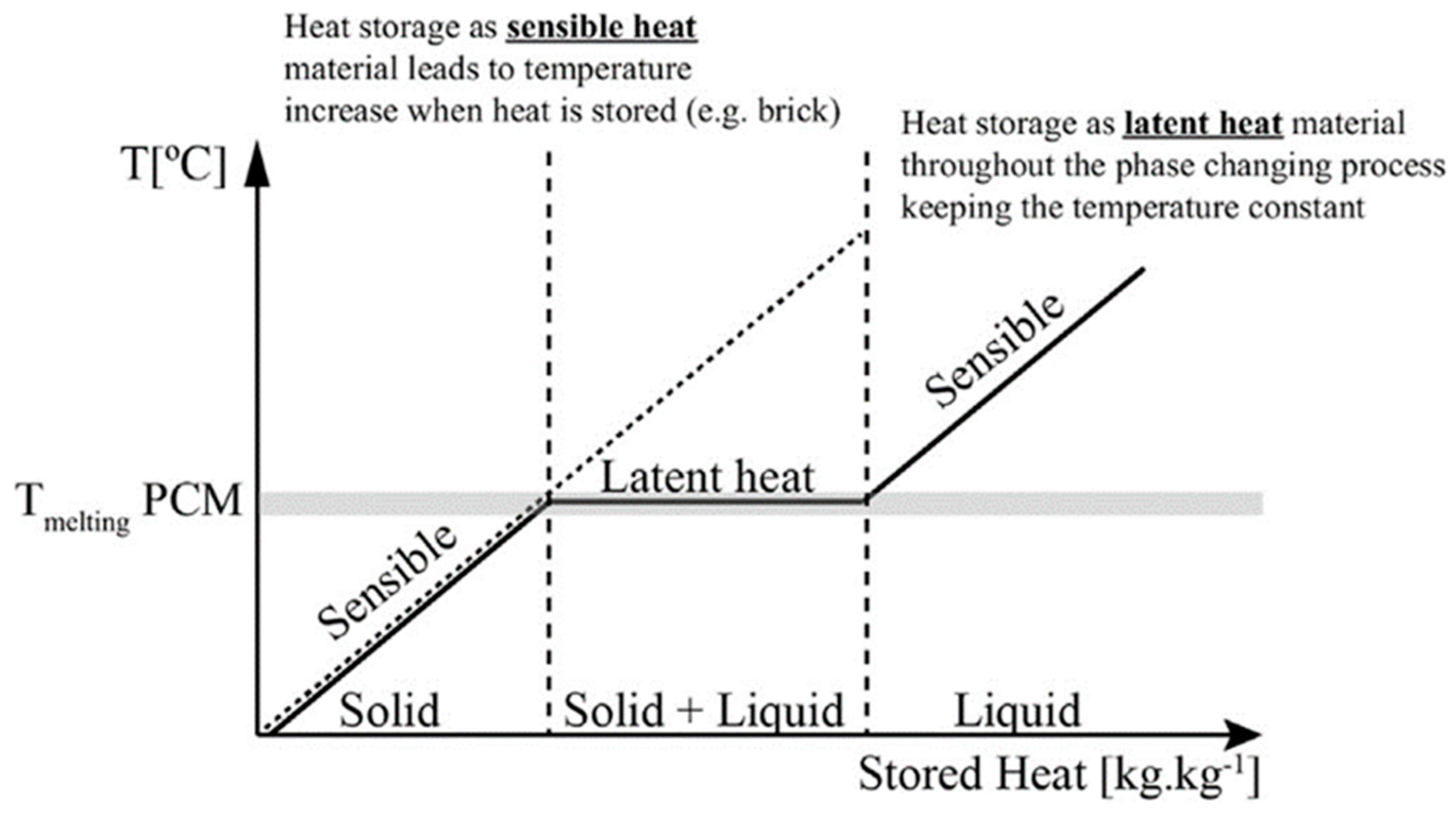
2.1. Definition of PCMs
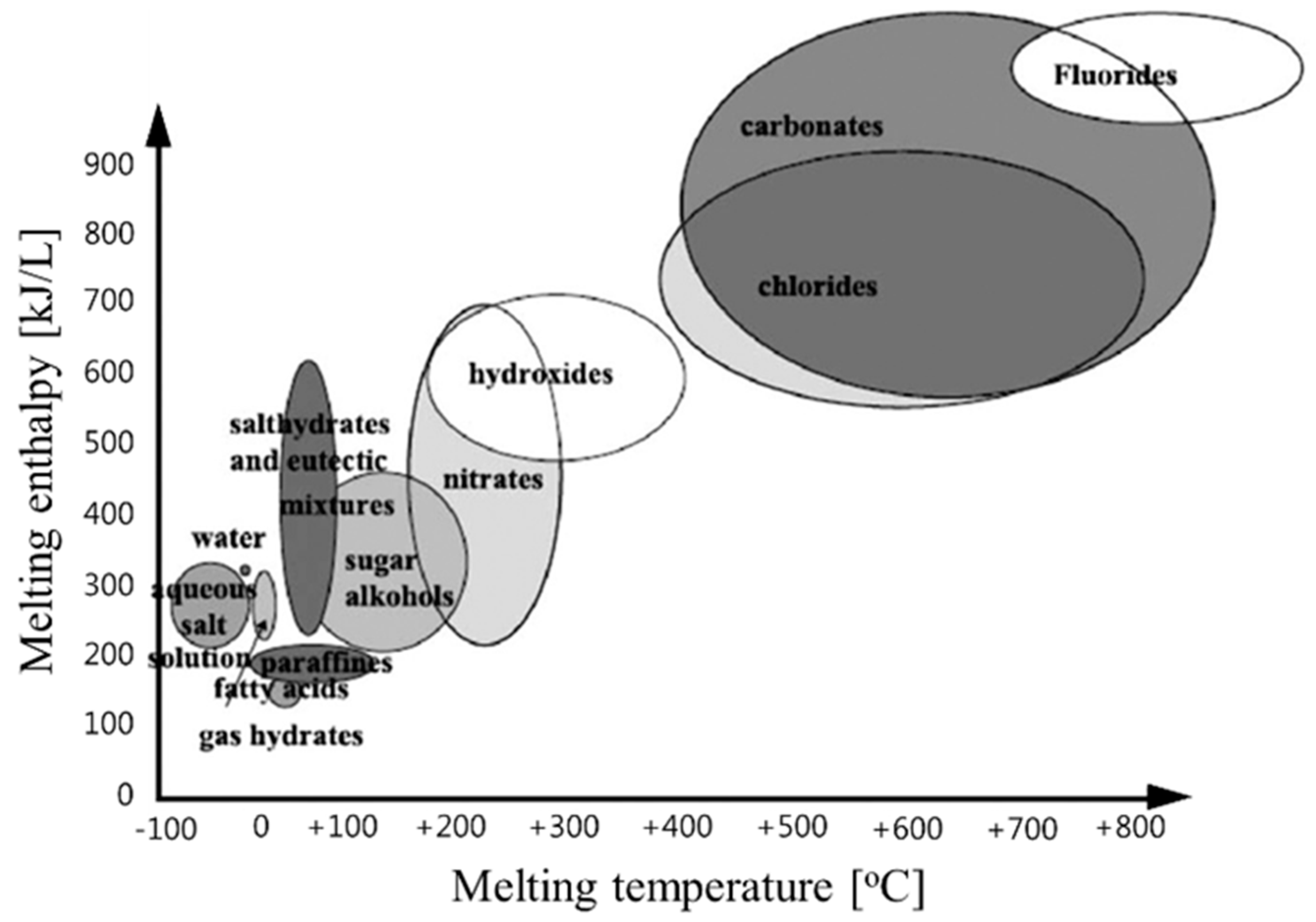
2.2. Classification of PCMs
3. Organic PCMs with Carbon Fillers
3.1. Organic PCMs
3.1.1. Paraffin
3.1.2. Fatty Acids
3.1.3. Others
3.2. Thermal Properties of Organic PCMs Filled with Carbon Materials
3.2.1. Thermal Properties of Organic PCMs Filled with Carbon Nanotubes
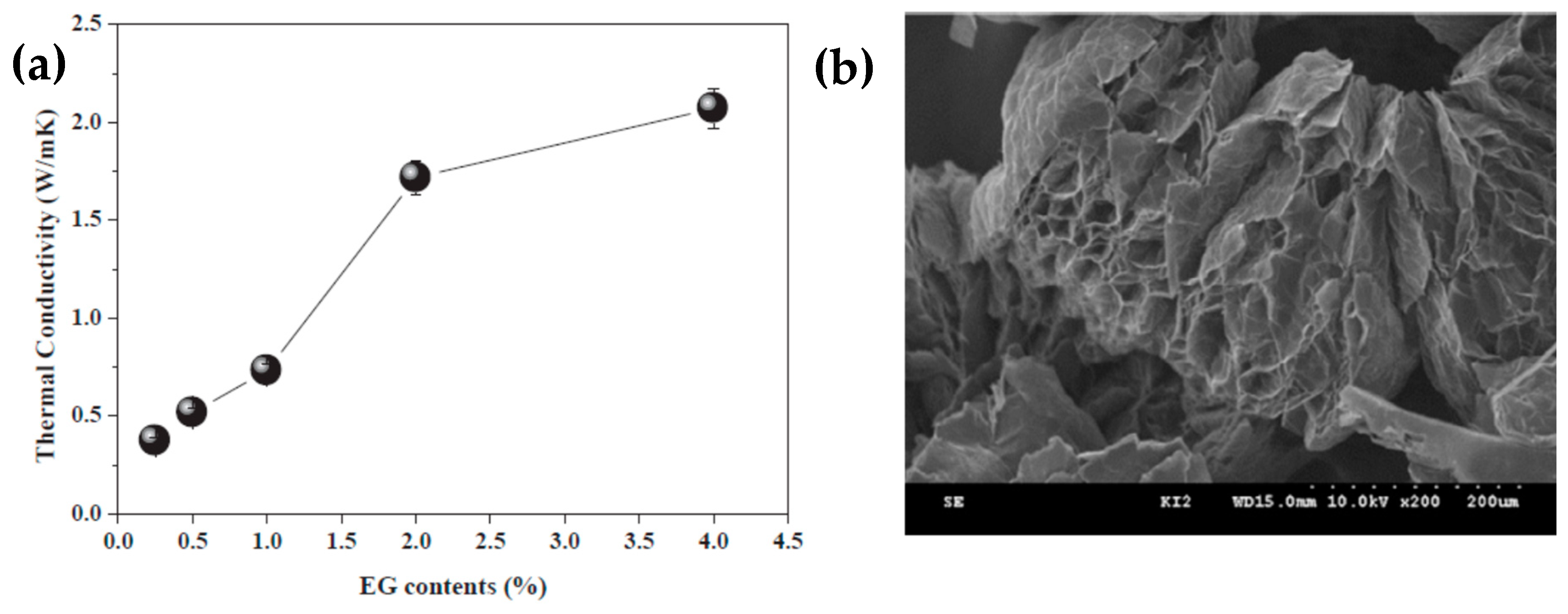
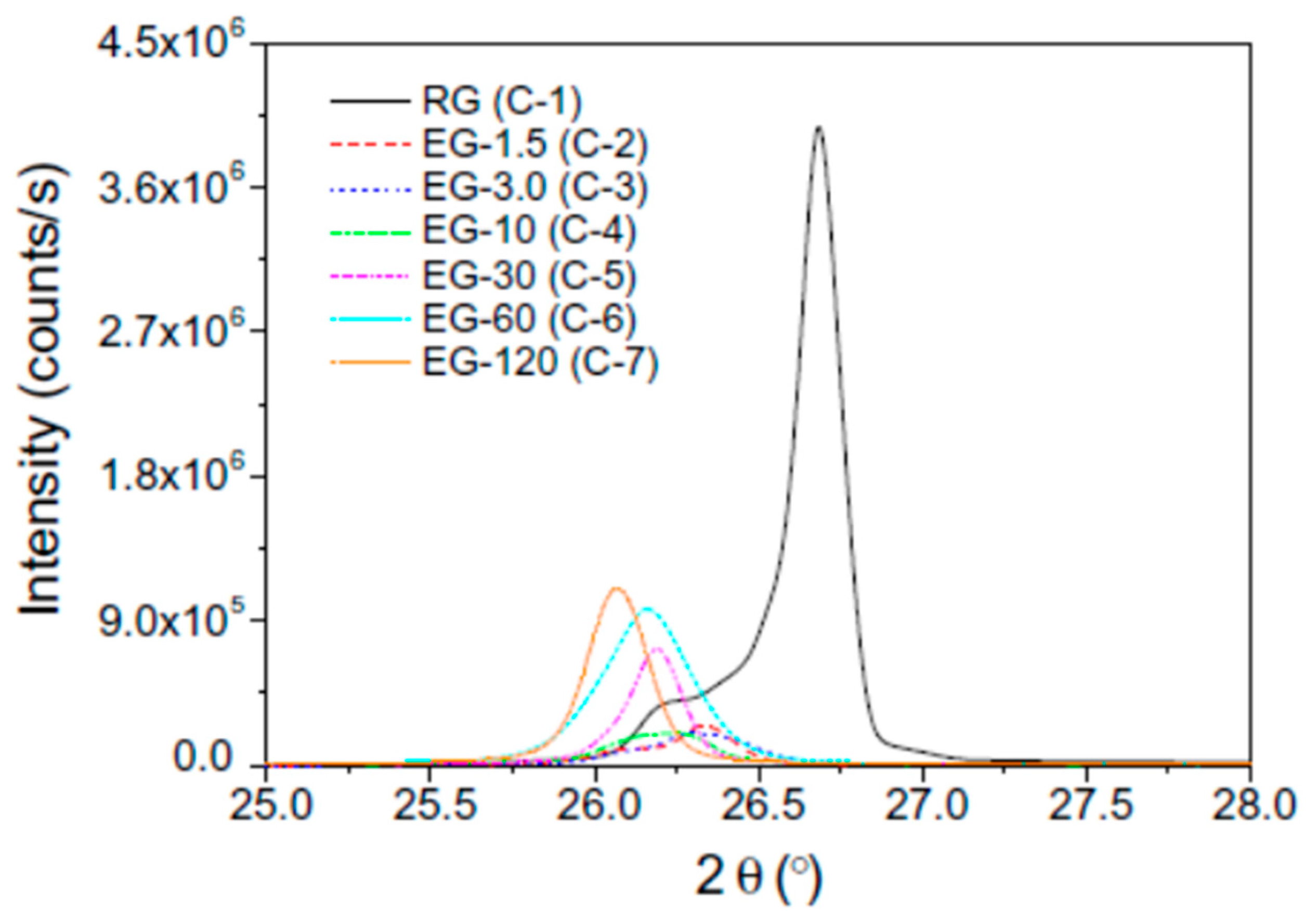
3.2.2. Thermal Properties of Organic PCMs Filled with Graphite Derivatives
3.2.3. Thermal Properties of Organic PCMs Filled with Other Carbon Materials
4. Applications of PCMs
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lin, Y.; Jia, Y.; Alva, G.; Fang, G. Review on thermal conductivity enhancement, thermal properties and applications of phase change materials in thermal energy storage. Renew. Sustain. Energy Rev. 2018, 82, 2730–2742. [Google Scholar] [CrossRef]
- Latibari, S.T.; Sadrameli, S.M. Carbon based material included-shaped stabilized phase change materials for sunlight-driven energy conversion and storage: An extensive review. Sol. Energy 2018, 170, 1130–1161. [Google Scholar] [CrossRef]
- Zhou, D.; Zhao, C.Y.; Tian, Y. Review on thermal energy storage with phase change materials (PCMs) in building applications. Appl. Energy 2012, 92, 593–605. [Google Scholar] [CrossRef]
- Oró, E.; de Gracia, A.; Castell, A.; Farid, M.M.; Cabeza, L.F. Review on phase change materials (PCMs) for cold thermal energy storage applications. Appl. Energy 2012, 99, 513–533. [Google Scholar] [CrossRef]
- Farid, M.M.; Khudhair, A.K.; Razackm, S.A.K.; Said, A.H. A review on phase change energy storage: Materials and applications. Energy Convers. Manag. 2004, 45, 1579–1615. [Google Scholar] [CrossRef]
- Jaguemont, J.; Omar, N.; Van den Bossche, P.; Mierlo, J. Phase-change materials (PCM) for automotive applications: A review. Appl. Therm. Eng. 2018, 132, 308–320. [Google Scholar] [CrossRef]
- Fan, L.; Khodadadi, J.M. Thermal conductivity enhancement of phase change materials for thermal energy storage: A review. Renew. Sustain. Energy Rev. 2011, 15, 24–46. [Google Scholar] [CrossRef]
- Hosseineizedh, S.F.; Khodadadi, J.M. Nanoparticle-enhanced phase change materials (NEPCM) with great potential for improved thermal energy storage. Int. Commun. Heat Mass Transf. 2007, 34, 345–354. [Google Scholar]
- Khodadadi, J.M.; Fan, L.; Babaei, H. Thermal conductivity enhancement of nanostructure-based colloidal suspensions utilized as phase change materials for thermal energy storage: A review. Renew. Sustain. Energy Rev. 2013, 24, 418–444. [Google Scholar] [CrossRef]
- Ettouney, H.; Alatiqi, I.; Al-Sahali, M.; Al-Hajirie, K. Heat transf. enhancement in energy storage in spherical capsules filled with paraffin wax and metal beads. Energy Convers. Manag. 2006, 47, 211–228. [Google Scholar]
- Mettawee, E.B.S.; Assassa, G.M.R. Thermal conductivity enhancement in a latent heat storage system. Sol. Energy 2007, 81, 839–845. [Google Scholar] [CrossRef]
- Molefi, J.A.; Luyt, A.S.; Krupa, I. Investigation of thermally conducting phase change materials based on polyethylene/wax blends filled with copper particles. J. Appl. Polym. Sci. 2010, 116, 1766–1774. [Google Scholar] [CrossRef]
- Zeng, J.L.; Sun, L.X.; Xu, F.; Tan, Z.C.; Zhang, Z.H.; Zhang, J.; Zhang, T. Study of a PCM based energy storage system containing Ag nanoparticles. J. Therm. Anal. Calorim. 2007, 87, 369–373. [Google Scholar] [CrossRef]
- Wu, S.; Zhu, D.; Li, X.; Li, H.; Lei, J. Thermal energy storage behavior of Al2O3-H2O nanofluids. Thermochim. Acta 2009, 483, 73–77. [Google Scholar] [CrossRef]
- Liu, Y.D.; Zhou, Y.G.; Tong, M.W.; Zhou, X.S. Experimental study of thermal diffusivity and phase change performance of nanofluids PCMs. Microfluid. Nanofluid. 2009, 7, 579–584. [Google Scholar] [CrossRef]
- Ho, C.J.; Gao, J.Y. Preparation and thermophysical properties of nanoparticle-inparaffin emulsion as phase change material. Int. Commun. Heat Mass Transf. 2009, 36, 467–470. [Google Scholar] [CrossRef]
- Wang, J.; Xie, H.; Li, Y.; Xin, Z. PW based phase change nanocomposites containing-Al2O3. J. Therm. Anal. Calorim. 2010, 102, 709–713. [Google Scholar] [CrossRef]
- Wu, S.; Zhu, D.; Zhang, X.; Huang, J. Preparation and melting/freezing characteristics of Cu/paraffin nanofluid as phase-change material (PCM). Energy Fuels 2010, 24, 1894–1898. [Google Scholar] [CrossRef]
- Harikrishnan, S.; Kalaiselvam, S. Preparation and thermal characteristics of CuO-oleic acid nanofluids as a phase change material. Thermochim. Acta 2012, 533, 46–55. [Google Scholar] [CrossRef]
- Fan, L.; Khodadadi, J.M. An experimental investigation of enhanced thermal conductivity and expedited unidirectional freezing of cyclohenxae-based nanoparticle suspensions utilized as nano-enhanced phase change materials. Int. J. Therm. Sci. 2012, 62, 120–126. [Google Scholar] [CrossRef]
- Fan, L.; Khodadadi, J.M. A theoretical and experimental investigation of unidirectional freezing of nanoparticle-enhanced phase change materials. J. Heat Transf. 2012, 134, 092301. [Google Scholar] [CrossRef]
- Jesumathy, S.; Udayakumar, M.; Suresh, S. Experimental study of enhanced heat transf. by addition of CuO nanoparticles. Heat Mass Transf. 2012, 48, 965–978. [Google Scholar] [CrossRef]
- Wu, S.Y.; Wang, H.; Xiao, S.; Zhu, D.S. An investigation of melting/freezing characteristics of nanoparticle-enhanced phase change materials. J. Therm. Anal. Calorim. 2012, 110, 1127–1231. [Google Scholar] [CrossRef]
- Zeng, J.L.; Cao, Z.; Yang, D.W.; Sun, L.X.; Zhang, L. Thermal conductivity enhancement of Ag nanowires on an organic phase change material. J. Therm. Anal. Calorim. 2010, 101, 385–389. [Google Scholar] [CrossRef]
- Zeng, J.L.; Zhu, F.R.; Yu, S.B.; Zhu, L.; Cao, Z.; Sun, L.X.; Deng, G.R.; Yan, W.P.; Zhang, L. Effects of copper nanowires on the properties of an organic phase change material. Sol. Energy Mater. Sol. Cells 2012, 105, 174–178. [Google Scholar] [CrossRef]
- Fukai, J.; Kanou, M.; Kodama, Y.; Miyatake, O. Thermal conductivity enhancement of energy storage media using carbon fibers. Energy Convers. Manag. 2000, 41, 1543–1556. [Google Scholar] [CrossRef]
- Frusteri, F.; Leonardi, V.; Vasta, S.; Restuccia, G. Thermal conductivity measurement of a PCM based storage system containing carbon fibers. Appl. Therm. Eng. 2005, 25, 1623–1633. [Google Scholar] [CrossRef]
- Zhang, Z.; Fang, X. Study on paraffin/expanded graphite composite phase change thermal energy storage material. Energy Convers. Manag. 2006, 47, 303–310. [Google Scholar] [CrossRef]
- Sarı, A.; Karaipekli, A. Thermal conductivity and latent heat thermal energy storage characteristics of paraffin/expanded graphite composite as phase change material. Appl. Therm. Eng. 2007, 27, 1271–1277. [Google Scholar] [CrossRef]
- Sarı, A.; Karaipekli, A. Preparation, thermal properties and thermal reliability of palmitic acid/expanded graphite composite as form-stable PCM for thermal energy storage. Sol. Energy Mater. Sol. Cells 2009, 93, 571–576. [Google Scholar] [CrossRef]
- Xia, L.; Zhang, P.; Wang, R.Z. Preparation and thermal characterization of expanded graphite/paraffin composite phase change material. Carbon 2010, 48, 2538–2548. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, N.; Peng, J.; Fang, X.; Gao, X.; Fang, Y. Preparation and thermal energy storage properties of paraffin/expanded graphite composite phase change material. Appl. Energy 2012, 91, 426–431. [Google Scholar] [CrossRef]
- Li, M.; Wu, Z.; Tan, J. Properties of form-stable paraffin/silicon dioxide/expanded graphite phase change composites prepared by sol-gel method. Appl. Energy 2012, 92, 456–461. [Google Scholar] [CrossRef]
- Elgafy, A.; Lafdi, K. Effect of carbon nanofiber additives on thermal behavior phase change materials. Carbon 2005, 43, 3067–3074. [Google Scholar] [CrossRef]
- Shaikh, S.; Lafdi, K.; Hallinan, K. Carbon nanoadditives to enhance latent energy storage of phase change materials. J. Appl. Phys. 2008, 103, 094302. [Google Scholar] [CrossRef]
- Sanusi, O.; Warzoha, R.; Fleischer, A.S. Energy storage and solidification of paraffin phase change material embedded with graphite nanofibers. Int. J. Heat Mass Transf. 2011, 54, 4429–4436. [Google Scholar] [CrossRef]
- Li, M.; Mu, B. Effect of different dimensional carbon materials on the properties and application of phase change materials: A review. Appl. Energy 2019, 242, 695–715. [Google Scholar] [CrossRef]
- Zeng, J.L.; Liu, Y.Y.; Cao, Z.X.; Zhang, J.; Zhang, Z.H.; Sun, L.X.; Xu, F. Thermal conductivity enhancement of MWNTs on the PANI/tetradecanol form-stable PCM. J. Therm. Anal. Calorim. 2008, 91, 443–446. [Google Scholar] [CrossRef]
- Wang, J.; Xie, H.; Xin, Z. Thermal properties of heat storage composites containing multiwalled carbon nanotubes. J. Appl. Phys. 2008, 104, 113537. [Google Scholar] [CrossRef]
- Wang, J.; Xie, H.; Xin, Z. Thermal properties of paraffin based composites containing multi-walled carbon nanotubes. Thermochim. Acta 2009, 488, 39–42. [Google Scholar] [CrossRef]
- Zeng, J.L.; Cao, Z.; Yang, D.W.; Xu, F.; Sun, L.X.; Zhang, X.F.; Zhang, L. Effects of MWNTs on phase change enthalpy and thermal conductivity of a solid-liquid organic PCM. J. Therm. Anal. Calorim. 2009, 95, 507–512. [Google Scholar] [CrossRef]
- Wang, J.; Xie, H.; Xin, Z.; Li, Y.; Chen, L. Enhancing thermal conductivity of palmitic acid base phase change materials with carbon nanotubes as fillers. Sol. Energy 2010, 84, 339–344. [Google Scholar] [CrossRef]
- Wang, J.; Xie, H.; Zhong, X.; Yang, L. Increasing the thermal conductivity of palmitic acid by the addition of carbon nanotubes. Carbon 2010, 48, 3979–3986. [Google Scholar] [CrossRef]
- Bayramoglu, E.Ç. Thermal properties and stability of n-octadecane based composites containing multiwalled carbon nanotubes. Polym. Compos. 2011, 32, 904–909. [Google Scholar] [CrossRef]
- Kumaresan, V.; Velraj, R.; Das, S.K. The effect of carbon nanotubes in enhancing the thermal transport properties of PCM during solidification. Heat Mass Transf. 2012, 48, 1345–1355. [Google Scholar] [CrossRef]
- Teng, T.P.; Cheng, C.M.; Cheng, C.P. Performance assessment of heat storage by phase change materials containing MWCNTs and graphite. Appl. Therm. Eng. 2013, 50, 637–644. [Google Scholar] [CrossRef]
- Kim, S.; Drzal, L.T. High latent heat storage and high conductive phase change materials using exfoliated graphite nanoplatelets. Sol. Energy Mater. Sol. Cells 2009, 93, 136–142. [Google Scholar] [CrossRef]
- Yavari, F.; Fard, H.R.; Pashayi, K.; Rafiee, M.A.; Zamiri, A.; Yu, Z.; Ozisik, R.; Tasciuc, T.; Koratkar, N. Enhanced thermal conductivity in a nanostructured phase change composite due to low concentration graphene additives. J. Phys. Chem. C 2011, 15, 8753–8758. [Google Scholar] [CrossRef]
- Xia, Y.; Zhang, H.; Huang, P. Graphene-oxide-induced lamellar structures used to fabricate novel composite solid-solid phase change materials for thermal energy storage. Chem. Eng. J. 2019, 362, 909–920. [Google Scholar] [CrossRef]
- Xue, F.; Lu, Y.; Qi, X. Melamine foam-templated graphene nanoplatelet framework toward phase change materials with multiple energy conversion abilities. Chem. Eng. J. 2019, 365, 20–29. [Google Scholar] [CrossRef]
- Chen, Y.J.; Nguyen, D.D.; Shen, M.Y.; Yip, M.C.; Tai, N.H. Thermal characterizations of the graphite nanosheets reinforced paraffin phase-change composites. Compos. A Appl. Sci. 2013, 44, 40–46. [Google Scholar] [CrossRef]
- Li, M. A nano-graphite/paraffin phase change material with high thermal conductivity. Appl. Energy 2013, 106, 25–30. [Google Scholar] [CrossRef]
- Yu, Z.T.; Fang, X.; Fan, L.W.; Wang, X.; Xiao, Y.Q.; Zeng, Y.; Xu, X.; Hu, Y.C.; Cen, K.F. Increased thermal conductivity of liquid paraffin-based suspensions in the presence of carbon nano-additives of various sizes and shapes. Carbon 2013, 53, 277–285. [Google Scholar] [CrossRef]
- Mittal, G.; Dhand, V.; Rhee, K.Y.; Park, S.J.; Lee, W.R. A review on carbon nanotubes and graphene as fillers in reinforced polymer nanocomposites. J. Ind. Eng. Chem. 2015, 21, 11–25. [Google Scholar] [CrossRef]
- Kim, H.S.; Jang, J.U.; Yu, J.; Kim, S.Y. Thermal conductivity of polymer composites based on the length of multi-walled carbon nanotubes. Compos. B Eng. 2015, 79, 505–512. [Google Scholar] [CrossRef]
- Yao, S.S.; Jin, F.L.; Rhee, K.Y.; Hui, D.; Park, S.J. Recent advances in carbon-fiber-reinforced thermoplastic composites: A review. Compos. B Eng. 2018, 142, 241–250. [Google Scholar] [CrossRef]
- Yang, G.; Park, M.; Park, S.J. Recent progresses of fabrication and characterization of fibers-reinforced composites: A review. Compos. Commun. 2019, 14, 34–42. [Google Scholar] [CrossRef]
- Jin, F.L.; Ma, C.J.; Park, S.J. Thermal and mechanical interfacial properties of epoxy composites based on functionalized carbon nanotubes. Mater. Sci. Eng. A 2011, 528, 8517–8522. [Google Scholar] [CrossRef]
- Shim, H.B.; Seo, M.K.; Park, S.J. Thermal conductivity and mechanical properties of various cross-section types carbon fiber-reinforced composites. J. Mater. Sci. 2002, 37, 1881–1885. [Google Scholar] [CrossRef]
- Fleming, E.; Du, F.; Ou, E.; Dai, L.; Shi, L. Thermal conductivity of carbon nanotubes grown by catalyst-free chemical vapor deposition in nanopores. Carbon 2019, 145, 195–200. [Google Scholar] [CrossRef]
- Ismail, K.A.R.; Henrí quez, J.R. Thermally effective windows with moving phase change material curtains. Appl. Therm. Eng. 2001, 21, 1909–1923. [Google Scholar] [CrossRef]
- Zhu, N.; Ma, Z.; Wang, S. Dynamic characteristics and energy performance of buildings using phase change materials: A review. Energy Convers. Manag. 2009, 50, 3169–3181. [Google Scholar] [CrossRef]
- Mehling, H.; Cabeza, L.F. Heat and Cold Storage with PCM: An up to Date Introduction into Basics and Applications; Springer: Berlin/Heidelberg, Germany, 2008. [Google Scholar]
- Sharma, S.D.; Kitano, H.; Sagara, K. Phase change materials for low temperature solar thermal applications. Res. Rep. Fac. Eng. Mie Univ. 2004, 29, 31–64. [Google Scholar]
- Isa, M.H.M.; Zhao, X.; Yoshino, H. Preliminary study of passive cooling strategy using a combination of PCM and copper foam to increase thermal heat storage in building facade. Sustainability 2010, 2, 2365–2381. [Google Scholar] [CrossRef]
- Jelle, B.P. Traditional, state-of-the-art and future thermal building insulation materials and solutions-properties, requirements and possibilities. Energy Build. 2011, 43, 2549–2563. [Google Scholar] [CrossRef]
- Cabeza, L.F.; Castell, A.; Barreneche, C.; de Gracia, A.; Fernández, A.I. Materials used as PCM in thermal energy storage in buildings: A review. Renew. Sustain. Energy Rev. 2011, 15, 1675–1695. [Google Scholar] [CrossRef]
- Baetens, R.; Jelle, B.P.; Gustavsen, A. Phase change materials for building applications: A state-of-the-art review. Energy Build. 2010, 42, 1361–1368. [Google Scholar] [CrossRef]
- Tyagi, V.V.; Buddhi, D. PCM thermal storage in buildings: A state of art. Renew. Sustain. Energy Rev. 2007, 11, 1146–1166. [Google Scholar] [CrossRef]
- Sharma, A.; Tyagi, V.V.; Chen, C.R.; Buddhi, D. Review on thermal energy storage with phase change materials and applications. Renew. Sustain. Energy Rev. 2009, 13, 318–345. [Google Scholar] [CrossRef]
- Kapsalis, V.; Karamanis, D. Solar thermal energy storage and heat pumps with phase change materials. Appl. Therm. Eng. 2016, 99, 1212–1224. [Google Scholar] [CrossRef]
- Yuan, Y.; Zhang, N.; Tao, W.; Cao, X.; He, Y. Fatty acids as phase change materials: A review. Renew. Sustain. Energy Rev. 2014, 29, 482–498. [Google Scholar] [CrossRef]
- Rathod, M.K.; Banerjee, J. Thermal stability of phase change materials used in latent heat energy storage systems: A review. Renew. Sustain. Energy Rev. 2013, 18, 246–258. [Google Scholar] [CrossRef]
- Tatsidjodoung, P.; Le Pierrès, N.; Luo, L. A review of potential materials for thermal energy storage in building applications. Renew. Sustain. Energy Rev. 2013, 18, 327–349. [Google Scholar] [CrossRef]
- Silva, T.; Vicente, R.; Rodrigues, F. Literature review on the use of phase change materials in glazing and shading solutions. Renew. Sustain. Energy Rev. 2016, 53, 515–535. [Google Scholar] [CrossRef]
- Kenisarin, M.; Mahkamov, K. Solar energy storage using phase change materials. Renew. Sustain. Energy Rev. 2007, 11, 1913–1965. [Google Scholar] [CrossRef]
- Nkwetta, D.N.; Haghighat, F. Thermal energy storage with phase change material-A state-of-the art review. Sustain. Cities Soc. 2014, 10, 87–100. [Google Scholar] [CrossRef]
- Narayanan, S.S.; Kardam, A.; Kumar, V.; Bhardwaj, N.; Madhwal, D.; Shukla, P.; Kumar, A.; Verma, A.; Jain, V.K. Development of sunlight-driven eutectic phase change material nanocomposite for applications in solar water heating. Resour. Effic. Technol. 2017, 3, 272–279. [Google Scholar] [CrossRef]
- Fang, G.; Tang, F.; Cao, L. Preparation, thermal properties and applications of shape-stabilized thermal energy storage materials. Renew. Sustain. Energy Rev. 2014, 40, 237–259. [Google Scholar] [CrossRef]
- Hong, H.; Kim, S.K.; Kim, Y.S. Accuracy improvement of T-history method for measuring heat of fusion of various materials. Int. J. Refrig. 2004, 27, 360–366. [Google Scholar] [CrossRef]
- El-Bassuoni, A.M.; Tayeb, A.M.; Helwa, N.H.; Fathy, A.M. Modification of urea-sodium acetate trihydrate mixture for solar energy storage. Renew. Energy 2003, 28, 1629–1643. [Google Scholar] [CrossRef]
- Kaizawa, A.; Maruoka, N.; Kawai, A.; Kamano, H.; Jozuka, T.; Senda, T.; Akiyama, T. Thermophysical and heat transf. properties of phase change material candidate for waste heat transportation system. Heat Mass Transf. 2008, 44, 763–769. [Google Scholar] [CrossRef]
- Zhao, L.; Xing, Y.; Liu, X.; Luo, Y. Thermal performance of sodium acetate trihydrate based composite phase change material for thermal energy storage. Appl. Therm. Eng. 2018, 143, 172–181. [Google Scholar] [CrossRef]
- Canik, G.; Alkan, C. Hexamethylene dilauroyl, dimyristoyl, and dipalmytoyl amides as phase change materials for thermal energy storage. Sol. Energy 2010, 84, 666–672. [Google Scholar] [CrossRef]
- Alkan, C.; Canik, G.; Dunya, H.; Sari, A. Synthesis and thermal energy storage properties of ethylene dilauroyl, dimyristoyl, and dipalmitoyl amides as novel solid-liquid phase change materials. Sol. Energy Mater. Sol. Cells 2011, 95, 1203–1207. [Google Scholar] [CrossRef]
- Kenar, J.A. Latent heat characteristics of biobased oleochemical carbonates as potential phase change materials. Sol. Energy Mater. Sol. Cells 2010, 94, 1697–1703. [Google Scholar] [CrossRef]
- Cui, Y.; Liu, C.; Hu, S.; Yu, X. The experimental exploration of carbon nanofiber and carbon nanotube additives on thermal behavior of phase change materials. Sol. Energy Mater. Sol. Cells 2011, 95, 1208–1212. [Google Scholar] [CrossRef]
- Tang, Q.; Sun, J.; Yu, S.; Wang, G. Improving thermal conductivity and decreasing supercooling of paraffin phase change materials by n-octadecylamine-functionalized multi-walled carbon nanotubes. RSC Adv. 2014, 4, 36584–38590. [Google Scholar] [CrossRef]
- Fan, L.W.; Fang, X.; Wang, X.; Zeng, Y.; Xiao, Y.Q.; Yu, Z.T.; Xu, X.; Hu, Y.C.; Cen, K.F. Effects of various carbon nanofillers on the thermal conductivity and energy storage properties of paraffin-based nanocomposite phase change materials. Appl. Energy 2013, 110, 163–172. [Google Scholar] [CrossRef]
- Xu, B.; Wang, B.; Zhang, C.; Zhou, J. Synthesis and light-heat conversion performance of hybrid particles decorated MWCNTs/paraffin phase change materials. Thermochim. Acta 2017, 652, 77–84. [Google Scholar] [CrossRef]
- Zhang, N.; Yuan, Y.; Yuan, Y.; Cao, X.; Yang, X. Effect of carbon nanotubes on the thermal behavior of palmitic-stearic acid eutectic mixtures as phase change materials for energy storage. Sol. Energy 2014, 110, 64–70. [Google Scholar] [CrossRef]
- Li, B.; Nie, S.; Hao, Y.; Liu, T.; Zhu, J.; Yan, S. Stearic-acid/carbon-nanotube composites with tailored shape-stabilized phase transitions and light-heat conversion for thermal energy storage. Energy Convers. Manag. 2015, 98, 314–321. [Google Scholar] [CrossRef]
- Tang, B.; Wei, H.; Zhao, D.; Zhang, S. Light-heat conversion and thermal conductivity enhancement of PEG/SiO2 composite PCM by in situ Ti4O7 doping. Sol. Energy Mater. Sol. Cells 2017, 161, 183–189. [Google Scholar] [CrossRef]
- Shi, J.N.; Ger, M.D.; Liu, Y.M.; Fan, Y.C.; Wen, N.T.; Lin, C.K.; Pu, N.W. Improving the thermal conductivity and shape-stabilization of phase change materials using nanographite additives. Carbon 2013, 51, 365–372. [Google Scholar] [CrossRef]
- Xiang, J.; Drzal, L.T. Investigation of exfoliated graphite nanoplatelets (xGnP) in improving thermal conductivity of paraffin wax-based phase change material. Sol. Energy Mater. Sol. Cells 2011, 95, 1811–1818. [Google Scholar] [CrossRef]
- Wang, S.; Qin, P.; Fang, X.; Zhang, Z.; Wang, S.; Liu, X. A novel sebacic acid/expanded graphite composite phase change material for solar thermal mediumtemperature applications. Sol. Energy 2014, 99, 283–290. [Google Scholar] [CrossRef]
- Zhou, S.; Zhang, X.; Liu, S.; Li, Y.; Xu, X. Performance study on expand graphite/organic composite phase change material for cold thermal energy storage. Energy Procedia 2019, 158, 5305–5310. [Google Scholar]
- Zeng, J.L.; Zheng, S.H.; Yu, S.B.; Zhu, F.R.; Gan, J.; Zhu, L.; Xiao, Z.L.; Zhu, X.Y.; Zhu, Z.; Sun, L.X.; et al. Preparation and thermal properties of palmitic acid/polyaniline/exfoliated graphite nanoplatelets form-stable phase change materials. Appl. Energy 2014, 115, 603–609. [Google Scholar] [CrossRef]
- Shin, H.K.; Rhee, K.Y.; Park, S.J. Effects of exfoliated graphite on the thermal properties of erythritol-based composites used as phase-change materials. Compos. B Eng. 2016, 96, 350–353. [Google Scholar] [CrossRef]
- Lee, S.Y.; Shin, H.K.; Park, M.; Rhee, K.Y.; Park, S.J. Thermal characterization of erythritol/expanded graphite composites for high thermal storage capacity. Carbon 2014, 68, 67–72. [Google Scholar] [CrossRef]
- Mehrali, M.; Latibari, S.T.; Mehrali, M.; Mahlia, T.M.I.; Metselaar, H.S.C.; Naghavi, M.S.; Sadeghinezhad, E.; Akhiani, A.R. Preparation and characterization of palmitic acid/graphene nanoplatelets composite with remarkable thermal conductivity as a novel shape-stabilized phase change material. Appl. Therm. Eng. 2013, 61, 633–640. [Google Scholar] [CrossRef]
- Li, B.; Liu, T.; Hu, L.; Wang, Y.; Nie, S. Facile preparation and adjustable thermal property of stearic acid-graphene oxide composite as shape-stabilized phase change material. Chem. Eng. J. 2013, 215, 819–826. [Google Scholar] [CrossRef]
- Mehrali, M.; Latibari, S.T.; Mehrali, M.; Mahlia, T.M.I.; Sadeghinezhad, E.; Metselaar, H.S.C. Preparation of nitrogen-doped graphene/palmitic acid shape stabilized composite phase change material with remarkable thermal properties for thermal energy storage. Appl. Energy 2014, 135, 339–349. [Google Scholar] [CrossRef]
- Li, Y.; Samad, Y.A.; Polychronopoulou, K.; Alhassan, S.M.; Liao, K. From biomass to high performance solar-thermal and electric-thermal energy conversion and storage materials. J. Mater. Chem. A 2014, 2, 7759–7765. [Google Scholar] [CrossRef]
- Zhang, S.; Wang, S.; Zhang, J.; Jiang, Y.; Ji, Q.; Zhang, Z.; Wang, Z. Increasing phase change latent heat of stearic acid via nanocapsule interface confinement. J. Phys. Chem. C 2013, 117, 23412–23417. [Google Scholar] [CrossRef]
- Zhang, L.; Li, R.; Tang, B.; Wang, P. Solar-thermal conversion and thermal energy storage of graphene foam-based composites. Nanoscale 2016, 8, 14600–14607. [Google Scholar] [CrossRef]
- Chen, Z.; Shan, F.; Cao, L.; Fang, G. Synthesis and thermal properties of shapestabilized lauric acid/activated carbon composites as phase change materials for thermal energy storage. Sol. Energy Mater. Sol. Cells 2012, 102, 131–136. [Google Scholar] [CrossRef]
- Huang, X.; Liu, Z.; Xia, W.; Zou, R.; Han, R.P.S. Alkylated phase change composites for thermal energy storage based on surface-modified silica aerogels. J. Mater. Chem. A 2015, 3, 1935–1940. [Google Scholar] [CrossRef]
- Feng, L.; Zheng, J.; Yang, H.; Guo, Y.; Li, W.; Li, X. Preparation and characterization of polyethylene glycol/active carbon composites as shape-stabilized phase change materials. Sol. Energy Mater. Sol. Cells 2011, 95, 644–650. [Google Scholar] [CrossRef]
- Tyagi, V.V.; Panwar, N.L.; Rahim, N.A.; Kothari, R. Review on solar air heating system with and without thermal energy storage system. Renew. Sustain. Energy Rev. 2012, 16, 2289–2303. [Google Scholar] [CrossRef]
- Pasupathy, A.; Velraja, R.; Seeniraj, R.V. Phase change material-based building architecture for thermal management in residential and commercial establishments. Renew. Sustain. Energy Rev. 2008, 12, 39–64. [Google Scholar] [CrossRef]
- Jeon, J.; Lee, J.H.; Seo, J.; Jeong, S.G.; Kim, S. Application of PCM thermal energy storage system to reduce building energy consumption. J. Therm. Anal. Calorim. 2013, 111, 279–288. [Google Scholar] [CrossRef]
- Biçer, A.; Sari, A. New kinds of energy-storing building composite PCMs for thermal energy storage. Energy Convers. Manag. 2013, 69, 148–156. [Google Scholar] [CrossRef]
- Waqas, A.; Din, Z.U. Phase change material (PCM) storage for free cooling of buildings—A review. Renew. Sustain. Energy Rev. 2013, 18, 607–625. [Google Scholar] [CrossRef]
- Dutil, Y.; Rousse, D.; Lassue, S.; Zalewski, L.; Joulin, A.; Virgone, J.; Kuznik, F.; Johannes, K.; Dumas, J.P.; Bédécarrats, J.P.; et al. Modeling phase change materials behavior in building applications: Comments on material characterization and model validation. Renew. Energy 2014, 61, 132–135. [Google Scholar] [CrossRef]
- Barba, A.; Spiga, M. Discharge mode for encapsulated PCMs in storage tanks. Sol. Energy 2003, 74, 141–148. [Google Scholar] [CrossRef]
- Cabeza, L.F.; Ibáñez, M.; Solé, C.; Roca, J.; Nogués, M. Experimentation with a water tank including a PCM module. Sol. Energy Mater. Sol. Cells 2006, 90, 1273–1282. [Google Scholar] [CrossRef]
- Sole, C.; Medrano, M.; Castell, A.; Nogue, M.; Mehling, H.; Cabeza, L.F. Energetic and exergetic analysis of a domestic water tank with phase change material. Int. J. Energy Res. 2008, 32, 204–214. [Google Scholar] [CrossRef]
- Mazmana, M.; Cabeza, L.F.; Mehling, H.; Nogues, M.; Evliya, H.; Paksoy, H.O. Utilization of phase change materials in solar domestic hot water systems. Renew. Energy 2009, 34, 1639–1643. [Google Scholar] [CrossRef]
- Yang, R.; Xu, H.; Zhang, Y. Preparation, physical property and thermal physical property of phase change microcapsule slurry and phase change emulsion. Sol. Energy Mater. Sol. Cells 2003, 80, 405–416. [Google Scholar] [CrossRef]
- Gschwander, S.; Schossig, P.; Henning, H.M. Micro-encapsulated paraffin in phase-change slurries. Sol. Energy Mater. Sol. Cells 2005, 89, 307–315. [Google Scholar] [CrossRef]
- Alvarado, J.L.; Marsh, C.; Sohn, C.; Phetteplace, G.; Newell, T. Thermal performance of microencapsulated phase change material slurry in turbulent flow under constant heat flux. Int. J. Heat Mass Transf. 2007, 50, 1938–1952. [Google Scholar] [CrossRef]
- Trinquet, F.; Karim, L.; Lefebvre, G.; Royon, L. Mechanical properties and melting heat transf. characteristics of shapestabilized paraffin slurry. Exp. Heat Transf. 2014, 27, 1–13. [Google Scholar] [CrossRef]
- Gil, A.; Oró, E.; Peiró, G.; Álvarez, S.; Cabeza, L.F. Material selection and testing for thermal energy storage in solar cooling. Renew. Energy 2013, 57, 366–371. [Google Scholar] [CrossRef]
- Kurklu, A.; Ozmerzi, A.; Bilgin, S. Thermal performance of a water-phase change material solar collector. Renew. Energy 2002, 26, 391–399. [Google Scholar] [CrossRef]
- Mondal, S. Phase change materials for smart textiles—An overview. Appl. Therm. Eng. 2008, 28, 1536–1550. [Google Scholar] [CrossRef]
- Nelson, G. Application of microencapsulation in textiles. Int. J. Pharm. 2002, 242, 55–62. [Google Scholar] [CrossRef]
- Wang, S.X.; Li, Y.; Hu, J.Y.; Tokura, H.; Song, Q.W. Effect of phase-change material on energy consumption of intelligent thermalprotective clothing. Polym. Test. 2006, 25, 580–587. [Google Scholar] [CrossRef]
- Chen, C.H.; Wang, L.; Huang, Y. Electrospinning of thermo-regulating ultrafine fibers based on polyethylene glycol/cellulose acetate composite. Polymer 2007, 48, 5202–5207. [Google Scholar] [CrossRef]
- Zhang, H.; Yang, S.; Liu, H.; Fang, Y.; Jiang, S.; Guo, J. Preparation of PNHMPA/PEG interpenetrating polymer networks gel and its application for phase change fibers. J. Appl. Polym. Sci. 2013, 129, 1563–1568. [Google Scholar] [CrossRef]
- Lafdi, K.; Mesalhy, O.; Elgafy, A. Graphite foams infiltrated with phase change materials as alternative materials for space and terrestrial thermal energy storage applications. Carbon 2008, 46, 159–168. [Google Scholar] [CrossRef]
- Cui, H.; Xing, Y.; Guo, Y.; Wang, Z.; Cui, H.; Yuan, X. Numerical simulation and experiment investigation on unit heat exchange tube for solar heat receiver. Sol. Energy 2008, 82, 1229–1234. [Google Scholar] [CrossRef]
- Cui, H.; Yuan, X.; Hou, X. Thermal performance analysis for a heat receiver using multiple phase change materials. Appl. Therm. Eng. 2003, 23, 2353–2361. [Google Scholar] [CrossRef]
- Wu, W.F.; Liu, N.; Cheng, W.L.; Liu, Y. Study on the effect of shape-stabilized phase change materials on spacecraft thermal control in extreme thermal environment. Energy Convers. Manag. 2013, 69, 174–180. [Google Scholar] [CrossRef]

| Materials | Melting Point (°C) | Latent Heat (kJ/kg) |
|---|---|---|
| Tetradecane | 5.9 | 258 |
| Pentadecane | 9.9 | 193.9 |
| Hexadecane | 18.1 | 236 |
| Heptadecane | 20.8 | 171 |
| Icosane | 36.7 | 246 |
| Tricosane | 47.5 | 232 |
| Hexacosane | 56.3 | 256 |
| Nonacosane | 63.4 | 240 |
| Dotriacontane | 69.5 | 170 |
| Tetratriacontane | 75.9 | 269 |
| Materials | Melting Point (°C) | Latent Heat (kJ/kg) |
|---|---|---|
| Capric acid | 30.1 | 158 |
| Lauric acid | 43.7 | 210.8 |
| Pentadecanoic acid | 52.5 | 158.6 |
| Myristic acid | 52.1 | 190 |
| Palmitic acid | 54.1 | 183 |
| Stearic acid | 64.5 | 196 |
| PCMs | Melting point Temperature (°C) | Thermal Conductivity (W/mK) | Thermal Conductivity Enhancement (%) |
|---|---|---|---|
| Soy wax/CNTs [87] | 37 | 0.403 | 24.38 |
| n-Octadecylamine/f-MWCNTs [88] | 54.6 | 0.532 | 86.66 |
| Paraffin/S-MWCNTs [89] | 57.9 | 0.43 | 63.49 |
| Paraffin/L-MWCNTs [89] | 58.4 | 0.39 | 48.28 |
| Paraffin/Cu2O–Cu-MWCNTs [90] | 52.32 | 0.34 | 17.24 |
| Palmitic acid-stearic acid/carbon nanotubes (CNTs) [91] | 53.59 | 0.341 | 29.7 |
| Stearic acid/acid treated carbon nanotubes (a-CNTs) [92] | 22 | NA | NA |
| PEG6000/SiO2/CNTs [93] | 53.3 | 0.421 | 51.3 |
| PEG10000-co-N,N′-dihydroxyethyl aniline/surface-modified SWCNTs [94] | 31.5 | 0.334 | 25.09 |
| PCMs | Melting Point (°C) | Thermal Conductivity (W/mK) | Thermal Conductivity Enhancement (%) |
|---|---|---|---|
| Paraffin/GNP [89] | 57.9 | 0.7 | 166.15 |
| Paraffin/graphene [94] | NA | 0.5 | 100 |
| Paraffin/xGNP [95] | 54.9 | 2.4 | 805.66 |
| Paraffin-oleic acid (eutectic gel)/nanographite [98] | 53.5 | 0.662 | 264 |
| Palmitic acid/polyaniline (PANI)/xGNP [99] | 61.57 | 1.08 | 237.5 |
| Erythritol/EG [100] | 116.32 | 2.06 | 564.5 |
| Erythritol/EG [101] | NA | 3.56 | 401.4 |
| Palmitic acid/graphene nanoplatelets [102] | 61.16 | 2.11 | 776.19 |
| Stearic acid/graphene oxide [103] | 32.57 | NA | NA |
| Palmitic acid/nitrogen doped graphene [104] | 66.52 | 1.73 | 517.85 |
| PCMs | Melting Point Temperature (°C) | Thermal Conductivity (W/mK) | Thermal Conductivity Enhancement (%) |
|---|---|---|---|
| Paraffin wax/CNFs [87] | 50 | 0.45 | 40.62 |
| Paraffin/CNFs [89] | 58.4 | 0.33 | 25.47 |
| Paraffin/carbon aerogel [104] | 53.5 | NA | NA |
| Octadecanoic acid/graphene aerogel [105] | 57 | 2.635 | 1332.06 |
| Paraffin/3D graphene foam [106] | 58 | 0.617 | 99.67 |
| Lauric acid/activated carbon [107] | 44.07 | 0.18 | 12.5 |
| Palmitic acid/carbonized alkylated silica aerogel [108] | 61 | NA | NA |
| Octadecanol/carbonized alkylated silica aerogel [108] | 59 | NA | NA |
| PEG1500/active carbon [109] | 48 | NA | NA |
| PEG10000-co-N,N′-dihydrox-yethyl aniline/carbon black [96] | 29.3 | 0.280 | 4.86 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, G.; Yim, Y.-J.; Lee, J.W.; Heo, Y.-J.; Park, S.-J. Carbon-Filled Organic Phase-Change Materials for Thermal Energy Storage: A Review. Molecules 2019, 24, 2055. https://doi.org/10.3390/molecules24112055
Yang G, Yim Y-J, Lee JW, Heo Y-J, Park S-J. Carbon-Filled Organic Phase-Change Materials for Thermal Energy Storage: A Review. Molecules. 2019; 24(11):2055. https://doi.org/10.3390/molecules24112055
Chicago/Turabian StyleYang, Guijun, Yoon-Ji Yim, Ji Won Lee, Young-Jung Heo, and Soo-Jin Park. 2019. "Carbon-Filled Organic Phase-Change Materials for Thermal Energy Storage: A Review" Molecules 24, no. 11: 2055. https://doi.org/10.3390/molecules24112055
APA StyleYang, G., Yim, Y.-J., Lee, J. W., Heo, Y.-J., & Park, S.-J. (2019). Carbon-Filled Organic Phase-Change Materials for Thermal Energy Storage: A Review. Molecules, 24(11), 2055. https://doi.org/10.3390/molecules24112055




