Antibacterial Properties of Propolis
Abstract
1. Introduction
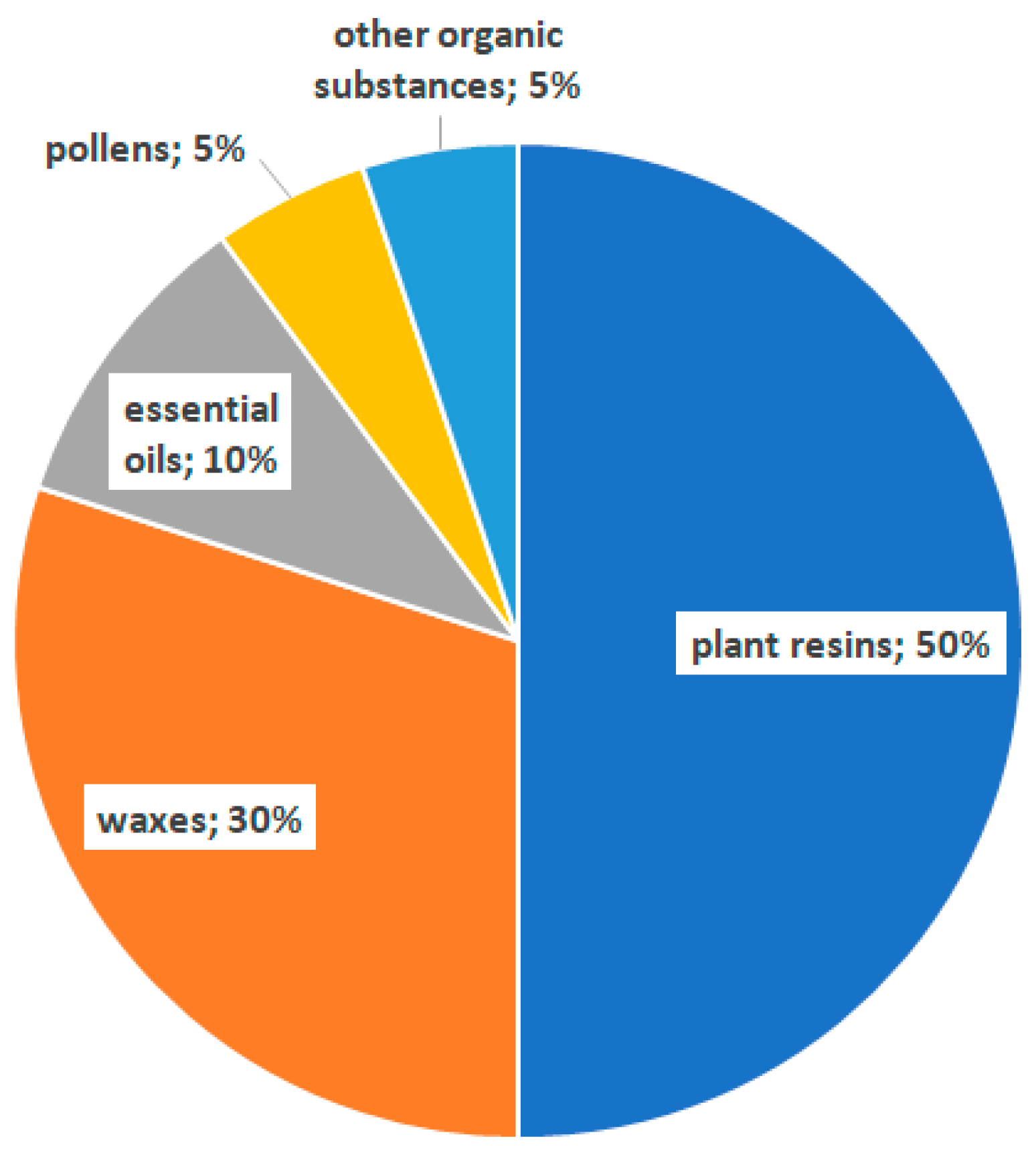
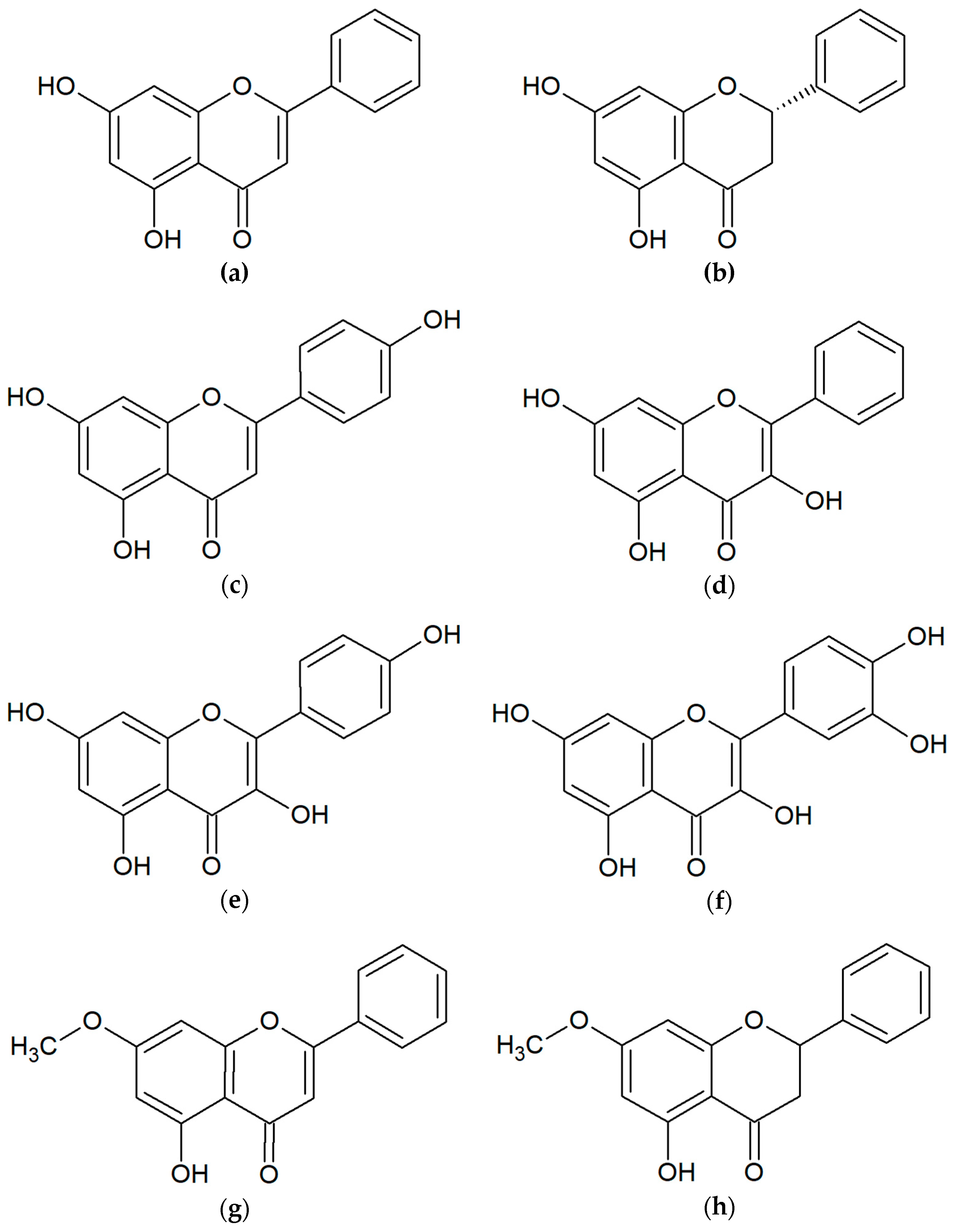
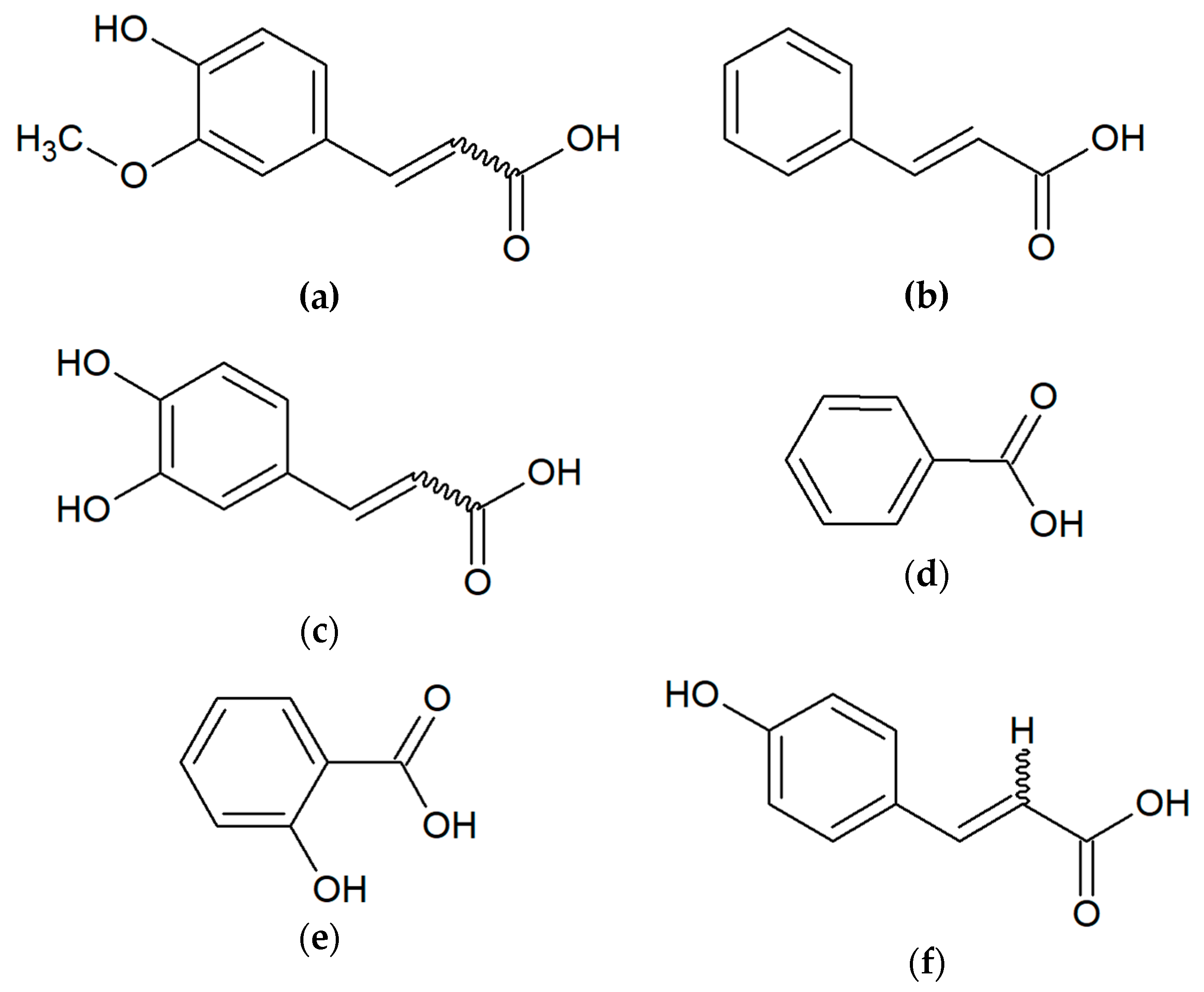
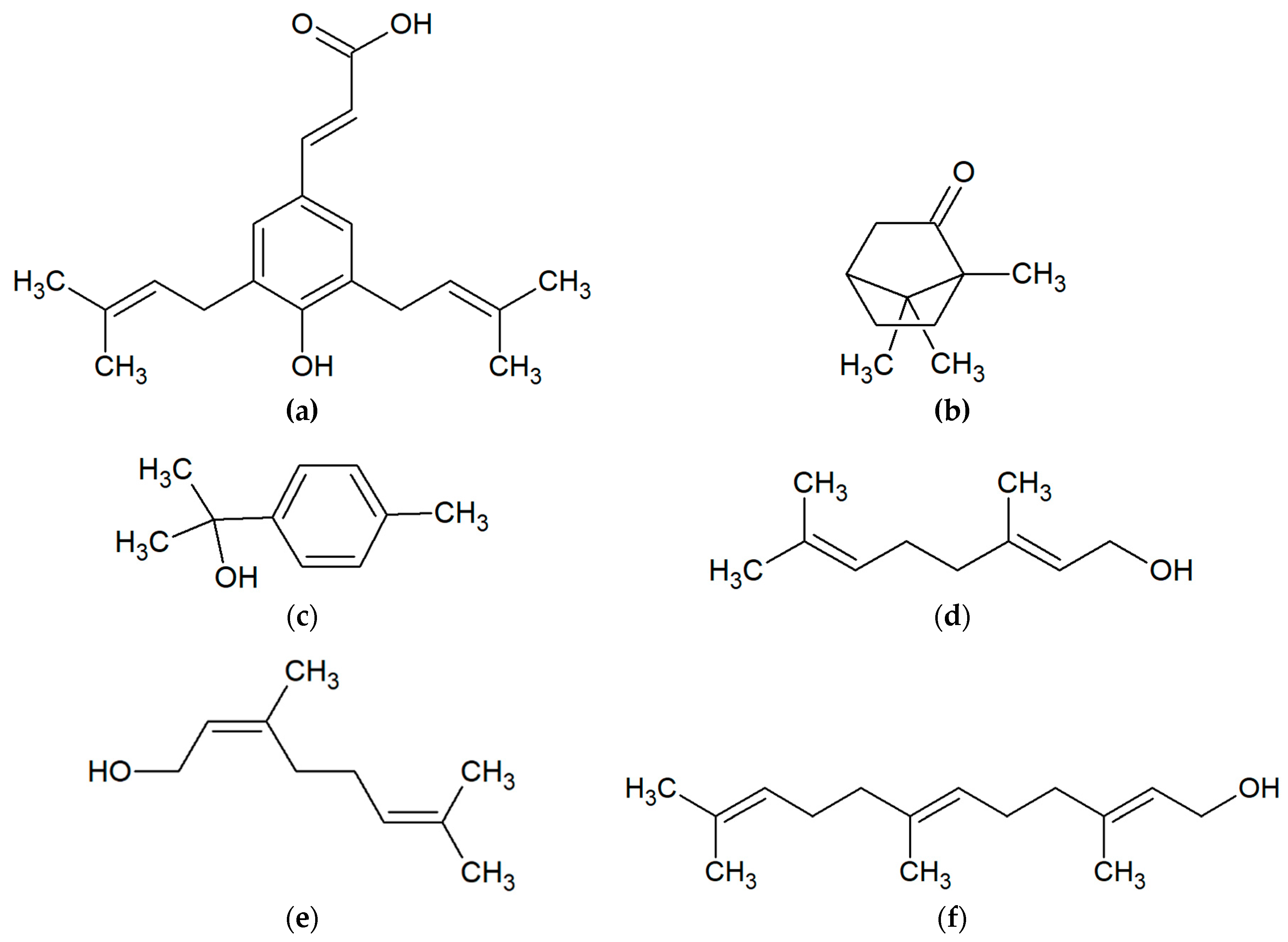
2. Chemical Compounds of Propolis
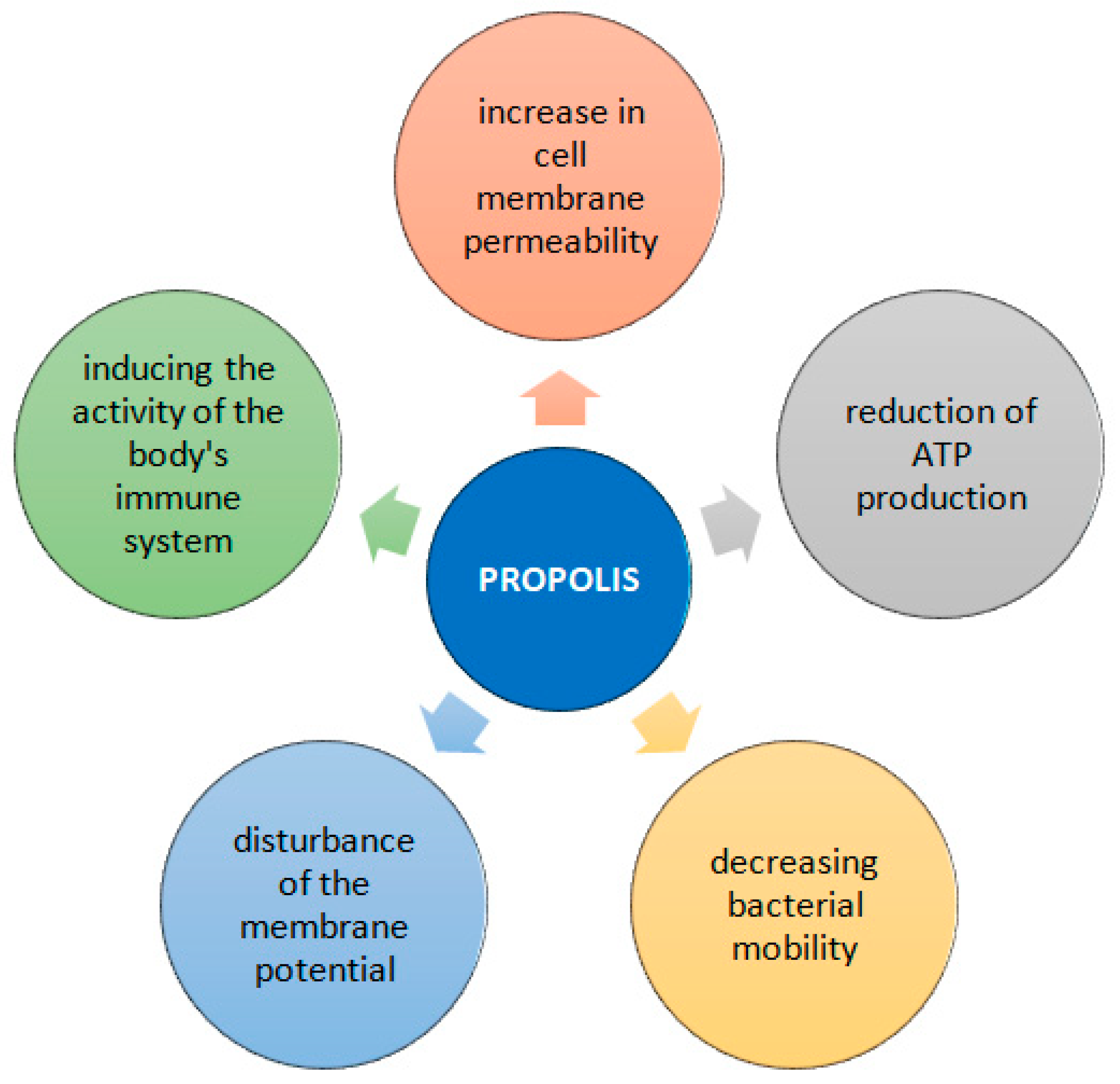
3. Antimicrobial Properties of Propolis
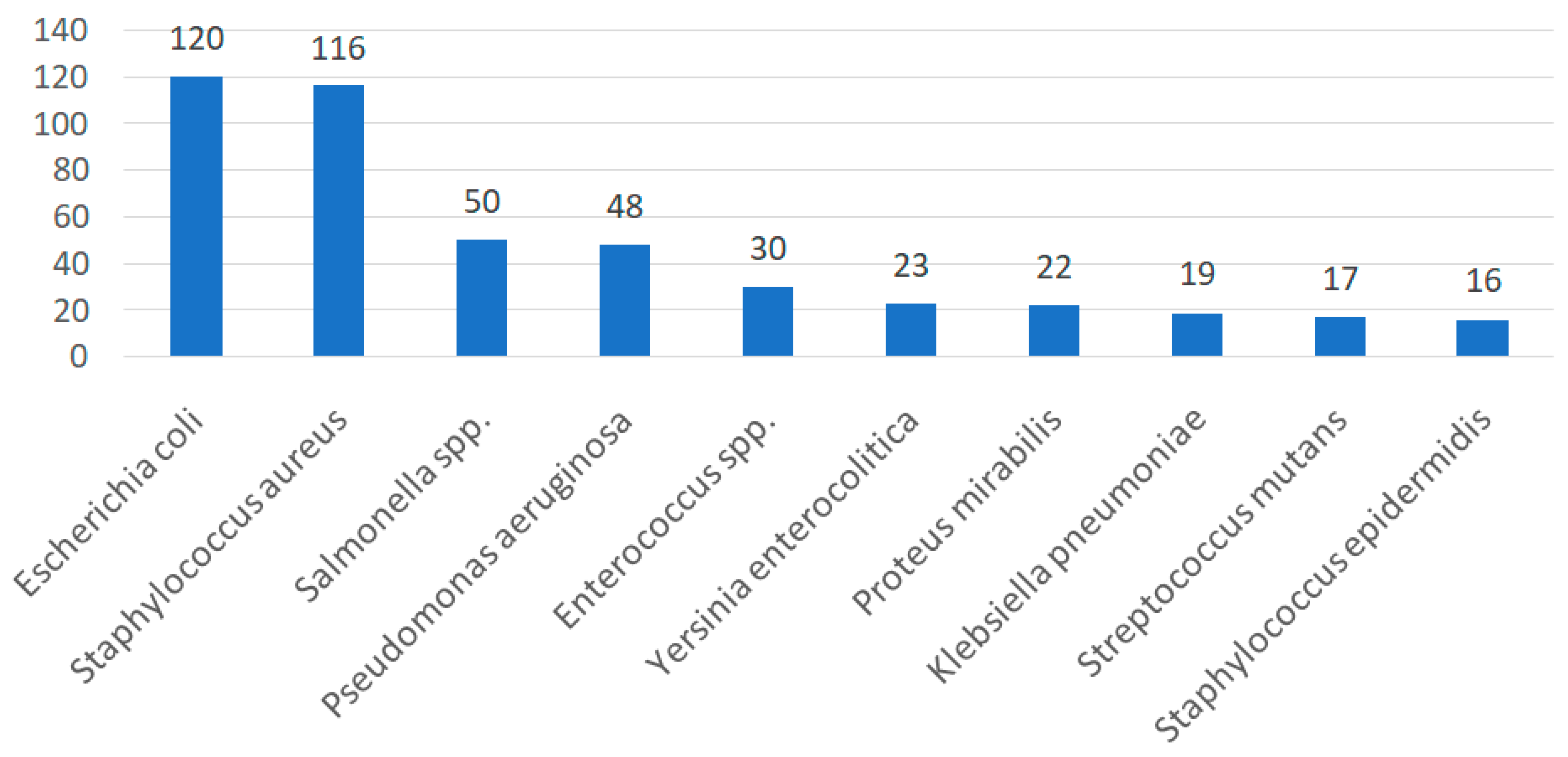
4. Data Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kędzia, B. Pochodzenie propolisu w świetle teorii i badań naukowych. The origin of propolis in the theories and scientific research. Herba Pol. 2008, 54, 179–186. [Google Scholar]
- Zabaiou, N.; Fouache, A.; Trousson, A.; Baron, S.; Zellagui, A.; Lahouel, M.; Lobaccaro, J.A. Biological properties of propolis extracts: Something new from an ancient product. Chem. Phys. Lipids 2017, 207, 214–222. [Google Scholar] [CrossRef] [PubMed]
- Wagh, V.D. Propolis: A wonder bees product and its pharmacological potentials. Adv. Pharmacol. Sci. 2013, 2013, 308249. [Google Scholar] [CrossRef] [PubMed]
- Toreti, V.C.; Sato, H.H.; Pastore, G.M.; Park, Y.K. Recent progress of propolis for its biological and chemical compositions and its botanical origin. Evid. Based Complement. Alternat. Med. 2013, 2013, 697390. [Google Scholar] [CrossRef]
- Sforcin, J.M.; Bankova, V. Propolis: Is there a potential for the development of new drugs? J. Ethnopharmacol. 2011, 133, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Silva, J.C.; Rodrigues, S.; Feás, X.; Estevinho, L.M. Antimicrobial activity, phenolic profile and role in the inflammation of propolis. Food Chem. Toxicol. 2012, 50, 1790–1795. [Google Scholar] [CrossRef] [PubMed]
- Schnitzler, P.; Neuner, A.; Nolkemper, S.; Zundel, C.; Nowack, H.; Sensch, K.H.; Reichling, J. Antiviral activity and mode of action of propolis extracts and selected compounds. Phytother. Res. 2010, 24 (Suppl. 1), S20–S28. [Google Scholar] [CrossRef]
- Ramanauskiene, K.; Inkeniene, A. Propolis oil extract: Quality analysis and evaluation of its antimicrobial activity. Nat. Prod. Res. 2011, 25, 1463–1468. [Google Scholar] [CrossRef]
- Franchin, M.; Cólon, D.F.; Castanheira, F.V.; da Cunha, M.G.; Bueno-Silva, B.; Alencar, S.M.; Cunha, T.M.; Rosalen, P.L. Vestitol isolated from Brazilian red propolis inhibits neutrophils migration in the inflammatory process: Elucidation of the mechanism of action. J. Nat. Prod. 2016, 79, 954–960. [Google Scholar] [CrossRef] [PubMed]
- De Freitas, M.C.D.; de Miranda, M.B.; de Oliveira, D.T.; Vieira-Filho, S.A.; Caligiorne, R.B.; de Figueiredo, S.M. Biological activities of red propolis: A review. Recent Pat. Endocr. Metab. Immun. Drug Discov. 2017, 11, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Warakomska, Z.; Maciejewicz, W. Microscopic analysis of propolis from Polish regions. Apidologie 1992, 23, 277. [Google Scholar] [CrossRef]
- Jabłoński, B.; Kołtowski, Z. Nectar secretion and honey potential of honey plants growing under Poland’s conditions. J. Api. Sci. 2005, 49, 59. [Google Scholar]
- Szeleszczuk, Ł.; Zielińska-Pisklak, M.; Goś, P. Propolis - panaceum prosto z ula. Lek W Polsce 2013, 23, 32–39. [Google Scholar]
- Şenel, E.; Demir, E. Bibliometric analysis of apitherapy in complementary medicine literature between 1980 and 2016. Complement. Ther. Clin. Pract. 2018, 31, 47–52. [Google Scholar] [CrossRef]
- Bueno-Silva, B.; Marsola, A.; Ikegaki, M.; Alencar, S.M.; Rosalen, P.L. The effect of seasons on Brazilian red propolis and its botanical source: Chemical composition and antibacterial activity. Nat. Prod. Res. 2016, 31, 1318–1324. [Google Scholar] [CrossRef]
- da Cunha, M.G.; Franchin, M.; de Carvalho Galvão, L.C.; de Ruiz, A.L.; de Carvalho, J.E.; Ikegaki, M.; de Alencar, S.M.; Koo, H.; Rosalen, P.L. Antimicrobial and antiproliferative activities of stingless bee Melipona scutellaris geopropolis. BMC Complement. Altern. Med. 2013, 13, 23. [Google Scholar] [CrossRef]
- Santos, T.L.A.D.; Queiroz, R.F.; Sawaya, A.C.H.F.; Lopez, B.G.; Soares, M.B.P.; Bezerra, D.P.; Rodrigues, A.C.B.C.; Paula, V.F.; Waldschmidt, A.M. Melipona mondury produces a geopropolis with antioxidant, antibacterial and antiproliferative activities. An. Acad. Bras. Cienc. 2017, 89, 2247–2259. [Google Scholar] [CrossRef]
- Torres, A.R.; Sandjo, L.P.; Friedemann, M.T.; Tomazzoli, M.M.; Maraschin, M.; Mello, C.F.; Santos, A.R.S. Chemical characterization, antioxidant and antimicrobial activity of propolis obtained from Melipona quadrifasciata quadrifasciata and Tetragonisca angustula stingless bees. Braz. J. Med. Biol. Res. 2018, 51, e7118. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Caravaca, A.M.; Gómez-Romero, M.; Arráez-Román, D.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Advances in the analysis of phenolic compounds in products derived from bees. J. Pharmac. Biomed. Anal. 2006, 41, 1220–1234. [Google Scholar] [CrossRef]
- Miguel, M.G.; Nunes, S.; Dandlen, S.A.; Cavaco, A.M.; Antunes, M.D. Phenols and antioxidant activity of hydro-alcoholic extracts of propolis from Algarve, South of Portugal. Food Chem. Toxicol. 2010, 48, 3418–3423. [Google Scholar] [CrossRef] [PubMed]
- Inui, S.; Hatano, A.; Yoshino, M.; Hosoya, T.; Shimamura, Y.; Masuda, S.; Ahn, M.R.; Tazawa, S.; Araki, Y.; Kumazawa, S. Identification of the phenolic compounds contributing to antibacterial activity in ethanol extracts of Brazilian red propolis. Nat. Prod. Res. 2014, 28, 1293–1296. [Google Scholar] [CrossRef] [PubMed]
- Brodowska, K.M. Natural flavonoids: Classification, potential role, and application of flavonoid analogues. Eur. J. Biol. Res. 2017, 7, 108–123. [Google Scholar]
- Górniak, I.; Bartoszewski, R.; Króliczewski, J. Comprehensive review of antimicrobial activities of plant flavonoids. Phytochem. Rev. 2019, 18, 241–272. [Google Scholar] [CrossRef]
- Devequi-Nunes, D.; Machado, B.A.S.; Barreto, G.A.; Rebouças Silva, J.; da Silva, D.F.; da Rocha, J.L.C.; Brandão, H.N.; Borges, V.M.; Umsza-Guez, M.A. Chemical characterization and biological activity of six different extracts of propolis through conventional methods and supercritical extraction. PLoS ONE 2018, 13, e0207676. [Google Scholar] [CrossRef]
- Wieczyńska, A.; Weżgowiec, J.; Więckiewicz, W.; Czarny, A.; Kulbacka, J.; Nowakowska, D.; Gancarz, R.; Wilk, K.A. Antimicrobial activity, cytotoxicity and total phenolic content of different extracts of propolis from the west Pomeranian region in Poland. Acta Pol. Pharm. 2017, 74, 715–722. [Google Scholar] [PubMed]
- Pimenta, H.C.; Violante, I.M.; Musis, C.R.; Borges, Á.H.; Aranha, A.M. In vitro effectiveness of Brazilian brown propolis against Enterococcus faecalis. Braz. Oral Res. 2015, 29, 1–6. [Google Scholar] [CrossRef]
- Kędzia, B.; Hołderna-Kędzia, E. Pinocembryna – flawonoidowy składnik krajowego propolisu o działaniu opóźniającym rozwój choroby Alzheimera. Pinocembrin—flavonoid component of domestic propolis with delaying effect of the development of Alzheimer’s disease. Post. Fitoter. 2017; 18, 223–228. [Google Scholar]
- Bankova, V.S.; Castro, D.S.L.; Marcucci, M.C. Propolis: Recent advances in chemistry and plant origin. Apidologie 2000, 31, 3–15. [Google Scholar] [CrossRef]
- Kędzia, B. Skład chemiczny i aktywność biologiczna propolisu pochodzącego z różnych rejonów świata. Post. Fitoter. 2006, 1, 23–35. [Google Scholar]
- Wolska, K.; Górska, A.; Adamiak, A. Właściwości przeciwbakteryjne propolisu. Post. Mikrobiol. 2016, 55, 343–350. [Google Scholar]
- Sforcin, J.M. Biological properties and therapeutic applications of propolis. Phytother. Res. 2016, 30, 894–905. [Google Scholar] [CrossRef]
- Pasupuleti, V.R.; Sammugam, L.; Ramesh, N.; Gan, S.H. Honey, propolis and royal jelly: A comprehensive review of their biological actions and health benefits. Oxid. Med. Cell Longev. 2017, 2017, 1259510. [Google Scholar] [CrossRef] [PubMed]
- Pamplona-Zomenhan, L.C.; Pamplona, B.C.; da Silva, C.B.; Marcucci, M.C.; Mimica, L.M. Evaluation of the in vitro antimicrobial activity of an ethanol extract of Brazilian classified propolis on strains of Staphylococcus aureus. Braz. J. Microbiol. 2011, 42, 1259–1264. [Google Scholar] [CrossRef]
- Kędzia, B.; Hołderna-Kędzia, E. Aktywność antybiotyczna propolisu krajowego i europejskiego. The antibiotic activity of native and European propolis. Post. Fitoter. 2013, 2, 97–107. [Google Scholar]
- Veiga, R.S.; De Mendonça, S.; Mendes, P.B.; Paulino, N.; Mimica, M.J.; Lagareiro Netto, A.A.; Marcucci, M.C. Artepillin C and phenolic compounds responsible for antimicrobial and antioxidant activity of green propolis and Baccharis dracunculifolia DC. J. Appl. Microbiol. 2017, 122, 911–920. [Google Scholar] [CrossRef]
- Yoshimasu, Y.; Ikeda, T.; Sakai, N.; Yagi, A.; Hirayama, S.; Morinaga, Y.; Furukawa, S.; Nakao, R. Rapid bactericidal action of propolis against Porphyromonas gingivalis. J. Dent. Res. 2018, 97, 928–936. [Google Scholar] [CrossRef]
- Paulino, N.; Abreu, S.R.L.; Uto, Y.; Koyama, D.; Nagasawa, H.; Hori, H.; Dirsch, V.M.; Vollmar, A.M.; Scremin, A.; Bretz, W.A. Anti-inflammatory effects of a bioavailable compound, artepillin C, in Brazilian propolis. Eur. J. Pharmacol. 2008, 587, 296–301. [Google Scholar] [CrossRef] [PubMed]
- Viuda-Martos, M.; Ruiz-Navajas, Y.; Fernández-López, J.; Pérez-Álvarez, J.A. Functional properties of honey, propolis, and royal jelly. J. Food Sci. 2008, 73, R117–R124. [Google Scholar] [CrossRef]
- Guz, N.R.; Stermitz, F.R.; Johnson, J.B.; Beeson, T.D.; Willen, S.; Hsiang, J.F.; Lewis, K. Flavonolignan and flavone inhibitors of a Staphylococcus aureus multidrug resistance pump: Structure-activity relationships. J. Med. Chem. 2001, 44, 261–268. [Google Scholar] [CrossRef]
- Seibert, J.B.; Bautista-Silva, J.P.; Amparo, T.R.; Petit, A.; Pervier, P.; Dos Santos Almeida, J.C.; Azevedo, M.C.; Silveira, B.M.; Brandão, G.C.; de Souza, G.H.B.; et al. Development of propolis nanoemulsion with antioxidant and antimicrobial activity for use as a potential natural preservative. Food Chem. 2019, 287, 61–67. [Google Scholar] [CrossRef]
- Veloz, J.J.; Alvear, M.; Salazar, L.A. Antimicrobial and antibiofilm activity against Streptococcus mutans of individual and mixtures of the main polyphenolic compounds found in Chilean propolis. Biomed. Res. Int. 2019, 2019, 7602343. [Google Scholar] [CrossRef]
- Soromou, L.W.; Zhang, Y.; Cui, Y.; Wei, M.; Chen, N.; Yang, X.; Huo, M.; Baldé, A.; Guan, S.; Deng, X.; et al. Subinhibitory concentrations of pinocembrin exert anti-Staphylococcus aureus activity by reducing α-toxin expression. J. Appl. Microbiol. 2013, 115, 41–49. [Google Scholar] [CrossRef]
- Joray, M.B.; Trucco, L.D.; González, M.L.; Napal, G.N.; Palacios, S.M.; Bocco, J.L.; Carpinella, M.C. Antibacterial and cytotoxic activity of compounds isolated from Flourensia oolepis. Evid. Based Complement. Alternat. Med. 2015, 2015, 912484. [Google Scholar] [CrossRef]
- Hernández Tasco, A.J.; Ramírez Rueda, R.Y.; Alvarez, C.J.; Sartori, F.T.; Sacilotto, A.C.B.C.; Ito, I.Y.; Vichnewski, W.; Salvador, M.J. Antibacterial and antifungal properties of crude extracts and isolated compounds from Lychnophora markgravii. Nat. Prod. Res. 2018, 1–5. [Google Scholar] [CrossRef]
- Kharsany, K.; Viljoen, A.; Leonard, C.; van Vuuren, S. The new buzz: Investigating the antimicrobial interactions between bioactive compounds found in South African propolis. J. Ethnopharmacol. 2019, 238, 111867. [Google Scholar] [CrossRef] [PubMed]
- Nayaka, H.B.; Londonkar, R.L.; Umesh, M.K.; Tukappa, A. Antibacterial attributes of apigenin, isolated from Portulaca oleracea L. Int. J. Bacteriol. 2014, 2014, 175851. [Google Scholar] [CrossRef]
- Akilandeswari, K.; Ruckmani, K. Synergistic antibacterial effect of apigenin with β-lactam antibiotics and modulation of bacterial resistance by a possible membrane effect against methicillin resistant Staphylococcus aureus. Cell Mol. Biol. 2016, 62, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Eumkeb, G.; Chukrathok, S. Synergistic activity and mechanism of action of ceftazidime and apigenin combination against ceftazidime-resistant Enterobacter cloacae. Phytomedicine 2013, 20, 262–269. [Google Scholar] [CrossRef]
- Heleno, S.A.; Ferreira, I.C.; Esteves, A.P.; Ćirić, A.; Glamočlija, J.; Martins, A.; Soković, M.; Queiroz, M.J. Antimicrobial and demelanizing activity of Ganoderma lucidum extract, p-hydroxybenzoic and cinnamic acids and their synthetic acetylated glucuronide methyl esters. Food Chem. Toxicol. 2013, 58, 95–100. [Google Scholar] [CrossRef]
- Guzman, J.D. Natural cinnamic acids, synthetic derivatives and hybrids with antimicrobial activity. Molecules 2014, 19, 19292–19349. [Google Scholar] [CrossRef]
- Yilmaz, S.; Sova, M.; Ergün, S. Antimicrobial activity of trans-cinnamic acid and commonly used antibiotics against important fish pathogens and nonpathogenic isolates. J. Appl. Microbiol. 2018, 125, 1714–1727. [Google Scholar] [CrossRef] [PubMed]
- Vasconcelos, N.G.; Croda, J.; Simionatto, S. Antibacterial mechanisms of cinnamon and its constituents: A review. Microb. Pathog. 2018, 120, 198–203. [Google Scholar] [CrossRef]
- Gopu, V.; Meena, C.K.; Shetty, P.H. Quercetin influences quorum sensing in food borne bacteria: In-vitro and in-silico evidence. PLoS ONE 2015, 10, e0134684. [Google Scholar] [CrossRef] [PubMed]
- Siriwong, S.; Teethaisong, Y.; Thumanu, K.; Dunkhunthod, B.; Eumkeb, G. The synergy and mode of action of quercetin plus amoxicillin against amoxicillin-resistant Staphylococcus epidermidis. BMC Pharmacol. Toxicol. 2016, 17, 39. [Google Scholar] [CrossRef]
- Harasstani, O.A.; Tham, C.L.; Israf, D.A. Kaempferol and chrysin synergies to improve septic mice survival. Molecules 2017, 22, 92. [Google Scholar] [CrossRef]
- Ming, D.; Wang, D.; Cao, F.; Xiang, H.; Mu, D.; Cao, J.; Li, B.; Zhong, L.; Dong, X.; Zhong, X.; et al. Kaempferol inhibits the primary attachment phase of biofilm formation in Staphylococcus aureus. Front. Microbiol. 2017, 8, 2263. [Google Scholar] [CrossRef]
- Wang, S.; Yao, J.; Zhou, B.; Yang, J.; Chaudry, M.T.; Wang, M.; Xiao, F.; Li, Y.; Yin, W. Bacteriostatic effect of quercetin as an antibiotic alternative in vivo and its antibacterial mechanism in vitro. J. Food Prot. 2018, 81, 68–78. [Google Scholar] [CrossRef]
- Park, H.; Bae, S.H.; Choi, H.; Suh, H.J. Lipase-mediated lipid removal from propolis extract and its antiradiacal and antimicrobial activity. J. Sci. Food Agric. 2015, 95, 1697–1705. [Google Scholar] [CrossRef]
- Kubiliene, L.; Laugaliene, V.; Paviloniis, A.; Maruska, A.; Majiene, D.; Barcauskaite, K.; Kunilus, R.; Kasparaviciene, G.; Savickas, A. Alternative preparation of propolis extracts: Comparison of their composition and biological activities. Complement. Altern. Med. 2015, 15, 156. [Google Scholar] [CrossRef]
- Bridi, R.; Montenegro, G.; Nuñez-Quijada, G.; Giordano, A.; Morán-Romero, F.M.; Jara-Pezoa, I.; Speisky, H.; Atala, E.; López-Alarcón, C. International regulations of propolis quality: Required assays do not necessarily reflect their polyphenolic-related in vitro activities. J. Food Sci. 2015, 80, C1188–C1195. [Google Scholar] [CrossRef]
- Al-Waili, N.; Al-Ghamdi, A.; Ansari, M.J.; Al-Attal, Y.; Salom, K. Synergistic effects of honey and propolis toward drug multi-resistant Staphylococcus aureus, Escherichia coli and Candida albicans isolates in single and polymicrobial cultures. Int. J. Med. Sci. 2012, 9, 793–800. [Google Scholar] [CrossRef]
- Boyanova, L.; Kolarov, R.; Gergova, G.; Mitov, I. In vitro activity of Bulgarian propolis against 94 clinical isolates of anaerobic bacteria. Anaerobe 2006, 12, 173–177. [Google Scholar] [CrossRef]
- Ferreira, F.B.; Torres, S.A.; Rosa, O.P.; Ferreira, C.M.; Garcia, R.B.; Marcucci, M.C.; Gomes, B.P. Antimicrobial effect of propolis and other substances against selected endodontic pathogens. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2007, 104, 709–716. [Google Scholar] [CrossRef]
- Koru, O.; Toksoy, F.; Acikel, C.H.; Tunca, Y.M.; Baysallar, M.; Uskudar Guclu, A.; Akca, E.; Ozkok Tuylu, A.; Sorkun, K.; Tanyuksel, M.; et al. In vitro antimicrobial activity of propolis samples from different geographical origins against certain oral pathogens. Anaerobe 2007, 13, 140–145. [Google Scholar] [CrossRef]
- Shabbir, A.; Rashid, M.; Tipu, H.N. Propolis, a hope for the future in treating resistant periodontal pathogens. Cureus 2016, 8, e682. [Google Scholar] [CrossRef]
- Kędzia, A. Działanie wyciągu etanolowego z propolisu na bakterie beztlenowe. Herba Pol. 1986, 32, 53–57. [Google Scholar]
- Kędzia, A.; Kałowski, M. Ocena skuteczności działania wyciągu etanolowego z propolisu na bakterie bezwzględnie beztlenowe jamy ustnej. Czas. Stomat. 1988, 41, 757–762. [Google Scholar]
- Kędzia, A. Wrażliwość bakterii bezwzględnie beztlenowych na wyciąg etanolowy z propolisu. Herba Pol. 1988, 34, 267–272. [Google Scholar]
- Clinical and Laboratory Standards Institute. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically. Approved Standards-Ninth Edition. M07-A9. 2012; 32, 3. [Google Scholar]
- Mavri, A.; Abramovič, H.; Polak, T.; Bertoncelj, J.; Jamnik, P.; Smole Možina, S.; Jeršek, B. Chemical properties and antioxidant and antimicrobial activities of Slovenian propolis. Chem. Biodiv. 2012, 9, 1545–1558. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Sankarapandian, K.; Cheng, Y.; Woo, S.O.; Kwon, H.W.; Perumalsamy, H.; Ahn, Y.-J. Relationship between total phenolic contents and biological properties of propolis from 20 different regions in South Korea. BMC Complement. Altern. Med. 2016, 16, 65. [Google Scholar] [CrossRef] [PubMed]
- Bittencourt, M.L.F.; Ribeiro, P.R.; Franco, R.L.P.; Hilhorst, H.W.M.; de Castro, R.D.; Fernandez, L.G. Metabolite profiling, antioxidant and antibacterial activities of Brazilian propolis: Use of correlation and multivariate analyses. Food Res. Int. 2015, 76, 449–457. [Google Scholar] [CrossRef]
- Popova, M.; Giannopoulou, E.; Skalicka-Woźniak, K.; Graikou, K.; Widelski, J.; Bankova, V.; Kalofonos, H.; Sivolapenko, G.; Gaweł-Bęben, K.; Antosiewicz, B.; et al. Characterization and biological evaluation of propolis from Poland. Molecules 2017, 22, 1159. [Google Scholar] [CrossRef] [PubMed]
- Moncla, B.J.; Guevara, P.W.; Wallace, J.A.; Marcucci, M.C.; Nor, J.E.; Bretz, W.A. The inhibitory activity of typified propolis against Enterococcus species. Z. Naturforsch. C. 2012, 67, 249–256. [Google Scholar] [PubMed]
- Uzel, A.; Sorkun, K.; Önçağ, Ö.; Çoğulu, D.; Gençay, Ö.; Salih, B. Chemical compositions and antimicrobial activities of four different Anatolian propolis samples. Microbiol. Res. 2005, 160, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Popova, M.P.; Chinou, I.B.; Marekov, I.N.; Bankova, V.S. Terpenes with antimicrobial activity from Cretan propolis. Phytochemistry. 2009, 70, 1262–1271. [Google Scholar] [CrossRef] [PubMed]
- Popova, M.; Dimitrova, R.; Al-Lawati, H.; Tsvetkova, I.; Najdenski, H.; Bankova, V. Omani propolis: Chemical profiling, antibacterial activity and new propolis plant sources. Chem. Cent. J. 2013, 7, 158. [Google Scholar] [CrossRef]
- Wojtyczka, R.D.; Dziedzic, A.; Idzik, D.; Kępa, M.; Kubina, R.; Kabała-Dzik, A.; Smoleń-Dzirba, J.; Stojko, J.; Sajewicz, M.; Wąsik, T.J. Susceptibility of Staphylococcus aureus clinical isolates to propolis extract alone or in combination with antimicrobial drugs. Molecules 2013, 18, 9623–9640. [Google Scholar] [CrossRef]
- Patel, J.; Ketkar, S.; Patil, S.; Fearnley, J.; Mahadik, K.R.; Paradkar, A.R. Potentiating antimicrobial efficacy of propolis through niosomal-based system for administration. Integr. Med. Res. 2014, 4, 94–101. [Google Scholar] [CrossRef]
- Schmidt, E.M.; Stock, D.; Chada, F.J.G.; Finger, D.; Sawaya, A.C.; Eberlin, M.N.; Felsner, M.L.; Quináia, S.P.; Monteiro, M.C.; Torres, Y.R. A Comparison between characterization and biological properties of Brazilian fresh and aged propolis. Biomed. Res. Int. 2014, 2014, 257617. [Google Scholar] [CrossRef]
- Campos, J.F.; dos Santos, U.P.; Macorini, L.F.B.; de Melo, A.M.M.F.; Balestieri, J.B.P.; Paredes-Gamero, E.J.; dos Santos, E.L. Antimicrobial, antioxidant and cytotoxic activities of propolis from Melipona orbignyi (Hymenoptera, Apidae). Food Chem. Toxicol. 2014, 65, 374–380. [Google Scholar] [CrossRef]
- Campos, J.F.; Dos Santos, U.P.; da Rocha Pdos, S.; Damião, M.J.; Balestieri, J.B.; Cardoso, C.A.; Paredes-Gamero, E.J.; Estevinho, L.M.; de Picoli Souza, K.; Dos Santos, E.L. Antimicrobial, antioxidant, anti-inflammatory, and cytotoxic activities of propolis from the stingless bee Tetragonisca fiebrigi (Jataí). Evid. Based Complement. Alternat. Med. 2015, 2015, 296186. [Google Scholar] [CrossRef]
- Massaro, C.F.; Simpson, J.B.; Powell, D.; Brooks, P. Chemical composition and antimicrobial activity of honeybee (Apis mellifera ligustica) propolis from subtropical. Sci. Nat. 2015, 102, 68. [Google Scholar] [CrossRef] [PubMed]
- Regueira, N.M.S.; Tintino, S.R.; da Silva, A.R.P.; Costa, M.D.S.; Boligon, A.A.; Matias, E.F.F.; de Queiroz Balbino, V.; Menezes, I.R.A.; Melo Coutinho, H.D. Seasonal variation of Brazilian red propolis: Antibacterial activity, synergistic effect and phytochemical screening. Food Chem. Toxicol. 2017, 107 Pt B, 572–580. [Google Scholar] [CrossRef] [PubMed]
- Dantas Silva, R.P.; Machado, B.A.; Barreto, G.A.; Costa, S.S.; Andrade, L.N.; Amaral, R.G.; Carvalho, A.A.; Padilha, F.F.; Barbosa, J.D.; Umsza-Guez, M.A. Antioxidant, antimicrobial, antiparasitic, and cytotoxic properties of various Brazilian propolis extracts. PLoS ONE 2017, 12, e0172585. [Google Scholar] [CrossRef]
- Al-Ani, I.; Zimmermann, S.; Reichling, J.; Wink, M. Antimicrobial activities of European propolis collected from various geographic origins alone and in combination with antibiotics. Medicine 2018, 5, 2. [Google Scholar] [CrossRef] [PubMed]
- El-Guendouz, S.; Aazza, S.; Lyoussi, B.; Bankova, V.; Popova, M.; Neto, L.; Faleiro, M.L.; Miguel, M.D.G. Moroccan propolis: A natural antioxidant, antibacterial, and antibiofilm against Staphylococcus aureus with no induction of resistance after continuous exposure. Evid. Based Complement. Alternat. Med. 2018, 2018, 9759240. [Google Scholar] [CrossRef] [PubMed]
- Yue-Wen, C.; Siou-Ru, Y.; Chieh, T.; Yu-Hsiang, Y. Antibacterial activity of propolins from Taiwanese green propolis. J. Food Drug Anal. 2018, 26, 761–768. [Google Scholar]
- Nina, N.; Quispe, C.; Jiménez-Aspee, F.; Theoduloz, C.; Feresín, G.E.; Lima, B.; Leiva, E.; Schmeda-Hirschmann, G. Antibacterial activity, antioxidant effect and chemical composition of propolis from the Región del Maule, Central Chile. Molecules 2015, 20, 18144–18167. [Google Scholar] [CrossRef] [PubMed]
- Wojtyczka, R.D.; Kępa, M.; Idzik, D.; Kubina, R.; Kabała-Dzik, A.; Dziedzic, A.; Wąsik, T.J. In vitro antimicrobial activity of ethanolic extract of Polish propolis against biofilm forming Staphylococcus epidermidis strains. Evid. Based Complement. Alternat. Med. 2013, 2013, 590703. [Google Scholar] [CrossRef]
- Kim, M.J.; Kim, C.S.; Kim, B.H.; Ro, S.B.; Lim, Y.K.; Park, S.N.; Cho, E.; Ko, J.H.; Kwon, S.S.; Ko, Y.M.; et al. Antimicrobial effect of Korean propolis against the mutans streptococci isolated from Korean. J. Microbiol. 2011, 49, 161. [Google Scholar] [CrossRef]
- Barrientos, L.; Herrera, C.L.; Montenegro, G.; Ortega, X.; Veloz, J.; Alvear, M.; Cuevas, A.; Saavedra, N.; Salazar, L.A. Chemical and botanical characterization of Chilean propolis and biological activity on cariogenic bacteria Streptococcus mutans and Streptococcus sobrinus. Braz. J. Microbiol. 2013, 44, 577–585. [Google Scholar] [CrossRef]
- Martins, M.L.; Leite, K.L.; Pacheco-Filho, E.F.; Pereira, A.F.; Romanos, M.T.V.; Maia, L.C.; Cavalcanti, Y.W. Efficacy of red propolis hydro-alcoholic extract in controlling Streptococcus mutans biofilm build-up and dental enamel demineralization. Arch. Oral Biol. 2018, 93, 56–65. [Google Scholar] [CrossRef] [PubMed]
- Righi, A.A.; Alves, T.R.; Negri, G.; Marques, L.M.; Breyer, H.; Salatino, A. Brazilian red propolis: Unreported substances, antioxidant and antimicrobial activities. J. Sci. Food Agric. 2011, 91, 2363–2370. [Google Scholar] [CrossRef] [PubMed]
- De Marco, S.; Piccioni, M.; Pagiotti, R.; Pietrella, D. Antibiofilm and antioxidant activity of propolis and bud poplar resins versus Pseudomonas aeruginosa. Evid. Based Complement. Alternat. Med. 2017, 2017, 5163575. [Google Scholar] [CrossRef] [PubMed]
- Campana, R.; Patrone, V.; Franzini, I.T.; Diamantini, G.; Vittoria, E.; Baffone, W. Antimicrobial activity of two propolis samples against human Campylobacter jejuni. J. Med. Food. 2009, 12, 1050–1056. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |
| Bacteria | Solvent | Average (min.–max.) MIC Value µg/mL | Ref. |
|---|---|---|---|
| Staphylococcus aureus | DCM | 364 (16–950) | [72,73] |
| DMSO | 930 (50–950) | [6,74] | |
| EEP | 457 (8–3100) | [16,60,70,72,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88] | |
| Hexan | 258 (16–500) | [72] | |
| MeEP | 266 (63–1000) | [89] | |
| WEP | 883 (565–1200) | [86] | |
| Staphylococcus epidermidis | DCM | 900 | [73] |
| EEP | 345 (8–1135) | [75,76,82,90] | |
| Streptococcus mutans | EEP | 511 (4–4025) | [16,75,76,91,92,93] |
| Streptococcus “viridans” | EEP | 682 (150–1370) | [76] |
| Streptococcus pyogenes | EEP | 534 (80–1556) | [60,70,86,94] |
| WEP | 1078 (600–1556) | [86] | |
| Streptococcus pneumoniae | EEP | 153 (80–300) | [86] |
| WEP | 1003 (600–1556) | [86] | |
| Streptococcus oralis | EEP | 167 (100–300) | [86] |
| WEP | 1070 (940–1200) | [86] | |
| Streptococcus agalactiae | EEP | 333 (100–600) | [86] |
| WEP | 2150 (600–3693) | [86] | |
| Streptococcus sobrinus | EEP | 5 (2–8) | [75] |
| Enterococcus spp. | DMSO | 1600 | [74] |
| EEP | 544 (2–1600) | [16,70,74,75,80,82,85,86,94] | |
| SCEP | 698 (63–1000) | [85] | |
| WEP | 250 | [86] | |
| Micrococcus luteus | DCM | 35 (8–63) | [72] |
| EEP | 117 (4–400) | [72,75,80] | |
| Hexan | 254 (8–901) | [72] | |
| Bacillus subtilis | DCM | 39 (16–62.5) | [72] |
| EEP | 180 (21–300) | [72,86,94] | |
| Hexan | 266 (31–500) | [72] | |
| WEP | 250 | [86] | |
| Clostridium difficile | EEP | 1840 | [71] |
| Strain of bacteria | Solvent | Average (min.–max.) MIC Value µg/mL | Ref. |
|---|---|---|---|
| Escherichia coli | DMC | 1340 | [73] |
| DSMO | 3648 (3190–4940) | [6,74] | |
| EEP | 784 (16–5000) | [70,71,74,75,76,77,84,86,88,94] | |
| MeEP | 303 (31–1000) | [89] | |
| WEP | 2500 | [86] | |
| Salmonella spp. | EEP | 2962 (32–14700) | [75,86,94] |
| MeEP | 265 (62–1000) | [89] | |
| WEP | 2500 | [86] | |
| Klebsiella spp. | DCM | 1030 | [73] |
| EEP | 1006 (32–3330) | [70,71,76,82,85,86,94] | |
| WEP | 2067 (1200–2500) | [86] | |
| Yersinia enterocolitica | EEP | 1633 (1200–2500) | [86] |
| MeEP | 171 (63–500) | [89] | |
| Proteus mirabilis | EEP | 1947 (512–3080) | [82,94] |
| MeEP | 618 (250–1000) | [89] | |
| Shigella flexneri | EEP | 1133 (300–2500) | [86] |
| WEP | 2500 | [86] | |
| Enterobacter cloacae | DMC | 1150 | [73] |
| EEP | 1926 (300–5000) | [76,86] | |
| WEP | 2500 | [86] | |
| Enterobacter aerogenes | EEP | 34 (8–64) | [75] |
| Pseudomonas aeruginosa | DCM | 1100 | [73] |
| DSMO | 2310 (1560–2810) | [6] | |
| EEP | 1252 (32–7910) | [75,76,82,84,86,94,95] | |
| MeEP | 180 (63–500) | [89] | |
| WEP | 2500 | [86] | |
| Acinetobacter baumannii | EEP | 5000 | [86] |
| Haemophilus influenzae | EEP | 1433 (600–2500) | [86] |
| WEP | 2500 | [86] | |
| Campylobacter jejuni | EEP | 256 (170–340) | [70,96] |
| Bacteroides fragilis | EEP | 2460 (1840–3700) | [71] |
| Burkholderia cepacia | EEP | 2467 (1200–5000) | [86] |
| WEP | 2500 | [86] |
| MIC Value [µg/mL] | Ref. | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Strain | B. subtilis | C. difficile | Enterococcus spp. | M. luteus | S. aureus | S. epidermidis | S. agalactiae | S. mutans | S. oralis | S. pneumoniae | S. pyogenes | S. sorbinus | S. “viridans” | ||
| Country | |||||||||||||||
| Australia | - | - | - | - | 1200 | - | - | - | - | - | - | - | - | [83] | |
| Brazil | 134 | - | 631 | 258 | 612 | 825 | - | 123 | - | - | 512 | - | - | [72,74,80–82,84,85,93,94] | |
| Bulgaria | - | - | - | - | 125 | - | - | - | - | - | - | - | - | [77] | |
| Chile | - | - | - | - | 1445 | - | - | 4 | - | - | 1470 | - | - | [60,92] | |
| Czech Republic | 300 | - | 250 | - | 600 | - | 300 | - | 100 | 80 | 80 | - | - | [86] | |
| Germany | 300 | - | 250 | - | 750 | - | 600 | - | 300 | 300 | 600 | - | - | [86] | |
| Greece | - | - | - | - | 393 | 296 | - | 602 | - | - | - | - | 682 | [76] | |
| India | - | - | - | - | 500 | - | - | - | - | - | - | - | - | [79] | |
| Ireland | 80 | - | 500 | - | 545 | - | 100 | - | 100 | 80 | 80 | - | - | [86] | |
| Korea | - | 1840 | - | - | - | - | - | 35 | - | - | - | - | - | [91] | |
| Morocco | - | - | - | - | 360 | - | - | - | - | - | - | - | - | [87] | |
| Oman | - | - | - | - | 81 | - | - | - | - | - | - | - | - | [77] | |
| Poland | - | - | - | - | 555 | 1135 | - | - | - | - | - | - | - | [78,90] | |
| Slovakia | 170 | - | 1400 | - | 255 | - | - | - | - | - | 1400 | - | - | [70] | |
| Taiwan | - | - | - | - | 10 | - | - | - | - | - | - | - | - | [88] | |
| Turkey | - | - | 19 | 11 | 8 | 20 | - | 42 | - | - | - | 5 | - | [75] | |
| MIC Value [µg/mL] | Ref. | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Strain | A. baumani | B. cepacia | B. frgilis | C. jejuni | E. cloacae | E. aerogenes | E. coli | H. influenzae | Klebsiella spp. | P. mirabilis | P. aeruginosa | Salmonella spp. | S. flexneri | Y. enterocolitica | ||
| Country | ||||||||||||||||
| Brazil | - | - | - | - | - | - | 571 | - | 961 | 1947 | 2293 | 512 | - | - | [74,82,84,85,94] | |
| Bulgaria | - | - | - | - | - | - | 1000 | - | - | - | - | - | - | - | [77] | |
| Czech Republic | 5000 | 1200 | - | - | 5000 | - | 600 | 1200 | 1850 | - | 1200 | 5000 | 600 | 1200 | [86] | |
| Germany | 5000 | 5000 | - | - | 5000 | - | 5000 | 2500 | 1500 | - | 2500 | 5000 | 2500 | 2500 | [86] | |
| Greece | - | - | - | - | 931 | - | 902 | - | 894 | - | - | - | - | - | [76] | |
| Ireland | 5000 | 1200 | - | - | 5000 | - | 1200 | 600 | 900 | - | 600 | 5000 | 300 | 1200 | [86] | |
| Italy | - | - | - | 260 | - | - | - | - | - | - | 125 | - | - | - | [95,96] | |
| Korea | - | - | 2460 | - | - | - | 1840 | - | - | - | - | 1470 | - | - | [71] | |
| Oman | - | - | - | - | - | - | 302 | - | - | - | - | - | - | - | [77] | |
| Slovakia | - | - | - | 255 | - | - | 510 | - | - | - | - | 1140 | - | - | [70] | |
| Taiwan | - | - | - | - | - | - | 640 | - | - | - | - | - | - | - | [88] | |
| Turkey | - | - | - | - | - | 34 | 116 | - | - | - | 120 | 72 | - | - | [75] | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Przybyłek, I.; Karpiński, T.M. Antibacterial Properties of Propolis. Molecules 2019, 24, 2047. https://doi.org/10.3390/molecules24112047
Przybyłek I, Karpiński TM. Antibacterial Properties of Propolis. Molecules. 2019; 24(11):2047. https://doi.org/10.3390/molecules24112047
Chicago/Turabian StylePrzybyłek, Izabela, and Tomasz M. Karpiński. 2019. "Antibacterial Properties of Propolis" Molecules 24, no. 11: 2047. https://doi.org/10.3390/molecules24112047
APA StylePrzybyłek, I., & Karpiński, T. M. (2019). Antibacterial Properties of Propolis. Molecules, 24(11), 2047. https://doi.org/10.3390/molecules24112047







