Chemical Analysis of Lepidium meyenii (Maca) and Its Effects on Redox Status and on Reproductive Biology in Stallions †
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Extraction and Chemical Analysis of Maca
2.3. Animals and Experimental Design
2.4. Systemic Oxidative Stress Status
2.5. Statistical Analysis
3. Results
3.1. Chemical Analysis of Maca
3.2. Oxidative Stress Measurements in Blood Samples
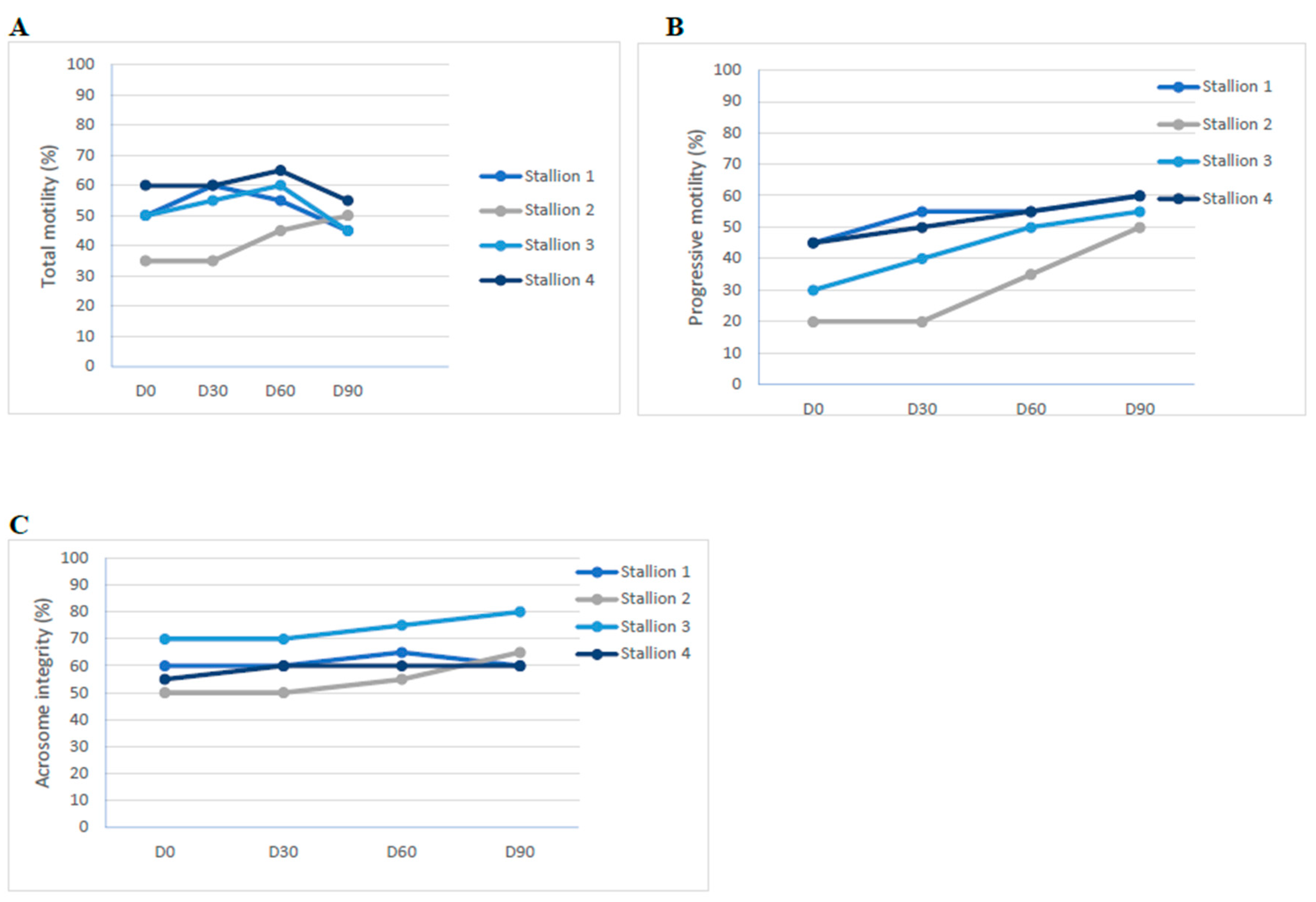
3.3. Semen Analysis
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gonzales, G.F.; Ruiz, A.; Gonzales, C.; Villegas, L.; Cordova, A. Effect of Lepidium meyenii (Maca) roots on spermatogenesis of male rats. Asian J. Androl. 2001, 3, 231–233. [Google Scholar]
- Canales, M.; Aguilar, J.; Prada, A.; Marcelo, A.; Huamán, C.; Carbajal, L. Nutritional evaluation of Lepidium meyenii (MACA) in albino mice and their descendants. Arch. Latinoam. Nutr. 2000, 50, 126–133. [Google Scholar]
- Gonzales, G.F. Ethnobiology and ethnopharmacology of Lepidium meyenii (Maca), a plant from the Peruvian Highlands. Evid. Based Compl. Alt. Med. 2012, 2012, 1–10. [Google Scholar] [CrossRef]
- Gonzales, G.F.; Miranda, S.; Nieto, J.; Fernandez, G.; Yucra, S.; Rubio, J.; Yi, P.; Gasco, M. Red maca (Lepidium meyenii) reduced prostate size in rats. Reprod. Biol. Endrocrinol. 2005, 20, 3–5. [Google Scholar]
- Oshima, M.; Gu, Y.; Tsukada, S. Effects of Lepidium meyenii Walp and Jatropha macrantha on blood levels of estradiol-17 beta, progesterone, testosterone and the rate of embryo implantation in mice. J. Vet. Med. Sci. 2003, 65, 1145–1146. [Google Scholar] [CrossRef]
- Valentova, K.; Buckiova, D.; Kren, V.; Peknicova, J.; Ulrichova, J.; Simanek, V. The in vitro biological activity of Lepidium meyenii extracts. Cell Biol. Toxicol. 2006, 22, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Gonzales, G.F.; Cordova, A.; Gonzales, C.; Chung, A.; Vega, K.; Villena, A. Lepidium meyenii (Maca) improved semen parameters in adult men. Asian J. Androl. 2001, 3, 301–303. [Google Scholar]
- Gonzales, G.F.; Gasco, M.; Córdova, A.; Chung, A.; Rubio, J.; Villegas, L. Effect of Lepidium meyenii (Maca) on spermatogenesis in male rats acutely exposed to high altitude (4340 m). J. Endocrinol. 2004, 180, 87–95. [Google Scholar] [CrossRef]
- Del Prete, C.; Cocchia, N.; Ciani, F.; Tafuri, S.; Carotenuto, D.; Napoleone, G.; Pasolini, M.P. Influence of a diet supplementation with Lepidium meyenii on quality of cooled equine semen. Reprod. Domest. Anim. 2015, 50, 30. [Google Scholar] [CrossRef]
- Del Prete, C.; Tafuri, S.; Ciani, F.; Pasolini, M.P.; Ciotola, F.; Albarella, S.; Carotenuto, D.; Peretti, V.; Cocchia, N. Influences of dietary supplementation with Lepidium meyenii (Maca) on stallion sperm production and on preservation of sperm quality during storage at 5 °C. Andrology 2018, 6, 351–361. [Google Scholar] [CrossRef] [PubMed]
- Ciani, F.; Cocchia, N.; d’Angelo, D.; Tafuri, S. Influence of ROS on Ovarian Functions. In New Discoveries in Embryology; Wu, B., Ed.; Intechopen: London, UK, 2015; pp. 41–73. [Google Scholar] [CrossRef]
- Mathur, P.P.; D’Cruz, S.C. The effect of environmental contaminants on testicular function. Asian J. Androl. 2011, 13, 585–591. [Google Scholar] [CrossRef]
- Tafuri, S.; Ciani, F.; Iorio, E.L.; Esposito, L.; Cocchia, N. Reactive oxygen species (ROS) and male fertility. In New Discoveries in Embryology; Wu, B., Ed.; Intechopen: London, UK, 2015; pp. 19–40. [Google Scholar] [CrossRef]
- Agarwal, A.; Said, T.M. Role of sperm chromatin abnormalities and DNA damage in male infertility. Hum. Reprod. Update 2003, 9, 331–345. [Google Scholar] [CrossRef]
- Sies, H.; Mehlhorn, R. Mutagenicity of nitroxide-free radicals. Arch. Biochem. Biophys. 1986, 251, 393–396. [Google Scholar] [CrossRef]
- Costantino, M.; Giuberti, G.; Caraglia, A.; Caraglia, M.; Lombardi, A.; Misso, G.; Abbruzzese, A.; Ciani, F.; Lampa, E. Possible antioxidant role of SPA therapy with chlorine-sulphur-bicarbonate mineral water. Amino Acids 2009, 36, 161–165. [Google Scholar] [CrossRef]
- Del Prete, C.; Ciani, F.; Tafuri, S.; Pasolini, M.P.; Valle, G.D.; Palumbo, V.; Abbondante, L.; Calamo, A.; Barbato, V.; Gualtieri, R.; et al. Effect of superoxide dismutase, catalase, and glutathione peroxidase supplementation in the extender on chilled semen of fertile and hypofertile dogs. J. Vet. Sci. 2018, 19, 667–675. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Li, P.; Brantner, A.; Wang, H.; Shu, X.; Yang, J.; Si, N.; Han, L.; Zhao, H.; Bian, B. Chemical profiling analysis of Maca using UHPLC-ESI-Orbitrap MS coupled with UHPLC-ESI-QqQ MS and the neuroprotective study on its active ingredients. Sci. Rep. 2017, 7, 44660. [Google Scholar] [CrossRef] [PubMed]
- Vinale, F.; Strakowska, J.; Mazzei, P.; Piccolo, A.; Marra, R.; Lombardi, N.; Manganiello, G.; Pascale, A.; Woo, S.L.; Lorito, M. Cremenolide, a new antifungal, 10-member lactone from Trichoderma cremeum with plant growth promotion activity. Nat. Prod. Res. 2016, 31, 2207. [Google Scholar] [CrossRef] [PubMed]
- Vinale, F.; Nicoletti, R.; Lacatena, F.; Marra, R.; Sacco, A.; Lombardi, N.; d’Errico, G.; Digilio, M.C.; Woo, S.L. Secondary metabolites from the endophytic fungus Talaromyces pinophilus. Nat. Prod. Res. 2017, 31, 1778–1785. [Google Scholar] [CrossRef]
- Bennet, R.N.; Mellon, F.A.; Kroon, P.A. Screening crucifer seeds as sources of specific intact glucosinolates using ion-pair High-Performance Liquid Chromatography Negative Ion Electrospray Mass Spectrometry. J. Agric. Food Chem. 2004, 52, 428–438. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Dou, J.; Wu, Z.; Yin, D.; Wu, W. Controlled release of biological control agents for preventing aflatoxin contamination from starch-alginate beads. Molecules 2019, 24, 1858. [Google Scholar] [CrossRef] [PubMed]
- Fahey, J.W.; Zalcmann, A.T.; Talalay, P. The chemical diversity and distribution of glucosinolates and isothiocyanates among plants. Phytochemistry 2001, 56, 5–51. [Google Scholar] [CrossRef]
- Gonzales, G.F.; Còrdova, A.; Vega, K.; Chung, A.; Villena, A.; Gònez, A.; Castillo, S. Effect of Lepidium meyenii (MACA) on sexual desire and its absent relationship with serum testosterone levels in adult healthy men. Andrologia 2002, 34, 367–372. [Google Scholar] [CrossRef] [PubMed]
- Zheng, B.L.; He, K.; Kim, C.H.; Rogers, L.; Shao, Y.; Huang, Z.Y.; Lu, Y.; Yan, S.J.; Qien, L.C.; Zheng, Q.Y. Effect of a lipidic extract from Lepidium meyenii on sexual behavior in mice and rats. Urology 2000, 55, 598–602. [Google Scholar] [CrossRef]
- Cicero, A.F.G.; Bandieri, E.; Arletti, R. Lepidium meyenii Walp. improves sexual behaviour in male rats independently from its action on spontaneous locomotor activity. J. Ethnopharmacol. 2001, 75, 225–229. [Google Scholar] [CrossRef]
- Cocchia, N.; Ciani, F.; El-Rass, R.; Russo, M.; Borzacchiello, G.; Esposito, V.; Montagnaro, S.; Avallone, L.; Tortora, G.; Lorizio, R. Cryopreservation of feline epididymal spermatozoa from dead and alive animals and its use in assisted reproduction. Zygote 2010, 18, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Cocchia, N.; Corteggio, A.; Altamura, G.; Tafuri, S.; Rea, S.; Rosapane, I.; Sica, A.; Landolfi, F.; Ciani, F. The effects of superoxide dismutase addition to the transport medium on cumulus–oocyte complex apoptosis and IVF outcome in cats (Felis catus). Reprod. Biol. 2015, 15, 56–64. [Google Scholar] [CrossRef]
- Tafuri, S.; Marullo, A.; Ciani, F.; Della Morte, R.; Montagnaro, S.; Fiorito, F.; De Martino, L. Reactive oxygen metabolites in alpha-herpesvirus-seropositive Mediterranean buffaloes (Bubalus bubalis): A preliminary study. Pol. J. Vet. Sci. 2018, 21, 639–642. [Google Scholar] [PubMed]
- Alberti, A.; Bolognini, L.; Macciantelli, D.; Carratelli, M. The radical cation of N,N-diethylpara-phenylendiamine: A possible indicator of oxidative stress in biological samples. Res. Chem. Intermediat. 2000, 26, 253–267. [Google Scholar] [CrossRef]
- Muhammad, I.; Zhao, J.; Khan, I.A. Maca (Lepidium meyenii). In Encyclopedia of Dietary Supplement, 2nd ed.; Coates, P., Blackman, M.R., Cragg, G., Levine, M., Moss, J., White, J., Eds.; Markel Dekker Inc.: New York, NY, USA, 2010; Supplement 1, pp. 522–531. [Google Scholar] [CrossRef]
- Piacente, S.; Carbone, V.; Plaza, A.; Zampelli, A.; Pizza, C. Investigation of the tuber constituents of Maca (Lepidium meyenii Walp.). J. Agric. Food Chem. 2002, 50, 5621–5625. [Google Scholar] [CrossRef]
- Esparza, E.; Hadzich, A.; Kofer, W.; Mithofer, A.; Cosio, E.G. Bioactive maca (Lepidium meyenii) alkamides are a result of traditional Andean post harvest drying practices. Phythochemistry 2015, 116, 138–148. [Google Scholar] [CrossRef]
- Zhao, J.; Muhammad, I.; Dunbar, C.; Mustafa, J.; Khan, I.A. New alkamides from Maca (Lepidium meyenii). J. Agric. Food Chem. 2005, 53, 690–693. [Google Scholar] [CrossRef]
- Chain, F.E.; Grau, A.; Martins, J.C.; Catalan, C.A.N. Macamides from wild “Maca”, Lepidium meyenii Walpers (Brassicaceae). Phytochem. Lett. 2014, 8, 145–148. [Google Scholar] [CrossRef]
- Martınez-Soto, J.C.; Landeras, J.; Gadea, J. Spermatozoa and seminal plasma fatty acids as predictors of cryopreservation success. Andrology 2013, 1, 365–375. [Google Scholar] [CrossRef]
- Fazio, F.; Casella, S.; Giannetto, C.; Caola, G.; Piccione, G. Serum homocysteine and oxidative stress evaluation during exercise in horse. Pol. J. Vet. Sci. 2009, 12, 169–174. [Google Scholar]
- Melnikovova, I.; Fait, T.; Kolarova, M.; Fernandez, E.C.; Milella, L. Effect of Lepidium meyenii Walp. on Semen Parameters and Serum Hormone Levels in Healthy Adult Men: A Double-Blind, Randomized, Placebo-Controlled Pilot Study. Evid. Based Complement. Alternat. Med. 2015, 324369. [Google Scholar]
Sample Availability: Not available. |

| No. | Name | RT (min) | ESI(+)/Expected (m/z) | ESI(+)/Measured (m/z) | Delta (ppm) | Main Fragment (m/z) | λmax (nm) |
|---|---|---|---|---|---|---|---|
| 1 * | (1R,3S)-1-methyltetrahydro-beta-5,6-hydride-carboline-3-carboxylic acid | 13.842 | 233.1284 | 233.1276 | 3.431585341 | 91.054 | 215.5 |
| 2 * | N-(3-hydroxy-benzyl)-2Z-fivecarbon acrylamide | 18.693 | 180.1019 | 180.1019 | 0.027 | 162.0914 | 220.1 |
| 3 * | 1-dibenzyl-2-propane-4,5-dimethylimidazilium | 18.997 | 227.1542 | 227.1546 | −1.76091835 | 91.0544 | 219.5 |
| 4 ** | 3-benzyl-1,2-dihydro-N-hydroxypyridine-4-carbaldehyde | 22.571 | 216.1175 | 216.1173 | 0.916936972 | 91.0543 | 213.2 |
| 5 * | 1,3-dibenzyl-2,4,5-trimethylimidazilium | 24.574 | 291.1854 | 291.1851 | 1.030271435 | 91.054 | 214.1 |
| 6 * | 1,3-dibenzyl-2-phenyl-4,5-dimethylimidazilium | 27.749 | 353.2014 | 353.2013 | 0.283124586 | 185.1072 | 221.1 |
| 7 ** | N-ethyl-tetradecene ester | 28.187 | 274.274 | 274.2743 | −1.09379671 | 256.2637 | 221.2 |
| 8 ** | 1,3-dibenzyl-2-pentyl-4,5-dimethylimidazilium | 29.578 | 347.2481 | 347.2482 | −0.28797854 | 277.1705 | 222.7 |
| 9 ** | 5-oxo-6E,8E-octadecadienoic acid | 40.444 | 295.2267 | 295.2266 | 0.338722751 | 277.2165 | 223.9 |
| 10 ** | N-benzyl-5-oxo-6E,8E-octadecadienamide | 43.789 | 384.2824 | 384.2822 | 0.520450585 | 306.2421 | 224.3 |
| 11 * | N-benzyl-9-oxo-12E-octadecenamide | 44.976 | 386.2981 | 386.2979 | 0.517734879 | 261.2212 | 223.8 |
| 12 * | N-(3-methoxybenzyl)-(9Z,12Z,15Z)-octadecatrienamide | 48.505 | 398.298 | 398.2976 | 1.004273182 | 381.2785 | 223.5 |
| 13 * | N-benzyl-(9Z,12Z,15Z)-octadecatrienamide | 48.863 | 368.2875 | 368.288 | −1.357635 | 351.2678 | 223.9 |
| 14 * | N-(3-Methoxybenzyl)-(9Z,12Z)-octadecadienamide | 51.403 | 400.3137 | 400.3133 | 0.999216364 | 365.2838 | 224.3 |
| 15 * | N-Benzyl-(9Z,12Z)-octadecadienamide | 51.868 | 370.3031 | 370.3034 | −0.81014715 | 232.1694 | 224.7 |
| 16 *˟ | N-benzylhexadecanamide | 54.34 | 346.3031 | 346.3034 | −0.86629314 | 221.2261 | 221.3 |
| 17 * | N-benzyl-9-Z-octadecenamide | 55.169 | 372.3188 | 372.3194 | −1.61152217 | 247.2423 | 225.9 |
| 18 * | N-benzyloctadecanamide | 59.184 | 374.3345 | 374.3344 | 0.267140752 | 296.3172 | 225.5 |
| No. | Name | RT (min) | ESI(-)/Expected (m/z) | ESI(-)/Measured (m/z) | Delta (ppm) | Main Fragment (m/z) | λmax (nm) |
|---|---|---|---|---|---|---|---|
| 1 ** ˟ | Benzyl glucosinolate | 14.174 | 408.0423 | 408.0424 | −0.24507263 | 274.9903 241.0027 166.0333 | 217.2 |
| 2 ** ˟ | m-methoxybenzyl glucosinolate | 15.421 | 438.0534 | 438.054 | −1.36969602 | 358.0956 259.0126 | 219.4 |
| d-ROMs (U CARR) | OXY-Ads (μmol HClO/mL) | BAP (μEq/L) | -SHp (μM/L) | |||||
|---|---|---|---|---|---|---|---|---|
| C | M | C | M | C | M | C | M | |
| D0 | 336 | 316.8 | 325 | 335.8 | 1759.3 | 1844.4 | 532.7 | 568.2 |
| (±34.4) | (±33.5) | (±9.9) | (±12.13) | (±58.8) | (±88.2) | (±57.5) | (±32.5) | |
| D30 | 342.6 | 329.8 | 355.2 | 375.6 | 1652.4 | 1722.6 | 602.4 | 645 |
| (±37.3) | (±31) | (±26.3) | (±27.4) | (±122.7) | (±151.2) | (±70.0) | (±48.6) | |
| D60 | 315.8 | 299.6 | 354 | 402 | 1746.4 | 1902.5 | 579.8 | 706.2 |
| (±12.1) | (±14.3) | (±58.5) | (±33.8) | (±193.2) | (±138.5) | (±67.7) | (±95.7) | |
| D90 | 349.8 | 336.8 | 364.8 | 379.8 | 1580.8 | 1683 * | 469.5 | 554.6 |
| (±28.7) | (±29.9) | (±43.2) | (±52.7) | (±51.6) | (±65.6) | (±49.6) | (±56.3) | |
| OSi (%) | ||
|---|---|---|
| C | M | |
| D0 | 103.4 | 94.3 |
| D30 | 96.5 | 87.8 |
| D60 | 89.2 | 74.5 |
| D90 | 95.9 | 88.7 |
| D0 | D30 | D60 | D90 | |
|---|---|---|---|---|
| Ejaculate volume (mL) | 38.38 ± 12.12 | 48.13 ± 17.50 * | 60.88 ± 29.09 * | 47.5 ± 23.63 * |
| Sperm concentration (×106/mL) | 124.38 ± 43.05 | 220.00 ± 90.69 * | 178.88 ± 86.54 * | 279 ± 126.87 * |
| Total Sperm Count (×109) | 3.68 ± 1.18 | 10.15 ± 6.22 * | 8.20 ± 7.65 * | 8.83 ± 4.36 * |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tafuri, S.; Cocchia, N.; Carotenuto, D.; Vassetti, A.; Staropoli, A.; Mastellone, V.; Peretti, V.; Ciotola, F.; Albarella, S.; Del Prete, C.; et al. Chemical Analysis of Lepidium meyenii (Maca) and Its Effects on Redox Status and on Reproductive Biology in Stallions. Molecules 2019, 24, 1981. https://doi.org/10.3390/molecules24101981
Tafuri S, Cocchia N, Carotenuto D, Vassetti A, Staropoli A, Mastellone V, Peretti V, Ciotola F, Albarella S, Del Prete C, et al. Chemical Analysis of Lepidium meyenii (Maca) and Its Effects on Redox Status and on Reproductive Biology in Stallions. Molecules. 2019; 24(10):1981. https://doi.org/10.3390/molecules24101981
Chicago/Turabian StyleTafuri, Simona, Natascia Cocchia, Domenico Carotenuto, Anastasia Vassetti, Alessia Staropoli, Vincenzo Mastellone, Vincenzo Peretti, Francesca Ciotola, Sara Albarella, Chiara Del Prete, and et al. 2019. "Chemical Analysis of Lepidium meyenii (Maca) and Its Effects on Redox Status and on Reproductive Biology in Stallions" Molecules 24, no. 10: 1981. https://doi.org/10.3390/molecules24101981
APA StyleTafuri, S., Cocchia, N., Carotenuto, D., Vassetti, A., Staropoli, A., Mastellone, V., Peretti, V., Ciotola, F., Albarella, S., Del Prete, C., Palumbo, V., Esposito, L., Vinale, F., & Ciani, F. (2019). Chemical Analysis of Lepidium meyenii (Maca) and Its Effects on Redox Status and on Reproductive Biology in Stallions. Molecules, 24(10), 1981. https://doi.org/10.3390/molecules24101981











