Lignans in Spirits: Chemical Diversity, Quantification, and Sensory Impact of (±)-Lyoniresinol
Abstract
:1. Introduction
2. Results and Discussion
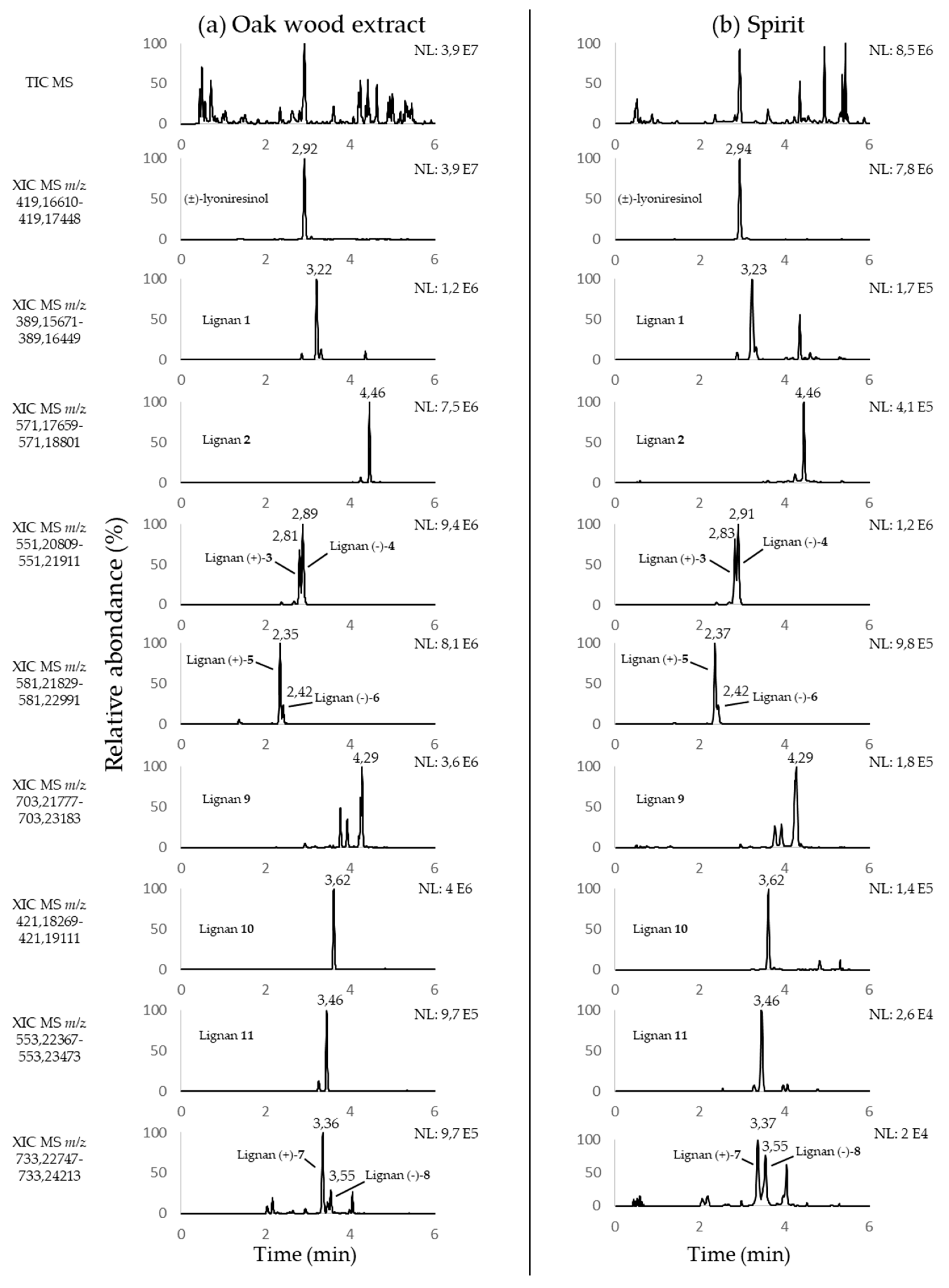
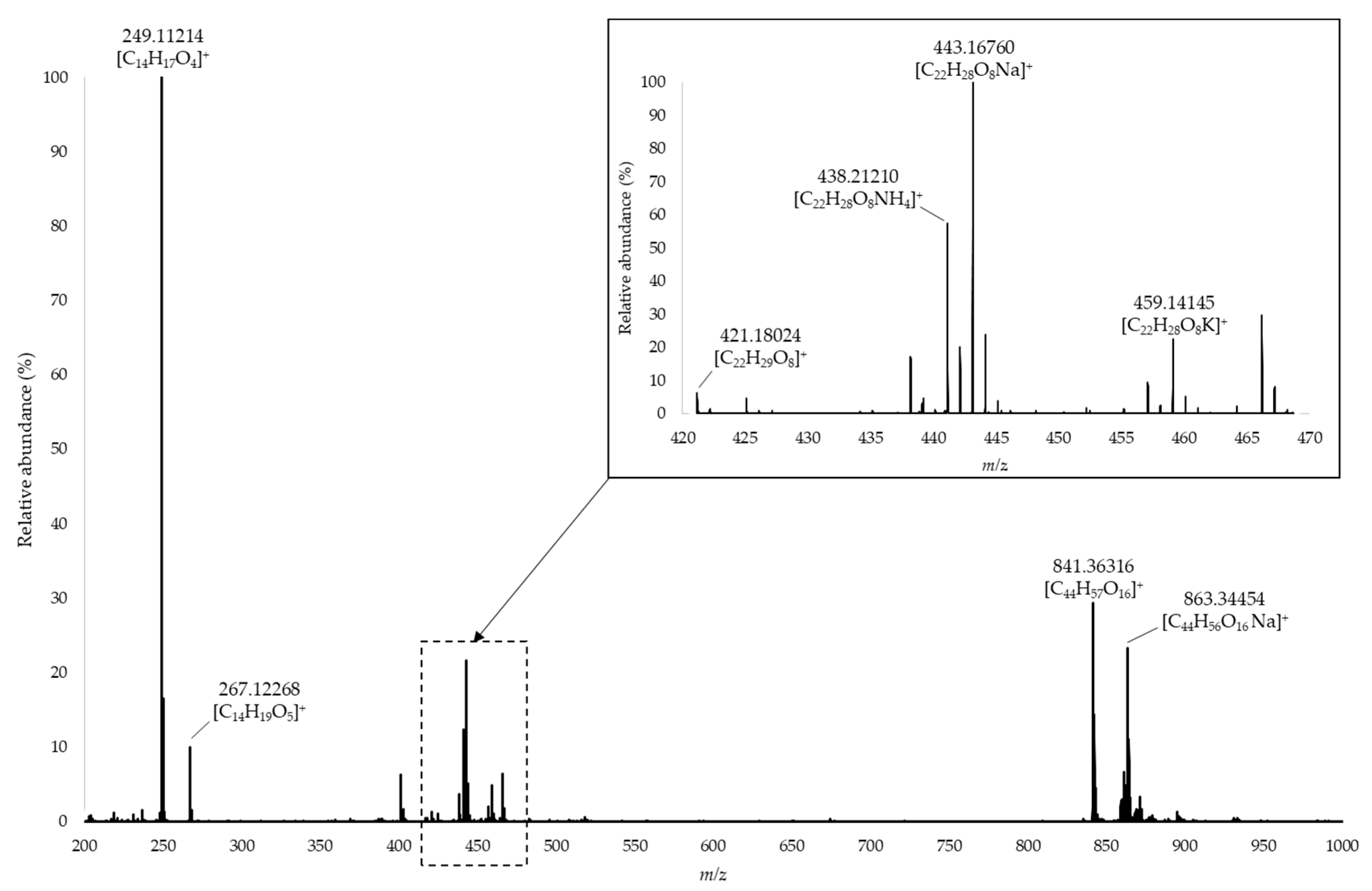
2.1. Chemical Diversity of Oak Lignans in Spirits
2.2. Sensory Impact and Quantification of (±)-Lyoniresinol in Spirits
2.2.1. Determination of (±)-Lyoniresinol Detection Threshold in Spirits
2.2.2. Development of an LC-HRMS Method to Quantitate Lyoniresinol in Spirits
Sensitivity
Linearity and Accuracy
Specificity
Repeatability and Trueness
2.2.3. Application of the Method to Quantitate Lyoniresinol in Spirits
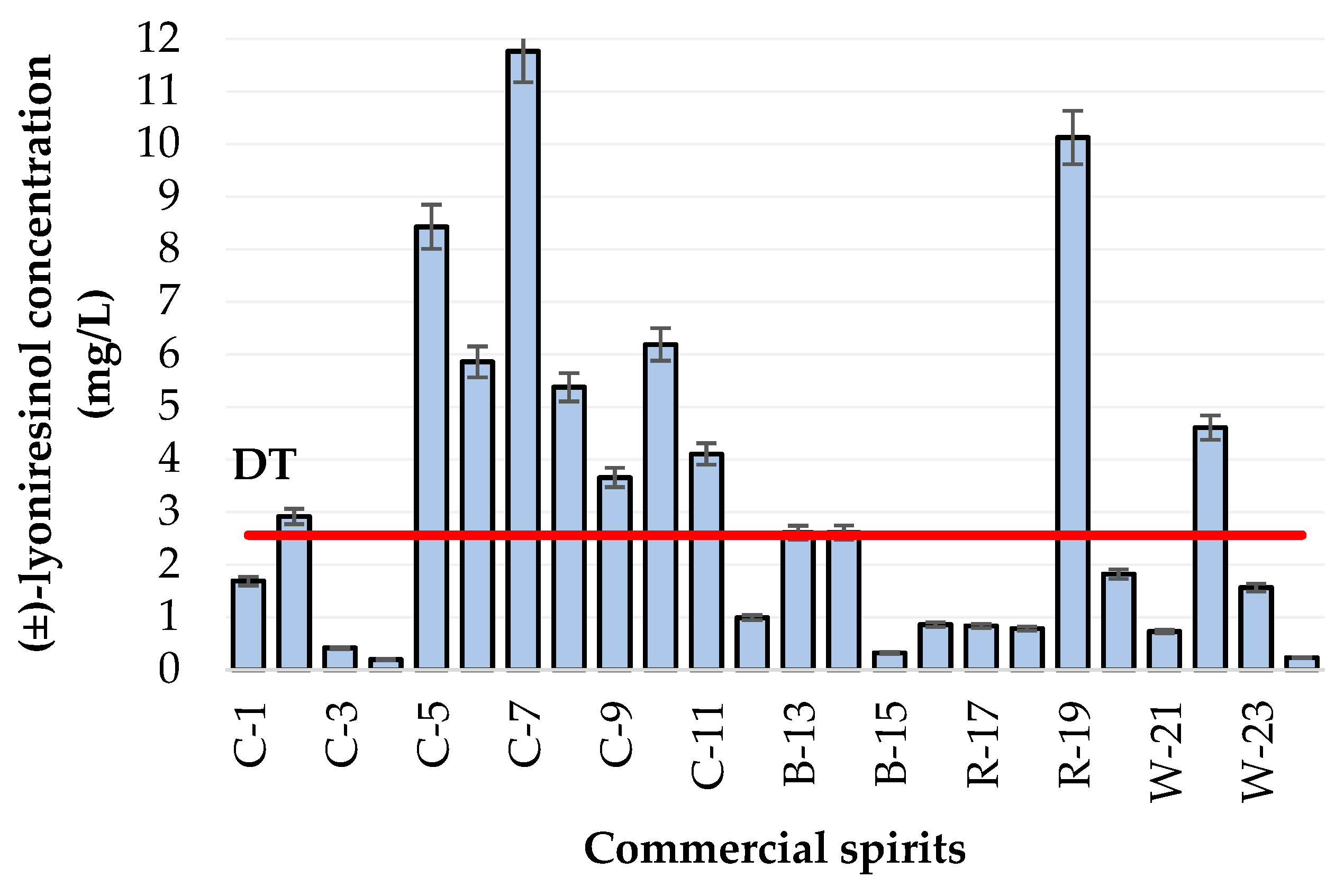
Content of Lyoniresinol in Various Commercial Spirits
Content of Lyoniresinol in Various Vintages of the Same Spirits
3. Materials and Methods
3.1. Chemicals
3.2. LC Analysis
3.3. HRMS
3.3.1. Screening
3.3.2. Quantification
3.4. Spirits and Sample Preparation
3.5. Preparation of Calibration Solution for Lignan Quantitation
3.6. Method Validation for Quantitation of Lyoniresinol on C18 Column
3.7. Sensory Analysis
3.7.1. Panel Training
3.7.2. Determination of Lyoniresinol Taste Threshold in Spirits
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cantagrel, R.; Mazerolles, G.; Vidal, J.; Galy, B. Élaboration et Connaissance des Spiritueux: Recherche de la qualité, tradition & innovation; Lavoisier—Tec & Doc.: Paris, France, 1992; ISBN 2-87777-357-4. [Google Scholar]
- MacNamara, K.; van Wyk, C.; Brunerie, P.; Augustyn, O.P.; Rapp, A. Flavour components of whiskey. III. ageing changes in the low-volatility fraction. South Afr. J. Enol. Vitic. 2001, 22, 82–92. [Google Scholar] [CrossRef]
- Piggott, J.R. Flavour of Distilled Beverages: Origin and Development; E. Horwood Limited: London, UK, 1983; ISBN 978-0-89573-131-9. [Google Scholar]
- Lafon, J.; Couillud, P.; Gaybellile, F. Le Cognac; J.B. Baillière: Paris, France, 1973. [Google Scholar]
- Ledauphin, J.; Le Milbeau, C.; Barillier, D.; Hennequin, D. Differences in the Volatile Compositions of French Labeled Brandies (Armagnac, Calvados, Cognac, and Mirabelle) Using GC-MS and PLS-DA. J. Agric. Food Chem. 2010, 58, 7782–7793. [Google Scholar] [CrossRef] [PubMed]
- Ledauphin, J.; Basset, B.; Cohen, S.; Payot, T.; Barillier, D. Identification of trace volatile compounds in freshly distilled Calvados and Cognac: Carbonyl and sulphur compounds. J. Food Compos. Anal. 2006, 19, 28–40. [Google Scholar] [CrossRef]
- Puentes, C.; Joulia, X.; Vidal, J.-P.; Esteban-Decloux, M. Simulation of spirits distillation for a better understanding of volatile aroma compounds behavior: Application to Armagnac production. Food Bioprod. Proc. 2018, 112, 31–62. [Google Scholar] [CrossRef]
- MacNamara, K.; Dabrowska, D.; Baden, M.; Helle, N. Advances in the ageing chemistry of distilled spirits matured in oak barrels. 2011, 6, 448–466. [Google Scholar]
- Mosedale, J.R.; Puech, J.-L. Wood maturation of distilled beverages. Trends Food Sci. Technol. 1998, 9, 95–101. [Google Scholar] [CrossRef]
- Piggott, J.R.; Conner, J.M.; Paterson, A.; Clyne, J. Effects on Scotch whisky composition and flavour of maturation in oak casks with varying histories. Int. J. Food Sci. Technol. 1993, 28, 303–318. [Google Scholar] [CrossRef]
- Puech, J.-L. Extraction of Phenolic Compounds from Oak Wood in Model Solutions and Evolution of Aromatic Aldehydes in Wines Aged in Oak Barrels. Am. J. Enol. Vitic. 1987, 38, 236–238. [Google Scholar]
- Puech, J.-L. Characteristics of Oak Wood and Biochemical Aspects of Armagnac Ageing. Am. J. Enol. Vitic. 1984, 35, 77–81. [Google Scholar]
- Chatonnet, P. Influence des procédés de tonnellerie et des conditions d’élevage sur la composition et la qualité des vins élevés en fûts de chêne. 1995. Available online: https://www.theses.fr/1995BOR20338 (accessed on 31 October 2018).
- Tominaga, T.; Blanchard, L.; Darriet, P.; Dubourdieu, D. A Powerful Aromatic Volatile Thiol, 2-Furanmethanethiol, Exhibiting Roast Coffee Aroma in Wines Made from Several Vitis v inifera Grape Varieties. J. Agric. Food Chem. 2000, 48, 1799–1802. [Google Scholar] [CrossRef]
- Marchal, A.; Génin, E.; Waffo-Téguo, P.; Bibès, A.; Da Costa, G.; Mérillon, J.-M.; Dubourdieu, D. Development of an analytical methodology using Fourier transform mass spectrometry to discover new structural analogs of wine natural sweeteners. Anal. Chem. Acta 2015, 853, 425–434. [Google Scholar] [CrossRef] [PubMed]
- Marchal, A.; Waffo-Téguo, P.; Génin, E.; Mérillon, J.-M.; Dubourdieu, D. Identification of New Natural Sweet Compounds in Wine Using Centrifugal Partition Chromatography–Gustatometry and Fourier Transform Mass Spectrometry. Anal. Chem. 2011, 83, 9629–9637. [Google Scholar] [CrossRef] [PubMed]
- Glabasnia, A.; Hofmann, T. Identification and Sensory Evaluation of Dehydro- and Deoxy-ellagitannins Formed upon Toasting of Oak Wood (Quercus alba L.). J. Agric. Food Chem. 2007, 55, 4109–4118. [Google Scholar] [CrossRef] [PubMed]
- Hufnagel, J.C.; Hofmann, T. Orosensory-Directed Identification of Astringent Mouthfeel and Bitter-Tasting Compounds in Red Wine. J. Agric. Food Chem. 2008, 56, 1376–1386. [Google Scholar] [CrossRef] [PubMed]
- Puech, J.-L. Phenolic compounds in oak wood extracts used in the ageing of brandies. J. Sci. Food Agric. 1988, 42, 165–172. [Google Scholar] [CrossRef]
- Puech, J.-L. Extraction and evolution of lignin products in armagnac matured in oak. 1981, 32, 111–114. [Google Scholar]
- Moutounet, M.; Puech, J.-L.; Rabier, P. Analysis by HPLC of Extractable Substances in Oak Wood. Available online: http://agris.fao.org/agris-search/search.do?recordID=FR19890112212 (accessed on 8 October 2018).
- Nabeta, K.; Yonekubo, J.; Miyake, M.; Obihiro, U.A.V.M. Phenolic Compounds from the Heartwood of European Oak (Quercus robur L.) and Brandy. Available online: http://agris.fao.org/agris-search/search.do?recordID=JP880129288 (accessed on 3 October 2018).
- Puech, J.L.; Moutonnet, M. Phenolic Compounds in an Ethanol-Water Extract of Oak Wood and in a Brandy. Available online: http://agris.fao.org/agris-search/search.do?recordID=US201301769211 (accessed on 8 October 2018).
- Ribéreau-Gayon, P.; Glories, Y.; Maujean, A.; Dubourdieu, D. Traité d’oenologie-Tome 2-6e éd.-Chimie du Vin. Stabilisation et Traitements; Dunod: Buchanan, NY, USA, 2012; ISBN 978-2-10-058875-6. [Google Scholar]
- Salagoity-Auguste, M.-H.; Tricard, C.; Sudraud, P. Dosage simultané des aldéhydes aromatiques et des coumarines par chromatographie liquide haute performance: Application aux vins et eaux-de-vie vieillis en fŭt de chĕne. J. Chromatogr. A 1987, 392, 379–387. [Google Scholar] [CrossRef]
- Marchal, A.; Cretin, B.N.; Sindt, L.; Waffo-Téguo, P.; Dubourdieu, D. Contribution of oak lignans to wine taste: Chemical identification, sensory characterization and quantification. Tetrahedron 2015, 71, 3148–3156. [Google Scholar] [CrossRef]
- Cretin, B.N.; Sallembien, Q.; Sindt, L.; Daugey, N.; Buffeteau, T.; Waffo-Teguo, P.; Dubourdieu, D.; Marchal, A. How stereochemistry influences the taste of wine: Isolation, characterization and sensory evaluation of lyoniresinol stereoisomers. Anal. Chem. Acta 2015, 888, 191–198. [Google Scholar] [CrossRef]
- Cretin, B.N.; Dubourdieu, D.; Marchal, A. Development of a quantitation method to assay both lyoniresinol enantiomers in wines, spirits, and oak wood by liquid chromatography-high resolution mass spectrometry. Anal. Bioanal. Chem. 2016, 408, 3789–3799. [Google Scholar] [CrossRef]
- Chatonnet, P.; Dubourdieu, D.; Boidron, J.; Pons, M. The origin of ethylphenols in wines. J. Sci. Food Agric. 1992, 60, 165–178. [Google Scholar] [CrossRef]
- Gammacurta, M.; Tempere, S.; Marchand, S.; Moine, V.; De Revel, G. Ethyl 2-hydroxy-3-methylbutanoate enantiomers: Quantitation and sensory evaluation in wine. OENO One 2018, 52, 57–65. [Google Scholar] [CrossRef]
- Mackey, A.; Valassi, K. The discernement of primary tastes in the presence of different food textures. Food Technol. 1956, 10, 238–240. [Google Scholar]
- Poisson, L.; Schieberle, P. Characterization of the Key Aroma Compounds in an American Bourbon Whisky by Quantitative Measurements, Aroma Recombination, and Omission Studies. J. Agric. Food Chem. 2008, 56, 5820–5826. [Google Scholar] [CrossRef] [PubMed]
- Nonier, M.-F.; Vivas, N.; Vivas de Gaulejac, N.; Fouquet, E. Origin of brown discoloration in the staves of oak used in cooperage–Characterization of two new lignans in oak wood barrels. Comptes Rendus Chim. 2009, 12, 291–296. [Google Scholar] [CrossRef]
- Sindt, L.; Gammacurta, M.; Waffo-Teguo, P.; Dubourdieu, D.; Marchal, A. Taste-Guided Isolation of Bitter Lignans from Quercus petraea and Their Identification in Wine. J. Nat. Prod. 2016, 79, 2432–2438. [Google Scholar] [CrossRef] [PubMed]
- Arramon, G. Les triterpènes et lignanes des bois de chêne européen Quercus robur L. Et Quercus Petraea Liebl.: Quantification et apports qualitatifs aux eaux de vie d’Armagnac. Available online: https://www.theses.fr/2001BOR20882 (accessed on 6 October 2018).
- Seikel, M.K.; Hostettler, F.D.; Niemann, G.J. Phenolics of Quercus rubra wood. Phytochemistry 1971, 10, 2249–2251. [Google Scholar] [CrossRef]
- Koga, K.; Taguchi, A.; Koshimizu, S.; Suwa, Y.; Yamada, Y.; Shirasaka, N.; Yoshizumi, H. Reactive Oxygen Scavenging Activity of Matured Whiskey and Its Active Polyphenols. J. Food Sci. 2007, 72, S212–S217. [Google Scholar] [CrossRef]
- ISO 4120:2007 Sensory analysis-Methodology-Triangle test. In Analyse Sensorielle: Recueil, Normes, Agroalimentaire; AFNOR: Geneva, Switzerland, 2007.
- García, J.; Prieto, L.; Guevara, A.; Malagon, D.; Osorio, C. Chemical Studies of Yellow Tamarillo (Solanum betaceum Cav.) Fruit Flavor by Using a Molecular Sensory Approach. Molecules 2016, 21, 1729. [Google Scholar] [CrossRef]
- Li, M.; Liu, D.; Cong, X.-S.; Wu, J.-H.; Wu, C.-C.; Xia, W. Identification of Lignin-Derived p-Bis(2,6-dimethoxyphenol)yl Compounds in Bio-oil with Mass Spectrometry. Energy Fuels 2016, 30, 10950–10953. [Google Scholar] [CrossRef]
- Jenkins, R.W.; Sutton, A.D.; Robichaud, D.J. Chapter 8-Pyrolysis of Biomass for Aviation Fuel. In Biofuels for Aviation; Chuck, C.J., Ed.; Academic Press: Cambridge, MA, USA, 2016; pp. 191–215. ISBN 978-0-12-804568-8. [Google Scholar]
- De Paepe, D.; Servaes, K.; Noten, B.; Diels, L.; De Loose, M.; Van Droogenbroeck, B.; Voorspoels, S. An improved mass spectrometric method for identification and quantification of phenolic compounds in apple fruits. Food Chem. 2013, 136, 368–375. [Google Scholar] [CrossRef] [PubMed]
- Center for Veterinary Medicine (CVM). Guidance for Industry Bioanalytical Method Validation; Center for Drug Evaluation and Research (CDER), Food and Drug Administration, U.S. Department of Health and Human Services: Washington, DC, USA, 2018.
- De Rosso, M.; Cancian, D.; Panighel, A.; Dalla Vedova, A.; Flamini, R. Chemical compounds released from five different woods used to make barrels for ageing wines and spirits: Volatile compounds and polyphenols. Wood Sci. Technol. 2009, 43, 375–385. [Google Scholar] [CrossRef]
- Marchal, A.; Prida, A.; Dubourdieu, D. New Approach for Differentiating Sessile and Pedunculate Oak: Development of a LC-HRMS Method To Quantitate Triterpenoids in Wood. J. Agric. Food Chem. 2016, 64, 618–626. [Google Scholar] [CrossRef] [PubMed]
- Prida, A.; Boulet, J.-C.; Ducousso, A.; Nepveu, G.; Puech, J.-L. Effect of species and ecological conditions on ellagitannin content in oak wood from an even-aged and mixed stand of Quercus robur L. and Quercus petraea Liebl. Ann. For. Sci. 2006, 63, 415–424. [Google Scholar] [CrossRef]
- Canas, S.; Caldeira, I.; Mateus, A.M.; Belchior, A.P.; Clímaco, M.C.; Bruno-de, R. Effect of natural seasoning on the chemical composition of chestnut wood used for barrel making. Ciência e técnica vitivinícola 2006, 21, 1–16. [Google Scholar]
- Chatonnet, P.; Boidron, J.-N.; Dubourdieu, D.; Pons, M. Evolution de certains composés volatils du bois de chêne au cours de son séchage premiers résultats. OENO One 1994, 28, 359. [Google Scholar] [CrossRef]
- Fernández de Simón, B.; Cadahía, E.; del Álamo, M.; Nevares, I. Effect of size, seasoning and toasting in the volatile compounds in toasted oak wood and in a red wine treated with them. Anal. Chem. Acta 2010, 660, 211–220. [Google Scholar] [CrossRef] [PubMed]
- Mosedale, J.R.; Puech, J.-L.; Feuillat, F. The Influence on Wine Flavor of the Oak Species and Natural Variation of Heartwood Components. Am. J. Enol. Vitic. 1999, 50, 503–512. [Google Scholar]
- Snakkers, G.; Boulesteix, J.-M.; Estréguil, S.; Gaschet, J.; Lablanquie, O.; Faure, A.; Cantagrel, R. Effect of oak wood heating on cognac spirit matured in new barrel: A pilot study. OENO One 2003, 37, 243. [Google Scholar] [CrossRef]
- Sarni, F.; Moutounet, M.; Puech, J.L.; Rabier, P. Effect of heat treatment of oak wood on extractable compounds. Holzforsch.-Int. J. Biol. Chem. Phys. Technol. Wood 1990, 44, 461–466. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds 1, 2, 3, 5, 6, 10 and lyoniresinol are available from the authors. |

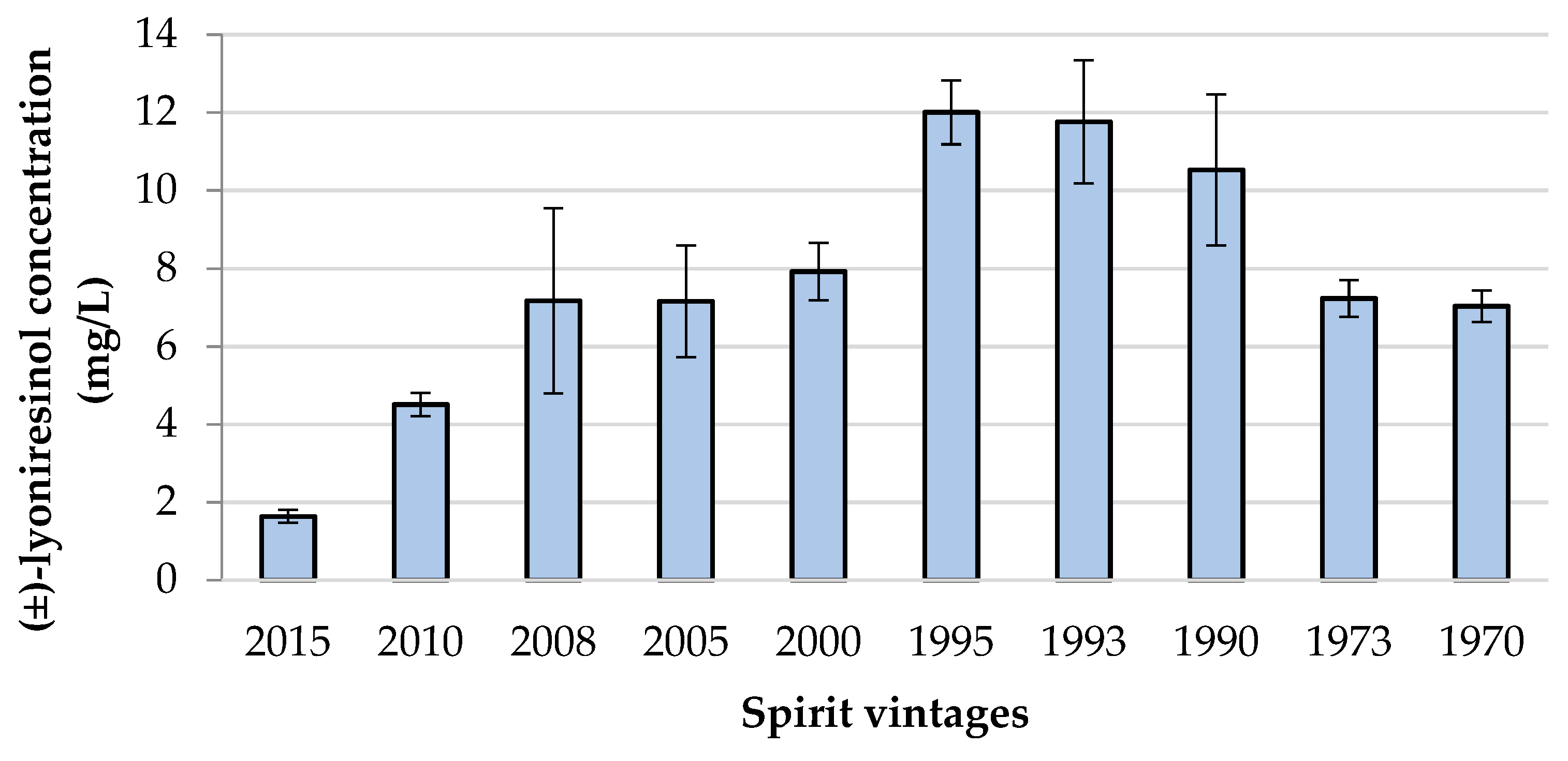
| Parameters | Matrix/Spirits | |||
|---|---|---|---|---|
| Sensitivity | IDL (µg/L) | IQL (µg/L) | LOD (µg/L) | LOQ (µg/L) |
| 5 | 10 | 25 | 50 | |
| Linearity and Accuracy | Working Range | R2 | ||
| 10 µg/L–10 mg/L | 0.9999 | |||
| Specificity | tR variation | Mass Accuracy | ||
| <0.04 min | <2.2 ppm | |||
| Repeatability and Trueness | Intraday Repeatability | |||
| 100 µg/L | 1 mg/L | |||
| 3.35% | 3.43% | |||
| Recovery | ||||
| Samples | 100 µg/L | 1 mg/L | 2 mg/L | |
| EDV-C7 | 94% | 91% | 98% | |
| EDV-C8 | 87% | 85% | 97% | |
| EDV-1995 | 88% | 86% | 87% | |
| Mass Spectrometer | Exactive | |
|---|---|---|
| Use | LC-MS Screening | LC-MS Quantification |
| Ionization Mode | Negative | Positive |
| Sheath gas flow a | 75 | 75 |
| Auxiliary gas flow a | 18 | 20 |
| HESI probe temperature | 320 °C | 320 °C |
| Capillary temperature | 350 °C | 350 °C |
| Electrospray voltage | −3 kV | 3.5 kV |
| Capillary voltage | −60 V | 35 V |
| Tube lens voltage offset | −135 V | 120 V |
| Skimmer voltage | −26 V | 18 V |
| Mass range (in Th) | 200–800 | 200–800 |
| Resolution b | 25 000 | 25 000 |
| Automatic gain control value | 106 | 106 |
| Samples | Brands | Type | Origin | ABV (Alcohol by Volume) |
|---|---|---|---|---|
| C-1 | Brand A | Cognac | France | 40 |
| C-2 | Brand A | Cognac | France | 40 |
| C-3 | Brand A | Cognac | France | 40 |
| C-4 | Brand A | Cognac | France | 40 |
| C-5 | Brand B | Cognac | France | 40 |
| C-6 | Brand B | Cognac | France | 40 |
| C-7 | Brand C | Cognac | France | 40 |
| C-8 | Brand C | Cognac | France | 40 |
| C-9 | Brand C | Cognac | France | 40 |
| C-10 | Brand D | Cognac | France | 40 |
| C-11 | Brand D | Cognac | France | 40 |
| C-12 | Brand E | Cognac | France | 42 |
| B-13 | Brand F | Brandy | South Africa | 38 |
| B-14 | Brand G | Brandy | France | 40 |
| B-15 | Brand H | Brandy | Germany | 38 |
| B-16 | Brand I | Brandy | France | 40 |
| R-17 | Brand J | Rum | Jamaica | 43 |
| R-18 | Brand K | Rum | Guyana | 40 |
| R-19 | Brand L | Rum | Barbados | 43 |
| W-20 | Brand M | Whisky | Ireland | 40 |
| W-21 | Brand N | Whisky | Scotland | 40 |
| W-22 | Brand O | Whisky | Scotland | 40 |
| W-23 | Brand O | Whisky | Scotland | 40 |
| Bo-24 | Brand P | Bourbon | United States | 50 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Winstel, D.; Marchal, A. Lignans in Spirits: Chemical Diversity, Quantification, and Sensory Impact of (±)-Lyoniresinol. Molecules 2019, 24, 117. https://doi.org/10.3390/molecules24010117
Winstel D, Marchal A. Lignans in Spirits: Chemical Diversity, Quantification, and Sensory Impact of (±)-Lyoniresinol. Molecules. 2019; 24(1):117. https://doi.org/10.3390/molecules24010117
Chicago/Turabian StyleWinstel, Delphine, and Axel Marchal. 2019. "Lignans in Spirits: Chemical Diversity, Quantification, and Sensory Impact of (±)-Lyoniresinol" Molecules 24, no. 1: 117. https://doi.org/10.3390/molecules24010117
APA StyleWinstel, D., & Marchal, A. (2019). Lignans in Spirits: Chemical Diversity, Quantification, and Sensory Impact of (±)-Lyoniresinol. Molecules, 24(1), 117. https://doi.org/10.3390/molecules24010117






