Identification, Recombinant Expression, and Characterization of LGH2, a Novel Antimicrobial Peptide of Lactobacillus casei HZ1
Abstract
1. Introduction
2. Results
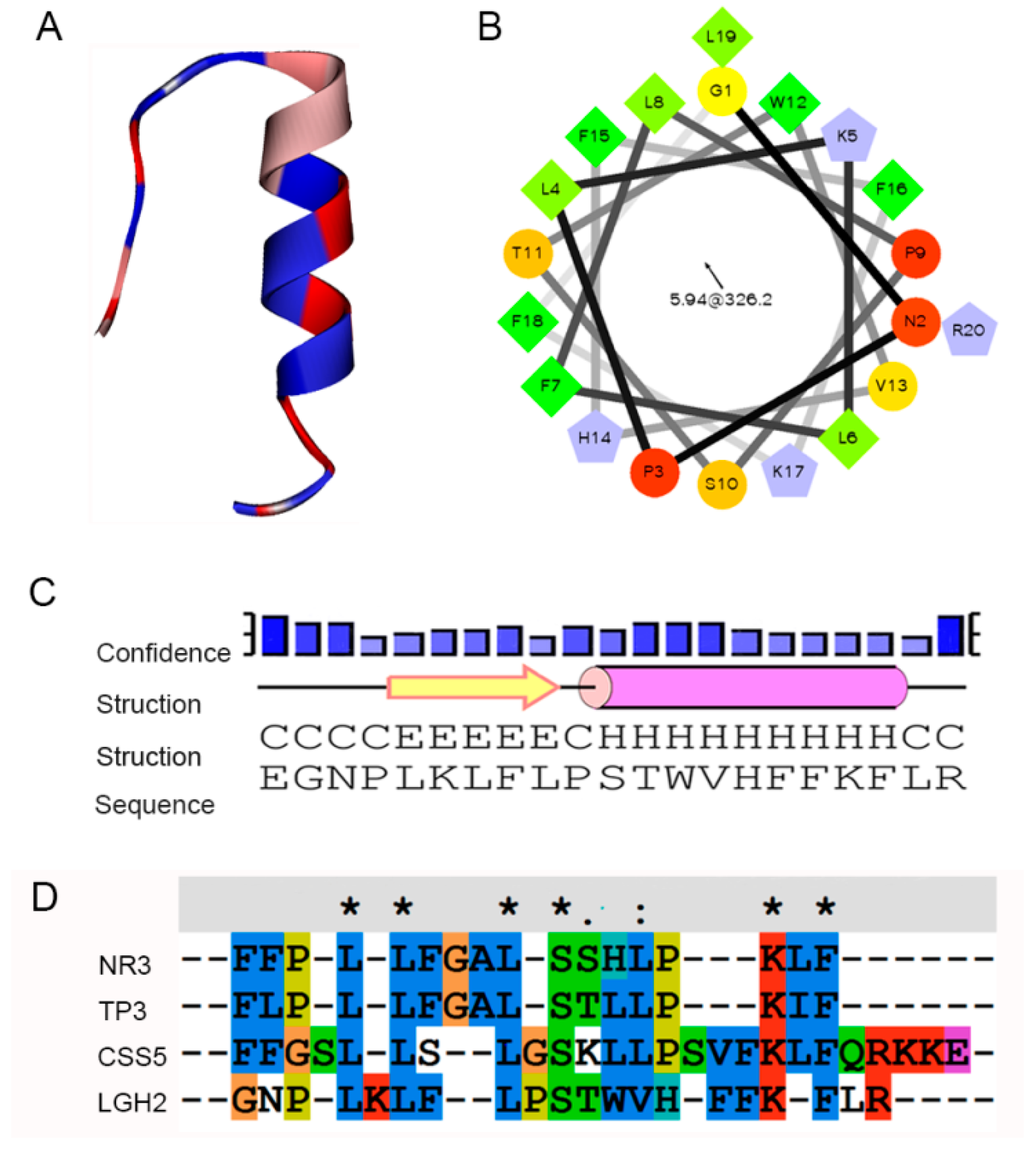
2.1. AMPs Prediction
2.2. Antimicrobial Test of the AMPs
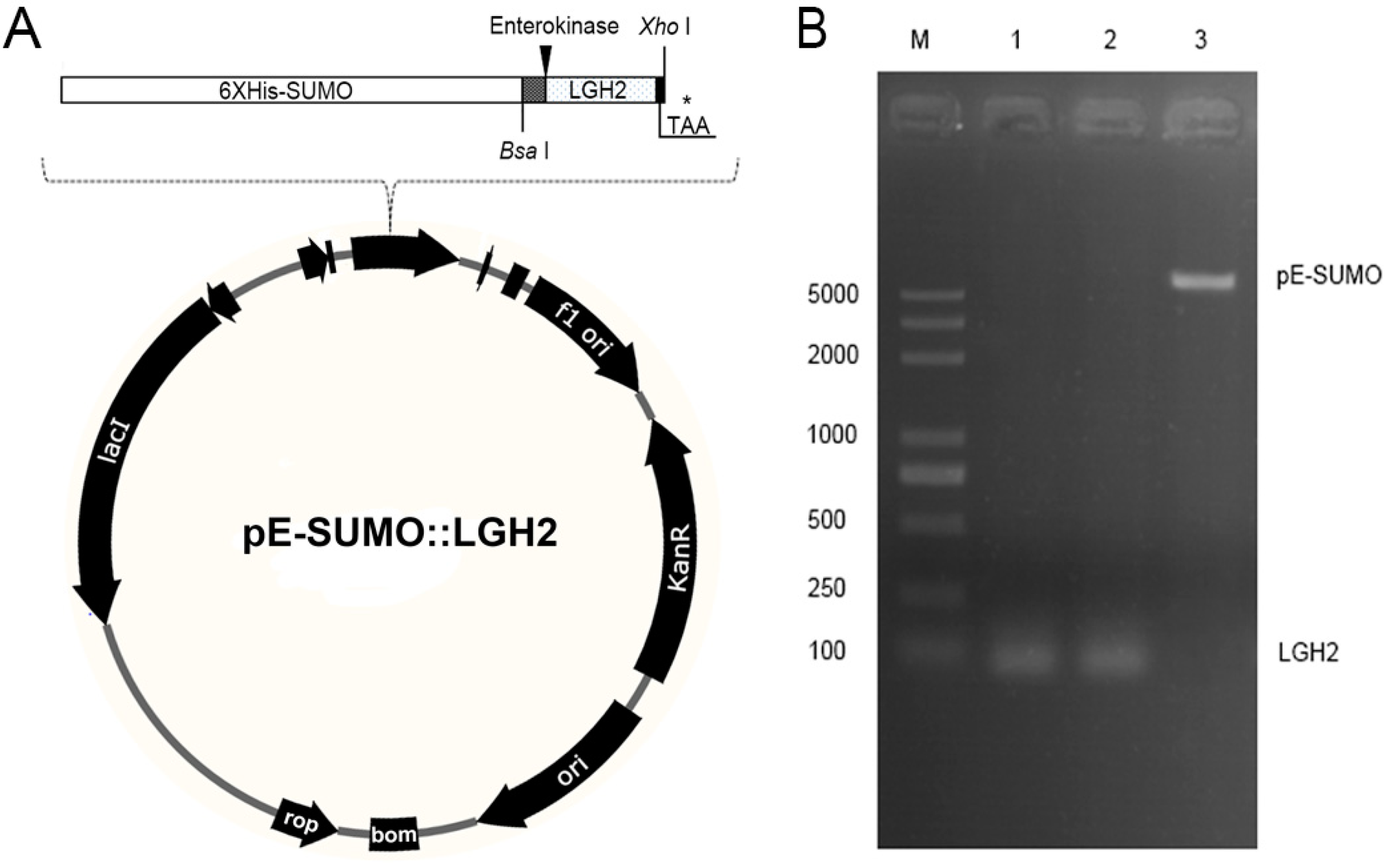
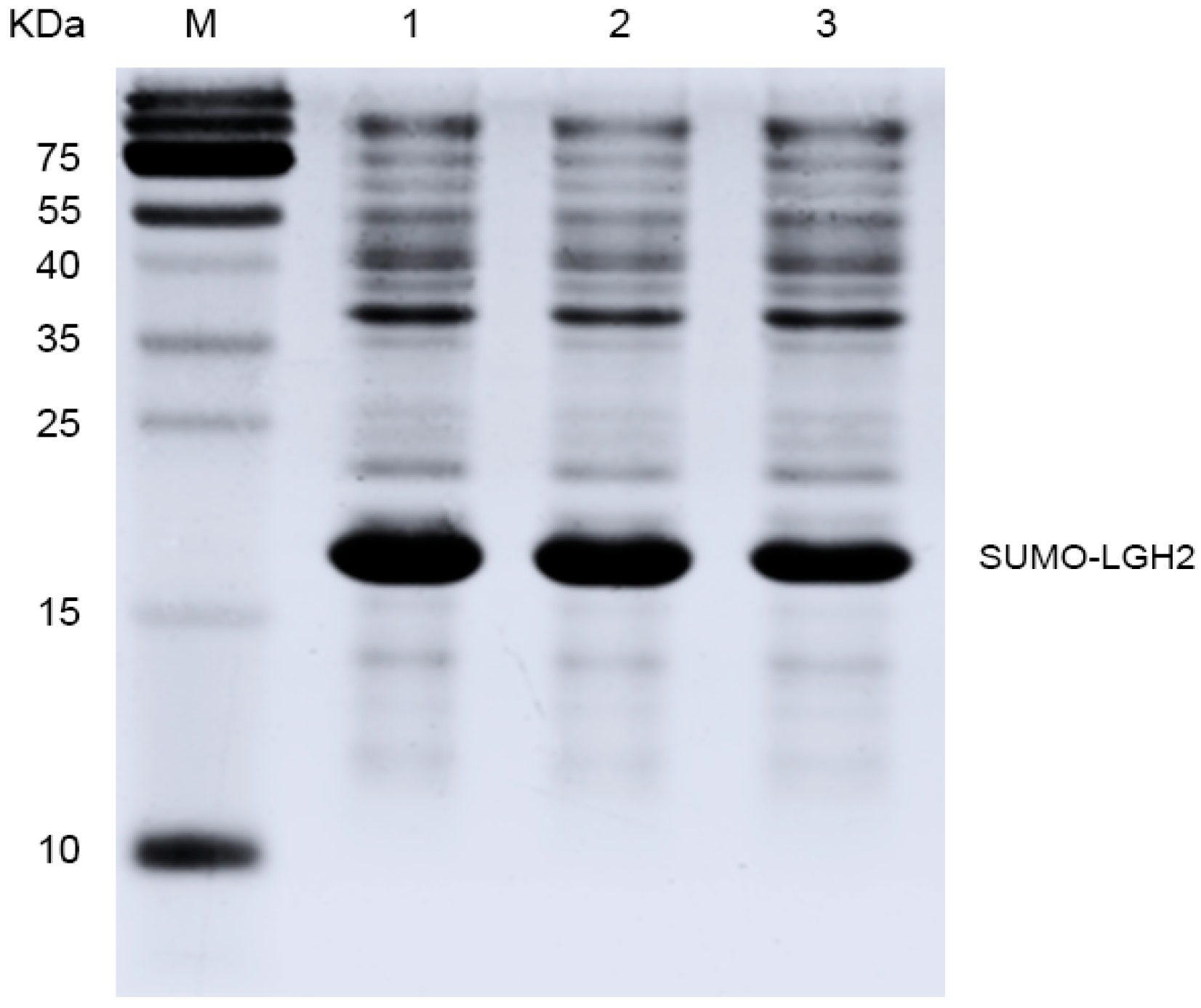
2.3. Expression of the Lgh2 Fusion Protein in E. coli
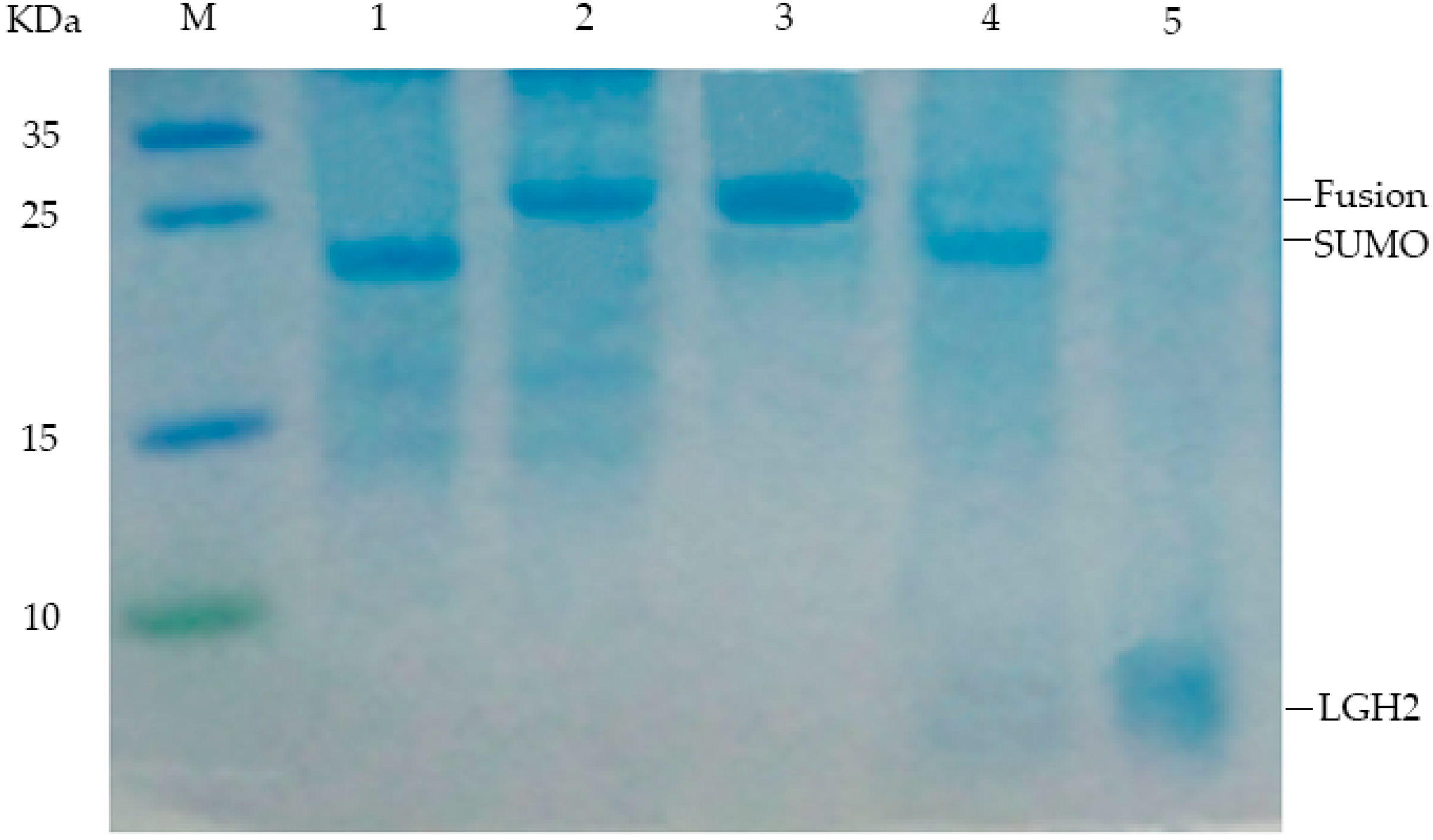
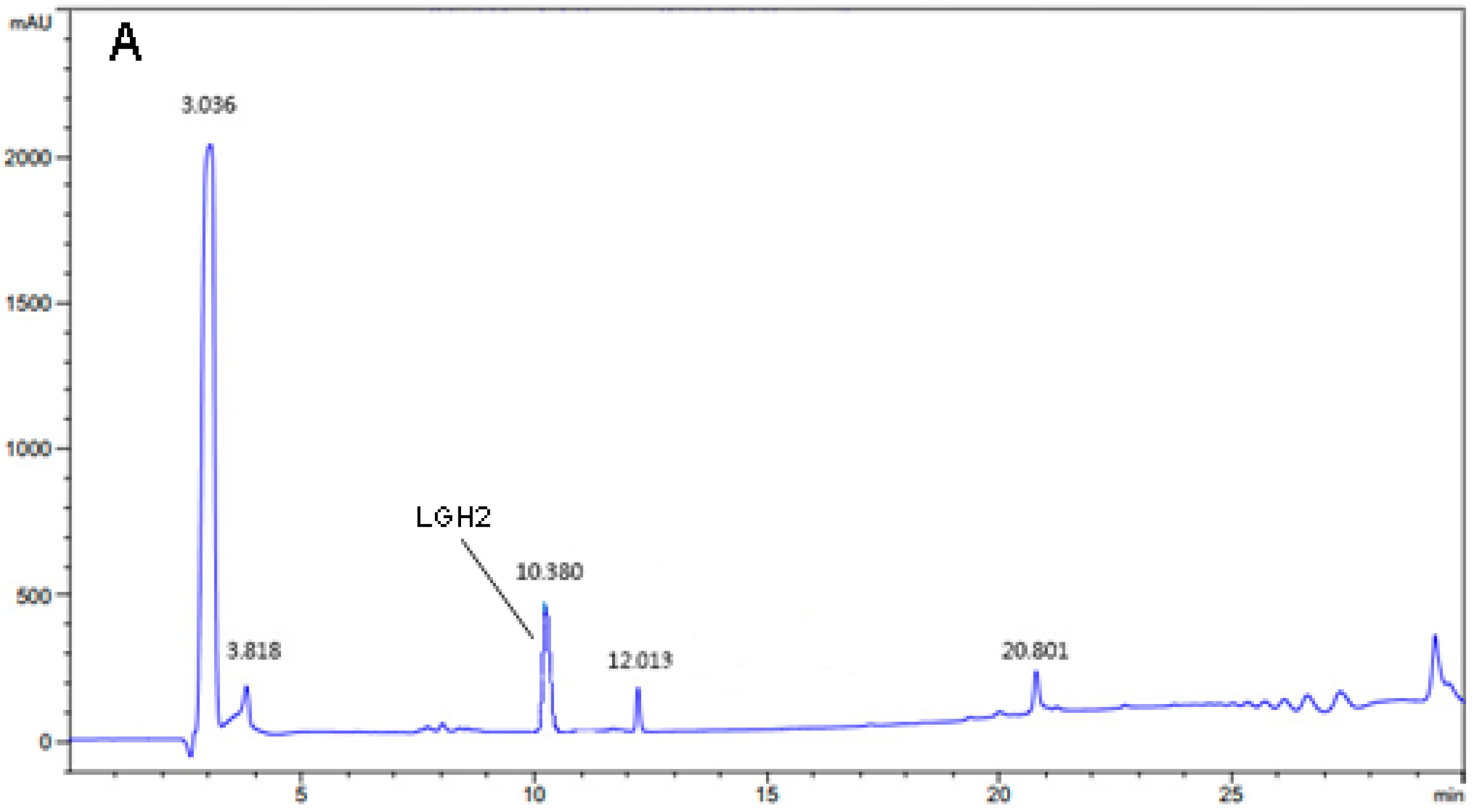
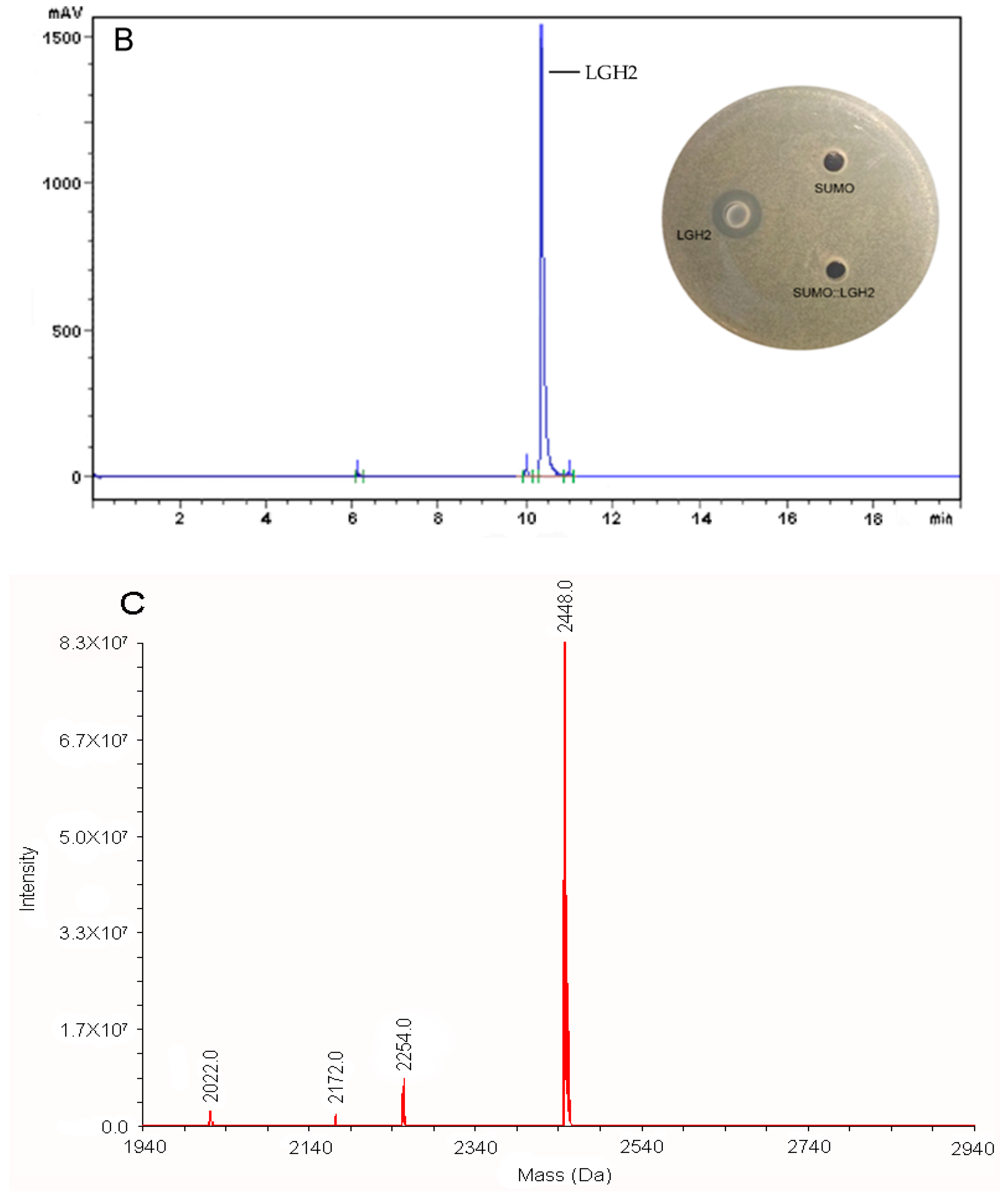
2.4. Purification and Analysis of LGH2
2.5. Antimicrobial Spectrum of the LGH2
2.6. Hemolysis Assays
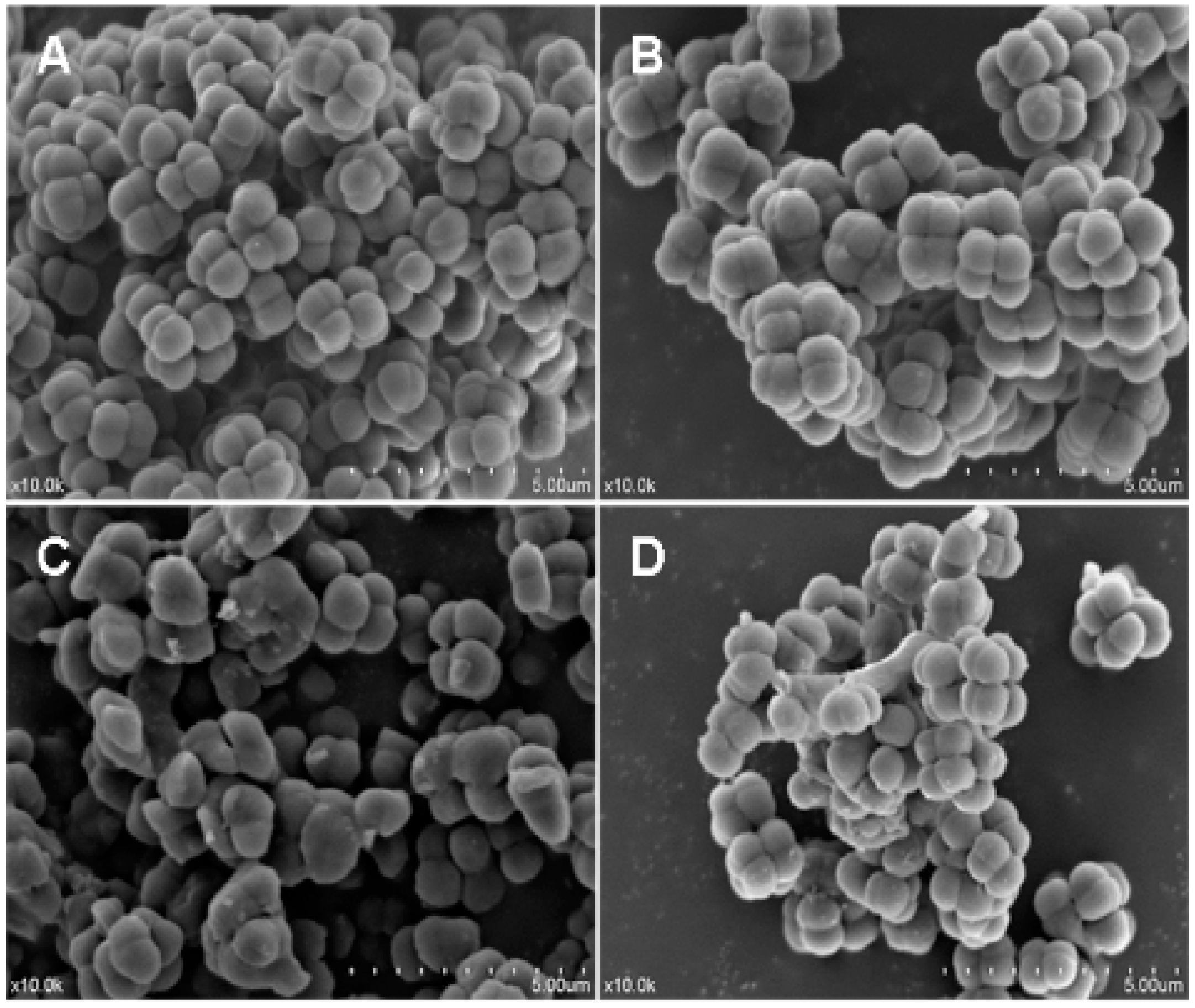
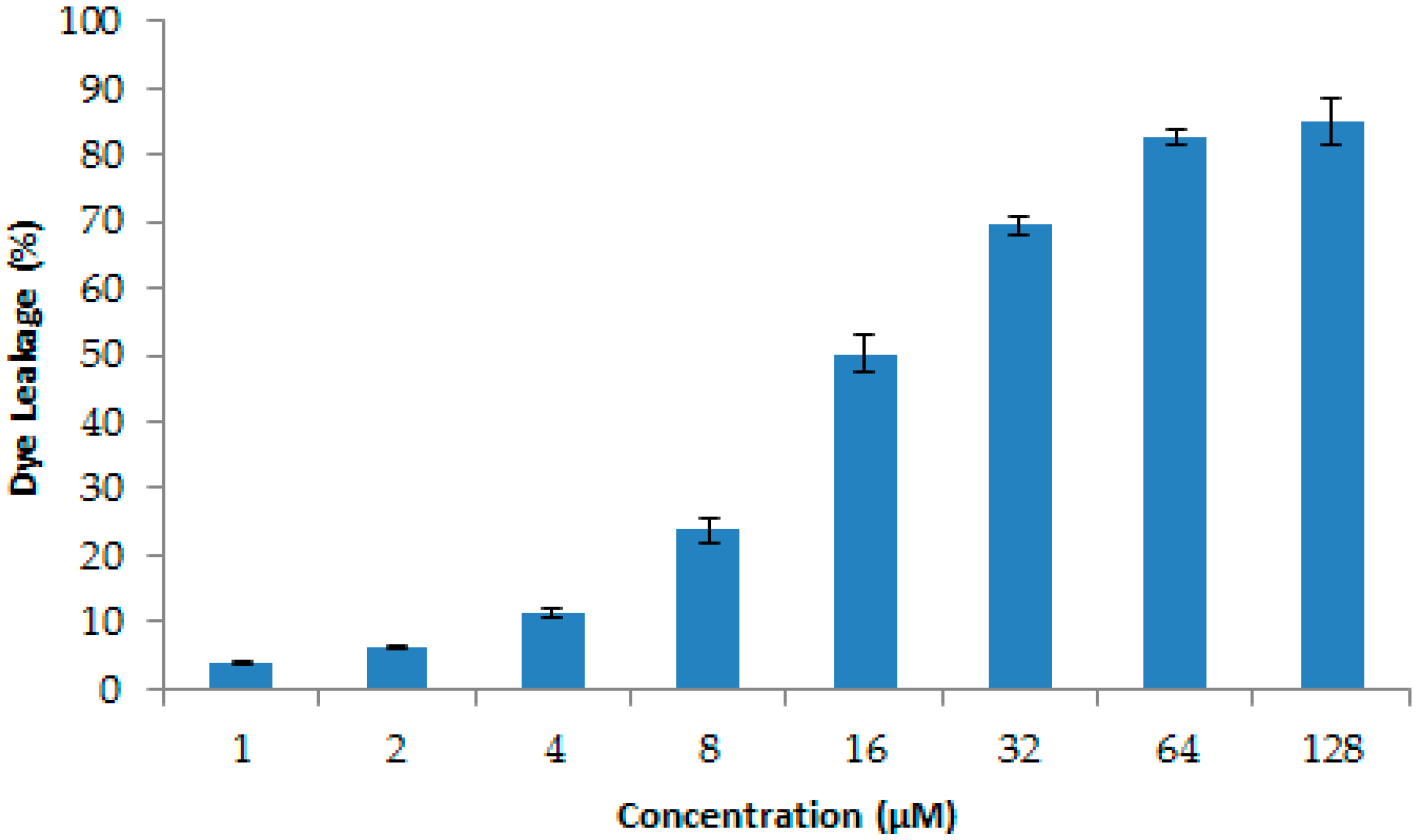
2.7. Effects of Recombinant LGH2 on the Cell Wall Integrity of S. aureus
3. Discussion
4. Materials and Methods
4.1. Bacterial Strains, Plasmid And Reagents
4.2. Identification of Potential AMPs from Genome of L. casei HZ1
4.3. Construction of pE-SUMO-LGH2 Expression Vector
4.4. Expression of the SUMO-LGH2 Fusion Protein
4.5. Purification of LGH2
4.6. Matrix-Assisted Laser Desorption/Ionization-Time-of-Flight Massspectrometry Analysis
4.7. Disk Diffusion Test
4.8. Inhibitory Concentration Determination
4.9. Hemolytic Activity of LGH2
4.10. Binding of LHH1 Fluorescently Labeled with Fluorescein
4.11. Scanning Electron Microscopy Assay
4.12. Flow Cytometry Assay
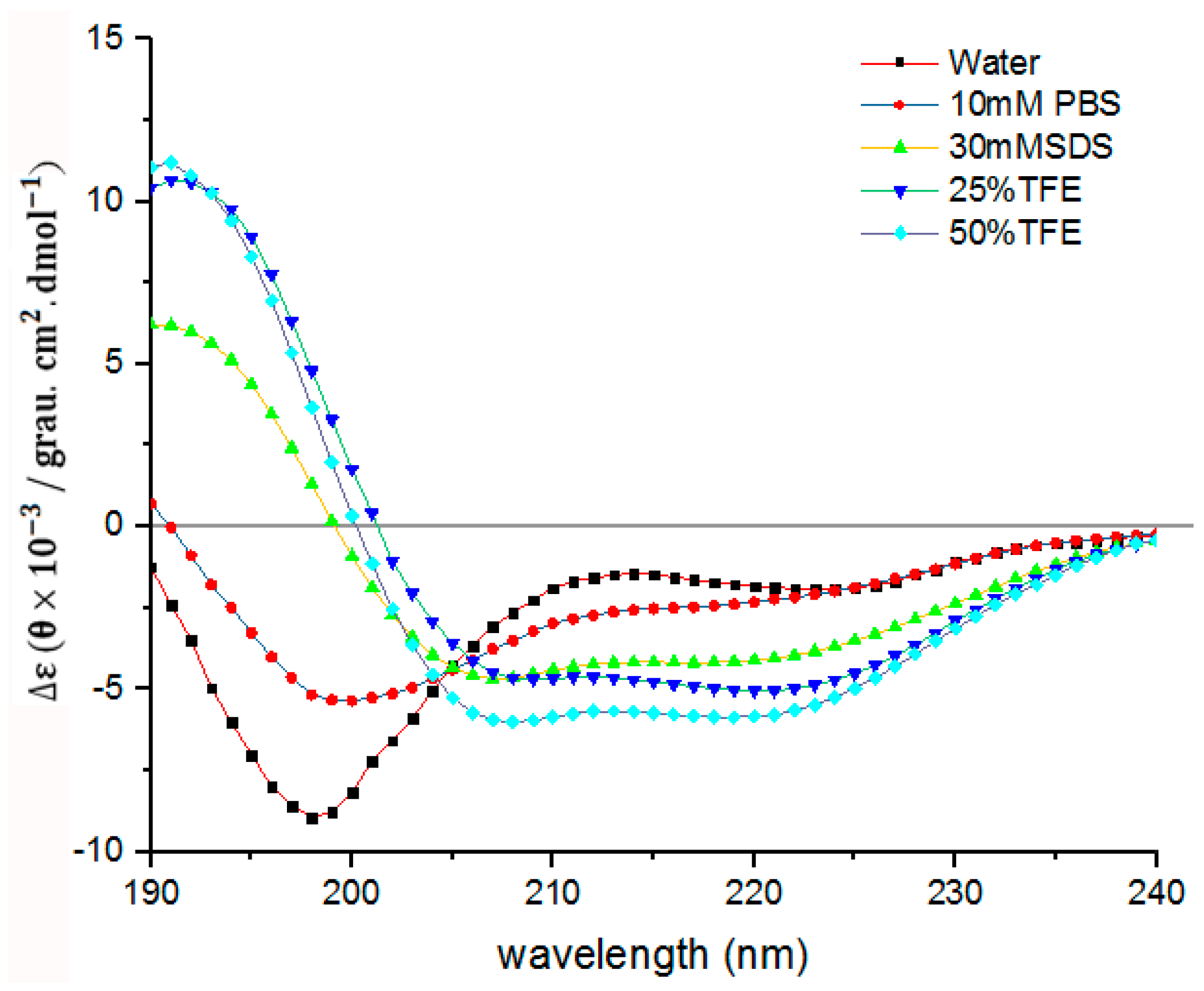
4.13. CD Analysis
4.14. Dye Leakage Assays
4.15. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Galdeano, C.M.; Perdigon, G. The Probiotic Bacterium Lactobacillus casei Induces Activation of the Gut Mucosal Immune System through Innate Immunity. Clin. Vaccine Immunol. 2006, 13, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Hancock, R.E.; Nijnik, A.; Philpott, D.J. Modulating immunity as a therapy for bacterial infections. Nat. Rev. Microbiol. 2012, 10, 243–254. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Tailhades, J.; O’Brien-Simpson, N.M.; Separovic, F.; Otvos, L., Jr.; Hossain, M.A.; Wade, J.D. Proline-rich antimicrobial peptides: Potential therapeutics against antibiotic-resistant bacteria. Amino Acids 2014, 46, 2287–2294. [Google Scholar] [CrossRef] [PubMed]
- Benkerroum, N. Antimicrobial peptides generated from milk proteins: A survey and prospects for application in the food industry. A review. Int. J. Dairy Technol. 2010, 63, 320–338. [Google Scholar] [CrossRef]
- Meisel, H. Multifunctional peptides encrypted in milk proteins. BioFactors 2004, 21, 55–61. [Google Scholar] [CrossRef] [PubMed]
- LeMessurier, K.S.; Lin, Y.; McCullers, J.A.; Samarasinghe, A.E. Antimicrobial peptides alter early immune response to influenza A virus infection in C57BL/6 mice. Antiviral Res. 2016, 133, 208–217. [Google Scholar] [CrossRef] [PubMed]
- Login, F.H.; Balmand, S.; Vallier, A.; Vincent-Monegat, C.; Vigneron, A.; Weiss-Gayet, M.; Rochat, D.; Heddi, A. Antimicrobial peptides keep insect endosymbionts under control. Science 2011, 334, 362–365. [Google Scholar] [CrossRef] [PubMed]
- Pu, C.; Tang, W. Affinity and selectivity of anchovy antibacterial peptide for Staphylococcus aureus cell membrane lipid and its application in whole milk. Food Control 2017, 72, 153–163. [Google Scholar] [CrossRef]
- Otvos, L., Jr. Antibacterial peptides isolated from insects. J. Pept. Sci. 2000, 6, 497–511. [Google Scholar] [CrossRef]
- Hurdle, J.G.; O’Neill, A.J.; Chopra, I.; Lee, R.E. Targeting bacterial membrane function: An underexploited mechanism for treating persistent infections. Nat. Rev. Microbiol. 2011, 9, 62–75. [Google Scholar] [CrossRef] [PubMed]
- Fjell, C.D.; Hiss, J.A.; Hancock, R.E.W.; Schneider, G. Designing antimicrobial peptides: Form follows function. Nat. Rev. Drug Discov. 2011, 16, 37–51. [Google Scholar] [CrossRef]
- Ageitos, J.M.; Sanchez-Perez, A.; Calo-Mata, P.; Villa, T.G. Antimicrobial peptides (AMPs): Ancient compounds that represent novel weapons in the fight against bacteria. Biochem. Pharmacol. 2016, 133, 117–138. [Google Scholar] [CrossRef] [PubMed]
- You, S.J.; Udenigwe, C.C.; Aluko, R.E.; Wu, J. Multifunctional peptides from egg white lysozyme. Food Res. Int. 2010, 43, 848–855. [Google Scholar] [CrossRef]
- Zhu, X.; Zhao, Y.; Sun, Y.; Gu, Q. Purification and characterisation of plantaricin ZJ008, a novel bacteriocin against Staphylococcus spp. from Lactobacillus plantarum ZJ008. Food Chem. 2014, 165, 216–223. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Feng, Y.; Kuang, Y.; Zeng, W.; Yang, Y.; Li, H.; Jiang, Z.; Li, M. Construction of eukaryotic expression vector with mBD1-mBD3 fusion genes and exploring its activity against influenza A virus. Viruses 2014, 6, 1237–1252. [Google Scholar] [CrossRef] [PubMed]
- Mechkarska, M.; Attoub, S.; Sulaiman, S.; Pantic, J.; Lukic, M.L.; Conlon, J.M. Anticancer, immunoregulatory, and antimicrobial activities of the frog skin host-defense peptides pseudhymenochirin-1Pb and pseudhymenochirin-2Pa. Regul. Pept. 2014, 194–195, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Shagaghi, N.; Palombo, E.A.; Clayton, A.H.; Bhave, M. Archetypal tryptophan-rich antimicrobial peptides: Properties and applications. World J. Microbiol. Biotechnol. 2016, 32, 31. [Google Scholar] [CrossRef] [PubMed]
- Han, Z.; Luo, X.G.; Tian, H.; Men, Y.; Wang, N.; Jiang, Y.; Zhang, T.C. Separation of Angiotensin-Converting Enzyme Inhibitors from Fermented Milk Prepared with Lactobacillus casei HZ1. Adv. Mater. Res. 2011, 345, 161–167. [Google Scholar] [CrossRef]
- O’Brien-Simpson, N.M.; Pantarat, N.; Attard, T.J.; Walsh, K.A.; Reynolds, E.C. A Rapid and Quantitative Flow Cytometry Method for the Analysis of Membrane Disruptive Antimicrobial Activity. PLoS ONE 2016, 11, e0151694. [Google Scholar] [CrossRef]
- Li, W.; O’Brien-Simpson, N.M.; Tailhades, J.; Pantarat, N.; Dawson Raymond, M.; Otvos, L.; Reynolds Eric, C.; Separovic, F.; Hossain Mohammed, A.; Wade John, D. Multimerization of a Proline-Rich Antimicrobial Peptide, Chex-Arg20, Alters Its Mechanism of Interaction with the Escherichia coli Membrane. Chem. Biol. 2015, 22, 1250–1258. [Google Scholar] [CrossRef] [PubMed]
- Lam, S.J.; O’Brien-Simpson, N.M.; Pantarat, N.; Sulistio, A.; Wong, E.H.; Chen, Y.Y.; Lenzo, J.C.; Holden, J.A.; Blencowe, A.; Reynolds, E.C.; et al. Combating multidrug-resistant Gram-negative bacteria with structurally nanoengineered antimicrobial peptide polymers. Nat. Microbiol. 2016, 1, 16162. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, D.I.; Lee, T.H.; Sani, M.A.; Aguilar, M.I.; Separovic, F. Proline facilitates membrane insertion of the antimicrobial peptide maculatin 1.1 via surface indentation and subsequent lipid disordering. Biophys. J. 2013, 104, 1495–1507. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Huang, H.; Yang, Z.; Liu, B.; Gou, S.; Zhong, C.; Han, X.; Zhang, Y.; Ni, J.; Wang, R. Design of novel antimicrobial peptide dimer analogues with enhanced antimicrobial activity in vitro and in vivo by intermolecular triazole bridge strategy. Peptides 2017, 88, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Park, I.Y.; Cho, J.H.; Kim, K.S.; Kim, Y.B.; Kim, M.S.; Kim, S.C. Helix stability confers salt resistance upon helical antimicrobial peptides. J. Biol. Chem. 2004, 279, 13896–13901. [Google Scholar] [CrossRef] [PubMed]
- Parachin, N.S.; Mulder, K.C.; Viana, A.A.; Dias, S.C.; Franco, O.L. Expression systems for heterologous production of antimicrobial peptides. Peptides 2012, 38, 446–456. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, J.; Borrero, J.; Gomez-Sala, B.; Basanta, A.; Herranz, C.; Cintas, L.M.; Hernandez, P.E. Cloning and heterologous production of Hiracin JM79, a Sec-dependent bacteriocin produced by Enterococcus hirae DCH5, in lactic acid bacteria and Pichia pastoris. Appl. Environ. Microbiol. 2008, 74, 2471–2479. [Google Scholar] [CrossRef] [PubMed]
- Ashcheulova, D.O.; Efimova, L.V.; Lushchyk, A.Y.; Yantsevich, A.V.; Baikov, A.N.; Pershina, A.G. Production of the recombinant antimicrobial peptide UBI18-35 in Escherichia coli. Protein Expr. Purif. 2018, 143, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Eppelmann, K.; Doekel, S.; Marahiel, M.A. Engineered biosynthesis of the peptide antibiotic bacitracin in the surrogate host Bacillus subtilis. J. Biol. Chem. 2001, 276, 34824–34831. [Google Scholar] [CrossRef] [PubMed]
- Kuthning, A.; Mosker, E.; Sussmuth, R.D. Engineering the heterologous expression of lanthipeptides in Escherichia coli by multigene assembly. Appl. Microbiol. Biotechnol. 2015, 99, 6351–6361. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.H.; Lee, S.Y. Secretory and extracellular production of recombinant proteins using Escherichia coli. Appl. Microbiol. Biotechnol. 2004, 64, 625–635. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Li, H.; Gao, D.; Gao, C.; Qi, Q. Secretory production of antimicrobial peptides in Escherichia coli using the catalytic domain of a cellulase as fusion partner. J. Biotechnol. 2015, 214, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Malakhov, M.P.; Mattern, M.R.; Malakhova, O.A.; Drinker, M.; Weeks, S.D.; Butt, T.R. SUMO fusions and SUMO-specific protease for efficient expression and purification of proteins. J. Struct. Funct. Genomics 2004, 5, 75–86. [Google Scholar] [CrossRef] [PubMed]
- Naimi, T.S.; LeDell, K.H.; Como-Sabetti, K.; Borchardt, S.M.; Boxrud, D.J.; Etienne, J.; Johnson, S.K.; Vandenesch, F.; Fridkin, S.; O’Boyle, C.; et al. Comparison of community- and health care-associated methicillin-resistant Staphylococcus aureus infection. JAMA 2003, 290, 2976–2984. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Dong, W.; Sun, L.; Ma, L.; Shang, D. Insights into the membrane interaction mechanism and antibacterial properties of chensinin-1b. Biomaterials 2015, 37, 299–311. [Google Scholar] [CrossRef] [PubMed]
- Sharma, G.; Dang, S.; Gupta, S.; Gabrani, R. Antibacterial Activity, Cytotoxicity and Mechanism of Action of Bacteriocin from Bacillus subtilis GAS101. Med. Princ. Pract. 2018, 27, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Epand, R.M.; Rotem, S.; Mor, A.; Berno, B.; Epand, R.F. Bacterial membranes as predictors of antimicrobial potency. J. Am. Chem. Soc. 2008, 130, 14346–14352. [Google Scholar] [CrossRef] [PubMed]
- Sablon, E.; Vandamme, E. Antimicrobial peptides of lactic acid bacteria: mode of action, genetics and biosynthesis. Adv. Biochem. Eng. Biotechnol. 2000, 68, 21–60. [Google Scholar] [CrossRef] [PubMed]
- van Belkum, M.J.; Worobo, R.W.; Stiles, M.E. Double-glycine-type leader peptides direct secretion of bacteriocins by ABC transporters: Colicin V secretion in Lactococcus lactis. Mol. Microbiol. 1997, 23, 1293–1301. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Li, X.; Wang, Z. APD3: The antimicrobial peptide database as a tool for research and education. Nucleic Acids Res. 2016, 44, 1087–1093. [Google Scholar] [CrossRef] [PubMed]
- Gasteiger, E. ExPASy: The proteomics server for in-depth protein knowledge and analysis. Nucleic Acids Res. 2003, 31, 3784–3788. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y. I-TASSER server for protein 3D structure prediction. BMC Bioinformatics 2008, 9, 40. [Google Scholar] [CrossRef] [PubMed]
- Gautam, A.; Chaudhary, K.; Kumar, R.; Raghava, G.P. Computer-Aided Virtual Screening and Designing of Cell-Penetrating Peptides. Methods Mol. Biol. 2015, 1324, 59–69. [Google Scholar] [CrossRef] [PubMed]
- Chaudhary, K.; Kumar, R.; Singh, S.; Tuknait, A.; Gautam, A.; Mathur, D.; Anand, P.; Varshney, G.C.; Raghava, G.P. A Web Server and Mobile App for Computing Hemolytic Potency of Peptides. Sci. Rep. 2016, 6, 22843. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Zhou, Y.; Liu, X.; Zhang, F.; Yan, L.; Chen, L.; Wang, X.; Ruan, H.; Ji, C.; Cui, X.; et al. Antimicrobial activity and mechanism of PDC213, an endogenous peptide from human milk. Biochem. Biophys. Res. Commun. 2017, 484, 132–137. [Google Scholar] [CrossRef] [PubMed]
- Yi, L.; Dang, J.; Zhang, L.; Wu, Y.; Liu, B.; Lü, X. Purification, characterization and bactericidal mechanism of a broad spectrum bacteriocin with antimicrobial activity against multidrug-resistant strains produced by Lactobacillus coryniformis XN8. Food Control 2016, 67, 53–62. [Google Scholar] [CrossRef]
- Mendes, M.A.; de Souza, B.M.; Palma, M.S. Structural and biological characterization of three novel mastoparan peptides from the venom of the neotropical social wasp Protopolybia exigua (Saussure). Toxicon 2005, 45, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Chou, S.; Wang, J.; Shao, C.; Li, W.; Zhu, X.; Shan, A. Antimicrobial activity and membrane-active mechanism of tryptophan zipper-like beta-hairpin antimicrobial peptides. Amino Acids 2015, 47, 2385–2397. [Google Scholar] [CrossRef] [PubMed]
- Verly, R.M.; Resende, J.M.; Junior, E.F.; de Magalhaes, M.T.; Guimaraes, C.F.; Munhoz, V.H.; Bemquerer, M.P.; Almeida, F.C.; Santoro, M.M.; Pilo-Veloso, D.; et al. Structure and membrane interactions of the homodimeric antibiotic peptide homotarsinin. Sci. Rep. 2017, 7, 40854. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds not available from the authors. |




| Amino Acid Sequence 1 | Characteristic of AMPs 2 | ||||||
|---|---|---|---|---|---|---|---|
| HR (%) | NC | pI | MW (Da) | HS | BI (kcal/mol) | ||
| LGH1 | MKHKFARGFFVGTLMTLGAIAGSVFAFKKLYIEPVETKVDEINDNRRKANRKRFSAHQG | 34 | +4 | 9.99 | 3989.47 | 0.46 | 4.23 |
| LGH2 | MDSQYPKRIFHIIKIWLMIALIALILGLLIGFALGEGNPLKLFLPSTWVHFFKFLR | 50 | +3 | 11.17 | 2447.95 | 0.55 | 0.21 |
| LGH3 | MQRGMIGAVIGIVLAFAWMRFGFMGMLLVLIFAGWLIERYLVPNWHAFTSWLTAGKDAFSKGK | 45 | +2 | 9.53 | 3551.07 | 0.60 | 0.76 |
| LGH4 | MAAGFIKEVFYSEWFVNSVFVRKKKWRMCVDYTGFNKVCSKVFYFLFRID | 50 | +3 | 9.24 | 3266.89 | 0.48 | 1.33 |
| Microbial Strains | LGH2 | Ampicillin | ||
|---|---|---|---|---|
| MIC () | MBC () | MIC () | MBC () | |
| Gram+ | ||||
| Staphylococcus aureus (ATCC 29213) | 4 | 8 | 0.25 | 0.50 |
| Listeria monocytogenes (ATCC 7611) | 4 | 8 | 0.125 | 0.25 |
| Lactobacillus casei HZ1 (our lab) | 32 | 64 | 0.50 | 1.00 |
| Gram- | ||||
| Klebsiella pneumoniae (ATCC 700603) | >256 | >256 | 4 | 8 |
| Enterobacter aerogenes (ATCC 13048) | 64 | 128 | 8 | 16 |
| Fungus | ||||
| Saccharomyces cerevisiae (our lab) | >256 | >256 | -- | -- |
| Pichia pastoris (our lab) | >256 | >256 | -- | -- |
| Solution | α-Helix (%) | β-Sheet (%) | Random Coil (%) |
|---|---|---|---|
| water | 4.35 | 9.66 | 85.99 |
| 10 mM PBS | 21.15 | 9.74 | 69.11 |
| 30 mM SDS | 44.32 | 5.98 | 49.70 |
| 25% TFE | 44.69 | 12.13 | 43.18 |
| 50% TFE | 52.76 | 6.75 | 40.49 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
He, J.; Luo, X.; Jin, D.; Wang, Y.; Zhang, T. Identification, Recombinant Expression, and Characterization of LGH2, a Novel Antimicrobial Peptide of Lactobacillus casei HZ1. Molecules 2018, 23, 2246. https://doi.org/10.3390/molecules23092246
He J, Luo X, Jin D, Wang Y, Zhang T. Identification, Recombinant Expression, and Characterization of LGH2, a Novel Antimicrobial Peptide of Lactobacillus casei HZ1. Molecules. 2018; 23(9):2246. https://doi.org/10.3390/molecules23092246
Chicago/Turabian StyleHe, Junfang, Xuegang Luo, Duxin Jin, Yunyang Wang, and Tongcun Zhang. 2018. "Identification, Recombinant Expression, and Characterization of LGH2, a Novel Antimicrobial Peptide of Lactobacillus casei HZ1" Molecules 23, no. 9: 2246. https://doi.org/10.3390/molecules23092246
APA StyleHe, J., Luo, X., Jin, D., Wang, Y., & Zhang, T. (2018). Identification, Recombinant Expression, and Characterization of LGH2, a Novel Antimicrobial Peptide of Lactobacillus casei HZ1. Molecules, 23(9), 2246. https://doi.org/10.3390/molecules23092246




