Daily Consumption of Chocolate Rich in Flavonoids Decreases Cellular Genotoxicity and Improves Biochemical Parameters of Lipid and Glucose Metabolism
Abstract
1. Introduction
2. Results
2.1. Study Population
2.2. Determination of Total Phenolic, Flavonoids Content and Antioxiant Capacity
2.3. Changes Caused by Dailyconsumption of Chocolate
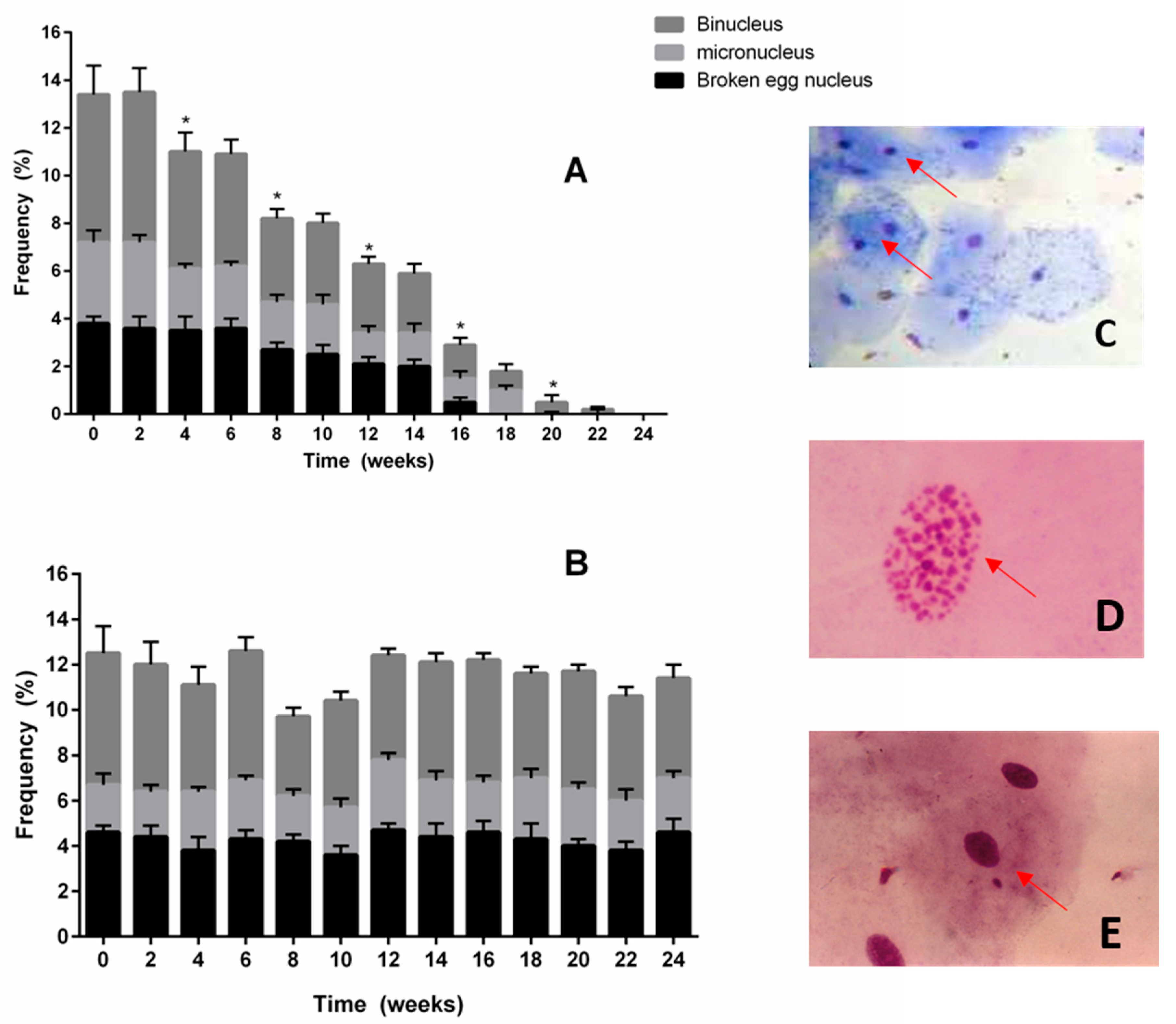
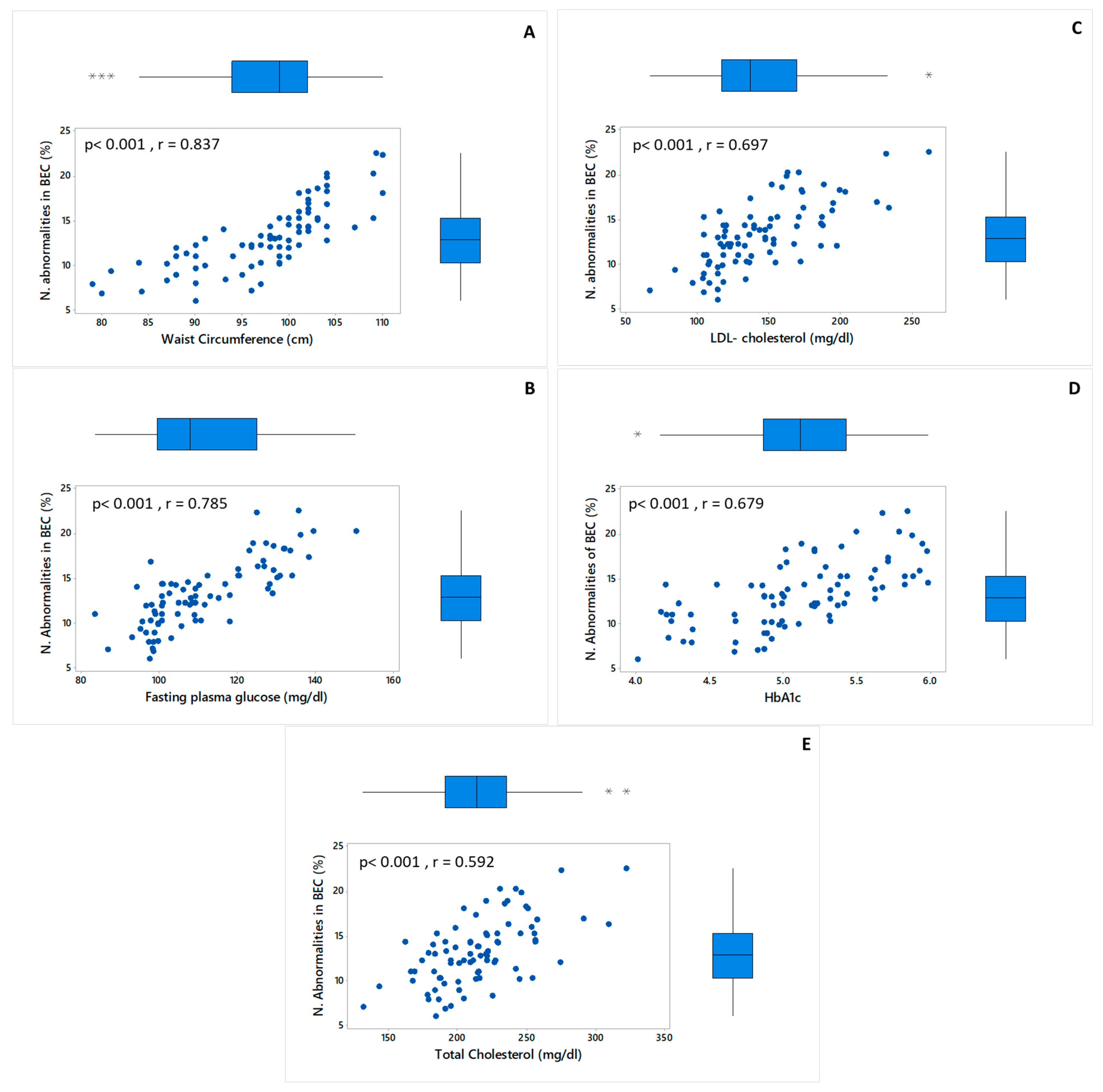
2.4. Frequency of Nuclear Abnormalities in Buccal Epithelial Cells
3. Discussion
3.1. Determination of Total Phenolic and Flavonoids Content
3.2. Characteristics of Study Population
3.3. Determination of Total Phenolic and Flavonoids Content
3.4. Changes in Measured Parameters by Daily Dark Chocolate Consumption
3.5. Frequency of Nuclear Abnormalities in Buccal Epithelial Cells
4. Materials and Methods
4.1. Study Design and Participants
4.2. Chocolate Consumption (Independent Variable)
4.3. Determination of Total Phenolic Content
4.4. Determination of Flavonoids Content
4.5. Antioxidant Capacity
4.6. Epithelial Genotoxicity, Biochemical and Anthropometric Parameters Measure (Dependent Variables)
4.7. Statistical Analyses
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Prevention of Cardiovascular Disease. 2011. Available online: http://www.who.int/cardiovascular_diseases/en/ (accessed on 28 August 2011).
- Esser, N.; Legrand-Poels, S.; Piette, J.; Scheen, A.J.; Paquot, N. Inflammation as a link between obesity, metabolic syndrome and type 2 diabetes. Diabetes Res. Clin. Pract. 2014, 105, 141–150. [Google Scholar] [CrossRef] [PubMed]
- Torres, N.; Torre-Villalvazo, I.; Tovar, A.R. Regulation of lipid metabolism by soy protein and its implication in diseases mediated by lipid disorders. J. Nutr. Biochem. 2006, 17, 365–373. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, C.J.; Elosua, R. Cardiovascular Risk Factors. Insights from Framingham Heart Study. Rev. Española Cardiol. 2008, 61, 299–310. [Google Scholar] [CrossRef]
- McGillicuddy, F.C.; Roche, H.M. Nutritional status, genetic susceptibility, and insulin resistance–important precedents to atherosclerosis. Mol. Nutr. Food Res. 2018, 56, 1173–1184. [Google Scholar] [CrossRef] [PubMed]
- Torres, N.T.; Palacios-Gonzalez, B.; Noriega-Lopez, L.; Tovar-Palacio, A.R. Glycaemic index, insulinemic index and glycaemic load of soya drinks with low and high levels of carbohydrates. Rev. Investig. Clin. 2006, 58, 487–497. [Google Scholar]
- Ginsberg, H.N. Insulin resistance and cardiovascular disease. J. Clin. Investig. 2000, 106, 453–458. [Google Scholar] [CrossRef] [PubMed]
- Surujdeo-Maharaj, S.; Sreenivasan, T.N.; Motilal, L.A.; Umaharan, P. Black pod and other phytophthora induced diseases of cacao: History, biology, and control. In Cacao Diseases; Bailey, B., Meinhardt, L., Eds.; Springer: Cham, Switzerland, 2016; pp. 213–266. ISBN 978-3-319-24789-2. [Google Scholar]
- Grassi, D.; Socci, V.; Tempesta, D.; Ferri, C.; De Gennaro, L.; Desideri, G.; Ferrara, M. Flavanol-rich chocolate acutely improves arterial function and working memory performance counteracting the effects of sleep deprivation in healthy individuals. J. Hypertens. 2016, 34, 1298–1308. [Google Scholar] [CrossRef]
- Flammer, A.J.; Sudano, I.; Wolfrum, M.; Thomas, R.; Enseleit, F.; Périat, D.; Kaiser, P.; Hirt, A.; Hermann, M.; Serafini, M.; et al. Cardiovascular effects of flavanol-rich chocolate in patients with heart failure. Eur. Heart J. 2012, 33, 2172–2180. [Google Scholar] [CrossRef] [PubMed]
- Stahl, L.; Miller, K.B.; Apgar, J.; Sweigart, D.S.; Stuart, D.A.; McHale, N.; Ou, B.; Kondo, M.; Hurst, W.J. Preservation of cocoa antioxidant activity, total polyphenols, flavan-3-ols, and procyanidin content in foods prepared with cocoa powder. J. Food Sci. 2009, 74, C456–C461. [Google Scholar] [CrossRef] [PubMed]
- Langer, S.; Marshall, L.J.; Day, A.J.; Morgan, M.R.A. Flavanols and methylxanthines in commercially available dark chocolate: A study of the correlation with nonfat cocoa solids. J. Agric. Food Chem. 2011, 59, 8435–8441. [Google Scholar] [CrossRef] [PubMed]
- Coussens, L.M.; Werb, Z. Inflammation and cancer. Nature 2002, 420, 860–867. [Google Scholar] [CrossRef] [PubMed]
- Gupta, P.K. Toxicological testing: Genesis. In Fundamentals of Toxicology: Essential Concepts and Applications; Erin Hill-Parks, Ed.; Elsevier Inc.: Bareilly, India, 2016; pp. 111–122. ISBN 978-0-12-805426-0. [Google Scholar]
- Arif, M.; Islam, M.R.; Waise, T.M.Z.; Hassan, F.; Mondal, S.I.; Kabir, Y. DNA damage and plasma antioxidant indices in Bangladeshi type 2 diabetic patients. Diabetes Metab. 2010, 36, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Erol, A. Systemic DNA damage response and metabolic syndrome as a premalignant state. Curr. Mol. Med. 2010, 10, 321–334. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Global Physical Activity Questionnaire (GPAQ) Analysis Guide. 2018. Available online: https://epi.grants.cancer.gov/paq/q078.html (accessed on 24 April 2018).
- National Center for Chronic Disease Prevention and Health Promotion. Body mass index—BMI. 2018. Available online: https://www.cdc.gov/healthyweight/assessing/bmi/adult_bmi/index.html (accessed on 24 April 2018).
- Lee, K.W.; Kim, Y.J.; Lee, H.J.; Lee, C.Y. Cocoa has more phenolic phytochemicals and a higher antioxidant capacity than teas and red wine. J. Agric. Food Chem. 2003, 51, 7292–7295. [Google Scholar] [CrossRef] [PubMed]
- Ioannone, F.; Sacchetti, G.; Serafini, M. Effect of dark chocolate extracts on phorbol 12-myristate 13-acetate-induced oxidative burst in leukocytes isolated by normo-weight and overweight/obese subjects. Front. Nutr. 2017, 4, 23. [Google Scholar] [CrossRef] [PubMed]
- Palacios-González, B.; Zarain-Herzberg, A.; Flores-Galicia, I.; Noriega, L.G.; Alemán-Escondrillas, G.; Zariñan, T.; Ulloa-Aguirre, A.; Torres, N.; Tovar, A.R. Genistein stimulates fatty acid oxidation in a leptin receptor-independent manner through the JAK2-mediated phosphorylation and activation of AMPK in skeletal muscle. Biochim. Biophys. Acta 2014, 1841, 132–140. [Google Scholar] [CrossRef] [PubMed]
- Chavez-Santoscoy, R.C.; Gutierrez-Uribe, J.A.; Granados, O.; Torre-Villalvazo, I.; Serna-Saldivar, S.O.; Torres, N.; Palacios-González, B.; Tovar, A.R. Flavonoids and saponins extracted from black bean (Phaseolus vulgaris L.) seed coats modulate lipid metabolism and biliary cholesterol secretion in C57BL/6 mice. Br. J. Nutr. 2014, 112, 886–899. [Google Scholar] [CrossRef] [PubMed]
- Function, H.D.L.; Millar, C.L.; Duclos, Q.; Blesso, C.N. Effects of dietary flavonoids on reverse cholesterol transport, HDL metabolism, and HDL function. Adv. Nutr. 2017, 8, 226–239. [Google Scholar]
- Alkerwi, A.; Sauvageot, N.; Crichton, G.E.; Elias, M.F.; Stranges, S. Daily chocolate consumption is inversely associated with insulin resistance and liver enzymes in the observation of cardiovascular risk factors in luxembourg study. Br. J. Nutr. 2016, 115, 1661–1668. [Google Scholar] [CrossRef] [PubMed]
- Ramos, S.; Rodríguez-Ramiro, I.; Martín, M.A.; Goya, L.; Bravo, L. Dietary flavanols exert different effects on antioxidant defenses and apoptosis/proliferation in Caco-2 and SW480 colon cancer cells. Toxicol. In Vitro 2011, 25, 1771–1781. [Google Scholar] [CrossRef] [PubMed]
- Rull, G.; Mohd-Zain, Z.N.; Shiel, J.; Lundberg, M.H.; Collier, D.J.; Johnston, A.; Warner, T.D.; Corder, R. Effects of high flavanol dark chocolate on cardiovascular function and platelet aggregation. Vascul. Pharmacol. 2015, 71, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Müllner, E.; Brath, H.; Nersesyan, A.; Nitz, M.; Petschnig, A.; Wallner, M.; Knasmüller, S.; Wagner, K.H. Nuclear anomalies in exfoliated buccal cells in healthy and diabetic individuals and the impact of a dietary intervention. Mutagenesis 2014, 29, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Demirbag, R.; Yilmaz, R.; Gur, M.; Celik, H.; Guzel, S.; Selek, S.; Kocyigit, A. DNA damage in metabolic syndrome and its association with antioxidative and oxidative measurements. Int. J. Clin. Pract. 2006, 60, 1187–1193. [Google Scholar] [CrossRef] [PubMed]
- Jain, N.; Naseem, I.; Ahmad, J. Evaluation of DNA damage and metabolic syndrome parameters in diabetic rabbits supplemented with antioxidants. Fundam. Clin. Pharmacol. 2009, 23, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Damasceno, D.C.; Sinzato, Y.K.; Bueno, A.; Dallaqua, B.; Lima, P.H.; Calderon, I.M.P.; Rudge, M.V.C.; Campos, K.E. Metabolic profile and genotoxicity in obese rats exposed to cigarette smoke. Obesity 2013, 21, 1596–1601. [Google Scholar] [CrossRef] [PubMed]
- Luca, V.S.; Miron, A.; Aprotosoaie, A.C. The antigenotoxic potential of dietary flavonoids. Phytochem. Rev. 2016, 15, 591–625. [Google Scholar] [CrossRef]
- Chakrabarti, M.; Ghosh, I.; Jana, A.; Ghosh, M.; Mukherjee, A. Genotoxicity of antiobesity drug orlistat and effect of caffeine intervention: An in vitro study. Drug Chem. Toxicol. 2017, 40, 339–343. [Google Scholar] [CrossRef] [PubMed]
- Serafini, M.; Peluso, I.; Raguzzini, A. Flavonoids as anti-inflammatory agents. Proc. Nutr. Soc. 2010, 69, 273–278. [Google Scholar] [CrossRef] [PubMed]
- Pimentel, F.A.; Nitzke, J.A.; Klipel, C.B.; De Jong, E.V. Chocolate and red wine–A comparison between flavonoids content. Food Chem. 2010, 120, 109–112. [Google Scholar] [CrossRef]
Sample Availability: not available. |
| Characteristics | n | (%) | FRC (n) | MC (n) | Observations | |
|---|---|---|---|---|---|---|
| Participants | 84 | (100) | 42 | 42 | ||
| Sex | Men | 47 | (55.90) | 23 | 24 | |
| Women | 37 | (44.10) | 19 | 18 | ||
| Education level (completed or in progress) | According to the information provided in the surveys conducted. | |||||
| Elementary school | 2 | (2.38) | 1 | 1 | ||
| High school | 3 | (3.57) | 2 | 1 | ||
| University | 79 | (94.05) | 39 | 40 | ||
| Current smokers | 32 | (38.09) | 16 | 16 | ||
| Physical activity | According to the information provided in the surveys conducted. | |||||
| Inactive | 17 | (20.24) | 8 | 9 | ||
| Moderate active | 55 | (65.48) | 28 | 27 | ||
| Active | 12 | (14.28) | 6 | 6 | ||
| Obesity (BMI range) | The classification was made according to of the world health organization (WHO) | |||||
| Underweight (Below 18.5) | 0 | (0.00) | 0 | 0 | ||
| Normal weight (18.5–24.9) | 9 | (10.71) | 4 | 5 | ||
| Pre-obesity (25.0–29.9) | 10 | (11.90) | 5 | 5 | ||
| Obesity class I (30.0–34.9) | 57 | (67.85) | 29 | 28 | ||
| Obesity class II (35.0–39.9) | 8 | (9.52) | 4 | 4 | ||
| Obesity class III (Above 40) | 0 | (0.00) | ||||
| Risk Factors | Hypertension | 18 | (21.41) | 9 | 9 | Defined as blood pressure ≥140 mm Hg and/or ≥90 mm Hg for systolic and diastolic pressures respectively. |
| Diabetes | 12 | (14.28) | 6 | 6 | Defined as taking antidiabetic medications and/or having fasting plasma glucose ≥120 mg/dL (≥7 mmol/L) | |
| Dyslipidemia | 52 | (61.90) | 26 | 26 | Defined as having at least one of the following anomalies: total cholesterol ≥190 mg/dL (≥4.9 mmol/L), TAG ≥150 mg/dL (≥1.7 mmol/L), LDL-cholesterol ≥115 mg/dL (≥3.0 mmol/L), HDL-cholesterol <40 mg/dL for men and <46 mg/dL for women. | |
| Treatment | Phenolic Content (µmol Gallic Aid Equivalents/g) | Flavonoid Content (µmol Catechin Equivalents/g) | Antioxidant Capacity (µmol Trolox Equivalents/g) | Flavonoids/Phenolics (%) |
|---|---|---|---|---|
| FRC | 63.70 ± 0.20 | 34.80 ± 0.50 | 3.52 ± 0.60 | 54.63 |
| MC | 56.30 ± 1.50 | 10.40 ± 0.80 | 0.98 ± 0.02 | 18.47 |
| Variables | Intervention Group | Beginning of the Study Mean ± SD | End of the Study Mean ± SD | |
|---|---|---|---|---|
| Age | FRC MC | 23.8 ± 3.4 23.6 ± 3.5 | 24.6 ± 3.1 23.8 ± 2.6 | |
| Number of Participants | FRC MC | 42 42 | 42 42 | |
| Dietary variables | Fruit and vegetable intake (g/day) | FRC MC | 523.7 ± 371.4 529.1 ± 329.6 | 548.2 ± 387.9 531.2 ± 356.21 |
| Total energy intake (kJ/day) | FRC MC | 2214 ± 323 2208 ± 350 | 2298 ± 236 2310 ± 120 | |
| Total carbohydrate (%E) | FRC MC | 45.3 ± 8.2 46.7 ± 6.3 | 43.8 ± 8.9 45.8 ± 7.2 | |
| Added sugar | FRC MC | 5.3 ± 3.2 5.8 ± 4.1 | 6.1 ± 2.7 5.8 ± 3.8 | |
| Total Fat (%E) | FRC MC | 36.9 ± 8.1 34.8 ± 6.2 | 37.1 ± 7.6 36.5 ± 5.1 | |
| Saturated Fat (%E) | FRC MC | 20.8 ± 6.1 21.2 ± 3.1 | 16.9 ± 6.9 22.3 ± 5.9 | |
| Unsaturated Fat (%E) | FRC MC | 14.6 ± 4.1 14.5 ± 3.9 | 14.8 ± 4.6 15.9 ± 3.1 | |
| Anthropometric variables | BMI (Kg/m2) | FRC MC | 32.1 ± 3.8 31.4 ± 3.2 | 30.1 ± 2.2 32.4 ± 2.5 |
| Waist Circumference (cm) | FRC MC | 98.7 ± 3.5 96.9 ± 4.1 | 90.4 ± 4.5 * 94.9 ± 3.9 | |
| Biochemical variables | Total Cholesterol (mg/dL) | FRC MC | 221.3 ± 16.7 224.3 ± 18.9 | 201.2 ± 19.5 * 227.4 ± 12.4 |
| LDL-Cholesterol (mg/dL) | FRC MC | 149.82 ± 18.4 147.23 ± 21.1 | 116.2 ± 21.1 * 138.9 ± 19.1 | |
| HDL-Cholesterol (mg/dL) | FRC MC | 46.3 ± 12.5 45.4 ± 12.1 | 43.2 ± 10.9 44.2 ± 13.5 | |
| Triglycerides (mg/dL) | FRC MC | 228.25 ± 17.9 223.5 ± 21.1 | 153.26 ± 18.95 * 224.1 ± 23.1 | |
| HOMA-IR | FRC MC | 2.3 ± 1.8 2.5 ± 1.6 | 1.93 ± 1.1 * 2.4 ± 1.5 | |
| Fasting plasma glucose (mg/dL) | FRC MC | 114.23 ± 13.56 112.31 ± 16.71 | 91.23 ± 9.25 * 111.67 ± 10.9 | |
| HbA1c (%) | FRC MC | 5.8 ± 1.0 4.7 ± 1.0 | 4.6 ± 1.1 4.5 ± 0.9 | |
| Systolic blood pressure (mmHg) | FRC MC | 139.2 ± 10.5 136.3 ± 21.5 | 127.8 ± 11.2 * 133.9 ± 12.7 | |
| Diastolic blood pressure (mmHg) | FRC MC | 87.24 ± 11.8 87.28 ± 9.18 | 84 ± 9.12 * 87.31 ± 9.44 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leyva-Soto, A.; Chavez-Santoscoy, R.A.; Lara-Jacobo, L.R.; Chavez-Santoscoy, A.V.; Gonzalez-Cobian, L.N. Daily Consumption of Chocolate Rich in Flavonoids Decreases Cellular Genotoxicity and Improves Biochemical Parameters of Lipid and Glucose Metabolism. Molecules 2018, 23, 2220. https://doi.org/10.3390/molecules23092220
Leyva-Soto A, Chavez-Santoscoy RA, Lara-Jacobo LR, Chavez-Santoscoy AV, Gonzalez-Cobian LN. Daily Consumption of Chocolate Rich in Flavonoids Decreases Cellular Genotoxicity and Improves Biochemical Parameters of Lipid and Glucose Metabolism. Molecules. 2018; 23(9):2220. https://doi.org/10.3390/molecules23092220
Chicago/Turabian StyleLeyva-Soto, Aldo, Rocio Alejandra Chavez-Santoscoy, Linda Ramona Lara-Jacobo, Ana Vianey Chavez-Santoscoy, and Lina Natalia Gonzalez-Cobian. 2018. "Daily Consumption of Chocolate Rich in Flavonoids Decreases Cellular Genotoxicity and Improves Biochemical Parameters of Lipid and Glucose Metabolism" Molecules 23, no. 9: 2220. https://doi.org/10.3390/molecules23092220
APA StyleLeyva-Soto, A., Chavez-Santoscoy, R. A., Lara-Jacobo, L. R., Chavez-Santoscoy, A. V., & Gonzalez-Cobian, L. N. (2018). Daily Consumption of Chocolate Rich in Flavonoids Decreases Cellular Genotoxicity and Improves Biochemical Parameters of Lipid and Glucose Metabolism. Molecules, 23(9), 2220. https://doi.org/10.3390/molecules23092220







