Anti-Bacterial Adhesion Activity of Tropical Microalgae Extracts
Abstract
1. Introduction
2. Results and Discussion
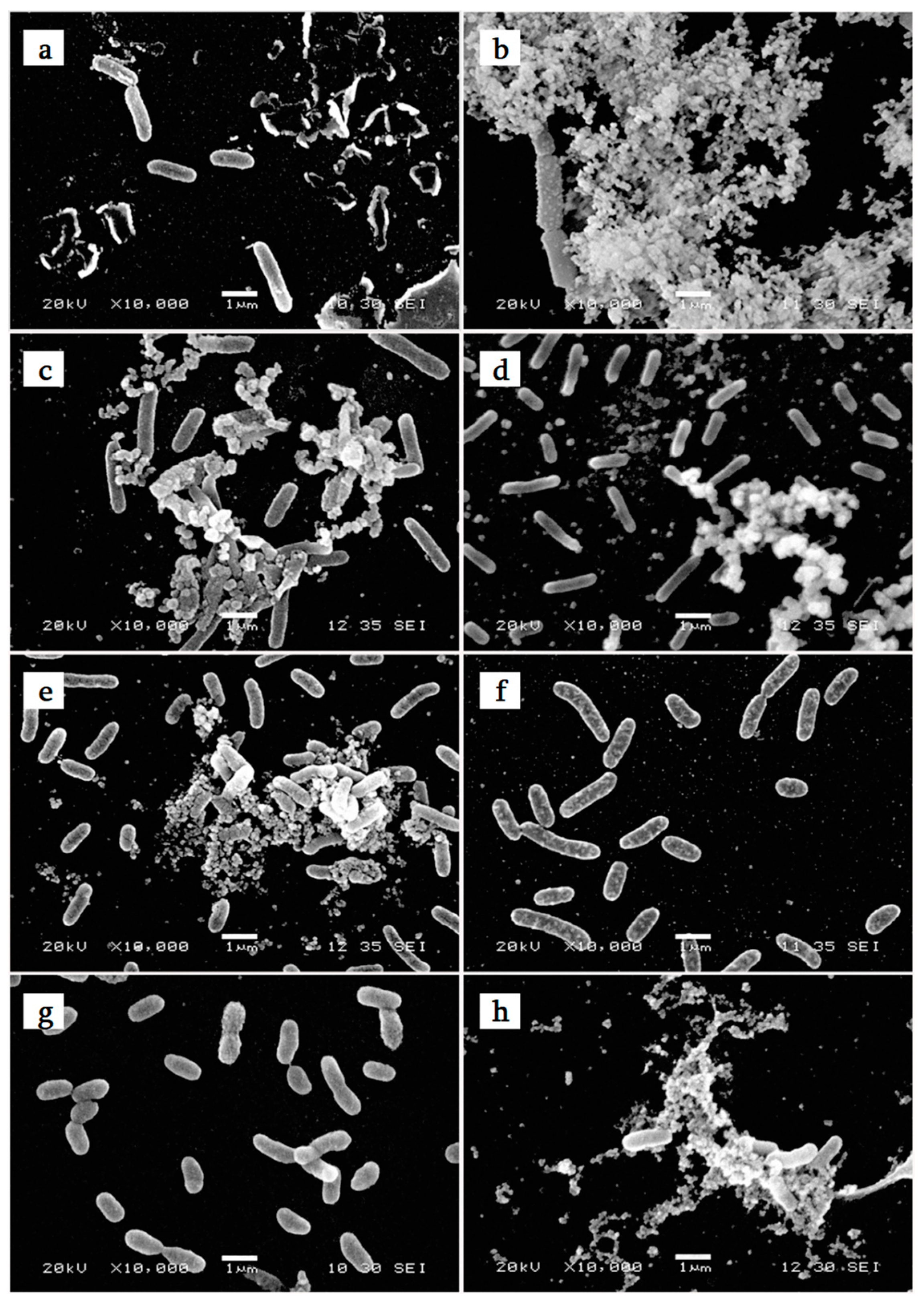
2.1. Bacterial Characterization
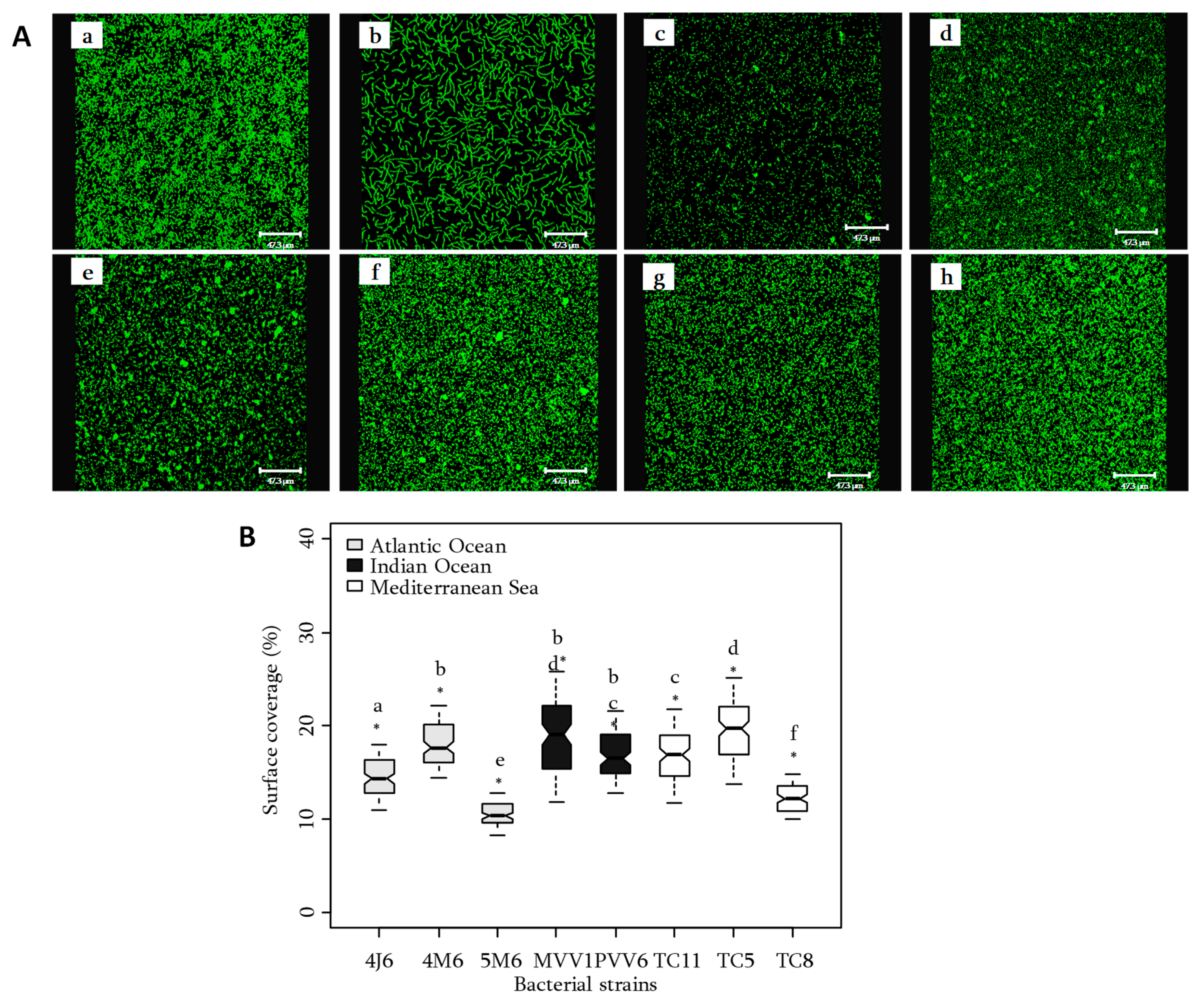
2.2. Bacterial Adhesion
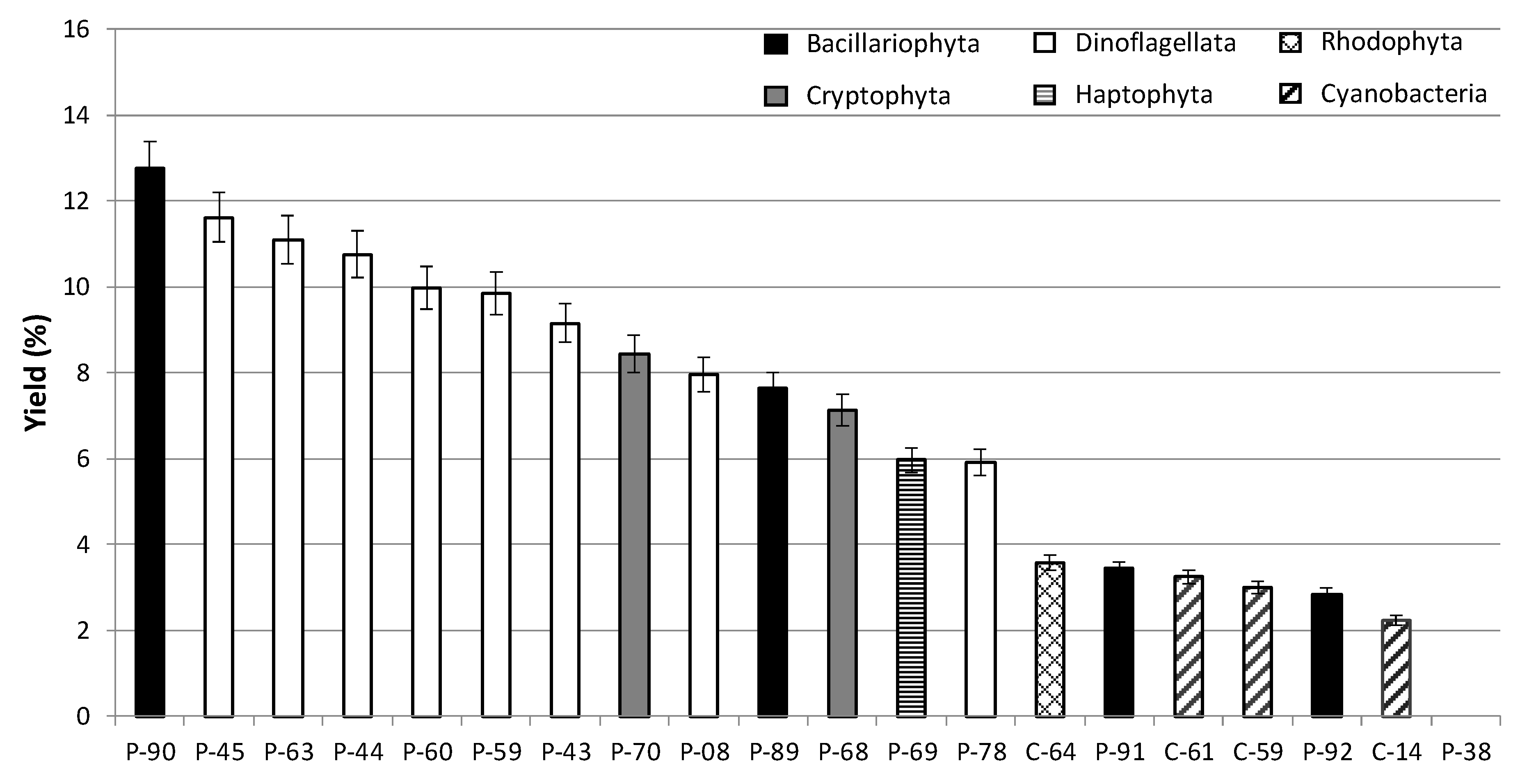
2.3. Extracts of Tropical Microalgae
2.4. Bioactivity of the Microalgal Extracts
2.4.1. Impact on Bacterial Growth and Viability
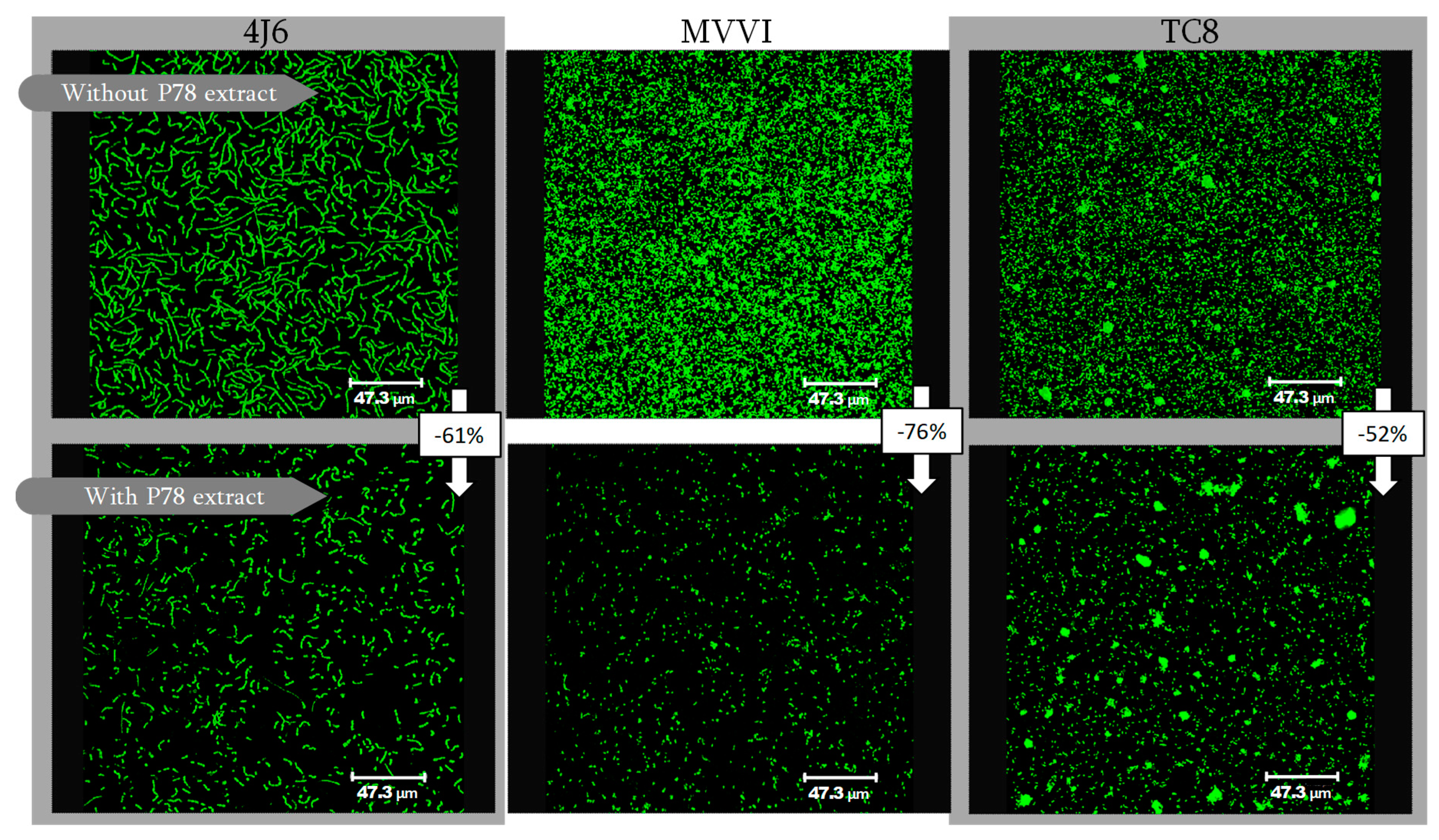
2.4.2. Impact on Bacterial Adhesion
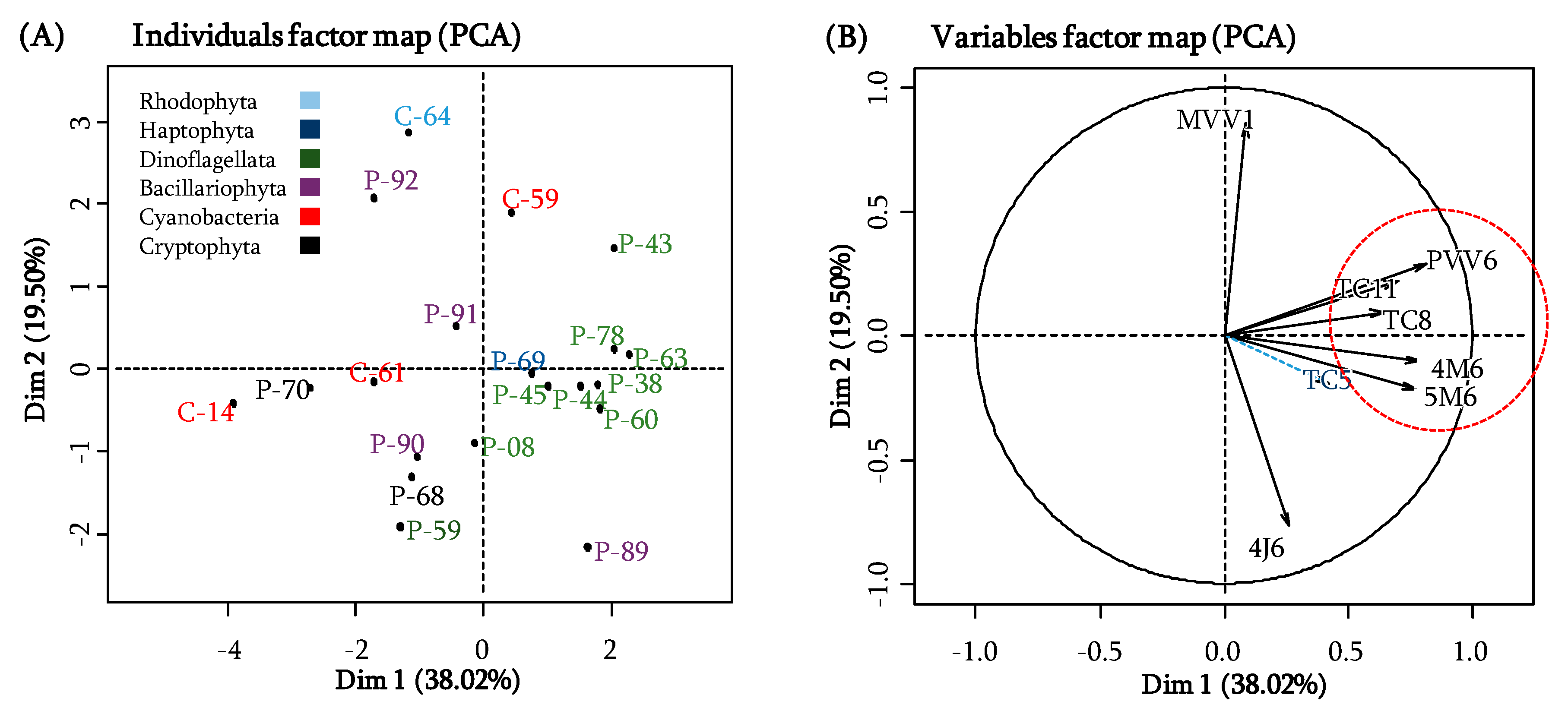
2.5. Which Microalgal Compounds Can Potentially Contribute to the Anti-adhesion Activity?
3. Materials and Methods
3.1. Bacterial Strains
3.1.1. Bacterial Growth and Morphology
3.1.2. Bacterial Adhesion in Flow Cell
3.2. Microalgal Strains
3.2.1. Isolation and Cultivation of Microalgae
3.2.2. Isolation and Cultivation of Cyanobacteria
3.2.3. Biomass Production
3.2.4. Extraction
3.3. Extract Bioactivity
3.3.1. Anti-Adhesion Assay
3.3.2. Determination of EC50 Values
3.3.3. Bacterial Growth Inhibition (EC50) and Viability Assays (LC50)
3.3.4. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Oliveira, I.B.; Groh, K.J.; Schönenberger, R.; Barroso, C.; Thomas, K.V.; Suter, M.J.-F. Toxicity of emerging antifouling biocides to non-target freshwater organisms from three trophic levels. Aquat. Toxicol. 2017, 191, 164–174. [Google Scholar] [CrossRef] [PubMed]
- Fusetani, N. Antifouling marine natural products. Nat. Prod. Rep. 2011, 28, 400–410. [Google Scholar] [CrossRef] [PubMed]
- Qian, P.Y.; Li, Z.; Xu, Y.; Li, Y.; Fusetani, N. Mini-review: Marine natural products and their synthetic analogs as antifouling compounds: 2009–2014. Biofouling 2015, 31, 101–122. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Xia, C.; Qian, P.Y. Optimization of antifouling coatings incorporating butenolide, a potent antifouling agent via field and laboratory tests. Prog. Org. Coat. 2017, 109, 22–29. [Google Scholar] [CrossRef]
- Qian, P.-Y.; Wong, Y.H.; Zhang, Y. Changes in the proteome and phosphoproteome expression in the bryozoan Bugula neritina larvae in response to the antifouling agent butanolide. Proteomics 2010, 10, 3435–3446. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.-F.; Xiao, K.; Chandramouli, K.H.; Xu, Y.; Pan, K.; Wang, W.X.; Qian, P.Y. Acute toxicity of the antifouling compound butenolide in non-target organisms. PLoS ONE 2011, 6, e23803. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhang, F.; Xu, Y.; Matsumura, K.; Han, Z.; Liu, L.; Lin, W.; Jia, Y.; Qian, P.Y. Structural optimization and evaluation of butenolides as potent antifouling agents: Modification of the side chain affects the biological activities of compounds. Biofouling 2012, 28, 857–864. [Google Scholar] [CrossRef] [PubMed]
- Bhatnagar, I.; Kim, S.-K. Immense essence of excellence: Marine microbial bioactive compounds. Mar. Drugs 2010, 8, 2673–2701. [Google Scholar] [CrossRef] [PubMed]
- Blunt, J.W.; Carroll, A.R.; Copp, B.R.; Davis, R.A.; Keyzers, R.A.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2018, 35, 8–53. [Google Scholar] [CrossRef] [PubMed]
- Bhadury, P.; Wright, P.C. Exploitation of marine algae: Biogenic compounds for potential antifouling applications. Planta 2004, 219, 561–578. [Google Scholar] [CrossRef] [PubMed]
- Amaro, H.M.; Guedes, A.C.; Malcata, F.X. Antimicrobial activities of microalgae: An invited review. Sci. Microb. Pathog. Commun. Curr. Res. Technol. Adv. 2011, 3, 1272–1284. [Google Scholar]
- Singh, S.; Kate, B.N.; Banerjee, U.C. Bioactive compounds from cyanobacteria and microalgae: An overview. Crit. Rev. Biotechnol. 2005, 25, 73–95. [Google Scholar] [CrossRef] [PubMed]
- Shannon, E.; Abu-Ghannam, N. Antibacterial derivatives of marine algae: An overview of pharmacological mechanisms and applications. Mar. Drugs 2016, 14, 81. [Google Scholar] [CrossRef] [PubMed]
- Desbois, A.P.; Mearns-Spragg, A.; Smith, V.J. A fatty acid from the diatom phaeodactylum tricornutum is antibacterial against diverse bacteria including multi-resistant Staphylococcus aureus (MRSA). Mar. Biotechnol. 2009, 11, 45–52. [Google Scholar] [CrossRef] [PubMed]
- ’t Lam, G.P.; Vermue, M.H.; Eppink, M.H.M.; Wijffels, R.H.; van den Berg, C. Multi-product microalgae biorefineries: From concept towards reality. Trends Biotechnol. 2018, 36, 216–227. [Google Scholar] [CrossRef] [PubMed]
- Qian, P.-Y.; Chen, L.; Xu, Y. Mini-review: Molecular mechanisms of antifouling compounds. Biofouling 2013, 29, 381–400. [Google Scholar] [CrossRef] [PubMed]
- Vilas-Boas, C.; Sousa, E.; Pinto, M.; Correia-da-Silva, M. An antifouling model from the sea: A review of 25 years of zosteric acid studies. Biofouling 2017, 33, 927–942. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.; Barrios, C.; Cutright, T.; Newby, B.Z. Assessment of antifouling effectiveness of two natural product antifoulants by attachment study with freshwater bacteria. Environ. Sci. Pollut. Res. 2005, 12, 278–284. [Google Scholar] [CrossRef]
- Wafar, M.; Venkataraman, K.; Ingole, B.; Ajmal Khan, S.; LokaBharathi, P. State of knowledge of coastal and marine biodiversity of Indian Ocean countries. PLoS ONE 2011, 6, e14613. [Google Scholar] [CrossRef] [PubMed]
- Franz, A.; Lehr, F.; Posten, C.; Schaub, G. Modeling microalgae cultivation productivities in different geographic locations–estimation method for idealized photobioreactors. Biotechnol. J. 2012, 7, 546–557. [Google Scholar] [CrossRef] [PubMed]
- Zubia, M.; Turquet, J.; Golubic, D. Benthic cyanobacterial diversity of iles eparses (scattered islands) in the Mozambique channel. Acta Oecologica. 2016, 72, 21–32. [Google Scholar] [CrossRef]
- Huerlimann, R.; De Nys, R.; Heimann, K. Growth, lipid content, productivity, and fatty acid composition of tropical microalgae for scale-up production. Biotechnol. Bioeng. 2010, 107, 245–257. [Google Scholar] [CrossRef] [PubMed]
- Zea Obando, C.; Linossier, I.; Dufossé, L.; Zubia, M.; Tunin-Ley, A.; Turquet, J.; Grondin, I.; Réjean, T.; Fay, F.; Réhel, K. Fatty acid profiling of tropical microalgae and cyanobacteria strains isolated from southwest indian Ocean islands. J. Marine Biol. Aquacult. 2017, 3, 1–14. [Google Scholar]
- Subrahmanyam, C.; Kumar, S.R.; Reddy, G.D. Bioactive compounds from the Indian ocean gorgonian Subergorgia suberosa (Pallas). Indian J. Chem. 2005, 44B, 2186–2188. [Google Scholar] [CrossRef]
- Kumar, C.S.; Sarada, D.V.; Gideon, T.P.; Rengasamy, R. Antibacterial activity of three South Indian seagrasses, Cymodocea serrulata, Halophila ovalis and Zostera capensis. World J. Microbiol. Biotechnol. 2008, 24, 1989–1992. [Google Scholar] [CrossRef]
- Shridhar, D.M.; Mahajan, G.B.; Kamat, V.P.; Naik, C.G.; Parab, R.R.; Thakur, N.R.; Mishra, P.D. Antibacterial activity of 2-(2′, 4′-Dibromophenoxy)-4, 6-dibromophenol from Dysidea granulosa. Mar. Drugs 2009, 7, 464–471. [Google Scholar] [CrossRef] [PubMed]
- Joel, E.L.; Bhimba, V. Isolation and characterization of secondary metabolites from the mangrove plant Rhizophora mucronata. Asian Pac. J. Trop. Med. 2010, 3, 602–604. [Google Scholar] [CrossRef]
- Beesoo, R.; Bhagooli, R.; Neergheen-Bhujun, V.S.; Li, W.-W.; Kagansky, A.; Bahorun, T. Antibacterial and antibiotic potentiating activities of tropical marine sponge extracts. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2017, 196, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Dixit, D.C.; Reddy, C.R.K.; Balar, N.; Suthar, P.; Gajaria, T.; Gadhavi, D.K. Assessment of the nutritive, biochemical, antioxidant and antibacterial potential of eight tropical macro algae along Kachchh coast, India as human food supplements. J. Aquat. Food Prod. Technol. 2018, 27, 61–79. [Google Scholar] [CrossRef]
- Nwodo, U.U.; Green, E.; Okoh, A.I. Bacterial exopolysaccharides: Functionality and prospects. Int. J. Mol. Sci. 2012, 13, 14002–14015. [Google Scholar] [CrossRef] [PubMed]
- Harimawan, A.; Ting, Y.-P. Investigation of extracellular polymeric substances (EPS) properties of P. aeruginosa and B. subtilis and their role in bacterial adhesion. Colloids Surf. B Biointerfaces 2016, 146, 459–467. [Google Scholar] [CrossRef] [PubMed]
- Tolker-Nielsen, T.; Sternberg, C. Methods for studying biofilm formation: Flow cells and confocal laser scanning microscopy. In Pseudomonas Methods and Protocols; Filloux, A., Ramos, J.L., Eds.; Springer: Berlin, Germany, 2014; pp. 615–629. [Google Scholar]
- Brian-Jaisson, F.; Ortalo-Magné, A.; Guentas-Dombrowsky, L.; Armougom, F.; Blache, Y.; Molmeret, M. Identification of bacterial strains isolated from the Mediterranean sea exhibiting different abilities of biofilm formation. Microb. Ecol. 2014, 68, 94–110. [Google Scholar] [CrossRef] [PubMed]
- Camps, M.; Briand, J.F.; Guentas-Dombrowsky, L.; Culioli, G.; Bazire, A.; Blache, Y. Antifouling activity of commercial biocides vs. natural and natural-derived products assessed by marine bacteria adhesion bioassay. Mar. Pollut. Bull 2011, 62, 1032–1040. [Google Scholar] [CrossRef] [PubMed]
- Faÿ, F.; Horel, G.; Linossier, I.; Vallée-Réhel, K. Effect of biocidal coatings on microfouling: In vitro and in situ results. Prog. Org. Coat. 2018, 114, 162–172. [Google Scholar] [CrossRef]
- Sánchez-Saavedra, M.; Licea-Navarro, A.; Bernáldez-Sarabia, J. Evaluation of the antibacterial activity of different species of phytoplankton. Rev. Biol. Mar. Oceanogr. 2010, 45, 531–536. [Google Scholar] [CrossRef]
- Al-Mola, H.F. Antibacterial activity of crude extracts and phlorotannin isolated from the diatom Cymbella spp. J. Pharm. Res. 2009, 2, 304–308. [Google Scholar]
- Dixit, R.B.; Suseela, M.R. Cyanobacteria: Potential candidates for drug discovery. Antonie Van Leeuwenhoek 2013, 103, 947–961. [Google Scholar] [CrossRef] [PubMed]
- Bajpai, V.K. Antimicrobial bioactive compounds from marine algae: A mini review. Ind. J. Geo-Mar. Sci. 2016, 45, 1076–1085. [Google Scholar]
- Daranas, A.H.; Norte, M.; Fernández, J.J. Toxic marine microalgae. Toxicon 2001, 39, 1101–1132. [Google Scholar] [CrossRef]
- Onodera, K.-I.; Nakamura, H.; Oba, Y.; Ohizumi, Y.; Ojika, M. Zooxanthellamice Cs: Vasoconstrictive polyhydroxylated Macrolides with the largest lactone ring size from a marine dinoflagellate of symbiodinium sp. J. Am. Chem. Soc. 2005, 127, 10406–10411. [Google Scholar] [CrossRef] [PubMed]
- Kita, M.; Kondo, M.; Komoyuki, T.; Yamada, K.; Matsumoto, T.; Lee, K.H.; Woo, J.T.; Uemura, D. Symbioimine exhibiting inhibitory effect of osteoclast differentiation, from the symbiotic marine dinoflagellate Symbiodium sp. J. Am. Chem. Soc. 2004, 126, 4794–4795. [Google Scholar] [CrossRef] [PubMed]
- Washida, K.; Koyama, T.; Yamada, K.; Kita, M.; Uemura, D. Karatungiols A and B, two novel antimicrobial polyol compounds, from the symbiotic marine dinoflagellate Amphidinium sp. Tetrahedron Lett. 2006, 47, 2521–2525. [Google Scholar] [CrossRef]
- Huang, S.J.; Kuo, C.M.; Lin, Y.C.; Chen, Y.M.; Lu, C.K.; Carteraol, E. A potent polyhydroxyl ichthyotoxin from the dinoflagellate Amphidinium carterae. Tetrahedron Lett. 2009, 50, 2512–2515. [Google Scholar] [CrossRef]
- Kobayashi, J.; Kubota, T. Bioactive macrolides and polyketides from marine dinoflagellates of the genus Amphidinium. J. Nat. Prod. 2007, 70, 451–460. [Google Scholar] [CrossRef]
- Kubota, T.; Iwai, T.; Ishiyama, H.; Sakai, K.; Gonoi, T.; Kobayashi, J. Amphidinin G, a putative biosynthetic precursor of amphidinin A from marine dinoflagellate Amphidinium sp. Tetrahedron Lett. 2015, 56, 990–993. [Google Scholar] [CrossRef]
- Bezić, N.; Skočibušić, M.; Dunkić, V.; Radonić, A. Composition and antimicrobial activity of Achillea clavennae L. essential oil. Phytother. Res. 2003, 17, 1037–1040. [Google Scholar] [CrossRef] [PubMed]
- Zea Obando, C.; Linossier, I.; Kervarec, N.; Zubia, M.; Turquet, J.; Faÿ, F.; Rehel, K. Rapid identification of osmolytes in tropical microalgae and cyanobacteria by 1H HR-MAS NMR spectroscopy. Talanta 2016, 153, 372–380. [Google Scholar] [CrossRef] [PubMed]
- Kirby, C.W.; McCallum, J.L.; Fofana, B. A 1H NMR study of the fatty acid distribution in developing flax bolls before and after a cooking treatment. Can. J. Chem. 2011, 89, 1138–1142. [Google Scholar] [CrossRef]
- Bergé, J.P.; Barnathan, G. Fatty Acids from Lipids of Marine Organisms: Molecular Biodiversity, Roles as Biomarkers, Biologically Active Compounds, and Economical Aspects. In Marine Biotechnology I. Advances in Biochemical Engineering/Biotechnology; Ulber, R., Le Gal, Y., Eds.; Springer: Berlin, Germany, 2005; Volume 96, pp. 49–125. [Google Scholar]
- Huang, C.B.; Ebersole, J.L. A novel bioactivity of omega-3 polyunsaturated fatty acids and their ester derivatives. Mol. Oral Microbiol. 2010, 25, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.B.; Alimova, Y.; Myers, T.M.; Ebersole, J.L. Short-and medium-chain fatty acids exhibit antimicrobial activity for oral microorganisms. Arch. Oral Biol. 2011, 56, 650–654. [Google Scholar] [CrossRef] [PubMed]
- Correia, M.; Michel, V.; Matos, A.A.; Carvalho, P.; Oliveira, M.J.; Ferreira, R.M.; Dillies, M.A.; Huerre, M.; Seruca, R.; Figueiredo, C.; et al. Docosahexaenoic acid inhibits Helicobacter pylori growth in vitro and mice gastric mucosa colonization. PLoS ONE 2012, 7, e35072. [Google Scholar] [CrossRef] [PubMed]
- Desbois, A.P.; Lawlor, K.C. Antibacterial activity of long-chain polyunsaturated fatty acids against Propionibacterium acnes and Staphylococcus aureus. Mar. Drugs 2013, 11, 4544–4557. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Zhou, Z.; Dong, J.; Zhang, J.; Xia, Y.; Shu, R. Antibacterial and antibiofilm activities of docosahexaenoic acid (DHA) and eicosapentaenoic acid (EPA) against periodontopathic bacteria. Microb. Pathog. 2016, 99, 196–203. [Google Scholar] [CrossRef] [PubMed]
- De Morais, M.G.; Vaz, B.S.; de Morais, E.G.; Costa, J.A.V. Biologically active metabolites synthesized by microalgae. BioMed Res. Int. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Desbois, A.P.; Smith, V.J. Antibacterial free fatty acids: Activities, mechanisms of action and biotechnological potential. Appl. Microbiol. Biotechnol. 2010, 85, 1629–1642. [Google Scholar] [CrossRef] [PubMed]
- Bazes, A.; Silkina, A.; Douzenel, P.; Faÿ, F.; Kervarec, N.; Morin, D.; Berge, J.P.; Bourgougnon, N. Investigation of the antifouling constituents from the brown alga Sargassum muticum (Yendo) Fensholt. J. Appl. Phycol. 2009, 21, 395–403. [Google Scholar] [CrossRef]
- Grasland, B.; Mitalane, J.; Briandet, R.; Quemener, E.; Meylheuc, T.; Linossier, I.; Vallée-Rehel, K.; Haras, D. Bacterial biofilm in seawater: Cell surface properties of early-attached marine bacteria. Biofouling 2003, 19, 307–313. [Google Scholar] [CrossRef] [PubMed]
- Mardèn, P.; Tunlid, A.; Malmcrona-Friberg, K.; Odham, G.; Kjelleberg, S. Physiological and morphological changes during short term starvation of marine bacterial isolates. Arch. Microbiol. 1985, 142, 326–332. [Google Scholar] [CrossRef]
- Andersen, R.A. Algal Culturing Techniques, 1st ed.; Elsevier: New York, NY, USA, 2005. [Google Scholar]
- Rippka, R. Isolation and purification of cyanobacteria. Methods Enzymol. 1988, 167, 3–27. [Google Scholar] [PubMed]
- Réveillon, D.; Tunin-Ley, A.; Grondin, I.; Othmani, A.; Zubia, M.; Bunet, R.; Turquet, J.; Culioli, G.; Briand, J.F. Exploring the chemodiversity of tropical microalgae for the discovery of natural antifouling compounds. J. Appl. Phycol. 2018. under revision. [Google Scholar]
Sample Availability: Samples of the compounds are available from the authors. |
| Growth | SEM Observation | ||||||
|---|---|---|---|---|---|---|---|
| Geographical Origin | Strain | Taxonomy | Neperian Growth Rate (h−1) | Generation Rate (h) | Beginning of the Stationary Phase (h) | Length (μm) | EPS Production |
| Atlantic Ocean | 4M6 | Paracoccus sp. | 0.72 | 0.97 | 11 | 1.76 ± 0.30 | − |
| 4J6 | Bacillus sp. | 0.47 | 1.47 | 10 | 2.05 ± 0.29 | ++ | |
| 5M6 | Pseudoalteromonas sp. | 0.51 | 1.35 | 10 | 1.98 ± 0.26 | + | |
| Mediterranean Sea | TC5 | Polibacter sp. | 0.87 | 0.80 | 7 | 1.35 ± 0.16 | + |
| TC8 | Pseudoalteromonas lipolityca | 0.76 | 0.91 | 5 | 1.33 ± 0.20 | + | |
| TC11 | Shewanella sp. | 0.72 | 0.96 | 8 | 1.63 ± 0.30 | − | |
| Indian Ocean | PVV6 | Shewanella sp. | 0.36 | 0.84 | 6 | 1.23 ± 0.07 | − |
| MVV1 | Shewanella sp. | 0.58 | 0.52 | 5 | 1.33 ± 0.11 | + | |
| Atlantic Ocean | Mediterranean Sea | Indian Ocean | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Bacillus sp. | Paracoccus sp. | Pseudoalt. | Polibacter sp. | Pseudoalt. | Shewanella sp. | Shewanella sp. | Shewanella sp. | ||
| Strain | Group | 4J6 | 4M6 | 5M6 | TC5 | TC8 | TC11 | PVV6 | MVV1 |
| P-78 | a | 61 ± 5 | 60 ± 3 | 87 ± 5 | 61 ± 4 | 52 ± 1 | 41 ± 4 | 61 ± 8 | 76 ± 4 |
| P-43 | a | 41 ± 5 | 40 ± 10 | 71 ± 6 | 51 ± 10 | 62 ± 2 | 53 ± 7 | 91 ± 4 | 82 ± 10 |
| P-63 | a | 38 ± 7 | 74 ± 1 | 68 ± 7 | 51 ± 3 | 78 ± 4 | 46 ± 1 | 70 ± 5 | 36 ± 10 |
| P-60 | b | 86 ± 3 | 46 ± 4 | 63 ± 6 | 37 ± 7 | 66 ± 6 | 49 ± 3 | 69 ± 6 | 56 ± 3 |
| P-89 | c | 68 ± 5 | 44 ± 7 | 64 ± 9 | 70 ± 4 | 32 ± 3 | 45 ± 5 | 86 ± 6 | −22 ± 2 |
| P-45 | d | 67 ± 9 | 25 ± 3 | 49 ± 6 | 49 ± 3 | 51 ± 1 | 42 ± 5 | 70 ± 7 | 48 ± 2 |
| P-69 | d | 54 ± 2 | 59 ± 9 | 86 ± 5 | 17 ± 15 | 43 ± 23 | 16 ± 15 | 53 ± 27 | 52 ± 8 |
| C-59 | e | 23 ± 2 | 19 ± 1 | 60 ± 8 | 12 ± 4 | 69 ± 3 | 40 ± 3 | 63 ± 8 | 72 ± 2 |
| P-38 | e | 58 ± 1 | 60 ± 2 | 72 ± 4 | 12 ± 12 | 67 ± 2 | 53 ± 2 | 75 ± 2 | 29 ± 2 |
| P-44 | e | 41 ± 5 | 48 ± 1 | 64 ± 26 | 41 ± 9 | 78 ± 5 | 48 ± 7 | 57 ± 5 | 19 ± 5 |
| P-90 | f | 75 ± 9 | −16 ± 7 | 29 ± 5 | 28 ± 8 | 34 ± 6 | 49 ± 12 | 17 ± 6 | 22 ± 7 |
| P-91 | f | 60 ± 2 | 46 ± 7 | −20 ± 5 | 1 ± 9 | 33 ± 11 | 32 ± 9 | 72 ± 13 | 48 ± 5 |
| P-08 | f | 48 ± 4 | 20 ± 4 | 43 ± 9 | 28 ± 3 | 56 ± 4 | 44 ± 4 | 25 ± 4 | 1 ± 5 |
| P-59 | f | 57 ± 2 | 11 ± 4 | 51 ± 10 | 45 ± 4 | 57 ± 7 | −50 ± 7 | 34 ± 4 | −1 ± 5 |
| C-64 | f | −7 ± 6 | 2 ± 6 | 28 ± 2 | 26 ± 5 | 22 ± 6 | 20 ± 5 | 64 ± 7 | 84 ± 7 |
| P-68 | g | 52 ± 4 | 46 ± 3 | 65 ± 7 | 19 ± 16 | 19 ± 12 | 3 ± 12 | −6 ± 13 | 19 ± 9 |
| P-70 | g | 36 ± 3 | −7 ± 11 | −41 ± 11 | 49 ± 3 | 42 ± 2 | −3 ± 9 | −6 ± 10 | 26 ± 7 |
| C-61 | g | 51 ± 5 | −16 ± 5 | −6 ± 4 | 47 ± 5 | 73 ± 8 | 21 ± 8 | −18 ± 2 | 43 ± 3 |
| C-14 | g | 55 ± 7 | 5 ± 3 | −2 ± 5 | −11 ± 8 | −37 ± 5 | −6 ± 15 | −12 ± 3 | 43 ± 4 |
| P-92 | g | 7 ± 2 | 2 ± 2 | −41 ± 4 | 37 ± 4 | 49 ± 2 | 21 ± 6 | 41 ± 3 | 62 ± 1 |
| Strain | 4M6 | 4J6 | 5M6 | TC5 | TC8 | TC11 | PVV6 | MVV1 |
|---|---|---|---|---|---|---|---|---|
| Extracts Number | 4 | 12 | 11 | 4 | 11 | 2 | 12 | 6 |
| Microalgal Strain | Bacterial Strain | |||
|---|---|---|---|---|
| 4J6 | 4M6 | MVV1 | ||
| P-43 | EC50 (μg mL−1) | 32 ± 6 | 73 ± 2 | 31 ± 7 |
| Hill slope | Median | Median | Low | |
| R2 | 0.9 ± 0.2 | 0.9 ± 0.1 | 0.9 ± 0.3 | |
| P-60 | EC50 (μg mL−1) | 25 ± 1 | 57 ± 13 | 39 ± 10 |
| Hill slope | High | Median | Low | |
| R2 | 0.9 ± 0.1 | 0.9 ± 0.1 | 0.9 ± 0 | |
| P-78 | EC50 (μg mL−1) | 37 ± 4. | 35 ± 6 | 21 ± 1 |
| Hill slope | Median | Median | Low | |
| R2 | 0.9 ± 0.1 | 0.8 ± 0.2 | 0.9 ± 0 | |
| Strain | Taxonomy | Origin | Gram | Source |
|---|---|---|---|---|
| 4M6 | Paracoccus sp. | Atlantic Ocean | − | [59] |
| 4J6 | Bacillus sp. | + | ||
| 5M6 | Pseudoalteromonas sp. | − | ||
| TC5 | Polibacter sp. | Mediterranean Sea | − | |
| TC8 | Pseudoalteromonas lipolityca | − | [33,34] | |
| TC11 | Shewanella sp. | − | ||
| PVV6 | Shewanella sp. | Indian Ocean | − | This study |
| MVV1 | Shewanella sp. | − |
| Strain Code a | Phylum | Family | Genus | Species | Origin | Growth Medium |
|---|---|---|---|---|---|---|
| C-64 | Rhodophyta | ND b | Porphyridium | sp. | Glorioso | BG11 c |
| P-69 | Haptophyta | Pavlovaceae | Pavlova | sp. | Glorioso | F/2 d |
| P-38 | Dinoflagellata | Gymnodiniaceae | Amphidinium | sp. | Reunion | F/2 |
| P-43 | Amphidinium | sp. | Europa | F/2 | ||
| P-44 | Amphidinium | massartii | Glorioso | F/2 | ||
| P-45 | Amphidinium | sp. | Glorioso | F/2 | ||
| P-59 | Amphidinium | sp. | Madagascar | F/2 | ||
| P-60 | Amphidinium | operculatum | Madagascar | F/2 | ||
| P-63 | Amphidinium | sp. | Madagascar | F/2 | ||
| P-08 | Prorocentraceae | Prorocentrum | lima | Reunion | F/2 | |
| P-78 | Symbiodiniaceae | Symbiodinium | sp. (Clade D) | Reunion | F/2 | |
| P-91 | Bacillariophyta | Bacillariaceae | Navicula | mollis | Reunion | F/2 + Si e |
| P-92 | Navicula | sp. | Reunion | F/2 + Si | ||
| P-89 | Psammodictyon | cf. constrictum | Reunion | F/2 + Si | ||
| P-90 | Nitzschia | sp. | Reunion | F/2 + Si | ||
| C-59 | Cyanobacteria | ND | ND b | sp5 | Glorioso | BG11 |
| C-61 | ND b | sp6 | Glorioso | BG11 | ||
| C-14 | ND b (LPP-group) | sp. LPP1 | Mayotte | BG11 | ||
| P-70 | Cryptophyta | ND | NDb | sp1 | Glorioso | F/2 |
| P-68 | NDb | sp3 | Glorioso | F/2 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zea-Obando, C.; Tunin-Ley, A.; Turquet, J.; Culioli, G.; Briand, J.-F.; Bazire, A.; Réhel, K.; Faÿ, F.; Linossier, I. Anti-Bacterial Adhesion Activity of Tropical Microalgae Extracts. Molecules 2018, 23, 2180. https://doi.org/10.3390/molecules23092180
Zea-Obando C, Tunin-Ley A, Turquet J, Culioli G, Briand J-F, Bazire A, Réhel K, Faÿ F, Linossier I. Anti-Bacterial Adhesion Activity of Tropical Microalgae Extracts. Molecules. 2018; 23(9):2180. https://doi.org/10.3390/molecules23092180
Chicago/Turabian StyleZea-Obando, Claudia, Alina Tunin-Ley, Jean Turquet, Gérald Culioli, Jean-François Briand, Alexis Bazire, Karine Réhel, Fabienne Faÿ, and Isabelle Linossier. 2018. "Anti-Bacterial Adhesion Activity of Tropical Microalgae Extracts" Molecules 23, no. 9: 2180. https://doi.org/10.3390/molecules23092180
APA StyleZea-Obando, C., Tunin-Ley, A., Turquet, J., Culioli, G., Briand, J.-F., Bazire, A., Réhel, K., Faÿ, F., & Linossier, I. (2018). Anti-Bacterial Adhesion Activity of Tropical Microalgae Extracts. Molecules, 23(9), 2180. https://doi.org/10.3390/molecules23092180








