Synthesis and Biological Evaluation of Novel Alkyl Amine Substituted Icariside II Derivatives as Potential Anticancer Agents
Abstract
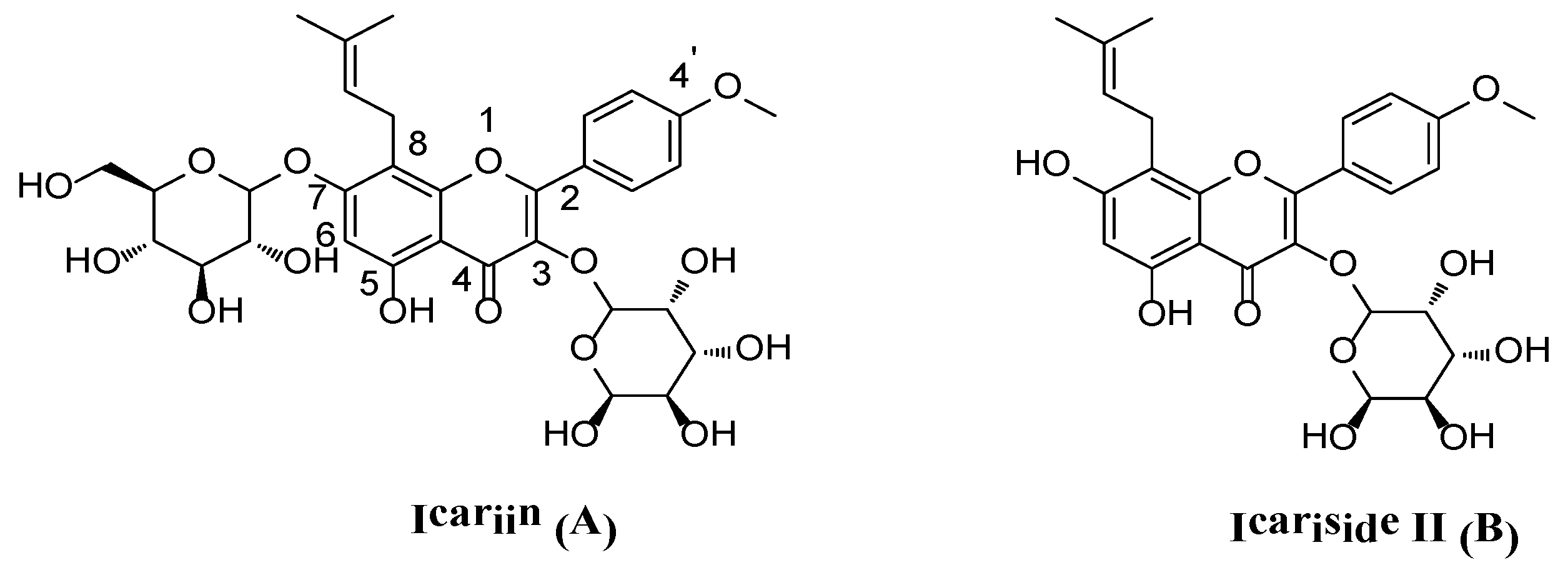
1. Introduction
2. Results and Discussion
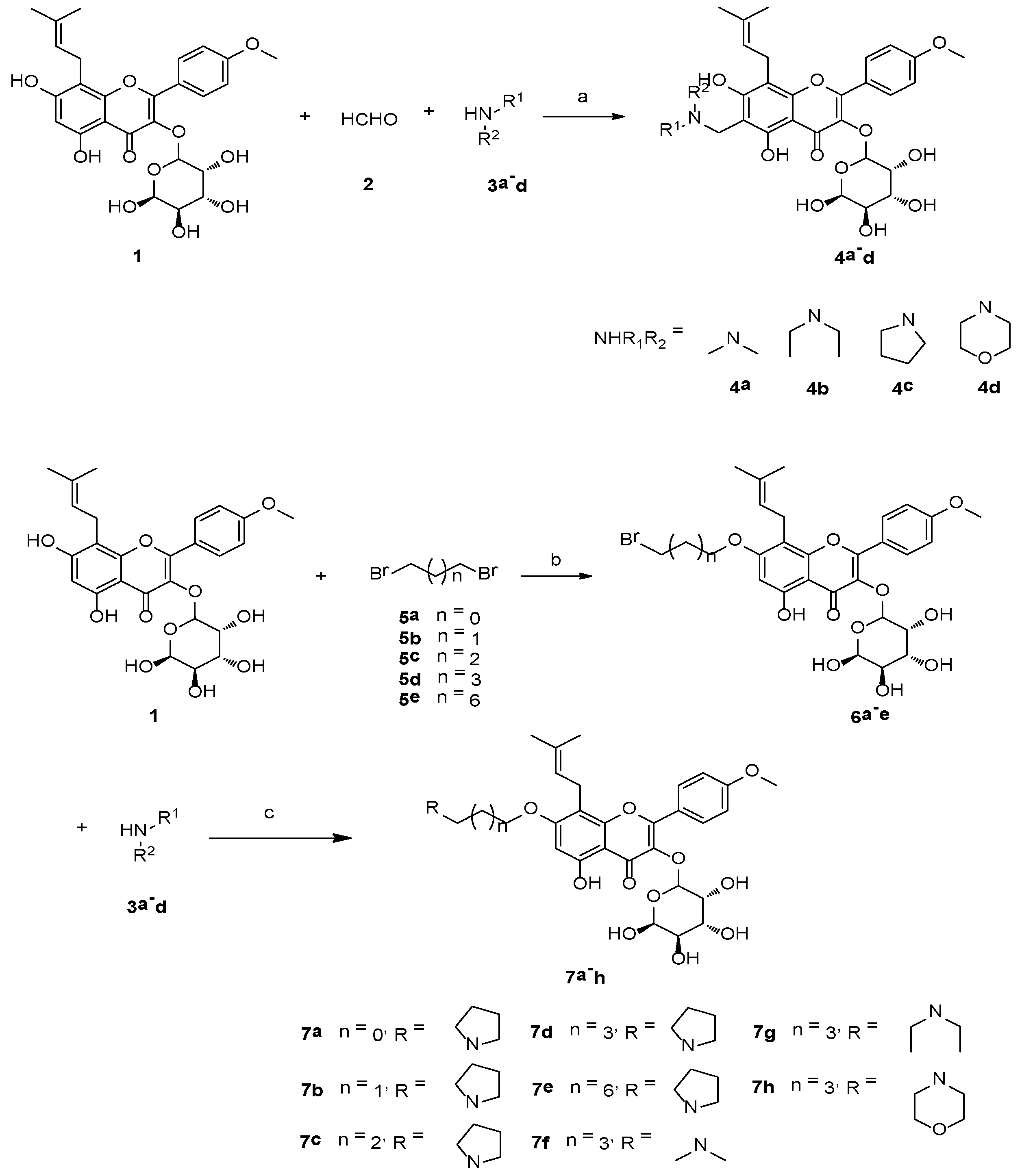
2.1. Chemistry
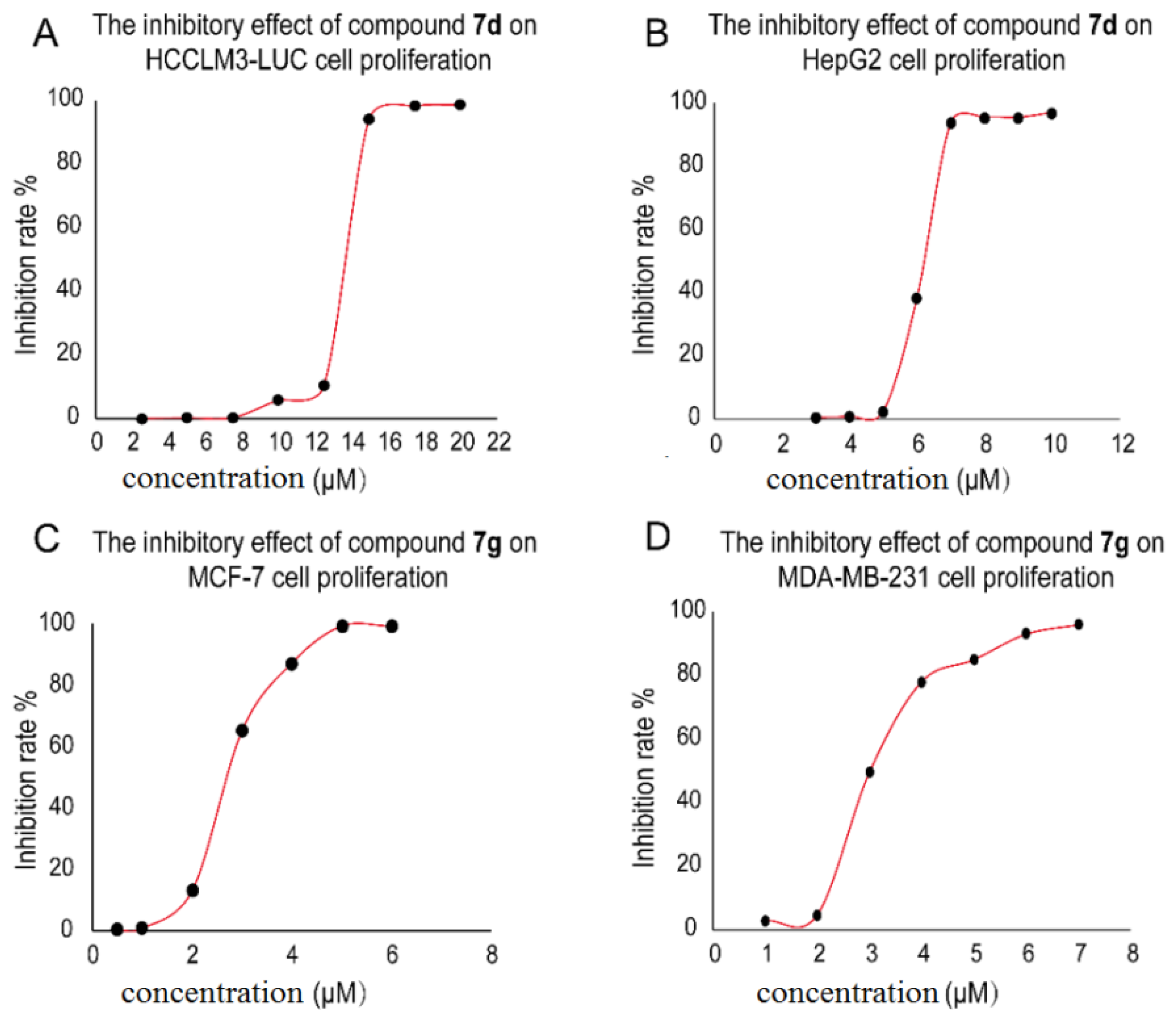
2.2. Cell Viability Analysis and Discussion on the Preliminary Structure-Activity Relationship (SAR)
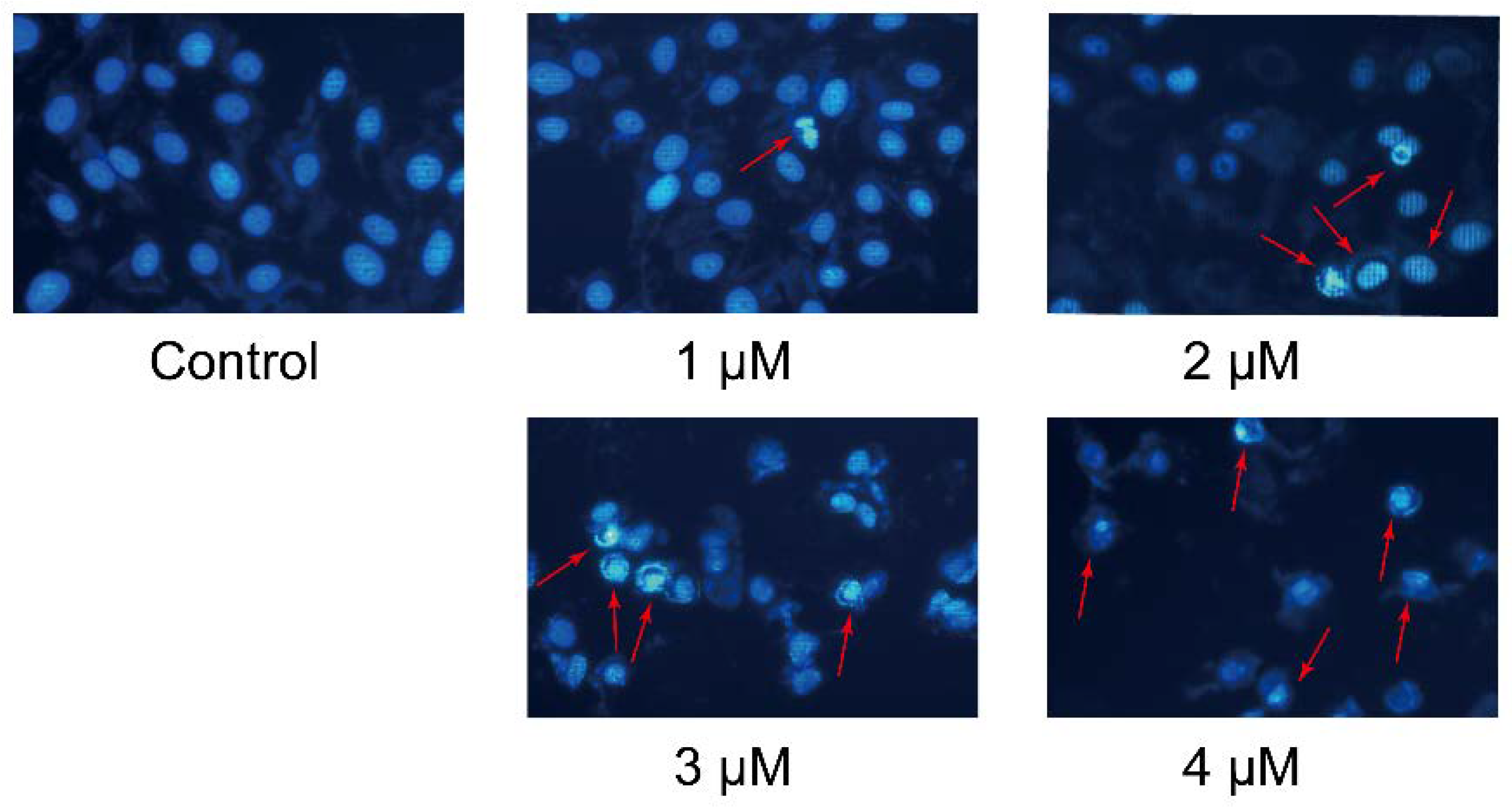
2.3. Cell Morphological Assessment
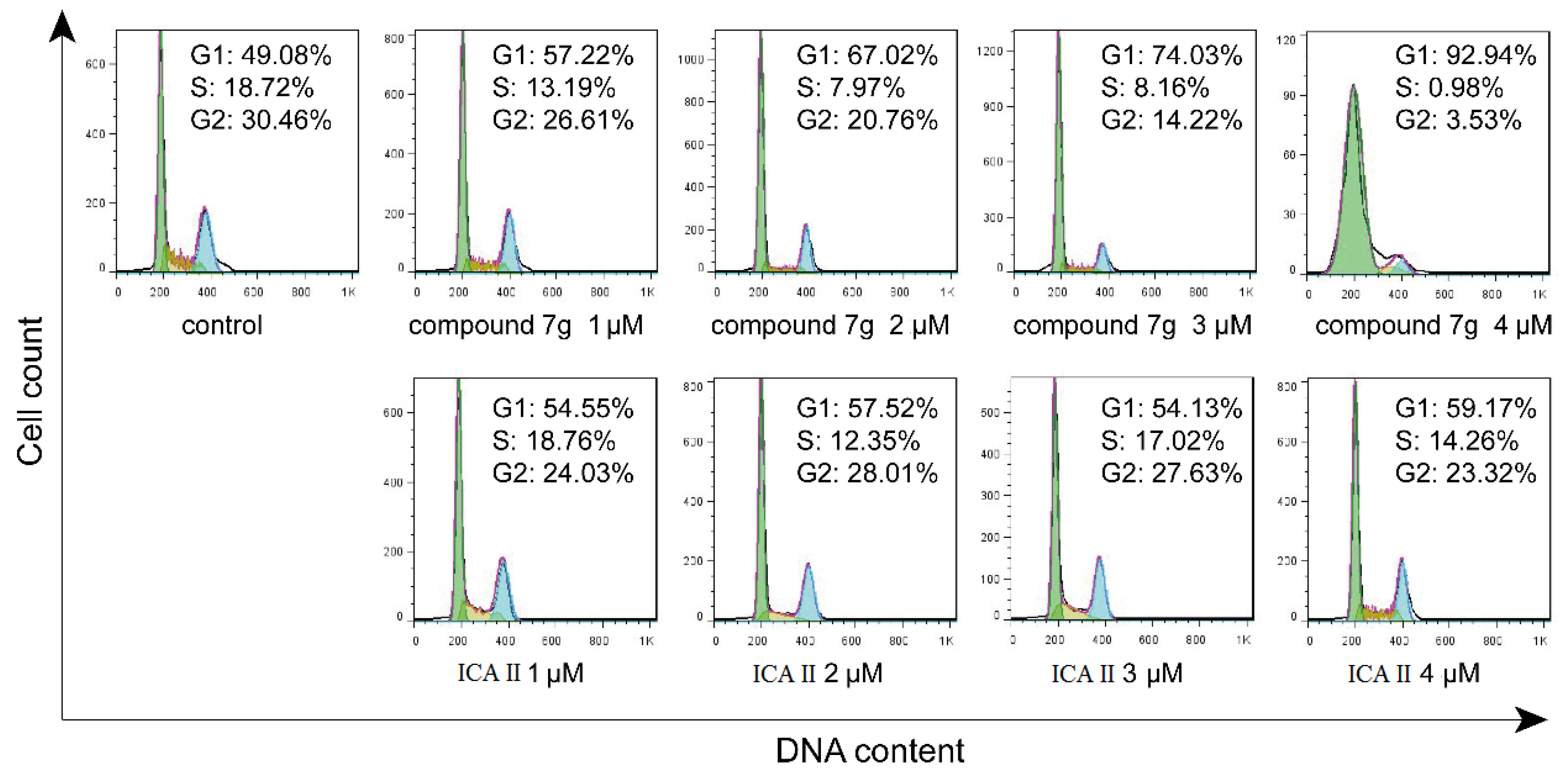
2.4. Cell Cycle Distribution Analysis
2.5. Assessment of Apoptotic Analysis
3. Conclusions
4. Experimental Section
4.1. Materials and Methods
4.2. General Experimental Procedure for Mannich Base Derivatives 4a–d
4.3. General Experimental Procedure for the 7-O-Alkylamino Derivatives 7a–h
4.4. Cell Lines and Cell Culture Conditions
4.5. Cell Viability Inhibition Assay
4.6. Cell Morphological Assessment
4.7. Cell Cycle Distribution Analysis
4.8. Assessment of Apoptotic Analysis
Author Contributions
Funding
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics. CA Cancer J. Clin. 2015, 65, 5–29. [Google Scholar] [CrossRef] [PubMed]
- Poustchi, H.; Sepanlou, S.; Esmaili, S.; Mehrabi, N.; Ansarymoghadam, A. Hepatocellular carcinoma in the world and the Middle East. Middle East J. Dig. Dis. 2010, 2, 31–41. [Google Scholar] [PubMed]
- Bugano, D.D.; Conforti-Froes, N.; Yamaguchi, N.H.; Baracat, E.C. Genetic polymorphisms, the metabolism of estrogens and breast cancer: A review. Eur. J. Gynaecol. Oncol. 2008, 29, 313–320. [Google Scholar] [PubMed]
- Gao, Y.; Chen, L.; Zhang, Z.; Chen, Y.; Li, Y. Reversal of multidrug resistance by reduction-sensitive linear cationic click polymer/iMDR1-pDNA complex nanoparticles. Biomaterials 2011, 32, 1738–1747. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Pan, Y.; Wang, H.; Li, H.; Peng, Q.; Wei, D.; Chen, C.; Zheng, J. Synthesis and evaluation of new pyrazoline derivatives as potential anticancer agents in HepG-2 cell line. Molecules 2017, 22, 467. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Gu, W.; Shari, Y.; Liu, F.; Ku, X.; Yang, Y.; Zhang, Q.; Zhang, Y.; Kuang, H.; Wang, Z.; Wang, S. Design, synthesis and anticancer activity of novel nopinone-based thiosemicarbazone derivatives. Bioorg. Med. Chem. Lett. 2017, 27, 2360–2363. [Google Scholar] [CrossRef] [PubMed]
- Galanski, M.; Arion, V.B.; Jakupec, M.A.; Keppler, B.K. Recent developments in the field of tumor-inhibiting metal complexes. Curr. Pharm. Des. 2003, 9, 2078–2089. [Google Scholar] [CrossRef] [PubMed]
- Yan, Z.; Lai, Z.; Lin, J. Anticancer properties of Traditional Chinese Medicine. Comb. Chem. High Throughput Screen. 2017, 20, 423–429. [Google Scholar] [CrossRef] [PubMed]
- Guo, B.L.; Li, W.K.; Yu, J.G.; Xiao, P.G. Brevicornin, a new flavone from Epimedium brevicornum. Phytochemistry 1996, 41, 991–992. [Google Scholar] [CrossRef]
- Liu, B.; Xu, C.; Wu, X.; Liu, F.; Du, Y.; Sun, J.; Tao, J.; Dong, J. Icariin exerts an antidepressant effect in an unpredictable chronic mild stress model of depression in rats and is associated with the regulation of hippocampal neuroinflammation. Neuroscience 2015, 294, 193–205. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.B.; Yang, Q.T. The testosterone mimetic properties of icariin. Asian J. Androl. 2006, 8, 601–605. [Google Scholar] [CrossRef] [PubMed]
- Wei, Z.; Wang, M.; Hong, M.; Diao, S.; Liu, A.; Huang, Y.; Yu, Q.; Peng, Z. Icariin exerts estrogen-like activity in ameliorating EAE via mediating estrogen receptor β, modulating HPA function and glucocorticoid receptor expression. Am. J. Transl. Res. 2016, 8, 1910–1918. [Google Scholar] [PubMed]
- Liu, P.; Jin, X.; Lv, H.; Li, J.; Xu, W.; Qian, H.; Yin, Z. Icaritin ameliorates carbon tetrachloride-induced acute liver injury mainly because of the antioxidative function through estrogen-like effects. In Vitro Cell. Dev. Biol. Anim. 2014, 50, 899–908. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Xiong, Y.; Zhu, X.; Gao, H.; Yin, S.; Wang, J.; Chen, G.; Wang, C.; Xiang, L.; Wang, P.; et al. Icariin improves osteoporosis, inhibits the expression of PPARγ, C/EBPα, FABP4 mRNA, N1ICD and jagged1 proteins, and increases Notch2 mRNA in ovariectomized rats. Exp. Ther. Med. 2017, 13, 1360–1368. [Google Scholar] [CrossRef] [PubMed]
- Zheng, C.M.; Liu, X.Z.; Li, Q.L.; Wang, J.F.; Tan, Z.; Ge, M.H. The bisphenol A-enhanced activity of thyroid carcinoma cell line B-CPAP is inhibited by Icarrin. Chin. J. Otorhinolaryngol. Head Neck Surg. 2017, 52, 458–462. [Google Scholar]
- Sun, Y.; Sun, X.H.; Fan, W.J.; Jiang, X.M.; Li, A.W. Icariin induces S-phase arrest and apoptosis in medulloblastoma cells. Cell Mol. Biol. 2016, 62, 123–129. [Google Scholar] [PubMed]
- Xia, Q.; Xu, D.; Huang, Z.; Liu, J.; Wang, X.; Wang, X.; Liu, S. Preparation of icariside II from icariin by enzymatic hydrolysis method. Fitoterapia 2010, 81, 437–442. [Google Scholar] [CrossRef] [PubMed]
- Cheng, T.; Zhang, Y.; Zhang, T.; Lu, L.; Ding, Y.; Zhao, Y. Comparative pharmacokinetics study of Icariin and Icariside II in rats. Molecules 2015, 20, 21274–21286. [Google Scholar] [CrossRef] [PubMed]
- Luo, G.; Gu, F.; Zhang, Y.; Liu, T.; Guo, P.; Huang, Y. Icariside II promotes osteogenic differentiation of bone marrow stromal cells in beagle canine. Int. J. Clin. Exp. Pathol. 2015, 8, 4367–4377. [Google Scholar] [PubMed]
- Gao, J.; Deng, Y.; Yin, C.; Liu, Y.; Zhang, W.; Shi, J.; Gong, Q. Icariside II, a novel phosphodiesterase 5 inhibitor, protects against H2O2-induced PC12 cells death by inhibiting mitochondria-mediated autophagy. J. Cell Mol. Med. 2017, 21, 375–386. [Google Scholar] [CrossRef] [PubMed]
- Bai, G.Y.; Zhou, F.; Hui, Y.; Xu, Y.D.; Lei, H.E.; Pu, J.X.; Xin, Z.C. Effects of Icariside II on corpus cavernosum and major pelvic ganglion neuropathy in streptozotocin-induced diabetic rats. Int. J. Mol. Sci. 2014, 15, 23294–23306. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Xie, M.; Jiang, N.; Huang, F.; Zhang, X.; Li, R.; Lu, J.; Liao, S.; Liu, Y. Icarisid II inhibits the proliferation of human osteosarcoma cells by inducing apoptosis and cell cycle arrest. Tumour Biol. 2017, 39. [Google Scholar] [CrossRef] [PubMed]
- Geng, Y.D.; Zhang, C.; Shi, Y.M.; Xia, Y.Z.; Guo, C.; Yang, L.; Kong, L.Y. Icariside II-induced mitochondrion and lysosome mediated apoptosis is counterbalanced by an autophagic salvage response in hepatoblastoma. Cancer Lett. 2015, 366, 19–31. [Google Scholar] [CrossRef] [PubMed]
- Cheng, T.; Yang, J.; Zhang, T.; Yang, Y.S.; Ding, Y. Optimized biotransformation of Icariin into Icariside II by β-Glucosidase from trichoderma viride using central composite design method. BioMed Res. Int. 2016. [Google Scholar] [CrossRef]
- Yan, H.; Song, J.; Jia, X.; Zhang, Z. Hyaluronic acid-modified didecyldimethylammonium bromide/d-a-tocopheryl polyethylene glycol succinate mixed micelles for delivery of baohuoside I against non-small cell lung cancer: In vitro and in vivo evaluation. Drug Deliv. 2017, 24, 30–39. [Google Scholar] [CrossRef] [PubMed]
- Joshi, D.; Field, J.; Murphy, J.; Abdelrahim, M.; Schönherr, H.; Sparrow, J.; Ellestad, G.; Nakanishi, K.; Zask, A. Synthesis of antioxidants for prevention of age-related macular degeneration. J. Nat. Prod. 2013, 76, 450–454. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Song, S.; Shen, M.; Tang, Y.; Shi, Z.; Tang, H.; Shi, Q.; Fu, Y.; Duan, J. Mannich bases of scutellarein as thrombin-inhibitors: Design, synthesis, biological activity and solubility. Bioorg. Med. Chem. 2012, 20, 6919–6923. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Wu, P.; Shi, J.F.; Jiang, Z.H.; Wei, X.Y. Synthesis and cancer cell growth inhibitory activity of icaritin derivatives. Eur. J. Med. Chem. 2015, 100, 139–150. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhang, Y.; Li, Y. β-elemene decreases cell invasion by upregulating E-cadherin expression in MCF-7 human breast cancer cells. Oncol. Rep. 2013, 30, 745–750. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Lu, N.; Dai, Q.; Wei, L.; Zhao, Q.; Li, Z.; He, Q.; Dai, Y.; Guo, Q. GL-V9, a newly synthetic flavonoid derivative, induces mitochondrial-mediated apoptosis and G2/M cell cycle arrest in human hepatocellular carcinoma HepG2 cells. Eur. J. Pharmacol. 2011, 670, 13–21. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds 4a–d and 7a–h are available from the authors. |

| Compound | IC50 (μM) a | |||
|---|---|---|---|---|
| HCCLM3-LUC | HepG2 | MCF-7 | MDA-MB-231 | |
| 4a | >50 | >50 | >50 | >50 |
| 4b | >50 | >50 | >50 | >50 |
| 4c | >50 | >50 | >50 | >50 |
| 4d | >50 | >50 | >50 | >50 |
| 7a | 18.72 ± 0.33 | 15.41 ± 0.60 | 14.97 ± 0.12 | 16.85 ± 0.24 |
| 7b | 14.23 ± 1.02 | 7.67 ± 0.83 | 10.53 ± 1.34 | 8.42 ± 0.45 |
| 7c | Nd b | Nd | Nd | Nd |
| 7d | 13.51 ± 0.93 | 7.67 ± 0.67 | 10.98 ± 0.22 | 10.15 ± 0.74 |
| 7e | 13.9 ± 1.12 | 7.12 ± 0.83 | 10.94 ± 0.94 | 8.58 ± 0.69 |
| 7f | 46.82 ± 5.94 | 19.92 ± 1.19 | 31.46 ± 2.37 | 31.08 ± 4.62 |
| 7g | 13.28 ±0.44 | 3.96 ± 0.37 | 2.44 ± 0.18 | 4.21 ± 0.31 |
| 7h | 15.31 ± 0.87 | 7.14 ± 0.69 | 14.61 ± 1.05 | 9.57 ± 0.59 |
| Icariin | >100 | >100 | >100 | >100 |
| Icariside II | 39.04 ± 1.92 | 21.92 ± 2.04 | 30.64 ± 3.46 | 32.53 ± 2.18 |
| doxorubicin | 1.97 ± 0.11 | 0.96 ± 0.06 | 16.16 ± 0.47 | 2.36 ± 0.14 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, T.; Li, T.; Kang, Y.-N.; Liu, L.; Wang, X.-M.; Lan, J.-S.; Ding, Y.; Zhang, T. Synthesis and Biological Evaluation of Novel Alkyl Amine Substituted Icariside II Derivatives as Potential Anticancer Agents. Molecules 2018, 23, 2146. https://doi.org/10.3390/molecules23092146
Wu T, Li T, Kang Y-N, Liu L, Wang X-M, Lan J-S, Ding Y, Zhang T. Synthesis and Biological Evaluation of Novel Alkyl Amine Substituted Icariside II Derivatives as Potential Anticancer Agents. Molecules. 2018; 23(9):2146. https://doi.org/10.3390/molecules23092146
Chicago/Turabian StyleWu, Tong, Ting Li, Ya-Nan Kang, Li Liu, Xi-Man Wang, Jin-Shuai Lan, Yue Ding, and Tong Zhang. 2018. "Synthesis and Biological Evaluation of Novel Alkyl Amine Substituted Icariside II Derivatives as Potential Anticancer Agents" Molecules 23, no. 9: 2146. https://doi.org/10.3390/molecules23092146
APA StyleWu, T., Li, T., Kang, Y.-N., Liu, L., Wang, X.-M., Lan, J.-S., Ding, Y., & Zhang, T. (2018). Synthesis and Biological Evaluation of Novel Alkyl Amine Substituted Icariside II Derivatives as Potential Anticancer Agents. Molecules, 23(9), 2146. https://doi.org/10.3390/molecules23092146





