Valorization of Olive Pomace-Based Nutraceuticals as Antioxidants in Chemical, Food, and Biological Models
Abstract
1. Introduction
2. Results and Discussion
2.1. Chemical Composition and Antioxidant Activity in Chemical Model Systems
2.1.1. Chemical Composition of DOPE
2.1.2. Antioxidant Activity of DOPE in Chemical Models
2.2. Antioxidant Activity of DOPE in Food Model Systems
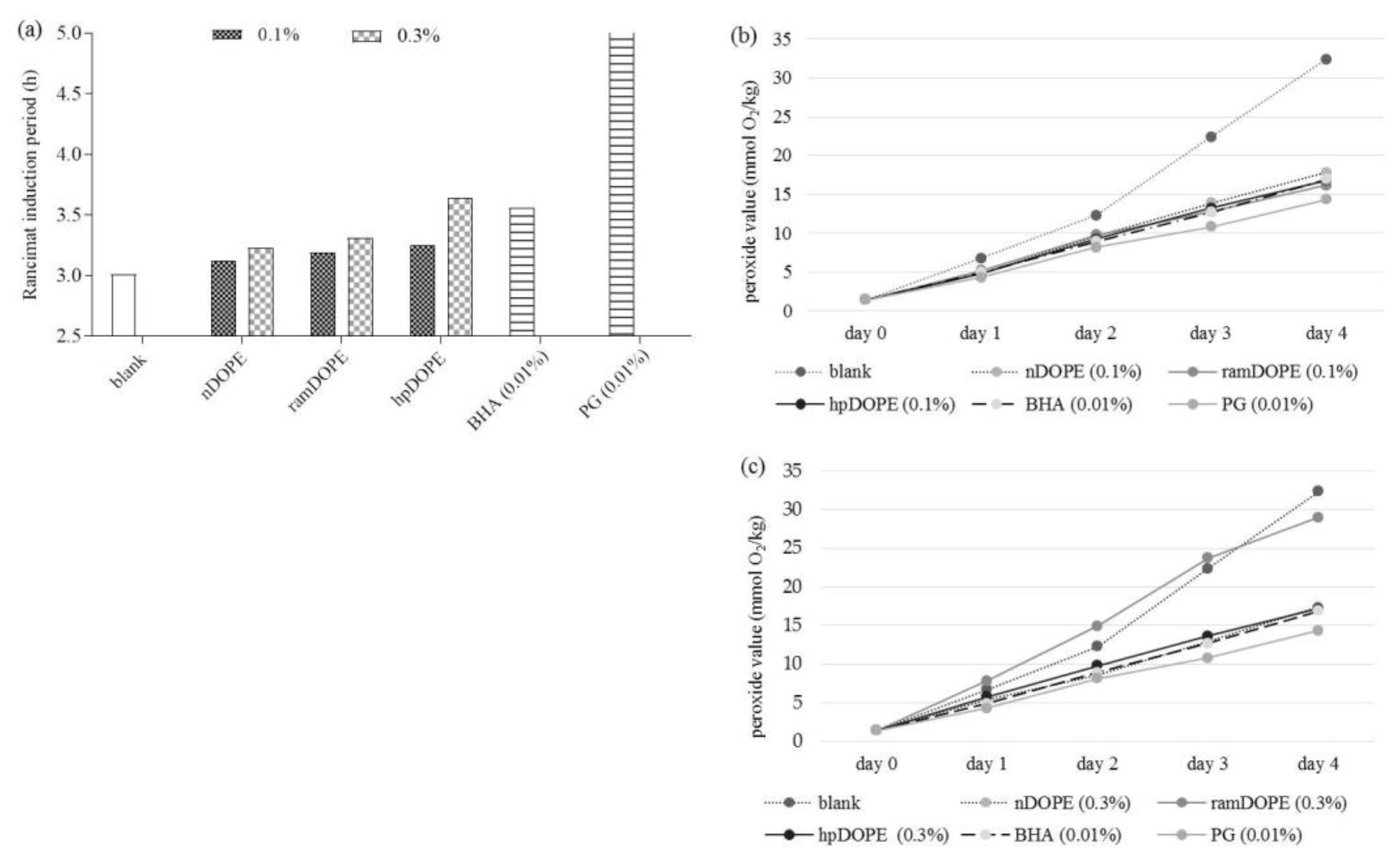
2.2.1. Antioxidant Activity of DOPE in Oil
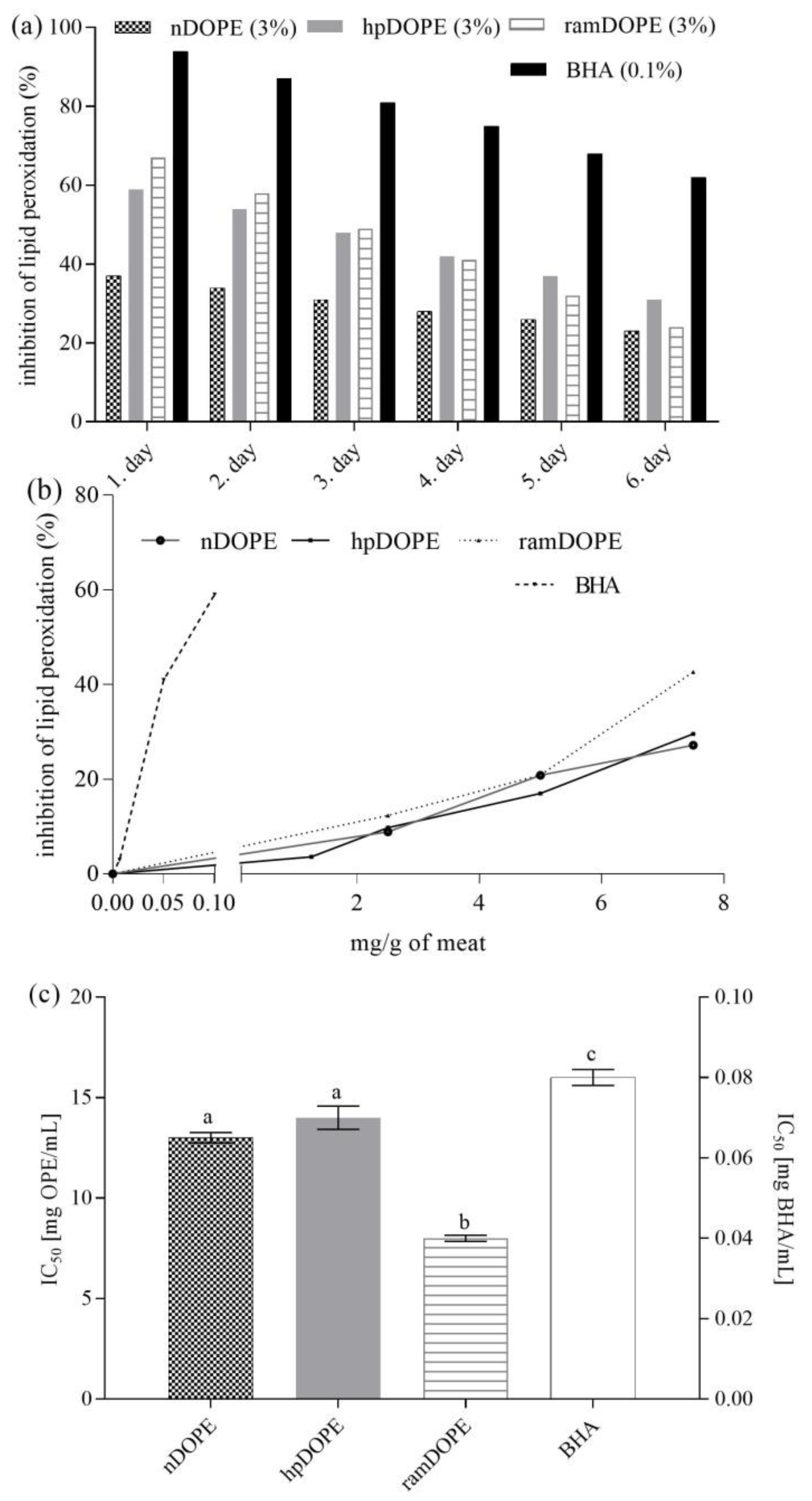
2.2.2. Antioxidant Activity of DOPE in Meat Model
2.2.3. Antioxidant Activity of DOPE in the β-Carotene Emulsion Model System
2.3. Antioxidant Activity of DOPE in Biological Model Systems
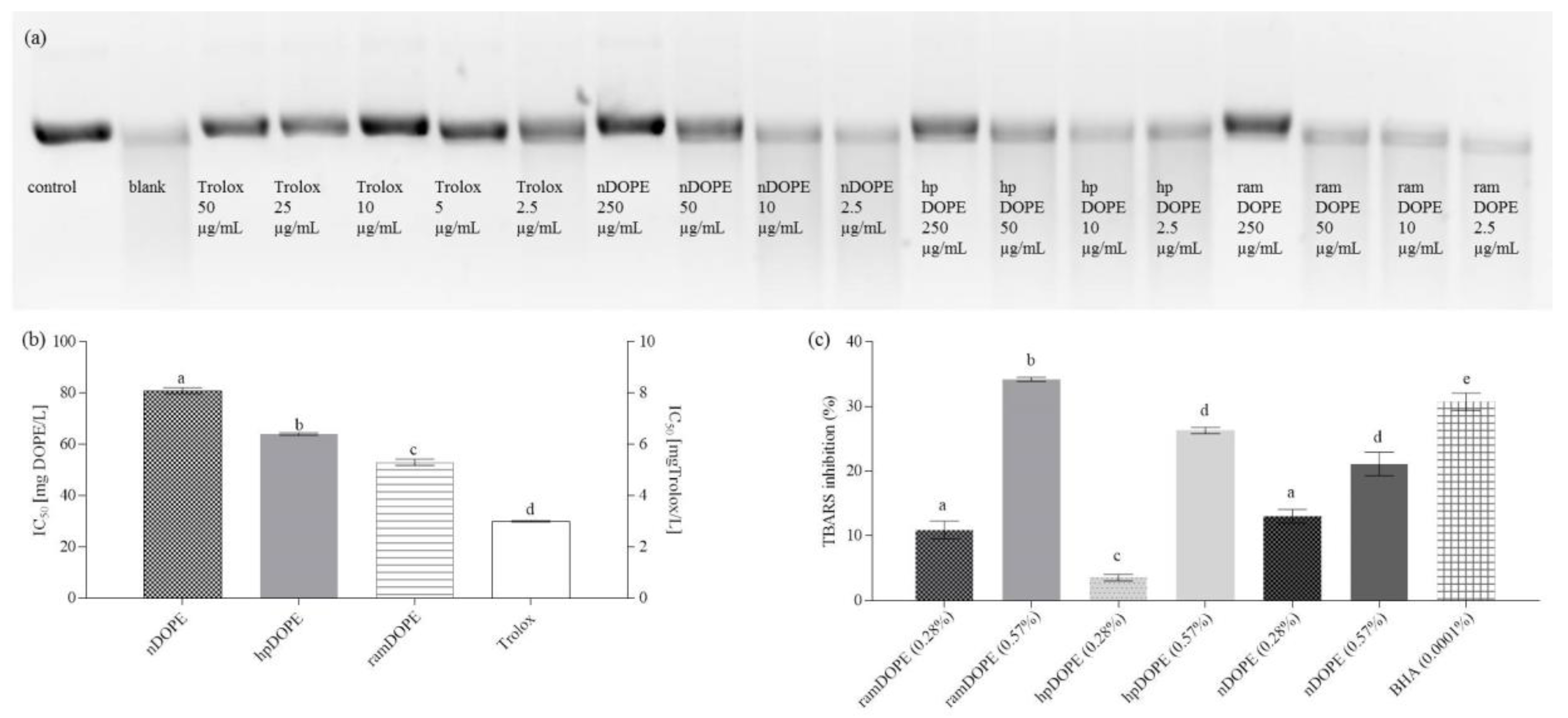
2.3.1. Inhibition of Plasmid DNA Strand Scission by DOPE
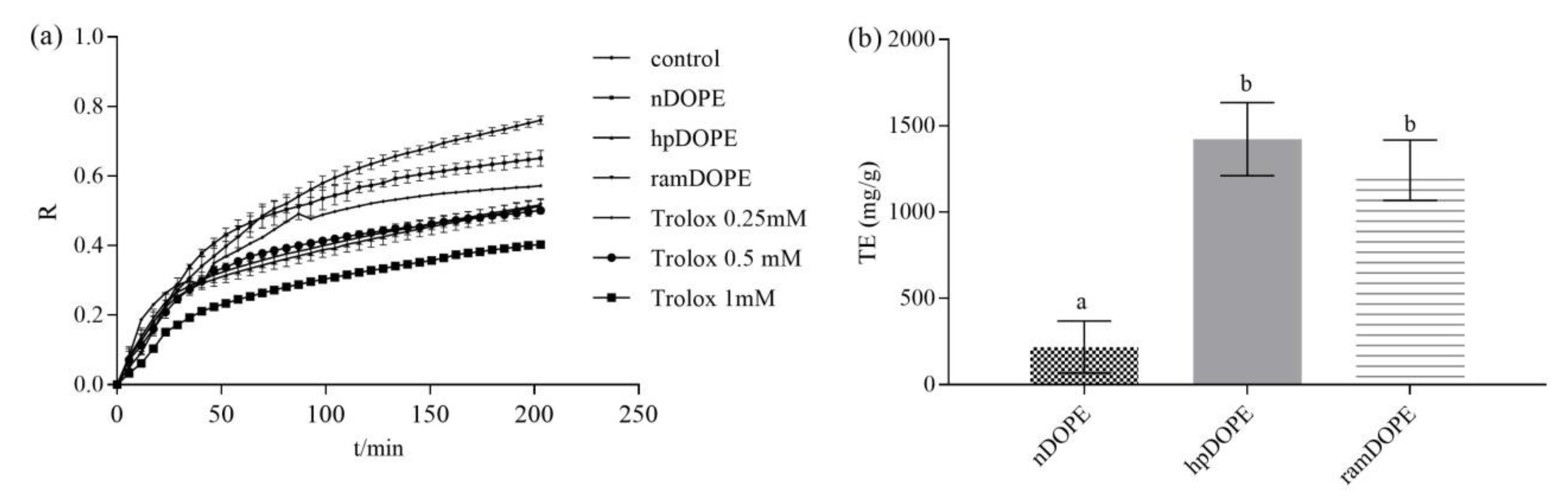
2.3.2. Antioxidant Activity of DOPE in the Liposome System
3. Materials and Methods
3.1. Chemicals and Reagents
3.2. Plant Material
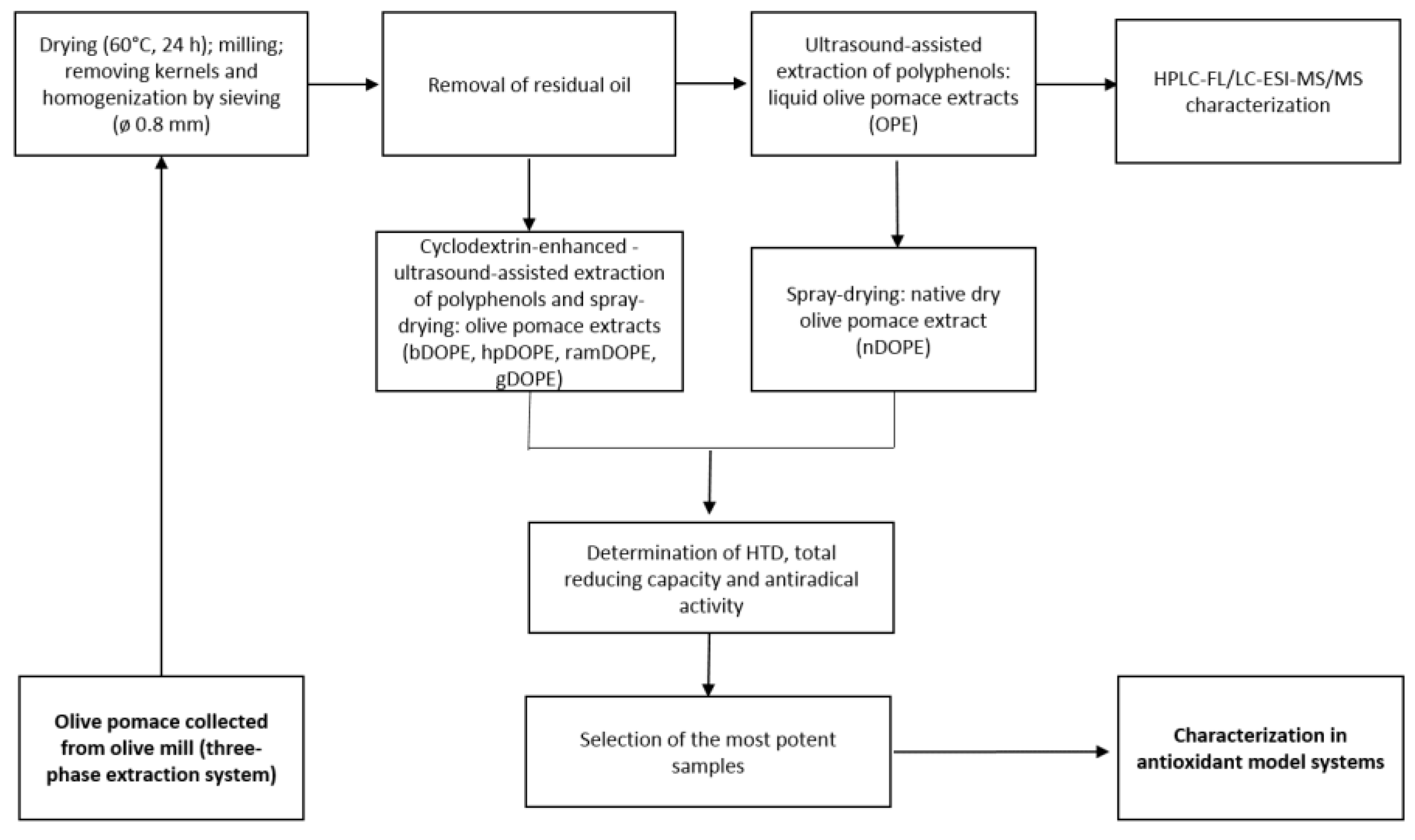
3.3. Preparation of Olive Pomace Extracts
3.4. Chromatographic Analysis of Olive Pomace Extract
3.4.1. HPLC Analysis of Hydroxytyrosol Derivatives
3.4.2. UPLC–ESI-MSMS Analysis of Phenolic Acids
3.5. Determination of Antioxidant Activity in Chemical Model Systems
3.5.1. Determination of Total Reducing Power
3.5.2. DPPH Radical-Scavenging Assay
3.5.3. Scavenging Activity against the ABTS Radical (TEAC Test)
3.5.4. Oxygen Radical Absorbance Capacity (ORAC)
3.6. Antioxidant Activity in Food Model Systems
3.6.1. Antioxidant Activity in Oil
3.6.2. Antioxidant Activity in Meat Model System
3.6.3. Antioxidant Activity in β-Carotene Emulsion Model System
3.7. Antioxidant Activity in Biological Model Systems
3.7.1. Inhibition of Plasmid DNA Strand Scission
3.7.2. Antioxidant Activity in Liposome Model System
3.8. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Clemente, A.; Sánchez-Viquore, R.; Viquore, J.; Bautista, J.; Millán, F. Chemical composition of extracted dried pomaces containing two and three phases. Food Biotechnol. 1997, 11, 273–291. [Google Scholar] [CrossRef]
- Pagnanelli, F.; Mainelli, S.; Vegliò, F.; Toro, L. Heavy metal removal by olive pomace: Biosorbent characterization and equilibrium modeling. Chem. Eng. Sci. 2003, 58, 4709–4717. [Google Scholar] [CrossRef]
- Sánchez Moral, P.; Ruiz Méndez, M.V. Production of pomace olive oil. Grasas Y Aceites 2006, 57, 47–55. [Google Scholar] [CrossRef]
- Suárez, M.; Romero, M.P.; Ramo, T.; Macià, A.; Motilva, M.J. Methods for preparing phenolic extracts from olive cake for potential application as food antioxidants. J. Agric. Food Chem. 2009, 57, 1463–1472. [Google Scholar] [CrossRef] [PubMed]
- Echeverría, F.; Ortiz, M.; Valenzuela, R.; Videla, L.A. Hydroxytyrosol and cytoprotection: A projection for clinical interventions. Int. J. Mol. Sci. 2017, 18, 930. [Google Scholar] [CrossRef] [PubMed]
- Nakiboglu, M.; Urek, O.R.; Kayali, A.H.; Tarhan, L. Antioxidant capacities of endemic Sideritis sipylea and Origanum sipyleum from Turkey. Food Chem. 2007, 104, 630–635. [Google Scholar] [CrossRef]
- Halliwell, B.; Gutteridge, J.M.C.; Cross, C.E. Free radicals, antioxidants and human disease: Where are we now? J. Lab. Clin. Med. 1992, 119, 598–620. [Google Scholar] [PubMed]
- Aruoma, O.I. Free radicals, oxidative stress and antioxidants in human health and disease. J. Am. Oil Chem. Soc. 1998, 75, 199–212. [Google Scholar] [CrossRef]
- Soto-Alarcon, S.A.; Valenzuela, R.; Valenzuela, A.; Videla, L.A. Liver protective effects of extra virgin olive oil: Interaction between its chemical composition and the cell-signaling pathways involved in protection. Endocr. Metab. Immune Disord. Drug Targets 2018, 18, 75–84. [Google Scholar] [PubMed]
- Grice, H.C. Safety evaluation of butylated hydroxytoluene (BHT) in the liver, lung and gastrointestinal tract. Food Chem. Toxicol. 1986, 24, 1127–1130. [Google Scholar]
- Wichi, H.P. Enhanced tumor development by butylated hydroxyanisole (BHA) from the perspective of effect on forestomach and oesophageal squamous epithelium. Food Chem. Toxicol. 1988, 26, 717–723. [Google Scholar]
- Huang, D.; Ou, B.; Prior, R.L. The chemistry behind antioxidant capacity assays. J. Agric. Food Chem. 2005, 53, 1841–1856. [Google Scholar] [CrossRef] [PubMed]
- Decker, E.A.; Warner, K.; Richards, M.P.; Shahidi, F. Measuring antioxidant effectiveness in food. J. Agric. Food Chem. 2005, 53, 4303–4310. [Google Scholar] [CrossRef] [PubMed]
- Sansone, F.; Mencherini, T.; Picerno, P.; d’Amore, M.; Aquino, R.P.; Lauro, M.R. Maltodextrin/pectin microparticles by spray drying as carrier for nutraceutical extracts. J. Food Eng. 2011, 105, 468–476. [Google Scholar] [CrossRef]
- Albahari, P.; Jug, M.; Radić, K.; Jurmanović, S.; Brnčić, M.; Rimac Brnčić, S.; Vitali Čepo, D. Characterization of olive pomace extract obtained by cyclodextrin-enhanced pulsed ultrasound assisted extraction. LWT-Food Sci. Tech. 2018, 92, 22–31. [Google Scholar] [CrossRef]
- Braithwaite, M.C.; Tyagi, C.; Tomar, L.K.; Kumar, P.; Chiinara, Y.E.; Pillay, V. Nutraceutical-based therapeutic and formulation strategies augmenting their efficiency to complement modern medicine: An overview. J. Funct. Food. 2014, 6, 82–99. [Google Scholar] [CrossRef]
- Aliakbarian, B.; Casazza, A.A.; Perego, P. Valorization of olive oil solid waste using high pressure–high temperature reactor. Food Chem. 2011, 128, 704–710. [Google Scholar] [CrossRef]
- Cioffi, G.; Pesca, M.S.; De Caprariis, P.; Braca, A.; Severino, L.; De Tommasi, N. Phenolic compounds in olive oil and olive pomace from Cilento (Campania, Italy) and their antioxidant activity. Food Chem. 2010, 121, 105–111. [Google Scholar] [CrossRef]
- Herrero, M.; Temirzoda, T.N.; Segura-Carretero, A.; Quirantes, R.; Plaza, M.; Ibañez, E. New possibilities for the valorization of olive oil by-products. J. Chromatogr. A 2011, 1218, 7511–7520. [Google Scholar] [CrossRef] [PubMed]
- Ryan, D.; Robards, K.; Lavee, S. Changes in phenolic content of olive during maturation. Int. J. Food Sci. Tech. 1999, 34, 265–274. [Google Scholar] [CrossRef]
- Cimato, A.; Mattei, A.; Osti, M. Variation of polyphenol composition with harvesting period. Acta Hortic. 1990, 286, 453–456. [Google Scholar] [CrossRef]
- Efmorfopoulou, E.; Rodis, P. Complexation of oleuropein and trans-cinnamic acid with cyclodextrins. Chem. Nat. Compd. 2004, 40, 362–366. [Google Scholar] [CrossRef]
- Efmorfopoulou, E.; Rodis, P. Application of cyclodextrins to the extraction of antioxidant compounds from olive oil. Chem. Nat. Compd. 2005, 41, 22–26. [Google Scholar] [CrossRef]
- García-Padial, M.; Martínez-Ohárriz, M.C.; Navarro-Blasco, I.; Zornoz, A. The role of cyclodextrins in ORAC-fluorescence assays. Antioxidant capacity of tyrosol and caffeic acid with hydroxypropyl-β-cyclodextrin. J. Agric. Food Chem. 2013, 61, 260–264. [Google Scholar] [CrossRef] [PubMed]
- Mourtzinos, I.; Makris, D.P.; Yannakopoulou, K.; Kalogeropoulos, N.; Michali, I.; Karathanos, V.T. Thermal stability of anthocyanin extract of Hibiscus sabdariffa L. in the Presence of beta-Cyclodextrin. J. Agric. Food Chem. 2008, 56, 10303–10310. [Google Scholar] [CrossRef] [PubMed]
- Mercader-Ros, M.T.; Lucas-Abellán, C.; Fortea, M.I.; Gabaldón, J.A.; Núñez-Delicado, E. Effect of HP-β-cyclodextrins complexation on the antioxidant activity of flavonols. Food Chem. 2010, 118, 769–773. [Google Scholar] [CrossRef]
- Prior, R.L.; Wu, X.; Schaich, K. Standardized Methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. J. Agric. Food Chem. 2005, 53, 4290–4302. [Google Scholar] [CrossRef] [PubMed]
- Terpinc, P.; Bezjak, M.; Abramovič, H. A kinetic model for evaluation of the antioxidant activity of several rosemary extracts. Food Chem. 2009, 115, 740–744. [Google Scholar] [CrossRef]
- Amorati, R.; Valgimigli, L. Advantages and limitations of common testing methods for antioxidants. Free Radical Res. 2015, 49, 633–649. [Google Scholar] [CrossRef] [PubMed]
- Pincemail, J.; Tabart, J.; Defraigne, J.O.; Dommes, J.; Kevers, C. The total antioxidant capacity of foods: A reappraisal. application to commercial orange juices. J. Anti. Act. 2015, 1, 1–7. [Google Scholar] [CrossRef]
- Barão, C.E.; Paiva-Martins, F.; Zanin, G.M.; Moraes, F.F.D. Determination of the inclusion complex constant between oleuropein and cyclodextrins by complexation theory. J. Incl. Phenom. Macrocycl. Chem. 2014, 78, 465–470. [Google Scholar] [CrossRef]
- Shahidi, F.; Zhong, Y. Lipid Oxidation and improving the oxidative stability. Chem. Soc. Rev. 2010, 39, 4067–4079. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, A.; Jafari, S.M.; Esfanjani, A.F.; Akhavan, S. Application of nano-encapsulated olive leaf extract in controlling the oxidative stability of soybean oil. Food Chem. 2016, 190, 513–519. [Google Scholar] [CrossRef] [PubMed]
- López-Nicolás, J.M.; Rodríguez-Bonilla, P.; García-Carmona, F. Cyclodextrins and antioxidants. Crit. Rev. Food Sci. Nutr. 2014, 54, 251–276. [Google Scholar] [CrossRef] [PubMed]
- Fasseas, M.K.; Mountzouris, K.C.; Tarantilis, P.A.; Polissiou, M.; Zervas, G. Antioxidant activity in meat treated with oregano and sage essential oils. Food Chem. 2008, 106, 1188–1194. [Google Scholar] [CrossRef]
- Tanabe, H.; Yoshida, M.; Tomita, N. Comparison of the antioxidant activities of 22 commonly used culinary herbs and spices on the lipid oxidation of pork meat. J. Anim. Sci. 2002, 73, 389–393. [Google Scholar] [CrossRef]
- Prieto, M.A.; Rodríguez-Amado, I.; Vázquez, J.A.; Murado, M.A. β-Carotene assay revisited. Application to characterize and quantify antioxidant and prooxidant activities in a microplate. J. Agric. Food Chem. 2012, 60, 8983–8993. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Y.; Shahidi, F. Methods for the assessment of antioxidant activity in foods. In Woodhead Publishing Series in Food Science, Technology and Nutrition, Handbook of Antioxidants for Food Preservation, 1st ed.; Shahidi, F., Woodhead, Eds.; Publisher: Sawston, Cambridge, UK, 2015; pp. 287–333. ISBN 9781782420897. [Google Scholar]
- Singh, R.P.; Chidambara Murthy, K.N.; Jayaprakasha, G.K. Studies on the antioxidant activity of pomegranate (Punica granatum) peel and seed extracts using in vitro models. J. Agric. Food Chem. 2002, 50, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Zengin, G.; Cakmak, Y.S.; Guler, G.O.; Aktumsek, A. In vitro antioxidant capacities and fatty acid compositions of three Centaurea species collected from Central Anatolia region of Turkey. Food Chem. Toxicol. 2010, 48, 2638–2641. [Google Scholar] [CrossRef] [PubMed]
- Dizdaroglu, M.; Jaruga, P.; Birincioglu, M.; Rodriguez, H. Free radical-induced damage to DNA: Mechanisms and measurement. Free Radical Biol. Med. 2002, 32, 1102–1115. [Google Scholar] [CrossRef]
- Chatti, I.B.; Boubaker, J.; Skandrani, I.; Bhouri, W.; Ghedira, K.; Ghedira, L.C. Antioxidant and antigenotoxic activities in Acacia salicina extracts and its protective role against DNA strand scission induced by hydroxyl radical. Food Chem. Toxicol. 2011, 49, 1753–1758. [Google Scholar] [CrossRef] [PubMed]
- Suksomtip, M.; Pongsamart, S. Protective effect against oxidation of human low-density lipoprotein and plasmid DNA strand scission of Tamarind seed coat extract in vitro. LW-Food Sci Technol. 2008, 41, 2002–2007. [Google Scholar] [CrossRef]
- Yuan, J.J.; Qin, F.G.F.; Tu, J.L.; Li, B. Preparation, characterization, and antioxidant activity evaluation of liposomes containing water-soluble hydroxytyrosol from olive. Molecules 2017, 22, 870. [Google Scholar] [CrossRef] [PubMed]
- Knobloch, J.; Suhendro, D.K.; Zieleniecki, J.L.; Shapter, J.G.; Köper, I. Membrane–drug interactions studied using model membrane systems. Saudi J. Biol. Sci. 2015, 22, 714–718. [Google Scholar] [CrossRef] [PubMed]
- Balducci, V.; Incerpi, S.; Stano, P.; Tofani, D. Antioxidant activity of hydroxytyrosyl esters studied in liposome models. BBA-Biomembr. 1860, 1860, 600–610. [Google Scholar] [CrossRef] [PubMed]
- Generalić Mekinić, I.; Gotovac, M.; Skroza, D.; Ljubenkov, I.; Burčul, F.; Katalinić, V. Effect of the extraction solvent on the oleuropein content and antioxidant properties of olive leaf (cv. Oblica, Lastovka and Levantinka) extracts. Cro. J. Food Sci. Tech. 2014, 6, 7–14. [Google Scholar]
- Kong, B.; Zhang, H.; Xiong, Y.L. Antioxidant activity of spice extracts in a liposome system and in cooked pork patties and the possible mode of action. Meat. Sci. 2010, 85, 772–778. [Google Scholar] [CrossRef] [PubMed]
- Tsarbopoulos, E.; Gikas, N.; Papadopoulos, N.; Aligiannis, N.; Kafatos, A. Simultaneous determination of oleuropein and its metabolites in plasma by high-performance liquid chromatography. J. Chromatogr. B 2003, 785, 157–164. [Google Scholar] [CrossRef]
- López de las Hazas, M.C.; Piñol, C.; Macià, A.; Romero, M.P.; Pedret, A.; Solà, R.; Rubió, L.; Motilva, M.J. Differential absorption and metabolism of hydroxytyrosol and its precursors oleuropein and secoiridoids. J. Funct. Food 2016, 22, 52–63. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Viticult. 1965, 16, 144–158. [Google Scholar]
- Kirigaya, N.; Kato, H.; Fujimaki, M. Studies on antioxidant activity of nonenzymatic browning reaction products. Part III: Fractionation of browning reaction solution between ammonia and d-glucose and antioxidant activity of the resulting fractions. Nippon Nogei. Kagaku Kaishi. 1971, 45, 292–298. [Google Scholar] [CrossRef]
- Shimada, K.; Fujikawa, K.; Yahara, K.; Nakamura, T. Antioxidative properties of xanthan on the antioxidation of soybean oil in cyclodextrin emulsion. J. Agric. Food Chem. 1992, 40, 945–948. [Google Scholar] [CrossRef]
- Chanda, S.; Dave, R. In vitro models for antioxidant activity evaluation and some medicinal plants possessing antioxidant properties: An overview. Afr. J. Microbiol. Res. 2009, 3, 981–996. [Google Scholar]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Rad. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Ou, B.; Huang, D.; Hampsch-Woodill, M.; Flanagan, J.A.; Deemer, E.K. Analysis of Antioxidant Activities of common vegetables employing oxygen radical absorbance capacity (ORAC) and ferric reducing antioxidant power (FRAP) assays: A comparative study. J. Agric. Food Chem. 2002, 50, 3122–3128. [Google Scholar] [CrossRef] [PubMed]
- Animal and vegetable fats and oils—Determination of oxidative stability (accelerated oxidation test). Available online: https://www.iso.org/standard/69594.html (accessed on 28 July 2018).
- Matthäus, B.W. Determination of the oxidative stability of vegetable oils by rancimat and conductivity and chemiluminescence measurements. J. Am. Oil Chem. Soc. 1996, 73, 1039–1043. [Google Scholar] [CrossRef]
- Pardun, H.; Kroll, E. Bestimmung der oxydationsstabilität von Ölen und fetten mit hilfe einer automatischen version des swift-testes. Fette Seifen Anstrichmittel Die Ernährungsindustrie 1972, 74, 366–372. [Google Scholar] [CrossRef]
- Kitts, D.D.; Wijewickreme, A.N.; Hu, C. Antioxidant properties of a North American ginseng extract. Mol. Cell Biochem. 2000, 203, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Cao, Y.; Zhao, M. Extraction optimization, purification and antioxidant activity of procyanidins from hawthorn (C. pinnatifida Bge. var. major) fruits. Food Chem. 2010, 119, 1656–1662. [Google Scholar] [CrossRef]
Sample Availability: Not available. |
| Hydroxytyrosol Derivatives (mg/100 g) | Phenolic Acids (mg/100 g) | ||
|---|---|---|---|
| Hydoxytyrosol | 51.26 ± 0.40 A | 3,4-dihydroxybenzoic acid | 3.72 ± 0.47 A |
| Tyrosol | 88.67 ± 1.17 B | vanillic acid | 2.16 ± 0.13 B |
| Oleuropein | 16.29 ± 0.39 C | homovanillic acid | 1.25 ± 0.12 C |
| p-hydroxybenzoic acid | 0.33 ± 0.058 D | ||
| nDOPE | hpDOPE | ramDOPE | bDOPE | gDOPE | |
|---|---|---|---|---|---|
| Hydroxytyrosol (mg/kg) | 513.1 ± 4.77 A | 716.8 ± 16.97 B | 663.6 ± 39.03 B | 497.4 ± 5.67 A | 527.1 ± 12.87 A |
| Tyrosol (mg/kg) | 870.1 ± 35.07 A | 715.7 ± 19.92 B | 950.9 ± 7.61 C | 804.9 ± 0.33 AC | 777.7 ± 7.65 BC |
| Oleuropein (mg/kg) | 166.0 ± 7.45 A | 249.4 ± 5.93 B | 157.6 ± 12.96 A | 171.4 ± 1.00 A | 226.8 ± 10.19 B |
| HTD (mg/100 g) | 1549.2 ± 22.9 A | 1682 ± 8.9 B | 1772.1 ± 44.4 B | 1473 ± 4.3 A | 1531 ± 30.7 A |
| TRC (mg/g GAE) | 39.24 ± 0.042 A | 50.97 ± 0.516 B | 53.16 ± 0.164 C | 44.21 ± 0.878 D | 42.71 ± 0.45 D |
| IC50 against DPPH (g/L) | 1.65 ± 0.129 A | 1.039 ± 0.129 B | 1.032 ± 0.094 B | 1.175 ± 0.007 B | 1.429 ± 0.083 A |
| TEAC (mg/g TE) | 12.86 ± 0.419 A | 15.44 ± 0.133 B | 17.07 ± 0.51 C | 14.00 ± 0.33 A | 12.71 ± 0.24 A |
| ORAC (mg/kg TE) | 152.8 ± 5.06 AB | 166.5 ± 7.51 A | 165.8 ± 6.34 A | 142.7 ± 10.21 B | 162.8 ± 10.24 A |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vitali Čepo, D.; Radić, K.; Jurmanović, S.; Jug, M.; Grdić Rajković, M.; Pedisić, S.; Moslavac, T.; Albahari, P. Valorization of Olive Pomace-Based Nutraceuticals as Antioxidants in Chemical, Food, and Biological Models. Molecules 2018, 23, 2070. https://doi.org/10.3390/molecules23082070
Vitali Čepo D, Radić K, Jurmanović S, Jug M, Grdić Rajković M, Pedisić S, Moslavac T, Albahari P. Valorization of Olive Pomace-Based Nutraceuticals as Antioxidants in Chemical, Food, and Biological Models. Molecules. 2018; 23(8):2070. https://doi.org/10.3390/molecules23082070
Chicago/Turabian StyleVitali Čepo, Dubravka, Kristina Radić, Sanja Jurmanović, Mario Jug, Marija Grdić Rajković, Sandra Pedisić, Tihomir Moslavac, and Petra Albahari. 2018. "Valorization of Olive Pomace-Based Nutraceuticals as Antioxidants in Chemical, Food, and Biological Models" Molecules 23, no. 8: 2070. https://doi.org/10.3390/molecules23082070
APA StyleVitali Čepo, D., Radić, K., Jurmanović, S., Jug, M., Grdić Rajković, M., Pedisić, S., Moslavac, T., & Albahari, P. (2018). Valorization of Olive Pomace-Based Nutraceuticals as Antioxidants in Chemical, Food, and Biological Models. Molecules, 23(8), 2070. https://doi.org/10.3390/molecules23082070