Berberine Inhibits Human Melanoma A375.S2 Cell Migration and Invasion via Affecting the FAK, uPA, and NF-κB Signaling Pathways and Inhibits PLX4032 Resistant A375.S2 Cell Migration In Vitro
Abstract
:1. Introduction
2. Results
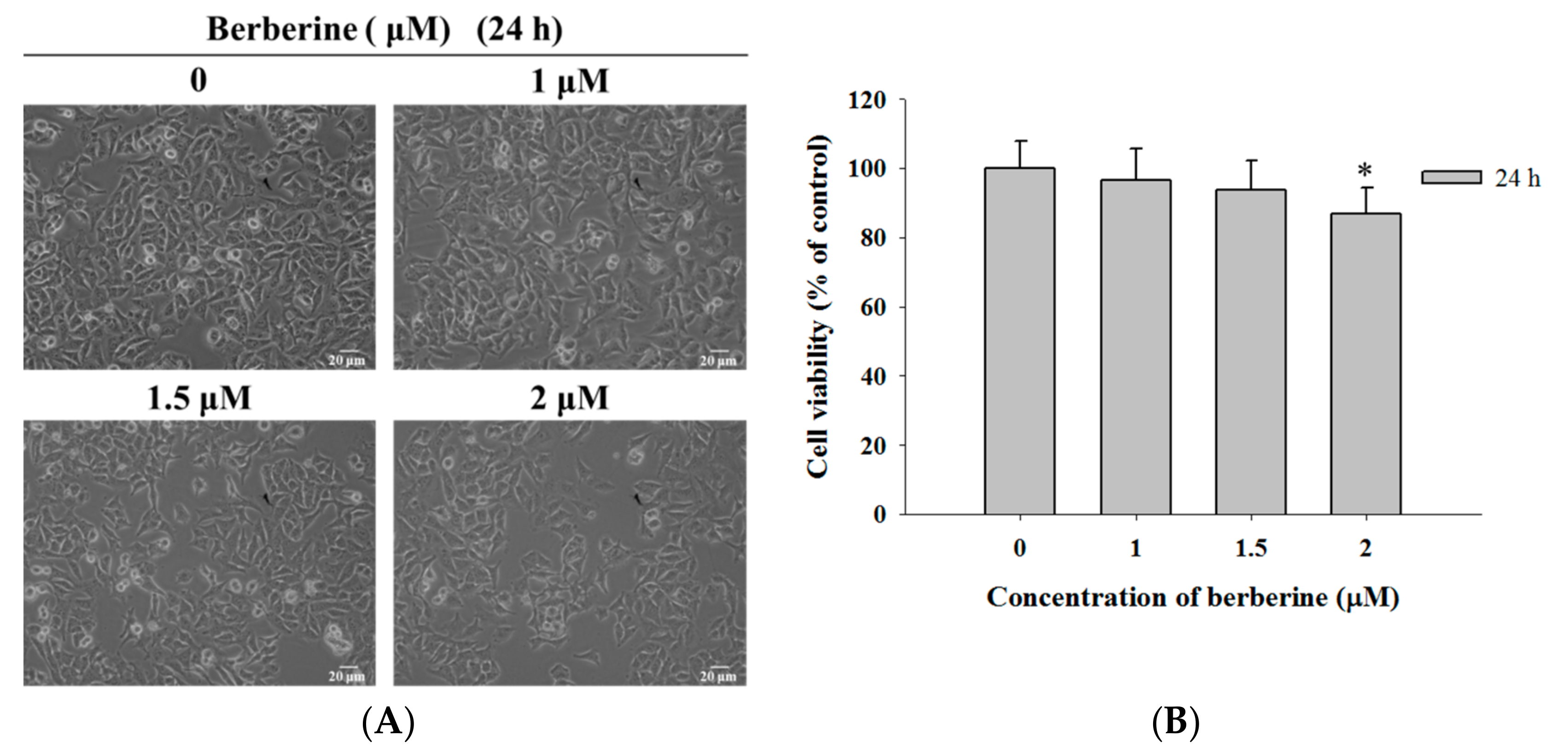
2.1. Berberine Induces Cell Morphological Changes and Decreases the Total Viability of A375.S2 Cells
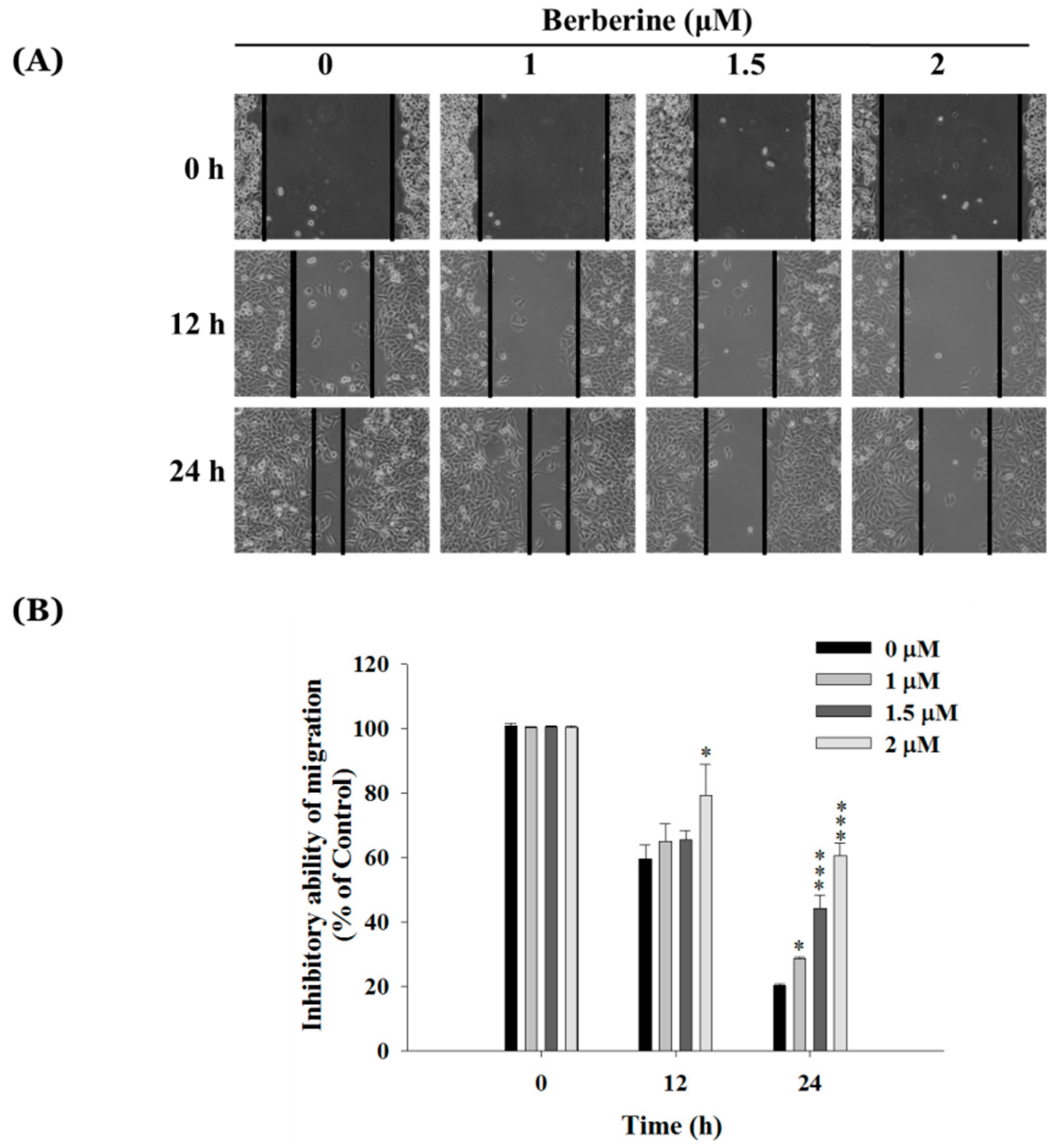
2.2. Berberine Inhibits Cell Mobility in A375.S2 Cells
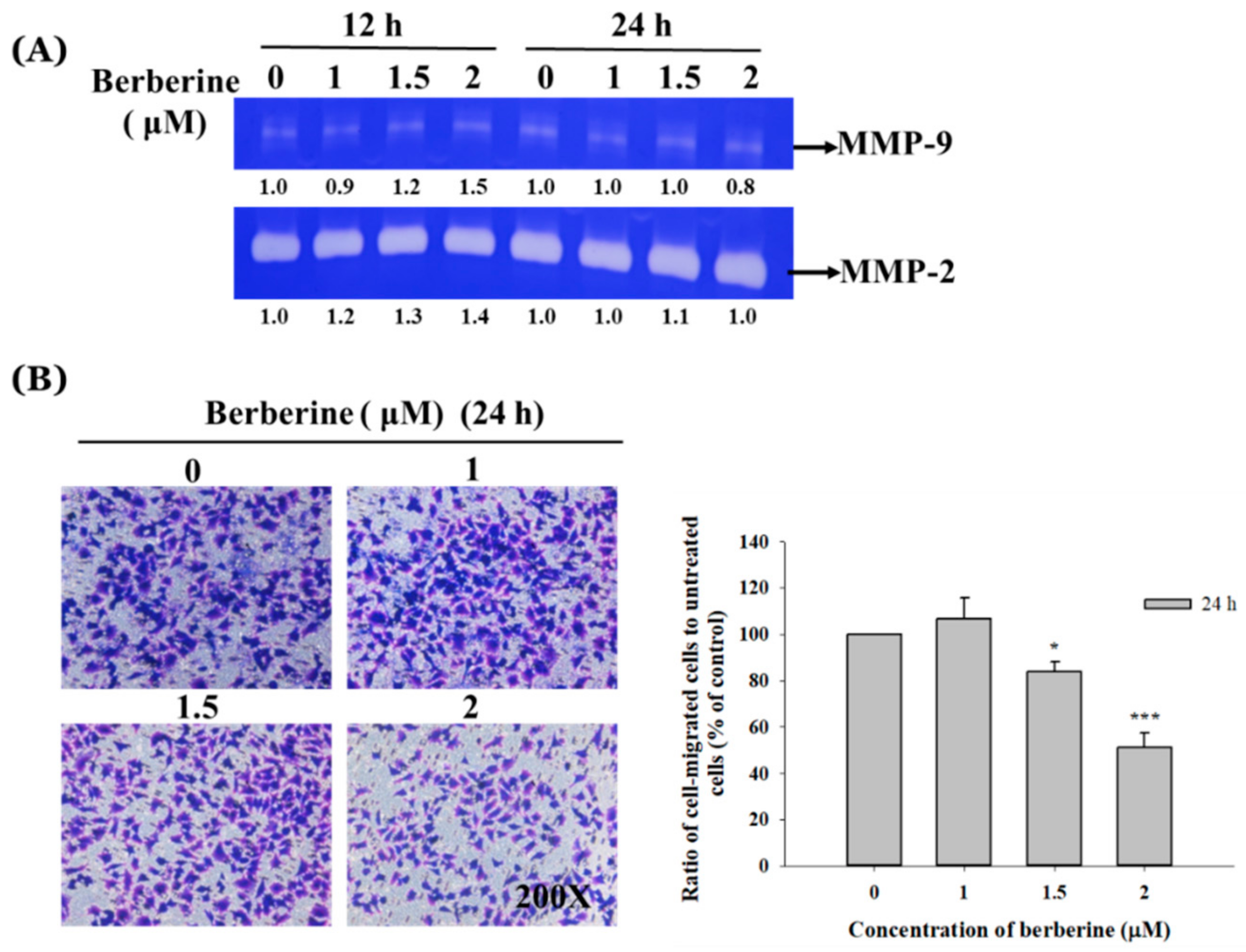
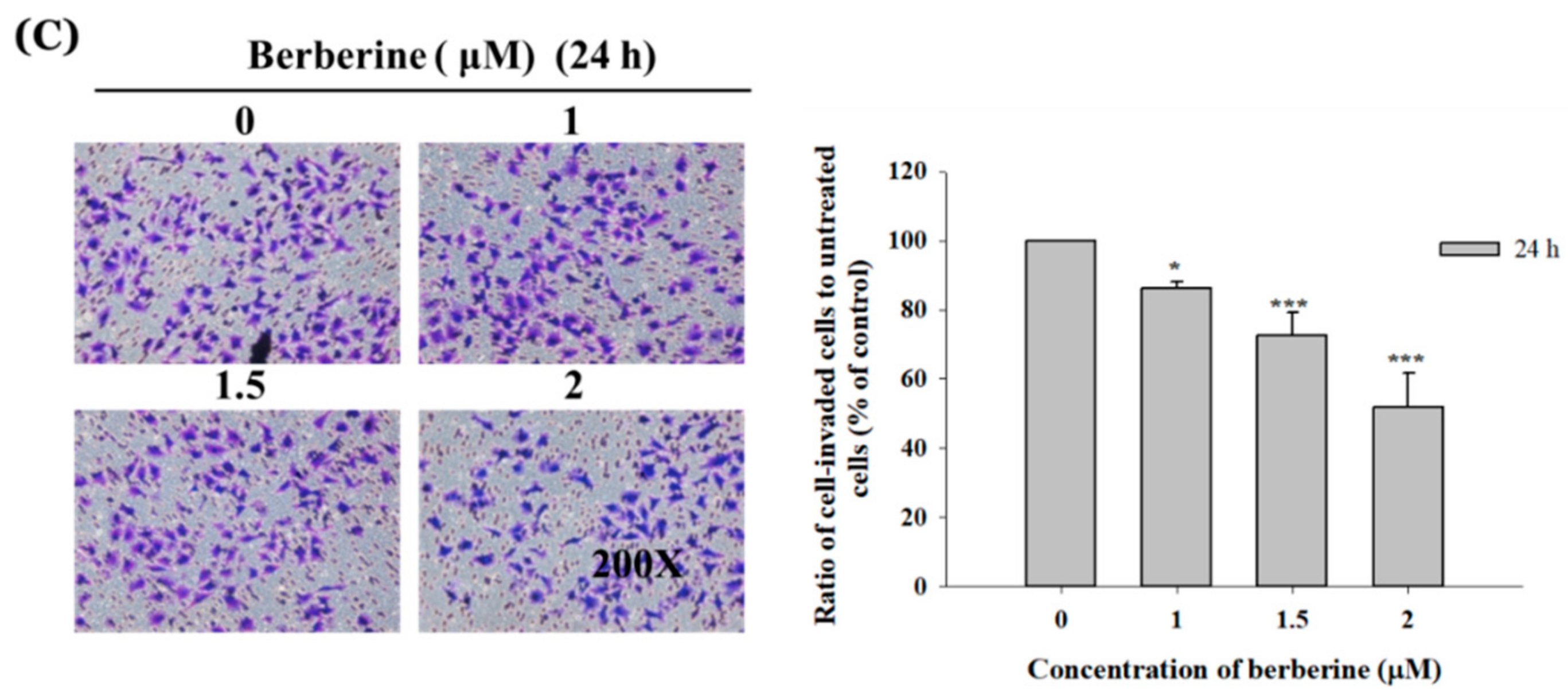
2.3. Berberine Affects the Matrix Metalloproteinase Activity and Cell Migration and Invasion in A375.S2 Cells
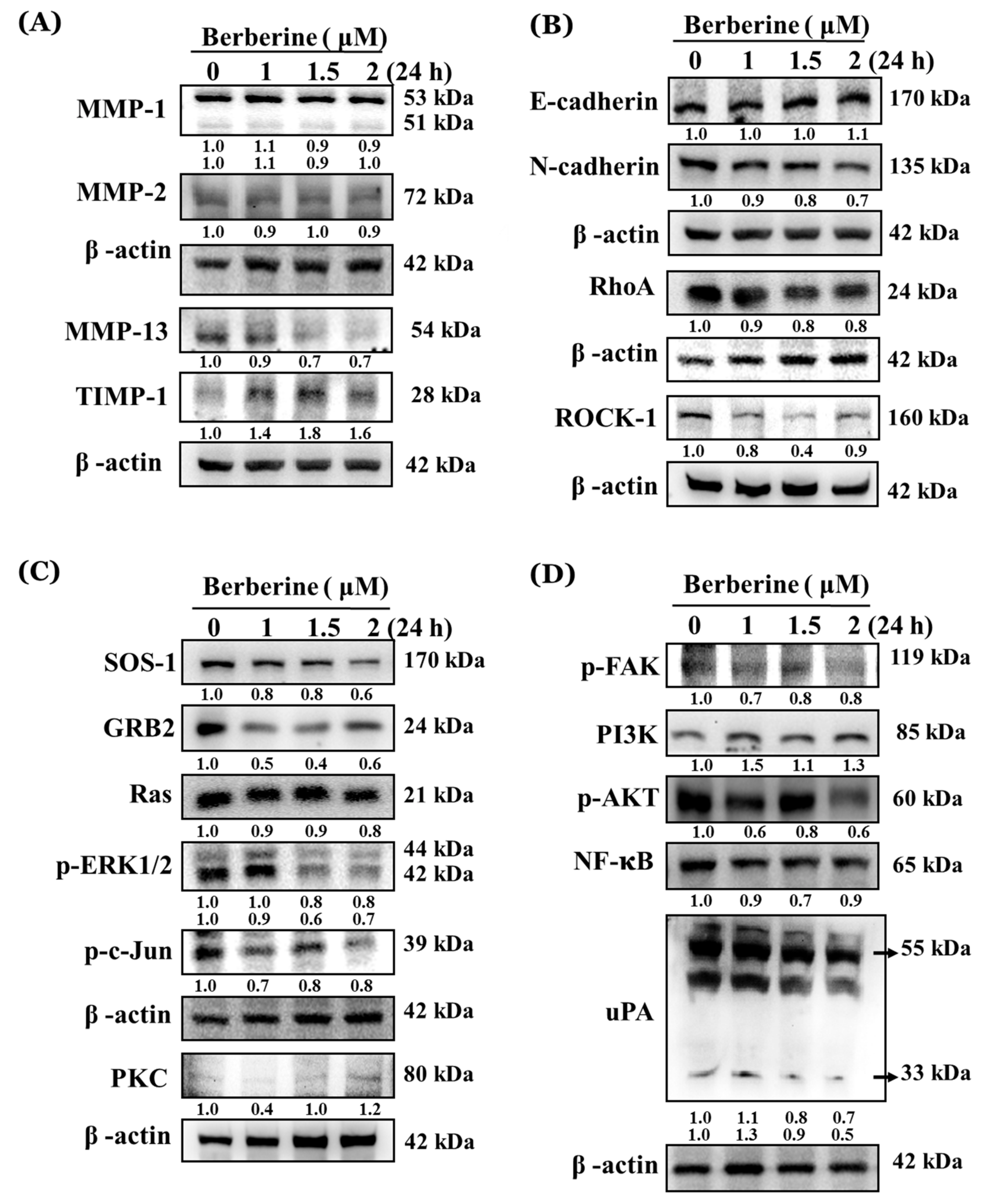
2.4. Berberine Affects Key Metastasis-Related Proteins in A375.S2 Cells
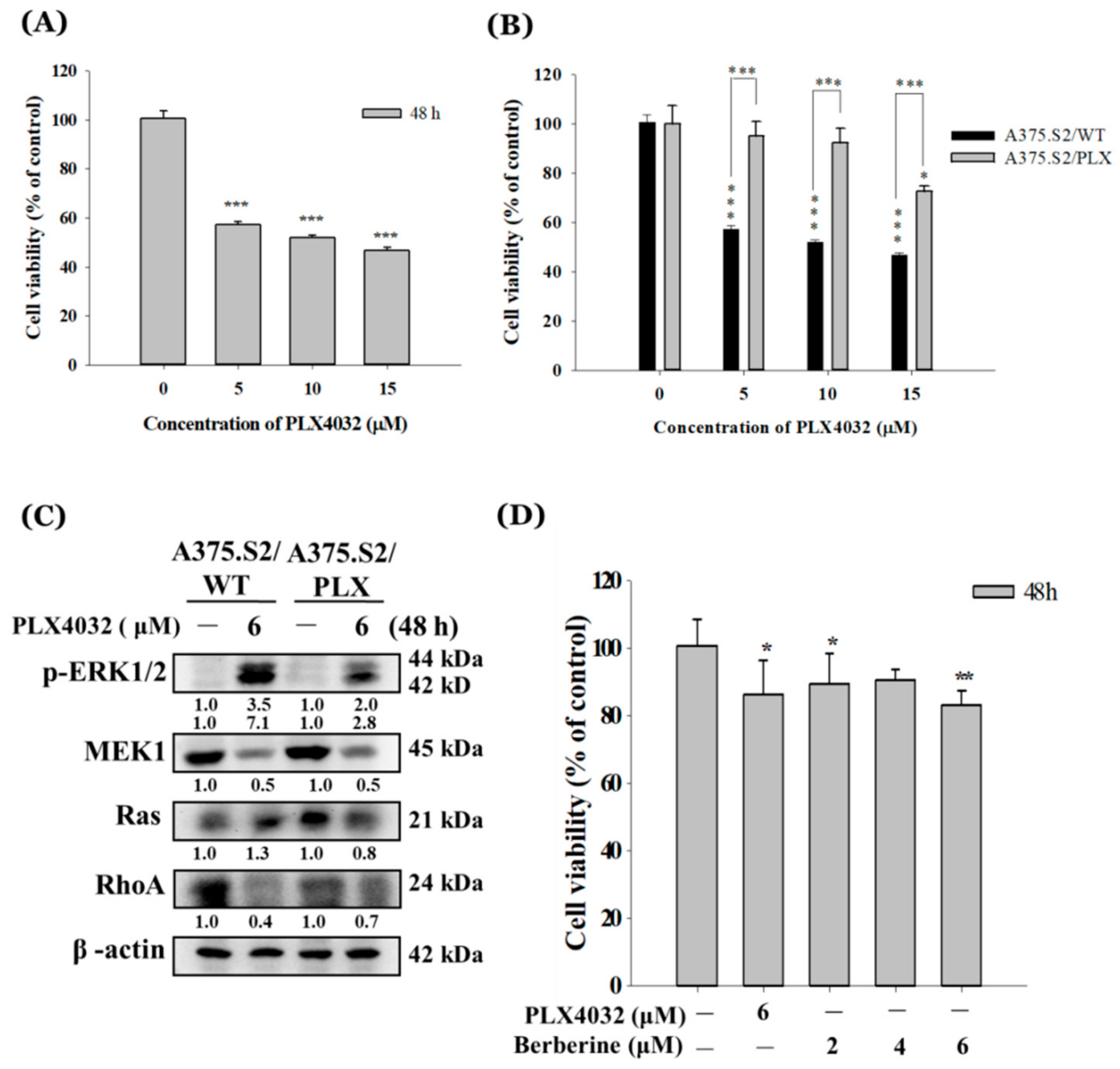
2.5. Berberine Decreases Cell Viability of A375.S2/PLX Resistant Cells
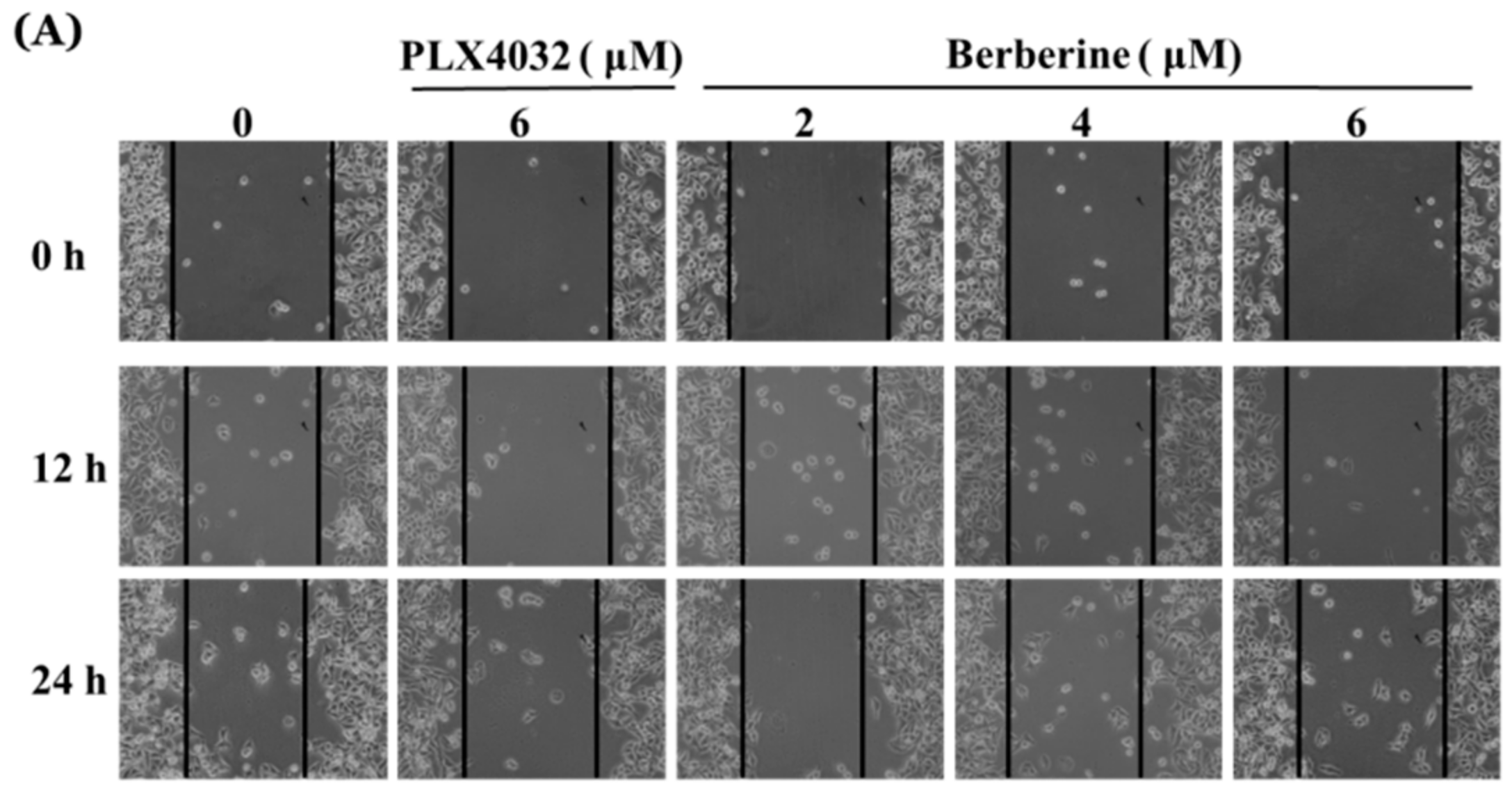
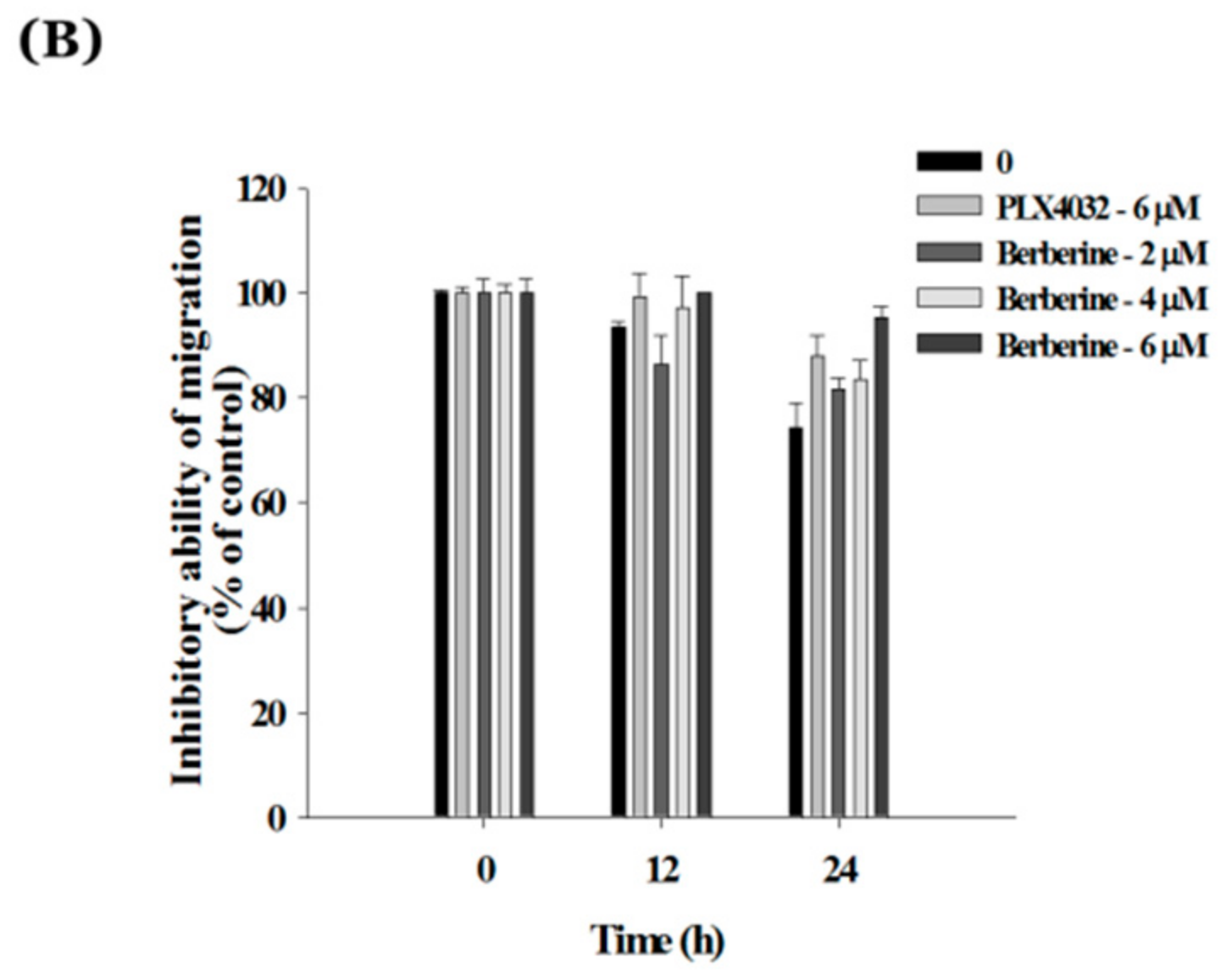
2.6. Berberine Suppresses Cell Mobility in A375.S2/PLX Resistant Cells
3. Discussion
4. Materials and Methods
4.1. Test Chemicals, Reagents and Culture Medium
4.2. Cell Line and Culture
4.3. Cell Morphological Examination and Viability Assay
4.4. In-Vitro Scratch Wound Healing Assay
4.5. Gelatin Zymography Assay
4.6. Transwell Assay for Cell Migration and Invasion Examinations
4.7. Western Blotting Analysis for Cell Metastasis-Associated Protein Expressions
4.8. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Leiter, U.; Eigentler, T.; Garbe, C. Epidemiology of skin cancer. Adv. Exp. Med. Biol. 2014, 810, 120–140. [Google Scholar] [PubMed]
- Linos, E.; Swetter, S.M.; Cockburn, M.G.; Colditz, G.A.; Clarke, C.A. Increasing burden of melanoma in the United States. J. Investig. Dermatol. 2009, 129, 1666–1674. [Google Scholar] [CrossRef] [PubMed]
- American Cancer Society. Available online: http://www.cancer.org/cancer/skincancer-melanoma/detailedguide/melanoma-skin-cancer-key-statistics (accessed on 3 June 2016).
- Rastrelli, M.; Tropea, S.; Rossi, C.R.; Alaibac, M. Melanoma: Epidemiology, risk factors, pathogenesis, diagnosis and classification. In Vivo 2014, 28, 1005–1011. [Google Scholar] [PubMed]
- Chen, S.T.; Geller, A.C.; Tsao, H. Update on the Epidemiology of Melanoma. Curr. Dermatol. Rep. 2013, 2, 24–34. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2015. CA A Cancer J. Clin. 2015, 65, 5–29. [Google Scholar] [CrossRef] [PubMed]
- Hamm, C.; Verma, S.; Petrella, T.; Bak, K.; Charette, M. Biochemotherapy for the treatment of metastatic malignant melanoma: A systematic review. Cancer Treat. Rev. 2008, 34, 145–156. [Google Scholar] [CrossRef] [PubMed]
- Martin, T.A.; Ye, L.; Sanders, A.J.; Lane, J.; Jiang, W.G. Cancer invasion and metastasis: Molecular and cellular perspective. In Metastatic Cancer Clinical Biological Perspectives; Landes Bioscience: Austin, TX, USA, 2013; pp. 135–168. [Google Scholar]
- Liu, P.L.; Tsai, J.R.; Charles, A.L.; Hwang, J.J.; Chou, S.H.; Ping, Y.H.; Lin, F.Y.; Chen, Y.L.; Hung, C.Y.; Chen, W.C.; et al. Resveratrol inhibits human lung adenocarcinoma cell metastasis by suppressing heme oxygenase 1-mediated nuclear factor-kappaB pathway and subsequently downregulating expression of matrix metalloproteinases. Mol. Nutr. Food Res. 2010, 54 (Suppl. 2), S196–S204. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, Y.; Sawada, S.; Yoshioka, I.; Ohashi, Y.; Matsuo, M.; Harimaya, Y.; Tsukada, K.; Saiki, I. Increased surgical stress promotes tumor metastasis. Surgery 2003, 133, 547–555. [Google Scholar] [CrossRef] [PubMed]
- Hainaut, P.; Plymoth, A. Targeting the hallmarks of cancer: Towards a rational approach to next-generation cancer therapy. Curr. Opin. Oncol. 2013, 25, 50–51. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, M.; Christofori, G.; Lehembre, F. Distinct mechanisms of tumor invasion and metastasis. Trends Mol. Med. 2007, 13, 535–541. [Google Scholar] [CrossRef] [PubMed]
- Folgueras, A.R.; Pendas, A.M.; Sanchez, L.M.; Lopez-Otin, C. Matrix metalloproteinases in cancer: From new functions to improved inhibition strategies. Int. J. Dev. Biol. 2004, 48, 411–424. [Google Scholar] [CrossRef] [PubMed]
- Kessenbrock, K.; Plaks, V.; Werb, Z. Matrix metalloproteinases: Regulators of the tumor microenvironment. Cell 2010, 141, 52–67. [Google Scholar] [CrossRef] [PubMed]
- Nikkola, J.; Vihinen, P.; Vuoristo, M.S.; Kellokumpu-Lehtinen, P.; Kahari, V.M.; Pyrhonen, S. High serum levels of matrix metalloproteinase-9 and matrix metalloproteinase-1 are associated with rapid progression in patients with metastatic melanoma. Clin. Cancer Res. 2005, 11, 5158–5166. [Google Scholar] [CrossRef] [PubMed]
- Malaponte, G.; Zacchia, A.; Bevelacqua, Y.; Marconi, A.; Perrotta, R.; Mazzarino, M.C.; Cardile, V.; Stivala, F. Co-regulated expression of matrix metalloproteinase-2 and transforming growth factor-beta in melanoma development and progression. Oncol. Rep. 2010, 24, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Bhadra, K.; Kumar, G.S. Therapeutic potential of nucleic acid-binding isoquinoline alkaloids: Binding aspects and implications for drug design. Med. Res. Rev. 2011, 31, 821–862. [Google Scholar] [CrossRef] [PubMed]
- Hwang, B.Y.; Roberts, S.K.; Chadwick, L.R.; Wu, C.D.; Kinghorn, A.D. Antimicrobial constituents from goldenseal (the Rhizomes of Hydrastis canadensis) against selected oral pathogens. Planta Med. 2003, 69, 623–627. [Google Scholar] [PubMed]
- Kuo, C.L.; Chi, C.W.; Liu, T.Y. The anti-inflammatory potential of berberine in vitro and in vivo. Cancer Lett. 2004, 203, 127–137. [Google Scholar] [CrossRef] [PubMed]
- Tomosaka, H.; Chin, Y.W.; Salim, A.A.; Keller, W.J.; Chai, H.; Kinghorn, A.D. Antioxidant and cytoprotective compounds from Berberis vulgaris (barberry). Phytother. Res. PTR 2008, 22, 979–981. [Google Scholar] [CrossRef] [PubMed]
- Abd El-Wahab, A.E.; Ghareeb, D.A.; Sarhan, E.E.; Abu-Serie, M.M.; El Demellawy, M.A. In vitro biological assessment of Berberis vulgaris and its active constituent, berberine: Antioxidants, anti-acetylcholinesterase, anti-diabetic and anticancer effects. BMC Complement. Altern. Med. 2013, 13, 218. [Google Scholar] [CrossRef] [PubMed]
- Tillhon, M.; Guaman Ortiz, L.M.; Lombardi, P.; Scovassi, A.I. Berberine: New perspectives for old remedies. Biochem. Pharmacol. 2012, 84, 1260–1267. [Google Scholar] [CrossRef] [PubMed]
- Zou, K.; Li, Z.; Zhang, Y.; Zhang, H.Y.; Li, B.; Zhu, W.L.; Shi, J.Y.; Jia, Q.; Li, Y.M. Advances in the study of berberine and its derivatives: A focus on anti-inflammatory and anti-tumor effects in the digestive system. Acta Pharmacol. Sin. 2017, 38, 157–167. [Google Scholar] [CrossRef] [PubMed]
- Eo, S.H.; Kim, J.H.; Kim, S.J. Induction of G(2)/M Arrest by Berberine via Activation of PI3K/Akt and p38 in Human Chondrosarcoma Cell Line. Oncol. Res. 2014, 22, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Yip, N.K.; Ho, W.S. Berberine induces apoptosis via the mitochondrial pathway in liver cancer cells. Oncol. Rep. 2013, 30, 1107–1112. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.M.; Li, T.M.; Tan, T.W.; Fong, Y.C.; Tang, C.H. Berberine Reduces the Metastasis of Chondrosarcoma by Modulating the alpha v beta 3 Integrin and the PKC delta, c-Src, and AP-1 Signaling Pathways. Evid.-Based Complement. Altern. Med. ECAM 2013, 2013, 423164. [Google Scholar]
- Liu, C.H.; Tang, W.C.; Sia, P.; Huang, C.C.; Yang, P.M.; Wu, M.H.; Lai, I.L.; Lee, K.H. Berberine inhibits the metastatic ability of prostate cancer cells by suppressing epithelial-to-mesenchymal transition (EMT)-associated genes with predictive and prognostic relevance. Int. J. Med. Sci. 2015, 12, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Ji, Q.; Ye, N.; Sui, H.; Zhou, L.; Zhu, H.; Fan, Z.; Cai, J.; Li, Q. Berberine Inhibits Invasion and Metastasis of Colorectal Cancer Cells via COX-2/PGE2 Mediated JAK2/STAT3 Signaling Pathway. PLoS ONE 2015, 10, e0123478. [Google Scholar] [CrossRef] [PubMed]
- Li, D.X.; Zhang, J.; Zhang, Y.; Zhao, P.W.; Yang, L.M. Inhibitory effect of berberine on human skin squamous cell carcinoma A431 cells. Genet. Mol. Res. 2015, 14, 10553–10568. [Google Scholar] [CrossRef] [PubMed]
- Ricci, M.S.; Zong, W.X. Chemotherapeutic approaches for targeting cell death pathways. Oncologist 2006, 11, 342–357. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Liang, Q.; Yang, X.; Yu, Y.; Shen, X.; Sun, G. Combination of sorafenib and Valproic acid synergistically induces cell apoptosis and inhibits hepatocellular carcinoma growth via down-regulating Notch3 and pAkt. Am. J. Cancer Res. 2017, 7, 2503–2514. [Google Scholar] [PubMed]
- Zanotto-Filho, A.; Rajamanickam, S.; Loranc, E.; Masamsetti, P.; Gorthi, A.; Romero, J.C.; Tonapi, S.; Goncalves, R.M.; Reddick, R.L.; Benavides, R.; et al. Sorafenib improves alkylating therapy by blocking induced inflammation, invasion and angiogenesis in breast cancer cells. Cancer Lett. 2018. [Google Scholar] [CrossRef] [PubMed]
- Yin, P.; Song, G.; Jiang, Z. Cisplatin suppresses proliferation, migration and invasion of nasopharyngeal carcinoma cells in vitro by repressing the Wnt/beta-catenin/Endothelin-1 axis via activating B cell translocation gene 1. Cancer Chemother. Pharmacol. 2018, 81, 863–872. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.; Zhang, N.; Feng, Y.; Cao, J.; Chen, X.; Liu, B. Aspirin Inhibits IKK-beta-mediated Prostate Cancer Cell Invasion by Targeting Matrix Metalloproteinase-9 and Urokinase-Type Plasminogen Activator. Cell. Physiol. Biochem. 2017, 41, 1313–1324. [Google Scholar] [CrossRef] [PubMed]
- Lai, K.C.; Hsiao, Y.T.; Yang, J.L.; Ma, Y.S.; Huang, Y.P.; Chiang, T.A.; Chung, J.G. Benzyl isothiocyanate and phenethyl isothiocyanate inhibit murine melanoma B16F10 cell migration and invasion in vitro. Int. J. Oncol. 2017, 51, 832–840. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Kim, M.J.; Kim, E.J.; Yang, Y.; Lee, M.S.; Lim, J.S. Berberine-induced AMPK activation inhibits the metastatic potential of melanoma cells via reduction of ERK activity and COX-2 protein expression. Biochem. Pharmacol. 2012, 83, 385–394. [Google Scholar] [CrossRef] [PubMed]
- Smalley, K.S. PLX-4032, a small-molecule B-Raf inhibitor for the potential treatment of malignant melanoma. Curr. Opin. Investig. Drugs 2010, 11, 699–706. [Google Scholar] [PubMed]
- Kou, Y.; Li, L.; Li, H.; Tan, Y.; Li, B.; Wang, K.; Du, B. Berberine suppressed epithelial mesenchymal transition through cross-talk regulation of PI3K/AKT and RARalpha/RARbeta in melanoma cells. Biochem. Biophys. Res. Commun. 2016, 479, 290–296. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, N.; Li, H.; Liu, M.; Cao, F.; Yu, X.; Zhang, J.; Tan, Y.; Xiang, L.; Feng, Y. Up-Regulation of PAI-1 and Down-Regulation of uPA Are Involved in Suppression of Invasiveness and Motility of Hepatocellular Carcinoma Cells by a Natural Compound Berberine. Int. J. Mol. Sci. 2016, 17, 577. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; You, D.; Lu, M.; He, Y.; Yan, S. Inhibitory effect of norcantharidin on melanoma tumor growth and vasculogenic mimicry by suppressing MMP-2 expression. Oncol. Lett. 2017, 13, 1660–1664. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fan, S.; Liao, Y.; Liu, C.; Huang, Q.; Liang, H.; Ai, B.; Fu, S.; Zhou, S. Estrogen promotes tumor metastasis via estrogen receptor beta-mediated regulation of matrix-metalloproteinase-2 in non-small cell lung cancer. Oncotarget 2017, 8, 56443–56459. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, H.F.; Ma, R.R.; He, J.Y.; Zhang, H.; Liu, X.L.; Guo, X.Y.; Gao, P. Protocadherin 7 inhibits cell migration and invasion through E-cadherin in gastric cancer. Tumour Biol. 2017, 39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Canel, M.; Serrels, A.; Frame, M.C.; Brunton, V.G. E-cadherin-integrin crosstalk in cancer invasion and metastasis. J. Cell Sci. 2013, 126 Pt 2, 393–401. [Google Scholar] [CrossRef]
- Labernadie, A.; Kato, T.; Brugues, A.; Serra-Picamal, X.; Derzsi, S.; Arwert, E.; Weston, A.; Gonzalez-Tarrago, V.; Elosegui-Artola, A.; Albertazzi, L.; et al. A mechanically active heterotypic E-cadherin/N-cadherin adhesion enables fibroblasts to drive cancer cell invasion. Nat. Cell Biol. 2017, 19, 224–237. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, D.; Kim, S.; Koh, H.; Yoon, S.O.; Chung, A.S.; Cho, K.S.; Chung, J. Akt/PKB promotes cancer cell invasion via increased motility and metalloproteinase production. FASEB J. 2001, 15, 1953–1962. [Google Scholar] [CrossRef] [PubMed]
- Shukla, S.; Maclennan, G.T.; Hartman, D.J.; Fu, P.; Resnick, M.I.; Gupta, S. Activation of PI3K-Akt signaling pathway promotes prostate cancer cell invasion. Int. J. Cancer 2007, 121, 1424–1432. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thang, N.D.; Yajima, I.; Kumasaka, M.Y.; Iida, M.; Suzuki, T.; Kato, M. Deltex-3-like (DTX3L) stimulates metastasis of melanoma through FAK/PI3K/AKT but not MEK/ERK pathway. Oncotarget 2015, 6, 14290–14299. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Veit, C.; Genze, F.; Menke, A.; Hoeffert, S.; Gress, T.M.; Gierschik, P.; Giehl, K. Activation of phosphatidylinositol 3-kinase and extracellular signal-regulated kinase is required for glial cell line-derived neurotrophic factor-induced migration and invasion of pancreatic carcinoma cells. Cancer Res. 2004, 64, 5291–5300. [Google Scholar] [CrossRef] [PubMed]
- Gunther, W.; Skaftnesmo, K.O.; Arnold, H.; Terzis, A.J. Molecular approaches to brain tumour invasion. Acta Neurochir. 2003, 145, 1029–1036. [Google Scholar] [CrossRef] [PubMed]
- Senapati, S.; Rachagani, S.; Chaudhary, K.; Johansson, S.L.; Singh, R.K.; Batra, S.K. Overexpression of macrophage inhibitory cytokine-1 induces metastasis of human prostate cancer cells through the FAK-RhoA signaling pathway. Oncogene 2010, 29, 1293–1302. [Google Scholar] [CrossRef] [PubMed]
- Zhong, D.; Ran, J.H.; Tang, W.Y.; Zhang, X.D.; Tan, Y.; Chen, G.J.; Li, X.S.; Yan, Y. Mda-9/syntenin promotes human brain glioma migration through focal adhesion kinase (FAK)-JNK and FAK-AKT signaling. Asian Pac. J. Cancer Prev. 2012, 13, 2897–2901. [Google Scholar] [CrossRef] [PubMed]
- Kwiatkowska, A.; Kijewska, M.; Lipko, M.; Hibner, U.; Kaminska, B. Downregulation of Akt and FAK phosphorylation reduces invasion of glioblastoma cells by impairment of MT1-MMP shuttling to lamellipodia and downregulates MMPs expression. Biochim. Biophys. Acta 2011, 1813, 655–667. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shih, Y.L.; Chou, H.M.; Chou, H.C.; Lu, H.F.; Chu, Y.L.; Shang, H.S.; Chung, J.G. Casticin impairs cell migration and invasion of mouse melanoma B16F10 cells via PI3K/AKT and NF-kappaB signaling pathways. Environ. Toxicol. 2017, 32, 2097–2112. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, L.; Yoon, C.H.; Sharma, G.; Bertagnolli, M.M.; Cho, N.L. Sorafenib inhibits proliferation and invasion in desmoid-derived cells by targeting Ras/MEK/ERK and PI3K/Akt/mTOR pathways. Carcinogenesis 2018, 39, 681–688. [Google Scholar] [CrossRef] [PubMed]
- Li, C.Y.; Wang, Q.; Shen, S.; Wei, X.L.; Li, G.X. Oridonin inhibits migration, invasion, adhesion and TGF-beta1-induced epithelial-mesenchymal transition of melanoma cells by inhibiting the activity of PI3K/Akt/GSK-3beta signaling pathway. Oncol. Lett. 2018, 15, 1362–1372. [Google Scholar] [PubMed]
- Chou, Y.C.; Chang, M.Y.; Wang, M.J.; Yu, F.S.; Liu, H.C.; Harnod, T.; Hung, C.H.; Lee, H.T.; Chung, J.G. PEITC inhibits human brain glioblastoma GBM 8401 cell migration and invasion through the inhibition of uPA, Rho A, and Ras with inhibition of MMP-2, -7 and -9 gene expression. Oncol. Rep. 2015, 34, 2489–2496. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yodkeeree, S.; Chaiwangyen, W.; Garbisa, S.; Limtrakul, P. Curcumin, demethoxycurcumin and bisdemethoxycurcumin differentially inhibit cancer cell invasion through the down-regulation of MMPs and uPA. J. Nutr. Biochem. 2009, 20, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Hsu, H.H.; Hu, W.S.; Lin, Y.M.; Kuo, W.W.; Chen, L.M.; Chen, W.K.; Hwang, J.M.; Tsai, F.J.; Liu, C.J.; Huang, C.Y. JNK suppression is essential for 17beta-Estradiol inhibits prostaglandin E2-Induced uPA and MMP-9 expressions and cell migration in human LoVo colon cancer cells. J. Biomed. Sci. 2011, 18, 61. [Google Scholar] [CrossRef] [PubMed]
- Perkins, N.D. The diverse and complex roles of NF-kappaB subunits in cancer. Nat. Rev. Cancer 2012, 12, 121–132. [Google Scholar] [CrossRef] [PubMed]
- Joseph, E.W.; Pratilas, C.A.; Poulikakos, P.I.; Tadi, M.; Wang, W.; Taylor, B.S.; Halilovic, E.; Persaud, Y.; Xing, F.; Viale, A.; et al. The RAF inhibitor PLX4032 inhibits ERK signaling and tumor cell proliferation in a V600E BRAF-selective manner. Proc. Natl. Acad. Sci. USA 2010, 107, 14903–14908. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vultur, A.; Villanueva, J.; Herlyn, M. Targeting BRAF in advanced melanoma: A first step toward manageable disease. Clin. Cancer Res. 2011, 17, 1658–1663. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.Y.; Lien, J.C.; Huang, Y.P.; Liao, C.L.; Lin, J.J.; Fan, M.J.; Ko, Y.C.; Hsiao, Y.P.; Lu, H.F.; Chung, J.G. Casticin Inhibits A375.S2 Human Melanoma Cell Migration/Invasion through Downregulating NF-kappaB and Matrix Metalloproteinase-2 and -1. Molecules 2016, 21, 384. [Google Scholar] [CrossRef] [PubMed]
- Shiue, Y.W.; Lu, C.C.; Hsiao, Y.P.; Liao, C.L.; Lin, J.P.; Lai, K.C.; Yu, C.C.; Huang, Y.P.; Ho, H.C.; Chung, J.G. Casticin Induced Apoptosis in A375.S2 Human Melanoma Cells through the Inhibition of NF-[Formula: See text]B and Mitochondria-Dependent Pathways In Vitro and Inhibited Human Melanoma Xenografts in a Mouse Model In Vivo. Am. J. Chin. Med. 2016, 44, 637–661. [Google Scholar] [CrossRef] [PubMed]
- Huang, A.C.; Yang, M.D.; Hsiao, Y.T.; Lin, T.S.; Ma, Y.S.; Peng, S.F.; Hsia, T.C.; Cheng, Y.D.; Kuo, C.L.; Chung, J.G. Bufalin inhibits gefitinib resistant NCI-H460 human lung cancer cell migration and invasion in vitro. J. Ethnopharmacol. 2016, 194, 1043–1050. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.S.; Hsiao, Y.T.; Lin, J.J.; Liao, C.L.; Lin, C.C.; Chung, J.G. Phenethyl Isothiocyanate (PEITC) and Benzyl Isothiocyanate (BITC) Inhibit Human Melanoma A375.S2 Cell Migration and Invasion by Affecting MAPK Signaling Pathway In Vitro. Anticancer Res. 2017, 37, 6223–6234. [Google Scholar] [PubMed]
- Huang, Y.P.; Chang, N.W. PPARalpha modulates gene expression profiles of mitochondrial energy metabolism in oral tumorigenesis. BioMedicine 2016, 6, 3. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.Y.; Lien, C.H.; Lee, M.F.; Huang, C.Y. Quercetin suppresses cellular migration and invasion in human head and neck squamous cell carcinoma (HNSCC). BioMedicine 2016, 6, 15. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.-M.; Velmurugan, B.K.; Kuo, W.-W.; Chen, Y.-S.; Ho, T.-J.; Tsai, C.-T.; Ye, C.-X.; Tsai, C.-H.; Tsai, F.-J.; Huang, C.-Y. Inhibitory effect of alpinate Oxyphyllae fructus extracts on Ang II-induced cardiac pathological remodeling-related pathways in H9c2 cardiomyoblast cells. BioMedicine 2013, 3, 148–152. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds berberine and PLX4032 are commercially available. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, J.-F.; Lai, K.C.; Peng, S.-F.; Maraming, P.; Huang, Y.-P.; Huang, A.-C.; Chueh, F.-S.; Huang, W.-W.; Chung, J.-G. Berberine Inhibits Human Melanoma A375.S2 Cell Migration and Invasion via Affecting the FAK, uPA, and NF-κB Signaling Pathways and Inhibits PLX4032 Resistant A375.S2 Cell Migration In Vitro. Molecules 2018, 23, 2019. https://doi.org/10.3390/molecules23082019
Liu J-F, Lai KC, Peng S-F, Maraming P, Huang Y-P, Huang A-C, Chueh F-S, Huang W-W, Chung J-G. Berberine Inhibits Human Melanoma A375.S2 Cell Migration and Invasion via Affecting the FAK, uPA, and NF-κB Signaling Pathways and Inhibits PLX4032 Resistant A375.S2 Cell Migration In Vitro. Molecules. 2018; 23(8):2019. https://doi.org/10.3390/molecules23082019
Chicago/Turabian StyleLiu, Jia-Fang, Kuang Chi Lai, Shu-Fen Peng, Pornsuda Maraming, Yi-Ping Huang, An-Cheng Huang, Fu-Shin Chueh, Wen-Wen Huang, and Jing-Gung Chung. 2018. "Berberine Inhibits Human Melanoma A375.S2 Cell Migration and Invasion via Affecting the FAK, uPA, and NF-κB Signaling Pathways and Inhibits PLX4032 Resistant A375.S2 Cell Migration In Vitro" Molecules 23, no. 8: 2019. https://doi.org/10.3390/molecules23082019
APA StyleLiu, J.-F., Lai, K. C., Peng, S.-F., Maraming, P., Huang, Y.-P., Huang, A.-C., Chueh, F.-S., Huang, W.-W., & Chung, J.-G. (2018). Berberine Inhibits Human Melanoma A375.S2 Cell Migration and Invasion via Affecting the FAK, uPA, and NF-κB Signaling Pathways and Inhibits PLX4032 Resistant A375.S2 Cell Migration In Vitro. Molecules, 23(8), 2019. https://doi.org/10.3390/molecules23082019




