Antiulcer Agents: From Plant Extracts to Phytochemicals in Healing Promotion
Abstract
1. Introduction
2. Epidemiology of Peptic Ulcer
3. Symptoms and Clinical Features of Peptic Ulcer
4. Pathogenesis
5. Pharmacological Agents Effective in Disease Management
6. Plant Products and Phytochemicals as Antiulcerogenic and Gastroprotective Agents
6.1. Materials and Methods
6.2. Plant Extracts with Antiulcerogenic Activity
6.2.1. Centella asiatica
6.2.2. Baccharis dracunculifolia
6.2.3. Baccharis trimera
6.2.4. Hieracium gymnocephalum
6.2.5. Tanacetum larvatum
6.2.6. Vernonia condensata
6.2.7. Solidago chilensis
6.2.8. Cordia dichotoma
6.2.9. Moringa oleifera
6.2.10. Capparis zeylanica
6.2.11. Salvadora indica
6.2.12. Maytenus robusta
6.2.13. Mukia maderaspatana
6.2.14. Momordica cymbalaria
6.2.15. Cibotium barometz
6.2.16. Cyperus rotundus
6.2.17. Caesalpinia sappan
6.2.18. Archidendron jiringa
6.2.19. Alhagi maurorum
6.2.20. Cassia sieberiana
6.2.21. Tamarindus indica
6.2.22. Parkia speciosa
6.2.23. Morinda citrifolia
6.2.24. Kigelia africana
6.2.25. Cordia verbenacea
6.2.26. Barleria lupulina
6.2.27. Eremomastax speciosa
6.2.28. Hyptis suaveolens
6.2.29. Tectona grandis
6.2.30. Calamintha officinalis
6.2.31. Myristica malabarica
6.2.32. Talinum portulacifolium
6.2.33. Cratoxylum arborescens
6.2.34. Mammea americana
6.2.35. Terminalia catappa
6.2.36. Terminalia coriacea
6.2.37. Terminalia arjuna
6.2.38. Terminalia belerica
6.2.39. Terminalia fagifolia
6.2.40. Terminalia chebula
6.2.41. Argemone mexicana
6.2.42. Piper betle
6.2.43. Ficus religiosa
6.2.44. Scutia buxifolia
6.2.45. Ziziphus jujuba
6.2.46. Cecropia glaziovii
6.2.47. Osyris quadripartita
6.2.48. Anacardium occidentale
6.2.49. Anacardium humile
6.2.50. Spondias mombin
6.2.51. Toona ciliata
6.2.52. Bryophyllum pinnatum
6.2.53. Aframomum pruinosum
6.3. Phytochemicals with Antiulcerogenic Activity
6.4. Principle Components and Their Antiulcer Inhibitory Effects
6.4.1. Alkaloids
6.4.2. Terpenes and Terpenoids
6.4.3. Flavonoids
6.4.4. Saponins
6.4.5. Phenolic Acids
6.4.6. Tannins
6.4.7. Fatty Acids
7. Safety of Plant Products Used as Antiulcerogenic Agents
8. Conclusions and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Da Silva, L.M.; Allemand, A.; Mendes, D.A.G.; dos Santos, A.C.; André, E.; de Souza, L.M.; Cipriani, T.R.; Dartora, N.; Marques, M.C.A.; Baggio, C.H. Ethanolic extract of roots from Arctium lappa L. accelerates the healing of acetic acid-induced gastric ulcer in rats: Involvement of the antioxidant system. Food Chem. Toxicol. 2013, 51, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Hamedi, S.; Arian, A.A.; Farzaei, M.H. Gastroprotective effect of aqueous stem bark extract of ziziphus jujuba l. Against hcl/ethanol-induced gastric mucosal injury in rats. J. Tradit. Chin. Med. 2015, 35, 666–670. [Google Scholar] [CrossRef]
- Van Zanten, S.J.V.; Dixon, M.F.; Lee, A. The gastric transitional zones: Neglected links between gastroduodenal pathology and helicobacter ecology. Gastroenterology 1999, 116, 1217–1229. [Google Scholar] [CrossRef]
- Tytgat, G. Etiopathogenetic principles and peptic ulcer disease classification. Digest. Dis. 2011, 29, 454–458. [Google Scholar] [CrossRef] [PubMed]
- Malfertheiner, P.; Chan, F.K.; McColl, K.E. Peptic ulcer disease. Lancet 2009, 374, 1449–1461. [Google Scholar] [CrossRef]
- Graham, D.Y. Changing patterns of peptic ulcer, gastrooesophageal reflux disease and Helicobacter pylori: A unifying hypothesis. Eur. J. Gastroen. Hepat. 2003, 15, 571–572. [Google Scholar] [CrossRef] [PubMed]
- Suerbaum, S.; Michetti, P. Helicobacter pylori infection. N. Engl. J. Med. 2002, 347, 1175–1186. [Google Scholar] [CrossRef] [PubMed]
- Bauer, B.; Meyer, T.F. The human gastric pathogen Helicobacter pylori and its association with gastric cancer and ulcer disease. Ulcers 2011, 2011. [Google Scholar] [CrossRef]
- Pounder, R.; Ng, D. The prevalence of Helicobacter pylori infection in different countries. Aliment. Pharm. Therap. 1995, 9, 33–39. [Google Scholar]
- Snowden, F.M. Emerging and reemerging diseases: A historical perspective. Immunol. Rev. 2008, 225, 9–26. [Google Scholar] [CrossRef] [PubMed]
- Najm, W.I. Peptic ulcer disease. Primary Care: Clin. Office Pract. 2011, 38, 383–394. [Google Scholar] [CrossRef] [PubMed]
- Milosavljevic, T.; Kostić-Milosavljević, M.; Jovanović, I.; Krstić, M. Complications of peptic ulcer disease. Digest. Dis. 2011, 29, 491–493. [Google Scholar] [CrossRef] [PubMed]
- Duggan, J.M.; Duggan, A.E. The possible causes of the pandemic of peptic ulcer in the late 19th and early 20th century. Med. J. Aust. 2006, 185, 667. [Google Scholar] [PubMed]
- Levenstein, S.; Rosenstock, S.; Jacobsen, R.K.; Jorgensen, T. Psychological stress increases risk for peptic ulcer, regardless of Helicobacter pylori infection or use of nonsteroidal anti-inflammatory drugs. Clin. Gastroen. Hepatol. 2015, 13, 498–506.e491. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.-Y.; Zhang, P.-Y.; Aboul-Soud, M.A. From inflammation to gastric cancer: Role of Helicobacter pylori. Oncol. lett. 2017, 13, 543–548. [Google Scholar] [CrossRef] [PubMed]
- Smoot, D.T. How does Helicobacter pylori cause mucosal damage? Direct mechanisms. Gastroenterology 1997, 113, S31–S34. [Google Scholar] [CrossRef]
- Semeraro, N.; Montemurro, P.; Piccoli, C.; Muoio, V.; Colucci, M.; Giuliani, G.; Fumarola, D.; Pece, S.; Moran, A.P. Effect of Helicobacter pylori lipopolysaccharide (LPS) and LPS derivatives on the production of tissue factor and plasminogen activator inhibitor type 2 by human blood mononuclear cells. J. Infect. Dis. 1996, 174, 1255–1260. [Google Scholar] [CrossRef] [PubMed]
- Dumrese, C.; Slomianka, L.; Ziegler, U.; Choi, S.S.; Kalia, A.; Fulurija, A.; Lu, W.; Berg, D.E.; Benghezal, M.; Marshall, B. The secreted helicobacter cysteine—rich protein a causes adherence of human monocytes and differentiation into a macrophage-like phenotype. FEBS lett. 2009, 583, 1637–1643. [Google Scholar] [CrossRef] [PubMed]
- Wallace, J.L. How do nsaids cause ulcer disease? Best Pract. Res.Clin. Gastroen. 2000, 14, 147–159. [Google Scholar] [CrossRef]
- Dajani, E.; Trotman, B. Drugs for treatment of peptic ulcers. J. Assoc. Acad. Minority Physic. Official Publ. Asso. Acad. Minority Physic. 1992, 3, 78–88. [Google Scholar]
- Garrigues-Gil, V. Antacids in the treatment of peptic ulcer disease. Method. Find. Exp. And Clin. Pharm. 1989, 11, 73–77. [Google Scholar]
- McMillan, D.E.; Freeman, R.B. The milk alkali syndrome: A study of the acute disorder with comments on the development of the chronic condition. Medicine 1965, 44, 485–501. [Google Scholar] [CrossRef] [PubMed]
- Levy, G.; Lampman, T.; Kamath, B.L.; Garrettson, L. Decreased serum salicylate concentrations in children with rheumatic fever treated with antacid. N. Engl. J. Med. 1975, 293, 323–325. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama, T. Proton pump inhibitors: Key ingredients in helicobacter pylori eradication treatment. In Proton Pump Inhibitors: A Balanced View; Chiba, T., Malfertheiner, P., Satoh, H., Eds.; Karger Publishers: Basel, Switzerland, 2013; Volume 32, pp. 59–67. [Google Scholar]
- Tarnawski, A. Cytoprotective drugs. Drug Invest. 1990, 2, 1–6. [Google Scholar] [CrossRef]
- D’souza, R.; Dhume, V.G. Gastric cytoprotection. Indian J. Physiol. Pharm. 1991, 35, 88–98. [Google Scholar]
- De Andrade, S.F.; Lemos, M.; Comunello, E.; Noldin, V.F.; Cechinel Filho, V.; Niero, R. Evaluation of the antiulcerogenic activity of Maytenus robusta (Celastraceae) in different experimental ulcer models. J. Ethnopharmacol. 2007, 113, 252–257. [Google Scholar] [CrossRef] [PubMed]
- De Andrade, S.F.; Comunello, E.; Noldin, V.F.; Delle Monache, F.; Cechinel Filho, V.; Niero, R. Antiulcerogenic activity of fractions and 3, 15-dioxo-21α-hydroxy friedelane isolated from Maytenus robusta (Celastraceae). Arch. Pharm. Res. 2008, 31, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Boligon, A.A.; de Freitas, R.B.; de Brum, T.F.; Waczuk, E.P.; Klimaczewski, C.V.; de Ávila, D.S.; Athayde, M.L.; de Freitas Bauermann, L. Antiulcerogenic activity of Scutia buxifolia on gastric ulcers induced by ethanol in rats. Acta Pharm. Sinica B 2014, 4, 358–367. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.M.; Choi, M.J.; Kim, J.M.; Cha, K.H.; Lee, K.W.; Park, Y.H.; Hong, S.S.; Lee, D.H. Centella asiatica leaf extract protects against indomethacin-induced gastric mucosal injury in rats. J. Med. Food 2016, 19, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Gohil, K.J.; Patel, J.A.; Gajjar, A.K. Pharmacological review on centella asiatica: A potential herbal cure-all. Indian J. Pharm. Sci. 2010, 72, 546–556. [Google Scholar] [CrossRef] [PubMed]
- Cestari, S.H.; Bastos, J.K.; Di Stasi, L.C. Intestinal anti-inflammatory activity of Baccharis dracunculifolia in the trinitrobenzenesulphonic acid model of rat colitis. Evid. Based Compl. Alternat. Med. 2011, 2011, 524349. [Google Scholar]
- Massignani, J.J.; Lemos, M.; Maistro, E.L.; Schaphauser, H.P.; Jorge, R.F.; Sousa, J.P.B.; Bastos, J.K.; de Andrade, S.F. Antiulcerogenic activity of the essential oil of Baccharis dracunculifolia on different experimental models in rats. Phytother. Res. 2009, 23, 1355–1360. [Google Scholar] [CrossRef] [PubMed]
- Biondo, T.M.A.; Tanae, M.M.; Della Coletta, E.; Lima-Landman, M.T.R.; Lapa, A.J.; Souccar, C. Antisecretory actions of Baccharis trimera (Less.) DC aqueous extract and isolated compounds: Analysis of underlying mechanisms. J. Ethnopharmacol. 2011, 136, 368–373. [Google Scholar] [CrossRef] [PubMed]
- De Toledo Dias, L.F.; de Melo, E.S.; Hernandes, L.S.; Bacchi, E.M. Antiulcerogenic and antioxidant activities of Baccharis trimera (Less) DC (Asteraceae). Revista Brasileira De Farmacognosia-Brazilian J. Pharm. 2009, 19, 309–314. [Google Scholar] [CrossRef]
- Lívero, F.A.D.; da Silva, L.M.; Ferreira, D.M.; Galuppo, L.F.; Borato, D.G.; Prando, T.B.L.; Lourenco, E.L.B.; Strapasson, R.L.B.; Stefanello, M.E.A.; Werner, M.F.D.; et al. Hydroethanolic extract of Baccharis trimera promotes gastroprotection and healing of acute and chronic gastric ulcers induced by ethanol and acetic acid. N-S Arch. Pharmacol. 2016, 389, 985–998. [Google Scholar] [CrossRef] [PubMed]
- Petrovic, S.; Dobric, S.; Mimica-Dukic, N.; Simin, N.; Kukic, J.; Niketic, M. The antiinflammatory, gastroprotective and antioxidant activities of Hieracium gymnocephalum extract. Phytother. Res. 2008, 22, 1548–1551. [Google Scholar] [CrossRef] [PubMed]
- Petrovic, S.D.; Dobric, S.; Bokonjic, D.B.; Niketic, M.; Garcia-Pineres, A.; Merfort, I. Evaluation of Tanacetum larvatum for an anti-inflammatory activity and for the protection against indomethacin-induced ulcerogenesis in rats. J. Ethnopharmacol. 2003, 87, 109–113. [Google Scholar] [CrossRef]
- Boeing, T.; da Silva, L.M.; Somensi, L.B.; Cury, B.J.; Costa, A.P.M.; Petreanu, M.; Niero, R.; de Andrade, S.F. Antiulcer mechanisms of Vernonia condensata Baker: A medicinal plant used in the treatment of gastritis and gastric ulcer. J. Ethnopharmacol. 2016, 184, 196–207. [Google Scholar] [CrossRef] [PubMed]
- Frutuoso, V.S.; Gurjao, M.R.R.; Cordeiro, R.S.B.; Martins, M.A. Analgesic and anti-ulcerogenic effects of a polar extract from leaves of Vernonia-condensata. Planta Med. 1994, 60, 21–25. [Google Scholar] [CrossRef] [PubMed]
- De Barros, M.; da Silva, L.M.; Boeing, T.; Somensi, L.B.; Cury, B.J.; Burci, L.D.; Santin, J.R.; de Andrade, S.F.; Delle Monache, F.; Cechinel, V. Pharmacological reports about gastroprotective effects of methanolic extract from leaves of Solidago chilensis (Brazilian arnica) and its components quercitrin and afzelin in rodents. N-S Arch. Pharmacol. 2016, 389, 403–417. [Google Scholar] [CrossRef] [PubMed]
- Bucciarelli, A.; Minetti, A.; Milczakowskyg, C.; Skliar, M. Evaluation of gastroprotective activity and acute toxicity of Solidago chilensis Meyen (Asteraceae). Pharm. Biol. 2010, 48, 1025–1030. [Google Scholar] [CrossRef] [PubMed]
- Ganjare, A.B.; Nirmal, S.A.; Rub, R.A.; Patil, A.N.; Pattan, S.R. Use of cordia dichotoma bark in the treatment of ulcerative colitis. Pharm. Biol. 2011, 49, 850–855. [Google Scholar] [CrossRef] [PubMed]
- Sini, K.R.; Sinha, B.N.; Rajasekaran, A. Protective effects of Capparis zeylanica Linn. Leaf extract on gastric lesions in experimental animals. Avicenna J. Med. Biotechnol. 2011, 3, 31–35. [Google Scholar] [PubMed]
- Debnath, S.; Biswas, D.; Ray, K.; Guha, D. Moringa oleifera induced potentiation of serotonin release by 5-ht(3) receptors in experimental ulcer model. Phytomedicine 2011, 18, 91–95. [Google Scholar] [CrossRef] [PubMed]
- Choudhary, M.K.; Bodakhe, S.H.; Gupta, S.K. Assessment of the antiulcer potential of Moringa oleifera root-bark extract in rats. J. Acupunct. Meridian Stud. 2013, 6, 214–220. [Google Scholar] [CrossRef] [PubMed]
- Minaiyan, M.; Asghari, G.; Taheri, D.; Saeidi, M.; Nasr-Esfahani, S. Anti-inflammatory effect of moringa oleifera lam. Seeds on acetic acid-induced acute colitis in rats. Avicenna J. Phytomed. 2014, 4, 127–136. [Google Scholar] [PubMed]
- Sahoo, S.K.; Sahoo, H.B.; Priyadarshini, D.; Soundarya, G.; Kumar, C.K.; Rani, K.U. Antiulcer activity of ethanolic extract of Salvadora indica (W.) leaves on albino rats. J. Clin. Diagn. Res. 2016, 10, FF7–FF10. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, L.M.; Boeing, T.; Somensi, L.B.; Cury, B.J.; Steimbach, V.M.B.; Silveria, A.C.D.; Niero, R.; Cechinel, V.; Santin, J.R.; de Andrade, S.F. Evidence of gastric ulcer healing activity of Maytenus robusta Reissek: In vitro and in vivo studies. J. Ethnopharmacol. 2015, 175, 75–85. [Google Scholar] [CrossRef] [PubMed]
- Benvenutti, D.F.; Della Monache, F.; Cechinel Filho, V.; de Andrade, S.F.; Niero, R. Phytochemical analysis and gastroprotective activity of the root bark from Maytenus robusta. Nat. Prod. Commun. 2016, 11, 597–599. [Google Scholar] [PubMed]
- Dhasan, P.B.; Jegadeesan, M.; Kavimani, S. Antiulcer activity of aqueous extract of fruits of Momordica cymbalaria hook f. In wistar rats. Pharm. Res. 2010, 2, 58–61. [Google Scholar]
- Gomathy, G.; Venkatesan, D.; Palani, S. Gastroprotective potentials of the ethanolic extract of Mukia maderaspatana against indomethacin-induced gastric ulcer in rats. Nat. Prod. Res. 2015, 29, 2107–2111. [Google Scholar] [CrossRef] [PubMed]
- Al-Wajeeh, N.S.; Hajrezaie, M.; Al-Henhena, N.; Kamran, S.; Bagheri, E.; Zahedifard, M.; Saremi, K.; Noor, S.M.; Ali, H.M.; Abdulla, M.A. The antiulcer effect of Cibotium barometz leaves in rats with experimentally induced acute gastric ulcer. Drug Des. Dev. Ther. 2017, 11, 995–1009. [Google Scholar] [CrossRef] [PubMed]
- Al-Wajeeh, N.S.; Hajerezaie, M.; Noor, S.M.; Halabi, M.F.; Al-Henhena, N.; Azizan, A.H.S.; Kamran, S.; Hassandarvish, P.; Shwter, A.N.; Karimian, H.; et al. The gastro protective effects of Cibotium barometz hair on ethanol-induced gastric ulcer in sprague-dawley rats. BMC Vet. Res. 2017, 13. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.; Govindhan, S.; Baiju, E.C.; Padmavathi, G.; Kunnumakkara, A.B.; Padikkala, J. Cyperus rotundus L. Prevents non-steroidal anti-inflammatory drug-induced gastric mucosal damage by inhibiting oxidative stress. J. Basic Clin. Physiol. Pharmacol. 2015, 26, 485–490. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, M.; MahayRookh; Rehman, A.B.; Muhammad, N.; Amber; Younus, M.; Wazir, A. Assessment of anti-inflammatory, anti-ulcer and neuro-pharmacological activities of Cyperus rotundus Linn. Pak. J. Pharm. Sci. 2014, 27, 2241–2246. [Google Scholar] [PubMed]
- Chellappan, D.R.; Purushothaman, A.K.; Brindha, P. Gastroprotective potential of hydro-alcoholic extract of pattanga (Caesalpinia sappan Linn.). J. Ethnopharmacol. 2017, 197, 294–305. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, I.A.A.; Qader, S.W.; Abdulla, M.A.; Nimir, A.R.; Abdelwahab, S.I.; Al-Bayaty, F.H. Effects of Pithecellobium jiringa ethanol extract against ethanol-induced gastric mucosal injuries in sprague-dawley rats. Molecules 2012, 17, 2796–2811. [Google Scholar] [CrossRef] [PubMed]
- Shaker, E.; Mahmoud, H.; Mnaa, S. Anti-inflammatory and anti-ulcer activity of the extract from Alhagi maurorum (Camelthorn). Food Chem. Toxicol. 2010, 48, 2785–2790. [Google Scholar] [CrossRef] [PubMed]
- Abbiw, D.K. Useful Plants of Ghana; Richmond Intermediate Technology Publications and Royal Botanic Gardens Kew: London, UK, 1990; pp. 154–157. [Google Scholar]
- Nartey, E.T.; Ofosuhene, M.; Kudzi, W.; Agbale, C.M. Antioxidant and gastric cytoprotective prostaglandins properties of Cassia sieberiana roots bark extract as an anti-ulcerogenic agent. BMC Complem. Altern. Med. 2012, 12, 65. [Google Scholar] [CrossRef] [PubMed]
- Al Batran, R.; Al-Bayaty, F.; Jamil Al-Obaidi, M.M.; Abdualkader, A.M.; Hadi, H.A.; Ali, H.M.; Abdulla, M.A. In vivo antioxidant and antiulcer activity of Parkia speciosa ethanolic leaf extract against ethanol-induced gastric ulcer in rats. PLoS ONE 2013, 8, e64751. [Google Scholar] [CrossRef] [PubMed]
- Kalra, P.; Sharma, S.; Suman; Kumar, S. Antiulcer effect of the methanolic extract of Tamarindus indica seeds in different experimental models. J. Pharm. Bioallied Sci. 2011, 3, 236–241. [Google Scholar] [PubMed]
- Mahattanadul, S.; Ridtitid, W.; Nima, S.; Phdoongsombut, N.; Ratanasuwon, P.; Kasiwong, S. Effects of Morinda citrifolia aqueous fruit extract and its biomarker scopoletin on reflux esophagitis and gastric ulcer in rats. J. Ethnopharmacol. 2011, 134, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, M.M.; Olaleye, M.T.; Ineu, R.P.; Boligon, A.A.; Athayde, M.L.; Barbosa, N.B.V.; Rocha, J.B.T. Antioxidant and antiulcer potential of aqueous leaf extract of Kigelia africana against ethanol-induced ulcer in rats. Excli J. 2014, 13, 323–330. [Google Scholar] [PubMed]
- Roldao, E.D.; Witaicenis, A.; Seito, L.N.; Hiruma-Lima, C.A.; Di Stasi, L.C. Evaluation of the antiulcerogenic and analgesic activities of Cordia verbenacea DC. (Boraginaceae). J. Ethnopharmacol. 2008, 119, 94–98. [Google Scholar] [CrossRef] [PubMed]
- Suba, V.; Murugesan, T.; Kumaravelrajan, R.; Mandal, S.C.; Saha, B.P. Antiinflammatory, analgesic and antiperoxidative efficacy of Barleria lupulina Lindl. Extract. Phytother. Res. 2005, 19, 695–699. [Google Scholar] [CrossRef] [PubMed]
- Suba, V.; Murugesan, T.; Pal, M.; Mandal, S.C.; Saha, B.P. Antiulcer activity of methanol fraction of barleria lupulina lindl. In animal models. Phytother. Res. 2004, 18, 925–929. [Google Scholar] [CrossRef] [PubMed]
- Tan, P.V.; Nditafon, N.G.; Yewah, M.P.; Dimo, T.; Ayafor, F.J. Eremomastax speciosa: Effects of leaf aqueous extract on ulcer formation and gastric secretion in rats. J. Ethnopharmacol. 1996, 54, 139–142. [Google Scholar] [CrossRef]
- Amang, P.A.; Tan, P.V.; Patamaken, S.A.; Mefe, M.N. Cytoprotective and antioxidant effects of the methanol extract of Eremomastax speciosa in rats. Afri. J. Trad. Complem. Altern. Med. 2014, 11, 165–171. [Google Scholar] [CrossRef]
- André Perfusion, A.; Tan, P.V.; Ernestine, N.; Barthélemy, N. Antisecretory action of the extract of the aerial parts of eremomastax speciosa (acanthaceae) occurs through antihistaminic and anticholinergic pathways. Adv. Pharmacol. Sci. 2014, 2014, 323470. [Google Scholar] [PubMed]
- Monforte, M.T.; Lanuzza, F.; Pergolizzi, S.; Mondello, F.; Tzakou, O.; Galati, E.M. Protective effect of Calamintha officinalis moench leaves against alcohol-induced gastric mucosa injury in rats. Macroscopic, histologic and phytochemical analysis. Phytother. Res. 2012, 26, 839–844. [Google Scholar] [CrossRef] [PubMed]
- Jesus, N.Z.T.; Falcao, H.S.; Lima, G.R.M.; Caldas, M.R.D.; Sales, I.R.P.; Gomes, I.F.; Santos, S.G.; Tavares, J.F.; Barbosa, J.M.; Batista, L.M. Hyptis suaveolens (L.) poit (Lamiaceae), a medicinal plant protects the stomach against several gastric ulcer models. J. Ethnopharmacol. 2013, 150, 982–988. [Google Scholar] [CrossRef] [PubMed]
- Vera-Arzave, C.; Antonio, L.C.; Arrieta, J.; Cruz-Hernández, G.; Velasquez-Mendez, A.M.; Reyes-Ramírez, A.; Sánchez-Mendoza, M.E. Gastroprotection of suaveolol, isolated from Hyptis suaveolens, against ethanol-induced gastric lesions in wistar rats: Role of prostaglandins, nitric oxide and sulfhydryls. Molecules 2012, 17, 8917–8927. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.; Shukla, N.; Singh, P.; Sharma, R.; Rajendran, S.; Maurya, R.; Palit, G. Verbascoside isolated from tectona grandis mediates gastric protection in rats via inhibiting proton pump activity. Fitoterapia 2010, 81, 755–761. [Google Scholar] [CrossRef] [PubMed]
- Jainu, M.; Devi, C.S.S. Antiulcerogenic and ulcer healing effects of Solanum nigrum (L.) on experimental ulcer models: Possible mechanism for the inhibition of acid formation. J. Ethnopharmacol. 2006, 104, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Maity, B.; Banerjee, D.; Bandyopadhyay, S.K.; Chattopadhyay, S. Myristica malabarica heals stomach ulceration by increasing prostaglandin synthesis and angiogenesis. Planta Med. 2008, 74, 1774–1778. [Google Scholar] [CrossRef] [PubMed]
- Maity, B.; Banerjee, D.; Bandyopadhyay, S.K.; Chattopadhyay, S. Regulation of arginase/nitric oxide synthesis axis via cytokine balance contributes to the healing action of malabaricone b against indomethacin-induced gastric ulceration in mice. Int. Immunopharmacol. 2009, 9, 491–498. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, D.; Bauri, A.K.; Guha, R.K.; Bandyopadhyay, S.K.; Chattopadhyay, S. Healing properties of malabaricone b and malabaricone c, against indomethacin-induced gastric ulceration and mechanism of action. Eur. J. Pharmacol. 2008, 578, 300–312. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, D.; Maity, B.; Bauri, A.K.; Bandyopadhyay, S.K.; Chattopadhyay, S. Gastroprotective properties of Myristica malabarica against indometacin-induced stomach ulceration: A mechanistic exploration. J. Pharm. Pharm. 2007, 59, 1555–1565. [Google Scholar] [CrossRef] [PubMed]
- Gundamaraju, R.; Maheedhar, K.; Hwi, K.K. Exploiting the phenomenal anti-ulcerogenic potential of talinum portulacifolium ethanolic extract whole plant on albino rats: The therapeutic potential of chinese herb-mǎ chǐ xiàn kē (Portulacaceae). Pharm. Res. 2014, 6, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Toma, W.; Hiruma-Lima, C.A.; Guerrero, R.O.; Brito, A. Preliminary studies of Mammea americana L. (Guttiferae) bark/latex extract point to an effective antiulcer effect on gastric ulcer models in mice. Phytomedicine 2005, 12, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Sidahmed, H.M.; Hashim, N.M.; Mohan, S.; Abdelwahab, S.I.; Taha, M.M.; Dehghan, F.; Yahayu, M.; Ee, G.C.; Loke, M.F.; Vadivelu, J. Evidence of the gastroprotective and anti- helicobacter pylori activities of β-mangostin isolated from Cratoxylum arborescens (Vahl) Blume. Drug Des. Dev. Ther. 2016, 10, 297–313. [Google Scholar] [CrossRef] [PubMed]
- Sidahmed, H.M.; Abdelwahab, S.I.; Mohan, S.; Abdulla, M.A.; Mohamed Elhassan Taha, M.; Hashim, N.M.; Hadi, A.H.; Vadivelu, J.; Loke Fai, M.; Rahmani, M.; et al. α-mangostin from Cratoxylum arborescens (Vahl) Blume demonstrates anti-ulcerogenic property: A mechanistic study. Evid. Based Compl. Alternat. Med. 2013, 2013, 450840. [Google Scholar]
- Devi, R.S.; Kist, M.; Vani, G.; Devi, C.S.S. Effect of methanolic extract of terminalia arjuna against Helicobacter pylori 26695 lipopolysaccharide-induced gastric ulcer in rats. J. Pharm. Pharmacol. 2008, 60, 505–514. [Google Scholar] [CrossRef] [PubMed]
- Devi, R.S.; Narayan, S.; Vani, G.; Srinivasan, P.; Mohan, K.V.; Sabitha, K.E.; Devi, C.S.S. Ulcer protective effect of Terminalia arjuna on gastric mucosal defensive mechanism in experimental rats. Phytother. Res. 2007, 21, 762–767. [Google Scholar] [CrossRef] [PubMed]
- Jawanjal, H.; Rajput, M.S.; Agrawal, P.; Dange, V. Pharmacological evaluation of fruits of Terminalia belerica Roxb. For antiulcer activity. J. Complem. Int. Med. 2012, 9, Article 9. [Google Scholar] [CrossRef] [PubMed]
- Silva, L.P.; de Angelis, C.D.; Bonamin, F.; Kushima, H.; Mininel, F.J.; dos Santos, L.C.; Delella, F.K.; Felisbino, S.L.; Vilegas, W.; da Rocha, L.R.M.; et al. Terminalia catappa L.: A medicinal plant from the caribbean pharmacopeia with anti-helicobacter pylori and antiulcer action in experimental rodent models. J. Ethnopharmacol. 2015, 159, 285–295. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Prakash, T.; Kotresha, D.; Ansari, M.A.; Sahrm, U.R.; Kumar, B.; Debnath, J.; Goli, D. Antiulcerogenic activity of Terminalia chebula fruit in experimentally induced ulcer in rats. Pharm. Biol. 2011, 49, 262–268. [Google Scholar] [CrossRef] [PubMed]
- Mishra, V.; Agrawal, M.; Onasanwo, S.A.; Madhur, G.; Rastogi, P.; Pandey, H.P.; Palit, G.; Narender, T. Anti-secretory and cyto-protective effects of chebulinic acid isolated from the fruits of Terminalia chebula on gastric ulcers. Phytomedicine 2013, 20, 506–511. [Google Scholar] [CrossRef] [PubMed]
- Ali Khan, M.S.; Nazan, S.; Mat Jais, A.M. Flavonoids and anti-oxidant activity mediated gastroprotective action of leathery murdah, Terminalia coriacea (Roxb.) wight & arn. Leaf methanolic extract in rats. Arch. Gastroen. 2017, 54, 183–191. [Google Scholar]
- Nunes, P.H.M.; Martins, M.D.C.; Oliveira, R.D.M.; Chaves, M.H.; Sousa, E.A.; Leite, J.; Veras, L.M.; Almeida, F.R.C. Gastric antiulcerogenic and hypokinetic activities of Terminalia fagifolia Mart. & Zucc. (Combretaceae). Biomed Res. Int. 2014, 2014, 261745. [Google Scholar] [PubMed]
- Das, P.K.; Pillai, S.; Kar, D.; Pradhan, D.; Sahoo, S. Pharmacological efficacy of argemone mexicana plant extract, against cysteamine-induced duodenal ulceration in rats. Indian J. Med. Sci. 2011, 65, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Majumdar, B.; Ray Chaudhuri, S.G.; Ray, A.; Bandyopadhyay, S.K. Effect of ethanol extract of Piper betle Linn leaf on healing of nsaid-induced experimental ulcer—A novel role of free radical scavenging action. Indian J. Exp. Biol. 2003, 41, 311–315. [Google Scholar] [PubMed]
- Majumdar, B.; Chaudhuri, S.R.; Ray, A.; Bandyopadhyay, S.K. Potent antiulcerogenic activity of ethanol extract of leaf of Piper betle Linn by antioxidative mechanism. Indian J. Clin. Biochem. 2002, 17, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, S.; Banerjee, D.; Bauri, A.K.; Chattopadhyay, S.; Bandyopadhyay, S.K. Healing property of the piper betel phenol, allylpyrocatechol against indomethacin-induced stomach ulceration and mechanism of action. World J. Gastroen. 2007, 13, 3705–3713. [Google Scholar] [CrossRef]
- Yadav, S.K.; Adhikary, B.; Maity, B.; Bandyopadhyay, S.K.; Chattopadhyay, S. The gastric ulcer-healing action of allylpyrocatechol is mediated by modulation of arginase metabolism and shift of cytokine balance. Eur. J. Pharm. 2009, 614, 106–113. [Google Scholar] [CrossRef] [PubMed]
- Yadav, S.K.; Adhikary, B.; Bandyopadhyay, S.K.; Chattopadhyay, S. Inhibition of tnf-α, and nf-κb and jnk pathways accounts for the prophylactic action of the natural phenolic, allylpyrocatechol against indomethacin gastropathy. BBA 2013, 1830, 3776–3786. [Google Scholar] [CrossRef] [PubMed]
- Chaudhary, A.; Yadav, B.S.; Singh, S.; Maurya, P.K.; Mishra, A.; Srivastva, S.; Varadwaj, P.K.; Singh, N.K.; Mani, A. Docking-based screening of Ficus religiosa phytochemicals as inhibitors of human histamine h2 receptor. Pharm. Mag. 2017, 13, S706. [Google Scholar]
- Souccar, C.; Cysneiros, R.M.; Tanae, M.M.; Torres, L.M.B.; Lima-Landman, M.T.R.; Lapa, A.J. Inhibition of gastric acid secretion by a standardized aqueous extract of Cecropia glaziovii Sneth and underlying mechanism. Phytomedicine 2008, 15, 462–469. [Google Scholar] [CrossRef] [PubMed]
- Abebaw, M.; Mishra, B.; Gelayee, D.A. Evaluation of anti-ulcer activity of the leaf extract of Osyris quadripartita Decne. (santalaceae) in rats. J. Exp. Pharm. 2017, 9, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Ajibola, E.S.; Adeleye, O.E.; Okediran, B.S.; Rahman, S.A. Effect of intragastric administration of crude aqueous leaf extract of anacardium occidentale on gastric acid secretion in rats. Niger. J. Physiol. Sci. 2010, 25, 59–62. [Google Scholar] [PubMed]
- Behravan, E.; Heidari, M.R.; Heidari, M.; Fatemi, G.; Etemad, L.; Taghipour, G.; Abbasifard, M. Comparison of gastric ulcerogenicity of percolated extract of Anacardium occidentale (cashew nut) with indomethacin in rats. Pak. J. Pharm. Sci. 2012, 25, 111–115. [Google Scholar] [PubMed]
- Konan, N.A.; Bacchi, E.M. Antiulcerogenic effect and acute toxicity of a hydroethanolic extract from the cashew (Anacardium occidentale L.) leaves. J. Ethnopharm. 2007, 112, 237–242. [Google Scholar] [CrossRef] [PubMed]
- Morais, T.C.; Pinto, N.B.; Carvalho, K.; Rios, J.B.; Ricardo, N.; Trevisan, M.T.S.; Rao, V.S.; Santos, F.A. Protective effect of anacardic acids from cashew (Anacardium occidentale) on ethanol-induced gastric damage in mice. Chem. Biol. Int. 2010, 183, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Luiz-Ferreira, A.; Cola-Miranda, M.; Barbastefano, V.; Hiruma-Lima, C.A.; Vilegas, W.; Souza Brito, A.R. Should anacardium humile st. Hil be used as an antiulcer agent? A scientific approach to the traditional knowledge. Fitoterapia 2008, 79, 207–209. [Google Scholar] [CrossRef] [PubMed]
- Luiz-Ferreira, A.; Almeida, A.C.; Cola, M.; Barbastefano, V.; Almeida, A.B.; Batista, L.M.; Farias-Silva, E.; Pellizzon, C.H.; Hiruma-Lima, C.A.; Santos, L.C.; et al. Mechanisms of the gastric antiulcerogenic activity of Anacardium humile st. Hil on ethanol-induced acute gastric mucosal injury in rats. Molecules 2010, 15, 7153–7166. [Google Scholar] [CrossRef] [PubMed]
- Abo, K.; Ogunleye, V.; Ashidi, J. Antimicrobial potential of Spondias mombin, Croton zambesicus and Zygotritonia crocea. Phytother. Res. Int. J. Devoted Pharm. Toxicol. Eval. Nat. Prod. Derivatives 1999, 13, 494–497. [Google Scholar]
- Sabiu, S.; Garuba, T.; Sunmonu, T.; Ajani, E.; Sulyman, A.; Nurain, I.; Balogun, A. Indomethacin-induced gastric ulceration in rats: Protective roles of spondias mombin and ficus exasperata. Toxicol. Rep. 2015, 2, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Malairajan, P.; Gopalakrishnan, G.; Narasimhan, S.; Veni, K.J.K.; Kavimani, S. Anti-ulcer activity of crude alcoholic extract of Toona ciliata roemer (heart wood). J. Ethnopharmacol. 2007, 110, 348–351. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.L.; Bhot, M.A.; Chandra, N. Gastroprotective effect of aqueous extract and mucilage from Bryophyllum pinnatum (Lam.) Kurz. Ancient Sci. Life 2014, 33, 252–258. [Google Scholar] [CrossRef] [PubMed]
- Pal, S.; Chaudhuri, A.K.N. Studies on the antiulcer activity of a Bryophyllum pinnatum leaf extract in experimental-animals. J. Ethnopharmacol. 1991, 33, 97–102. [Google Scholar] [CrossRef]
- Mabeku, L.B.K.; Nana, B.N.; Bille, B.E.; Tchuenguem, R.T.; Nguepi, E. Anti-helicobacter pylori and antiulcerogenic activity of Aframomum pruinosum seeds on indomethacin-induced gastric ulcer in rats. Pharm. Biol. 2017, 55, 929–936. [Google Scholar] [CrossRef] [PubMed]
- Tournier, H.; Schinella, G.; De Balsa, E.M.; Buschiazzo, H. Effect of the chloroform extract of tanacetum vulgare and one of its active principles, parthenolide, on experimental gastric ulcer in rats. J. Pharm. Pharmacol. 1999, 51, 215–219. [Google Scholar] [CrossRef] [PubMed]
- Maria, A.O.M.; Franchi, A.M.; Wendel, G.H.; Gimeno, M.; Guzman, J.A.; Giordano, O.S.; Guerreiro, E. Gastric cytoprotective activity of dehydroleucodine in rats. Role of prostaglandins. Biol. Pharm. Bull. 1998, 21, 335–338. [Google Scholar] [CrossRef] [PubMed]
- Niero, R.; Mafra, A.P.; Lenzi, A.C.; Cechinel-Filho, V.; Tischer, C.A.; Malheiros, A.; De Souza, M.M.; Yunes, R.A.; Delle Monache, F. A new triterpene with antinociceptive activity from Maytenus robusta. Nat. Prod. Res. 2006, 20, 1315–1320. [Google Scholar] [CrossRef] [PubMed]
- Sousa, G.F.; Duarte, L.P.; Alcântara, A.F.; Silva, G.D.; Vieira-Filho, S.A.; Silva, R.R.; Oliveira, D.M.; Takahashi, J.A. New triterpenes from Maytenus robusta: Structural elucidation based on NMR experimental data and theoretical calculations. Molecules 2012, 17, 13439–13456. [Google Scholar] [CrossRef] [PubMed]
- Klein-Júnior, L.C.; Santin, J.R.; Niero, R.; de Andrade, S.F.; Cechinel-Filho, V. The therapeutic lead potential of metabolites obtained from natural sources for the treatment of peptic ulcer. Phytochem. Rev. 2012, 11, 567–616. [Google Scholar] [CrossRef]
- Niero, R.; Moser, R.; Busato, A.C.; Chechinel Filho, V.; Yunes, R.A.; Reis, A. A comparative chemical study of Maytenus ilicifolia Mart. Reiss and Maytenus robusta Reiss (Celastraceae). Zeitschrift für Naturforschung C 2001, 56, 158–162. [Google Scholar] [CrossRef]
- Häkkinen, S.H.; Kärenlampi, S.O.; Heinonen, I.M.; Mykkänen, H.M.; Törrönen, A.R. Content of the flavonols quercetin, myricetin, and kaempferol in 25 edible berries. J. Agric. Food Chem. 1999, 47, 2274–2279. [Google Scholar] [CrossRef]
- Pandian, R.S.; Anuradha, C.V.; Viswanathan, P. Gastroprotective effect of fenugreek seeds (Trigonella foenum graecum) on experimental gastric ulcer in rats. J. Ethnopharmacol. 2002, 81, 393–397. [Google Scholar] [CrossRef]
- Sharifi-Rad, M.; Varoni, E.M.; Salehi, B.; Sharifi-Rad, J.; Matthews, K.R.; Ayatollahi, S.A.; Kobarfard, F.; Ibrahim, S.A.; Mnayer, D.; Zakaria, Z.A.; et al. Plants of the genus Zingiber as a source of bioactive phytochemicals: From tradition to pharmacy. Molecules 2017, 22, 2145. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Rad, J.; Salehi, B.; Varoni, E.M.; Sharopov, F.; Yousaf, Z.; Ayatollahi, S.A.; Kobarfard, F.; Sharifi-Rad, M.; Afdjei, M.H.; Sharifi-Rad, M.; et al. Plants of the Melaleuca genus as antimicrobial agents: From farm to pharmacy. Phytother. Res. 2017, 31, 1475–1494. [Google Scholar] [CrossRef] [PubMed]
- Salehi, B.; Ayatollahi, S.A.; Segura-Carretero, A.; Kobarfard, F.; Contreras, M.D.M.; Faizi, M.; Sharifi-Rad, M.; Tabatabai, S.A.; Sharifi-Rad, J. Bioactive chemical compounds in Eremurus persicus (Joub. & Spach) Boiss. Essential oil and their health implications. Cell. Mol. Biol. 2017, 63, 1–17. [Google Scholar] [PubMed]
- Sharifi-Rad, J.; Ayatollahi, S.A.; Varoni, E.M.; Salehi, B.; Kobarfard, F.; Sharifi-Rad, M.; Iriti, M.; Sharifi-Rad, M. Chemical composition and functional properties of essential oils from Nepeta schiraziana Boiss. Farmacia 2017, 65, 802–812. [Google Scholar]
- Sharifi-Rad, J.; Salehi, B.; Schnitzler, P.; Ayatollahi, S.A.; Kobarfard, F.; Fathi, M.; Eisazadeh, M.; Sharifi-Rad, M. Susceptibility of herpes simplex virus type 1 to monoterpenes thymol, carvacrol, p-cymene and essential oils of Sinapis arvensis L., Lallemantia royleana Benth. and Pulicaria vulgaris gaertn. Cell. Mol. Biol. 2017, 63, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Rad, J.; Hoseini-Alfatemi, S.M.; Sharifi-Rad, M.; Miri, A. Phytochemical screening and antibacterial activity of different parts of the Prosopis farcta extracts against methicillin-resistant Staphylococcus aureus (MRSA). Minerva Biotecnol. 2014, 26, 287–293. [Google Scholar]
- Sharifi-Rad, M.; Tayeboon, G.S.; Sharifi-Rad, J.; Iriti, M.; Varoni, E.M.; Razazi, S. Inhibitory activity on type 2 diabetes and hypertension key-enzymes, and antioxidant capacity of Veronica persica phenolic-rich extracts. Cell. Mol. Biol. 2016, 62, 80–85. [Google Scholar] [PubMed]
- Sharifi-Rad, M.; Tayeboon, G.S.; Miri, A.; Sharifi-Rad, M.; Setzer, W.N.; Fallah, F.; Kuhestani, K.; Tahanzadeh, N.; Sharifi-Rad, J. Mutagenic, antimutagenic, antioxidant, anti-lipoxygenase and antimicrobial activities of scandix pecten-veneris L. Cell. Mol. Biol. 2016, 62, 8–16. [Google Scholar] [PubMed]
- Sahraie-Rad, M.; Izadyari, A.; Rakizadeh, S.; Sharifi-Rad, J. Preparation of strong antidandruff shampoo using medicinal plant extracts: A clinical trial and chronic dandruff treatment. Jundishapur J. Nat. Pharm. Prod. 2015, 10. [Google Scholar] [CrossRef]
- Bagheri, G.; Mirzaei, M.; Mehrabi, R.; Sharifi-Rad, J. Cytotoxic and antioxidant activities of Alstonia scholaris, Alstonia venenata and Moringa oleifera plants from india. Jundishapur J. Nat. Pharm. Prod. 2016, 11, e31129. [Google Scholar] [CrossRef]
- Stojanović-Radić, Z.; Pejčić, M.; Stojanović, N.; Sharifi-Rad, J.; Stanković, N. Potential of Ocimum basilicum L. and Salvia officinalis L. Essential oils against biofilms of P. aeruginosa clinical isolates. Cell. Mol. Biol. 2016, 62, 27–32. [Google Scholar] [PubMed]
- Sharifi-Rad, M.; Nazaruk, J.; Polito, L.; Morais-Braga, M.F.B.; Rocha, J.E.; Coutinho, H.D.M.; Salehi, B.; Tabanelli, G.; Montanari, C.; del Mar Contreras, M. Matricaria genus as a source of antimicrobial agents: From farm to pharmacy and food applications. Microbiol. Res. 2018, 215, 76–88. [Google Scholar] [CrossRef]
- Salehi, B.; Mishra, A.P.; Shukla, I.; Sharifi-Rad, M.; Contreras, M.d.M.; Segura-Carretero, A.; Fathi, H.; Nasrabadi, N.N.; Kobarfard, F.; Sharifi-Rad, J. Thymol, thyme, and other plant sources: Health and potential uses. Phytother. Res. 2018. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Rad, M.; Mnayer, D.; Morais-Braga, M.F.B.; Carneiro, J.N.P.; Bezerra, C.F.; Coutinho, H.D.M.; Salehi, B.; Martorell, M.; del Mar Contreras, M.; Soltani-Nejad, A. Echinacea plants as antioxidant and antibacterial agents: From traditional medicine to biotechnological applications. Phytother. Res. 2018. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Rad, M.; Varoni, E.M.; Iriti, M.; Martorell, M.; Setzer, W.N.; del Mar Contreras, M.; Salehi, B.; Soltani-Nejad, A.; Rajabi, S.; Tajbakhsh, M. Carvacrol and human health: A comprehensive review. Phytother. Res. 2018. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Rad, J.; Sureda, A.; Tenore, G.C.; Daglia, M.; Sharifi-Rad, M.; Valussi, M.; Tundis, R.; Sharifi-Rad, M.; Loizzo, M.R.; Oluwaseun Ademiluyi, A.; et al. Biological activities of essential oils: From plant chemoecology to traditional healing systems. Molecules 2017, 22, 70. [Google Scholar] [CrossRef] [PubMed]
- Efferth, T.; Koch, E. Complex interactions between phytochemicals. The multitarget therapeutic concept of phytotherapy. Curr. Drug Targets 2011, 12, 122–132. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Rad, J.; Salehi, B.; Stojanović-Radić, Z.Z.; Fokou, P.V.T.; Sharifi-Rad, M.; Mahady, G.B.; Sharifi-Rad, M.; Masjedi, M.R.; Lawal, T.O.; Ayatollahi, S.A.; et al. Medicinal plants used in the treatment of tuberculosis ethnobotanical and ethnopharmacological approaches. Biotechnol. Adv. 2017. [Google Scholar] [CrossRef] [PubMed]
- Salehi, B.; Kumar, N.; Şener, B.; Sharifi-Rad, M.; Kılıç, M.; Mahady, G.; Vlaisavljevic, S.; Iriti, M.; Kobarfard, F.; Setzer, W.; et al. Medicinal plants used in the treatment of human immunodeficiency virus. Int. J. Mol. Sci. 2018, 19, 1459. [Google Scholar] [CrossRef] [PubMed]
- Salehi, B.; Zucca, P.; SharifiRad, M.; Pezzani, R.; Rajabi, S.; Setzer, W.N.; Varoni, E.M.; Iriti, M.; Kobarfard, F.; Sharifi-Rad, J. Phytotherapeutics in cancer invasion and metastasis. Phytother. Res. 2018. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Rad, M.; Iriti, M.; Sharifi-Rad, M.; Gibbons, S.; Sharifi-Rad, J. Anti-methicillin-resistant Staphylococcus aureus (MRSA) activity of rubiaceae, fabaceae and poaceae plants: A search for new sources of useful alternative antibacterials against MRSA infections. Cell. Mol. Biol. 2016, 62, 39–45. [Google Scholar] [PubMed]
- Setzer, M.S.; Sharifi-Rad, J.; Setzer, W.N. The search for herbal antibiotics: An in-silico investigation of antibacterial phytochemicals. Antibiotics 2016, 5, 30. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Rad, J.; Mnayer, D.; Roointan, A.; Shahri, F.; Ayatollahi, S.A.M.; Sharifi-Rad, M.; Molaee, N.; Sharifi-Rad, M. Antibacterial activities of essential oils from Iranian medicinal plants on extended-spectrum β-lactamase-producing Escherichia coli. Cell. Mol. Biol. 2016, 62, 75–82. [Google Scholar] [PubMed]
- Sharifi-Rad, M.; Mnayer, D.; Tabanelli, G.; Stojanovic-Radic, Z.Z.; Sharifi-Rad, M.; Yousaf, Z.; Vallone, L.; Setzer, W.N.; Iriti, M. Plants of the genus Allium as antibacterial agents: From tradition to pharmacy. Cell. Mol. Biol. 2016, 62, 57–68. [Google Scholar] [PubMed]
- Sharifi-Rad, J.; Miri, A.; Hoseini-Alfatemi, S.M.; Sharifi-Rad, M.; Setzer, W.N.; Hadjiakhoondi, A. Chemical composition and biological activity of Pulicaria vulgaris essential oil from iran. Nat. Prod. Commun. 2014, 9, 1633–1636. [Google Scholar] [PubMed]
- Sharifi-Rad, J.; Hoseini-Alfatemi, S.M.; Sharifi-Rad, M.; Sharifi-Rad, M.; Iriti, M.; Sharifi-Rad, M.; Sharifi-Rad, R.; Raeisi, S. Phytochemical compositions and biological activities of essential oil from Xanthium strumarium L. Molecules 2015, 20, 7034–7047. [Google Scholar] [CrossRef] [PubMed]
- Beserra, F.P.; Rozza, A.L.; Vieira, A.J.; Gushiken, L.F.; Pellizzon, C.H. Antiulcerogenic compounds isolated from medicinal plants. In Studies in Natural Products Chemistry; Atta-ur-Rahman; Elsevier: Amsterdam, The Netherlands, 2016; Volume 47, pp. 215–234. [Google Scholar]
- Harsha, C.; Banik, K.; Bordoloi, D.; Kunnumakkara, A.B. Antiulcer properties of fruits and vegetables: A mechanism based perspective. Food Chem. Toxicol. 2017, 108, 104–119. [Google Scholar] [CrossRef] [PubMed]
- Hamasaki, N.; Ishii, E.; Tominaga, K.; Tezuka, Y.; Nagaoka, T.; Kadota, S.; Kuroki, T.; Yano, I. Highly selective antibacterial activity of novel alkyl quinolone alkaloids from a chinese herbal medicine, Gosyuyu (wu-chu-yu), against Helicobacter pylori in vitro. Microbiol. Immunol. 2000, 44, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Breviglieri, E.; da Silva, L.M.; Boeing, T.; Somensi, L.B.; Cury, B.J.; Gimenez, A.; Filho, V.C.; Andrade, S.F. Gastroprotective and anti-secretory mechanisms of 2-phenylquinoline, an alkaloid isolated from Galipea longiflora. Phytomedicine 2017, 25, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Wang, X.; Zhang, H.; He, Z.; Zhi, W.; Liu, F.; Wang, Y.; Niu, X. Anti-ulcerogenic effect of cavidine against ethanol-induced acute gastric ulcer in mice and possible underlying mechanism. Int. Immunopharm. 2016, 38, 450–459. [Google Scholar] [CrossRef] [PubMed]
- Li, W.F.; Hao, D.J.; Fan, T.; Huang, H.M.; Yao, H.; Niu, X.F. Protective effect of chelerythrine against ethanol-induced gastric ulcer in mice. Chem. Biol. Int. 2014, 208, 18–27. [Google Scholar] [CrossRef] [PubMed]
- Nicolau, L.A.D.; Carvalho, N.S.; Pacífico, D.M.; Lucetti, L.T.; Aragao, K.S.; Véras, L.M.C.; Souza, M.L.; Leite, J.; Medeiros, J.V. Epiisopiloturine hydrochloride, an imidazole alkaloid isolated from Pilocarpus microphyllus leaves, protects against naproxen-induced gastrointestinal damage in rats. Biomed. Pharmacother. 2017, 87, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Dey, A.; Mukherjeex, A.; Chaudhury, M. Alkaloids from apocynaceae: Origin, pharmacotherapeutic properties, and structureactivity studies. In Studies in Natural Products Chemistry; Atta-ur-Rahman; Elsevier: Amsterdam, The Netherlands, 2017; Volume 52, pp. 376–478. [Google Scholar]
- Zhang, S.L.; Li, H.; He, X.; Zhang, R.Q.; Sun, Y.H.; Zhang, C.F.; Wang, C.Z.; Yuan, C.S. Alkaloids from Mahonia bealei posses anti -H+/K+-atpase anti-gastrin effects on pyloric ligation-induced gastric ulcer in rats. Phytomedicine 2014, 21, 1356–1363. [Google Scholar] [CrossRef] [PubMed]
- Nam, J.H.; Jung, H.J.; Choi, J.; Lee, K.T.; Park, H.J. The anti-gastropathic and anti-rheumatic effect of niga-ichigoside f1 and 23-hydroxytormentic acid isolated from the unripe fruits of Rubus coreanus in a rat model. Biol. Pharm. Bull. 2006, 29, 967–970. [Google Scholar] [CrossRef] [PubMed]
- Memariani, Z.; Sharifzadeh, M.; Bozorgi, M.; Hajimahmoodi, M.; Farzaei, M.H.; Gholami, M.; Siavoshi, F.; Saniee, P. Protective effect of essential oil of Pistacia atlantica desf. On peptic ulcer: Role of α-pinene. J. Tradit. Chin. Med. 2017, 37, 57–63. [Google Scholar] [CrossRef]
- Takayama, C.; Faria, F.M.; Almeida, A.C.; Valim-Araujo, D.A.; Rehen, C.S.; Dunder, R.J.; Socca, E.A.; Manzo, L.P.; Rozza, A.L.; Salvador, M.J.; et al. Gastroprotective and ulcer healing effects of essential oil from Hyptis spicigera lam (Lamiaceae). J. Ethnopharmacol. 2011, 135, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Diniz, L.R.L.; Vieira, C.F.X.; Santos, E.C.; Lima, G.C.; Aragao, K.K.V.; Vasconcelos, R.P.; Araújo, P.C.; Vasconcelos, Y.A.G.; Oliveira, A.C.; Oliveira, H.D.; et al. Gastroprotective effects of the essential oil of Hyptis crenata pohl ex benth on gastric ulcer models. J. Ethnopharmacol. 2013, 149, 694–700. [Google Scholar] [CrossRef] [PubMed]
- Arunachalam, K.; Balogun, S.O.; Pavan, E.; Almeida, G.V.B.; Oliveira, R.G.; Wagner, T.; Filho, V.C.; Martins, D.O. Chemical characterization, toxicology and mechanism of gastric antiulcer action of essential oil from Gallesia integrifolia (spreng.) harms in the in vitro and in vivo experimental models. Biomed. Pharmacother. 2017, 94, 292–306. [Google Scholar] [CrossRef] [PubMed]
- Bonamin, F.; Moraes, T.M.; Dos Santos, R.C.; Kushima, H.; Faria, F.M.; Silva, M.A.; Junior, I.V.; Nogueira, L.; Bauab, T.M.; Brito, A.S.; et al. The effect of a minor constituent of essential oil from Citrus aurantium: The role of β-myrcene in preventing peptic ulcer disease. Chem. Biol. Int. 2014, 212, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Vidal, C.S.; Martins, A.O.; Silva, A.A.; Oliveira, M.R.; Ribeiro-Filho, J.; Albuquerque, T.R.; Coutinho, H.D.; Almeida, J.R.; Junior, L.J.; Menezes, I.R.A. Gastroprotective effect and mechanism of action of Croton rhamnifolioides essential oil in mice. Biomed. Pharmacother. 2017, 89, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.J.; Lee, H.J.; Kim, B.S.; Lee, D.; Lee, S.J.; Yoo, S.H.; Chang, H.I. Antiulcer activity of anthocyanins from Rubus coreanus via association with regulation of the activity of matrix metalloproteinase-2. J. Agric. Food Chem. 2011, 59, 11786–11793. [Google Scholar] [CrossRef] [PubMed]
- Minozzo, B.R.; Lemes, B.M.; Justo, A.S.; Lara, J.E.; Petry, V.E.K.; Fernandes, D.; Bello, C.; Vellosa, J.C.R.; Campagnoli, E.B.; Nunes, O.C.; et al. Anti-ulcer mechanisms of polyphenols extract of euphorbia umbellata (pax) bruyns (euphorbiaceae). J. Ethnopharmacol. 2016, 191, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Abdelwahab, S.I. Protective mechanism of gallic acid and its novel derivative against ethanol-induced gastric ulcerogenesis: Involvement of immunomodulation markers, hsp70 and bcl-2-associated x protein. Int. Immunopharm. 2013, 16, 296–305. [Google Scholar] [CrossRef] [PubMed]
- Gomez, R.D.; Lopez-Solis, R.; Obreque-Slier, E.; Toledo-Araya, H. Comparative antibacterial effect of gallic acid and catechin against helicobacter pylori. LWT-Food Sci. Technol. 2013, 54, 331–335. [Google Scholar] [CrossRef]
- Asokkumar, K.; Sen, S.; Umamaheswari, M.; Sivashanmugam, A.T.; Subhadradevi, V. Synergistic effect of the combination of gallic acid and famotidine in protection of rat gastric mucosa. Pharmacol. Rep. 2014, 66, 594–599. [Google Scholar] [CrossRef] [PubMed]
- Neto, L.J.L.; Ramos, A.G.B.; Sales, V.S.; Souza, S.D.G.; Santos, A.T.L.; Oliverira, L.R.; Kerntopf, M.R.; Albuquerque, T.R.; Coutinho, H.D.M.; Quintans-Junior, L.J.; et al. Gastroprotective and ulcer healing effects of hydroethanolic extract of leaves of Caryocar coriaceum: Mechanisms involved in the gastroprotective activity. Chem. Biol. Int. 2017, 261, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, F.; Saito, M.; Ariga, T.; Yoshimura, Y.; Nakazawa, H. Free radical scavenging activity and antiulcer activity of garcinol from garcinia indica fruit rind. J. Agric. Food Chem. 2000, 48, 2320–2325. [Google Scholar] [CrossRef]
- Bigoniya, P.; Singh, K. Ulcer protective potential of standardized hesperidin, a citrus flavonoid isolated from Citrus sinensis. Rev. Bras. Farmacogn. 2014, 24, 330–334. [Google Scholar] [CrossRef]
- Izzo, A.A.; Carlo, G.D.; Mascolo, N.; Capasso, F.; Autore, G. Antiulcer effect of flavonoids. Role of endogenous paf. Phytother. Res. 1994, 8, 179–181. [Google Scholar] [CrossRef]
- Junior, I.F.S.; Balogun, S.O.; Oliveira, R.G.; Damazo, A.S.; Martins, D.T.O. Piper umbellatum l.: A medical plant with gastric-ulcer protective and ulcer healing effects in experimental rodent models. J. Ethnopharmacol. 2016, 192, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Panda, V.; Suresh, S. Gastro-protective effects of the phenolic acids of Macrotyloma uniflorum (horse gram) on experimental gastric ulcer models in rats. Food Biosci. 2015, 12, 34–46. [Google Scholar] [CrossRef]
- Al-Sayeda, E.; El-Naga, R.N. Protective role of ellagitannins from Eucalyptus citriodora against ethanol-induced gastric ulcer in rats: Impact on oxidative stress, inflammation and calcitonin-gene related peptide. Phytomedicine 2014, 30, 358–361. [Google Scholar] [CrossRef] [PubMed]
- Vasconcelos, P.C.P.; Andreo, M.A.; Vilegas, W.; Hiruma-Lima, C.A.; Pellizzon, C.H. Effect of Mouriri pusa tanins and flavonoids on prevention and treatment against experimental gastric ulcer. J. Ethnopharmacol. 2010, 131, 146–153. [Google Scholar] [CrossRef] [PubMed]
- Donato-Trancoso, A.; Monte-Alto-Costa, A.; Romana-Souza, B. Olive oil-induced reduction of oxidative damage and inflammation promotes wound healing of pressure ulcers in mice. J. Dermatol. Sci. 2016, 83, 60–69. [Google Scholar] [CrossRef] [PubMed]
- Falcao, H.S.; Leite, J.A.; Barbosa-Filho, J.M.; Athayde-Filho, P.F.; Chaves, M.C.O.; Moura, M.D.; Ferreira, M.F.; Almeida, A.B.A.; Souza-Brito, A.R.M.; Daniz, M.F.F.; et al. Gastric and duodenal antiulcer activity of alkaloids: A review. Molecules 2008, 13, 3198–3223. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Singh, V.; Chaudhary, A.K. Gastric antisecretory and antiulcer activities of Cedrus deodara (Roxb.) loud. In wistar rats. J. Ethnopharmacol. 2011, 134, 294–297. [Google Scholar] [CrossRef] [PubMed]
- Paguigan, N.D.; Castillo, D.H.B.; Chichioco-Hernandez, C.L. Anti-ulcer activity of Leguminosae plants. Arq. Gastroenterol. 2014, 51, 64–68. [Google Scholar] [CrossRef] [PubMed]
- Somensi, L.B.; Boeing, T.; Cury, B.J.; Steimbach, V.M.; Niero, R.; Souza, L.M.; Silva, L.M.; Andrade, S.F. Hydroalcoholic extract from bark of Persea major (meisn.) l.E. Kopp (Lauraceae) exerts antiulcer effects in rodents by the strengthening of the gastric protective factors. J. Ethnopharmacol. 2017, 209, 294–304. [Google Scholar] [CrossRef] [PubMed]
- Kuruuzum-Uz, A.; Suleyman, H.; Cadirci, E.; Guvenalp, Z.; Demirezer, L.O. Investigation on anti-inflammatory and antiulcer activities of Anchusa azurea extracts and their major constituent rosmarinic acid. Zeitschrift fur Naturforschung. C. J. Biosci. 2012, 67, 360–366. [Google Scholar] [CrossRef]
- Chemicals, D. OECD Guideline for Testing of Chemicals; The Organisation for Economic Co-operation and Development: Paris, France, 2005; pp. 1–13. [Google Scholar]
- Zakaria, Z.A.; Abdul Hisam, E.E.; Rofiee, M.S.; Norhafizah, M.; Somchit, M.N.; Teh, L.K.; Salleh, M.Z. In vivo antiulcer activity of the aqueous extract of Bauhinia purpurea leaf. J. Ethnopharmacol. 2011, 137, 1047–1054. [Google Scholar] [CrossRef] [PubMed]
- Perico, L.L.; Heredia-Vieira, S.C.; Beserra, F.P.; de Cassia Dos Santos, R.; Weiss, M.B.; Resende, F.A.; Dos Santos Ramos, M.A.; Bonifacio, B.V.; Bauab, T.M.; Varanda, E.A.; et al. Does the gastroprotective action of a medicinal plant ensure healing effects? An integrative study of the biological effects of Serjania marginata casar. (Sapindaceae) in rats. J. Ethnopharmacol. 2015, 172, 312–324. [Google Scholar] [CrossRef] [PubMed]
- Moshi, M.J.; Nondo, R.S.; Haule, E.E.; Mahunnah, R.L.; Kidukuli, A.W. Antimicrobial activity, acute toxicity and cytoprotective effect of Crassocephalum vitellinum (Benth.) s. Moore extract in a rat ethanol-hcl gastric ulcer model. BMC Res. Notes 2014, 7, 91. [Google Scholar] [CrossRef] [PubMed]
- Litchfield, J.T., Jr.; Wilcoxon, F. A simplified method of evaluating dose-effect experiments. J. Pharmacol. Exp. Ther. 1949, 96, 99–113. [Google Scholar] [PubMed]
- Raymundo, T.M.; Favilla, M.; Niero, R.; Andrade, S.F.; Maistro, E.L. Genotoxicity of the medicinal plant Maytenus robusta in mammalian cells in vivo. Genet. Mol. Res. 2012, 11, 2847–2854. [Google Scholar] [CrossRef] [PubMed]
- Hisam, E.E.; Zakaria, Z.A.; Mohtaruddin, N.; Rofiee, M.S.; Hamid, H.A.; Othman, F. Antiulcer activity of the chloroform extract of Bauhinia purpurea leaf. Pharm. Biol. 2012, 50, 1498–1507. [Google Scholar] [CrossRef] [PubMed]
- Albaayit, S.F.; Abba, Y.; Abdullah, R.; Abdullah, N. Evaluation of antioxidant activity and acute toxicity of Clausena excavata leaves extract. Evid-Based Complem. Altern. Med. 2014, 2014, 975450. [Google Scholar] [CrossRef] [PubMed]
- Moghadamtousi, S.Z.; Rouhollahi, E.; Karimian, H.; Fadaeinasab, M.; Abdulla, M.A.; Kadir, H.A. Gastroprotective activity of Annona muricata leaves against ethanol-induced gastric injury in rats via hsp70/bax involvement. Drug Des. Dev. Ther. 2014, 8, 2099–2110. [Google Scholar]
- Lemos, M.; Santin, J.R.; Junior, L.C.; Niero, R.; Andrade, S.F. Gastroprotective activity of hydroalcoholic extract obtained from the leaves of Brassica oleracea Var. Acephala DC in different animal models. J. Ethnopharmacol. 2011, 138, 503–507. [Google Scholar] [CrossRef] [PubMed]
- Burci, L.M.; Pereira, I.T.; da Silva, L.M.; Rodrigues, R.V.; Facundo, V.A.; Militao, J.S.; Santos, A.R.; Marques, M.C.; Baggio, C.H.; Werner, M.F. Antiulcer and gastric antisecretory effects of dichloromethane fraction and piplartine obtained from fruits of Piper tuberculatum Jacq. In rats. J. Ethnopharmacol. 2013, 148, 165–174. [Google Scholar] [CrossRef] [PubMed]
- Cavalcanti, A.M.; Baggio, C.H.; Freitas, C.S.; Rieck, L.; de Sousa, R.S.; Da Silva-Santos, J.E.; Mesia-Vela, S.; Marques, M.C. Safety and antiulcer efficacy studies of Achillea millefolium L. After chronic treatment in wistar rats. J. Ethnopharmacol. 2006, 107, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Carrasco, V.; Pinto, L.A.; Cordeiro, K.W.; Cardoso, C.A.; Freitas Kde, C. Antiulcer activities of the hydroethanolic extract of Sedum dendroideum Moc et Sesse ex DC. (balsam). J. Ethnopharmacol. 2014, 158 (Pt A), 345–351. [Google Scholar] [CrossRef] [PubMed]
- Muniappan, M.; Sundararaj, T. Antiinflammatory and antiulcer activities of Bambusa arundinacea. J. Ethnopharmacol. 2003, 88, 161–167. [Google Scholar] [CrossRef]
- Rodriguez, J.A.; Astudillo, L.; Schmeda-Hirschmann, G. Oleanolic acid promotes healing of acetic acid-induced chronic gastric lesions in rats. Pharmacol. Res. 2003, 48, 291–294. [Google Scholar] [CrossRef]
| Drug Classes | Characteristics | Types | Reference |
|---|---|---|---|
| Antacids | Help in neutralizing gastric acid, reducing acid delivery in duodenum and pepsin activity, besides to bind bile acids | Calcium and magnesium carbonates, aluminum hydroxide and magnesium trisilicate | [21,22,23] |
| Anti-secretory agents | Reduce gastric acid secretion, help relieve ulcer pain and stimulate ulcer healing, inhibit H. pylori growth and proliferation in gastric tissues | Histamine H2-receptor antagonist (cimetidine, famotidine, nizatidine and ranitidine), proton pump inhibitors (esomeprazole, lansoprazole, omeprazole, pantoprazole and rabeprazole) | [24] |
| Cytoprotective agents | Reduce/prevent gastric mucosal damage (increase mucus and bicarbonate secretion, strengthen gastric mucosal barrier, decrease gastric motility, increase blood flow to gastric mucosa, increase prostaglandins and sulfhydryl biosynthesis, scavenge free radicals, stimulate cell growth and repair and decrease leukotrienes release) | Prostaglandins, fatty acids, sulfhydryl compounds, aluminum-containing antacids, sucralfate, bismuth chelate and liquorice | [25,26] |
| Order | Family | Binomial Name | Mechanism of Gastroprotection | Reference |
|---|---|---|---|---|
| Apiales | Apiaceae | Centella asiatica | Wound healing, mucus production, antioxidant, anti-inflammatory | [30,31] |
| Asterales | Asteraceae | Baccharis dracunculifolia | Wound healing, antioxidant, mucus production | [32,33] |
| Baccharis trimera | Wound healing, anti-secretory, antioxidant | [34,35,36] | ||
| Hieracium gymnocephalum | Wound healing, anti-inflammatory | [37] | ||
| Tanacetum larvatum | Wound healing, anti-inflammatory, antioxidant | [38] | ||
| Vernonia condensata | Wound healing, inhibition of gastric secretion, antioxidant, mucus production, cytoprotective | [39,40] | ||
| Solidago chilensis | Wound healing, antioxidant, anti-secretory, mucus production | [41,42] | ||
| Boraginales | Boraginaceae | Cordia dichotoma | Wound healing, antioxidant, anti-inflammatory | [43] |
| Brassicales | Capparaceae | Capparis zeylanica | Wound healing | [44] |
| Moringaceae | Moringa oleifera | Wound healing, serotonin release, anti-secretory, cytoprotective, anti-inflammatory | [45,46,47] | |
| Salvadoraceae | Salvadora indica | Wound healing, cytoprotective | [48] | |
| Celastrales | Celastraceae | Maytenus robusta | Wound healing, mucus production, antioxidant, anti-inflammatory, cytoprotective, gastroprotective, anti-secretory | [27,28,49,50] |
| Cucurbitales | Cucurbitaceae | Momordica cymbalaria | Wound healing, anti-secretory | [51] |
| Mukia maderaspatana | Wound healing, antioxidant, anti-inflammatory, mucus production | [52] | ||
| Cyatheales | Cibotiaceae | Cibotium barometz | Wound healing, antioxidant, HSP70 up-regulation, Bax protein down-regulation, mucus production | [53,54] |
| Cyperales | Cyperaceae | Cyperus rotundus | Wound healing, antioxidant activity, anti-inflammatory | [55,56] |
| Fabales | Caesalpinieae | Caesalpinia sappan | Wound healing, antioxidant, cytoprotective, anti-inflammatory | [57] |
| Fabaceae | Archidendron jiringa | Wound healing, mucus production, antioxidant | [58] | |
| Alhagi maurorum | Antioxidant, antiapoptotic | [59] | ||
| Cassia sieberiana | Wound healing, cytoprotective, antioxidant, anti-inflammatory | [60,61] | ||
| Parkia speciosa | Wound healing, mucus production, antioxidant, anti-inflammatory, HSP70 up-regulation, Bax protein down-regulation | [62] | ||
| Tamarindus indica | Wound healing, anti-secretory | [63] | ||
| Gentianales | Rubiaceae | Morinda citrifolia | Wound healing, anti-secretory, antioxidant, anti-inflammatory | [64] |
| Lamiales | Bignoniaceae | Kigelia africana | Wound healing, antioxidant | [65] |
| Boraginaceae | Cordia verbenacea | Wound healing, antioxidant, cytoprotective | [66] | |
| Acanthaceae | Barleria lupulina | Anti-inflammatory, wound healing, anti-secretory | [67,68] | |
| Eremomastax speciosa | Wound healing, anti-secretory, mucus production, antioxidant, cytoprotective | [69,70,71] | ||
| Lamiaceae | Calamintha officinalis | Wound healing, antioxidant | [72] | |
| Hyptis suaveolens | Wound healing, cytoprotective, anti-inflammatory | [73,74] | ||
| Tectona grandis | Wound healing, inhibition of gastric secretion | [75,76] | ||
| Magnoliales | Myristicaceae | Myristica malabarica | Wound healing, anti-inflammatory, angiogenesis, cytoprotective | [77,78,79,80] |
| Magnoliopsida | Talinaceae | Talinum portulacifolium | Wound healing, anti-secretory | [81] |
| Malpighiales | Calophyllaceae | Mammea americana | Wound healing, anti-secretory | [82] |
| Hypericaceae | Cratoxylum arborescens | Wound healing, anti-H. pylori, anti-secretory, mucus production, antioxidant, antiapoptotic, anti-inflammatory, cytoprotective | [83,84] | |
| Myrtales | Combretaceae | Terminalia arjuna | Wound healing, anti-H. pylori, anti-secretory | [85,86] |
| Terminalia belerica | Wound healing, anti-secretory, mucus production | [87] | ||
| Terminalia catappa | Wound healing, anti-H. pylori, mucus production, anti-inflammatory, cytoprotective | [88] | ||
| Terminalia chebula | Wound healing, anti-secretory, cytoprotective | [89,90] | ||
| Terminalia coriacea | Wound healing, anti-secretory, mucus production, antioxidant | [91] | ||
| Terminalia fagifolia | Wound healing, anti-secretory, antioxidant | [92] | ||
| Papaverales | Papaveraceae | Argemone mexicana | Wound healing | [93] |
| Piperales | Piperaceae | Piper betle | Wound healing, antioxidant, mucus production, anti-inflammatory | [94,95,96,97,98] |
| Rosales | Moraceae | Ficus religiosa | Wound healing, inhibition of gastric secretion | [99] |
| Santalales | Rhamnaceae | Scutia buxifolia | Wound healing, antioxidant | [29] |
| Ziziphus jujuba | Anti-H. pylori, mucus production, antioxidant | [2] | ||
| Urticaceae | Cecropia glaziovii | Wound healing, anti-secretory | [100] | |
| Santalaceae | Osyris quadripartita | Wound healing | [101] | |
| Sapindales | Anacardiaceae | Anacardium occidentale | Wound healing, antioxidant, anti-inflammatory, cytoprotective | [102,103,104,105] |
| Anacardium humile | Wound healing, mucus production, anti-inflammatory, cytoprotective | [106,107] | ||
| Meliaceae | Spondias mombin | Wound healing, antioxidant, anti-inflammatory, inhibition of gastric secretion | [108,109] | |
| Toona ciliata | Wound healing, anti-secretory, cytoprotective | [110] | ||
| Saxifragales | Crassulaceae | Bryophyllum pinnatum | Wound healing, anti-inflammatory | [111,112] |
| Zingiberales | Zingiberaceae | Aframomum pruinosum | Wound healing, anti-H. pylori, mucus production, anti-inflammatory, cytoprotective | [113] |
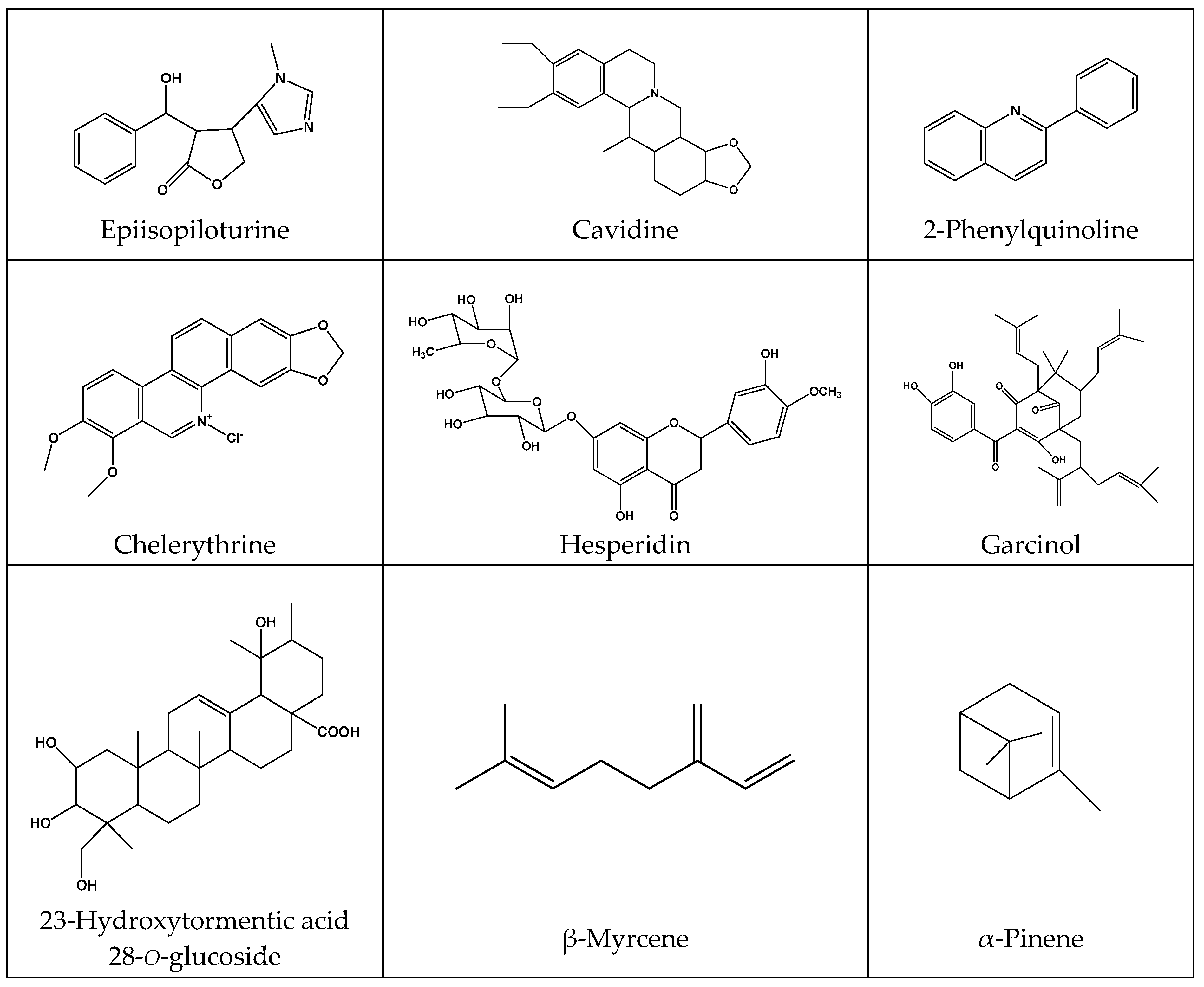
| Phytochemicals | Plant Source | Model | Dose/Results | Mode of Action | Reference |
|---|---|---|---|---|---|
| Alkaloids | |||||
| 1-methyl-2-[(z)-8-tridecenyl]-4-(1H)-quinolone and 1-methyl-2-[(z)-7-tridecenyl]-4-(1H)-quinolone | Evodia rutaecarpa | In vitro antibacterial activity against H. pylori | 0.05 μg/mL | Highly selective activity against H. pylori | [150] |
| 2-Phenylquinoline | Galipea longiflora | Gastric ulcer induced by 60% ethanol/0.03 M HCl, indomethacin-induced acute lesions in rats | 10–100 mg/kg | SOD and GST activity normalization, increased GSH and reduced LPO and TNF-α levels in gastric mucosa | [151] |
| Cavidine | Corydalis impatiens | Ethanol-induced acute gastric ulcer in mice | 10 mg/kg | Increased mucosa GSH, SOD and PGE2 levels, decreased IL-6 and TNF-α levels | [152] |
| Chelerythrine | Papaveraceae and Rutaceae family | Ethanol-induced gastric ulcer in mice | 1.5, 10 mg/kg | Reduced myeloperoxidase activity, IL-6 and TNF-α levels and inhibited NO | [153] |
| Epiisopiloturine | Pilocarpus microphyllus | Naproxen-induced gastrointestinal damage in rats | 10 mg/kg | Reduced pro-inflammatory cytokines, oxidative stress and increased gastric mucosal blood flow | [154] |
| Uleine | Himatanthus lancifolius | Ethanol-induced acute gastric ulcer and pylorus ligature-induced ulcer in rats | 30, 82 mg/kg | Increased GSH level, antioxidant response and decreased H+/K+-ATPase activity | [155] |
| Alkaloid fraction extract | Tylophora conpicua | Gastric acid secretion and ulceration in rat | 40 mg/kg | Decreased histamine insulted gastric acid secretion | [155] |
| Alkaloid fraction (columbamine, jatrorrhizine, palmatine and berberine) | Mahonia bealei | Pyloric ligation-induced gastric ulcer in rats | 18.6 mg/kg | Anti-H+/K+-ATPase anti-gastrin effects | [156] |
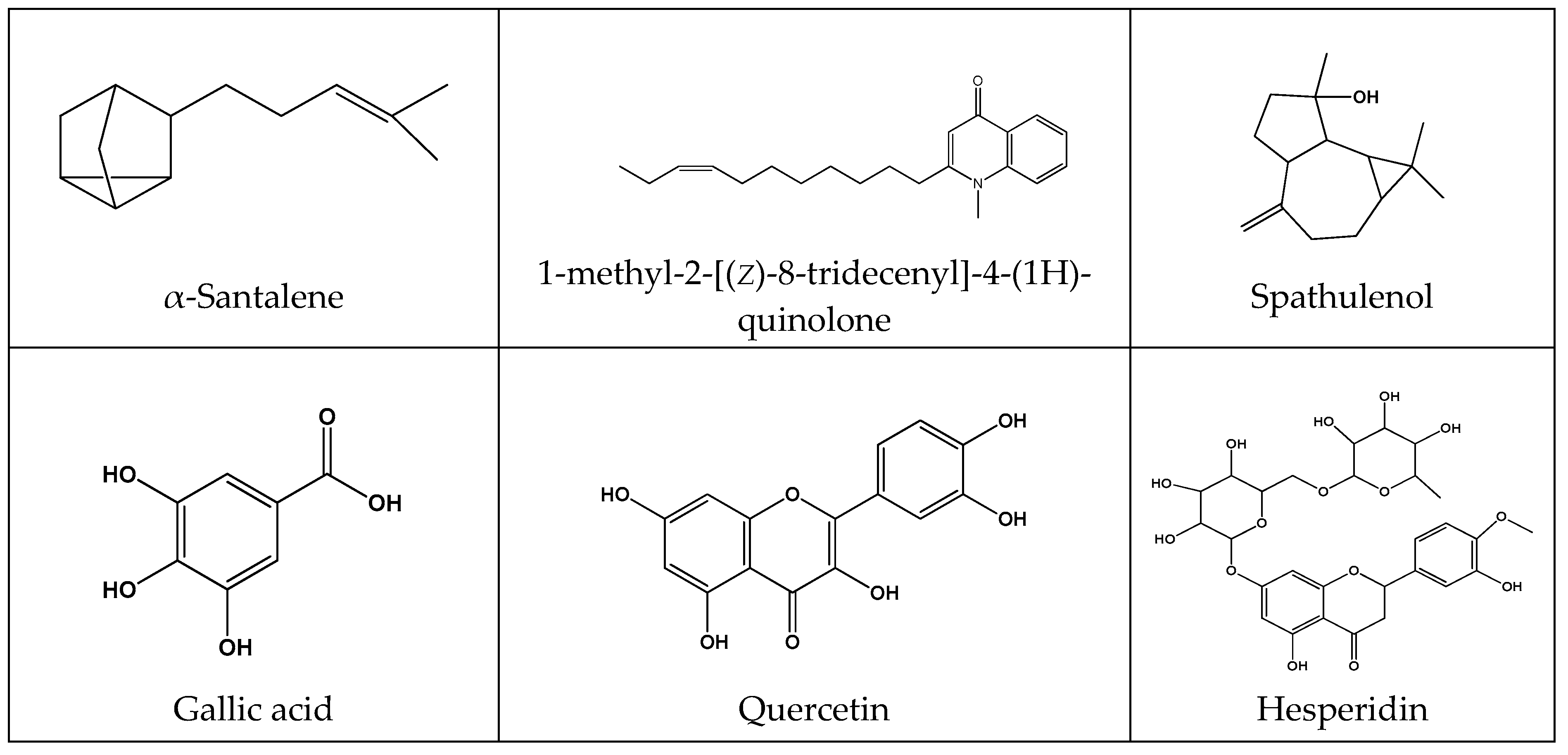
| Terpenes and Terpenoids | |||||
| 23-hydroxytormentic acid 28-o-glucoside and its aglycone | Rubus coreanus | Gastric ulcer induced by oral administration of ethanol + sodium salicylate | 10, 30 mg/kg | Increased SOD and GPx activity | [157] |
| α-Pinene | Pistacia atlantica | Ethanol-induced gastric ulcer | 12.32 mg/kg (EC50) | Antibacterial activity on metronidazole-resistant H. pylori | [158] |
| α-Pinene (50.8%), cineole (20.3%), β-pinene (18.3%) | Hyptis spicigera | Ethanol and NSAIDs rodent models | 100 mg/kg | Increased gastric mucus production and induced PGE2 | [159] |
| α-Pinene (13.4%), 1,8-cineole (18%), camphor (32.8%), β-caryophyllene (12.9%) | Hyptis crenata | Gastric ulcer induced by oral administration of absolute ethanol or indomethacin | 30, 100, 300 mg/kg | Accelerated gastric emptying effect and reduced oxidative damages | [160] |
| α-Santalene | Gallesia integrifolia | In vivo and in vitro experimental models | 80 mg/kg | Gastroprotective and curative effects, probably due to antioxidant, anti-inflammatory, anti-secretory, mucogenic and nitrergic and activity | [161] |
| β-Myrcene | Citrus aurantium | Ethanol, NSAIDs, stress, H. pylori, ischemia reperfusion injury and cysteamine-induced ulcers | 7.5 mg/kg | Decreased gastric and duodenal lesions, SOD activity, increased gastric mucus production, mucosal MDA levels and GPx and GR activity | [162] |
| Spathulenol (22.5%), 1,8-cineole (18.3%) | Croton rhamnifolioides | Gastric ulcer induced by administration of absolute ethanol, acidified ethanol or indomethacin | 100 mg/kg | Modulation of opioid receptors and NO | [163] |
| Phenolics and Favonoids | |||||
| Anthocyanins | Rubus coreanus | Naproxen-induced gastric ulcer | 20, 50 and 80 mg/kg | Via association with regulation of matrix metalloproteinase-2 activity; prevented lipid peroxidation and increased CAT, SOD and GPx activities | [164] |
| Ellagic and gallic acids | Euphorbia umbellata (Pax) Bruyns | Ethanol-induced acute gastric lesions in rats | 50, 100, 200 mg/kg | Potent antioxidant activity (PG, NO/cyclic guanosine monophosphate pathway related to antiulcer action) | [165] |
| Gallic acid | Widespread in plant kingdom | Ethanol-induced gastric ulcerogenesis | 25–50 mg/kg | Inhibited gastric acid secretion or through antioxidant action | [166] |
| Gallic acid and catechin | Widespread in plant kingdom | In vitro antimicrobial test on two H. pylori strains | 1 mg/mL | High inhibitory effect against H. pylori | [167] |
| Gallic acid + famotidine | Aspirin plus pyloric ligation induced ulcer in rat | 50 + 10 mg/kg (respectively) | Increased SOD, CAT, GR, GSH and G6PD levels and decreased myeloperoxidase and lipid peroxidation in stomach tissue | [168] | |
| Gallic, chlorogenic and caffeic acids, rutin and quercetin | Caryocar coriaceum | Gastric ulcer induced by ethanol, acidified ethanol, acetic acid or indomethacin | 100 mg/kg | Opioid receptors, α2-adrenergic receptors and primary afferent neurons sensitive to capsaicin | [169] |
| Garcinol | Garcinia indica | Acute ulceration in rats induced by indomethacin and water immersion stress | 200 mg/kg | Suppressed superoxide anion, hydroxyl and methyl radicals | [170] |
| Hesperidin | Citrus sinensis | Indomethacin plus hypothermic restrain stress-induced ulceration in rats | 150, 300 and 450 mg/kg | Increased GSH and mucin levels, prevented oxidative cell injury | [171] |
| Quercetin, rutin and kaempferol | Widespread in plant kingdom | gastric damage produced by acidified ethanol in rats | 25–100 mg/kg | Inhibited mucosal content of platelet-activating factor | [172] |
| Rutin and quercetin | Piper umbellatum L. | Experimental rodent models | 30, 100 and 300 mg/kg | Antioxidant, anti-secretory, anti-inflammatory and mucosa regeneration | [173] |
| p-Coumaric acid | Macrotyloma uniflorum | Indomethacin and ethanol-induced ulcer in rats | 250 mg/kg | Underlying antioxidant activity (attenuated ulcer elevated MDA levels and reduced GSH, SOD, CAT, GPx and GR levels) | [174] |
| Tannins | |||||
| Ellagitannin-rich fraction | Eucalyptus citriodora | Ethanol-induced gastric ulceration in rats | 25, 50 and 100 mg/kg | Increased GSH and SOD levels | [175] |
| Tannins (TF) and flavonoids (FF) fractions | Mouriri pusa | Gastric ulcer induced by ethanol and acetic acid | 25 mg/kg (TF) or 50 mg/kg (FF) | Cell proliferation improved, anti-inflammatory activity by reducing COX-2 levels, increased mucus secretion and angiogenesis | [176] |
| Fatty Acids | |||||
| Oleic acid (C18:1 cis 9) | Olive oil | Ulcer created in mice using ischemia-reperfusion-induced skin injury | 1500 mg/kg | Accelerated ROS and NO synthesis and reduced oxidative damage, inflammatory cells infiltration and TNF-α expression | [177] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sharifi-Rad, M.; Fokou, P.V.T.; Sharopov, F.; Martorell, M.; Ademiluyi, A.O.; Rajkovic, J.; Salehi, B.; Martins, N.; Iriti, M.; Sharifi-Rad, J. Antiulcer Agents: From Plant Extracts to Phytochemicals in Healing Promotion. Molecules 2018, 23, 1751. https://doi.org/10.3390/molecules23071751
Sharifi-Rad M, Fokou PVT, Sharopov F, Martorell M, Ademiluyi AO, Rajkovic J, Salehi B, Martins N, Iriti M, Sharifi-Rad J. Antiulcer Agents: From Plant Extracts to Phytochemicals in Healing Promotion. Molecules. 2018; 23(7):1751. https://doi.org/10.3390/molecules23071751
Chicago/Turabian StyleSharifi-Rad, Mehdi, Patrick Valere Tsouh Fokou, Farukh Sharopov, Miquel Martorell, Adedayo Oluwaseun Ademiluyi, Jovana Rajkovic, Bahare Salehi, Natália Martins, Marcello Iriti, and Javad Sharifi-Rad. 2018. "Antiulcer Agents: From Plant Extracts to Phytochemicals in Healing Promotion" Molecules 23, no. 7: 1751. https://doi.org/10.3390/molecules23071751
APA StyleSharifi-Rad, M., Fokou, P. V. T., Sharopov, F., Martorell, M., Ademiluyi, A. O., Rajkovic, J., Salehi, B., Martins, N., Iriti, M., & Sharifi-Rad, J. (2018). Antiulcer Agents: From Plant Extracts to Phytochemicals in Healing Promotion. Molecules, 23(7), 1751. https://doi.org/10.3390/molecules23071751











