Pubescenosides E–K, Seven New Triterpenoid Saponins from the Roots of Ilex pubescens and Their Anti-Inflammatory Activity
Abstract
1. Introduction
2. Results
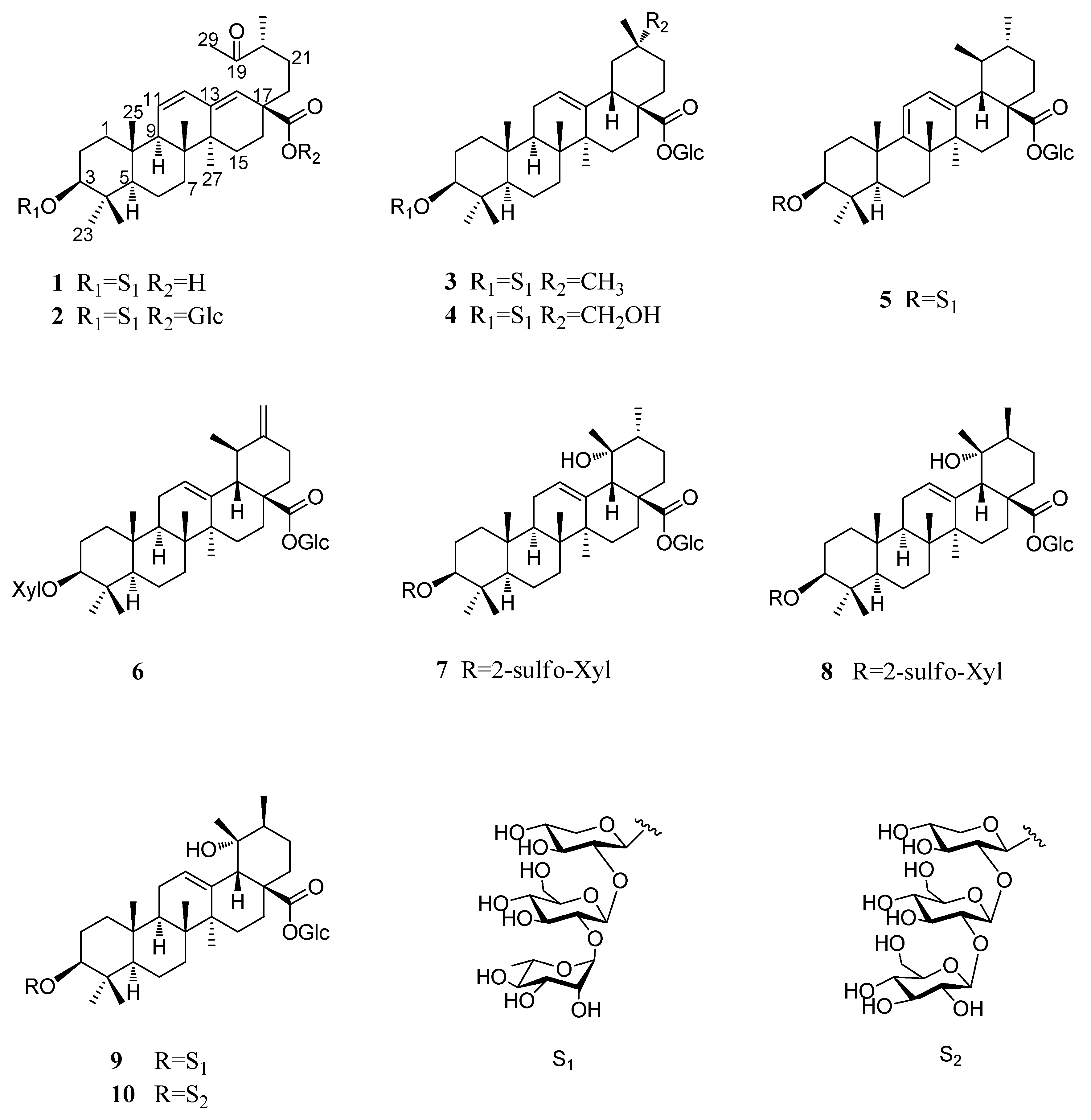
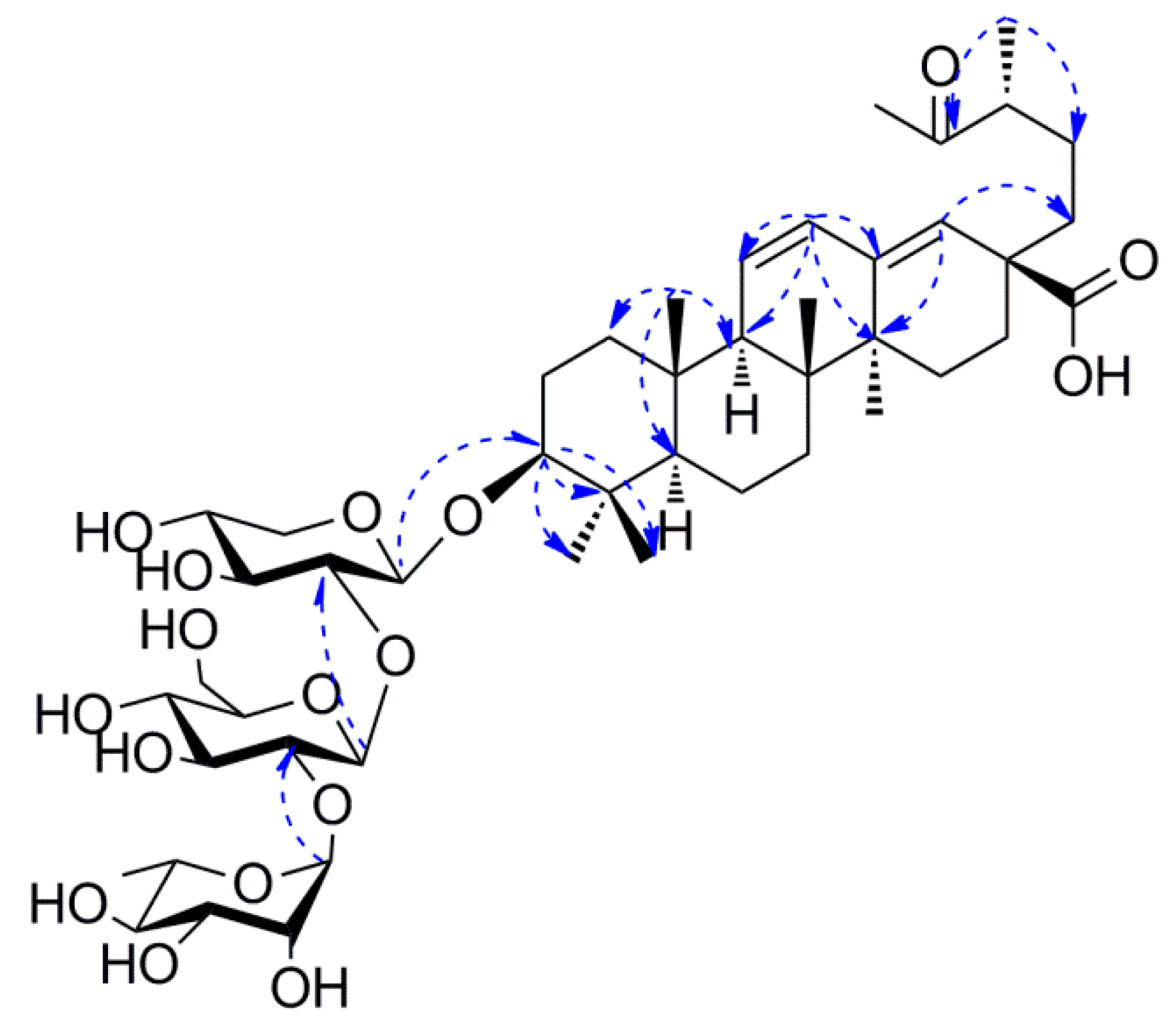
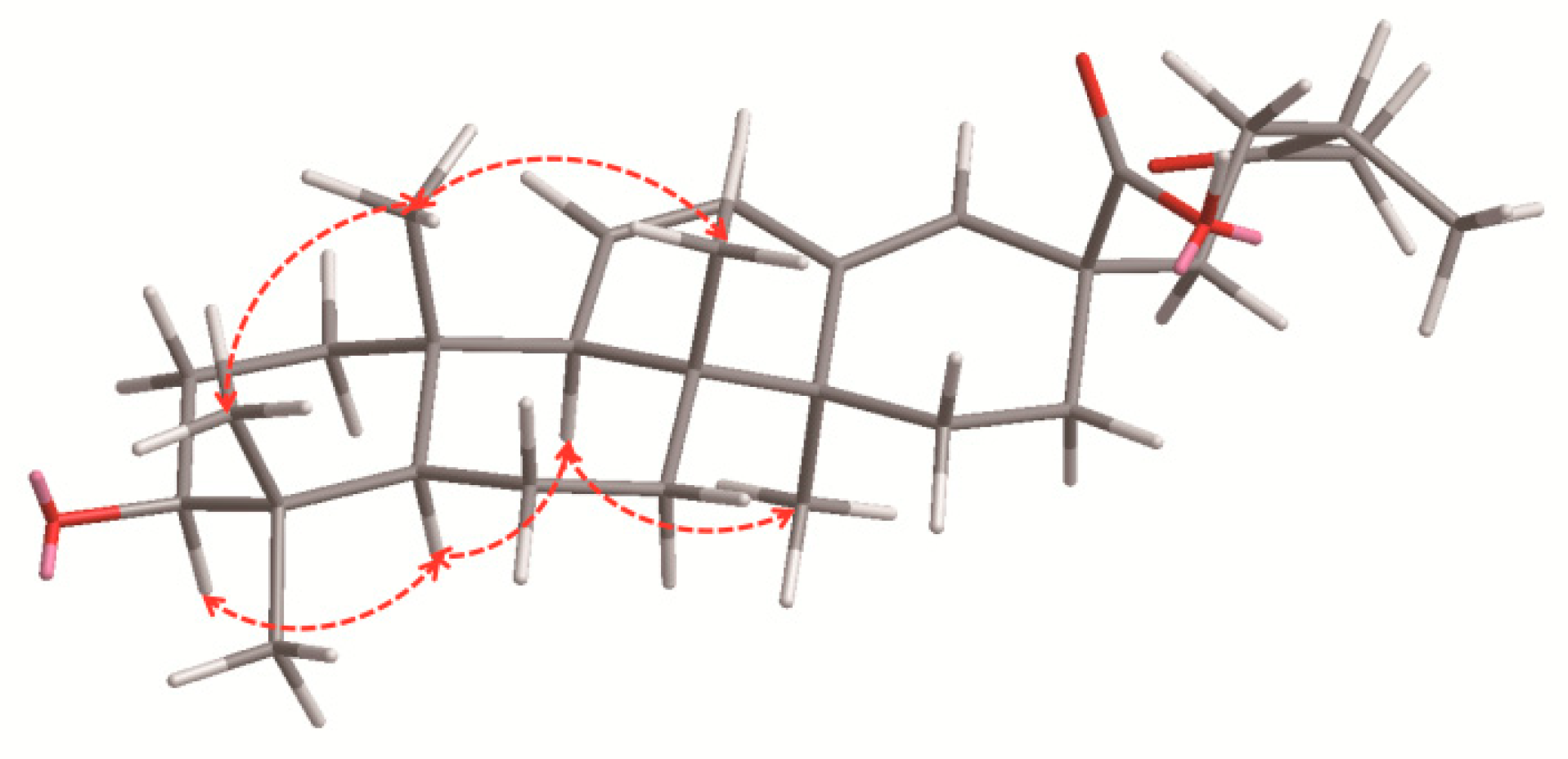
2.1. Characterization of the Compounds
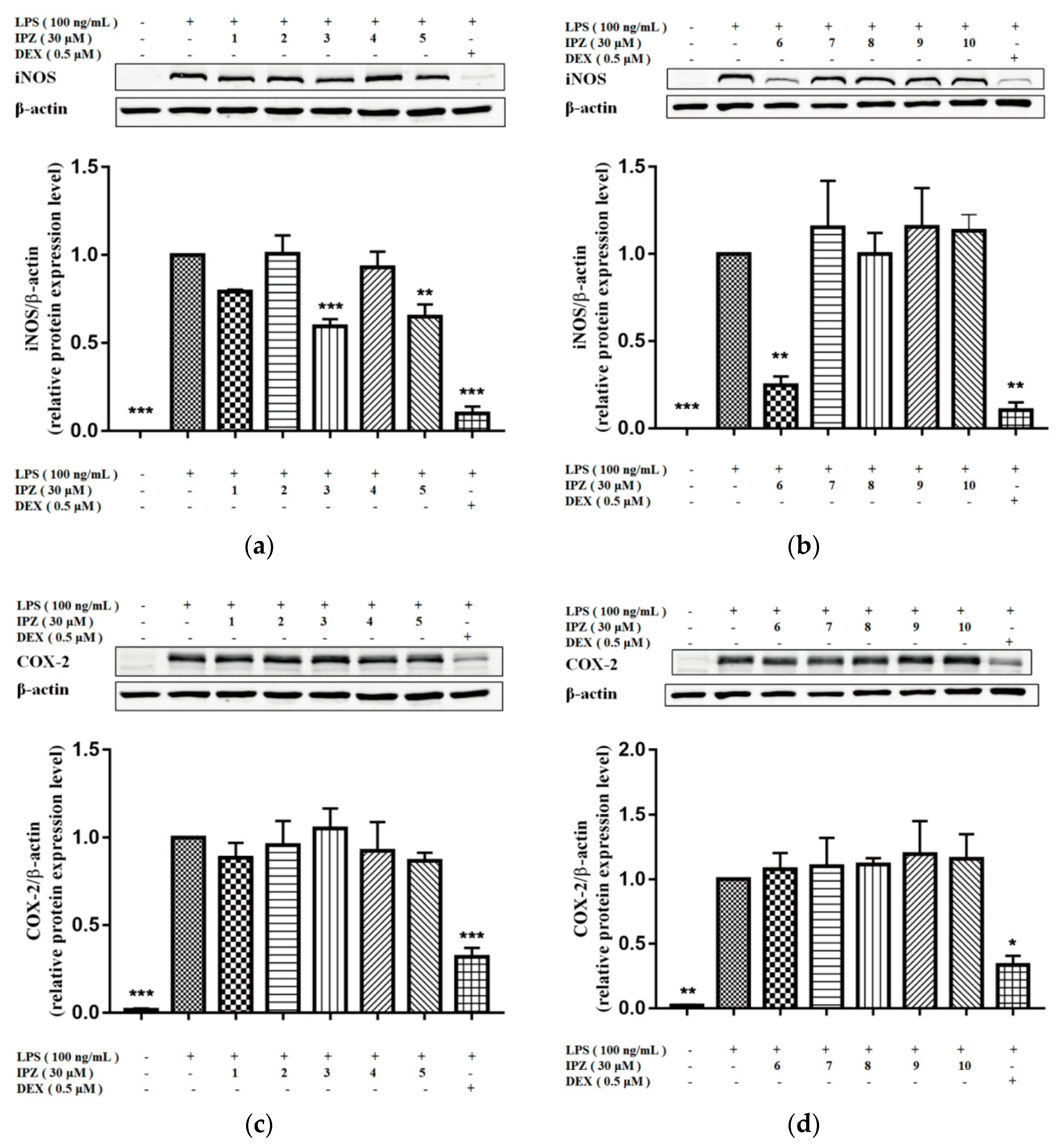
2.2. Anti-Inflammatory Activity
3. Discussion
4. Materials and Methods
4.1. General Experimental Procedures
4.2. Plant Material
4.3. Extraction and Isolation
4.3.1. Pubescenoside E (1)
4.3.2. Pubescenoside F (2)
4.3.3. Pubescenoside G (3)
4.3.4. Pubescenoside H (4)
4.3.5. Pubescenoside I (5)
4.3.6. Pubescenoside J (6)
4.3.7. Pubescenoside K (7)
4.4. Acid Hydrolysis
4.5. Cell Culture and Western Blot
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Lin, L.P.; Wei, Q.U.; Liang, J.Y. Chemical constituents from the stems of Ilex pubescens var. glabra. Chin. J. Nat. Med. 2011, 9, 176–179. [Google Scholar]
- Han, Y.N.; Bail, S.K.; Kim, T.H.; Han, B.H. New triterpenoid saponins from Ilex pubescens. Arch. Pharm. Res. 1987, 10, 132–141. [Google Scholar] [CrossRef]
- Jiangsu New Medical College. Directory of Chinese Material Medica, 1st ed.; Shanghai Scientific and Technological Press: Shanghai, China, 1997; p. 441. [Google Scholar]
- Zhou, Y.; Chai, X.Y.; Zeng, K.W.; Zhang, J.Y.; Li, N.; Jiang, Y.; Tu, P.F. Ilex publesnins C–M, eleven new triterpene saponins from the roots of Ilex pubescens. Planta Med. 2013, 79, 70–77. [Google Scholar] [PubMed]
- Xu, Z.R.; Chai, X.Y.; Bai, C.C.; Ren, H.Y.; Lu, Y.N.; Shi, H.M.; Tu, P.F. Xylocosides A–G, phenolic glucosides from the stems of xylosma controversum. Helv. Chim. Acta 2010, 91, 1346–1354. [Google Scholar] [CrossRef]
- Gohari, A.R.; Saeidnia, S.; Bayati-Moghadam, M.; Gh, A. Lignans and neolignans from Stelleropsis antoninae. DRAU 2011, 19, 74–79. [Google Scholar]
- Jiang, Z.H.; Wang, J.R.; Li, M.; Liu, Z.Q.; Chau, K.Y.; Zhao, C.; Liu, L. Hemiterpene glucosides with anti-platelet aggregation activities from Ilex pubescens. J. Nat. Prod. 2008, 68, 397–399. [Google Scholar] [CrossRef] [PubMed]
- Cao, L.H.; Zheng, Y.; Xin, W.Y.; Xu, K.; Miao, M.S. Study on intervention action of total flavonoids from Ilex Pubescens Radix on animal models of cerebral ischemic tolerance with blood stasis. China J. Chin. Mater. Med. 2016, 41, 3419–3424. [Google Scholar]
- Wang, J.R.; Zhou, H.; Jiang, Z.H.; Liu, L. Two new triterpene saponins from the anti-inflammatory saponin fraction of Ilex pubescens root. Chem. Biodivers. 2008, 5, 1369–1376. [Google Scholar] [CrossRef] [PubMed]
- Fabio, G.D.; Romanucci, V.; Marco, A.D.; Zarrelli, A. Triterpenoids from Gymnema sylvestre and their pharmacological activities. Molecules 2014, 19, 10956–10981. [Google Scholar] [CrossRef] [PubMed]
- Fabio, G.D.; Romanucci, V.; Zarrelli, M.; Giordano, M.; Zarrelli, A. C-4 gem-dimethylated oleanes of Gymnema sylvestre and their pharmacological activities. Molecules 2013, 18, 14892–14919. [Google Scholar] [CrossRef] [PubMed]
- Armo, Z.; Marina, D.G.; Afef, L.; Rabiaa, H.; Lucio, P. New triterpenes from Gymnema sylvestre. Helv. Chim. Acta 2013, 96, 1036–1045. [Google Scholar]
- Han, Y.N.; Song, J.I.; Rhee, I.K. Anticoagulant activity of ilexoside D, a triterpenoid saponin from Ilex pubescens. Arch. Pharm. Res. 1993, 16, 209–212. [Google Scholar] [CrossRef]
- Han, Y.N.; Baik, S.K.; Kim, T.H.; Han, B.H. Antithrombotic activities of saponins from Ilex pubescens. Arch. Pharm. Res. 1987, 10, 115–120. [Google Scholar] [CrossRef]
- Ryu, H.W.; Su, U.L.; Lee, S.; Song, H.H.; Son, T.H.; Kim, Y.U.; Yuk, H.J.; Ro, H.; Lee, C.K.; Hong, S.T. 3-Methoxy-catalposide inhibits inflammatory effects in lipopolysaccharide-stimulated RAW264.7 macrophages. Cytokine 2017, 91, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.S.; Li, J.J.; Cui, L.Y.; Wang, Y.F.; Lin, J.Q.; Qu, Y.; Wang, H. Cortisol modulates inflammatory responses in LPS-stimulated RAW264.7 cells via the NF-κB and MAPK pathways. BMC Vet. Res. 2018, 14, 30–40. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Wang, Q.L.; Hou, S.B.; Chen, G. Chemical constituents from the rhizomes of Polygonatum sibiricum Red. and anti-inflammatory activity in RAW264.7 macrophage cells. Nat. Prod. Res. 2018, 8, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Choy, C.S.; Hu, C.M.; Chiu, W.T.; Lam, C.S.; Ting, Y.; Tsai, S.H.; Wang, T.C. Suppression of lipopolysaccharide-induced of inducible nitric oxide synthase and cyclooxygenase-2 by Sanguis Draconis, a dragon’s blood resin, in RAW 264.7 cells. J. Ethnopharmacol. 2008, 115, 455–462. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.; Gao, H.; Liu, J.X.; Liu, L.; Zhou, H.; Liu, Z.Q. Triterpenoid saponins with anti-inflammatory activities from Ilex pubescens roots. Phytochemistry 2017, 134, 122–132. [Google Scholar] [CrossRef] [PubMed]
- Kakuno, T.; Yoshikawa, K.; Arihara, S. Ilexosides A, B, C and D, anti-allergic 18,19-seco-ursane glycosides from fruit of Ilex crenata. Tetrahedron Lett. 1991, 32, 3535–3538. [Google Scholar] [CrossRef]
- Abdel-Sattar, E. Saponin glycosides from Osteospermum vaillantii. Pharm. Biol. 2001, 39, 440–444. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds 1–10 are available from the authors. |
| Position | 1 a | 2 b | 3 b | 4 b | 5 b | 6 b | 7 b |
|---|---|---|---|---|---|---|---|
| 1 | 0.95 m; 1.77 m | 0.94 m;1.71 m | 0.95 m; 1.50 m | 0.91 m; 1.46 m | 0.97 m; 1.75 m | 0.98 m; 1.48 m | 0.90 m; 1.54 m |
| 2 | 1.68 m; 2.56 m | 1.75 m; 2.12 m | 1.88 m; 2.06 m | 1.86 m; 2.09 m | 1.96 m; 2.13 m | 1.89 m; 2.17 m | 1.93 m; 2.12 m |
| 3 | 3.30 dd (11.6, 3.7) | 3.28 dd (11.5, 4.1) | 3.28 dd (11.4, 3.6) | 3.27 dd (11.4, 4.1) | 3.27 dd (10.7, 3.1) | 3.40 dd (11.5, 3.8) | 3.29 dd (10.8, 2.5) |
| 5 | 0.83 m | 0.79 m | 0.80 m | 0.76 m | 0.76 m | 0.81 m | 0.80 m |
| 6 | 1.33 m; 1.54 m | 1.27 m; 1.52 m | 1.30 m; 1.49 m | 1.25 m; 1.44 m | 1.26 m; 1.47 m | 1.27 m; 1.46 m | 1.27 m; 1.45 m |
| 7 | 1.33 m | 1.26 m | 1.46 m | 1.31 m; 1.43 m | 1.16 m | 1.37 m; 1.46 m | 1.40 m; 1.54 m |
| 9 | 2.04 m | 1.98 m | 1.63 m | 1.61 m | - | 1.76 m | 1.78 m |
| 11 | 5.62 d (10.2) | 5.59 d (9.8) | 1.93 m | 1.87 m | 6.58 d (10.1) | 1.91 m; 1.98 m | 2.0 m; 2.06 m |
| 12 | 6.18 d (8.5) | 6.03 d (10.1) | 5.42 s | 5.45 s | 5.61 d (10.6) | 5.48 s | 5.57 s |
| 15 | 1.24 m; 1.92 m | 1.16 m; 1.90 m | 1.17 m; 2.33 m | 1.15 m; 2.32 m | 0.88 m; 1.93 m | 1.17 m; 2.35 m | 1.26 m; 2.44 m |
| 16 | 1.23 m; 2.15 m | 1.30 m; 2.13 m | 1.17 m; 1.26 m | 1.99 m; 2.16 m | 1.64 m; 2.15 m | 1.85 m; 1.99 m | 3.12 m |
| 18 | 5.88s | 5.72 s | 3.19 m | 3.28 m | 1.92 m | 3.99 s | 2.92 m |
| 19 | - | - | 1.25 m; 1.75 m | 1.40 m; 2.11 m | 2.30 m | 2.59 m | - |
| 20 | 2.53 m | 2.50 m | - | - | 1.44 m | - | 1.36 m |
| 21 | 1.33 m; 1.66 m | 1.32 m; 1.59 m | 1.08 m; 1.33 m | 1.24 m; 1.74 m | 1.50 m; 1.58 m | 1.28 m; 1.76 m | 2.04 m; 2.12 m |
| 22 | 1.99 m | 1.95 m | 1.75 m; 1.97 m | 1.73 m; 1.84 m | 1.28 m; 2.62 m | 1.76 m; 2.08 m | 1.79 m; 2.04 m |
| 23 | 1.34 s | 1.32 s | 1.33 s | 1.32 s | 1.27 s | 1.31 s | 1.42 s |
| 24 | 1.06 s | 1.03 s | 1.07 s | 1.08 s | 1.04 s | 0.99 s | 1.18 s |
| 25 | 0.82 s | 0.76 s | 0.82 s | 0.83 s | 0.81 s | 0.88 s | 0.89 s |
| 26 | 0.85 s | 0.82 s | 1.07 s | 1.09 s | 0.98 s | 1.09 s | 1.14 s |
| 27 | 1.06 s | 1.03 s | 1.24 s | 1.25 s | 0.99 s | 1.27 s | 1.73 s |
| 29 | 2.11 s | 2.13 s | 0.91 s | 3.56 m | 1.32 d (4.0) | 1.06 d (6.7) | 1.42 s |
| 30 | 1.08 d (8.7) | 1.03 d (7.1) | 0.87 s | 1.08 s | 0.76 d (5.9) | 5.04 c | 1.11 d (6.2) |
| 3-O- | Xyl | Xyl | Xyl | Xyl | Xyl | Xyl | 2-sulfo-Xyl |
| 1 | 4.92 d (5.2) | 4.89 d (6.7) | 4.88 d (6.6) | 4.88 d (6.6) | 4.84 d (6.1) | 4.85 d (7.4) | 4.97 d (6.6) |
| 2 | 4.42 e | 4.01 c | 4.29 d | 4.28 d | 4.22 d | 4.03 m | 5.05 m |
| 3 | 3.87 m | 3.87 m | 3.88 m | 3.86 m | 3.84 m | 4.17 m | 4.43 m |
| 4 | 4.08 c | 4.06 c | 4.08 c | 4.06 c | 4.02 c | 4.21 m | 4.21 m |
| 5 | 3.73 m; 4.28 d | 3.72 m; 4.28 d | 3.72 m; 4.28 d | 3.72 m; 4.27 d | 3.68 m; 4.25 d | 3.78 m; 4.38 m | 3.74 m; 4.21 m |
| Intermediate | Glc | Glc | Glc | Glc | Glc | ||
| 1 | 5.79 d (5.8) | 5.80 d (7.3) | 5.81 d (7.8) | 5.82 d (7.2) | 5.71 d (6.8) | ||
| 2 | 4.24 d | 4.27 d | 4.24 d | 4.22 d | 4.22 d | ||
| 3 | 4.48 e | 4.44 e | 4.44 c | 4.41 e | 4.39 e | ||
| 4 | 4.07 c | 4.06 c | 4.04 c | 4.03 c | 3.83 m | ||
| 5 | 4.28 d | 4.27 d | 4.43 e | 4.29 d | 4.39 e | ||
| 6 | 4.27 d; 4.50 e | 4.28 d; 4.51 e | 4.28 d; 4.51 e | 4.27 d; 4.48 e | 4.21 d; 4.48 e | ||
| Terminal | Rha | Rha | Rha | Rha | Rha | ||
| 1 | 6.39 br s | 6.38 br s | 6.39 br s | 6.40 br s | 6.30 br s | ||
| 2 | 4.71 m | 4.71 m | 4.75 m | 4.76 m | 4.69 s | ||
| 3 | 4.04 c | 4.05 c | 4.04 c | 4.04 c | 3.95 c | ||
| 4 | 4.33 d | 4.34 d | 4.34 e | 4.23 d | 4.29 d | ||
| 5 | 5.03 m | 5.04 m | 5.04 m | 5.04 m | 4.98 m | ||
| 6 | 1.81 f | 1.78 f | 1.79 d (6.0) | 1.8 d (6.0) | 1.75 d (5.5) | ||
| 28-O- | Glc | Glc | Glc | Glc | Glc | Glc | |
| 1 | 6.32 d (8.2) | 6.32 d (8.4) | 6.35 d (7.8) | 6.23 d (7.8) | 6.34 d (8.1) | 6.25 d (7.8) | |
| 2 | 4.48 e | 4.21 d | 4.33 e | 4.13 d | 4.23 m | 4.24 m | |
| 3 | 4.44 e | 4.29 d | 4.41 e | 4.39 e | 4.04 m | 4.33 c | |
| 4 | 4.29 d | 4.35 e | 4.35 e | 4.23 d | 4.37 m | 4.33 c | |
| 5 | 4.44 e | 4.29 d | 4.28 d | 4.22 d | 4.30 m | 4.08 m | |
| 6 | 4.29 d | 4.43 e | 4.39 e; 4.45 e | 4.27 d; 4.39 e | 4.38 m; 4.47 m | 4.39 m; 4.50 m |
| Position | 1 a | 2 b | 3 b | 4 b | 5 b | 6 b | 7 b |
|---|---|---|---|---|---|---|---|
| 1 | 38.6 | 38.5 | 39.1 | 39.1 | 38.3 | 39.1 | 38.8 |
| 2 | 27.9 | 26.6 | 26.8 | 26.8 | 26.6 | 27.1 | 26.5 |
| 3 | 89.9 | 89.8 | 89.9 | 89.9 | 89.9 | 89.0 | 89.7 |
| 4 | 40.1 | 40.1 | 40.0 | 40.0 | 39.9 | 39.9 | 39.6 |
| 5 | 55.7 | 55.7 | 56.2 | 56.2 | 55.5 | 56.4 | 55.8 |
| 6 | 18.6 | 18.5 | 18.8 | 18.8 | 18.6 | 18.9 | 18.6 |
| 7 | 32.8 | 32.6 | 32.8 | 33.4 | 32.8 | 33.7 | 33.4 |
| 8 | 41.1 | 41.1 | 40.1 | 40.2 | 43.4 | 40.1 | 40.5 |
| 9 | 54.8 | 54.8 | 48.3 | 48.3 | 134.6 | 48.4 | 47.6 |
| 10 | 37.0 | 36.9 | 37.2 | 37.3 | 36.7 | 37.4 | 36.9 |
| 11 | 127.7 | 128.4 | 24.1 | 24.1 | 128.6 | 24.2 | 24.0 |
| 12 | 130.8 | 130.4 | 123.1 | 123.1 | 125.8 | 127.9 | 128.4 |
| 13 | 142.5 | 143.9 | 144.4 | 144.6 | 138.6 | 137.7 | 139.3 |
| 14 | 41.8 | 41.7 | 42.4 | 42.4 | 41.6 | 43.1 | 42.0 |
| 15 | 26.8 | 26.7 | 28.5 | 28.6 | 25.4 | 28.7 | 29.2 |
| 16 | 27.0 | 27.9 | 23.7 | 23.7 | 33.4 | 26.6 | 26.0 |
| 17 | 48.0 | 48.0 | 47.3 | 47.7 | 51.8 | 49.6 | 48.6 |
| 18 | 129.6 | 127.0 | 42.0 | 41.5 | 55.4 | 47.5 | 54.4 |
| 19 | 212 | 212.3 | 46.5 | 41.2 | 45.0 | 37.6 | 72.6 |
| 20 | 47.8 | 47.6 | 31.0 | 36.7 | 39.7 | 153.6 | 42.1 |
| 21 | 28.5 | 28.0 | 34.2 | 29.1 | 33.6 | 28.2 | 26.6 |
| 22 | 39.3 | 38.9 | 33.4 | 32.3 | 40.2 | 31.9 | 37.7 |
| 23 | 28.4 | 28.4 | 28.6 | 28.6 | 28.1 | 28.5 | 28.3 |
| 24 | 16.5 | 16.4 | 17.0 | 17.0 | 16.2 | 17.3 | 16.9 |
| 25 | 18.3 | 18.3 | 15.8 | 15.8 | 18.4 | 16.1 | 15.6 |
| 26 | 17.1 | 16.9 | 17.7 | 17.8 | 16.9 | 17.6 | 17.3 |
| 27 | 20.4 | 20.2 | 26.3 | 26.3 | 19.3 | 26.0 | 24.6 |
| 28 | 178.9 | 175.1 | 176.8 | 176.8 | 176.5 | 176.5 | 177.3 |
| 29 | 28.2 | 28.3 | 33.4 | 74.0 | 21.1 | 21.1 | 27.0 |
| 30 | 16.6 | 16.5 | 23.9 | 20.0 | 20.5 | 113.1 | 16.7 |
| 3-O- | Xyl | Xyl | Xyl | Xyl | Xyl | Xyl | 2-sulfo-Xyl |
| 1 | 106.2 | 106.2 | 106.1 | 106.1 | 106.1 | 108 | 104.8 |
| 2 | 79.3 | 79.7 | 79.7 | 79.7 | 79.0 | 75.9 | 80.2 |
| 3 | 78.1 | 78.2 | 78.2 | 78.2 | 78.0 | 79.0 | 77.2 |
| 4 | 71.6 | 71.6 | 71.6 | 71.6 | 71.4 | 71.6 | 70.7 |
| 5 | 67.0 | 66.9 | 66.9 | 67.0 | 66.8 | 67.4 | 65.9 |
| Intermediate | Glc | Glc | Glc | Glc | Glc | ||
| 1 | 102.6 | 102.5 | 102.5 | 102.5 | 102.2 | ||
| 2 | 79.7 | 79.6 | 79.6 | 79.6 | 79.5 | ||
| 3 | 79.4 | 79.3 | 79.4 | 79.4 | 79.1 | ||
| 4 | 72.7 | 72.9 | 72.9 | 73.0 | 72.4 | ||
| 5 | 78.9 | 79.0 | 79.2 | 79.3 | 78.7 | ||
| 6 | 63.6 | 63.6 | 63.6 | 63.6 | 63.4 | ||
| Terminal | Rha | Rha | Rha | Rha | Rha | ||
| 1 | 102.3 | 102.4 | 102.3 | 102.3 | 102.1 | ||
| 2 | 72.9 | 72.7 | 72.7 | 72.7 | 72.6 | ||
| 3 | 73.0 | 72.9 | 72.9 | 72.9 | 72.7 | ||
| 4 | 74.6 | 74.6 | 74.6 | 74.4 | 74.3 | ||
| 5 | 69.8 | 69.8 | 69.7 | 69.8 | 69.5 | ||
| 6 | 19.2 | 19.2 | 19.2 | 19.3 | 19.0 | ||
| 28-O- | Glc | Glc | Glc | Glc | Glc | Glc | |
| 1 | 95.6 | 96.0 | 96.0 | 96.2 | 96.3 | 95.8 | |
| 2 | 74.5 | 74.4 | 74.7 | 74.3 | 74.4 | 73.9 | |
| 3 | 78.9 | 79.1 | 79.2 | 79.4 | 79.7 | 78.7 | |
| 4 | 71.5 | 71.3 | 71.4 | 71.3 | 71.4 | 71.2 | |
| 5 | 78.9 | 78.8 | 78.8 | 78.8 | 79.2 | 79.1 | |
| 6 | 62.6 | 62.4 | 62.5 | 62.3 | 62.5 | 62.3 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qiao, X.; Ji, M.; Yao, Y.; Ma, L.; Wu, J.; Liao, G.; Zhou, H.; Liu, Z.; Wu, P. Pubescenosides E–K, Seven New Triterpenoid Saponins from the Roots of Ilex pubescens and Their Anti-Inflammatory Activity. Molecules 2018, 23, 1426. https://doi.org/10.3390/molecules23061426
Qiao X, Ji M, Yao Y, Ma L, Wu J, Liao G, Zhou H, Liu Z, Wu P. Pubescenosides E–K, Seven New Triterpenoid Saponins from the Roots of Ilex pubescens and Their Anti-Inflammatory Activity. Molecules. 2018; 23(6):1426. https://doi.org/10.3390/molecules23061426
Chicago/Turabian StyleQiao, Xiaoxu, Mengying Ji, Yunda Yao, Leilei Ma, Jinjun Wu, Guochao Liao, Hua Zhou, Zhongqiu Liu, and Peng Wu. 2018. "Pubescenosides E–K, Seven New Triterpenoid Saponins from the Roots of Ilex pubescens and Their Anti-Inflammatory Activity" Molecules 23, no. 6: 1426. https://doi.org/10.3390/molecules23061426
APA StyleQiao, X., Ji, M., Yao, Y., Ma, L., Wu, J., Liao, G., Zhou, H., Liu, Z., & Wu, P. (2018). Pubescenosides E–K, Seven New Triterpenoid Saponins from the Roots of Ilex pubescens and Their Anti-Inflammatory Activity. Molecules, 23(6), 1426. https://doi.org/10.3390/molecules23061426






