In Vitro Antioxidant Activities of Phenols and Oleanolic Acid from Mango Peel and Their Cytotoxic Effect on A549 Cell Line
Abstract
1. Introduction
2. Results and Discussion
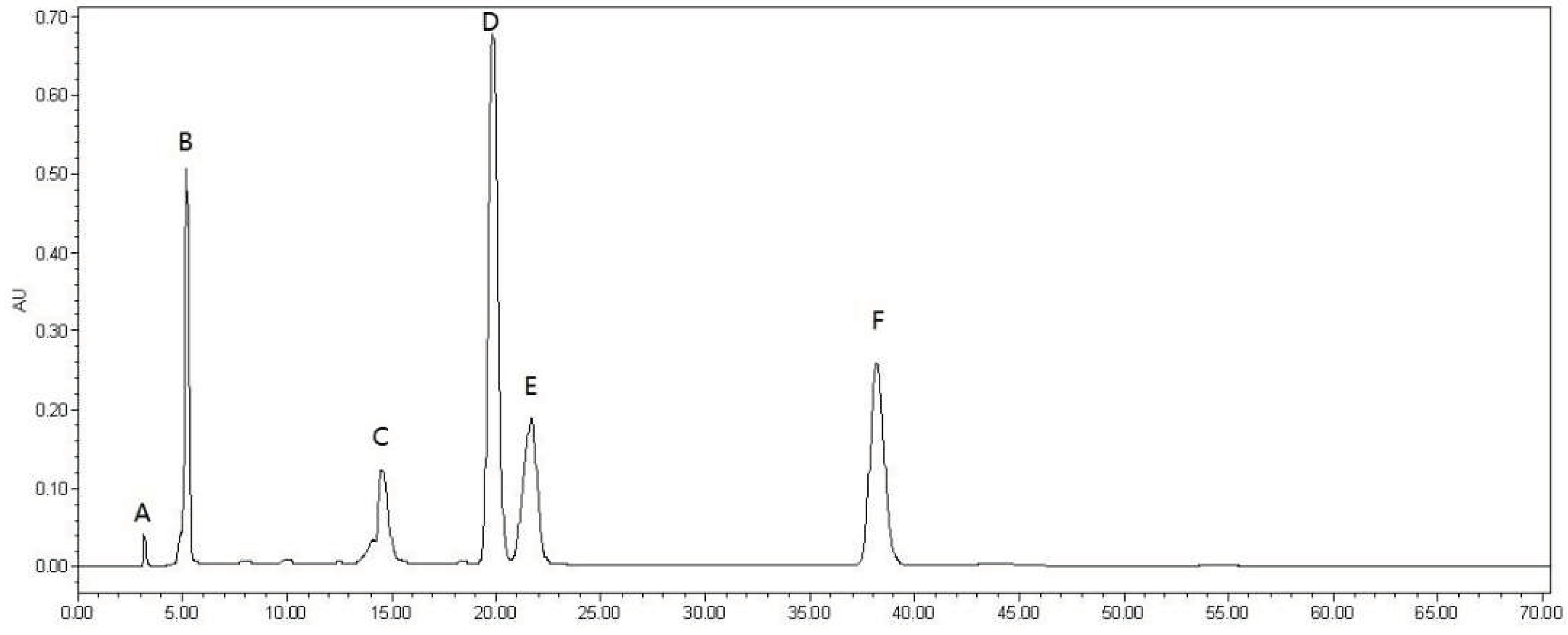
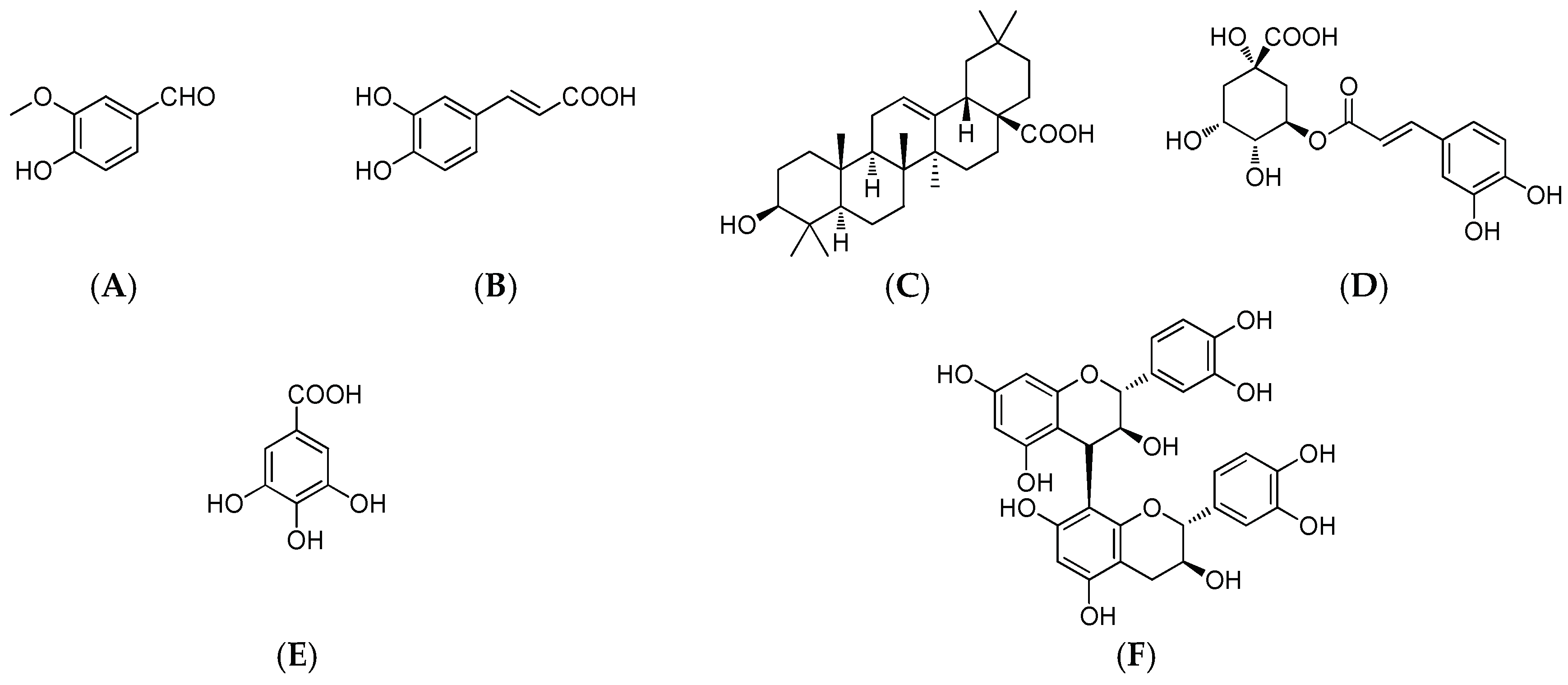
2.1. Total Phenolic Content and HPLC Analysis
2.2. Antioxidant Activity of MPE and MPPs
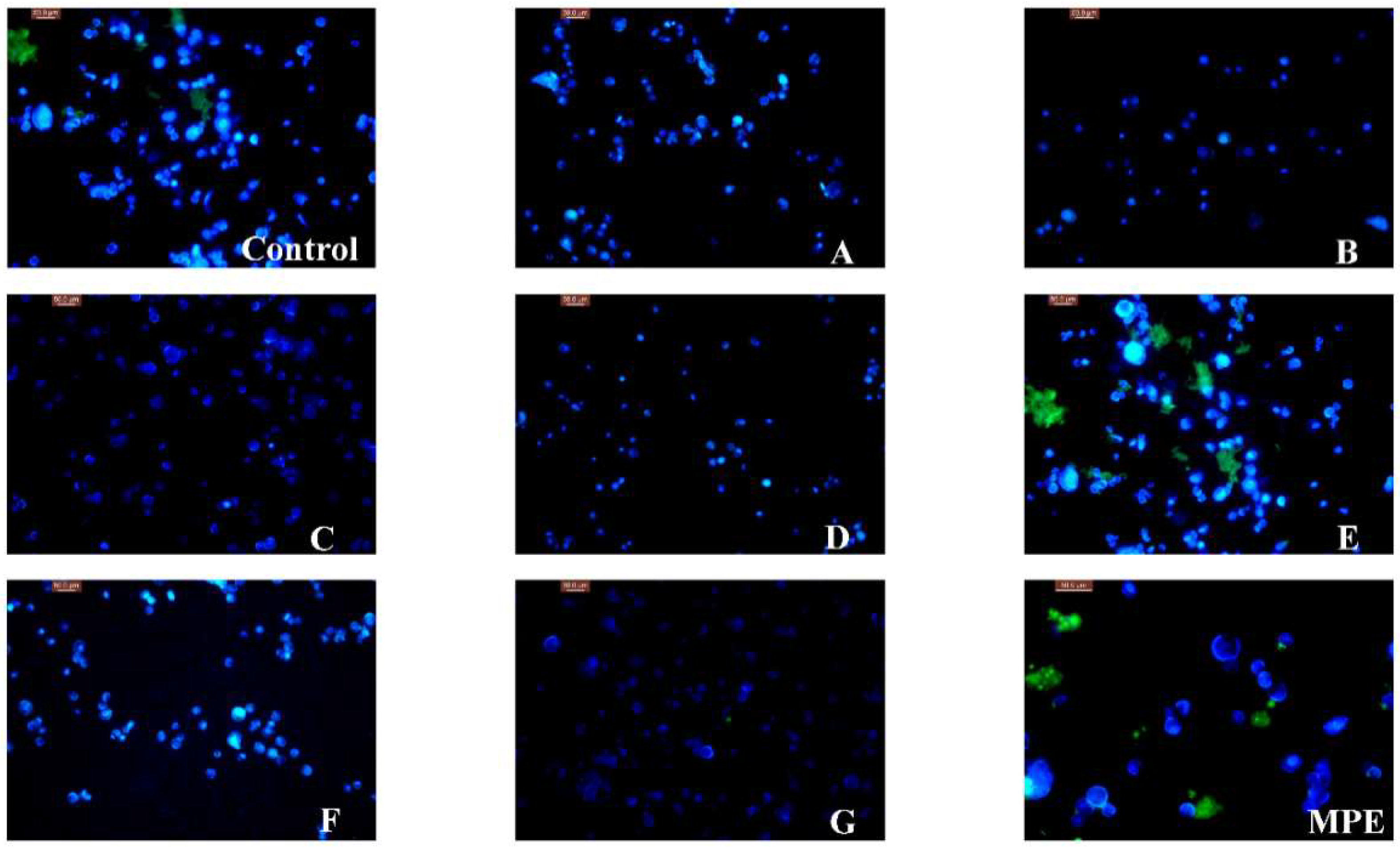
2.3. Cytotoxic Effect of MPE and MPPs
3. Experimental Section
3.1. Determination of Total Phenolic Content
3.2. HPLC Analysis of MPPs and Oleanolic Acid
3.3. Antioxidant Assay of MPPs
3.3.1. DPPH Assay
3.3.2. ABTS Assay
3.3.3. FRAP Assay
3.4. Cytotoxic Effect of MPPs and Oleanolic Acid on the A549 Cell Line
3.4.1. Inhibitory Activity
3.4.2. Morphological Observation of the Nucleus of Lung Cancer A549 Cells
3.5. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kaur, B.; Srivastav, P.P. Effect of cryogenic grinding on chemical and morphological characteristics of mango (Mangifera indica L.) peel powder. J. Food Process. Pres. 2018, 42, e13583. [Google Scholar] [CrossRef]
- Ndoye, S.F.; Fraisse, D.; Akendengue, B.; Dioum, M.D.; Gueye, R.S.; Sall, C.; Seck, I.; Felgines, C.; Seck, M.; Senejoux, F. Antioxidant and antiglycation properties of two mango (Mangifera indica L.) cultivars from Senegal. Asian Pac. J. Trop. Biomed. 2018, 3, 137–141. [Google Scholar]
- Fernandez-Ponce, M.T.; Casas, L.; Mantell, C.; De la Ossa, E.M. Use of high pressure techniques to produce Mangifera indica L. leaf extracts enriched in potent antioxidant phenolic compounds. Innov. Food Sci. Emerg. 2015, 29, 94–106. [Google Scholar] [CrossRef]
- Pierson, J.T.; Monteith, G.R.; Roberts-Thomson, S.J.; Dietzgen, R.G.; Gidley, M.J.; Shaw, P.N. Phytochemical extraction, characterisation and comparative distribution across four mango (Mangifera indica L.) fruit varieties. Food Chem. 2014, 149, 253–263. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Wang, C.T.; Shen, L.Q.; Shin, H.C.; Lee, K.B.; Ji, B.P. Comparative analysis of oxidative mechanisms of phloroglucinol and dieckol by electrochemical, spectroscopic, cellular and computational methods. RSC Adv. 2018, 8, 1963–1972. [Google Scholar] [CrossRef]
- Sui, X.N.; Sun, H.N.; Qi, B.K.; Zhang, M.; Li, Y.; Jiang, L.Z. Functional and conformational changes to soy proteins accompanying anthocyanins: Focus on covalent and non-covalent interactions. Food Chem. 2018, 245, 871–878. [Google Scholar] [CrossRef] [PubMed]
- Pacheco-Ordaz, R.; Antunes-Ricardo, M.; Gutierrez-Uribe, J.A.; Gonzalez-Aguilar, G.A. Intestinal permeability and cellular antioxidant activity of phenolic compounds from mango (Mangifera indica cv. Ataulfo) peels. Int. J. Mol. Sci. 2018, 19, 514. [Google Scholar] [CrossRef] [PubMed]
- Suvarnakuta, P.; Chaweerungrat, C.; Devahastin, S. Effects of drying methods on assay and antioxidant activity of xanthones in mangosteen rind. Food Chem. 2011, 125, 240–247. [Google Scholar] [CrossRef]
- Hu, K.; Dars, A.G.; Liu, Q.D.; Xie, B.J.; Sun, Z.D. Phytochemical profiling of the ripening of Chinese mango (Mangifera indica L.) cultivars by real-time monitoring using UPLC-ESI-QTOF-MS and its potential benefits as prebiotic ingredients. Food Chem. 2018, 256, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Yen, G.C.; Duh, P.D.; Tsai, H.L. Antioxidant and pro-oxidant properities of ascorbic acid and gallic acid. Food Chem. 2002, 79, 307–313. [Google Scholar] [CrossRef]
- Wijngaard, H.H.; Brunton, N. The optimisation of solid–liquid extraction of antioxidants from apple pomace by response surface methodology. J. Food Eng. 2010, 96, 134–140. [Google Scholar] [CrossRef]
- Bai, X.L.; Yue, T.L.; Yuan, Y.H.; Zhang, H.W. Optimization of microwave-assisted extraction of polyphenols from apple pomace. J. Sep. Sci. 2010, 33, 3571–3578. [Google Scholar] [CrossRef] [PubMed]
- Lily, Z.P.A.; Rokiah, H.; Shaida, F.S.; Ahmed, Y.C.; Othman, S.; Fumio, K.; Kushairi, M.S. In vitro antioxidant and antidiabetic activites of Gluta torquata. Ind. Crop Prod. 2015, 76, 755–760. [Google Scholar]
- Bao, Y.T.; Qu, Y.; Li, J.H.; Li, Y.F.; Ren, X.D.; Maffucci, K.G.; Li, R.P.; Wang, Z.G.; Zeng, R. In vitro and in vivo antioxidant activities of the flowers and leaves from Paeonia rockii and identification of their antioxidant constituents by UHPLC-ESI-HRMSn via pre-column DPPH reaction. Molecules 2018, 23, 392. [Google Scholar] [CrossRef] [PubMed]
- Iris, F.F.B.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar]
- Huang, W.S.; Mao, S.Q.; Zhang, L.Q.; Lu, B.Y.; Zheng, L.F.; Zhou, F.; Zhao, Y.J.; Li, M.Q. Phenolic compounds, antioxidant potential and antiproliferative potential of 10 common edible flowers from China assessed using a simulated in vitro digestion-dialysis process combined with cellular assays. J. Sci. Food Agric. 2017, 97, 4760–4769. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.F.; Zhou, L.; Jin, W.D.; Yang, W.J.; Wang, Y.; Yan, B.; Du, W.L.; Zhang, Q.; Zhang, L.; Guo, Y.H. Anti-proliferative and apoptosis-inducing effect of theabrownin against non-small cell lung adenocarcinoma A549 cells. Front. Pharmacol. 2016, 7, 465. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Li, J.Z.; Lu, A.X.; Zhang, K.F.; Li, B.J. Anticancer effect of salidroside on A549 lung cancer cells through inhibition of oxidative stress and phospho-p38 expression. Oncol. Lett. 2014, 7, 1159–1164. [Google Scholar] [CrossRef] [PubMed]
- Ramalingam, V.; Rajaram, R. Enhanced antimicrobial, antioxidant and anticancer activity of Rhizophora apiculata: An experimental report. Biotech 2018, 8, 200. [Google Scholar] [CrossRef] [PubMed]
- Maciela, L.G.; do Carmo, M.A.V.; Azevedo, L.; Daguer, H.; Molognoni, L.; De Almeida, M.M.; Granato, D.; Rosso, N.D. Hibiscus sabdariffa anthocyanins-rich extract: Chemical stability, in vitro antioxidant and antiproliferative activities. Food Chem. Toxicol. 2018, 113, 187–197. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds (vanillic aldehyde, caffeic acid, chlorogenic acid, gallic acid, procyanidin B2 and oleanolic acid) are available from the authors. |
| Phenol | Antioxidant Activity | ||
|---|---|---|---|
| DPPH• Scavenging Rate (%) | ABTS• + Inhibition Rate (%) | FRAP Assay (TE, µmol·kg−1) | |
| A | 60 ± 1.0 | 80 ± 3.2 | 8.0 ± 0.96 |
| B | 29 ± 1.0 | 55 ± 4.5 | 6.0 ± 0.65 |
| C | 2.7 ± 0.17 | 11 ± 1.5 | 1.2 ± 0.09 |
| D | 80 ± 1.2 | 76 ± 3.6 | 8.0 ± 0.25 |
| E | 97 ± 0.4 | 86 ± 3.0 | 12 ± 0.52 |
| F | 78 ± 1.2 | 76 ± 2.4 | 8.0 ± 0.22 |
| MPE * | 92 ± 4.2 | 79 ± 2.5 | 4.7 ± 0.53 |
| A | B | C | D | E | F | G | MPE | |
|---|---|---|---|---|---|---|---|---|
| IC50 value (μM) | 7.2 | 8.9 | 4.7 | 9.8 | 51.8 | 14.3 | 3.8 | 15 mg·mL−1 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bai, X.; Lai, T.; Zhou, T.; Li, Y.; Li, X.; Zhang, H. In Vitro Antioxidant Activities of Phenols and Oleanolic Acid from Mango Peel and Their Cytotoxic Effect on A549 Cell Line. Molecules 2018, 23, 1395. https://doi.org/10.3390/molecules23061395
Bai X, Lai T, Zhou T, Li Y, Li X, Zhang H. In Vitro Antioxidant Activities of Phenols and Oleanolic Acid from Mango Peel and Their Cytotoxic Effect on A549 Cell Line. Molecules. 2018; 23(6):1395. https://doi.org/10.3390/molecules23061395
Chicago/Turabian StyleBai, Xuelian, Tongfei Lai, Ting Zhou, Yicong Li, Xiuting Li, and Huawei Zhang. 2018. "In Vitro Antioxidant Activities of Phenols and Oleanolic Acid from Mango Peel and Their Cytotoxic Effect on A549 Cell Line" Molecules 23, no. 6: 1395. https://doi.org/10.3390/molecules23061395
APA StyleBai, X., Lai, T., Zhou, T., Li, Y., Li, X., & Zhang, H. (2018). In Vitro Antioxidant Activities of Phenols and Oleanolic Acid from Mango Peel and Their Cytotoxic Effect on A549 Cell Line. Molecules, 23(6), 1395. https://doi.org/10.3390/molecules23061395







