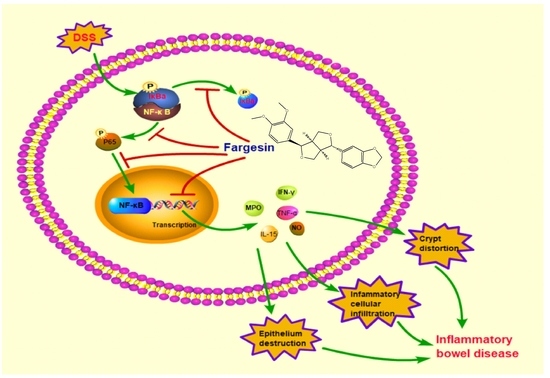
Anti-Inflammatory Effects of Fargesin on Chemically Induced Inflammatory Bowel Disease in Mice
Abstract
1. Introduction
2. Results
2.1. In Vivo Study
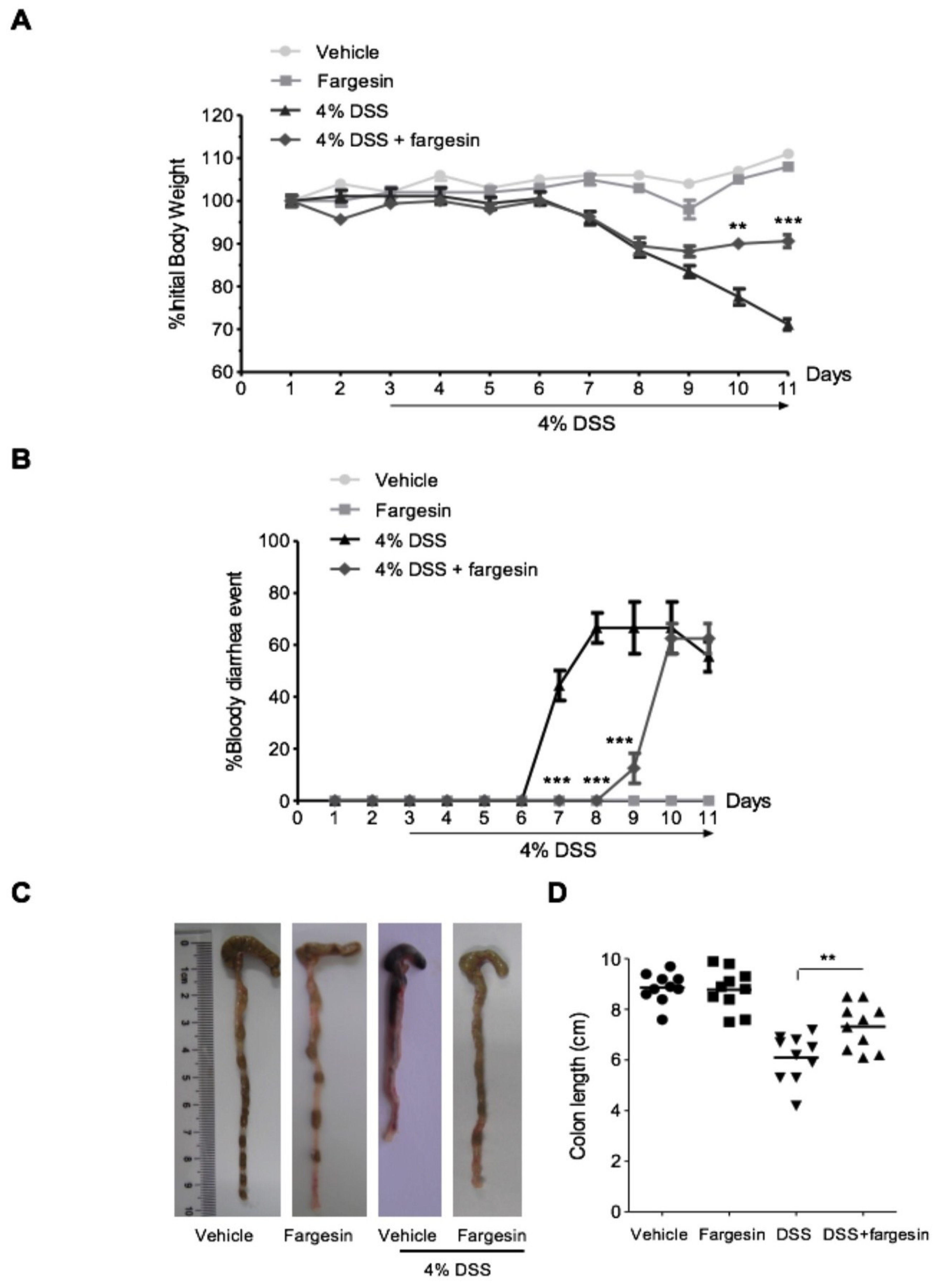
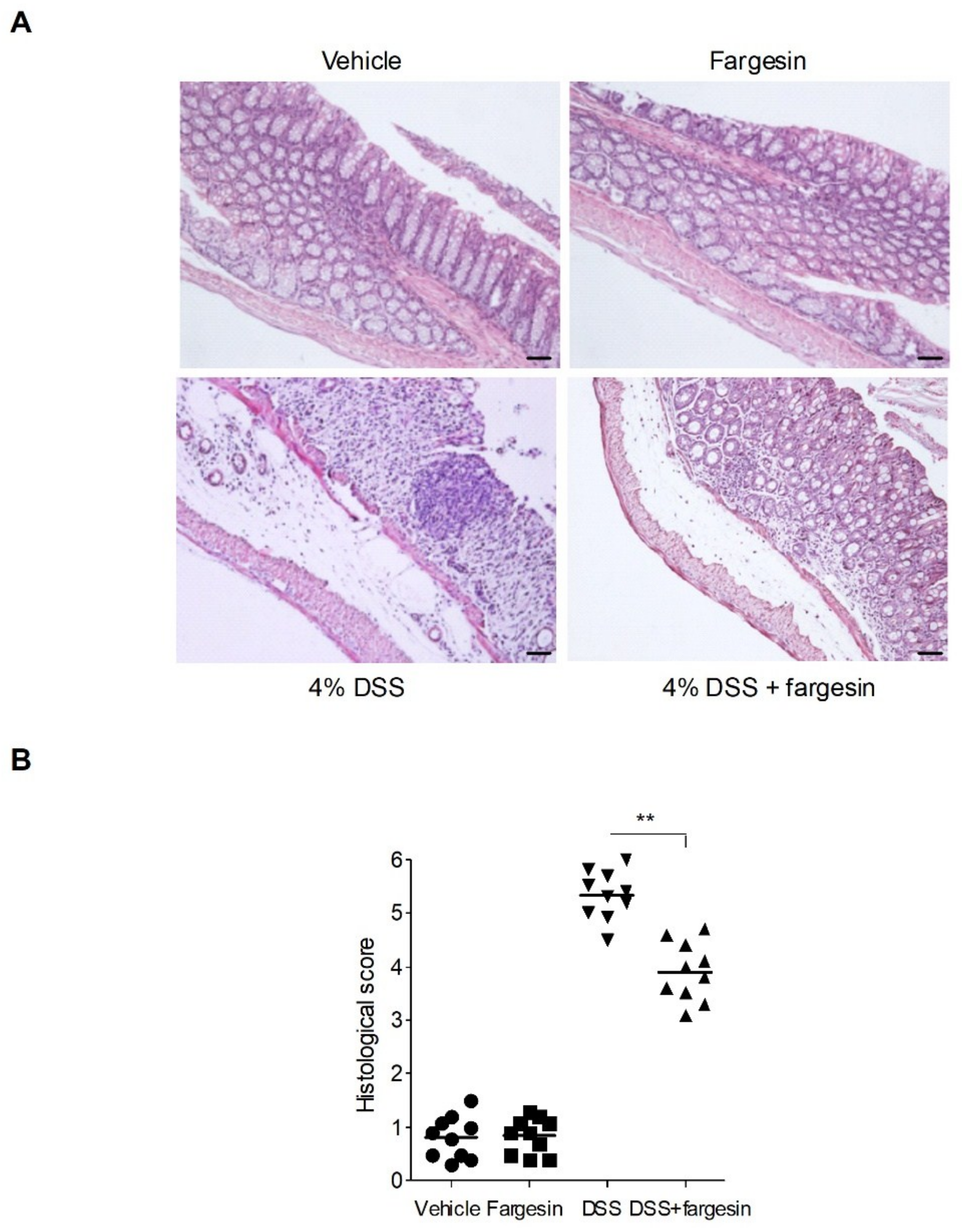
2.1.1. Effects of Fargesin on Dextran Sulfate Sodium (DSS)-Induced Colitis
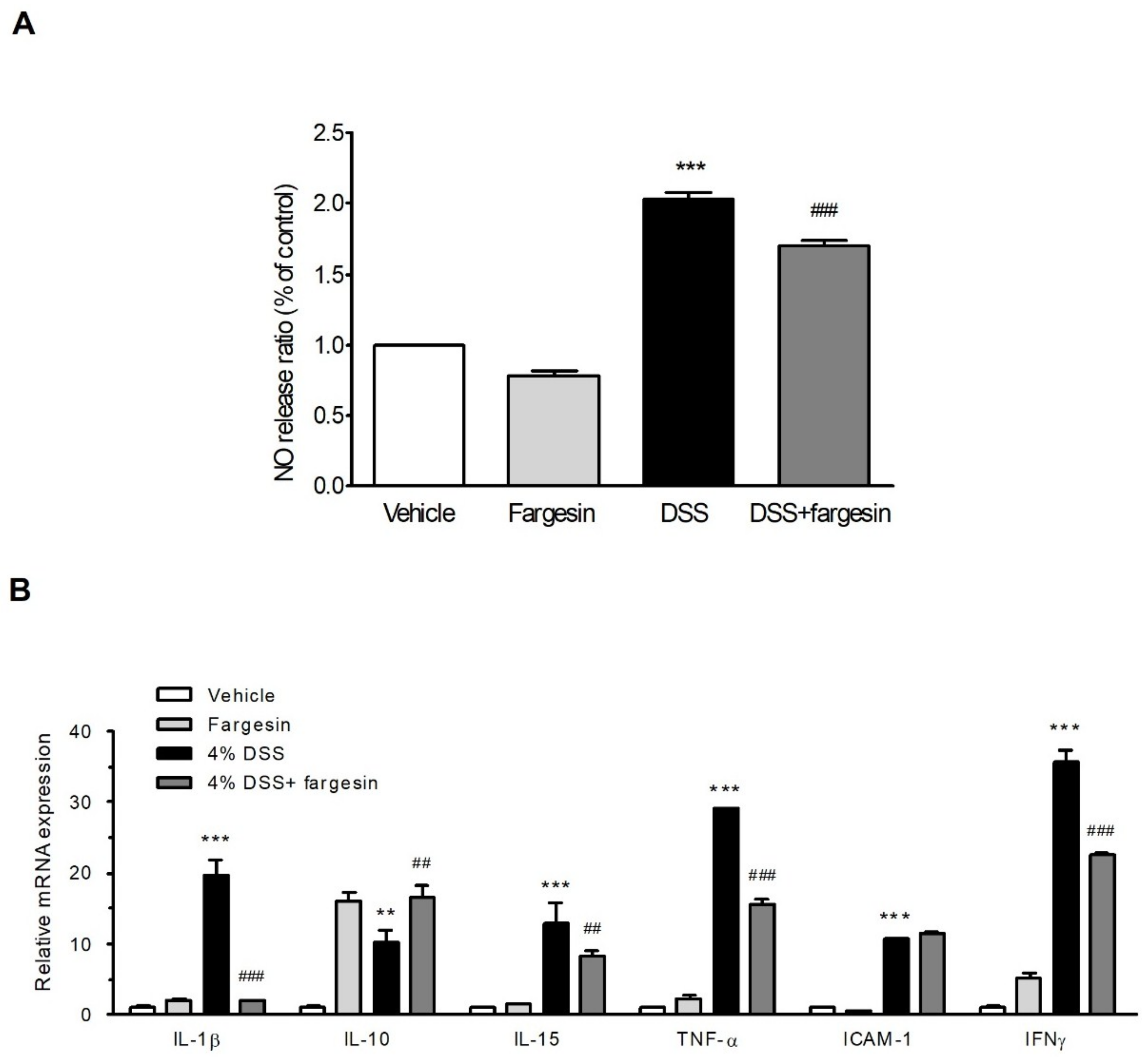
2.1.2. Effects of Fargesin on the Activity of MPO and the Level of Inflammatory Mediators
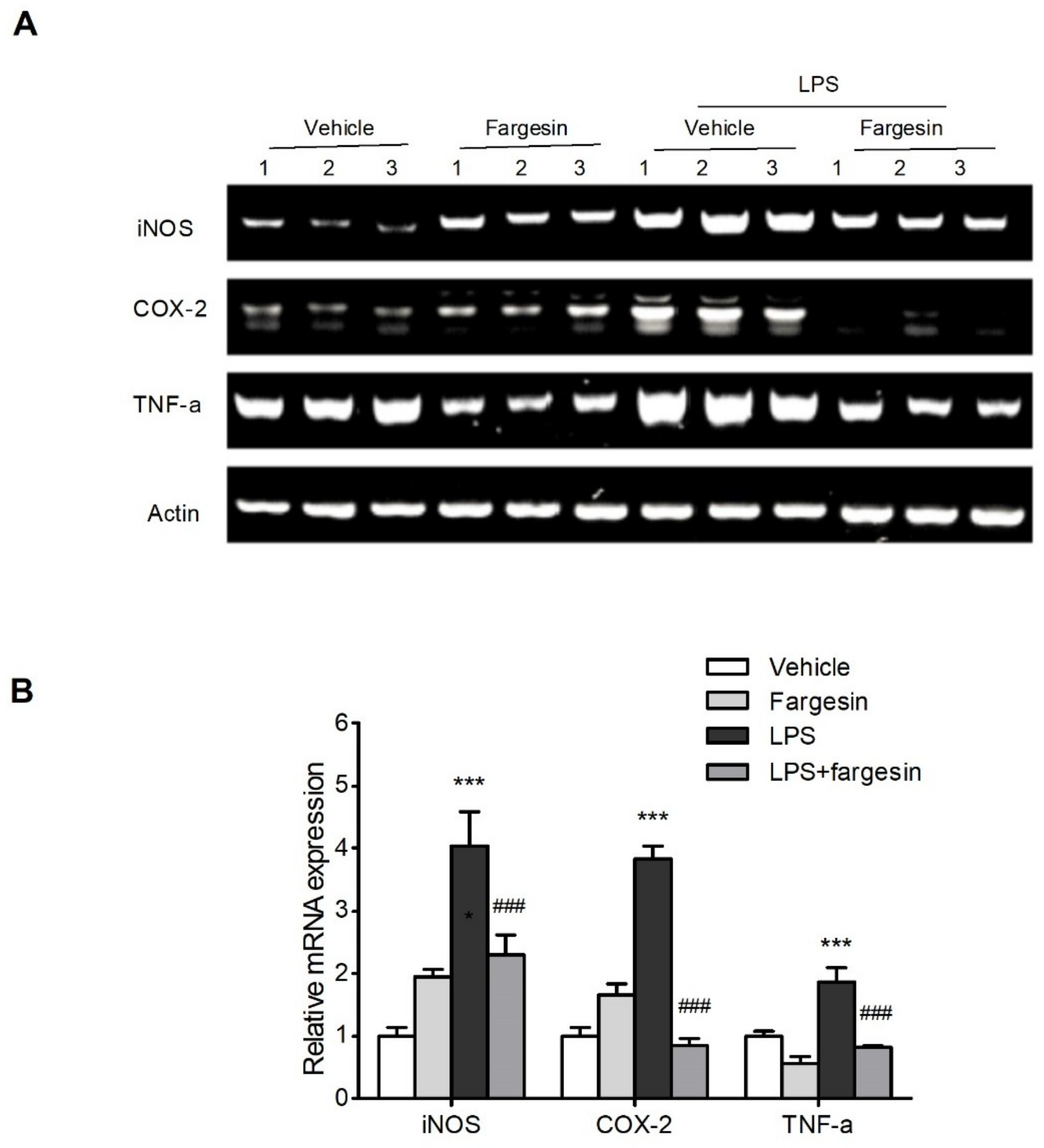
2.1.3. Fargesin Inhibited Pro-Inflammatory Gene Expression in the Colon
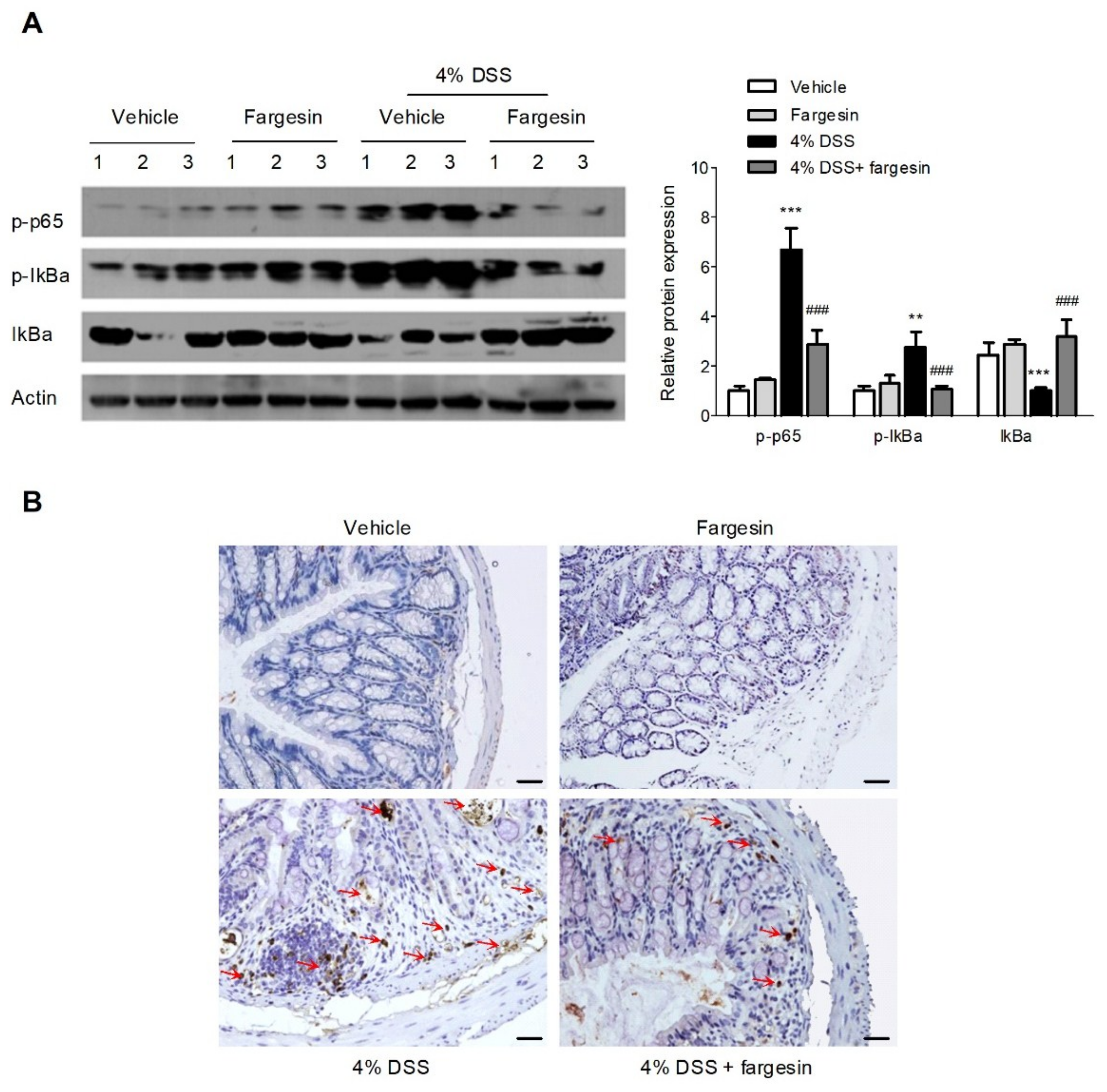
2.1.4. Fargesin Inhibited the Activation of NF-κB in the Colon
2.2. In Vitro Study
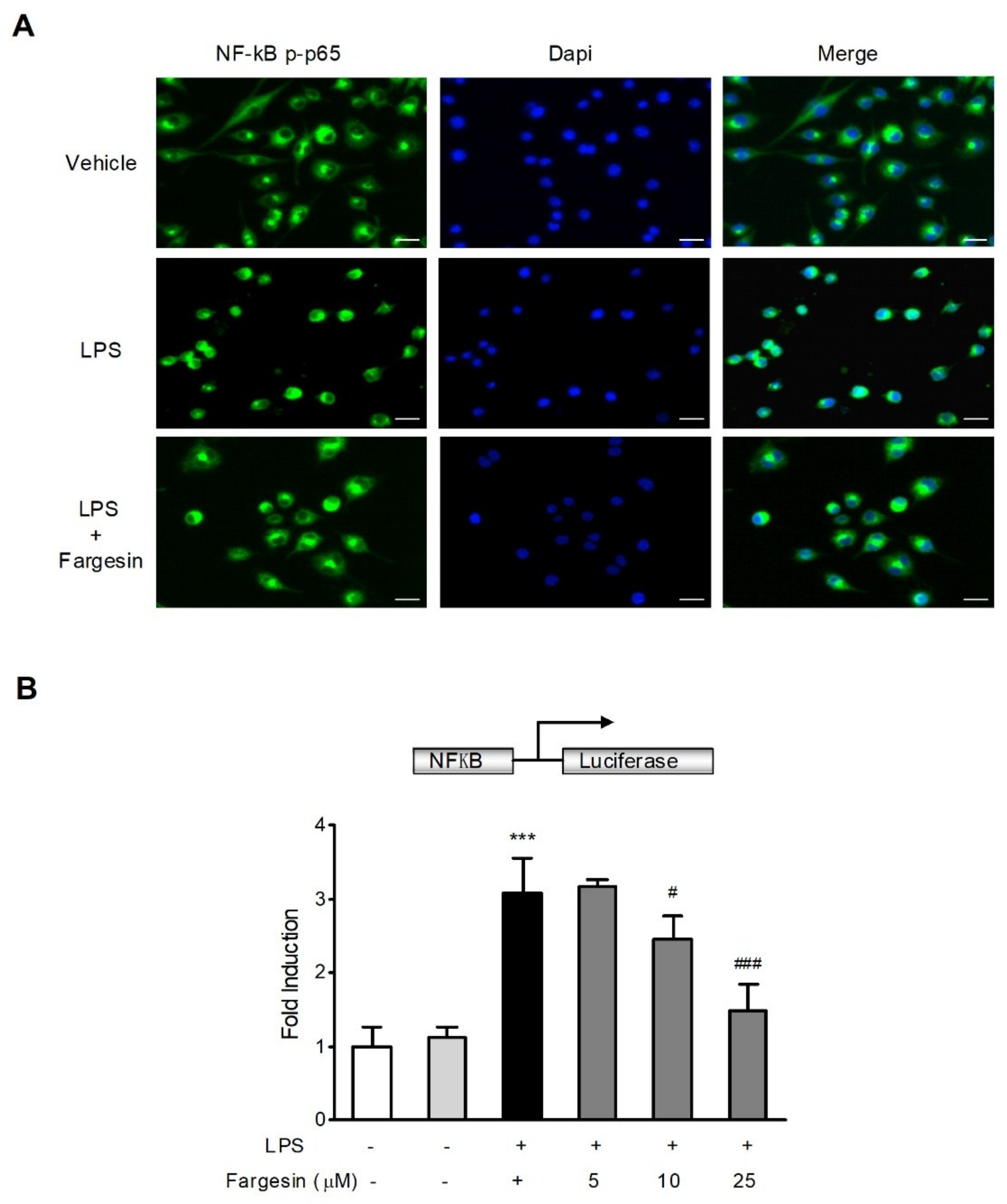
2.2.1. Fargesin Inhibited the Nuclear Translocation of p-p65 in RAW264.7 Cells
2.2.2. Fargesin Suppressed NF-κB-Luciferase Activity in RAW264.7 Cells
2.2.3. Fargesin Downregulated Proinflammatory Mediator Gene Expression in RAW264.7 Cells
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Animals and Cell Lines
4.3. Experimental Design and Assessment of the Intestinal Inflammatory Process
4.4. Western Blot and RNA Analysis
4.5. NF-κB Immunohistochemistry and Immunofluorescence
4.6. Determination of TNF-α and NO Levels and MPO Activity
4.7. NF-κB Luciferase Reporter Assay
4.8. Statistics Analyses
Author Contributions
Funding
Conflicts of Interest
References
- Rossignol, D.A. Hyperbaric oxygen treatment for inflammatory bowel disease: A systematic review and analysis. Med. Gas Res. 2012, 2, 6. [Google Scholar] [CrossRef] [PubMed]
- Jobin, C. Probiotics and ileitis: Could augmentation of TNF/NFκB activity be the answer? Gut Microbes 2010, 1, 196–199. [Google Scholar] [CrossRef] [PubMed]
- Sun, A.; Ren, A.; Deng, C.; Zhang, J.; Luo, X.; Wu, X.; Mani, S.; Dou, W.; Wang, Z. C-glycosyl flavonoid orientin improves chemically induced inflammatory bowel disease in mice. J. Funct. Foods 2016, 21, 418–430. [Google Scholar] [CrossRef]
- Liu, X.; Wang, J.M. Iridoid glycosides fraction of folium syringae leaves modulates NF-κB signal pathway and intestinal epithelial cells apoptosis in experimental colitis. PLoS ONE 2011, 6, e24740. [Google Scholar] [CrossRef] [PubMed]
- Dou, W.; Zhang, J.; Sun, A.; Zhang, E.; Ding, L.; Mukherjee, S.; Wei, X.; Chou, G.; Wang, Z.T.; Mani, S. Protective effect of naringenin against experimental colitis via suppression of toll-like receptor4/NF-κB signalling. Br. J. Nutr. 2013, 110, 599–608. [Google Scholar] [CrossRef] [PubMed]
- Schreiber, S.; Nikolaus, S.; Hampe, J. Activation of nuclear factor-kB in inflammatory bowel disease. Gut 1998, 42, 477–484. [Google Scholar] [CrossRef] [PubMed]
- Esposito, G.; Capoccia, E.; Turco, F.; Palumbo, I.; Lu, J.; Steardo, A.; Cuomo, R.; Sarnelli, G.; Steardo, L. Palmitoylethanolamide improves colon inflammation through an enteric glia/toll like receptor 4-dependent PPAR-α activation. Gut 2014, 63, 1300–1312. [Google Scholar] [CrossRef] [PubMed]
- Pham, T.H.; Kim, M.S.; Le, M.Q.; Song, Y.S.; Bak, Y.; Ryu, H.W.; Oh, S.R.; Yoon, D.Y. Fargesin exerts anti-inflammatory effects in THP-1 monocytes by suppressing PKC-dependent AP-1 and NF-kB signaling. Phytomedicine 2017, 24, 96–103. [Google Scholar] [CrossRef] [PubMed]
- Kolios, G.; Valatas, V.; Ward, S.G. Nitric oxide in inflammatory bowel disease: A universal messenger in an unsolved puzzle. Immunology 2004, 113, 427–437. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Dou, W.; Zhang, E.; Sun, A.; Ding, L.; Wei, X.; Chou, G.; Mani, S.; Wang, Z. Paeoniflorin abrogates DSS-induced colitis via a TLR4-dependent pathway. Am. J. Physiol. Gastrointest. Liver Physiol. 2014, 306, G27–G36. [Google Scholar] [CrossRef] [PubMed]
- Qin, X.; Jiang, X.; Jiang, X.; Wang, Y.; Miao, Z.; He, W.; Yang, G.; Lv, Z.; Yu, Y.; Zheng, Y. Micheliolide inhibits LPS-induced inflammatory response and protects mice from LPS challenge. Sci. Rep. 2016, 6, 23240. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Wang, L.; Wu, Z.; Yao, L.; Wu, Y.; Huang, L.; Liu, K.; Zhou, X.; Gou, D. Anthocyanin-rich fractions from red raspberries attenuate inflammation in both RAW264.7macrophages and a mouse model of colitis. Sci. Rep. 2014, 4, 6234. [Google Scholar] [CrossRef] [PubMed]
- Goyal, N.; Rana, A.; Ahlawat, A.; Bijjem, K.R.; Kumar, P. Animal models of inflammatory bowel disease: A review. Inflammopharmacology 2014, 22, 219–233. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.K.; Cheng, L.N.; Huang, X.L.; Shi, W.; Xiang, J.Y.; Gan, H.T. Tetrandrine ameliorates dextran-sulfate-sodium-induced colitis in mice through inhibition of nuclear factor-kappaB activation. Int. J. Colorectal Dis. 2009, 24, 5–12. [Google Scholar] [CrossRef] [PubMed]
- Dou, W.; Zhang, J.; Ren, G.; Ding, L.; Sun, A.; Deng, C.; Wu, X.; Wei, X.; Mani, Z.; Wang, Z. Mangiferin attenuates the symptoms of dextran sulfate sodium-induced colitis in mice via NF-kB and MAPK signaling inactivation. Int. Immunopharmacol. 2014, 23, 170–178. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Wu, X.; Wang, C.L.; Wang, L.; Sun, C.; Zhang, D.B.; Liu, J.L.; Liang, Y.N.; Tang, D.X.; Tang, Z.S. Tryptanthrin protects mice against dextran sulfate sodium-induced colitis through inhibition of TNF-α/NF-κB and IL-6/STAT3 pathways. Molecules 2018, 23, 1062. [Google Scholar] [CrossRef] [PubMed]
- Singh, U.P.; Singh, N.P.; Murphy, E.A.; Price, R.L.; Fayad, R.; Nagarkatti, M.; Nagarkatti, P.S. Chemokine and cytokine levels in inflammatory bowel disease patients. Cytokine 2016, 77, 44–49. [Google Scholar] [CrossRef] [PubMed]
- Kwon, H.S.; Oh, S.M.; Kim, J.K. Glabridin, a functional compound of liquorice, attenuates colonic inflammation in mice withdextran sulphate sodium-induced colitis. Clin. Exp. Immunol. 2008, 151, 165–173. [Google Scholar] [CrossRef] [PubMed]
- Baek, J.A.; Lee, Y.D.; Lee, C.B.; Go, H.K.; Kim, J.P.; Seo, J.J.; Rhee, Y.K.; Kim, A.M.; Na, D.J. Extracts of magnoliae flos inhibit inducible nitric oxide synthase via ERK in human respiratory epithelial cells. Nitric. Oxide. 2009, 20, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Debnath, T.; Kim, D.H.; Lim, B.O. Natural products as a source of anti-inflammatory agents associated with inflammatory bowel disease. Molecules 2013, 18, 7253–7270. [Google Scholar] [CrossRef] [PubMed]
- Shin, E.K.; Kwon, H.S.; Kim, Y.H.; Shin, H.K.; Kim, J.K. Chrysin, a natural flavone, improves murine inflammatory bowel diseases. Biochem. Biophys. Res. Commun. 2009, 381, 502–507. [Google Scholar] [CrossRef] [PubMed]
- Dou, W.; Zhang, J.; Li, H.; Kortagere, S.; Sun, K.; Ding, L.; Ren, G.; Wang, Z.; Mani, S. Plant flavonol isorhamnetin attenuates chemically induced inflammatory bowel disease via a PXR-dependent pathway. J. Nutr. Biochem. 2014, 25, 923–933. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.S.; Cha, B.Y.; Choi, S.S.; Harada, Y.; Choi, B.K.; Yonezawa, T.; Teruya, T.; Nagai, K.; Woo, J.T. Fargesin improves lipid and glucose metabolism in 3T3-L1 adipocytes and high-fat diet-induced obese mice. Biofactors 2012, 38, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Yu, Z.; Deng, C.; Zhang, J.; Ren, G.; Sun, A.; Mani, S.; Wang, Z. Baicalein ameliorates TNBS-induced colitis by suppressing TLR4/MyD88 signaling cascade and NLRP3 inflammasome activation in mice. Sci. Rep. 2017, 7, 16374. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: not available. |
| Group | TNF-α (pg/mg pr.) | MPO (U/mg pr.) |
|---|---|---|
| Vehicle | 1.37 ± 0.05 | 0.39 ± 0.08 |
| 4% DSS | 1.86 ± 0.08 *** | 0.77 ± 0.27 ** |
| 4% DSS + fargesin | 1.34 ± 0.18 # | 0.37 ± 0.04 # |
| S.No. | Gene | Primer Sequence | Annealing Temperature |
|---|---|---|---|
| 1 | iNOS | F:5′-GGGAATCTTGGAGCGAGTTG-3′ R:5′-GTGAGGGCTTGGCTGAGTGA-3′ | 58 °C |
| 2 | COX-2 | F:5′-GAAGTCTTTGGTCTGGTGCCT-3′ R:5′-GCTCCTGCTTGAGTATGTCG-3′ | 58 °C |
| 3 | TNF-α | F: 5′-CGTGGAACTGGCAGAAGAGG-3′ R: 5′-AGACAGAAGAGCGTGGTGGC-3′ | 58 °C |
| 4 | ICAM-1 | F: 5′-CGCTGTGCTTTGAGAACTGT-3′ R:5′-AGGTCCTTGCCTACTTGCTG-3′ | 60 °C |
| 5 | IFNγ | F: 5′-AAGTGGCATAGATGTGGAAG-3′ R: 5′-AAGTGGCATAGATGTGGAAG-3′ | 60 °C |
| 6 | IL-1β | F: 5′-GGCTGGACTGTTTCTAATGC-3′ R: 5′-ATGGTTTCTTGTGACCCTGA-3′ | 60 °C |
| 7 | IL-10 | F:5′-ACAACATACTGCTAACCGACTC-3′ R: 5′-CACTCTTCACCTGCTCCACT-3′ | 60 °C |
| 8 | IL-15 | F: 5′-CAGAATGGGAGGTGGTAGTGC-3′ R: 5′-AAGAGTGGCTGGACAGAAGG-3′ | 60 °C |
| 9 | β-actin | F:5′-CAGCCTTCCTTCTTGGGTAT-3′ R: 5′-TGGCATAGAGGTCTTTACGG-3′ | 58 °C/60 °C |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yue, B.; Ren, Y.-J.; Zhang, J.-J.; Luo, X.-P.; Yu, Z.-L.; Ren, G.-Y.; Sun, A.-N.; Deng, C.; Wang, Z.-T.; Dou, W. Anti-Inflammatory Effects of Fargesin on Chemically Induced Inflammatory Bowel Disease in Mice. Molecules 2018, 23, 1380. https://doi.org/10.3390/molecules23061380
Yue B, Ren Y-J, Zhang J-J, Luo X-P, Yu Z-L, Ren G-Y, Sun A-N, Deng C, Wang Z-T, Dou W. Anti-Inflammatory Effects of Fargesin on Chemically Induced Inflammatory Bowel Disease in Mice. Molecules. 2018; 23(6):1380. https://doi.org/10.3390/molecules23061380
Chicago/Turabian StyleYue, Bei, Yi-Jing Ren, Jing-Jing Zhang, Xiao-Ping Luo, Zhi-Lun Yu, Gai-Yan Ren, A-Ning Sun, Chao Deng, Zheng-Tao Wang, and Wei Dou. 2018. "Anti-Inflammatory Effects of Fargesin on Chemically Induced Inflammatory Bowel Disease in Mice" Molecules 23, no. 6: 1380. https://doi.org/10.3390/molecules23061380
APA StyleYue, B., Ren, Y.-J., Zhang, J.-J., Luo, X.-P., Yu, Z.-L., Ren, G.-Y., Sun, A.-N., Deng, C., Wang, Z.-T., & Dou, W. (2018). Anti-Inflammatory Effects of Fargesin on Chemically Induced Inflammatory Bowel Disease in Mice. Molecules, 23(6), 1380. https://doi.org/10.3390/molecules23061380