Sporulosol, a New Ketal from the Fungus Paraconiothyrium sporulosum
Abstract
:1. Introduction
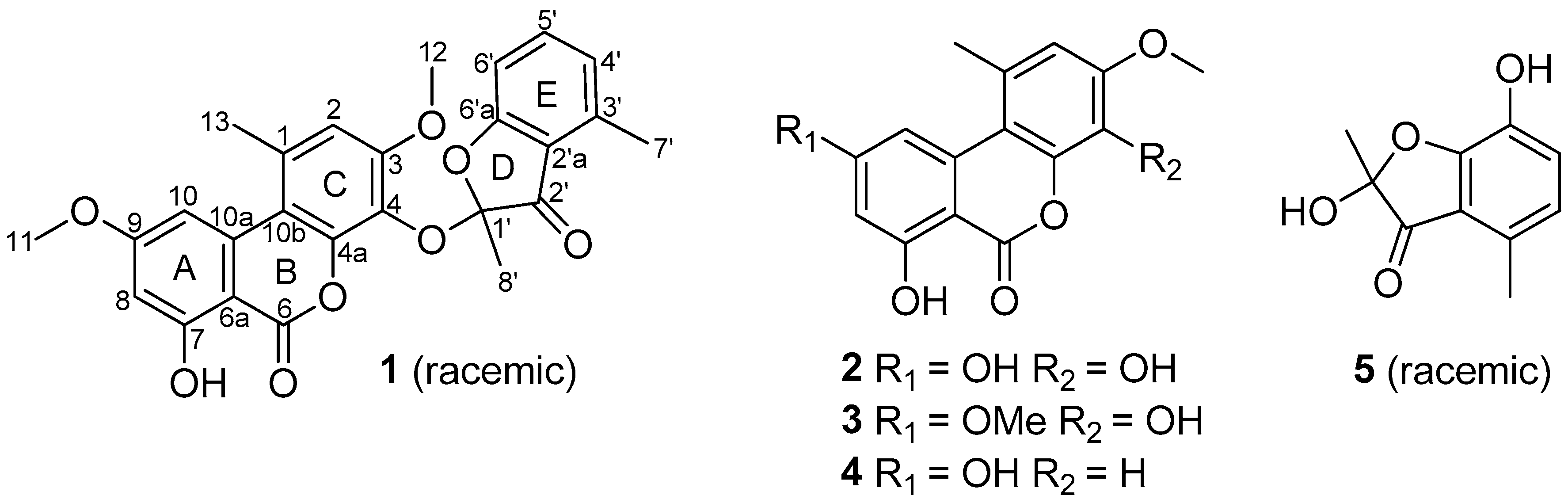
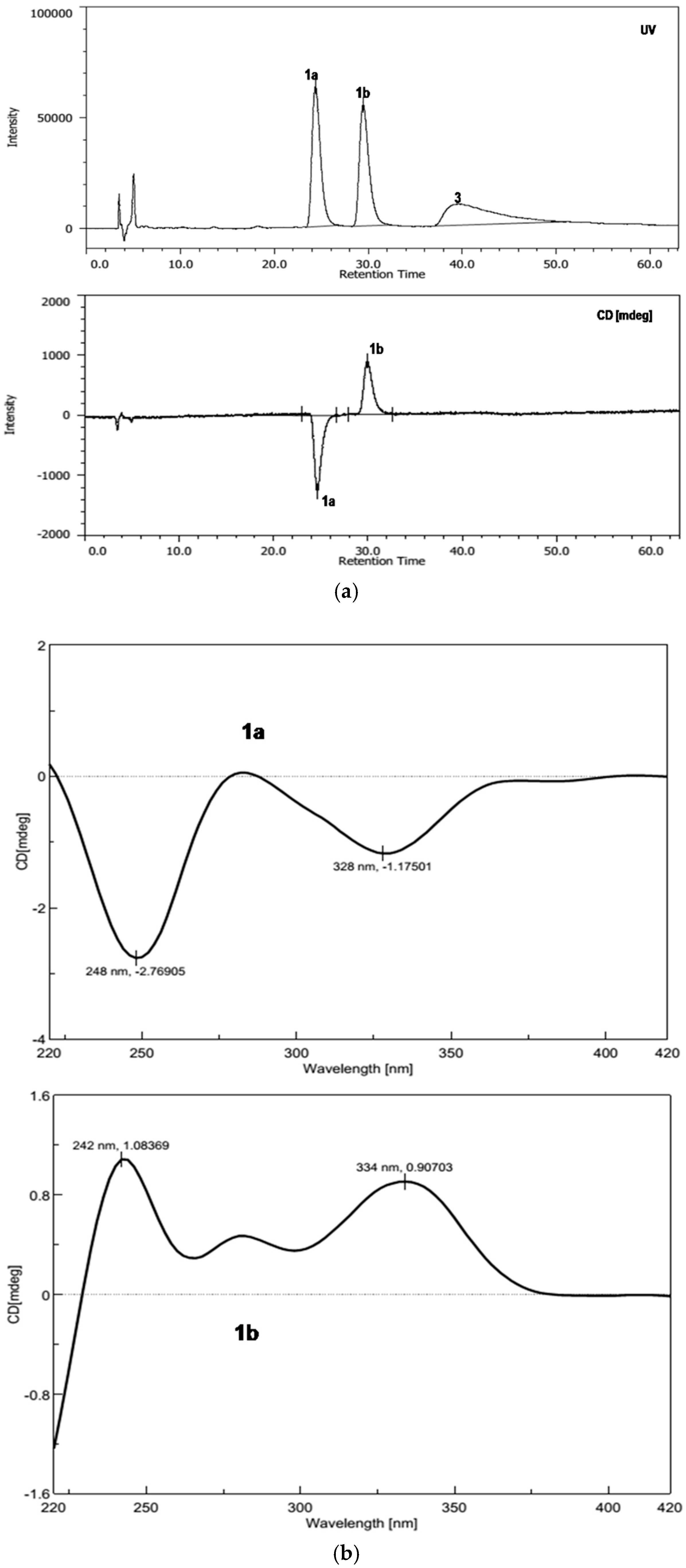
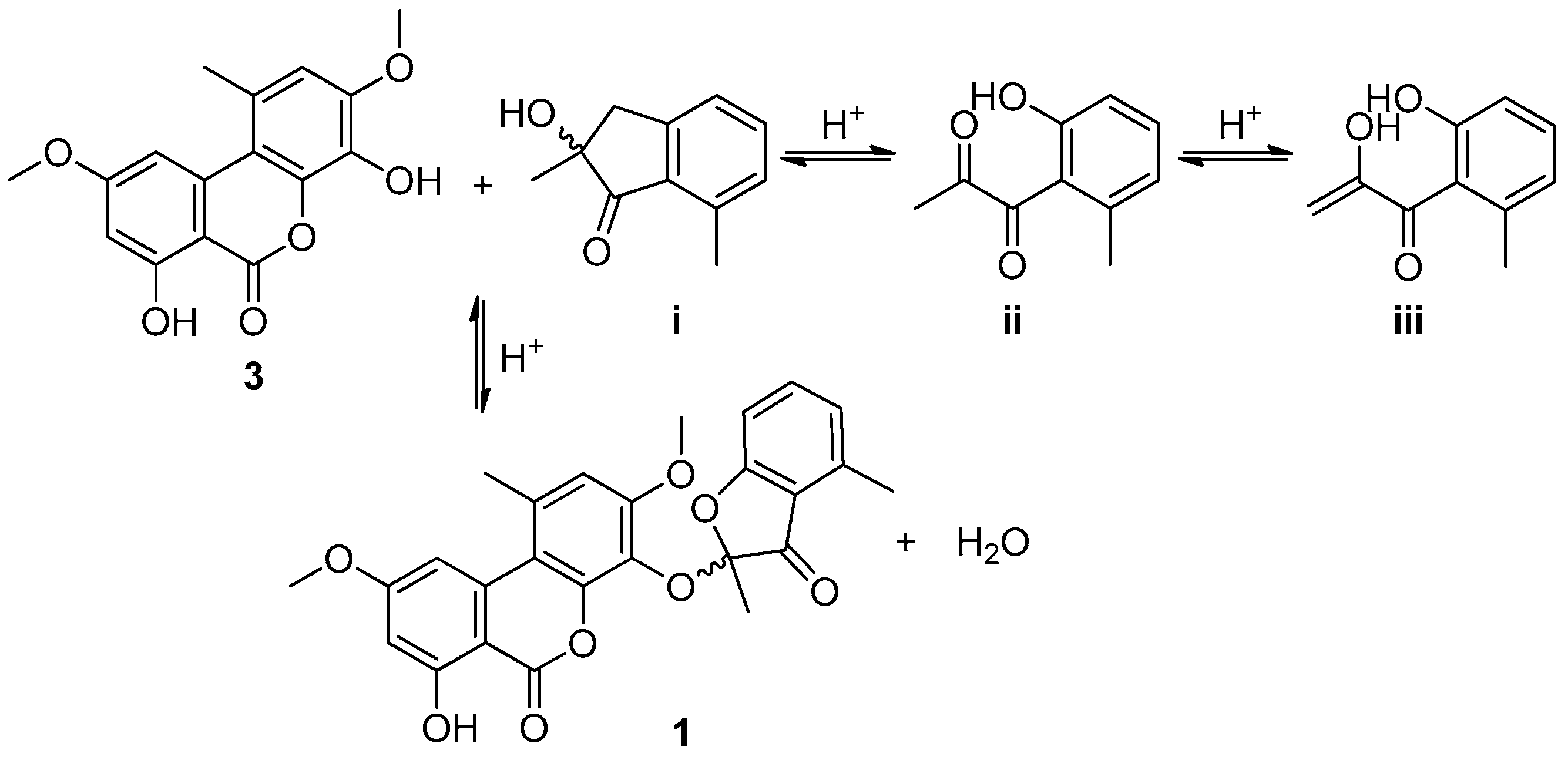
2. Results and Discussion
3. Materials and Methods
3.1. General Experimental Procedures
3.2. Fungal Material
3.3. Extraction and Isolation
3.4. Sporulosol (1)
3.5. 4,7,9-Trihydroxy-3-methoxy-1-methyl-6H-benzo[c]chromen-6-one (2)
3.6. MTT Assay
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References and Notes
- Schueffler, A.; Anke, T. Fungal natural products in research and development. Nat. Prod. Rep. 2014, 31, 1425–1448. [Google Scholar] [CrossRef] [PubMed]
- Bao, Y.; Chen, G.; Wu, Y.; Li, X.; Hu, D.; Liu, X.; Li, X.; Yao, X.; Gao, H. Stachybisbins A and B, the first cases of seco-bisabosquals from Stachybotrys bisbyi. Fitoterapia 2015, 105, 151–155. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Feng, B.; Sun, Y.; Wu, H.; Li, S.; Liu, B.; Liu, F.; Zhang, W.; Chen, G.; Bai, J.; et al. Flaviphenalenones A–C, three new phenalenone derivatives from the fungus Aspergillus flavipes PJ03-11. Tetrahedron Lett. 2016, 57, 645–649. [Google Scholar] [CrossRef]
- Zhang, L.; Feng, B.; Zhao, Y.; Sun, Y.; Liu, B.; Liu, F.; Chen, G.; Bai, J.; Hua, H.; Wang, H.; et al. Polyketide butenolide, diphenyl ether, and benzophenone derivatives from the fungus Aspergillus flavipes PJ03-11. Bioorg. Med. Chem. Lett. 2016, 26, 346–350. [Google Scholar] [CrossRef] [PubMed]
- Tian, J.; Li, P.; Li, X.; Sun, P.; Gao, H.; Liu, X.; Huang, P.; Tang, J.; Yao, X. New antibacterial isocoumarin glycosides from a wetland soil derived fungal strain Metarhizium anisopliae. Bioorg. Med. Chem. Lett. 2016, 26, 1391–1396. [Google Scholar] [CrossRef] [PubMed]
- Verkley, G.J.; da Silva, M.; Wicklow, D.T.; Crous, P.W. Paraconiothyrium, a new genus to accommodate the mycoparasite Coniothyrium minitans, anamorphs of Paraphaeosphaeria, and four new species. Stud. Mycol. 2004, 50, 323–335. [Google Scholar]
- Tsuda, M.; Mugishima, T.; Komatsu, K.; Sone, T.; Tanaka, M.; Mikami, Y.; Kobayashi, J. Modiolides A and B, two new 10-membered macrolides from a marine-derived fungus. J. Nat. Prod. 2003, 66, 412–415. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, I.E.; Kehraus, S.; Krick, A.; König, G.M.; Kelter, G.; Maier, A.; Fiebig, H.H.; Kalesse, M.; Malek, N.P.; Gross, H. Mode of action of epoxyphomalins A and B and characterization of related metabolites from the marine-derived fungus Paraconiothyrium sp. J. Nat. Prod. 2010, 73, 2053–2056. [Google Scholar] [CrossRef] [PubMed]
- Shiono, Y.; Kikuchi, M.; Koseki, T.; Murayama, T.; Kwon, E.; Aburai, N.; Kimura, K. Isopimarane diterpene glycosides, isolated from endophytic fungus Paraconiothyrium sp. MY-42. Phytochemistry 2011, 72, 1400–1405. [Google Scholar] [CrossRef] [PubMed]
- Almeida, C.; Aouad, N.E.; Martín, J.; Pérez-Victoria, I.; González-Menéndez, V.; Platas, G.; de la Cruz, M.; Monteiro, M.C.; de Pedro, N.; Bills, G.F.; et al. Graminin B, a furanone from the fungus Paraconiothyrium sp. J. Antibiot. 2014, 67, 421–423. [Google Scholar] [CrossRef] [PubMed]
- Amand, S.; Vallet, M.; Guedon, L.; Genta-Jouve, G.; Wien, F.; Mann, S.; Dupont, J.; Prado, S.; Nay, B. A reactive eremophilane and its antibacterial 2(1H)-naphthalenone rearrangement product, witnesses of a microbial chemical warfare. Org. Lett. 2017, 19, 4038–4041. [Google Scholar] [CrossRef] [PubMed]
- Soliman, S.S.; Tsao, R.; Raizada, M.N. Chemical inhibitors suggest endophytic fungal paclitaxel is derived from both mevalonate and non-mevalonate-like pathways. J. Nat. Prod. 2011, 74, 2497–2504. [Google Scholar] [CrossRef] [PubMed]
- Somjaipeng, S.; Medina, A.; Kwaśna, H.; Ordaz Ortiz, J.; Magan, N. Isolation, identification, and ecology of growth and taxol production by an endophytic strain of Paraconithyrium variabile from English yew trees (Taxus baccata). Fungal Biol. 2015, 119, 1022–1031. [Google Scholar] [CrossRef] [PubMed]
- De Gusmño, N.B.; Kaouadji, M.; Steiman, R.; Seigle-murandi, F.; Ulrichc, J. Coniothyriol, an uncommon polyketide from Coniothyrium sporulosum. Nat. Prod. Lett. 1993, 2, 287–292. [Google Scholar] [CrossRef]
- Guiraud, P.; Steiman, R.; Seigle-murandi, F.; de Gusmño, N.B. Antimicrobial and antitumor activities of mycosporulone. J. Nat. Prod. 1999, 62, 1222–1224. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Feng, B.; Chen, G.; Li, S.; Sun, Y.; Wu, H.; Bai, J.; Hua, H.; Wang, H.; Pei, Y. Sporulaminals A and B: A pair of unusual epimeric spiroaminal derivatives from a marine-derived fungus Paraconiothyrium sporulosum YK-03. RSC Adv. 2016, 6, 42361–42366. [Google Scholar] [CrossRef]
- Zhang, L.; Li, S.; Wu, H.; Chen, G.; Li, L.; Bai, J.; Hua, H.; Wang, H.; Pei, Y. 3,4-Dihydroisocoumarin derivatives from the marine-derived fungus Paraconiothyrium sporulosum YK-03. Phytochem. Lett. 2017, 20, 200–203. [Google Scholar] [CrossRef]
- Liu, L.; Gao, H.; Chen, X.; Cai, X.; Yang, L.; Guo, L.; Yao, X.; Che, Y. Brasilamides A–D: Sesquiterpenoids from the plant endophytic fungus Paraconiothyrium brasiliense. Eur. J. Org. Chem. 2010, 3302–3306. [Google Scholar] [CrossRef]
- Liu, L.; Chen, X.; Li, D.; Zhang, Y.; Li, L.; Guo, L.; Cao, Y.; Che, Y. Bisabolane sesquiterpenoids from the plant endophytic fungus Paraconiothyrium brasiliense. J. Nat. Prod. 2015, 78, 746–753. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Ren, F.; Che, Y.; Liu, G.; Liu, L. New bergamotane sesquiterpenoids from the plant endothytic fungus Paraconiothyrium brasiliense. Molecules 2015, 20, 14611–14620. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Zhang, Y.; Niu, S.; Liu, X.; Che, Y. Cytotoxic cleistanthane and cassane diterpenoids from the entomogenous fungus Paraconiothyrium hawaiiense. J. Nat. Prod. 2014, 77, 1513–1518. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Zhang, Y.; Zhao, C.; Ren, F.; Liu, X.; Che, Y. Hawaiinolides E–G, cytotoxic cassane and cleistanthane diterpenoids from the entomogenous fungus Paraconiothyrium hawaiiense. Fitoterapia 2014, 99, 236–242. [Google Scholar] [CrossRef] [PubMed]
- Tanahashi, T.; Kuroishi, M.; Kuwahara, A.; Nagakura, N.; Hamada, N. Four phenolics from the cultured lichen mycobiont of Graphis scripta var. pulverulenta. Chem. Pharm. Bull. 1997, 45, 1183–1185. [Google Scholar] [CrossRef]
- Machida, K.; Trifonov, L.S.; Ayer, W.A.; Lu, Z.; Laroche, A.; Huang, H.C.; Cheng, K.J.; Zantige, J.L. 3(2H)-Benzofuranones and chromanes from liquid cultures of the mycoparasitic fungus Coniothyrium minitans. Phytochemistry 2001, 58, 173–177. [Google Scholar] [CrossRef]
- Bekker, R.; Li, X.; ElSohly, H.N.; Clark, A.M.; Brandt, E.V.; Ferreira, D. Resolution and absolute configuration of naturally occurring auronols. J. Nat. Prod. 2001, 64, 345–347. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Wu, X.; Deng, Z.; Wang, J.; Zhou, S.; Vrijmoed, L.L.; Jones, E.B. The metabolites of the mangrove fungus Verruculina enalia No. 2606 from a salt lake in the Bahamas. Phytochemistry 2002, 59, 469–471. [Google Scholar] [CrossRef]
- Strobel, G.; Ford, E.; Worapong, J.; Harper, J.K.; Arif, A.M.; Grant, D.M.; Fung, P.C.; Chau, R.M. Isopestacin, an isobenzofuranone from Pestalotiopsis microspora, possessing antifungal and antioxidant activities. Phytochemistry 2002, 60, 179–183. [Google Scholar] [CrossRef]
- Harper, J.K.; Arif, A.M.; Ford, E.J.; Strobel, G.A.; Porco, J.A., Jr.; Tomer, D.P.; Oneill, K.L.; Heider, E.M.; Grant, D.M. Pestacin: A, 1,3-dihydro isobenzofuran from Pestalotiopsis microspora possessing antioxidant and antimycotic activities. Tetrahedron 2003, 59, 2471–2476. [Google Scholar] [CrossRef]
- Li, E.; Jiang, L.; Guo, L.; Zhang, H.; Che, Y. Pestalachlorides A–C, antifungal metabolites from the plant endophytic fungus Pestalotiopsis adusta. Bioorg. Med. Chem. 2008, 16, 7894–7899. [Google Scholar] [CrossRef] [PubMed]
- Li, E.; Zhang, F.; Niu, S.; Liu, X.; Liu, G.; Che, Y. A spiro[chroman-3,7′-isochromene]-4,6′(8′H)-dione from the Cordyceps-colonizing fungus Fimetariella sp. Org. Lett. 2012, 14, 3320–3323. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.; Kuang, R.; Chen, G.; Qin, S.; Wang, C.; Hu, D.; Wu, B.; Liu, X.; Yao, X.; Gao, H. Three pairs of new isopentenyl dibenzo[b,e]oxepinone enantiomers from Talaromyces flavus, a wetland soil-derived fungus. Molecules 2016, 21, 1184–1195. [Google Scholar] [CrossRef] [PubMed]
- Dellafiora, L.; Dall’Asta, C.; Cruciani, G.; Galaverna, G.; Cozzini, P. Molecular modelling approach to evaluate poisoning of topoisomerase I by alternariol derivatives. Food Chem. 2015, 189, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Harris, T.M.; Hay, J.V. Biogenetically modeled syntheses of heptaacetate metabolites alternariol and lichexanthone. J. Am. Chem. Soc. 1977, 99, 1631–1637. [Google Scholar] [CrossRef]
- Crystallographic data for 1 have been deposited with the Cambridge Crystallographic Data Centre (deposition number CCDC 1585447, accessed on 14/11/2017). Copies of the data can be obtained, free of charge, on application to the director, CCDC 12 Union Road, Cambridge CB2 1EZ, UK (fax: +44 1223 336033 or email: deposit@ccdc.cam.ac.uk).
- Sheldrick, G.M. SHELXL-97, Program for X-ray Crystal Structure Solution and Refinement; University of Göttingen: Göttingen, Germany, 1997. [Google Scholar]
- Sheldrick, G.M. SADABS, Program for Empirical Absorption Correction of Area Detector Data; University of Göttingen: Göttingen, Germany, 1999. [Google Scholar]
- Zhang, N.; Chen, Y.; Jiang, R.; Li, E.; Chen, X.; Xi, Z.; Guo, Y.; Liu, X.; Zhou, Y.; Che, Y.; et al. PARP and RIP 1 are required for autophagy induced by 11′-deoxyverticillin A, which precedes caspase-dependent apoptosis. Autophagy 2011, 7, 598–612. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds 1–5 are available from the authors. |

| Pos. | 1 | 2 | ||||
|---|---|---|---|---|---|---|
| δCa, Mult. | δHb (J in Hz) | HMBC | δCc Mult. | δHd (J in Hz) | HMBC | |
| 1 | 134.0, qC | 126.6, qC | ||||
| 2 | 112.8, CH | 6.90, s | 1, 3, 4, 4a, 10b | 112.8, CH | 6.93, s | 1, 3, 4, 10a, 10b, 12 |
| 3 | 153.8, qC | 147.8, qC | ||||
| 4 | 128.0, qC | 140.6, qC | ||||
| 4a | 147.0, qC | 138.8, qC | ||||
| 6 | 164.4, qC | 164.8, qC | ||||
| 6a | 98.9, qC | 98.2, qC | ||||
| 7 | 165.0, qC | 165.0, qC | ||||
| 8 | 99.5, CH | 6.60, d (2.0) | 7, 6a, 10a | 101.3, CH | 6.46, d (2.0) | 6a, 9, 10 |
| 9 | 166.7, qC | 165.3, qC | ||||
| 10 | 104.2, CH | 7.28, d (2.0) | 6a, 9, 10a | 104.9, CH | 7.37, d (2.0) | 6a, 8, 9, 10b |
| 10a | 137.9, qC | 132.8, qC | ||||
| 10b | 111.0, qC | 111.2, qC | ||||
| 11 | 55.4, CH3 | 3.96, s | 9 | 55.7, CH3 | 3.67, s | 3 |
| 12 | 55.2, CH3 | 3.67, s | 3 | 24.5, CH3 | 2.75, s | 1, 2, 10, 10a |
| 13 | 24.8, CH3 | 2.78, s | 1, 2 | |||
| 1′ | 105.4, qC | |||||
| 2′ | 195.9, qC | |||||
| 2′a | 117.4, qC | |||||
| 3′ | 139.8, qC | |||||
| 4′ | 123.8, CH | 6.88, d (7.8) | 5′, 6′, 7′ | |||
| 5′ | 137.7, CH | 7.45, t (7.8) | 4′, 6′a | |||
| 6′ | 109.8, CH | 6.72, d (7.8) | 2′a, 5′ | |||
| 6′a | 170.0, qC | |||||
| 7′ | 16.7, CH3 | 2.53, s | 2′a, 3′, 4′ | |||
| 8′ | 19.4, CH3 | 1.80, s | 1′, 2′ | |||
| OH-7 | 11.89, s | 7, 8 | 11.95, s | 6a, 7, 8 | ||
| Compound | IC50 (μM) | ||||
|---|---|---|---|---|---|
| HeLa | T24 | A549 | HCT116 | SH-SY5Y | |
| 1 | 31.8 ± 0.2 | 18.2 ±5.3 | 23.1 ± 2.9 | 50.1 ± 7.0 | >50 |
| 2 | 11.7 ± 0.7 | 22.8 ± 1.7 | 25.1 ± 1.8 | 81.5 ± 3.4 | 41.5 ± 2.0 |
| 3 | >50 | 77.8 ± 8.0 | >50 | >50 | 71.7 ± 3.2 |
| 4 | 5.4 ± 0.1 | 4.5 ± 0.4 | 93.7 ± 5.6 | 20.9 ± 2.6 | 38.8 ± 4.9 |
| cisplatin | 8.7 ± 0.3 | 10.9 ± 0.7 | 17.0 ± 0.5 | 19.9 ± 0.7 | 11.2 ± 2.0 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, C.; Fu, P.; Zhang, Y.; Liu, X.; Ren, F.; Che, Y. Sporulosol, a New Ketal from the Fungus Paraconiothyrium sporulosum. Molecules 2018, 23, 1263. https://doi.org/10.3390/molecules23061263
Zhao C, Fu P, Zhang Y, Liu X, Ren F, Che Y. Sporulosol, a New Ketal from the Fungus Paraconiothyrium sporulosum. Molecules. 2018; 23(6):1263. https://doi.org/10.3390/molecules23061263
Chicago/Turabian StyleZhao, Chen, Peinan Fu, Yang Zhang, Xingzhong Liu, Fengxia Ren, and Yongsheng Che. 2018. "Sporulosol, a New Ketal from the Fungus Paraconiothyrium sporulosum" Molecules 23, no. 6: 1263. https://doi.org/10.3390/molecules23061263
APA StyleZhao, C., Fu, P., Zhang, Y., Liu, X., Ren, F., & Che, Y. (2018). Sporulosol, a New Ketal from the Fungus Paraconiothyrium sporulosum. Molecules, 23(6), 1263. https://doi.org/10.3390/molecules23061263




