Pyrrole Alkaloids from the Edible Mushroom Phlebopus portentosus with Their Bioactive Activities
Abstract
:1. Introduction
2. Results
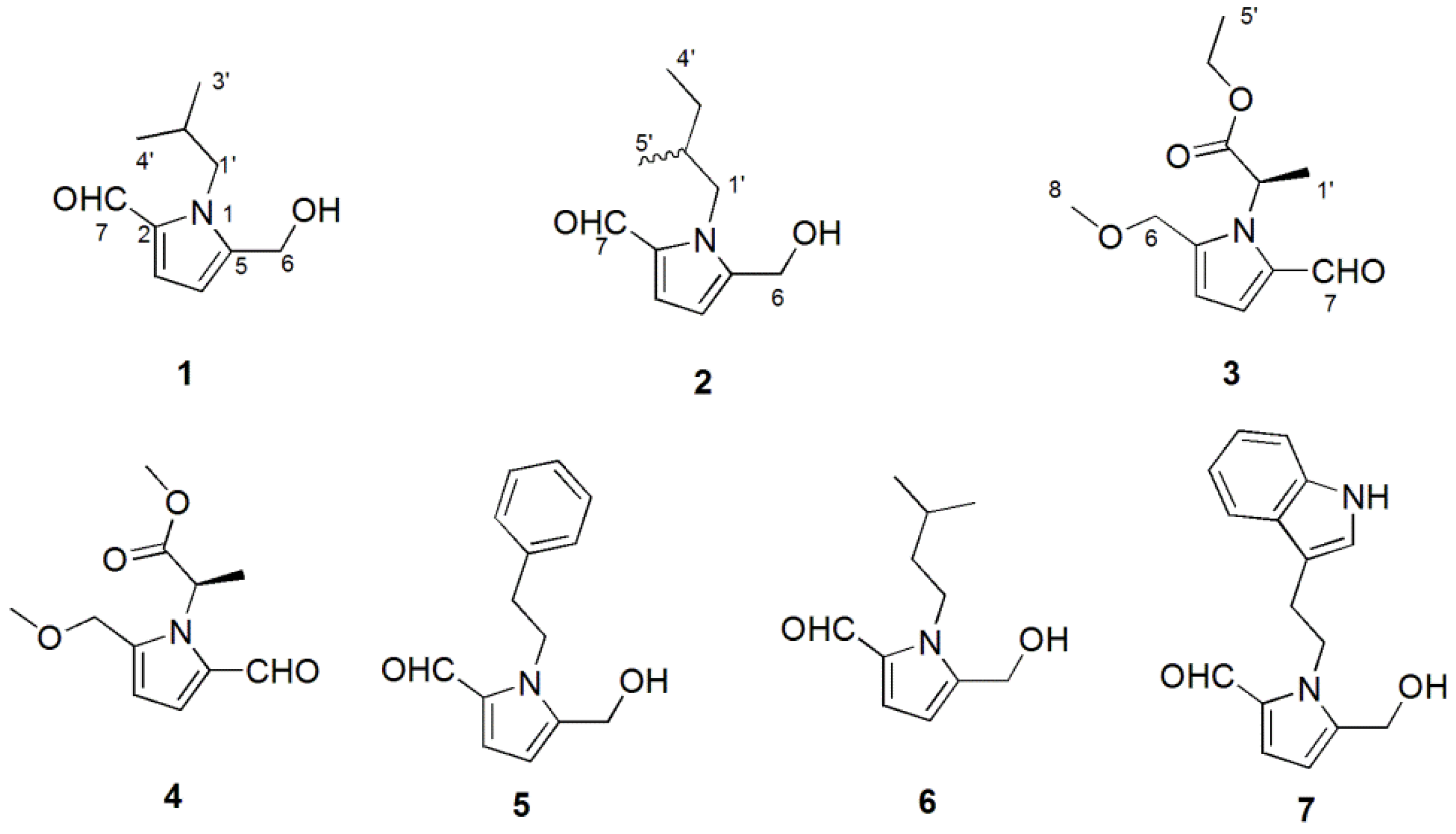
2.1. Structure Elucidation
2.2. Bioactive Activity
3. Discussion
4. Materials and Methods
4.1. General Experimental Procedures
4.2. Fungal Material
4.3. Isolation and Purification of Compounds 1–7
4.4. Characterization of Compounds 1–3
4.5. Neuroprotective Activity Assay
4.6. Acetylcholinesterase Inhibitory Assay
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Longvah, T.; Deosthale, Y.G. Compositional and nutritional studies on edible wild mushroom from northeast India. Food Chem. 1998, 63, 331–334. [Google Scholar] [CrossRef]
- Manzi, P.; Aguzzi, A.; Pizzoferrato, L. Nutritional value of mushrooms widely consumed in Italy. Food Chem. 2001, 73, 321–325. [Google Scholar] [CrossRef]
- Wani, B.A.; Bodha, R.H.; Wani, A.H. Nutritional and medicinal importance of mushrooms. J. Med. Plant Res. 2010, 4, 2598–2604. [Google Scholar]
- Kawagishi, H.; Shimada, A.; Shirai, R.; Okamoto, K.; Ojima, F.; Sakamoto, H.; Ishiguro, Y.; Furukawa, S. Erinacines, A, B and C, strong stimulators of nerve growth factor (NGF)-synthesis, from the mycelia of Hericium erinaceum. Tetrahedron Lett. 1994, 35, 1569–1572. [Google Scholar] [CrossRef]
- Yin, X.; Feng, T.; Li, Z.H.; Dong, Z.J.; Li, Y.; Liu, J.K. Highly oxygenated meroterpenoids from fruiting bodies of the mushroom Tricholoma terreum. J. Nat. Prod. 2013, 76, 1365–1368. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.Z.; Chen, H.P.; Wu, B.; Zhang, L.; Li, Z.H.; Feng, T.; Liu, J.K. Matsutakone and Matsutoic Acid, two (nor)steroids with unusual skeletons from the edible mushroom Tricholoma matsutake. J. Organ. Chem. 2017, 82, 7974–7979. [Google Scholar] [CrossRef] [PubMed]
- Ji, K.P.; Cao, Y.; Zhang, C.X.; He, M.X.; Liu, J.; Wang, W.B.; Wang, Y. Cultivation of Phlebopus portentosus in southern China. Mycol. Prog. 2011, 10, 293–300. [Google Scholar] [CrossRef]
- Ji, K.; Zhang, C.; Zeng, Y.; Liu, C.; He, M.; Wang, W. Artificial fungal colony and its fruiting of Phlebopus portentosus (Boletaceae) in pot. Acta Botanica Yunnanica 2007, 29, 554–558. [Google Scholar]
- Yang, Z.; Zang, M. Tropical Affinities of Higher Fungi in Southern China. Acta Botanica Yunnanica 2003, 25, 129–144. [Google Scholar]
- Joung Youn, U.; Kil, Y.S.; Nam, J.W.; Jin Lee, Y.; Kim, J.; Lee, D.; Lee, J.H.; Seo, E.K. New Pyrrole Alkaloids with Bulky N-Alkyl Side Chains Containing Stereogenic Centers from Lycium chinense. Helv. Chim. Acta 2013, 96, 1482–1487. [Google Scholar] [CrossRef]
- Zhan, Z.J.; Shan, W.G.; Bai, H.B.; Zhang, L.Y. A new alkaloid from the mycelium of Inonotus obliquus. J. Chem. Res. 2014, 38, 245–246. [Google Scholar] [CrossRef]
- Yang, J.J.; Yu, D.Q. Synthesis of ganoderma alkaloid A and B. Yao Xue Xue Bao 1990, 25, 555–559. [Google Scholar] [PubMed]
- Shan, W.-G.; Wang, Y.; Ma, L.-F.; Zhan, Z.-J. A new pyrrole alkaloid from the mycelium of Inonotus obliquus. J. Chem. Res. 2017, 41, 393. [Google Scholar] [CrossRef]
- Li, M.; Xiong, J.; Huang, Y.; Wang, L.-J.; Tang, Y.; Yang, G.-X.; Liu, X.-H.; Wei, B.-G.; Fan, H.; Zhao, Y. Xylapyrrosides A and B, two rare sugar-morpholine spiroketal pyrrole-derived alkaloids from Xylaria nigripes: Isolation, complete structure elucidation, and total syntheses. Tetrahedron 2015, 71, 5285–5295. [Google Scholar] [CrossRef]
- Shigematsu, H.; Kurata, T.; Kato, H.; Fujimaki, M. Formation of 2-(5-Hyclroxymethyl-2-formylpyrrol-1-yl) alkyl Acid Lactones on Roasting Alkyl-α-amino Acid with d-Glucose. Agric. Biol. Chem. 1971, 35, 2097–2105. [Google Scholar] [CrossRef]
- Xiong, J.; Huang, Y.; Wu, X.Y.; Liu, X.H.; Fan, H.; Wang, W.; Zhao, Y.; Yang, G.X.; Zhang, H.Y.; Hu, J.F. Chemical constituents from the fermented mycelia of the medicinal fungus Xylaria nigripes. Helv. Chim. Acta 2016, 99, 83–89. [Google Scholar] [CrossRef]
- Xiong, L.; Peng, C.; Xie, X.F.; Guo, L.; He, C.J.; Geng, Z.; Wan, F.; Dai, O.; Zhou, Q.M. Alkaloids isolated from the lateral root of Aconitum carmichaelii. Molecules 2012, 17, 9939–9946. [Google Scholar] [CrossRef] [PubMed]
- Compagnone, R.S.; Oliveri, M.C.; Piãna, I.C.; Marques, S.; Rangel, H.R.; Dagger, F.; Suárez, A.I.; Gómez, M. 5-alkylpyrrole-2-carboxaldehydes from the caribbean sponges mycale microsigmatosa and desmapsamma anchorata. Nat. Prod. Lett. 1999, 13, 203–211. [Google Scholar] [CrossRef]
- Mao, S.C.; Liu, Y.; Morgan, J.B.; Jekabsons, M.B.; Zhou, Y.D.; Nagle, D.G. Lipophilic 2,5-disubstituted pyrroles from the marine sponge mycale sp. inhibit mitochondrial respiration and hif-1 activation. J. Nat. Prod. 2009, 72, 1927–1936. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Fu, Y.; Xiong, J.; Li, M.; Ma, G.-L.; Yang, G.-X.; Wei, B.-G.; Zhao, Y.; Zhang, H.-Y.; Hu, J.-F.; et al. lycodine-type alkaloids from Lycopodiastrum casuarinoides. J. Nat. Prod. 2013, 76, 1475–1484. [Google Scholar] [CrossRef] [PubMed]
- Taveira, M.; Sousa, C.; Valentão, P.; Ferreres, F.; Teixeira, J.P.; Andrade, P.B. Neuroprotective effect of steroidal alkaloids on glutamate-induced toxicity by preserving mitochondrial membrane potential and reducing oxidative stress. J. Steroid Biochem. Mol. Biol. 2014, 140, 106–115. [Google Scholar] [CrossRef] [PubMed]
- Atanasova, M.; Stavrakov, G.; Philipova, I.; Zheleva, D.; Yordanov, N.; Doytchinova, I. Galantamine derivatives with indole moiety: Docking, design, synthesis and acetylcholinesterase inhibitory activity. Bioorganic Med. Chem. 2015, 23, 5382–5389. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, P.K.; Kumar, V.; Mal, M.; Houghton, P.J. Acetylcholinesterase inhibitors from plants. Phytomedicine 2007, 14, 289–300. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds 1–7 are available from the authors. |

| No. | 1 a | 2 a | 3 b | |||
|---|---|---|---|---|---|---|
| δC, Type | δH (J in Hz) | δC, Type | δH (J in Hz) | δC, Type | δH (J in Hz) | |
| 1 | -- | -- | -- | -- | -- | -- |
| 2 | 132.4, C | -- | 132.5, C | -- | 132.5, C | -- |
| 3 | 124.6 c, CH | 6.98, d (4.2) | 124.7 c, CH | 6.98, d (4.2) | 125.0, CH | 6.95, d (4.2) |
| 4 | 110.0, CH | 6.23, d (4.2) | 110.0, CH | 6.22, d (4.2) | 111.8, CH | 6.26, d (4.2) |
| 5 | 144.5, C | -- | 144.6, C | -- | 139.2 c, C | -- |
| 6 | 55.6, CH2 | 4.50, s | 55.6, CH2 | 4.49, s | 65.8, CH2 | 4.48, d (13.2); 4.42, d (13.2) |
| 7 | 179.5, CH | 9.44, s | 179.5, CH | 9.44, s | 178.9, CH | 9.43, s |
| 8 | -- | -- | -- | -- | 57.6, CH3 | 3.31, s |
| 1′ | 51.9, CH | 4.11, d (7.8) | 50.8, CH2 | 4.12, m; 4.20, m | 17.7, CH3 | 1.70, d (7.2) |
| 2′ | 30.1, CH | 2.00, m | 36.5, CH | 1.79, m | 54.7, CH | 4.00, m |
| 3′ | 19.9, CH3 | 0.80, d (7.2) | 26.7, CH2 | 1.12, m; 1.25, m | 170.1 c, C | -- |
| 4′ | 19.9, CH3 | 0.80, d (7.2) | 11.7, CH3 | 0.84, t (7.2) | 61.5, CH2 | 4.17, m |
| 5′ | 16.7, CH3 | 0.73, d (6.6) | 14.1, CH3 | 1.21, t (7.2) | ||
| Compounds | Cell Viability (% of Control) |
|---|---|
| 1 | 54.57 ± 0.97 a |
| 2 | 57.62 ± 3.42 |
| 3 | 60.27 ± 2.38 |
| 4 | 62.58 ± 3.32 |
| 5 | 64.43 ± 1.57 |
| 6 | 59.31 ± 2.01 |
| 7 | 75.23 ± 1.76 |
| N-acetyl-l-cysteine b | 73.03 ± 1.49 |
| H2O2 control | 48.73 ± 1.08 |
| Blank control | 100.00 ± 2.43 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, Z.; Hu, M.; Sun, Z.; Zhu, N.; Yang, J.; Ma, G.; Xu, X. Pyrrole Alkaloids from the Edible Mushroom Phlebopus portentosus with Their Bioactive Activities. Molecules 2018, 23, 1198. https://doi.org/10.3390/molecules23051198
Sun Z, Hu M, Sun Z, Zhu N, Yang J, Ma G, Xu X. Pyrrole Alkaloids from the Edible Mushroom Phlebopus portentosus with Their Bioactive Activities. Molecules. 2018; 23(5):1198. https://doi.org/10.3390/molecules23051198
Chicago/Turabian StyleSun, Zhaocui, Meigeng Hu, Zhonghao Sun, Nailiang Zhu, Junshan Yang, Guoxu Ma, and Xudong Xu. 2018. "Pyrrole Alkaloids from the Edible Mushroom Phlebopus portentosus with Their Bioactive Activities" Molecules 23, no. 5: 1198. https://doi.org/10.3390/molecules23051198
APA StyleSun, Z., Hu, M., Sun, Z., Zhu, N., Yang, J., Ma, G., & Xu, X. (2018). Pyrrole Alkaloids from the Edible Mushroom Phlebopus portentosus with Their Bioactive Activities. Molecules, 23(5), 1198. https://doi.org/10.3390/molecules23051198




