Transient Sulfenic Acids in the Synthesis of Biologically Relevant Products
Abstract
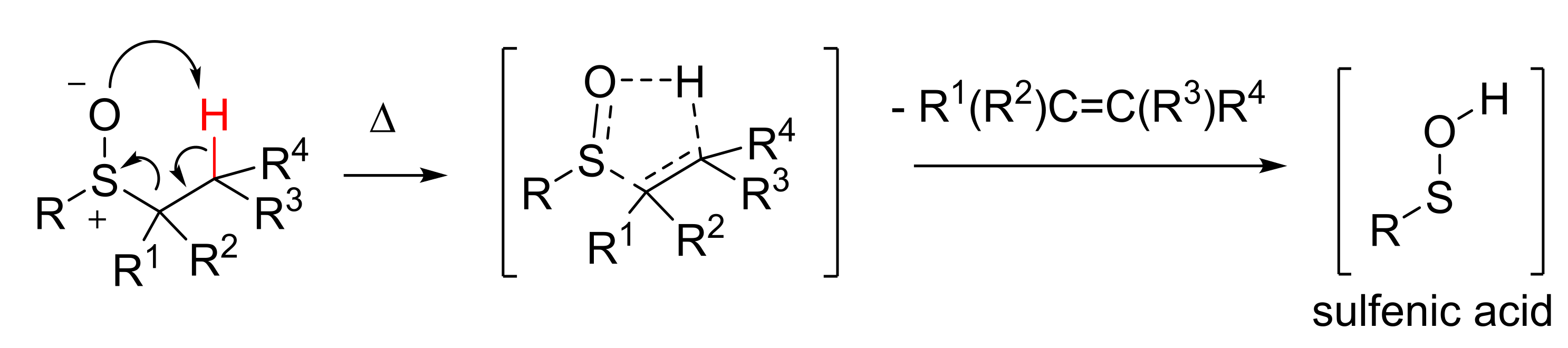
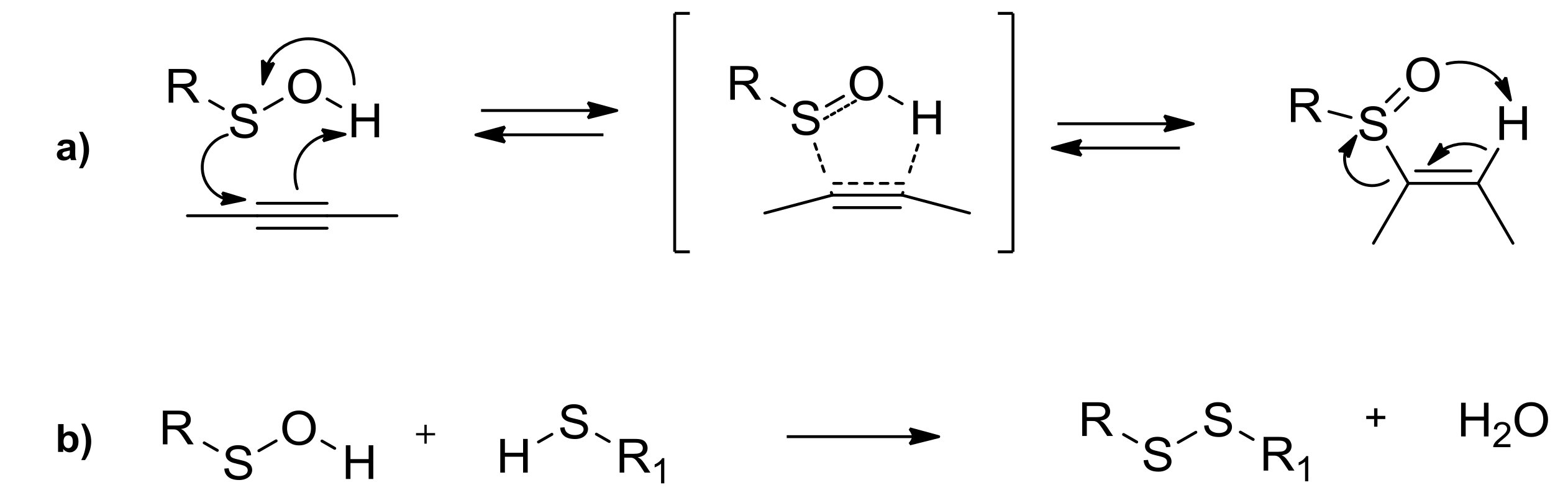
1. Introduction
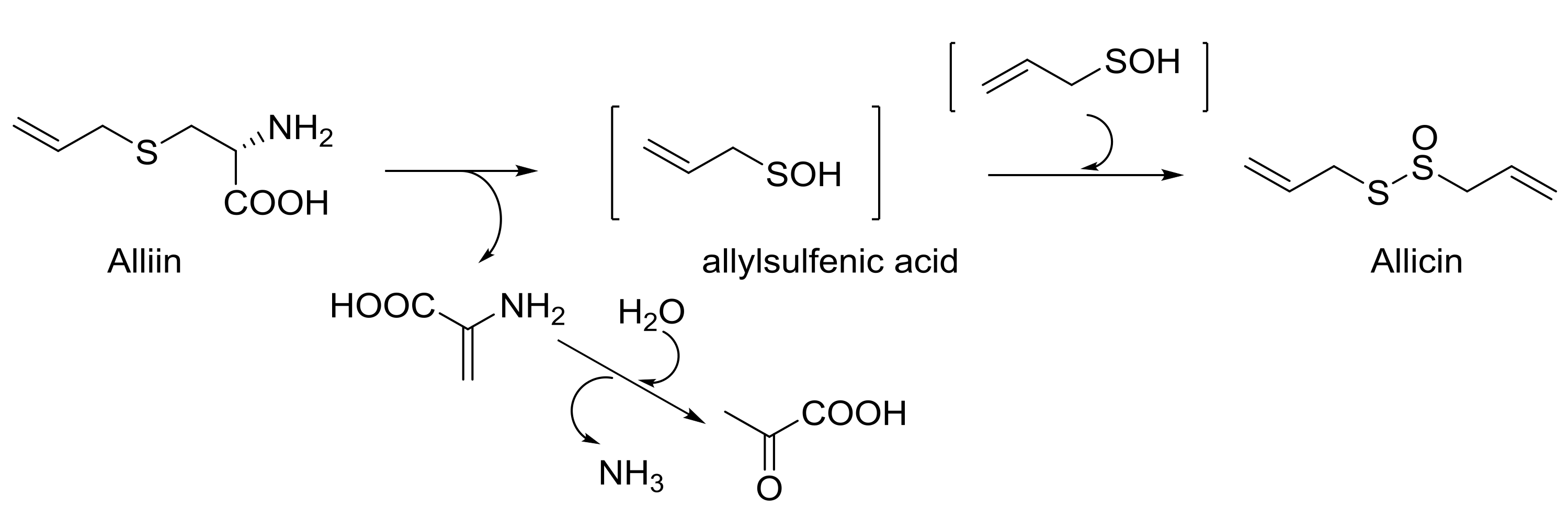
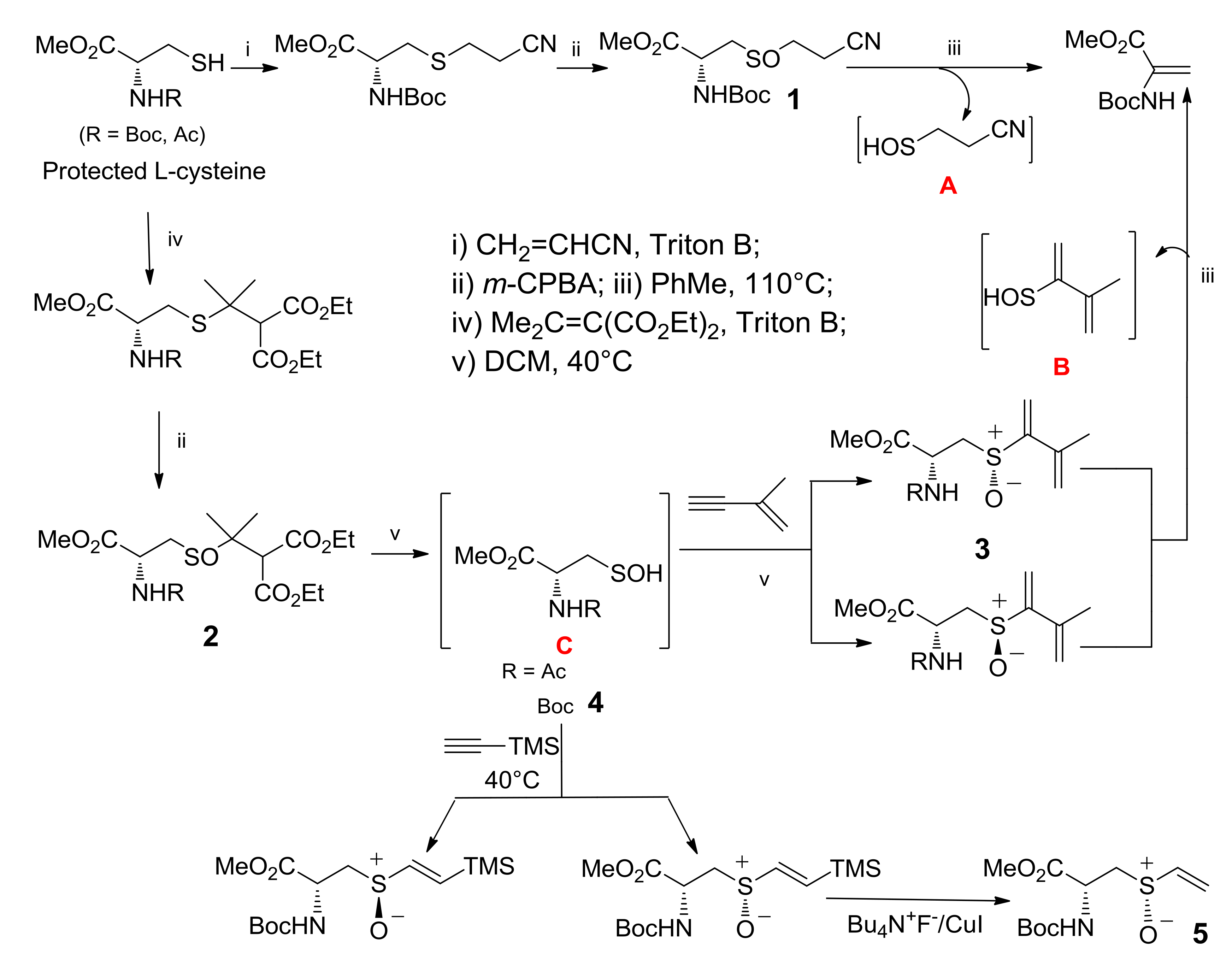
2. Alliin Derivatives
3. Thiosaccharides
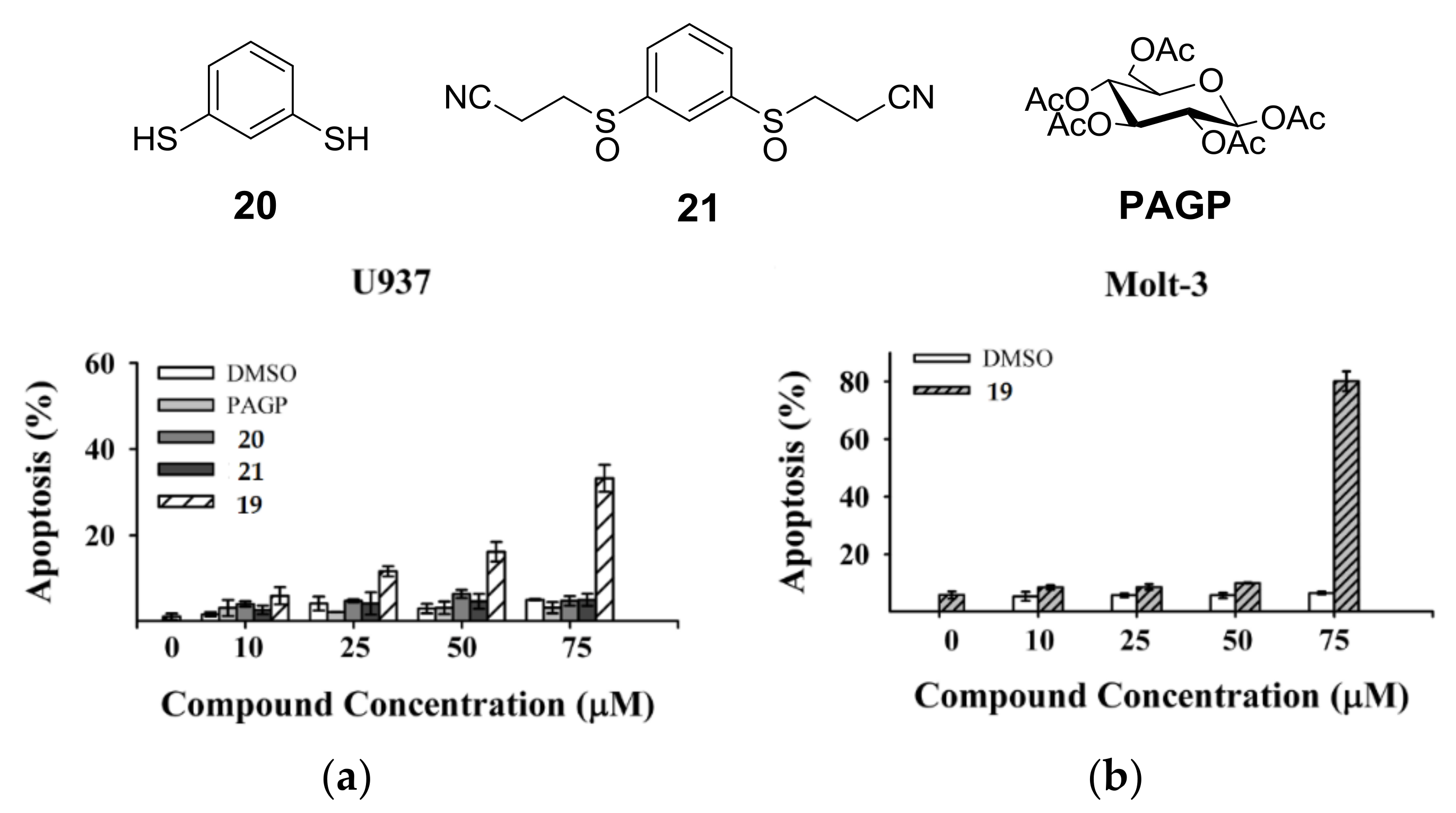
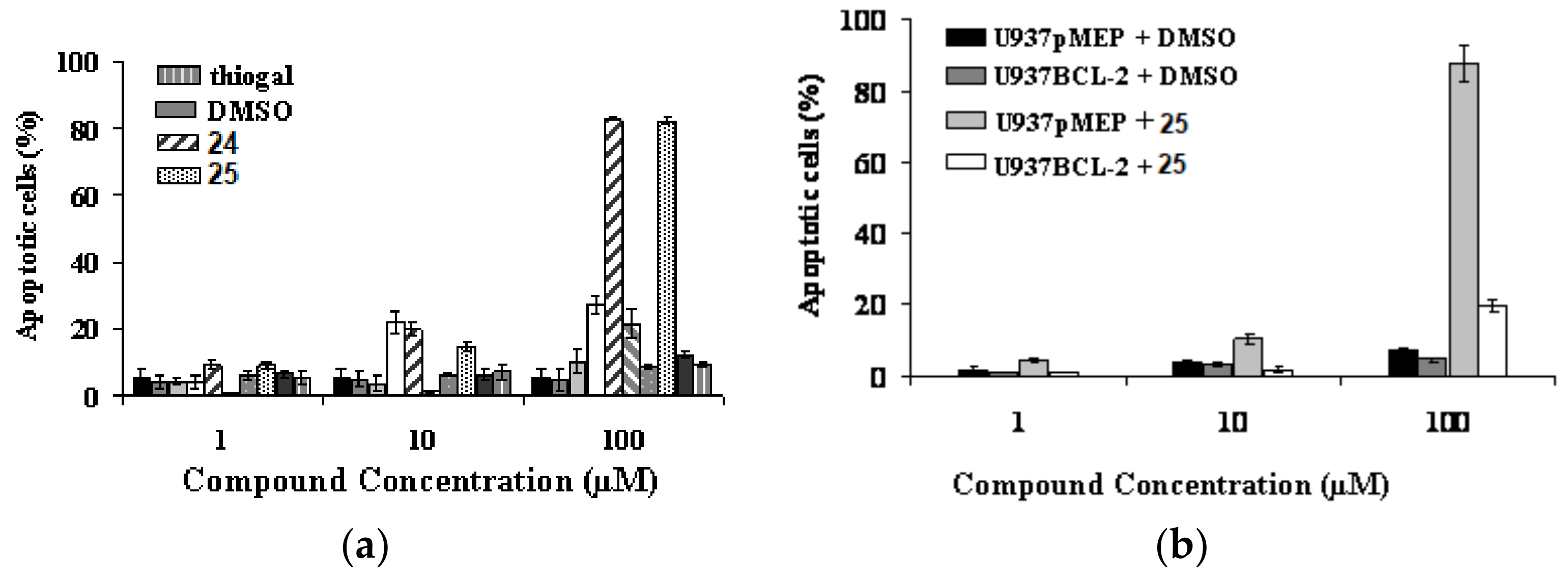
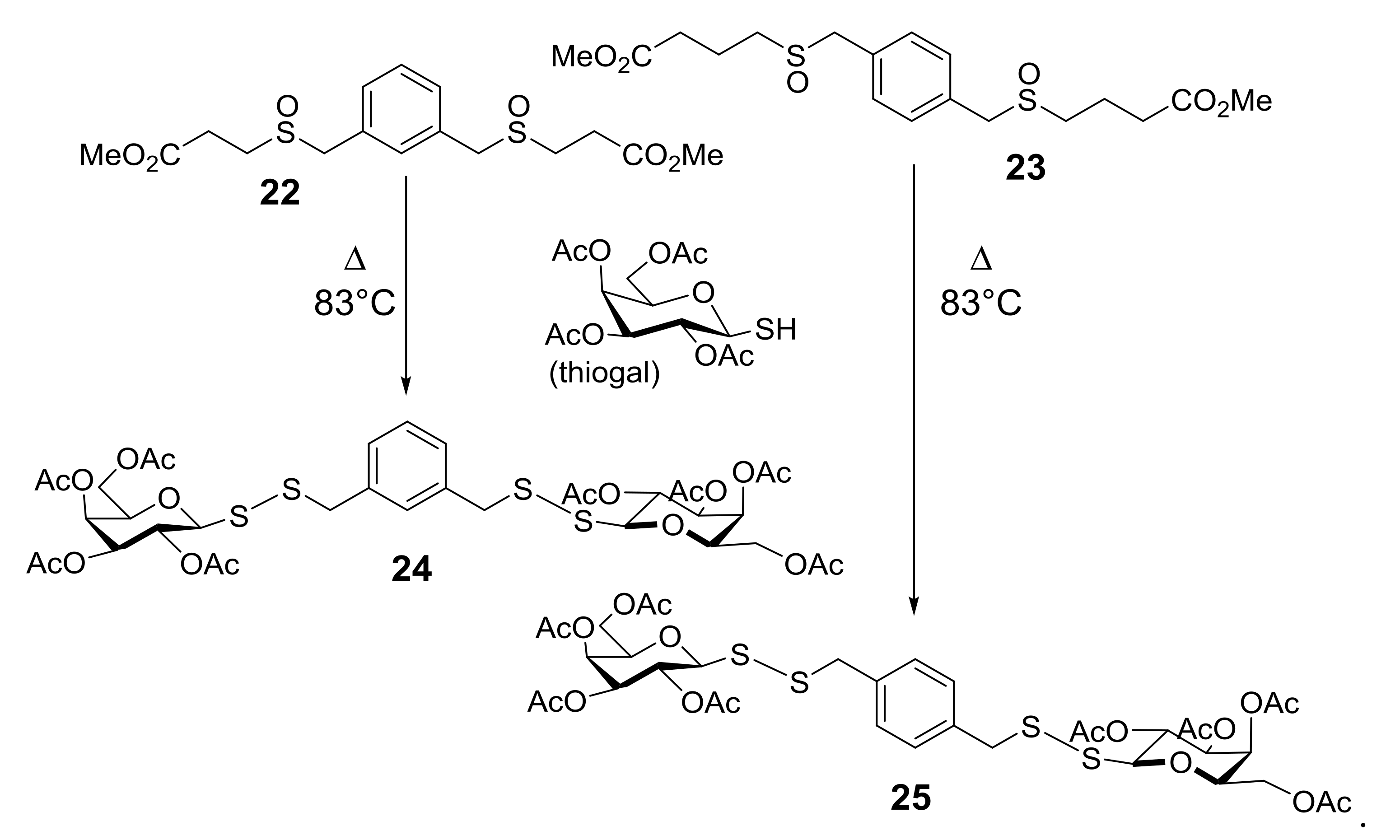
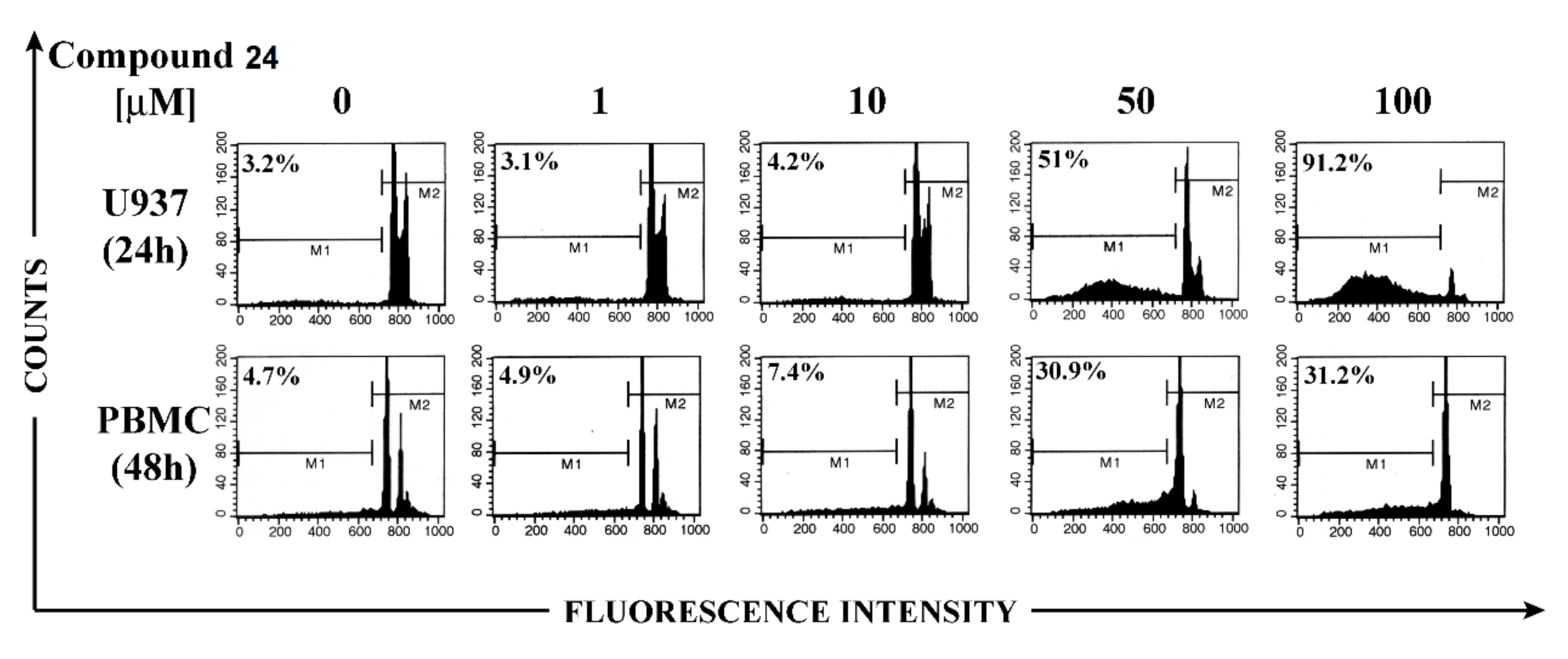
4. Glycoconjugated Disulfoxides and Disulfides
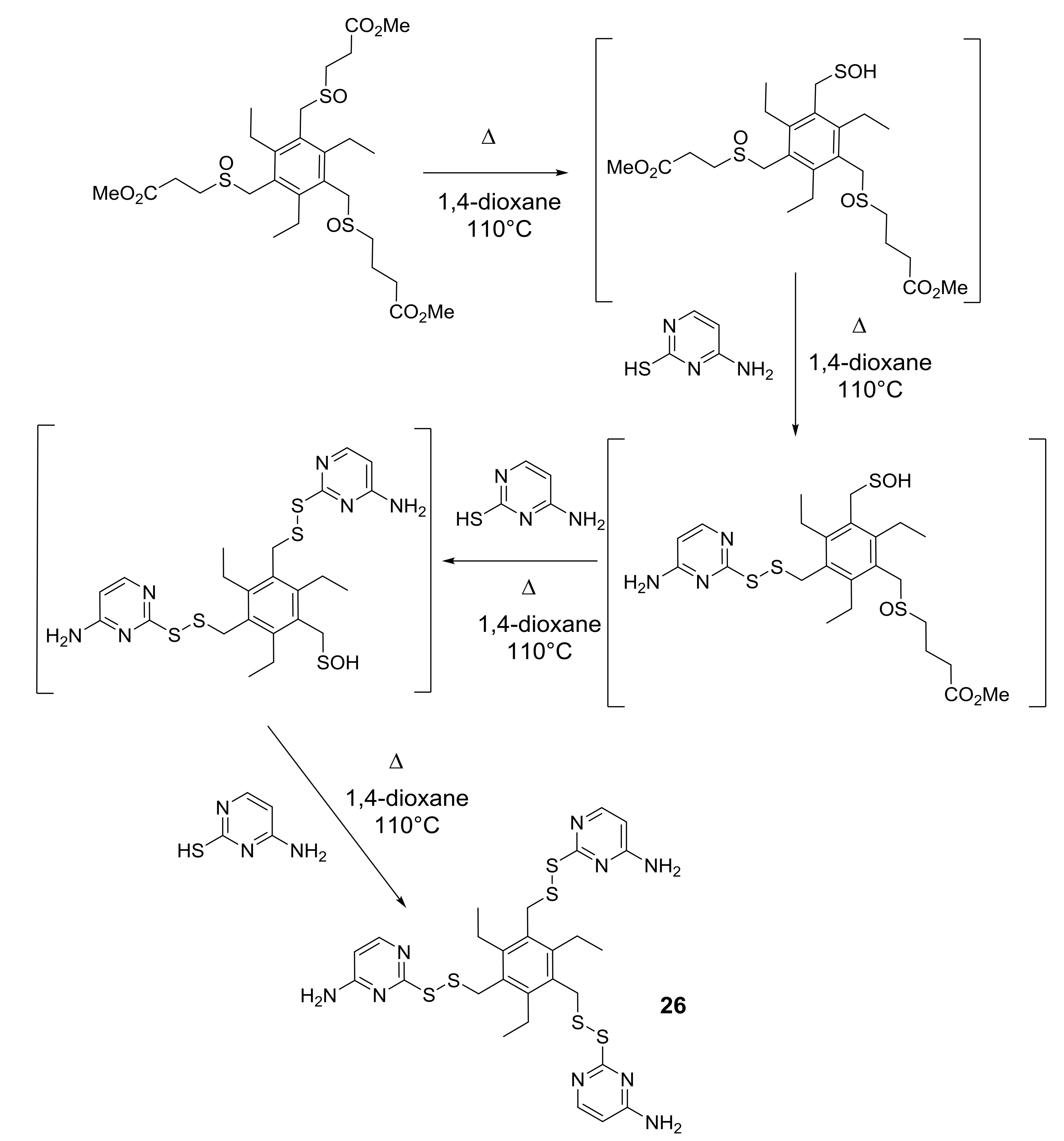
5. Pyrimidine-Derived Disulfides
6. Conclusions
Conflicts of Interest
Abbreviations
| DCE | 1,2-dichloroethane |
| DCM | dichloromethane |
| DMSO | dimethylsulfoxide |
| MTS | 5-[3-(carboxymethoxy)phenyl]-3-(4,5-dimethyl-2-thiazolyl)-2-(4-sulfophenyl)-2H-tetrazolium inner salt |
| PAGP | 1,2,3,4,6-penta-O-acetyl-β-d-glucopyranose |
| RT | room temperature |
| THF | tetrahydrofuran |
| Triton B | benzyltrimethylammonium hydroxide |
References
- Feng, M.; Tang, B.; Liang, S.H.; Jiang, X. Sulfur Containing Scaffolds in Drugs: Synthesis and Application in Medicinal Chemistry. Curr. Top. Med. Chem. 2016, 16, 1200–1216. [Google Scholar] [CrossRef] [PubMed]
- Ilardi, E.A.; Vitaku, E.; Njardarson, J.T. Data-Mining for Sulfur and Fluorine: An Evaluation of Pharmaceuticals to Reveal Opportunities for Drug Design and Discovery. J. Med. Chem. 2014, 57, 2832–2842. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Soloshonok, V.A.; Klika, K.D.; Drabowicz, J.; Wzorek, A. Chiral sulfoxides: Advances in asymmetric synthesis and problems with the accurate determination of the stereochemical outcome. Chem. Soc. Rev. 2018, 47, 1307–1350. [Google Scholar] [CrossRef] [PubMed]
- Bentley, R. Role of sulfur chirality in the chemical processes of biology. Chem. Soc. Rev. 2005, 34, 609–624. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.-W.; Liang, S.; Manolikakes, G. Recent Advances in the Synthesis of Sulfones. Synthesis 2016, 48, 1939–1973. [Google Scholar] [CrossRef]
- Meadows, D.C.; Gervay-Hague, J. Vinyl sulfones: Synthetic preparations and medicinal chemistry applications. Med. Res. Rev. 2006, 26, 793–814. [Google Scholar] [CrossRef] [PubMed]
- Shiri, L.; Ghorbani-Choghamarani, A.; Kazemi, M. S–S Bond Formation: Nanocatalysts in the Oxidative Coupling of Thiols. Aust. J. Chem. 2016, 70, 9–25. [Google Scholar] [CrossRef]
- Lee, M.H.; Yang, Z.; Lim, C.W.; Lee, Y.H.; Dongbang, S.; Kang, C.; Kim, J.S. Disulfide-Cleavage-Triggered Chemosensors and Their Biological Applications. Chem. Rev. 2013, 113, 5071–5109. [Google Scholar] [CrossRef] [PubMed]
- Rahman, I.; MacNee, W. Regulation of redox glutathione levels and gene transcription in lung inflammation: Therapeutic approaches. Free Radic. Biol. Med. 2000, 28, 1405–1420. [Google Scholar] [CrossRef]
- Block, E. The Organosulfur Chemistry of the Genus Allium—Implications for the Organic Chemistry of Sulfur. Angew. Chem. Int. Ed. Engl. 1992, 31, 1135–1178. [Google Scholar] [CrossRef]
- Gupta, V.; Carroll, K.S. Sulfenic acid chemistry, detection and cellular lifetime. Biochim. Biophys. Acta 2014, 1840, 847–875. [Google Scholar] [CrossRef] [PubMed]
- Goto, K.; Tokitoh, N.; Okazaki, R. Synthesis of a Stable Arenesulfenic Acid Bearing a Bowl-Shaped Macrobicyclic Cyclophane Skeleton. Angew. Chem. Int. Ed. 1995, 34, 1124–1126. [Google Scholar] [CrossRef]
- Aversa, M.C.; Barattucci, A.; Bonaccorsi, P.; Temperini, A. Regio- and Stereocontrolled Synthesis of (Z)-α-(Phenylseleno)sulfinyl and -sulfonyl Alkenes via Sulfenic Acids, and a Study of their Reactivity. Eur. J. Org. Chem. 2011, 5668–5673. [Google Scholar] [CrossRef]
- Minuti, L.; Barattucci, A.; Bonaccorsi, P.M.; Di Gioia, M.L.; Leggio, A.; Siciliano, C.; Temperini, A. Intramolecular Displacement of Phenylselenone by a Hydroxy Group: Stereoselective Synthesis of 2-Substituted Tetrahydrofurans. Org. Lett. 2013, 15, 3906–3909. [Google Scholar] [CrossRef] [PubMed]
- Aversa, M.C.; Barattucci, A.; Bonaccorsi, P.; Giannetto, P. Recent Advances and Perspectives in the Chemistry of Sulfenic Acids. Curr. Org. Chem. 2007, 11, 1034–1052. [Google Scholar] [CrossRef]
- Pan, J.; Carroll, K.S. Light-Mediated Sulfenic Acid Generation from Photocaged Cysteine Sulfoxide. Org. Lett. 2015, 17, 6014–6017. [Google Scholar] [CrossRef] [PubMed]
- Adams, H.; Anderson, J.C.; Bell, R.; Jones, D.N.; Peel, M.R.; Tomkinson, N.C.O. The synthesis and Diels–Alder reactions of (E)- and (Z)-1-methoxy-3-(phenylsulfinyl)buta-1,3-dienes. J. Chem. Soc. Perkin Trans. 1 1998, 3967–3974. [Google Scholar] [CrossRef]
- Aversa, M.C.; Barattucci, A.; Bonaccorsi, P.; Rollin, P.; Tatibouët, A. 2,2-Bis(phenylsulfonyl)ethyl sulfides as efficient precursors of sulfenic acids. Arkivoc 2009, 8, 187–198. [Google Scholar] [CrossRef]
- Aversa, M.C.; Barattucci, A.; Bonaccorsi, P.; Contini, A. Addition of sulfenic acids to monosubstituted acetylenes: A theoretical and experimental study. J. Phys. Org. Chem. 2009, 22, 1048–1057. [Google Scholar] [CrossRef]
- Gelat, F.; Jayashankaran, J.; Lohier, J.F.; Gaumont, A.C.; Perrio, S. Organocatalytic Asymmetric Synthesis of Sulfoxides from Sulfenic Acid Anions Mediated by a Cinchona-Derived PhaseTransfer Reagent. Org. Lett. 2011, 13, 3170–3173. [Google Scholar] [CrossRef] [PubMed]
- Aversa, M.C.; Barattucci, A.; Bonaccorsi, P.; Faggi, C.; Gacs-Baitz, E.; Marrocchi, A.; Minuti, L.; Taticchi, A. Maleimide cycloadditions by sulfinyldienes: Is the sulfur configuration the only controller of the diastereofacial selectivity? Tetrahedron 2005, 61, 7719–7726. [Google Scholar] [CrossRef]
- Aversa, M.C.; Barattucci, A.; Bonaccorsi, P.; Faggi, C.; Papalia, T. Thiacyclophane Cages and Related Bi- and Tripodal Molecules via Transient Polysulfenic Acids. J. Org. Chem. 2007, 72, 4486–4496. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Lei, X.; Fan, E.; Sun, Z. Thermolysis-Induced Two- or Multicomponent Tandem Reactions Involving Isocyanides and Sulfenic-Acid-Generating Sulfoxides: Access to Diverse Sulfur-Containing Functional Scaffolds. Org. Lett. 2018, 20, 522–525. [Google Scholar] [CrossRef] [PubMed]
- Aversa, M.C.; Barattucci, A.; Bonaccorsi, P. Efficient Synthesis of Unsymmetrical Disulfides through Sulfenic Acids. Eur. J. Org. Chem. 2009, 6355–6359. [Google Scholar] [CrossRef]
- Gu, X.; Zhu, Y.Z. Therapeupic applications of organosulfur compounds as novel hydrogen sulfide donors and/or mediators. Expert Rev. Clin. Pharmacol. 2011, 4, 123–133. [Google Scholar] [CrossRef] [PubMed]
- Augusti, K.T.; Sheela, C.G. Antiperoxide effect of S-allyl cysteine sulfoxide, an insulin secretagogue, in diabetic rats. Experientia 1996, 52, 115–120. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Cheng, B.; Meng, Z.; Fang, B.; Li, T.; Sun, M.; Liu, M.; Guan, S. Alliin attenuates 1,3-dichloro-2-propanol-induced lipogenesis in HepG2 cells through activation of the AMP-activated protein kinase-dependent pathway. Life Sci. 2018, 195, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Granroth, B. Biosynthesis and decomposition of cysteine derivatives in onion and other Allium species. Ann. Acad. Sci. Fenn. Chem. 1970, 154, 4–71. [Google Scholar]
- Borlinghaus, J.; Albrecht, F.; Gruhlke, M.C.H.; Nwachukwu, I.D.; Slusarenko, A.J. Allicin: Chemistry and Biological Properties. Molecules 2014, 19, 12591–12618. [Google Scholar] [CrossRef] [PubMed]
- Aversa, M.C.; Barattucci, A.; Bonaccorsi, P.; Giannetto, P. l-Cysteine, a Versatile Source of Sulfenic Acids. Synthesis of Enantiopure Alliin Analogues. J. Org. Chem. 2005, 70, 1986–1992. [Google Scholar] [CrossRef] [PubMed]
- Brás, N.F.; Cerqueira, N.M.; Ramos, M.J.; Fernandes, P.A. Glycosidase inhibitors: A patent review (2008–2013). Expert Opin. Ther. Pat. 2014, 24, 857–874. [Google Scholar] [CrossRef] [PubMed]
- Wadood, A.; Ghufran, M.; Khan, A.; Azam, S.S.; Jelani, M.; Uddin, R. Selective glycosidase inhibitors: A patent review (2012-present). Int. J. Biol. Macromol. 2018, 111, 82–91. [Google Scholar] [CrossRef] [PubMed]
- Tamura, Y.; Miyagawa, H.; Yoshida, T.; Chuman, H. Binding interaction of SGLT with sugar and thiosugar by the molecular dynamics simulation. Biochim. Biophys. Acta Biomembr. 2015, 1848, 2799–2804. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Witczak, Z.J. Thio Sugars: Biological Relevance as Potential New Therapeutics. Curr. Med. Chem. 1999, 6, 165–178. [Google Scholar] [CrossRef] [PubMed]
- Aucagne, V.; Aversa, M.C.; Barattucci, A.; Bonaccorsi, P.; Giannetto, P.; Rollin, P.; Tatibouët, A. Sulfenic Acids in the Carbohydrate Field. Synthesis of Transient Glycosulfenic Acids and Their Addition to Unsaturated Acceptors. J. Org. Chem. 2002, 67, 6925–6930. [Google Scholar] [CrossRef] [PubMed]
- Aversa, M.C.; Barattucci, A.; Bilardo, M.C.; Bonaccorsi, P.; Giannetto, P.; Rollin, P.; Tatibouët, A. Sulfenic Acids in the Carbohydrate Field. An Example of Straightforward Access to Novel Multivalent Thiosaccharides. J. Org. Chem. 2005, 70, 7389–7396. [Google Scholar] [CrossRef] [PubMed]
- Ramström, O.; Lehn, J.-M. In Situ Generation and Screening of a Dynamic Combinatorial Carbohydrate Library against Concanavalin A. ChemBioChem 2000, 1, 41–48. [Google Scholar] [CrossRef]
- Barattucci, A.; Di Gioia, M.L.; Leggio, A.; Minuti, L.; Papalia, T.; Siciliano, C.; Temperini, A.; Bonaccorsi, P. Stereoselective Synthesis of Dithia[3.3]cyclophane S,S′-Dioxides with Planar and Central Chirality. Eur. J. Org. Chem. 2014, 2099–2104. [Google Scholar] [CrossRef]
- Barattucci, A.; Bonaccorsi, P.; Papalia, T.; Manganaro, N.; Gattuso, G. Kinetic control in the formation of meso-dithia[3.3]-paracyclophane S,S′-dioxide. Tetrahedron Lett. 2014, 55, 5096–5100. [Google Scholar] [CrossRef]
- Galan, M.C.; Benito-Alifonso, D.; Watt, G.M. Carbohydrate chemistry in drug discovery. Org. Biomol. Chem. 2011, 9, 3598–3610. [Google Scholar] [CrossRef] [PubMed]
- Aversa, M.C.; Barattucci, A.; Bonaccorsi, P.; Marino-Merlo, F.; Mastino, A.; Sciortino, M.T. Synthesis and biological testing of thioalkane- and thioarene-spaced bis-d-glucopyranosides. Bioorg. Med. Chem. 2009, 17, 1456–1463. [Google Scholar] [CrossRef] [PubMed]
- Bonaccorsi, P.; Marino-Merlo, F.; Barattucci, A.; Battaglia, G.; Papaianni, E.; Papalia, T.; Aversa, M.C.; Mastino, A. Synthesis and biological evaluation of a new class of glycoconjugated disulfides that exhibit potential anticancer properties. Bioorg. Med. Chem. 2012, 20, 3186–3195. [Google Scholar] [CrossRef] [PubMed]
- Siciliano, C.; Barattucci, A.; Bonaccorsi, P.; Di Gioia, M.L.; Leggio, A.; Minuti, L.; Romio, E.; Temperini, A. Synthesis of d-erythro-Sphinganine through Serine-Derived α-Amino Epoxides. J. Org. Chem. 2014, 79, 5320–5326. [Google Scholar] [CrossRef] [PubMed]
- Kaźmierczak, Z.; Górski, A.; Dąbrowska, K. Facing antibiotic resistance: Staphylococcus aureus phages as a medical tool. Viruses 2014, 6, 2551–2570. [Google Scholar] [CrossRef] [PubMed]
- Jubeen, F.; Iqbal, S.Z.; Shafiq, N.; Khan, M.; Parveen, S.; Iqbal, M.; Nazir, A. Eco-friendly synthesis of pyrimidines and its derivatives: A review on broad spectrum bioactive moiety with huge therapeutic profile. Synth. Commun. 2018, 48, 601–625. [Google Scholar] [CrossRef]
- Aversa, M.C.; Barattucci, A.; Bonaccorsi, P. From Transient Sulfenic Acids to Disulfide-Functionalized Tripodal Structures. Synlett 2011, 254–258. [Google Scholar] [CrossRef]
- Bonaccorsi, P.; Barattucci, A.; Papalia, T.; Criseo, G.; Faggio, C.; Romeo, O. Pyrimidine-derived disulfides as potential antimicrobial agents: Synthesis and evaluation in vitro. J. Sulfur Chem. 2015, 36, 317–325. [Google Scholar] [CrossRef]



© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barattucci, A.; Aversa, M.C.; Mancuso, A.; Salerno, T.M.G.; Bonaccorsi, P. Transient Sulfenic Acids in the Synthesis of Biologically Relevant Products. Molecules 2018, 23, 1030. https://doi.org/10.3390/molecules23051030
Barattucci A, Aversa MC, Mancuso A, Salerno TMG, Bonaccorsi P. Transient Sulfenic Acids in the Synthesis of Biologically Relevant Products. Molecules. 2018; 23(5):1030. https://doi.org/10.3390/molecules23051030
Chicago/Turabian StyleBarattucci, Anna, Maria Chiara Aversa, Aurora Mancuso, Tania Maria Grazia Salerno, and Paola Bonaccorsi. 2018. "Transient Sulfenic Acids in the Synthesis of Biologically Relevant Products" Molecules 23, no. 5: 1030. https://doi.org/10.3390/molecules23051030
APA StyleBarattucci, A., Aversa, M. C., Mancuso, A., Salerno, T. M. G., & Bonaccorsi, P. (2018). Transient Sulfenic Acids in the Synthesis of Biologically Relevant Products. Molecules, 23(5), 1030. https://doi.org/10.3390/molecules23051030






