Profiling of Heterobranchia Sea Slugs from Portuguese Coastal Waters as Producers of Anti-Cancer and Anti-Inflammatory Agents
Abstract
:1. Introduction
2. Results
2.1. Chemical Characterization
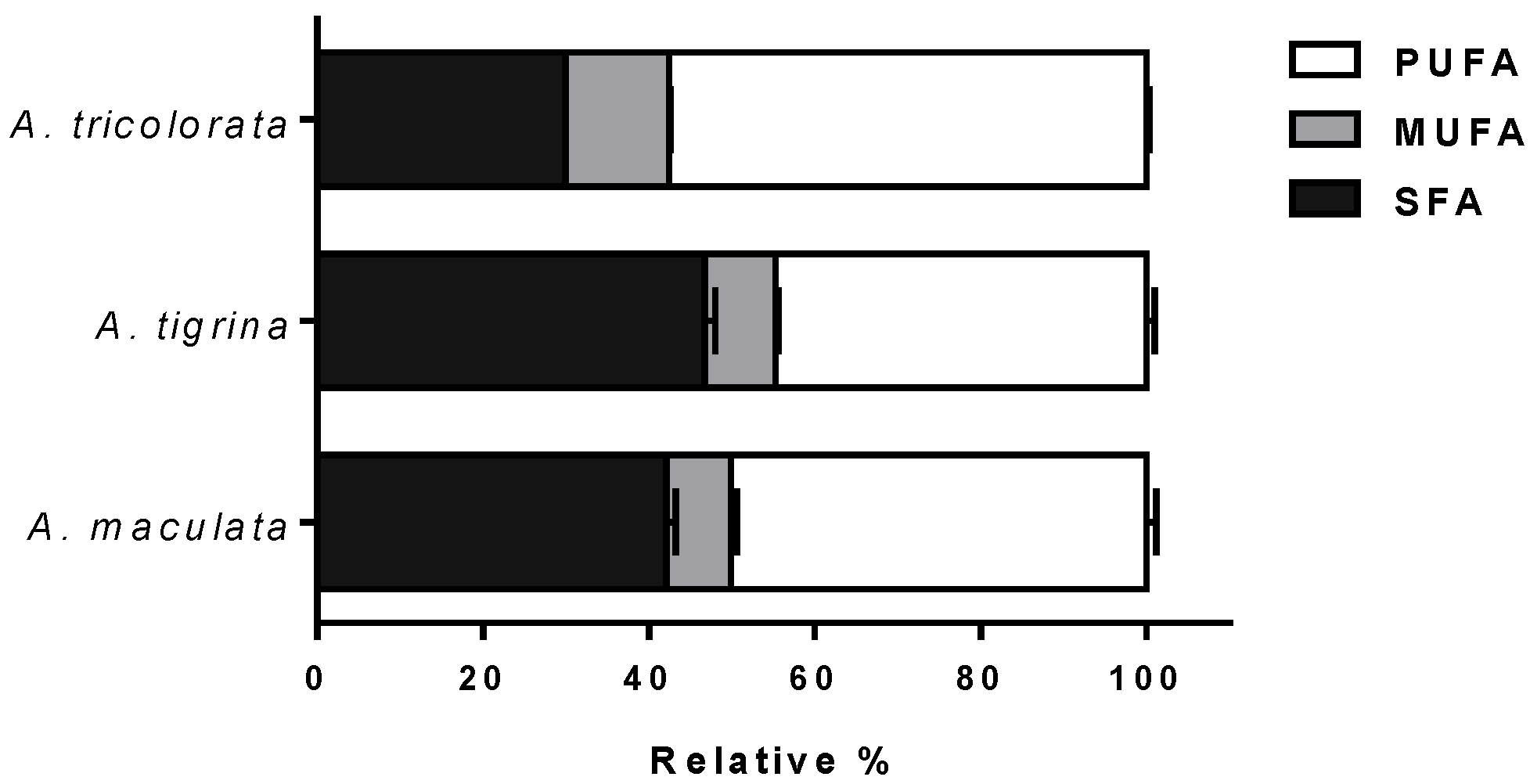
2.1.1. Fatty Acid Profiles
2.1.2. Homarine
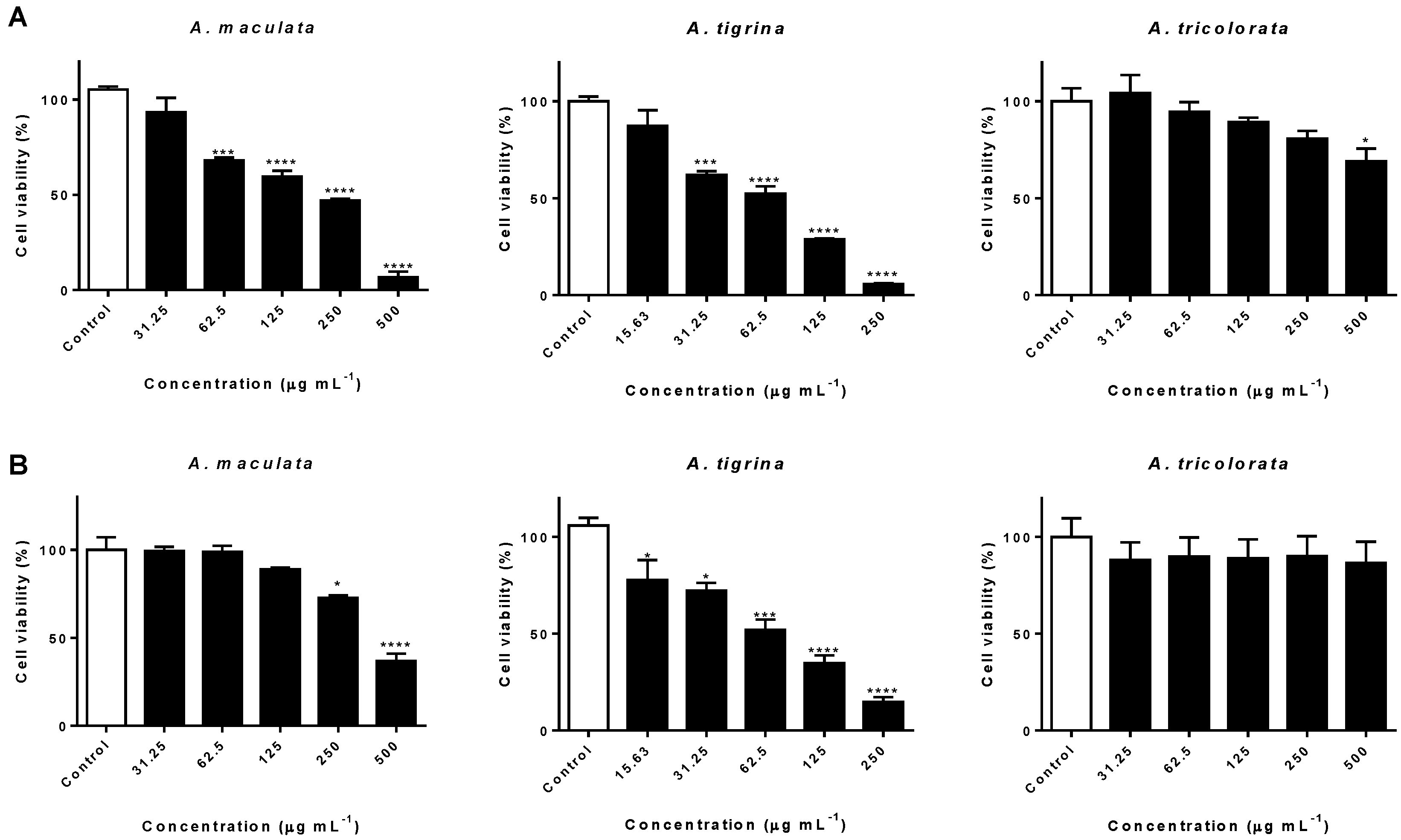
2.2. Impact on AGS and A549 Cancer Cells
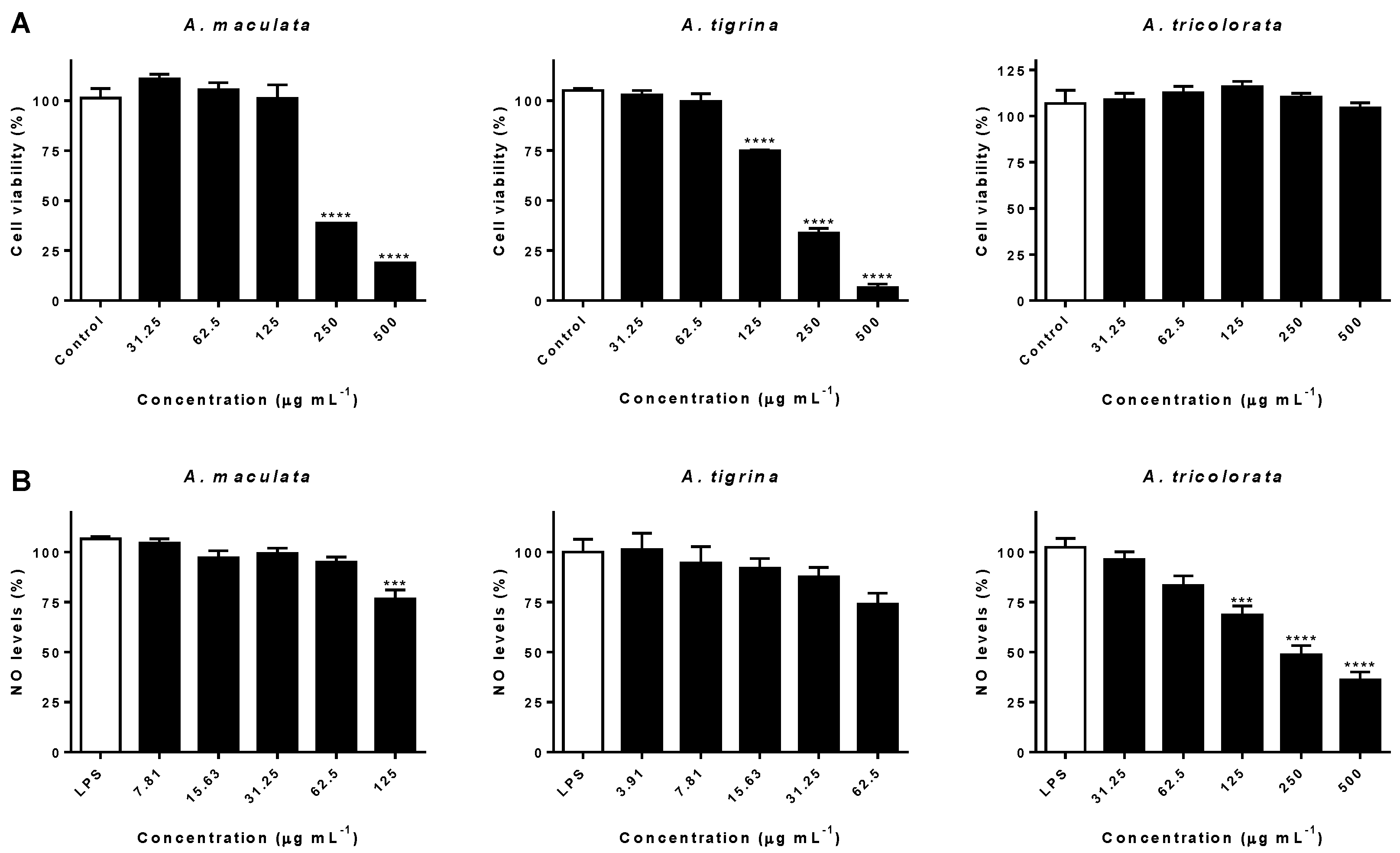
2.3. Impact on RAW 264.7 Macrophages and Interference on NO Levels
2.4. Screening for Antimicrobial Activity
3. Discussion
3.1. Chemical Characterization
3.1.1. Fatty Acid Profiles
3.1.2. Homarine
3.2. Impact on AGS and A549 Cancer Cells
3.3. Impact on RAW 264.7 Macrophages and Interference on NO Levels
4. Materials and Methods
4.1. General Chemicals and Materials
4.2. Animal Material and Extraction
4.3. Chemical Characterization
4.3.1. Standards
4.3.2. GC-FID Qualitative and Quantitative Analysis
4.3.3. HPLC-DAD Qualitative and Quantitative Analysis
4.4. Cell Culture and Cytotoxicity
4.5. Determination of NO Levels
4.6. Antimicrobial Activity
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| ATCC | American Type Culture Collection |
| CLSI | Clinical Laboratory Standards Institute |
| DAD | Diode-array detector |
| DMSO | Dimethyl sulfoxide |
| DMEM | Dulbecco’s modified Eagle medium |
| ECACC | European Collection of Authenticated Cell Cultures |
| FAME | Fatty acid methyl esters |
| FBS | Fetal bovine serum |
| GC-FID | Gas-chromatography-Flame Ionization Detection |
| HPLC | High-performance liquid chromatography |
| IC50 | Half maximal inhibitory concentration |
| LPS | Lipopolysaccharide |
| MIC | Minimum inhibitory concentration |
| MTT | Thiazolyl blue tetrazolium bromide |
| MUFA | Monounsaturated fatty acids |
| NO | Nitric oxide |
| PCA | Principal Component Analysis |
| PPAR | Peroxisome proliferator-activated receptors |
| PUFA | Polyunsaturated fatty acids |
| ROS | Reactive oxygen species |
| SEM | Standard error of the mean |
| SFA | Saturated fatty acids |
| UFA | Unsaturated fatty acids |
References
- Gomes, N.G.M.; Dasari, R.; Chandra, S.; Kiss, R.; Kornienko, A. Marine invertebrate metabolites with anticancer activities: Solutions to the “supply problem”. Mar. Drugs 2015, 98. [Google Scholar] [CrossRef] [PubMed]
- Pereira, R.B.; Dasari, R.; Lefranc, F.; Kornienko, A.; Kiss, R.; Gomes, N.G.M. Targeting enzymatic pathways with marine-derived clinical agents. In Natural Products Targeting Clinically Relevant Enzymes; Andrade, P.B., Valentão, P., Pereira, D.M., Eds.; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2017; pp. 255–275. [Google Scholar] [CrossRef]
- Benkendorff, K. Molluscan biological and chemical diversity: Secondary metabolites and medicinal resources produced by marine molluscs. Biol. Rev. Camb. Philos. Soc. 2010, 85, 757–775. [Google Scholar] [CrossRef] [PubMed]
- Pereira, R.B.; Andrade, P.B.; Valentão, P. Chemical diversity and biological properties of secondary metabolites from sea hares of Aplysia genus. Mar. Drugs 2016, 14, 39. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Chen, J.; Hu, G.; Yu, J.; Zhu, X.; Lin, Y.; Chen, S.; Yuan, J. Statistical research on the bioactivity of new marine natural products discovered during the 28 years from 1985 to 2012. Mar. Drugs 2015, 13, 202–221. [Google Scholar] [CrossRef] [PubMed]
- Leal, M.C.; Puga, J.; Serôdio, J.; Gomes, N.C.M.; Calado, R. Trends in the discovery of new marine natural products from invertebrates over the last two decades—Where and what are we bioprospecting? PLoS ONE 2012, 7, e30580. [Google Scholar] [CrossRef] [PubMed]
- Báez, D.P.; Ardila, N.; Valdés, Á.; Acero, A. Taxonomy and phylogeny of Armina (Gastropoda: Nudibranchia: Arminidae) from the Atlantic and eastern Pacific. J. Mar. Biol. Assoc. UK 2011, 91, 1107–1121. [Google Scholar] [CrossRef]
- Zamora-Silva, A.; Malaquias, A.E. Diet preferences of the Aglajidae: A family of cephalaspidean gastropod predators on tropical and temperate shores. J. Mar. Biol. Assoc. UK 2015, 96, 1101–1112. [Google Scholar] [CrossRef]
- Camacho-García, Y.E.; Ornelas-Gatdula, E.; Gosliner, T.M.; Valdés, Á. Phylogeny of the family Aglajidae (Pilsbry, 1895) (Heterobranchia: Cephalaspidea) inferred from mtDNA and nDNA. Mol. Phylogenet. Evol. 2014, 71, 113–126. [Google Scholar] [CrossRef] [PubMed]
- Domènech, A.; Avila, C.; Ballesteros, M. Opisthobranch molluscs from the subtidal trawling grounds off Blanes (Girona, north-east Spain). J. Mar. Biol. Assoc. UK 2006, 86, 383–389. [Google Scholar] [CrossRef]
- Guerriero, A.; D’Ambrosio, M.; Pietra, F. Isolation of the cembranoid preverecynarmin alongside some briaranes, the verecynarmins, from both the nudibranch mollusc Armina maculata and the octocoral Veretillum cynomorium of the east Pyrenean Mediterranean sea). Helv. Chim. Acta 1990, 73, 277–283. [Google Scholar] [CrossRef]
- Pereira, D.M.; Vinholes, J.; Correia-da-Silva, G.; Valentão, P.; Teixeira, N.; Andrade, P.B. Fatty acids in marine organisms: In the pursuit of bioactive agents. Curr. Pharm. Anal. 2011, 7, 108–119. [Google Scholar] [CrossRef]
- Figueiredo, C.; Baptista, M.; Rosa, I.C.; Lopes, A.R.; Dionísio, G.; Rocha, R.J.M.; Cruz, I.C.S.; Kikuchi, R.K.P.; Simões, N.; Leal, M.C.; et al. 3D chemoecology and chemotaxonomy of corals using fatty acid biomarkers: Latitude, longitude and depth. Biochem. Syst. Ecol. 2017, 70, 35–42. [Google Scholar] [CrossRef]
- Lopes, A.R.; Baptista, M.; Rosa, I.C.; Dionísio, G.; Gomes-Pereira, J.; Paula, J.R.; Figueiredo, C.; Bandarra, N.; Calado, R.; Rosa, R. “Gone with the wind”: Fatty acid biomarkers and chemotaxonomy of stranded pleustonic hydrozoans (Velella velella and Physalia physalis). Biochem. Syst. Ecol. 2016, 66, 297–306. [Google Scholar] [CrossRef]
- Gleissman, H.; Johnsen, J.I.; Kogner, P. Omega-3 fatty acids in cancer, the protectors of good and the killers of evil? Exp. Cell Res. 2010, 316, 1365–1373. [Google Scholar] [CrossRef] [PubMed]
- Vaughan, V.C.; Hassing, M.R.; Lewandowski, P.A. Marine polyunsaturated fatty acids and cancer therapy. Br. J. Cancer 2013, 108, 486–492. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Marine omega-3 fatty acids and inflammatory processes: Effects, mechanisms and clinical relevance. Biochim. Biophys. Acta 2015, 1851, 469–484. [Google Scholar] [CrossRef] [PubMed]
- Putz, A.; König, G.M.; Wägele, H. Defensive strategies of Cladobranchia (Gastropoda, Opisthobranchia). Nat. Prod. Rep. 2010, 27, 1386–1402. [Google Scholar] [CrossRef] [PubMed]
- Mariotto, S.; Menegazzi, M.; Suzuki, H. Biochemical aspects of nitric oxide. Curr. Pharm. Des. 2004, 10, 1627–1645. [Google Scholar] [CrossRef] [PubMed]
- Zhukova, N.V. Lipids and fatty acids of nudibranch mollusks: Potential sources of bioactive compounds. Mar. Drugs 2014, 12, 4578–4592. [Google Scholar] [CrossRef] [PubMed]
- Monroig, Ó.; Tocher, D.R.; Navarro, J.C. Biosynthesis of polyunsaturated fatty acids in marine invertebrates: Recent advances in molecular mechanisms. Mar. Drugs 2013, 11, 3998–4018. [Google Scholar] [CrossRef] [PubMed]
- Imbs, A.B.; Latyshev, N.A.; Dautova, T.N.; Latypov, Y.Y. Distribution of lipids and fatty acids in corals by their taxonomic position and presence of zooxanthellae. Mar. Ecol. Prog. Ser. 2010, 409, 65–75. [Google Scholar] [CrossRef]
- Akyol, O.; Sağlam, C. Armina maculata Rafinesque, 1814 (Nudibranchia: Arminidae): A new record for the Turkish mollusc fauna. Turk. J. Zool. 2014, 38, 508–509. [Google Scholar] [CrossRef]
- Baptista, M.; Lopes, V.M.; Pimentel, M.S.; Bandarra, N.; Narciso, L.; Marques, A.; Rosa, R. Temporal fatty acid dynamics of the octocoral Veretillum cynomorium. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2012, 161, 178–1121. [Google Scholar] [CrossRef] [PubMed]
- Corsolini, S.; Borghesi, N. A comparative assessment of fatty acids in Antarctic organisms from the Ross Sea: Occurrence and distribution. Chemosphere 2017, 174, 747–753. [Google Scholar] [CrossRef] [PubMed]
- Zhukova, N.V. Lipid classes and fatty acid composition of the tropical nudibranch mollusks Chromodoris sp. and Phyllidia coelestis. Lipids 2007, 42, 1169–1175. [Google Scholar] [CrossRef] [PubMed]
- Affeld, S.; Wägele, H.; Avila, X.; Kehraus, S.; König, G.M. Distribution of homarine in some Opisthobranchia (Gastropoda: Mollusca). Bonner Zool. Beitr. 2007, 55, 181–190. [Google Scholar]
- Polychronopoulos, P.; Magiatis, P.; Skaltsounis, A.L.; Tillequin, F.; Vardala-Theodorou, E.; Tsarbopoulos, A. Homarine, a common metabolite in edible Mediterranean molluscs: Occurrence, spectral data and revision of a related structure. Nat. Prod. Lett. 2001, 15, 411–418. [Google Scholar] [CrossRef] [PubMed]
- Dai, J.; Shen, J.; Pan, W.; Shen, S.; Das, U.N. Effects of polyunsaturated fatty acids on the growth of gastric cancer cells in vitro. Lipids Health Dis. 2013, 12, 71. [Google Scholar] [CrossRef] [PubMed]
- Riediger, N.D.; Othman, R.A.; Suh, M.; Moghadasian, M.H. A systemic review of the roles of n-3 fatty acids in health and disease. J. Am. Diet. Assoc. 2009, 109, 668–679. [Google Scholar] [CrossRef] [PubMed]
- D’Eliseo, D.; Velotti, F. Omega-3 fatty acids and cancer cell cytotoxicity: Implications for multi-targeted cancer therapy. J. Clin. Med. 2016, 5, 15. [Google Scholar] [CrossRef] [PubMed]
- Gao, K.; Liang, Q.; Zhao, Z.-H.; Li, Y.-F.; Wang, S.-F. Synergistic anticancer properties of docosahexaenoic acid and 5-fluorouracil through interference with energy metabolism and cell cycle arrest in human gastric cancer cell line AGS cells. World J. Gastroenterol. 2016, 22, 2971–2980. [Google Scholar] [CrossRef] [PubMed]
- Silva, T.C.; de Andrade, P.B.; Paiva-Martins, F.; Valentão, P.; Pereira, D.M. In vitro anti-inflammatory and cytotoxic effects of aqueous extracts from the edible sea anemones Anemonia sulcata and Actinia equina. Int. J. Mol. Sci. 2017, 18, 653. [Google Scholar] [CrossRef] [PubMed]
- Yao, Q.; Fu, T.; Wang, L.U.; Lai, Y.; Wang, Y.; Xu, C.; Huang, L.; Guo, Y. Role of autophagy in the ω-3 long chain polyunsaturated fatty acid-induced death of lung cancer A549 cells. Oncol. Lett. 2015, 9, 2736–2742. [Google Scholar] [CrossRef] [PubMed]
- Zajdel, A.; Wilczok, A.; Tarkowski, M. Toxic effects of n-3 polyunsaturated fatty acids in human lung A549 cells. Toxicol. In Vitro 2015, 30, 486–491. [Google Scholar] [CrossRef] [PubMed]
- Trombetta, A.; Maggiora, M.; Martinasso, G.; Cotogni, P.; Canuto, R.A.; Muzio, G. Arachidonic and docosahexaenoic acids reduce the growth of A549 human lung-tumor cells increasing lipid peroxidation and PPARs. Chem. Biol. Interact. 2007, 165, 239–250. [Google Scholar] [CrossRef] [PubMed]
- Das, U. Essential fatty acids enhance free radical generation and lipid peroxidation to induce apoptosis of tumor cells. Clin. Lipidol. 2011, 6, 463–489. [Google Scholar] [CrossRef]
- Schmitz, G.; Ecker, J. The opposing effects of n-3 and n-6 fatty acids. Prog. Lipid Res. 2008, 47, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Pallarès, V.; Calay, D.; Cedó, L.; Castell-Auví, A.; Raes, M.; Pinent, M.; Ardévol, A.; Arola, L.; Blay, M. Additive, antagonistic, and synergistic effects of procyanidins and polyunsaturated fatty acids over inflammation in RAW 264.7 macrophages activated by lipopolysaccharide. Nutrition 2012, 28, 447–457. [Google Scholar] [CrossRef] [PubMed]
- Ren, J.; Chung, S.H. Anti-inflammatory effect of α-linolenic acid and its mode of action through the inhibition of nitric oxide production and inducible nitric oxide synthase gene expression via NF-ĸB and mitogen-activated protein kinase pathways. J. Agric. Food Chem. 2007, 55, 5073–5080. [Google Scholar] [CrossRef] [PubMed]
- Ambrozova, G.; Pekarova, M.; Lojek, A. Effect of polyunsaturated fatty acids on the reactive oxygen and nitrogen species production by raw 264.7 macrophages. Eur. J. Nutr. 2010, 49, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Ebada, S.S.; Edrada, R.A.; Lin, W.; Proksch, P. Methods for isolation, purification and structural elucidation of bioactive secondary metabolites from marine invertebrates. Nat. Protoc. 2008, 3, 1820–1831. [Google Scholar] [CrossRef] [PubMed]
- Metcalfe, L.D.; Schmitz, A.A. The rapid preparation of fatty acid esters for gas chromatographic analysis. Anal. Chem. 1961, 33, 363–364. [Google Scholar] [CrossRef]
- Pereira, R.B.; Pinto, D.C.G.A.; Pereira, D.M.; Gomes, N.G.M.; Silva, A.M.S.; Andrade, P.B.; Valentão, P. UHPLC-MS/MS profiling of Aplysia depilans and assessment of its potential therapeutic use: Interference on iNOS expression in LPS-stimulated RAW 264.7 macrophages and caspase-mediated pro-apoptotic effect on SH-SY5Y cells. J. Funct. Foods 2017, 37, 164–175. [Google Scholar] [CrossRef]
- Ferreres, F.; Duangsrisai, S.; Gomes, N.G.M.; Suksungworn, R.; Pereira, D.M.; Gil-Izquierdo, A.; Valentão, P.; Choowongkomon, K.; Andrade, P.B. Anti-inflammatory properties of the stem bark from the herbal drug Vitex peduncularis Wall. ex Schauer and characterization of its polyphenolic profile. Food Chem. Toxicol. 2017, 106, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Pereira, D.M.; Noites, A.; Valentão, P.; Ferreres, F.; Pereira, J.A.; Vale-Silva, L.; Pinto, E.; Andrade, P.B. Targeted metabolite analysis and biological activity of Pieris brassicae fed with Brassica rapa var. rapa. J. Agric. Food Chem. 2009, 57, 483–489. [Google Scholar] [CrossRef] [PubMed]
- Granato, D.; Santos, J.S.; Escher, G.B.; Ferreira, B.L.; Maggio, R.M. Use of principal component analysis (PCA) and hierarchical cluster analysis (HCA) for multivariate association between bioactive compounds and functional properties in foods: A critical perspective. Trends Food Sci. Technol. 2018, 72, 83–90. [Google Scholar] [CrossRef]
Sample Availability: Homarine and the standard solution of fatty acid methyl esters (Supelco® 37 Component Fame mix) are available from the authors. |

| Compound | A. maculata | A. tigrina | A. tricolorata | |||
|---|---|---|---|---|---|---|
| µg mg−1 dry extract | Relative % | µg mg−1 dry extract | Relative % | µg mg−1 dry extract | Relative % | |
| SFA | ||||||
| C12:0 | nq | nq | nq | nq | - | - |
| C13:0 | nq | nq | nq | nq | - | - |
| C14:0 | 1.20 (0.05) | 0.75 | 1.64 (0.04) | 1.20 | 1.18 (0.16) | 0.71 |
| C15:0 | 1.14 (0.04) | 0.87 | 1.14 (0.06) | 0.84 | 1.69 (0.07) | 1.02 |
| C16:0 | 30.43 (2.82) | 19.08 | 25.73 (1.10) | 18.89 | 21.28 (1.67) | 12.84 |
| C17:0 | 14.60 (0.38) | 9.16 | 13.38 (0.50) | 9.82 | 9.71 (0.61) | 5.86 |
| C18:0 | 18.42 (0.49) | 11.55 | 15.20 (0.69) | 11.16 | 15.99 (1.05) | 9.64 |
| C20:0 | 1.36 (0.02) | 0.85 | 3.70 (0.11) | 2.72 | nq | nq |
| C21:0 | - | - | nq | nq | - | - |
| C22:0 | - | - | 2.79 (0.14) | 2.05 | - | - |
| MUFA | ||||||
| C14:1n-5 | nq | nq | nq | nq | nq | nq |
| C15:1n-5 | 1.39 (0.14) | 0.87 | 1.48 (0.17) | 1.09 | - | - |
| C16:1n-7 | 1.72 (0.07) | 1.08 | 2.78 (0.33) | 2.04 | 3.46 (0.17) | 2.09 |
| C17:1n-7 | 1.13 (0.17) | 0.71 | 1.79 (0.11) | 1.32 | - | - |
| C18:1n-9c | 3.91 (0.20) | 2.45 | 3.91 (0.42) | 2.87 | 9.81 (0.64) | 5.92 |
| C18:1n-9t | 3.56 (0.28) | 2.23 | 1.71 (0.22) | 1.25 | 7.43 (0.46) | 4.48 |
| C22:1n-9 | 1.07 (0.14) | 0.67 | nq | nq | - | - |
| PUFA | ||||||
| C18:2n-6c | 4.27 (0.27) | 2.68 | 2.36 (0.15) | 1.74 | 3.16 (0.14) | 1.91 |
| C18:2n-6t | - | - | nq | nq | - | - |
| C20:2n-6 | 11.05 (1.23) | 6.93 | 6.68 (0.89) | 4.91 | 19.38 (0.81) | 11.69 |
| C18:3n-6c | nq | nq | nq | nq | 2.18 (0.09) | 1.31 |
| C18:3n-3c | 4.37 (0.44) | 2.74 | 1.99 (0.24) | 1.46 | 4.20 (0.25) | 2.53 |
| C20:3n-6 | 5.95 (0.72) | 3.73 | 4.30 (0.78) | 3.16 | 9.63 (0.33) | 5.81 |
| C20:4n-6 | 30.34 (2.18) | 19.03 | 25.54 (0.93) | 18.75 | 11.38 (0.72) | 6.87 |
| C20:5n-3 | 20.00 (1.22) | 12.54 | 18.42 (0.87) | 13.52 | 24.02 (1.59) | 14.49 |
| C22:6n-3 | 3.56 (0.09) | 2.23 | 1.67 (0.13) | 1.22 | 21.28 (1.53) | 12.84 |
| Total | 159.47 (8.58) | 100 | 136.21 (6.90) c | 100 | 165.81 (8.61) | 100 |
| Σ SFA | 67.15 (2.75) bbb | 42.11 | 63.57 (1.44) cc | 46.67 | 49.86 (2.58) | 30.07 |
| Σ MUFA | 12.78 (1.08) bbb | 8.01 | 11.67 (1.04) ccc | 8.57 | 20.71 (1.20) | 12.49 |
| Σ PUFA | 79.55 (6.11) a,b | 49.88 | 60.97 (4.41) cc | 44.76 | 95.24 (4.92) | 57.44 |
| Σ n-3 | 27.93 (1.83) bbb | 17.51 | 22.07 (0.98) cccc | 16.21 | 49.51 (2.85) | 29.86 |
| Σ n-5 | 1.39 (0.14) bb | 0.87 | 1.48 (0.17) cc | 1.09 | nq | - |
| Σ n-6 | 51.62 (4.28) a | 32.37 | 38.89 (3.44) | 28.55 | 45.74 (2.07) | 27.59 |
| Σ n-7 | 2.86 (0.20) aaa,b | 1.79 | 4.57 (0.23) cc | 3.36 | 3.46 (0.17) | 2.09 |
| Σ n-9 | 8.53 (0.59) a,bbb | 5.35 | 5.62 (0.65) cccc | 4.12 | 17.24 (1.07) | 10.40 |
| n-6/n-3 | 1.85 (0.03) bbbb | 1.76 (0.08) cccc | 0.92 (0.01) | |||
| Compound | A. maculata | A. tigrina | A. tricolorata |
|---|---|---|---|
| Homarine | 969.74 (43.90) aaaa,bbb | nq cccc | 1237.15 (35.08) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gomes, N.G.M.; Fernandes, F.; Madureira-Carvalho, Á.; Valentão, P.; Lobo-da-Cunha, A.; Calado, G.; Andrade, P.B. Profiling of Heterobranchia Sea Slugs from Portuguese Coastal Waters as Producers of Anti-Cancer and Anti-Inflammatory Agents. Molecules 2018, 23, 1027. https://doi.org/10.3390/molecules23051027
Gomes NGM, Fernandes F, Madureira-Carvalho Á, Valentão P, Lobo-da-Cunha A, Calado G, Andrade PB. Profiling of Heterobranchia Sea Slugs from Portuguese Coastal Waters as Producers of Anti-Cancer and Anti-Inflammatory Agents. Molecules. 2018; 23(5):1027. https://doi.org/10.3390/molecules23051027
Chicago/Turabian StyleGomes, Nelson G. M., Fátima Fernandes, Áurea Madureira-Carvalho, Patrícia Valentão, Alexandre Lobo-da-Cunha, Gonçalo Calado, and Paula B. Andrade. 2018. "Profiling of Heterobranchia Sea Slugs from Portuguese Coastal Waters as Producers of Anti-Cancer and Anti-Inflammatory Agents" Molecules 23, no. 5: 1027. https://doi.org/10.3390/molecules23051027
APA StyleGomes, N. G. M., Fernandes, F., Madureira-Carvalho, Á., Valentão, P., Lobo-da-Cunha, A., Calado, G., & Andrade, P. B. (2018). Profiling of Heterobranchia Sea Slugs from Portuguese Coastal Waters as Producers of Anti-Cancer and Anti-Inflammatory Agents. Molecules, 23(5), 1027. https://doi.org/10.3390/molecules23051027









