Multi-Response Extraction Optimization Based on Anti-Oxidative Activity and Quality Evaluation by Main Indicator Ingredients Coupled with Chemometric Analysis on Thymus quinquecostatus Celak
Abstract
:1. Introduction
2. Results and Discussions
2.1. Optimization of Extraction Conditions
24.24 X12 + 122.96 X22 + 35.14 X32
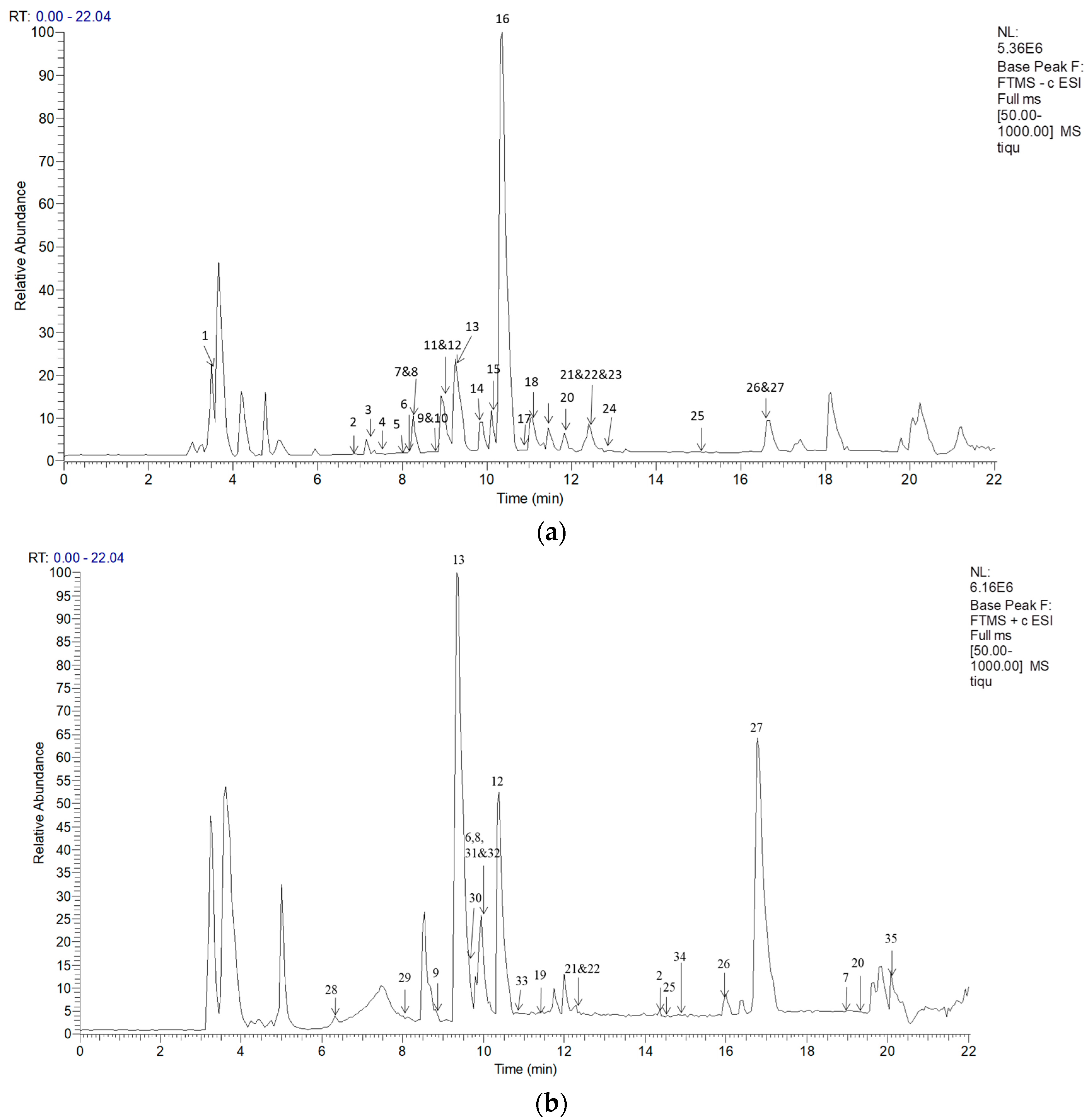
2.2. Identification of the Main Constituents of T. quinquecostatus Extract
2.2.1. Characterization of Phenolic Acids
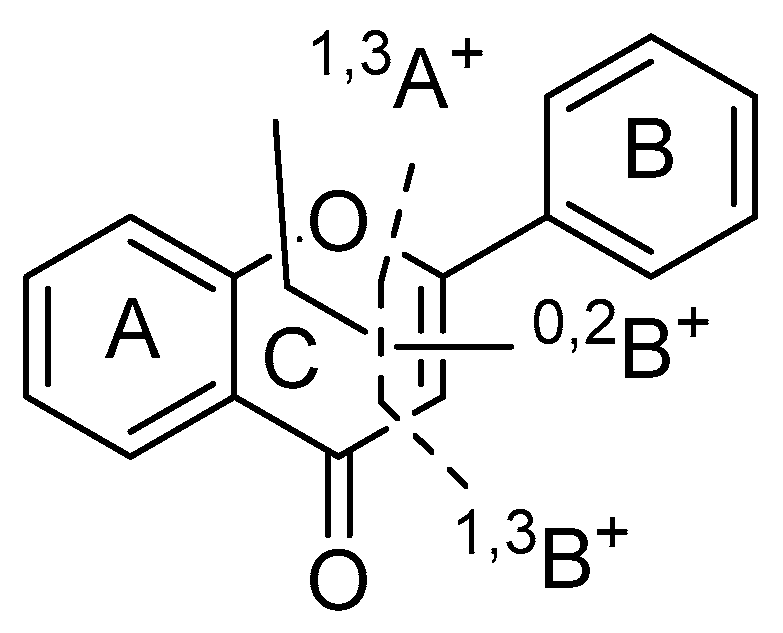
2.2.2. Characterization of Flavonoids
2.3. Optimization of HPLC Conditions
2.4. Validation of HPLC
2.4.1. Linearity
2.4.2. Precision, Repeatability, Stability and Accuracy
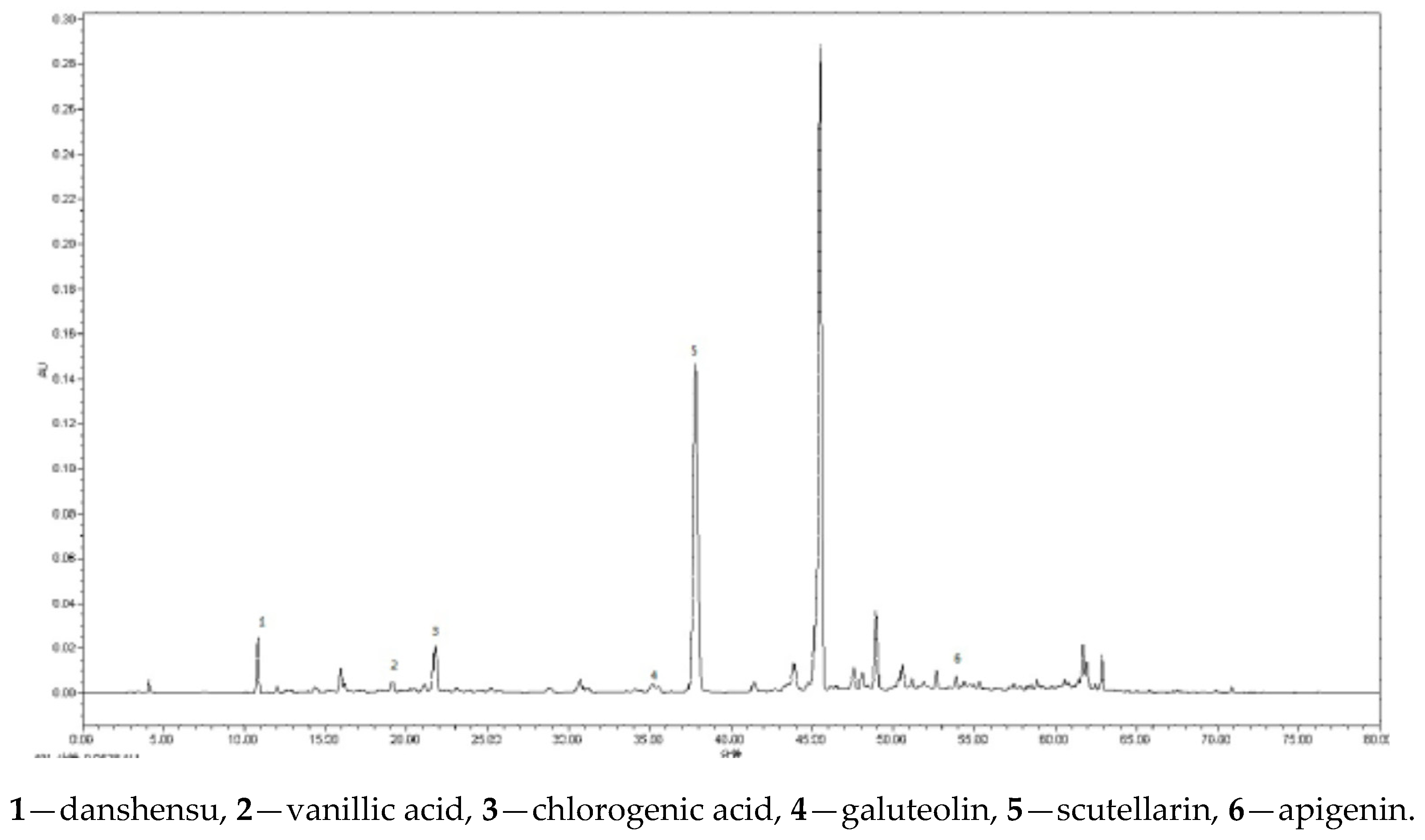
2.5. Quantitative Determination of 6 Compounds
2.6. Chemometric Analysis
2.6.1. PCA
2.6.2. HCA
3. Materials and Methods
3.1. Materials and Reagents
3.2. Optimization of Extraction Conditions
3.2.1. Design of the Extraction Method
3.2.2. DPPH Radical Scavenging Activity Assay
3.3. Preparation of Sample Solution
3.4. Preparation of Standard Solution
3.5. Instrument and UPLC-MS/MS Conditions
3.6. Instrument and HPLC Conditions
3.7. HPLC Method Validation
3.7.1. Linearity
3.7.2. Precision, Repeatability, and Stability
3.7.3. Accuracy
3.8. Identification and Determination of 6 Components
3.9. Data Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Jurca, T.; Vicas, L.; Marian, E.; Vicaş, S.; Muresan, M. A new natural antioxidant supplement-design and development. Farmacia 2016, 64, 135–142. [Google Scholar]
- Inagi, R. Oxidative stress in cardiovascular disease: A new avenue toward future therapeutic approaches. Recent. Pat. Cardiovasc. Drug. Discov. 2006, 1, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Slemmer, J.E.; Shacka, J.J.; Sweeney, M.I.; Weber, J.T. Antioxidants and free radical scavengers for the treatment of stroke, traumatic brain injury and aging. Curr. Med. Chem. 2008, 15, 404–414. [Google Scholar] [PubMed]
- Paul, S.; Das, S.; Tanvir, E.M.; Hossen, S.; Saha, M.; Afroz, R.; Islam, A.; Hossain, S.; Gan, S.H.; Khalil, I. Protective effects of ethanolic peel and pulp extracts of Citrus macroptera fruit against isoproterenol-induced myocardial infarction in rats. Biomed. Pharmacother. 2017, 94, 256–264. [Google Scholar] [CrossRef] [PubMed]
- Nieto, G.; Huvaere, K.; Skibsted, L.H. Antioxidant activity of rosemary and thyme by-products and synergism with added antioxidant in a liposome system. Eur. Food. Res. Technol. 2011, 233, 11–18. [Google Scholar] [CrossRef]
- Mohiseni, M.; Sepidnameh, M.; Bagheri, D.; Banaee, M.; Haghi, B.N. Comparative effects of Shirazi thyme and vitamin E on some growth and plasma biochemical changes in common carp (Cyprinuscarpio) during cadmium exposure. Aquac. Res. 2017. [Google Scholar] [CrossRef]
- Miliauskas, G.; Venskutonis, P.R.; van Beek, T.A. Screening of radical scavenging activity of some medicinal and aromatic plant extracts. Food Chem. 2004, 85, 231–237. [Google Scholar] [CrossRef]
- Ortiz-Viedma, J.; Romero, N.; Puente, L.; Burgos, K.; Toro, M.; Ramirez, L.; Rodriguez, A.; Barros-Velazquez, J.; Aubourg, S.P. Antioxidant and antimicrobial effects of stevia (Stevia rebaudiana Bert.) extracts during preservation of refrigerated salmon paste. Eur. J. Lipid. Sci. Tech. 2017. [Google Scholar] [CrossRef]
- Lin, J.N. Production and Application of Natural and Practical Spices; China Light Industry Press: Beijing, China, 1991; ISBN 7-5019-0957-1. [Google Scholar]
- Mancini, E.; Senatore, F.; Monte, D.D.; Martino, L.D.; Grulova, D.; Scognamiglio, M.; Snoussi, M.; Feo, V.D. Studies on chemical composition, antimicrobial and antioxidant activities of five Thymus vulgaris L. essential oils. Molecules 2015, 20, 12016–12028. [Google Scholar] [CrossRef] [PubMed]
- Tohidi, B.; Rahimmalek, M.; Arzani, A. Essential oil composition, total phenolic, flavonoid contents, and antioxidant activity of Thymus species collected from different regions of Iran. Food. Chem. 2017, 220, 153–161. [Google Scholar] [CrossRef] [PubMed]
- Khadir, A.; Sobeh, M.; Gad, H.A.; Benbelaid, F.; Bendahou, M.; Peixoto, H.; Sporer, F.; Ashour, M.L.; Wink, M. Chemical composition and biological activity of the essential oil from Thymus lanceolatus. Z. Naturforsch. C. 2016, 71, 155–163. [Google Scholar] [CrossRef] [PubMed]
- The Editorial Committee of the Flora of China; Chinese Academy of Sciences. The Flora of China; Science and Technology Press: Beijing, China, 1977; ISBN 9787030271662. [Google Scholar]
- National Herbal Medicine Editorial Group. National Herbal Medicine Collection, 3rd ed.; People’s Medical Publishing House: Beijing, China, 1988; ISBN 9787117180351. [Google Scholar]
- Nie, H.H.; Qiu, Y.F.; Xing, G.Q. The anti-inflammatory effect of volatile oil of thyme. J. Taishan Med. Coll. 1993, 14, 262–265. [Google Scholar]
- Zhao, Q.; Ren, C.; Han, Z.M. Drinking the Thymus quinquecostatus Celak and Fu tea for the treatment of cattle enteritis. Anim. Husb. Vet. Med. 2001, 30, 44–45. [Google Scholar]
- Tschiggerl, C.; Bucar, F. Influence of saponin plants on the volatile fraction of thyme in herbal teas. Fitoterapia 2011, 82, 903–910. [Google Scholar] [CrossRef] [PubMed]
- Li, J. Study on the Effects of Using Properties of Forsythia Suspensa and Thymus in Chinese-Style Sausage. Master’s Thesis, Shaanxi Normal University, Xi’an, China, 2006. [Google Scholar]
- Wang, Z.S. Comprehensive Research advances on Thymus mongolicus. Mod. Agric. Sci. Technol. 2016, 672, 63–66. [Google Scholar]
- Zhang, Y.X.; Shang, Z.J. Jia You Ben Cao Series of Copy; TCM Ancient Books Publishing House: Beijing, China, 2009; ISBN 9787801745903. [Google Scholar]
- Zhidan County Zhifang Commune Health Station. The preliminary observation of acute gastroenteritis and chronic stomachache treated by Thymus quinquecostatus Celak. Shaanxi Med. J. 1972, 5, 49–50. [Google Scholar]
- Hyun, T.K.; Kim, H.; Kim, J. Antioxidant and antidiabetic activity of Thymus quinquecostatus Celak. Ind. Crop Prod. 2014, 52, 611–616. [Google Scholar] [CrossRef]
- Sun, Z.X.; Sun, J.H.; Cheng, S.; Ma, Q.W.; Guo, S.L.; Zhang, J.B. The antitumor effects and the influence on immunity function in mice of the extraction of Thymus quinquecostatus Celak. J. Beijing Univ. Chin. Med. 2003, 1, 209–210. [Google Scholar]
- Sun, Z.X.; Zhang, H.Y.; Cheng, S.; Ma, Q.W.; Guo, S.L.; Zhang, J.B. Anti-tumor effect of ethanol extracts of Thymus quinquecostatus Celak on human leukemia cell line. J. Chin. Integr. Med. 2005, 3, 382–385. [Google Scholar] [CrossRef]
- Yan, C.; Chen, X.Y.; Sui, H.; Yu, X.T.; Wang, Y.; Bai, S.J.; Zhao, Y.C.; Shi, R.B.; She, G.M. Antioxidant activities and chemical compositions of Thymus quinquecostatus Celak. J. Beijing Univ. Trad. Chin. Med. 2016, 39, 383–389. [Google Scholar]
- Cheng, S.; Ma, Q.W.; Sun, Z.X.; Dai, G.Z. The extract of Thymuson scavenging activities to free radicals in vitro. Sci. Technol. Food. Ind. 2004, 25, 53–55. [Google Scholar]
- Liao, K.T.; Su, M.; Huang, S.S.; Chih, C.L.; Tsai, S.K. Honokiol Ameliorates Cerebral Infarction from Ischemia-Reperfusion Injury in Rats. Planta Med. 2003, 69, 130–134. [Google Scholar] [CrossRef] [PubMed]
- Akhtar, M.; Maikiyo, A.M.; Najmi, A.K.; Khanam, R.; Mujeeb, M.; Aqil, M. Neuroprotective effects of chloroform and petroleum ether extracts of Nigella sativa seeds in stroke model of rat. J. Pharm. Bioallied Allied Sci. 2013, 5, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Hu, L.M.; Wang, S.X.; Wang, Y.L.; Shi, F.; Li, H.; Liu, Y.; Kang, L.Y.; Gao, X.M. Neuroprotective Effects of Scutellarin against Hypoxic-Ischemic-Induced Cerebral Injury via Augmentation of Antioxidant Defense Capacity. Chin. J. Physiol. 2011, 54, 399–405. [Google Scholar] [PubMed]
- Mou, Q.Q.; He, J.X.; Yin, R.L.; Yang, B.; Fu, M.H.; Fang, J.; Li, H. Response surface optimized infrared-assisted extraction and UHPLC determination of flavonoid types from Flos Sophorae. Molecules 2017, 22, E1000. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Xue, A.; Niu, H.; Jia, Z.; Wang, J. Optimized ultrasonic-assisted extraction of flavonoids from Folium eucommiae and evaluation of antioxidant activity in multi-test systems in vitro. Food Chem. 2009, 114, 1147–1154. [Google Scholar] [CrossRef]
- Li, J.H.; Zeng, R.; Qu, Y.; Huang, L.F. Rapid identification on chemical constituents in roots of Paeonia delavayi var. lutea by UPLC-Q-TOF-MSE combined with UNIFI informatics platform. Chin. Tradit. Herbal. Drug. 2017, 8, 1529–1536. [Google Scholar]
- Zhang, Y.; Ma, H.L.; Mai, X.; Wang, H.L. Identification of components and metabolites of Mentha haplocalyx in rats plasma by UHPLC-Q-TOF-MS/MS. Chin. Tradit. Herbal. Drug. 2017, 19, 3927–3934. [Google Scholar]
- Chi, M.Y.; Xiang, W.Y.; Yang, W.; Sun, J.; Liao, S.G.; Li, Y.J.; Huang, Y. Analysis on Metabolites of Polygonum capitatum Extract in R at Urine by UPLC-Q-TOF/MS. Chin. J. Exp. Tradit. Med. Form. 2016, 17, 77–80. [Google Scholar]
- Wei, W.F.; Chen, H.C.; Liu, Y.; Ren, X.L.; Wang, W.M. Preliminary study on serum pharmacochemistry of leaves of acanthopanax senticosus based on UPLC-Q-TOF-MS. Chin. Tradit. Herbal. Drug. 2017, 7, 1306–1313. [Google Scholar]
- Zhou, Y.N.; Zhao, L.; Zheng, L.; Lv, L. Rapid identification of chemical constituents in Salvia chinensis Benth. by HPLC-TOF-MS. Chin. J. Chin. Mater. Med. 2013, 23, 4109–4112. [Google Scholar]
- Huang, H.; Ji, L.X.; Song, S.Y.; Wang, J.; Wei, N.; Jiang, M.; Bai, G.; Luo, G.A. Identification of the Major Constituents in Xuebijing Injection by HPLC-ESI-MS. Phytochem. Anal. 2011, 22, 330–338. [Google Scholar] [CrossRef] [PubMed]
- Kou, X.L.; Yang, D.H.; Su, Y.Y.; Liu, F. ESI-MS/MS Analysis of salvianolic acid A and salvianolic acid B from Salvia miltiorrhiza. Mod. Chin. Med. 2015, 10, 1026–1029. [Google Scholar]
- Wang, Y.; Li, X.B.; Li, J.J.; Xiong, H.Y.; Peng, C.S. Fragmentation pathway comparison of 5,6,7,4′-tetrahydroxy-flavone and 5,6,7,4′-tetramethoxy-flavone by high resolution electrospray ionization tandem mass spectroscopy. J. Chin. Mass. Spectrom. Soc. 2016, 5, 385–392. [Google Scholar]
- Li, K.; Song, J.; Zhao, Q.; Wang, B.; Zhang, Y.; Wang, X.; Tang, T.; Li, S. Effective component of Salvia miltiorrhiza in promoting cardiomyogenic differentiation of human placenta-derived mesenchymal stem cells. Int. J. Mol. Med. 2018, 2, 962–968. [Google Scholar]
- Hao, Y.Z. Protective Effect and Mechanism of Ligustrazine—Vanillic Acid on Experimental Cerebral Ischemia. Master’s Thesis, Beijing University of Chinese Medicine, Beijing, China, 2011. [Google Scholar]
- Liu, M.; Xia, X.H.; Zhang, Z.M.; Wu, Z.J.; Pan, S.Y. Comparative study on antioxidant activity of danshensu, protocatechuic aldehyde, caffeic acid and salvianolic acid B in vitro. J. Chin. Med. Mater. [CrossRef]
- Zhou, L. The research of cardiovascular pharmacological and clinical of breviscapine. Chin. J. Inf. Tradit. Chin. Med. 2013, 30, 134–136. [Google Scholar]
- Zhang, N.; Su, J.; Jin, L.; Zou, H.; Zhang, C. The research situation of cardiovascular function mechanism of tanshinol. J. Pharm. Pract. 2009, 27, 404–406. [Google Scholar]
- Bro, R.A.; Smilde, K. Principal component analysis. Anal. Methods 2014, 6, 2812–2831. [Google Scholar] [CrossRef]
- Da Silva Torres, E.A.F.; Garbelotti, M.L.; Moita Neto, J.M. The application of hierarchical clusters analysis to the study of the composition of foods. Food Chem. 2006, 99, 622–629. [Google Scholar] [CrossRef]
- Sharfifar, F.; Moshafi, M.H.; Mansouri, S.H.; Khodashenas, M.; Khoshnoodi, M. In vitro evaluation of antibacterial and antioxidant activities of the essential oil and methanol extract of endemic Zataria multiflora Boiss. Food Control. 2007, 18, 800–805. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |



| No. | X1/mL·g−1 | X2/% | X3/h | Y (IC50 (μg/mL)) |
|---|---|---|---|---|
| 1 | 1 | 0 | 1 | 218.55 |
| 2 | 0 | 0 | 0 | 205.04 |
| 3 | 0 | 0 | 0 | 205.09 |
| 4 | 0 | 0 | 0 | 208.40 |
| 5 | 0 | 1 | −1 | 338.87 |
| 6 | 0 | −1 | −1 | 382.21 |
| 7 | 0 | −1 | 1 | 257.91 |
| 8 | −1 | −1 | 0 | 208.50 |
| 9 | 0 | 0 | 0 | 202.47 |
| 10 | 1 | −1 | 0 | 312.52 |
| 11 | 0 | 0 | 0 | 204.07 |
| 12 | 1 | 1 | 0 | 321.01 |
| 13 | −1 | 0 | −1 | 197.13 |
| 14 | 0 | 1 | 1 | 473.46 |
| 15 | −1 | 1 | 0 | 372.92 |
| 16 | −1 | 0 | 1 | 216.58 |
| 17 | 1 | 0 | −1 | 231.38 |
| Source | Quadratic Sum | Degree of Freedom | Mean Sum of Square | F Value | p Value |
|---|---|---|---|---|---|
| model | 111,000 | 9 | 12,330.94 | 1655.45 | <0.0001 |
| X1 | 975.27 | 1 | 975.27 | 130.93 | <0.0001 |
| X2 | 14,888.48 | 1 | 14,888.48 | 1998.80 | <0.0001 |
| X3 | 35.74 | 1 | 35.74 | 4.80 | 0.0646 |
| X1X2 | 6078.54 | 1 | 6078.54 | 816.05 | <0.0001 |
| X1X3 | 260.50 | 1 | 260.50 | 34.97 | 0.0006 |
| X2X3 | 16,756.01 | 1 | 16,756.01 | 2249.52 | <0.0001 |
| X12 | 2473.91 | 1 | 2473.91 | 332.13 | <0.0001 |
| X22 | 63,662.73 | 1 | 63,662.73 | 8546.83 | <0.0001 |
| X32 | 5197.91 | 1 | 5197.91 | 697.83 | <0.0001 |
| residual | 52.14 | 7 | 9.94 | - | - |
| lack of fit | 33.31 | 3 | 11.10 | 2.36 | 0.2128 |
| pure error | 18.83 | 4 | 9.07 | - | - |
| Total | 111,000 | 16 | - | - | - |
| Peak NO. | Retention Time (min) | Formula Empirical | MW | Precurser Ions [M − H]− | Fragmentation | Tentative Structural Elucidation |
|---|---|---|---|---|---|---|
| 1 | 3.49 | C9H10O3 | 166.17 | 165.0395 | 165.0395, 147.0298, 129.0191 | Paeonol |
| 2 | 6.88 | C16H12O7 | 316.26 | 315.0700 | 297.0604, 246.9443, 153.0193, 135.0450 | Isorhamnetin |
| 3 | 7.15 | C9H10O5 | 198.17 | 197.0441 | 179.0347, 153.0552, 135.0447 | Danshensu |
| 4 | 7.33 | C8H8O4 | 168.15 | 167.0338 | 149.0239, 123.0449 | Vanillic acid |
| 5 | 7.87 | C7H5O4 | 154.12 | 153.0182 | 135.0447, 109.0293, 91.07836 | Gentisic acid or Protocatechuic acid |
| 6 | 8.00 | C16H18O9 | 354.31 | 353.0844 | 191.0561, 173.0456, 161.0241, 135.0450 | Chlorogenic acid |
| 7 | 8.26 | C20H20O8 | 388.37 | 387.1634 | 225.1128, 207.1027, 179.0557, 163.1128 | Desmethylnobiletin |
| 8 | 8.42 | C21H20O10 | 432.38 | 431.1884 | 385.1869, 341.1600, 311.0554, 279.0715, 223.1341 | Apigenin-7-O-glucoside |
| 9 | 8.61 | C22H21O10 | 478.40 | 477.0628 | 459.0562, 415.0665, 397.0566, 373.0568, 343.0461, 301.0355 | Isorhamnetin-3-O-glucoside |
| 10 | 8.67 | C21H20O11 | 448.38 | 447.0887 | 357.0616, 327.0513, 285.0406 | Galuteolin |
| 11 | 8.92 | C7H5O3 | 138.12 | 137.0235 | 93.0343 | Hydroxybenzoic acid |
| 12 | 9.02 | C9H8O4 | 180.16 | 179.0336 | 135.0450 | Caffeic acid |
| 13 | 9.26 | C21H18O12 | 462.36 | 461.0702 | 327.0508, 285.0405 | Scutellarin |
| 14 | 9.84 | C21H18O11 | 446.36 | 445.0749 | 269.0454, 240.9279, 175.0248 | Apigenin-7-O-glucuronide |
| 15 | 10.21 | C26H22O10 | 494.45 | 493.1100 | 449.1244, 383.0775, 359.0776, 313.0723, 295.0620 | Salvianolic acid A |
| 16 | 10.35 | C18H16O8 | 360.31 | 359.0750 | 341.0663, 315.0870, 197.0458, 179.0353, 161.0247, | Rosmarinic acid |
| 17 | 10.61 | C26H20O10 | 492.43 | 491.0946 | 311.0568, 295.0614, 267.0664, 223.0249, 179.0351 | Salvianolic acid C |
| 18 | 11.07 | C15H12O6 | 288.25 | 287.0540 | 269.0448, 259.0608, 243.0660, 151.0033 | Eridioctyol |
| 19 | 11.45 | C15H10O6 | 286.24 | 285.0386 | 257.0459, 241.0497, 133.0269 | Luteolin |
| 20 | 11.84 | C18H16O6 | 328.32 | 327.2159 | 309.2064, 291.1958, 165.0920 | Salvigenin |
| 21 | 12.30 | C17H14O7 | 330 | 329.0644 | 314.0432, 229.1438, 211.1333 | Thymusin |
| 22 | 12.40 | C11H12O4 | 208.21 | 207.0651 | 179.0346, 161.0240, 135.0449 | Ethyl caffeate |
| 23 | 12.57 | C15H10O5 | 270.24 | 269.0450 | 225.0551, 149.0241, 121.0293 | Apigenin |
| 24 | 12.71 | C16H12O6 | 300.26 | 299.0540 | 284.0326, 271.0421, 137.0270 | Sorbifolin |
| 25 | 14.97 | C17H14O6 | 314.289 | 313.0696 | 298.0483, 283.0241, 245.0448 | Ladanein or Cirsimaritin |
| 26 or 27 | 16.62 | C18H16O7 | 344.32 | 343.0803 | 328.0591, 313.0349 | Cirsilineol or Xanthomicrol |
| Peak NO. | Retention Time (min) | Molecular Formula | MW | Precurser Ions [M + H]+ | Fragmentation | Tentative Structural Elucidation |
|---|---|---|---|---|---|---|
| 28 | 6.31 | C15H12O5 | 272.25 | 272.9965 | 254.9872, 245.0031, 229.0079, 210.9974, 198.9973, 185.0181, 118.9713 | Naringenin |
| 29 | 8.11 | C27H30O15 | 594.52 | 595.1621 | 577.1669, 559.1462, 541.1353, 529.1353, 511.1248, 475.1426, 457.1143 | Luteolin-O-rutinoside |
| 9 | 8.81 | C22H22O12 | 478.406 | 479.0795 | 317.0836, 303.0502 | Isorhamnetin-O-glucoside |
| 13 | 9.33 | C21H18O12 | 462.36 | 463.0854 | 287.0551, 251.1245 | Scutellarin |
| 30 | 9.62 | C16H12O6 | 300.26 | 301.0693 | 286.0468, 273.0385, 242.0498, 227.0236, 167.7154 | Sorbifolin |
| 6 | 9.80 | C16H18O9 | 354.31 | 355.1710 | 337.1066, 203.0522, 193.1195 | Chlorogenic acid |
| 8 | 9.93 | C21H18O11 | 446.36 | 447.0904 | 429.0816, 271.0603 | Apigenin-7-O-glucuronide |
| 31 | 10.15 | C27H30O14 | 578.52 | 579.1078 | 517.1119, 471.1556, 399.0691, 381.0586, 337.0686, 319.0580 | Apigenin-7-O-rutinoside |
| 32 | 10.25 | C20H20O5 | 340.375 | 341.0640 | 323.0548, 297.0756, 279.0649, 187.0387 | Prenylnaringenin |
| 12 | 10.38 | C9H8O4 | 180.16 | 181.0492 | 163.0388 | Caffeic acid |
| 33 | 10.83 | C21H22O10 | 434.39 | 435.1234 | 391.1365, 373.1256, 349.1255, 271.0421, 227.0523 | Naringenin-7-O-glucoside |
| 19 | 11.44 | C15H10O6 | 286.24 | 287.0532 | 153.0182, 137.8446 | Luteolin or Scutellarein |
| 21 | 12.18 | C17H14O7 | 330 | 331.0792 | 316.0573, 298.0466, 255.0646, 213.0387 | Thymusin |
| 22 | 12.28 | C15H10O5 | 270.24 | 271.0584 | 135.0026 | Apigenin |
| 2 | 14.35 | C16H12O7 | 316.26 | 317.1001 | 275.0909, 197.0441, 147.0437, 125.0232, | Isorhamnetin |
| 25 | 14.41 | C17H14O6 | 314.289 | 315.0845 | 300.0626, 282.0520, 196.0326, 175.6378, 154.7404 | Ladanein or Cirsimaritin |
| 34 | 14.83 | C19H18O8 | 374.345 | 375.1054 | 360.0840, 345.0603,213.0390, 165.0342 | Methyl rosmarinate |
| 26 | 15.99 | C18H16O7 | 344.319 | 345.0952 | 330.0732, 312.0626, 297.0390, 227.0547, 212.8993 | Xanthomicrol |
| 27 | 16.76 | C18H16O7 | 344.319 | 345.0953 | 330.0733, 315.0498, 149.0233, 135.0439 | Cirsilineol |
| 7 | 18.98 | C20H20O8 | 388.372 | 389.1206 | 374.0994, 359.0758, 341.0652, 328.0936, 227.0544 | Desmethylnobiletin |
| 20 | 19.35 | C18H16O6 | 328.32 | 329.1000 | 314.0782, 301.1792, 287.1273, 269.1168, 163.0750, 135.0803 | Salvigenin |
| 35 | 20.08 | C19H18O7 | 358.346 | 359.1108 | 344.0889, 329.0654, 311.0547, 227.0548, 182.2784 | Gardenin B |
| Analyte | Regression Equation | R2 | Linear Range (μg/mL) | Precision (RSD, %, n = 6) | Repeatability (RSD, %, n = 6) | Stability (RSD, %, n = 6) | Recovery (%, n = 9) | ||
|---|---|---|---|---|---|---|---|---|---|
| Intra-Day (n = 6) | Inter-Day (n = 3) | Mean | RSD | ||||||
| danshensu (1) | y = 1126x + 724.5 | 0.9990 | 4.68–150.0 | 1.82 | 1.29 | 1.49 | 1.28 | 99.98 | 0.75 |
| vanillic acid (2) | y = 29710x − 1791.2 | 0.9998 | 0.40–12.9 | 1.28 | 1.96 | 1.84 | 1.95 | 99.72 | 1.49 |
| chlorogenic acid (3) | y = 21533x − 76485 | 0.9995 | 6.25–100.0 | 1.67 | 1.11 | 1.84 | 1.21 | 98.34 | 0.91 |
| galuteolin (4) | y = 13898x − 5771 | 0.9990 | 1.25–20.0 | 1.53 | 0.49 | 1.92 | 1.85 | 100.84 | 1.24 |
| scutellarin (5) | y = 17290x − 63063 | 0.9999 | 6.25–200.0 | 1.61 | 0.21 | 1.97 | 1.25 | 98.94 | 0.67 |
| apigenin (6) | y = 30618x − 5312 | 0.9999 | 0.625–20.0 | 1.88 | 1.70 | 1.56 | 1.60 | 101.67 | 1.30 |
| Sample | Content of Each Compound (mg/g) | |||||||
|---|---|---|---|---|---|---|---|---|
| No. | Habitat | Harvest Time | Danshensu | Vanillic Acid | Chlorogenic Acid | Galuteolin | Scutellarin | Apigenin |
| S1 | qingyang | 7.1 | 11.800 | 0.072 | 0.450 | 0.152 | 3.650 | 0.034 |
| S2 | 7.15 | 5.750 | 0.027 | 0.450 | 0.095 | 1.650 | 0.019 | |
| S3 | 8.1 | 4.380 | 0.018 | 0.850 | 0.062 | 3.310 | 0.013 | |
| S4 | 8.15 | 5.600 | 0.041 | 0.700 | 0.050 | 2.520 | 0.029 | |
| S5 | 9.1 | 5.550 | 0.036 | 1.050 | 0.052 | 2.050 | 0.037 | |
| S6 | jingbian | 8.1 | 2.650 | 0.015 | 0.330 | 0.073 | 0.630 | 0.036 |
| S7 | 8.15 | 0.840 | 0.016 | 1.210 | 0.065 | 0.490 | 0.019 | |
| S8 | 9.1 | 1.230 | 0.007 | 0.550 | 0.019 | 0.580 | 0.017 | |
| S9 | 9.15 | 1.670 | 0.007 | 1.300 | 0.064 | 1.270 | 0.016 | |
| S10 | yanchi | 6 | 7.035 | 0.075 | 1.181 | - | 4.472 | - |
| S11 | 7 | 9.021 | 0.056 | 1.034 | - | 3.612 | - | |
| S12 | 8 | 9.386 | 0.066 | 1.179 | - | 4.057 | - | |
| Factors | Level | ||
|---|---|---|---|
| −1 | 0 | 1 | |
| X1/mL·g−1 | 15 | 22.5 | 30 |
| X2/% | 0 | 50 | 100 |
| X3/h | 1 | 1.5 | 2 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chang, Y.-L.; Shen, M.; Ren, X.-Y.; He, T.; Wang, L.; Fan, S.-S.; Wang, X.-H.; Li, X.; Wang, X.-P.; Chen, X.-Y.; et al. Multi-Response Extraction Optimization Based on Anti-Oxidative Activity and Quality Evaluation by Main Indicator Ingredients Coupled with Chemometric Analysis on Thymus quinquecostatus Celak. Molecules 2018, 23, 957. https://doi.org/10.3390/molecules23040957
Chang Y-L, Shen M, Ren X-Y, He T, Wang L, Fan S-S, Wang X-H, Li X, Wang X-P, Chen X-Y, et al. Multi-Response Extraction Optimization Based on Anti-Oxidative Activity and Quality Evaluation by Main Indicator Ingredients Coupled with Chemometric Analysis on Thymus quinquecostatus Celak. Molecules. 2018; 23(4):957. https://doi.org/10.3390/molecules23040957
Chicago/Turabian StyleChang, Yan-Li, Meng Shen, Xue-Yang Ren, Ting He, Le Wang, Shu-Sheng Fan, Xiu-Huan Wang, Xiao Li, Xiao-Ping Wang, Xiao-Yi Chen, and et al. 2018. "Multi-Response Extraction Optimization Based on Anti-Oxidative Activity and Quality Evaluation by Main Indicator Ingredients Coupled with Chemometric Analysis on Thymus quinquecostatus Celak" Molecules 23, no. 4: 957. https://doi.org/10.3390/molecules23040957
APA StyleChang, Y.-L., Shen, M., Ren, X.-Y., He, T., Wang, L., Fan, S.-S., Wang, X.-H., Li, X., Wang, X.-P., Chen, X.-Y., Sui, H., & She, G.-M. (2018). Multi-Response Extraction Optimization Based on Anti-Oxidative Activity and Quality Evaluation by Main Indicator Ingredients Coupled with Chemometric Analysis on Thymus quinquecostatus Celak. Molecules, 23(4), 957. https://doi.org/10.3390/molecules23040957





