A Novel Synthesis of Highly Functionalized Pyridines by a One-Pot, Three-Component Tandem Reaction of Aldehydes, Malononitrile and N-Alkyl-2-cyanoacetamides under Microwave Irradiation
Abstract
1. Introduction
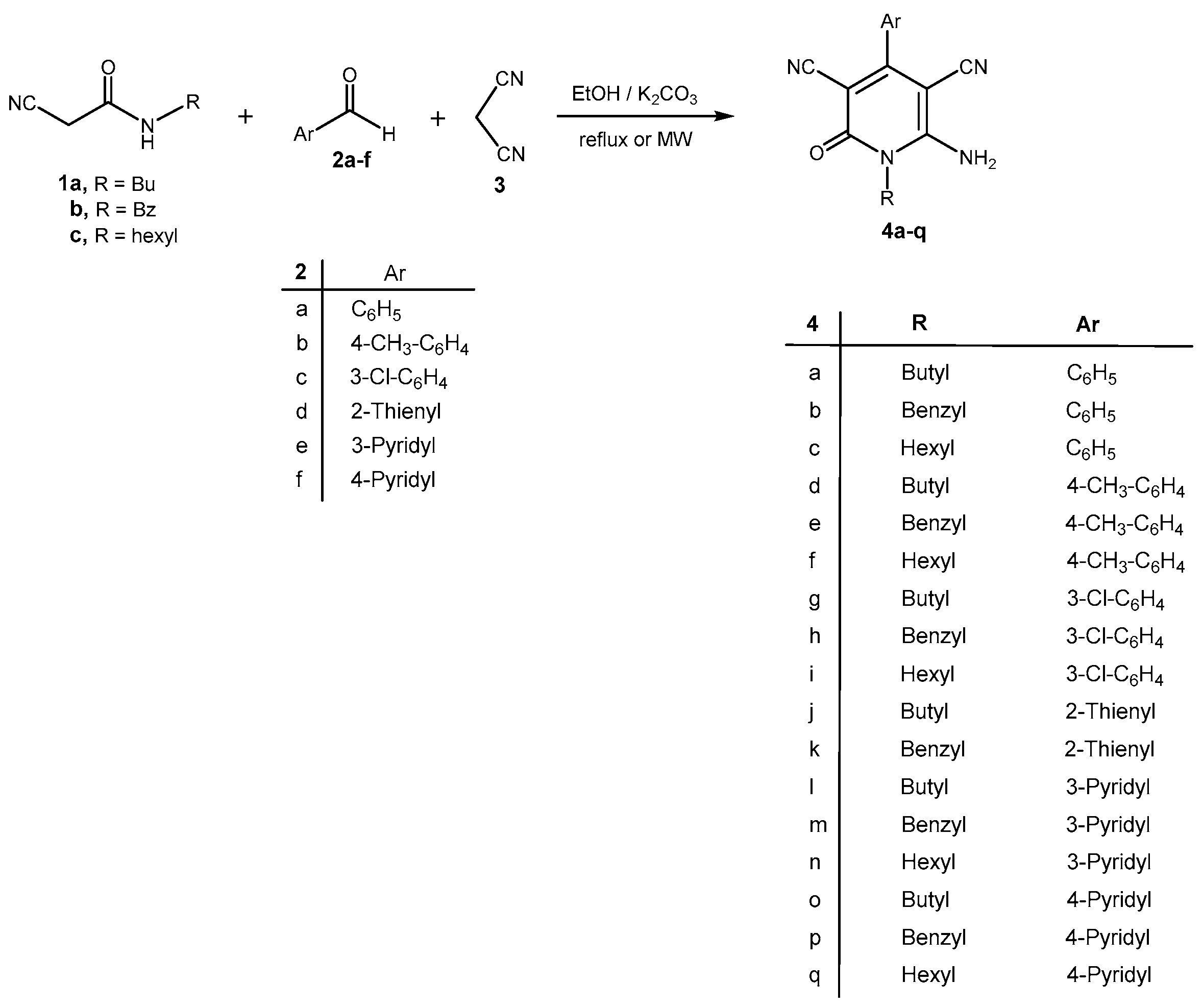
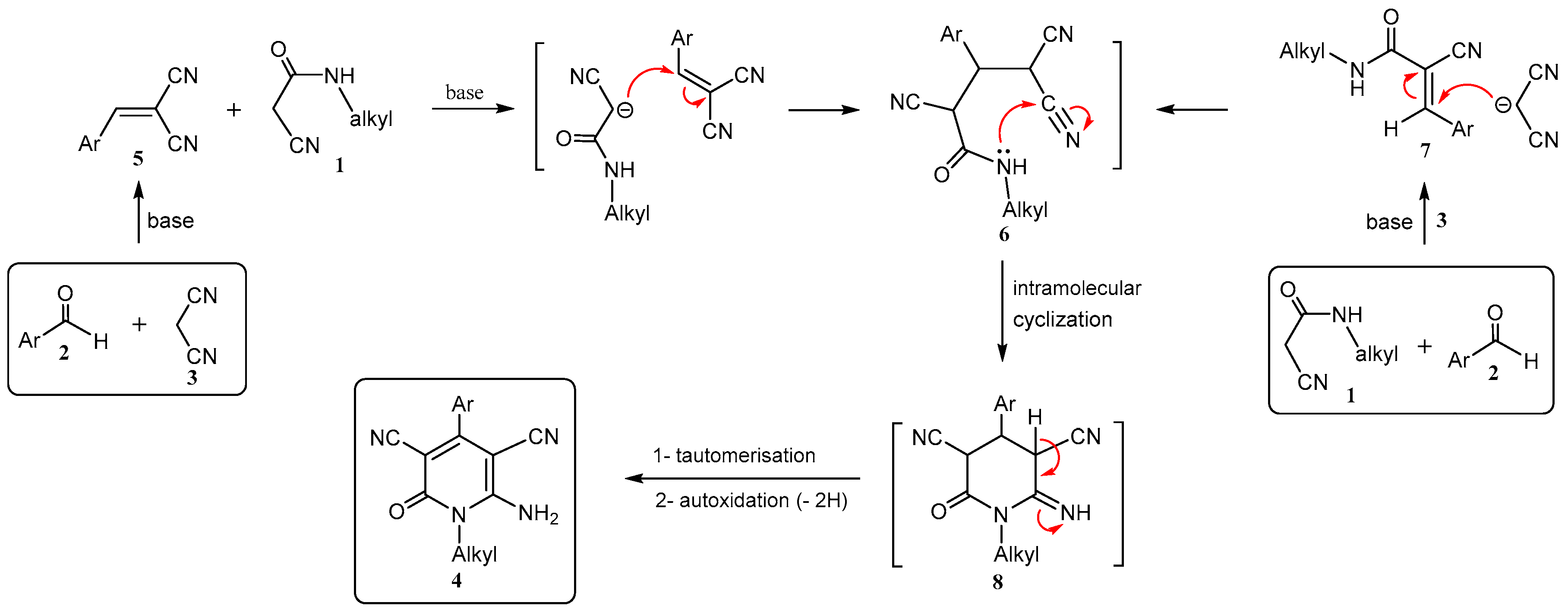
2. Results and Discussion
3. Experimental
3.1. General Information
3.2. General Procedure for the Synthesis of 1-Alkyl-6-amino-4-aryl(or het)-2-oxo-1,2-dihydro-pyridine-3,5-dicarbonitriles 4a–q
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Bayat, M.; Nasri, S.; Notash, B. Synthesis of new 3-cyanoacetamide pyrrole and 3-acetonitrile pyrrole derivatives. Tetrahedron 2017, 73, 1522–1527. [Google Scholar] [CrossRef]
- Xin, X.; Wang, Y.; Kumar, S.; Liu, X.; Lin, Y.; Dong, D. Efficient one-pot synthesis of substituted pyridines through multicomponent reaction. Org. Biomol. Chem. 2010, 8, 3078–3082. [Google Scholar] [CrossRef] [PubMed]
- Pathak, S.; Kundu, A.; Pramanik, A. Regioselective synthesis of two types of highly substituted 2-pyridones through similar multicomponent reactions. Tetrahedron Lett. 2012, 53, 3030–3034. [Google Scholar] [CrossRef]
- Dӧmling, A. Recent developments in isocyanide based multicomponent reactions in applied chemistry. Chem. Rev. 2006, 106, 17–89. [Google Scholar] [CrossRef] [PubMed]
- Esmaeili, A.A.; Amini-Ghalandarabad, S.; Mesbah, F.; Tasmimi, M.; Izadyar, M.; Fakhari, A.R.; Salimi, A.R. Efficient synthesis of novel spiro[indole-3,6′-pyrano[2,3-d][1,3]-thiazolo[3,2-a]pyrimidine derivatives through an organobase-catalyzed, three-component reaction. Tetrahedron 2015, 71, 2458–2462. [Google Scholar] [CrossRef]
- Hadjebi, M.; Hashtroudi, M.S.; Bijanzadeh, H.R.; Balalaie, S. Novel four-component approach for the synthesis of polyfunctionalized 1,4-dihydropyridines in aqueous media. Helv. Chim. Acta 2011, 94, 382–388. [Google Scholar] [CrossRef]
- Azzam, S.H.S.; Pasha, M.A. Microwave-assisted, mild, facile, and rapid one-pot three-component synthesis of some novel pyrano[2,3-d]pyrimidine-2,4,7-triones. Tetrahedron Lett. 2012, 53, 7056–7059. [Google Scholar] [CrossRef][Green Version]
- Kappe, C.O.; Dallinger, D. Controlled microwave heating in modern organic synthesis: Highlights from the 2004–2008 literature. Mol. Divers. 2009, 13, 71–193. [Google Scholar] [CrossRef] [PubMed]
- Jiang, B.; Shi, F.; Tu, S.J. Microwave-assisted multicomponent reactions in the heterocyclic chemistry. Curr. Org. Chem. 2010, 14, 357–378. [Google Scholar] [CrossRef]
- Kappe, C.O. Controlled microwave heating in modern organic synthesis. Angew. Chem. Int. Ed. 2004, 43, 6250–6284. [Google Scholar] [CrossRef] [PubMed]
- Torres, M.; Gil, S.; Parra, M. New synthetic methods to 2-pyridone rings. Curr. Org. Chem. 2005, 9, 1757–1779. [Google Scholar] [CrossRef]
- Henry, G.D. De novo synthesis of substituted pyridines. Tetrahedron 2004, 60, 6043–6061. [Google Scholar] [CrossRef]
- Khaksar, S.; Yaghoobi, M. A concise and versatile synthesis of 2-amino-3-cyanopyridine derivatives in 2,2,2-trifluoroethanol. J. Fluorine Chem. 2012, 142, 41–44. [Google Scholar] [CrossRef]
- Movassaghi, M.; Hill, M.D.; Ahmad, O.K. Direct synthesis of pyridine derivatives. J. Am. Chem. Soc. 2007, 129, 10096–10097. [Google Scholar] [CrossRef] [PubMed]
- Teague, S.J. Synthesis of heavily substituted 2-aminopyridines by displacement of a 6-methylsulfinyl group. J. Org. Chem. 2008, 73, 9765–9766. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Zhang, Q.; Yuan, H.; Liu, Q. Temperature-controlled synthesis of substituted pyridine derivatives via the [5C + 1N] annulation of 1,1-bisalkylthio-1,4-pentanedienes and ammonium acetate. J. Org. Chem. 2008, 73, 2442–2445. [Google Scholar] [CrossRef] [PubMed]
- Mobinikhaledi, A.; Asadbegi, S.; Bodaghifard, M.A. Convenient, multicomponent, one-pot synthesis of highly substituted pyridines under solvent-free conditions. Synth. Commun. 2016, 19, 1605–1611. [Google Scholar] [CrossRef]
- Reddy, T.R.; Mutter, R.; Heal, W.; Guo, K.; Gillet, V.J.; Pratt, S.; Chen, B. Library design, synthesis, and screening: Pyridine dicarbonitriles as potential prion disease therapeutics. J. Med. Chem. 2006, 49, 607–615. [Google Scholar] [CrossRef] [PubMed]
- Cocco, M.T.; Congiu, C.; Lilliu, V.; Onnis, V. Synthesis and antiproliferative activity of 2,6-dibenzylamino-3,5-dicyanopyridines on human cancer cell lines. Eur. J. Med. Chem. 2005, 40, 1365–1372. [Google Scholar] [CrossRef] [PubMed]
- May, B.C.H.; Zorn, J.A.; Witkop, J.; Sherrill, J.; Wallace, A.C.; Legname, G.; Prusiner, S.B.; Cohen, F.E. Structure-activity relationship study of prion inhibition by 2-amino-pyridine-3,5-dicarbonitrile-based compounds: Parallel synthesis, bioactivity, and in vitro Pharmacokinetics. J. Med. Chem. 2007, 50, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Beukers, M.W.; Chang, L.C.W.; Von Frijtag Drabbe Künzel, J.K.; Mulder-Krieger, T.; Spanjersberg, R.F.; Brussee, J.; Ijzerman, A.P. New, non-adenosine, high-potency agonists for the human adenosine A2B receptor with an improved selectivity profile compared to the reference agonist N-ethylcarboxamidoadenosine. J. Med. Chem. 2004, 47, 3707–3709. [Google Scholar] [CrossRef] [PubMed]
- Chang, L.C.W.; Von Frijtag Drabbe Künzel, J.K.; Mulder-Krieger, T.; Spanjersberg, R.F.; Roerink, S.F.; van den Hout, G.; Beukers, M.W.; Brussee, J.; Ijzerman, A.P. A Series of ligands displaying a remarkable agonistic-antagonistic profile at the adenosine A1 receptor. J. Med. Chem. 2005, 48, 2045–2053. [Google Scholar] [CrossRef] [PubMed]
- Samadi, A.; Chioua, M.; Alvarez Perez, M.; Soriano Santamaria, E.; Valderas Cortina, C.; Marco Contelles, J.L.; De Los Rios, S.C.; Romero Martinez, A.; Villarroya Sanchez, M.; Garcia Lopez, M.; et al. 2-Amino-3,5-dicyanopyridine and 2-Chloro-3,5-dicyanopyridine Derivatives as Cholinesterase Enzymes Inhibitors and with Neuroprotective Capacity. Spanish Patent ES 2,365,233 A1, 27 September 2011. [Google Scholar]
- Chen, H.; Zhang, W.; Tam, R.; Raney, A.K. Novel Heterocyclic Compounds as ikk2 Inhibitors with Anti-Hbv Activity. Patent No. WO 2005,058,315 A1, 30 June 2005. [Google Scholar]
- Levy, S.B.; Alekshun, M.N.; Podlogar, B.L.; Ohemeng, K.; Verma, A.K.; Warchol, T.; Bhatia, B.; Bowser, T.; Grier, M. Transcription Factor Modulating Compounds and Methods of Use Thereof. US Patent 20,050,124,678 A1, 4 May 2005. [Google Scholar]
- Harada, H.; Watanuki, S.; Takuwa, T.; Kawaguchi, K.; Okazaki, T.; Hirano, Y.; Saitoh, C. Medicin Comprising Dicyanopyridine Derivative. PCT Int. Appl. WO 2,002,006,237 A1, 24 January 2002. [Google Scholar]
- Perrier, V.; Wallace, A.C.; Kaneko, K.; Safar, J.; Prusiner, S.B.; Cohen, F.E. Mimicking dominant negative inhibition of prion replication through structure-based drug design. Proc. Nat. Acad. Sci. USA 2000, 97, 6073–6078. [Google Scholar] [CrossRef] [PubMed]
- Guo, K.; Mutter, R.; Heal, W.; Reddy, T.R.K.; Cope, H.; Pratt, S.; Thompson, M.J.; Chen, B. Synthesis and evaluation of a focused library of pyridine dicarbonitriles against prion disease. Eur. J. Med. Chem. 2008, 43, 93–106. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Xue, S.; Yang, Y.; Feng, J.; Liu, P.; Zhang, Y.; Zhu, J.; Xu, Z.; Hall, A.; Zhao, B.; et al. Regioselectivity and mechanism of synthesizing N-substituted 2-pyridones and 2-substituted pyridines via metal-free C-O and C-N bond-cleaving of oxazoline[3,2-a]pyridiniums. Sci. Rep. 2017, 7, 41287. [Google Scholar] [CrossRef] [PubMed]
- Sorbera, L.A.; Castaner, J. Laquinimod. Treatment of multiple sclerosis, Immuno-modulator. Drugs Future 2003, 28, 1059–1063. [Google Scholar] [CrossRef]
- Ferraz, A.C.; Angelucci, M.E.M.; Da Costa, M.L.; Batista, I.R.; De Olivera, B.H.; Da Cunha, C. Pharmacological evaluation of ricinine, a central nervous system stimulant isolated from Ricinus communis. Pharmacol. Biochem. Behav. 1999, 63, 367–375. [Google Scholar] [CrossRef]
- Tkachova, V.P.; Gorobets, N.Y.; Tkachov, R.P.; Musatov, V.I.; Dyachenko, V.D. Competing transformations of 2-cyanoacetanilides in the reactions with derivatives of ethoxymethylenemalonic acid. Arkivoc 2012, 6, 398–411. [Google Scholar]
- Ando, M.; Wada, T.; Sato, N. Facile one-pot synthesis of N-difluoromethyl-2-pyridone derivatives. Org. Lett. 2006, 8, 3805–3808. [Google Scholar] [CrossRef] [PubMed]
- Conreaux, D.; Bossharth, E.; Monteiro, N.; Desbordes, P.; Balme, G. A practical procedure for the selective N-alkylation of 4-alkoxy-2-pyridones and its use in a sulfone-mediated synthesis of N-methyl-4-methoxy-2-pyridone. Tetrahedron Lett. 2005, 46, 7917–7920. [Google Scholar] [CrossRef]
- Zhang, X.; Yang, Z.-P.; Huang, L.; You, S.-L. Highly regio- and enantioselective synthesis of N-substituted 2-pyridones: Iridium-catalyzed intermolecular asymmetric allylic amination. Angew. Chem. Int. Ed. 2015, 54, 1873–1876. [Google Scholar] [CrossRef] [PubMed]
- Mekheimer, R.A.; Abdel Hameed, A.M.; Sadek, K.U. Solar thermochemical reactions: Four-component synthesis of polyhydroquinoline derivatives induced by solar thermal energy. Green Chem. 2008, 10, 592–593. [Google Scholar] [CrossRef]
- Mekheimer, R.A.; Ameen, M.A.; Sadek, K.U. Solar thermochemical reactions II: Synthesis of 2-aminothiophenes via Gewald reaction induced by solar thermal energy. Chin. Chem. Lett. 2008, 19, 788–790. [Google Scholar] [CrossRef]
- Mekheimer, R.A.; Sadek, K.U. Microwave-assisted reactions: Three component process for the synthesis of 2-amino-2-chromenes under microwave heating. J. Heterocycl. Chem. 2009, 46, 149–151. [Google Scholar] [CrossRef]
- Al-Zaydi, K.M.; Borik, R.M.; Mekheimer, R.A.; Elnagdi, M.H. Green chemistry: A facile synthesis of polyfunctionally substituted thieno[3,4-c]pyridinones and thieno[3,4-d]pyridazinones under neat reaction conditions. Ultrason. Sonochem. 2010, 17, 909–915. [Google Scholar] [CrossRef] [PubMed]
- Mekheimer, R.A.; Abdel Hameed, A.M.; Mohamed, S.M.; Sadek, K.U. Green, three component highly efficient synthesis of 2-amino-5,6,7,8-tetrahydro-4H-chromen-3-carbonitriles in water at ambient temperature. Green Chem. Lett. Rev. 2010, 3, 161–163. [Google Scholar] [CrossRef]
- Mekheimer, R.A.; Hilmy, N.M.; Abdel Hameed, A.; Dacrory, S.; Sadek, K.U. Simple, three component, highly efficient green synthesis of thiazolo[3,2-a]pyridine derivatives under neat conditions. Synth. Commun. 2011, 41, 2511–2516. [Google Scholar] [CrossRef]
- Mekheimer, R.A.; Al-Zaydi, K.M.; Al-Shamary, A.; Sadek, K.U. Solar thermochemical reactions IV: Unusual reaction of nitrones with acetonitrile derivatives induced by solar thermal energy. Green Sustain. Chem. 2011, 1, 176–181. [Google Scholar] [CrossRef][Green Version]
- Sadek, K.U.; Mekheimer, R.A.; Mohamed, T.M.; Moustafa, M.S.; Elnagdi, M.H. Regioselectivity in the multicomponent reaction of 5-aminopyrazoles, cyclic 1,3-diketones and dimethylformamide dimethylacetal under controlled microwave heating. Beilstein J. Org. Chem. 2012, 8, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Sadek, K.U.; Mekheimer, R.A.; Abdel Hameed, A.M.; Elnahas, F.; Elnagdi, M.H. Green and highly efficient synthesis of 2-arylbenzothiazoles using glycerol without catalyst at ambient temperature. Molecules 2012, 17, 6011–6019. [Google Scholar] [CrossRef] [PubMed]
- Sadek, K.U.; Abdel Hameed, A.M.; Mekheimer, R.A.; abd Elmonem, M.; Elnagdi, M.H. Zn (L-proline)2: An efficient and recyclable catalytic system for the asymmetric multicomponent synthesis of 2-amino-4H-chromenes in water under controlled microwave heating. Curr. Microw. Chem. 2016, 3, 227–232. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds 4a–q are available from the authors. |
| No. | Heat Time | µω Time | ||
|---|---|---|---|---|
| (min) | Yield (%) | (min) | Yield (%) | |
| 4a | 60 | 70 | 10 | 91 |
| 4b | 90 | 77 | 10 | 94 |
| 4c | 90 | 71 | 10 | 87 |
| 4d | 90 | 73 | 10 | 91 |
| 4e | 90 | 76 | 10 | 92 |
| 4f | 90 | 70 | 10 | 88 |
| 4g | 90 | 65 | 10 | 81 |
| 4h | 180 | 69 | 12 | 90 |
| 4i | 90 | 67 | 10 | 83 |
| 4j | 180 | 71 | 12 | 87 |
| 4k | 120 | 73 | 11 | 92 |
| 4l | 180 | 69 | 15 | 85 |
| 4m | 180 | 72 | 13 | 93 |
| 4n | 240 | 70 | 15 | 83 |
| 4o | 120 | 72 | 11 | 88 |
| 4p | 120 | 75 | 11 | 92 |
| 4q | 120 | 71 | 11 | 85 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mekheimer, R.A.; Al-Sheikh, M.A.; Medrasi, H.Y.; Alsofyani, N.H.H. A Novel Synthesis of Highly Functionalized Pyridines by a One-Pot, Three-Component Tandem Reaction of Aldehydes, Malononitrile and N-Alkyl-2-cyanoacetamides under Microwave Irradiation. Molecules 2018, 23, 619. https://doi.org/10.3390/molecules23030619
Mekheimer RA, Al-Sheikh MA, Medrasi HY, Alsofyani NHH. A Novel Synthesis of Highly Functionalized Pyridines by a One-Pot, Three-Component Tandem Reaction of Aldehydes, Malononitrile and N-Alkyl-2-cyanoacetamides under Microwave Irradiation. Molecules. 2018; 23(3):619. https://doi.org/10.3390/molecules23030619
Chicago/Turabian StyleMekheimer, Ramadan Ahmed, Mariam Abdullah Al-Sheikh, Hanadi Yousef Medrasi, and Najla Hosain Hassan Alsofyani. 2018. "A Novel Synthesis of Highly Functionalized Pyridines by a One-Pot, Three-Component Tandem Reaction of Aldehydes, Malononitrile and N-Alkyl-2-cyanoacetamides under Microwave Irradiation" Molecules 23, no. 3: 619. https://doi.org/10.3390/molecules23030619
APA StyleMekheimer, R. A., Al-Sheikh, M. A., Medrasi, H. Y., & Alsofyani, N. H. H. (2018). A Novel Synthesis of Highly Functionalized Pyridines by a One-Pot, Three-Component Tandem Reaction of Aldehydes, Malononitrile and N-Alkyl-2-cyanoacetamides under Microwave Irradiation. Molecules, 23(3), 619. https://doi.org/10.3390/molecules23030619




