Study of the Anti-Staphylococcal Potential of Honeys Produced in Northern Poland
Abstract
:1. Introduction
2. Results
2.1. Investigation of Antimicrobial Potential of Honey Samples—Determination of MIC (Minimum Inhibitory Concentration) and MBC (Minimum Bactericidal Concentration) Parameters
2.2. Time-Kill Assay, Determination of Kinetic of Bactericidal Effect of Honey Against Staphylococci
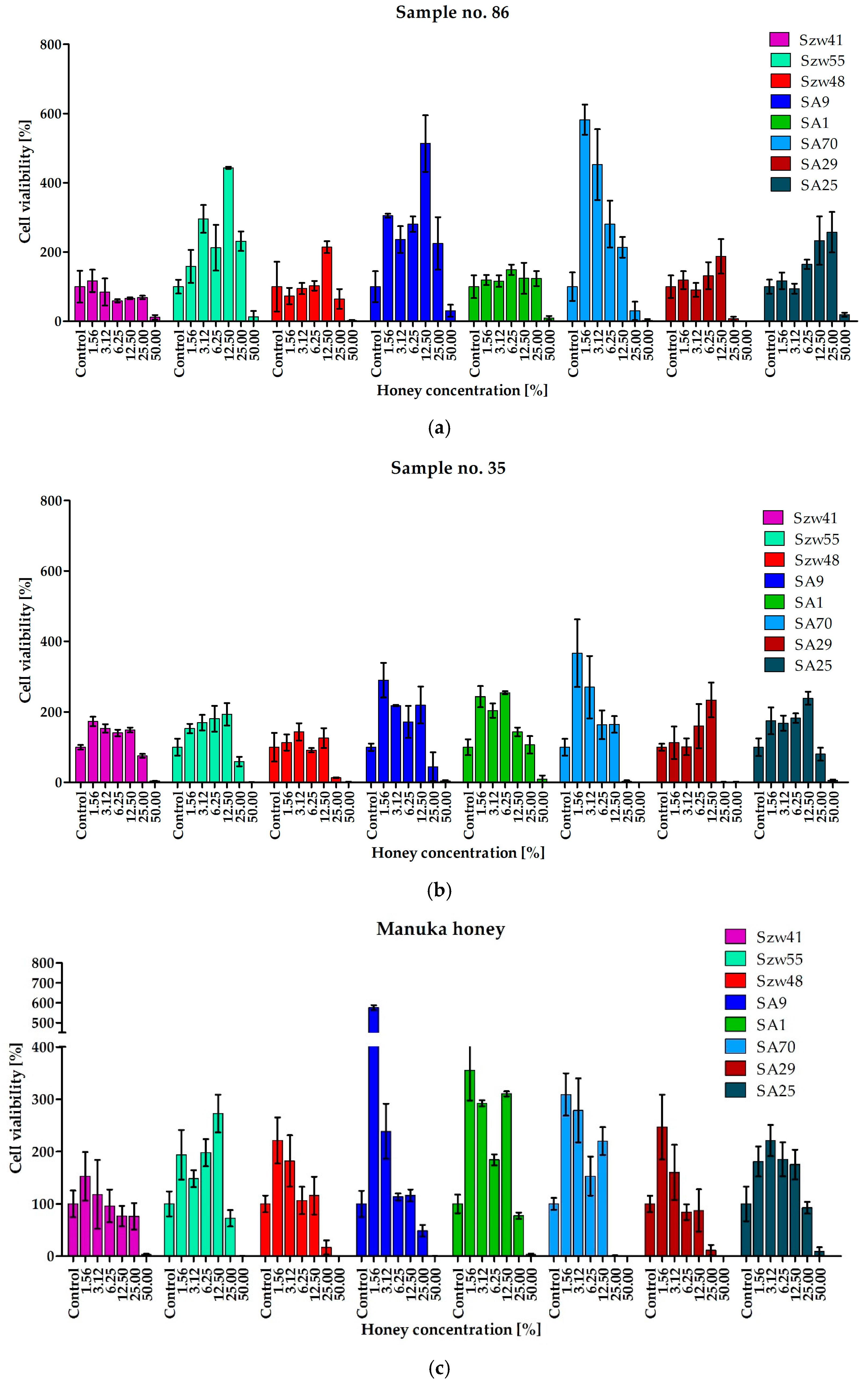
2.3. Activity of Honeys against Staphylococci Growing in the Form of Biofilm
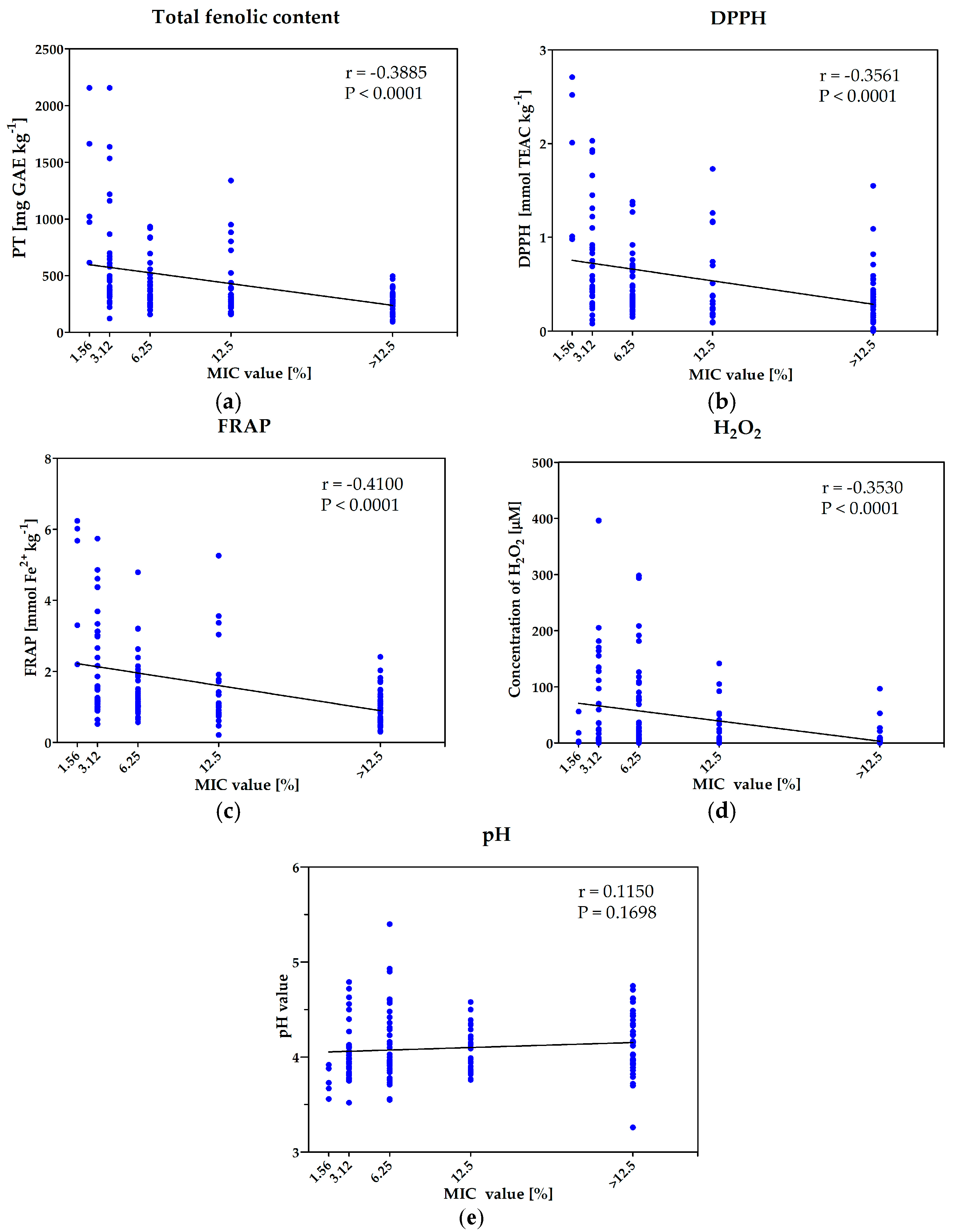
2.4. Determination of Mechanisms of Antimicrobial Activity of Investigated Honeys
3. Discussion
4. Materials and Methods
4.1. Bacterial Strains and Media
4.2. Chemicals and Reagents
4.3. Honey Samples
4.4. Investigation of Antimicrobial Potential of Honey Samples—Determination of MIC (Minimum Inhibitory Concentration) and MBC (Minimum Bactericidal Concentration) Parameters
4.5. Activity against Staphylococci Growing in the Form of Biofilm
4.5.1. Biofilm Formation Assay
4.5.2. MBEC Assay
4.5.3. Biofilm MTT-Staining
4.6. Time-Kill Assay, Determination of Kinetic of Bactericidal Effect of Honey Against Staphylococci
4.7. Determination of Hydrogen Peroxide Generation
4.8. Preliminary Determination of Mechanism of Antimicrobial Activity of Honeys—Heat and Catalase Treatment
4.9. Total Phenolics Determination
4.10. Total Antioxidant Activity (FRAP Assay)
4.11. Antiradical Activity (DPPH Assay)
4.12. Determination of Water Content
4.13. Determination of pH
4.14. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Samarghandian, S.; Farkhondeh, T.; Samini, F. Honey and health: A review of recent clinical research. Pharmacogn. Res. 2017, 9, 121–127. [Google Scholar]
- Hołderna-Kędzia, E.; Kędzia, B. Miody odmianowe i ich Znaczenie Lecznicze, 1st ed.; WDR: Włocławek, Poland, 2002; p. 112. [Google Scholar]
- Saikaly, S.K.; Khachemoune, A. Honey and wound healing: An update. Am. J. Clin. Dermatol. 2017, 18, 237–251. [Google Scholar] [CrossRef] [PubMed]
- Kwakman, P.H.; Van den Akker, J.P.; Guclu, A.; Aslami, H.; Binnekade, J.M.; de Boer, L.; Boszhard, L.; Paulus, F.; Middelhoek, P.; te Velde, A.A.; et al. Medical-grade honey kills antibiotic-resistant bacteria in vitro and eradicates skin colonization. Clin. Infect. Dis. 2008, 46, 1677–1682. [Google Scholar] [CrossRef] [PubMed]
- Kwakman, P.H.; te Velde, A.A.; de Boer, L.; Speijer, D.; Vandenbroucke-Grauls, C.M.; Zaat, S.A. How honey kills bacteria. FASEB J. 2010, 24, 2576–2582. [Google Scholar] [CrossRef] [PubMed]
- Anthimidou, E.; Mossialos, D. Antibacterial activity of Greek and Cypriot honeys against Staphylococcus aureus and Pseudomonas aeruginosa in comparison to Manuka honey. J. Med. Food 2013, 16, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Brudzynski, K.; Lannigan, R. Mechanism of honey bacteriostatic action against MRSA and VRE involves hydroxyl radicals generated from honey’s hydrogen peroxide. Front. Microbiol. 2012, 3, 36. [Google Scholar] [CrossRef] [PubMed]
- Kuncic, M.K.; Jaklic, D.; Lapanje, A.; Gunde-Cimerman, N. Antibacterial and antimycotic activities of Slovenian honeys. Br. J. Biomed. Sci. 2012, 69, 154–158. [Google Scholar] [PubMed]
- Kus, P.M.; Szweda, P.; Jerkovic, I.; Tuberoso, C.I. Activity of Polish unifloral honeys against pathogenic bacteria and its correlation with colour, phenolic content, antioxidant capacity and other parameters. Lett. Appl. Microbiol. 2016, 62, 269–276. [Google Scholar] [CrossRef] [PubMed]
- Hammond, E.N.; Duster, M.; Musuuza, J.S.; Safdar, N. Effect of United States Buckwheat honey on antibiotic-resistant hospital acquired pathogens. Pan Afr. Med. J. 2016, 25, 212. [Google Scholar] [CrossRef] [PubMed]
- Mandal, M.D.; Mandal, S. Honey: Its medicinal property and antibacterial activity. Asian Pac. J. Trop. Med. 2011, 1, 154–160. [Google Scholar] [CrossRef]
- Israili, Z.H. Antimicrobial properties of honey. Am. J. Ther. 2014, 21, 304–323. [Google Scholar] [CrossRef] [PubMed]
- Bose, B. Honey or sugar in treatment of infected wounds? Lancet 1982, 1, 963. [Google Scholar] [CrossRef]
- Taormina, P.J.; Niemira, B.A.; Beuchat, L.R. Inhibitory activity of honey against foodborne pathogens as influenced by the presence of hydrogen peroxide and level of antioxidant power. Int. J. Food Microbiol. 2001, 69, 217–225. [Google Scholar] [CrossRef]
- Mundo, M.A.; Padilla-Zakour, O.I.; Worobo, R.W. Growth inhibition of foodborne pathogens and food spoilage organisms by select raw honeys. Int. J. Food Microbiol. 2004, 97, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Brudzynski, K. Effect of hydrogen peroxide on antibacterial activities of Canadian honeys. Can. J. Microbiol. 2006, 52, 1228–1237. [Google Scholar] [CrossRef] [PubMed]
- Aljadi, A.M.; Yusoff, K.M. Isolation and identification of phenolic acids in Malaysian honey with antibacterial properties. Turkish J. Med. Sci. 2003, 33, 229–236. [Google Scholar]
- Bilikova, K.; Huang, S.C.; Lin, I.P.; Simuth, J.; Peng, C.C. Structure and antimicrobial activity relationship of royalisin, an antimicrobial peptide from royal jelly of Apis mellifera. Peptides 2015, 68, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Valachová, I.; Bučeková, M.; Majtán, J. Quantification of bee-derived peptide defensin-1 in honey by competitive enzyme-linked immunosorbent assay, a new approach in honey quality control. Czech J. Food Sci. 2016, 34, 233–243. [Google Scholar]
- Kwakman, P.H.; Zaat, S.A. Antibacterial components of honey. IUBMB Life 2012, 64, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Mavric, E.; Wittmann, S.; Barth, G.; Henle, T. Identification and quantification of methylglyoxal as the dominant antibacterial constituent of manuka (Leptospermum scoparium) honeys from New Zealand. Mol. Nutr. Food Res. 2008, 52, 483–489. [Google Scholar] [CrossRef] [PubMed]
- Brudzynski, K.; Miotto, D. Honey melanoidins: Analysis of the compositions of the high molecular weight melanoidins exhibiting radical-scavenging activity. Food Chem. 2011, 127, 1023–1030. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Churey, J.J.; Worobo, R.W. Antimicrobial activity of bacterial isolates from different floral sources of honey. Int. J. Food Microbiol. 2008, 126, 240–244. [Google Scholar] [CrossRef] [PubMed]
- Brudzynski, K.; Abubaker, K.; Miotto, D. Unraveling a mechanism of honey antibacterial action: Polyphenol/h(2)o(2)-induced oxidative effect on bacterial cell growth and on DNA degradation. Food Chem. 2012, 133, 329–336. [Google Scholar] [CrossRef] [PubMed]
- Irish, J.; Blair, S.; Carter, D.A. The antibacterial activity of honey derived from Australian flora. PLoS ONE 2011, 6, e18229. [Google Scholar] [CrossRef] [PubMed]
- Zainol, M.I.; Mohd Yusoff, K.; Mohd Yusof, M.Y. Antibacterial activity of selected Malaysian honey. BMC Complement. Altern. Med. 2013, 13, 129. [Google Scholar] [CrossRef] [PubMed]
- Brudzynski, K.; Abubaker, K.; Wang, T. Powerful bacterial killing by Buckwheat honeys is concentration-dependent, involves complete DNA degradation and requires hydrogen peroxide. Front. Microbiol. 2012, 3, 242. [Google Scholar] [CrossRef] [PubMed]
- Cao, G.; Sofic, E.; Prior, R.L. Antioxidant and prooxidant behavior of flavonoids: Structure-activity relationships. Free Radic. Biol. Med. 1997, 22, 749–760. [Google Scholar] [CrossRef]
- Sakihama, Y.; Cohen, M.F.; Grace, S.C.; Yamasaki, H. Plant phenolic antioxidant and prooxidant activities: Phenolics-induced oxidative damage mediated by metals in plants. Toxicology 2002, 177, 67–80. [Google Scholar] [CrossRef]
- Jantakee, K.; Tragoolpua, Y. Activities of different types of Thai honey on pathogenic bacteria causing skin diseases, tyrosinase enzyme and generating free radicals. Biol. Res. 2015, 48, 4. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.; Baghdad, K.; Aissat, S.; Djebli, N. Colour intensity, polyphenol content and antibacterial capacity of unheated and heat-treated Sahara honey. J. Food Process. Technol. 2016, 7, 589. [Google Scholar] [CrossRef]
- Brudzynski, K.; Miotto, D.; Kim, L.; Sjaarda, C.; Maldonado-Alvarez, L.; Fukś, H. Active macromolecules of honey form colloidal particles essential for honey antibacterial activity and hydrogen peroxide production. Sci. Rep. 2017, 7, 7637. [Google Scholar] [CrossRef] [PubMed]
- Archer, N.K.; Mazaitis, M.J.; Costerton, J.W.; Leid, J.G.; Powers, M.E.; Shirtliff, M.E. Staphylococcus aureus biofilms: Properties, regulation and roles in human disease. Virulence 2011, 2, 445–459. [Google Scholar] [CrossRef] [PubMed]
- Donlan, R.M.; Costerton, J.W. Biofilms: Survival mechanisms of clinically relevant microorganisms. Clin. Microbiol. Rev. 2002, 15, 167–193. [Google Scholar] [CrossRef] [PubMed]
- Hegazi, A.G.; Guthami, F.M.; Gethami, A.F.; Allah, F.M.; Saleh, A.A.; Fouad, E.A. Potential antibacterial activity of some Saudi Arabia honey. Vet. World 2017, 10, 233–237. [Google Scholar] [CrossRef] [PubMed]
- Salonen, A.; Virjamo, V.; Tammela, P.; Fauch, L.; Julkunen-Tiitto, R. Screening bioactivity and bioactive constituents of Nordic unifloral honeys. Food Chem. 2017, 237, 214–224. [Google Scholar] [CrossRef] [PubMed]
- Nishio, E.K.; Ribeiro, J.M.; Oliveira, A.G.; Andrade, C.G.; Proni, E.A.; Kobayashi, R.K.; Nakazato, G. Antibacterial synergic effect of honey from two stingless bees: Scaptotrigona bipunctata lepeletier, 1836, and S. postica latreille, 1807. Sci. Rep. 2016, 6, 21641. [Google Scholar] [CrossRef] [PubMed]
- Carnwath, R.; Graham, E.M.; Reynolds, K.; Pollock, P.J. The antimicrobial activity of honey against common equine wound bacterial isolates. Vet. J. 2014, 199, 110–114. [Google Scholar] [CrossRef] [PubMed]
- Sherlock, O.; Dolan, A.; Athman, R.; Power, A.; Gethin, G.; Cowman, S.; Humphreys, H. Comparison of the antimicrobial activity of Ulmo honey from Chile and Manuka honey against methicillin-resistant staphylococcus aureus, Escherichia coli and Pseudomonas aeruginosa. BMC Complement. Altern. Med. 2010, 10, 47. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Suarez, J.M.; Giampieri, F.; Brenciani, A.; Mazzoni, L.; Gasparrini, M.; González-Paramás, A.M.; Santos-Buelga, C.; Morroni, G.; Simoni, S.; Forbes-Hernández, T.Y.; et al. Apis mellifera vs Melipona beecheii Cuban polifloral honeys: A comparison based on their physicochemical parameters, chemical composition and biological properties. LWT Food Sci. Technol. 2018, 87, 272–279. [Google Scholar] [CrossRef]
- Ng, W.J.; Lim, M.S. Anti-staphylococcal activity of Melaleuca honey. Southeast Asian J. Trop. Med. Public Health 2015, 46, 472–479. [Google Scholar] [PubMed]
- Ronsisvalle, S.; Lissandrello, E.; Fuochi, V.; Petronio Petronio, G.; Straquadanio, C.; Crasci, L.; Panico, A.; Milito, M.; Cova, A.M.; Tempera, G.; et al. Antioxidant and antimicrobial properties of Casteanea sativa Miller Chestnut honey produced on Mount Etna (Sicily). Nat. Prod. Res. 2017, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Moussa, A.; Noureddine, D.; Mohamed, H.S.; Abdelmelek, M.; Saad, A. Antibacterial activity of various honey types of Algeria against staphylococcus aureus and Streptococcus pyogenes. Asian Pac. J. Trop. Med. 2012, 5, 773–776. [Google Scholar] [CrossRef]
- Al-Waili, N.; Al-Ghamdi, A.; Ansari, M.J.; Al-Attal, Y.; Salom, K. Synergistic effects of honey and propolis toward drug multi-resistant Staphylococcus aureus, Escherichia coli and Candida albicans isolates in single and polymicrobial cultures. Int. J. Med. Sci. 2012, 9, 793–800. [Google Scholar] [CrossRef] [PubMed]
- Ewnetu, Y.; Lemma, W.; Birhane, N. Antibacterial effects of Apis mellifera and stingless Bees honeys on susceptible and resistant strains of Escherichia coli, Staphylococcus aureus and Klebsiella pneumoniae in Gondar, Northwest Ethiopia. BMC Complement. Altern. Med. 2013, 13, 269. [Google Scholar] [CrossRef] [PubMed]
- Cooper, R. Honey for wound care in the 21st century. J. Wound Care 2016, 25, 544–552. [Google Scholar] [CrossRef] [PubMed]
- Hussain, M.B. Role of honey in topical and systemic bacterial infections. J. Altern. Complement. Med. 2017, 24, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Henatsch, D.; Wesseling, F.; Kross, K.W.; Stokroos, R.J. Honey and beehive products in otorhinolaryngology: A narrative review. Clin. Otolaryngol. 2016, 41, 519–531. [Google Scholar] [CrossRef] [PubMed]
- Kwakman, P.H.; Muller, M.C.; Binnekade, J.M.; van den Akker, J.P.; de Borgie, C.A.; Schultz, M.J.; Zaat, S.A. Medical-grade honey does not reduce skin colonization at central venous catheter-insertion sites of critically ill patients: A randomized controlled trial. Crit. Care 2012, 16, R214. [Google Scholar] [CrossRef] [PubMed]
- Henatsch, D.; Nabuurs, C.H.; van de Goor, R.M.; Wolffs, P.F.; Stokroos, R.J. Treatment of recurrent eczematous external otitis with honey eardrops: A proof-of-concept study. Arch. Otolaryngol. Head Neck Surg. 2017, 157, 696–699. [Google Scholar] [CrossRef] [PubMed]
- García-Tenesaca, M.; Navarrete, E.; Iturralde, G.; Villacrés Granda, I.; Tejera, E.; Beltrán-Ayala, P.; Giampieri, F.; Battino, M.; Alvarez-Suarez, J. Influence of botanical origin and chemical composition on the protective effect against oxidative damage and the capacity to reduce in vitro bacterial biofilms of Monofloral honeys from the Andean region of Ecuador. Int. J. Mol. Sci. 2017, 19, 45. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Turnbull, L.; Burke, C.M.; Liu, M.; Carter, D.A.; Schlothauer, R.C.; Whitchurch, C.B.; Harry, E.J. Manuka-type honeys can eradicate biofilms produced by staphylococcus aureus strains with different biofilm-forming abilities. PeerJ 2014, 2, e326. [Google Scholar] [CrossRef] [PubMed]
- Huttunen, S.; Riihinen, K.; Kauhanen, J.; Tikkanen-Kaukanen, C. Antimicrobial activity of different Finnish Monofloral honeys against human pathogenic bacteria. APMIS 2013, 121, 827–834. [Google Scholar] [CrossRef] [PubMed]
- Kędzia, B.; Hołderna-Kędzia, E.; Dutkowiak, A. The antibiotic activity of polish Monofloral honeys. Post. Fitoter. 2014, 2, 67–70. [Google Scholar]
- Kretavičius, J.; Kurtinaitiene, B.; Račys, J.; Čekstery, V. Inactivation of glucose oxidase during heat-treatment de-crystallization of honey. Zemdirbyste 2010, 97, 115–122. [Google Scholar]
- Brudzynski, K.; Abubaker, K.; St-Martin, L.; Castle, A. Re-examining the role of hydrogen peroxide in bacteriostatic and bactericidal activities of honey. Front. Microbiol. 2011, 2, 213. [Google Scholar] [CrossRef] [PubMed]
- Szczęsna, T.; Rybak-Chmielewska, H. Antybakteryjne Właściwości Miodu. W: Uzupełniające Zagadnienia Jakości Miodu; Wyd Inst Sadown Kwiac Oddz Pszczeln: Puławy, Poland, 1997. [Google Scholar]
- Semkiw, P. Sektor Pszczelarski w Polsce w 2016 Roku; Zakład Pszczelnictwa w Puławach: Puławy, Poland, 2016. [Google Scholar]
- Sjolund, M.; Kahlmeter, G. Staphylococci in primary skin and soft tissue infections in a Swedish county. Scand. J. Infect. Dis. 2008, 40, 894–898. [Google Scholar] [CrossRef] [PubMed]
- Szweda, P.; Schielmann, M.; Frankowska, A.; Kot, B.; Zalewska, M. Antibiotic resistance in Staphylococcus aureus strains isolated from cows with mastitis in Eastern Poland and analysis of susceptibility of resistant strains to alternative nonantibiotic agents: Lysostaphin, nisin and polymyxin B. J. Vet. Med. Sci. 2014, 76, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Szweda, P. Antimicrobial activity of honey. In Honey Analysis; Toledo, A.A., Ed.; InTech: Rijeka, Croatia, 2017. [Google Scholar]
- Walencka, E.; Sadowska, B.; Rozalska, S.; Hryniewicz, W.; Rozalska, B. Lysostaphin as a potential therapeutic agent for staphylococcal biofilm eradication. Pol. J. Microbiol. 2005, 54, 191–200. [Google Scholar] [PubMed]
- Kairo, S.K.; Bedwell, J.; Tyler, P.C.; Carter, A.; Corbel, M.J. Development of a tetrazolium salt assay for rapid determination of viability of BCG vaccines. Vaccine 1999, 17, 2423–2428. [Google Scholar] [CrossRef]
- Kuś, P.; Congiu, F.; Teper, D.; Sroka, Z.; Jerkovi, C.I.; Tuberoso, C.I.G. Antioxidant activity, color characteristics, total phenol content and general HPLC fingerprints of six Polish unifloral honey types. LWT Food Sci. Technol. 2013, 55, 124–130. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are available from the authors. |


| No. | MIC (%) (v/v) against Different Strains of Bacteria | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| S. aureus ATCC 25923 | S. aureus ATCC 29213 | S. epidermidis ATCC 12228 | E. coli ATCC 25922 | P. aeruginosa ATCC 27853 | ||||||
| MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | |
| 1 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 | >12.5 | >12.5 | 12.5 | 12.5 |
| 2 | 6.25 | >12.5 | 6.25 | >12.5 | 6.25 | >12.5 | >12.5 | >12.5 | 12.5 | 12.5 |
| 3 | 3.125 | 3.125 | 6.25 | 6.25 | 3.125 | 3.125 | 12.5 | 12.5 | 6.25 | 6.25 |
| 4 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 | >12.5 | >12.5 | >12.5 | >12.5 |
| 5 | 3.125 | 6.25 | 3.125 | 3.125 | 3.125 | 6.25 | 12.5 | 12.5 | 12.5 | 12.5 |
| 6 | 12.5 | >12.5 | 12.5 | 12.5 | 12.5 | 12.5 | >12.5 | >12.5 | 12.5 | 12.5 |
| 7 | 3.125 | 12.5 | 6.25 | 6.25 | 3.125 | 3.125 | 12.5 | 12.5 | 6.25 | 12.5 |
| 8 | 3.125 | 3.125 | 3.125 | 3.125 | 3.125 | 3.125 | 12.5 | 12.5 | 6.25 | 12.5 |
| 9 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 12.5 | 12.5 | 12.5 | 12.5 |
| 10 | 12.5 | 12.5 | 12.5 | >12.5 | 12.5 | 12.5 | >12.5 | >12.5 | >12.5 | >12.5 |
| 11 | 6.25 | 6.25 | 6.25 | 6.25 | 3.125 | 6.25 | 12.5 | 12.5 | 12.5 | 12.5 |
| 12 | 12.5 | >12.5 | 6.25 | >12.5 | 6.25 | >12.5 | >12.5 | >12.5 | 12.5 | 12.5 |
| 13 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 | >12.5 | 12.5 | 12.5 |
| 15 | >12.5 | >12.5 | >12.5 | >12.5 | 12.5 | >12.5 | >12.5 | >12.5 | >12.5 | >12.5 |
| 16 | 3.125 | 3.125 | 3.125 | 3.125 | 1.56 | 1.56 | 6.25 | 6.25 | 6.25 | 6.25 |
| 20 | 6.25 | 6.25 | 6.25 | 12.5 | 6.25 | 6.25 | 12.5 | 12.5 | 12.5 | >12.5 |
| 21 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 | >12.5 | >12.5 | 12.5 | >12.5 |
| 22 | 3.125 | 6.25 | 3.125 | 3.125 | 3.125 | 3.125 | 12.5 | 12.5 | 6.25 | 12.5 |
| 23 | 3.125 | 6.25 | 3.125 | 6.25 | 3.125 | 6.25 | 12.5 | 12.5 | 6.25 | 12.5 |
| 24 | 3.125 | 6.25 | 3.125 | 3.125 | 3.125 | 3.125 | 6.25 | 6.25 | 6.25 | 6.25 |
| 25 | 3.125 | 6.25 | 3.125 | 6.25 | 3.125 | 3.125 | 12.5 | 12.5 | 6.25 | 12.5 |
| 27 | 6.25 | 6.25 | 12.5 | 12.5 | 6.25 | 6.25 | 12.5 | 12.5 | 12.5 | 12.5 |
| 28 | 3.125 | 3.125 | 3.125 | 6.25 | 3.125 | 3.125 | 12.5 | 12.5 | 6.25 | 6.25 |
| 30 | 6.25 | 6.25 | 6.25 | 12.5 | 6.25 | 12.5 | 12.5 | >12.5 | 12.5 | 12.5 |
| 31 | 3.125 | 3.125 | 3.125 | 3.125 | 3.125 | 3.125 | 12.5 | 12.5 | 6.25 | 6.25 |
| 32 | 12.5 | >12.5 | >12.5 | >12.5 | 12.5 | >12.5 | >12.5 | >12.5 | 12.5 | >12.5 |
| 33 | 12.5 | >12.5 | 12.5 | >12.5 | 12.5 | >12.5 | >12.5 | >12.5 | 12.5 | >12.5 |
| 34 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 12.5 | 12.5 | 12.5 | 12.5 |
| 35 | 1.56 | 1.56 | 1.56 | 1.56 | 1.56 | 1.56 | 3.125 | 3.125 | 3.125 | 3.125 |
| 37 | 6.25 | >12.5 | 6.25 | >12.5 | 12.5 | >12.5 | >12.5 | >12.5 | 12.5 | >12.5 |
| 39 | >12.5 | >12.5 | >12.5 | >12.5 | 12.5 | >12.5 | >12.5 | >12.5 | >12.5 | >12.5 |
| 40 | 6.25 | 12.5 | 6.25 | 6.25 | 6.25 | 6.25 | 12.5 | 12.5 | 12.5 | 12.5 |
| 41 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 12.5 | 12.5 | 12.5 | 12.5 |
| 42 | 12.5 | >12.5 | 12.5 | >12.5 | 12.5 | >12.5 | >12.5 | >12.5 | >12.5 | >12.5 |
| 43 | 3.125 | 6.25 | 3.125 | 6.25 | 3.125 | 6.25 | 12.5 | >12.5 | 12.5 | 12.5 |
| 44 | 6.25 | 6.25 | 3.125 | 3.125 | 3.125 | 6.25 | 12.5 | 12.5 | 6.25 | 12.5 |
| 45 | 3.125 | 6.25 | 6.25 | 6.25 | 3.125 | 6.25 | 12.5 | >12.5 | 12.5 | >12.5 |
| 46 | 12.5 | >12.5 | 12.5 | >12.5 | 12.5 | >12.5 | >12.5 | >12.5 | 12.5 | >12.5 |
| 47 | 3.125 | 6.25 | 3.125 | 6.25 | 3.125 | 3.125 | 12.5 | 12.5 | 6.25 | 12.5 |
| 49 | >12.5 | >12.5 | >12.5 | >12.5 | 12.5 | >12.5 | >12.5 | >12.5 | 12.5 | >12.5 |
| 50 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 12.5 | 12.5 | 12.5 | 12.5 |
| 51 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 12.5 | 12.5 | 12.5 | 12.5 |
| 52 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 12.5 | 12.5 | 12.5 | 12.5 |
| 53 | 6.25 | 6.25 | 6.25 | 12.5 | 6.25 | >12.5 | >12.5 | >12.5 | 12.5 | >12.5 |
| 54 | 3.125 | 6.25 | 6.25 | 6.25 | 3.125 | 6.25 | >12.5 | >12.5 | 12.5 | >12.5 |
| 55 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 |
| 57 | 3.125 | 3.125 | 3.125 | 3.125 | 3.125 | 6.25 | 12.5 | 12.5 | 6.25 | 12.5 |
| 60 | 6.25 | 6.25 | 6.25 | 12.5 | 6.25 | 12.5 | 12.5 | >12.5 | 12.5 | >12.5 |
| 61 | 12.5 | >12.5 | >12.5 | >12.5 | 12.5 | >12.5 | 12.5 | >12.5 | 12.5 | >12.5 |
| 62 | 6.25 | 6.25 | 6.25 | 6.25 | 3.125 | 3.125 | 12.5 | 12.5 | 12.5 | 12.5 |
| 63 | 1.56 | 3.125 | 1.56 | 3.125 | 1.56 | 1.56 | 6.25 | 12.5 | 6.25 | 6.25 |
| 65 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 | >12.5 | >12.5 | 12.5 | >12.5 |
| 66 | 6.25 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 |
| 67 | >12.5 | >12.5 | >12.5 | >12.5 | 12.5 | 12.5 | >12.5 | >12.5 | >12.5 | >12.5 |
| 69 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 |
| 71 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 12.5 | 12.5 | 12.5 | 12.5 |
| 72 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | >12.5 | >12.5 | 12.5 | >12.5 |
| 73 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 12.5 | 12.5 | 12.5 | 12.5 |
| 74 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | >12.5 | >12.5 | 12.5 | >12.5 |
| 76 | 3.125 | 3.125 | 3.125 | 3.125 | 3.125 | 3.125 | 6.25 | 12.5 | 6.25 | 6.25 |
| 77 | 3.125 | 6.25 | 6.25 | 12.5 | 6.25 | 6.25 | 12.5 | 12.5 | 12.5 | 12.5 |
| 78 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 12.5 | 12.5 | 12.5 | 12.5 |
| 80 | 12.5 | >12.5 | >12.5 | >12.5 | 12.5 | >12.5 | >12.5 | >12.5 | 12.5 | >12.5 |
| 81 | 1.56 | 1.56 | 1.56 | 1.56 | 1.56 | 1.56 | 6.25 | 6.25 | 3.125 | 6.25 |
| 82 | 12.5 | >12.5 | 12.5 | >12.5 | 6.25 | 12.5 | >12.5 | >12.5 | 12.5 | >12.5 |
| 83 | 1.56 | 6.25 | 1.56 | 1.56 | 1.56 | 1.56 | 6.25 | 6.25 | 3.125 | 6.25 |
| 84 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 | >12.5 | >12.5 | >12.5 | >12.5 |
| 85 | 3.125 | 3.125 | 6.25 | 6.25 | 3.125 | 3.125 | 12.5 | 12.5 | 6.25 | 12.5 |
| 86 | 1.56 | 3.125 | 1.56 | 1.56 | 1.56 | 1.56 | 3.125 | 12.5 | 3.125 | 6.25 |
| 87 | 3.125 | 3.125 | 3.125 | 6.25 | 3.125 | 3.125 | >12.5 | >12.5 | 6.25 | >12.5 |
| 88 | 3.125 | 3.125 | 3.125 | 3.125 | 3.125 | 3.125 | 6.25 | 6.25 | 6.25 | 6.25 |
| 89 | 3.125 | 3.125 | 3.125 | 3.125 | 1.56 | 3.125 | 6.25 | 6.25 | 6.25 | 6.25 |
| 91 | 12.5 | >12.5 | >12.5 | >12.5 | 12.5 | >12.5 | >12.5 | >12.5 | 12.5 | >12.5 |
| 92 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 12.5 | 12.5 | 12.5 | 12.5 |
| 94 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | >12.5 | >12.5 | 12.5 | >12.5 |
| 95 | 12.5 | >12.5 | 12.5 | 12.5 | 12.5 | 12.5 | >12.5 | >12.5 | 12.5 | >12.5 |
| 96 | 6.25 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 | >12.5 | 12.5 | >12.5 |
| 98 | 6.25 | 12.5 | 6.25 | 6.25 | 6.25 | 6.25 | 12.5 | 12.5 | 12.5 | 12.5 |
| 99 | 6.25 | 12.5 | 12.5 | 12.5 | 6.25 | 12.5 | 12.5 | >12.5 | 12.5 | 12.5 |
| 104 | 12.5 | >12.5 | 12.5 | >12.5 | 12.5 | 12.5 | >12.5 | >12.5 | >12.5 | >12.5 |
| 105 | 3.125 | 6.25 | 3.125 | 3.125 | 3.125 | 3.125 | 6.25 | 12.5 | 6.25 | 6.25 |
| 106 | 12.5 | 12.5 | 12.5 | >12.5 | 12.5 | 12.5 | >12.5 | >12.5 | >12.5 | >12.5 |
| 107 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 12.5 | >12.5 | 12.5 | 12.5 |
| 108 | 3.125 | 3.125 | 3.125 | 3.125 | 3.125 | 3.125 | 6.25 | 6.25 | 6.25 | 6.25 |
| 109 | 6.25 | 12.5 | 6.25 | 6.25 | 6.25 | 6.25 | 12.5 | >12.5 | 12.5 | 12.5 |
| 112 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 | >12.5 | >12.5 | 12.5 | >12.5 |
| 114 | 12.5 | 12.5 | 12.5 | >12.5 | 12.5 | 12.5 | >12.5 | >12.5 | >12.5 | >12.5 |
| 117 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 12.5 | >12.5 | 12.5 | 12.5 |
| 122 | 6.25 | 12.5 | 12.5 | 12.5 | 6.25 | 12.5 | 12.5 | 12.5 | 12.5 | >12.5 |
| 123 | 3.125 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 12.5 | 12.5 | 12.5 | 12.5 |
| 124 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 12.5 | 12.5 | 12.5 | 12.5 |
| 125 | 3.125 | 6.25 | 3.125 | 12.5 | 3.125 | 3.125 | 12.5 | 12.5 | 6.25 | >12.5 |
| 127 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 12.5 | 12.5 | 12.5 | 12.5 |
| 128 | 3.125 | 3.125 | 3.125 | 3.125 | 3.125 | 3.125 | 6.25 | 12.5 | 6.25 | 6.25 |
| 129 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 | 12.5 | >12.5 | 12.5 | >12.5 |
| 130 | 3.125 | 3.125 | 3.125 | 3.125 | 3.125 | 3.125 | 12.5 | 12.5 | 6.25 | 12.5 |
| 131 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 12.5 | 12.5 | 12.5 | 12.5 |
| 132 | 6.25 | >12.5 | 12.5 | >12.5 | 12.5 | 12.5 | >12.5 | >12.5 | 12.5 | >12.5 |
| 133 | 3.125 | 6.25 | 3.125 | 6.25 | 3.125 | 3.125 | 12.5 | 12.5 | 6.25 | 12.5 |
| 134 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 12.5 | >12.5 | 12.5 | 12.5 |
| 135 | 3.125 | 3.125 | 3.125 | 3.125 | 3.125 | 3.125 | 12.5 | 12.5 | 12.5 | 12.5 |
| 137 | 6.25 | 6.25 | 6.25 | 6.25 | 3.125 | 3.125 | 12.5 | 12.5 | 12.5 | 12.5 |
| 138 | 3.125 | 6.25 | 3.125 | 3.125 | 3.125 | 3.125 | 12.5 | 12.5 | 12.5 | 12.5 |
| 139 | 3.125 | 3.125 | 3.125 | 6.25 | 3.125 | 3.125 | 12.5 | 12.5 | 6.25 | 12.5 |
| 140 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 6.25 | 12.5 | >12.5 | 12.5 | 12.5 |
| 142 | >12.5 | >12.5 | 12.5 | >12.5 | 12.5 | 12.5 | 12.5 | 12.5 | >12.5 | >12.5 |
| 143 | >12.5 | >12.5 | >12.5 | >12.5 | 12.5 | 12.5 | >12.5 | >12.5 | 12.5 | >12.5 |
| 144 | 3.125 | 6.25 | 3.125 | 3.125 | 3.125 | 6.25 | 6.25 | 6.25 | 6.25 | 12.5 |
| No. | MIC and MBC (%) (v/v) against Staphylococcus aureus Isolates | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| SA1 | SA9 | SA70 | SA103 | SA105 | SA102 | |||||||
| MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | |
| 35 | 1.56 | 1.56 | 0.78 | 3.125 | 0.78 | 1.56 | 1.56 | 6.25 | 1.56 | 6.25 | 1.56 | 6.25 |
| 24 | 1.56 | 1.56 | 1.56 | 6.25 | 1.56 | 3.125 | 1.56 | >12.5 | 1.56 | 12.5 | 1.56 | >12.5 |
| 25 | 3.125 | 6.25 | 3.125 | 12.5 | 3.125 | 3.125 | 3.125 | >12.5 | 3.125 | >12.5 | 3.125 | >12.5 |
| 86 | 1.56 | 1.56 | 1.56 | 1.56 | 1.56 | 1.56 | 1.56 | 6.25 | 1.56 | 6.25 | 1.56 | 6.25 |
| 76 | 3.125 | 3.125 | 3.125 | 6.25 | 3.125 | 3.125 | 3.125 | >12.5 | 1.56 | >12.5 | 3.125 | >12.5 |
| 128 | 3.125 | 3.125 | 3.125 | 6.25 | 3.125 | 3.125 | 3.125 | >12.5 | 3.125 | >12.5 | 3.125 | >12.5 |
| 105 | 3.125 | 3.125 | 3.125 | 12.5 | 3.125 | 3.125 | 3.125 | >12.5 | 3.125 | >12.5 | 3.125 | 12.5 |
| 81 | 1.56 | 1.56 | 1.56 | 3.125 | 1.56 | 3.125 | 1.56 | 12.5 | 1.56 | 6.25 | 1.56 | 12.5 |
| 108 | 3.125 | 3.125 | 3.125 | 6.25 | 3.125 | 3.125 | 3.125 | 12.5 | 3.125 | 12.5 | 3.125 | 12.5 |
| 89 | 1.56 | 3.125 | 1.56 | 1.56 | 1.56 | 3.125 | 1.56 | >12.5 | 1.56 | 12.5 | 1.56 | >12.5 |
| 139 | 3.125 | 3.125 | 3.125 | 6.25 | 3.125 | 3.125 | 3.125 | >12.5 | 3.125 | >12.5 | 3.125 | >12.5 |
| 83 | 1.56 | 1.56 | 1.56 | 6.25 | 3.125 | 3.125 | 1.56 | 6.25 | 1.56 | 6.25 | 1.56 | 6.25 |
| No. | MIC and MBC (%) (v/v) against Staphylococcus aureus Isolates | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Szw35 | Szw41 | Szw17 | Szw48 | Szw55 | Szw16 | |||||||
| MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | |
| 35 | 1.56 | 6.25 | 1.56 | 6.25 | 1.56 | 3.125 | 1.56 | 6.25 | 1.56 | 6.25 | 1.56 | 3.125 |
| 24 | 1.56 | 12.5 | 1.56 | 12.5 | 1.56 | 6.25 | 1.56 | 12.5 | 1.56 | 12.5 | 1.56 | 6.25 |
| 25 | 3.125 | >12.5 | 3.125 | >12.5 | 3.125 | >12.5 | 3.125 | >12.5 | 3.125 | >12.5 | 3.125 | >12.5 |
| 86 | 1.56 | 3.125 | 1.56 | 6.25 | 1.56 | 3.125 | 1.56 | 3.125 | 1.56 | 6.25 | 1.56 | 3.125 |
| 76 | 3.125 | >12.5 | 3.125 | 12.5 | 3.125 | 12.5 | 3.125 | >12.5 | 3.125 | 12.5 | 3.125 | 12.5 |
| 128 | 3.125 | >12.5 | 3.125 | >12.5 | 3.125 | >12.5 | 3.125 | >12.5 | 3.125 | >12.5 | 3.125 | >12.5 |
| 105 | 3.125 | >12.5 | 3.125 | >12.5 | 3.125 | 12.5 | 3.125 | >12.5 | 3.125 | >12.5 | 3.125 | 12.5 |
| 81 | 1.56 | 6.25 | 1.56 | 6.25 | 1.56 | 6.25 | 1.56 | 6.25 | 1.56 | 6.25 | 1.56 | 6.25 |
| 108 | 3.125 | 12.5 | 3.125 | 12.5 | 3.125 | 12.5 | 3.125 | 12.5 | 3.125 | 12.5 | 3.125 | 12.5 |
| 89 | 3.125 | 12.5 | 1.56 | 12.5 | 1.56 | 12.5 | 3.125 | 12.5 | 1.56 | 12.5 | 1.56 | 12.5 |
| 139 | 3.125 | >12.5 | 3.125 | >12.5 | 3.125 | >12.5 | 3.125 | >12.5 | 3.125 | >12.5 | 3.125 | >12.5 |
| 83 | 1.56 | 6.25 | 1.56 | 6.25 | 1.56 | 6.25 | 1.56 | 6.25 | 1.56 | 6.25 | 1.56 | 6.25 |
| MBEC50 (%) (v/v) against Staphylococcus aureus Isolates | ||||||||
|---|---|---|---|---|---|---|---|---|
| Szw41 | Szw55 | Szw48 | SA9 | SA1 | SA70 | S. aureus ATCC 25923 | S. aureus ATCC 29213 | |
| Sample No. 35 | 50 | 50 | 25 | 25 | 50 | 25 | 50 | 25 |
| Sample No. 86 | 50 | 50 | 50 | 50 | 50 | 25 | 50 | 25 |
| Manuka honey | 50 | 50 | 25 | 25 | 50 | 25 | 50 | 25 |
| No. | MIC and MBC (%) (v/v) against Staphylococcus aureus ATCC 25923 | |||||||
|---|---|---|---|---|---|---|---|---|
| 40 °C | 60 °C | 80 °C | Catalase | Untreated Control | ||||
| 10’ | 20’ | 10’ | 20’ | 10’ | 20’ | |||
| 35 | 1.56 | 1.56 | 6.25 | 6.25 | >12.5 | >12.5 | 12.5 | 1.56 |
| 24 | 6.25 | 6.25 | >12.5 | >12.5 | >12.5 | >12.5 | >12.5 | 3.125 |
| 25 | 3.125 | 3.125 | 6.25 | 12.5 | >12.5 | >12.5 | >12.5 | 3.125 |
| 86 | 1.56 | 1.56 | 6.25 | 12.5 | >12.5 | >12.5 | >12.5 | 1.56 |
| 76 | 3.125 | 3.125 | >12.5 | >12.5 | >12.5 | >12.5 | >12.5 | 3.125 |
| 128 | 3.125 | 3.125 | 3.125 | 12.5 | >12.5 | >12.5 | >12.5 | 3.125 |
| 105 | 6.25 | 6.25 | 12.5 | >12.5 | >12.5 | >12.5 | >12.5 | 3.125 |
| 81 | 1.56 | 1.56 | 12.5 | >12.5 | >12.5 | >12.5 | >12.5 | 1.56 |
| 108 | 3.125 | 3.125 | >12.5 | >12.5 | >12.5 | >12.5 | >12.5 | 3.125 |
| 89 | 3.125 | 3.125 | >12.5 | >12.5 | >12.5 | >12.5 | >12.5 | 3.125 |
| 139 | 3.125 | 3.125 | 12.5 | >12.5 | >12.5 | >12.5 | >12.5 | 3.125 |
| 83 | 1.56 | 1.56 | 6.25 | 12.5 | >12.5 | >12.5 | >12.5 | 1.56 |
| Sample Code | Honey Plant Source | n | Location |
|---|---|---|---|
| Professional Beekeepers; n = 95 | |||
| 1–58 | Multi-floral | n = 58 | Poland |
| 59–62 | Multi-floral collected from forest area | n = 4 | Poland |
| 63–64 | Multi-floral collected from peatland | n = 2 | Poland |
| 65–68 | Rapeseed (Brassica napus L.) 1 | n = 4 | Poland |
| 69–70 | Black locust (Robinia pseudoacacia L.) 1 | n = 2 | Poland |
| 71–76 | Lime tree (Tilia spp.) 1 | n = 6 | Poland |
| 77–80 | Honeydew 1 | n = 4 | Poland |
| 81–91 | Buckwheat (Fagopyrum esculentum Moench) 1 | n = 11 | Poland |
| 92 | Sunflowers (Helianthus spp.) 1 | n = 1 | Poland |
| 93 | Cowberry. chokeberry (Vaccinium spp., Aronia spp) 1 | n = 1 | Poland |
| 94 | Raspberry (Rubus spp.) 1 | n = 1 | Poland |
| 95 | Dandelion (Taraxacum officinale F.H. Wigg) 1 | n = 1 | Poland |
| Organic Grocery Store; n = 22 | |||
| 96–97 | Multi-floral | n = 2 | Poland |
| 98 | Multi-floral collected from forest area | n = 1 | Poland |
| 99–100 | Rapeseed (Brassica napus L.) 2 | n = 2 | Poland |
| 101–104 | Black locust (Robinia pseudoacacia L.) 2 | n = 4 | Poland |
| 105–107 | Lime tree (Tilia spp.) 2 | n = 3 | Poland |
| 108–110 | Honeydew 2 | n = 3 | Poland |
| 111–112 | Buckwheat (Fagopyrum esculentum Moench) 2 | n = 2 | Poland |
| 113 | Sunflowers (Helianthus spp.) 2 | n = 1 | Poland |
| 114 | Raspberry (Rubus spp.) 2 | n = 1 | Poland |
| 115 | Dandelion (Taraxacum officinale F.H. Wigg) 2 | n = 1 | Poland |
| 116 | Tansy phacelia (Phacelia tanacetifolia Benth.) 2 | n = 1 | Poland |
| 117 | Clover (Trifolium spp.) 2 | n = 1 | Poland |
| Supermarket; n = 12 | |||
| 118–119 | Multi-floral | n = 2 | Poland |
| 120 | Rapeseed (Brassica napus L.) 2 | n = 1 | Poland |
| 121 | Black locust (Robinia pseudoacacia L.)2 | n = 1 | Poland |
| 122 | Lime tree (Tilia spp.) 2 | n = 1 | Poland |
| 123 | Lime tree (Tilia spp.) 2 | n = 1 | Poland. Bulgaria |
| 124–126 | Honeydew 2 | n = 3 | Poland |
| 127–128 | Buckwheat (Fagopyrum esculentum Moench) 2 | n = 2 | Poland |
| 129 | Buckwheat (Fagopyrum esculentum Moench) 2 | n = 1 | UE countries |
| Foreign; n = 15 | |||
| 130 | Multi-floral | n = 1 | Italy |
| 131–133 | Multi-floral | n = 3 | Kazakhstan |
| 134–136 | Multi-floral | n = 3 | Ukraine |
| 137 | Multi-floral | n = 1 | Ethiopia |
| 138 | Multi-floral | n = 1 | Crete |
| 139 | Multi-floral | n = 1 | Kazakhstan |
| t140 | Chestnut (Aesculus spp.) 2 | n = 1 | Italy |
| 141 | Eucalyptus (Eucalyptus spp.) 2 | n = 1 | Italy |
| 142 | Orange tree (Citrus spp.) 2 | n = 1 | Italy |
| 143 | Sunflowers (Helianthus spp.) 2 | n = 1 | Kazakhstan |
| 144 | Manuka (Leptospermum scoparium J.R. Forst. & G. Forst) 2 | n = 1 | New Zealand |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grecka, K.; Kuś, P.M.; Worobo, R.W.; Szweda, P. Study of the Anti-Staphylococcal Potential of Honeys Produced in Northern Poland. Molecules 2018, 23, 260. https://doi.org/10.3390/molecules23020260
Grecka K, Kuś PM, Worobo RW, Szweda P. Study of the Anti-Staphylococcal Potential of Honeys Produced in Northern Poland. Molecules. 2018; 23(2):260. https://doi.org/10.3390/molecules23020260
Chicago/Turabian StyleGrecka, Katarzyna, Piotr M. Kuś, Randy W. Worobo, and Piotr Szweda. 2018. "Study of the Anti-Staphylococcal Potential of Honeys Produced in Northern Poland" Molecules 23, no. 2: 260. https://doi.org/10.3390/molecules23020260
APA StyleGrecka, K., Kuś, P. M., Worobo, R. W., & Szweda, P. (2018). Study of the Anti-Staphylococcal Potential of Honeys Produced in Northern Poland. Molecules, 23(2), 260. https://doi.org/10.3390/molecules23020260





