Effects of Toona sinensis Leaf Extract and Its Chemical Constituents on Xanthine Oxidase Activity and Serum Uric Acid Levels in Potassium Oxonate-Induced Hyperuricemic Rats
Abstract
1. Introduction
2. Results and Discussion
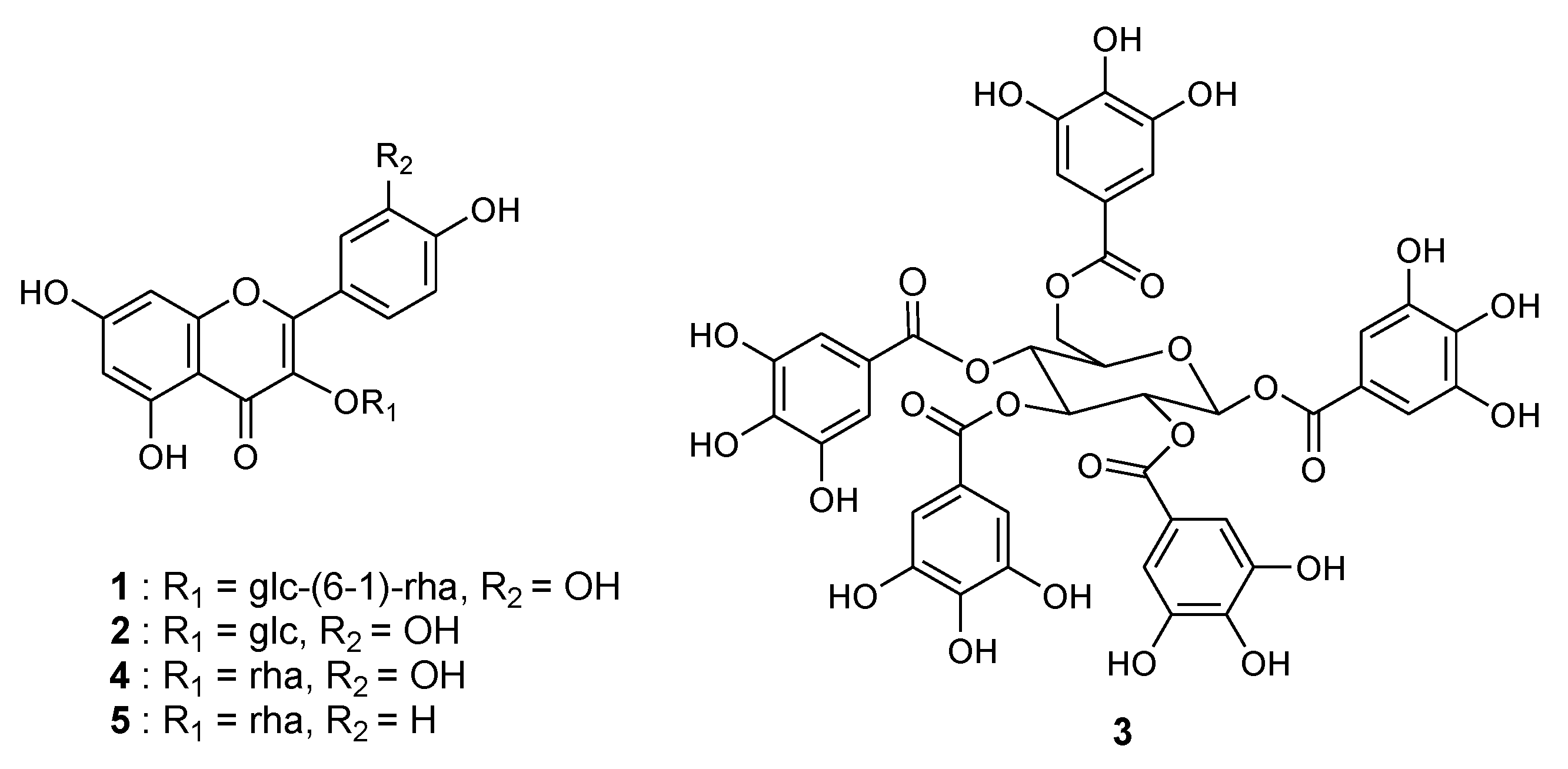
2.1. Bioassay-Guided Isolation and Structural Identification
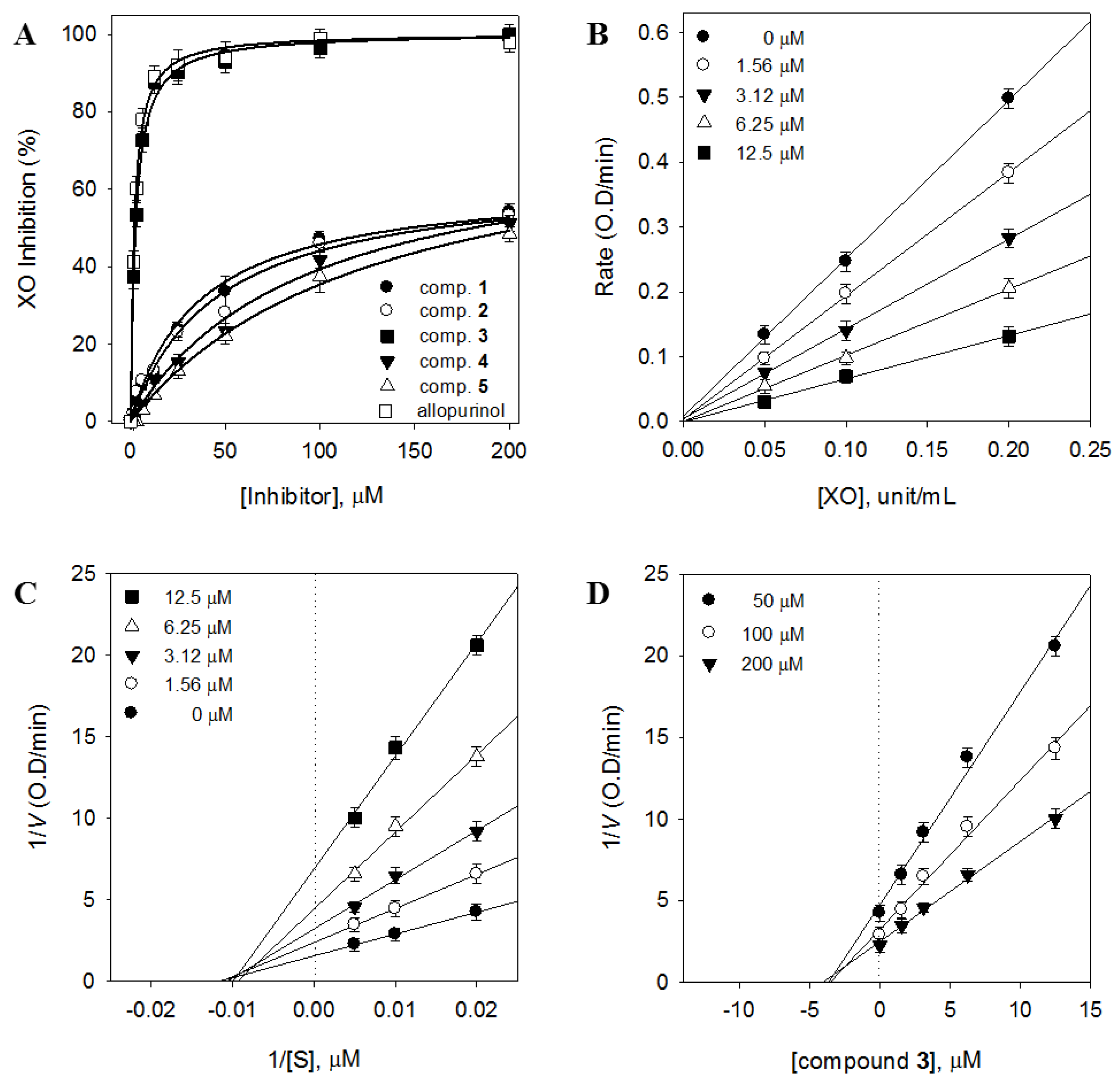
2.2. Contribution of the Identified Compounds to XO Inhibitory Activity
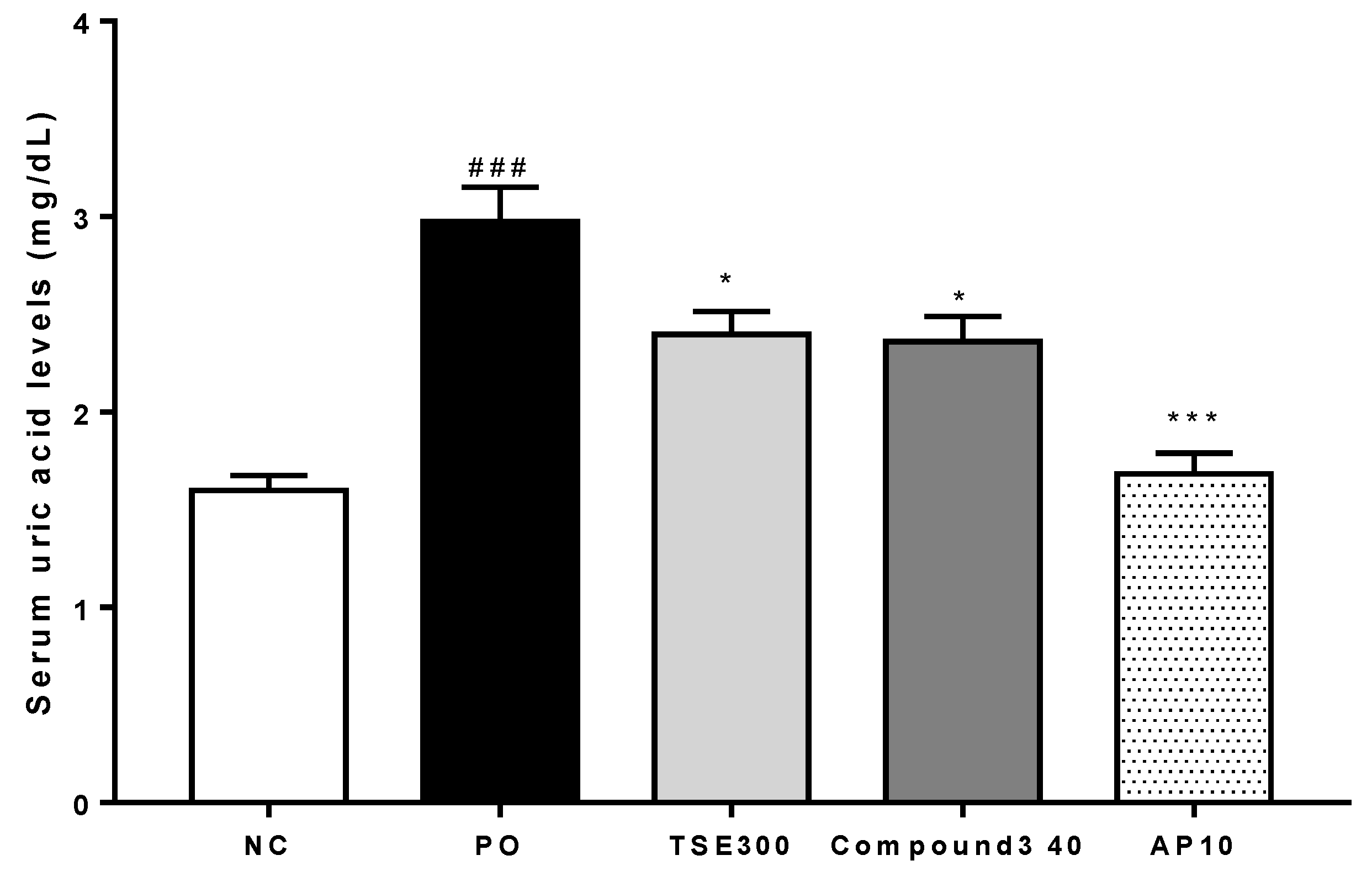
2.3. Serum Uric Acid Levels in Rats with PO-Induced Hyperuricemia
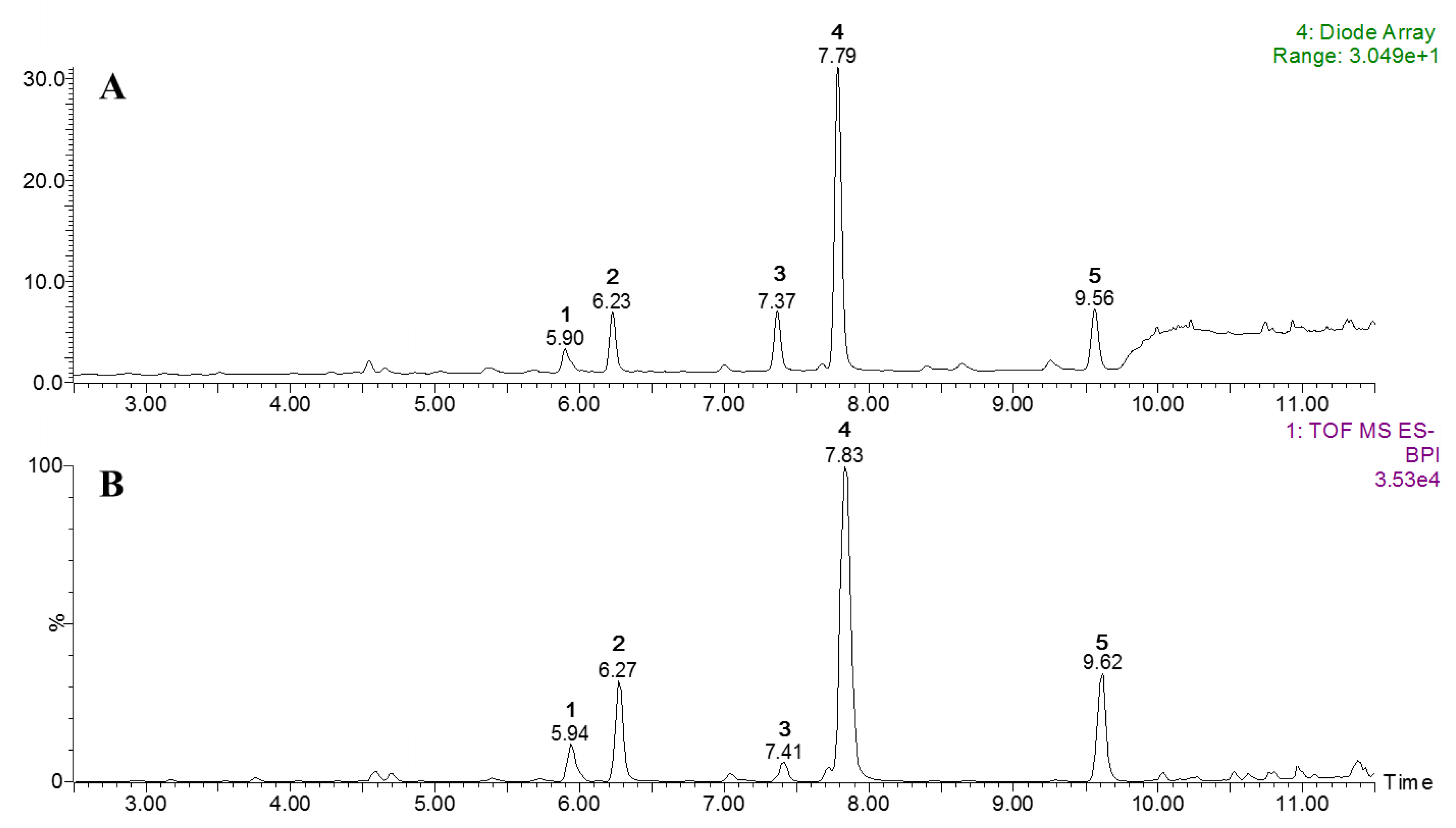
2.4. Ultraperformance Liquid Chromatography-Quadrupole Time-of-Flight Mass Spectrometry (UPLC-qToF MS) Profiles
3. Materials and Methods
3.1. Plant Materials and Sample Preparation
3.2. Instruments
3.3. Sample Extraction, Fractionation, and Isolation
3.4. UPLC-qToF MS Analysis
3.5. In Vitro XO Assay
3.6. Induction of Hyperuricemia and Sample Treatment
3.7. Serum Uric Acid Analysis
3.8. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Gibson, T. Hyperuricemia, gout and the kidney. Curr. Opin. Rheumatol. 2012, 24, 127–131. [Google Scholar] [CrossRef] [PubMed]
- Bitik, B.; Öztürk, M.A. An old disease with new insights: Update on diagnosis and treatment of gout. Eur. J. Rheumatol. 2014, 1, 72–77. [Google Scholar] [CrossRef] [PubMed]
- Martillo, M.A.; Nazzal, L.; Crittenden, D.B. The crystallization of monosodium urate. Curr. Rheumatol. Rep. 2014, 16, 400. [Google Scholar] [CrossRef] [PubMed]
- Redon, P.; Maloberti, A.; Facchetti, R.; Redon, J.; Lurbe, E.; Bombelli, M.; Mancia, G.; Grassi, G. Gender-related differences in serum uric acid in treated hypertensive patients from central and east European countries: findings from the blood pressure control rate and cardiovascular risk profile study. J. Hypertens. 2018. [Google Scholar] [CrossRef] [PubMed]
- Maloberti, A.; Maggioni, S.; Occhi, L.; Triglione, N.; Panzeri, F.; Nava, S.; Signorini, S.; Falbo, R.; Casati, M.; Grassi, G.; Giannattasio, C. Sex-related relationships between uric acid and target organ damage in hypertension. J. Clin. Hypertens. (Greenwich). 2018, 20, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Fukunari, A.; Okamoto, K.; Nishino, T.B.; Eger, T.; Pai, E.F.; Kamezawa, M.; Yamada, I.; Kato, N. Y-700 [1-[3-Cyano-4-(2,2-dimethylpropoxy)phenyl]-1H-pyrazole-4-carboxylic acid]: A potent xanthine oxidoreductase inhibitor with hepatic excretion. J. Pharmacol. Exp. Ther. 2004, 311, 519–528. [Google Scholar] [CrossRef] [PubMed]
- Terkeltaub, R.A. Clinical practice. Gout. N. Engl. J. Med. 2003, 349, 1647–1655. [Google Scholar] [CrossRef]
- Huo, L.N.; Wang, W.; Zhang, C.Y.; Shi, H.B.; Liu, Y.; Liu, X.H.; Guo, B.H.; Zhao, D.M.; Gao, H. Bioassy-guided isolation and identification of xanthine oxidase inhibitory constituents from the leaves of Perilla frutescens. Molecules 2015, 20, 17848–17859. [Google Scholar] [CrossRef]
- Chen, C.M.; Lin, C.Y.; Lin, L.C.; Wan, T.C. Antioxidation activity and total phenolic contents of various Toona sinensis extracts. Afr. J. Biotechnol. 2012, 11, 13831–13837. [Google Scholar] [CrossRef]
- Hsieh, T.J.; Wang, J.C.; Hu, C.Y.; Li, C.T.; Kuo, C.M.; Hsieh, S.L. Effects of rutin from Toona sinensis on the immune and physiological responses of white shrimp (Litopenaeus vannamei) under Vibrio alginolyticus challenge. Fish Shellfish Immunol. 2008, .25, 581–588. [Google Scholar] [CrossRef]
- Hsieh, T.J.; Liu, T.Z.; Chia, Y.C.; Chern, C.L.; Lu, F.J.; Chuang, M.C.; Mau, S.Y.; Chen, S.H.; Syu, Y.H.; Chen, C.H. Protective effect of methyl gallate from Toona sinensis (Meliaceae) against hydrogen peroxide-induced oxidative stress and DNA damage in MDCK cells. Food Chem. Toxicol. 2004, 42, 843–850. [Google Scholar] [CrossRef] [PubMed]
- You, H.L.; Chen, C.J.; Eng, H.L.; Liao, P.L.; Huang, S.T. The effectiveness and mechanism of Toona sinensis extract inhibit attachment of pandemic influenza A (H1N1) virus. Evid Based Complement Alternat. Med. 2013. [Google Scholar] [CrossRef] [PubMed]
- Gomes, C.A.; da Cruz, T.G.; Andrade, J.L.; Milhazes, N.; Borges, F.; Marques, M.P. Anticancer activity of phenolic acids of natural or synthetic origin: A structure-activity study. J. Med. Chem. 2003, 46, 5395–5401. [Google Scholar] [CrossRef] [PubMed]
- Serafini, M.; Peluso, I.; Raguzzini, A. Flavonoids as anti-inflammatory agents. Proc. Nutr. Soc. 2010, 69, 273–278. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.W.; Curtis-Long, M.J.; Yuk, H.J.; Wang, Y.; Song, Y.H.; Jeong, S.H.; Park, K.H. Quantitative analysis of phenolic metabolites from different parts of Angelica keiskei by HPLC-ESI MS/MS and their xanthine oxidase inhibition. Food Chem. 2014, 153, 20–27. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.H.; Yang, S.J.; Liang, N.; Hu, D.Y.; Jin, L.H.; Xue, W.; Yang, S. Chemical constituents of Caesalpinia decapetala (Roth) Alston. Molecules 2013, 18, 1325–1336. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Mou, Y.; Zhao, J.; Wang, J.; Zhou, L.; Wang, M.; Wang, D.; Han, J.; Yu, Z.; Yang, F. Flavonoids from Halostachys caspica and their antimicrobial and antioxidant activities. Molecules 2010, 15, 7933–7945. [Google Scholar] [CrossRef]
- Beretta, G.; Artali, R.; Caneva, E.; Maffei Facino, R. Conformation of the tridimensional structure of 1,2,3,4,6-pentagalloyl-β-D-glucopyranose (PGG) by 1H NMR, NOESY and theoretical study and membrane interaction in a simulated phospholipid bilayer: a first insight. Magn. Reson. Chem. 2011, 49, 132–136. [Google Scholar] [CrossRef]
- Saldanha, L.L.; Vilegas, W.; Dokkedal, A.L. Characterization of flavonoids and phenolic acids in Myrcia bella Cambess. using FIA-ESI-IT-MS(n) and HPLC-PAD-ESI-IT-MS combined with NMR. Molecules 2013, 18, 8402–8416. [Google Scholar] [CrossRef]
- Lee, S.Y.; So, Y.J.; Shin, M.S.; Cho, J.Y.; Lee, J. Antibacterial effects of afzelin isolated from Cornus macrophylla on Pseudomonas aeruginosa, a leading cause of illness in immunocompromised individuals. Molecules 2014, 19, 3173–3180. [Google Scholar] [CrossRef]
- Serna, D.M.; Martínez, J.H. Phenolics and Polyphenolics from Melastomataceae Species. Molecules 2015, 20, 17818–17847. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Liu, X.; Yang, T.; Slovin, J.; Chen, P. Profiling polyphenols of two diploid strawberry (Fragaria vesca) inbred lines using UHPLC-HRMSn. Food Chem. 2014, 146, 289–298. [Google Scholar] [CrossRef] [PubMed]
- Cos, P.; Ying, L.; Calomme, M.; Hu, J.P.; Cimanga, K.; Van Poel, B.; Pieters, L.; Vlietinck, A.J.; Vanden Berghe, D. Structure-activity relationship and classification of flavonoids as inhibitors of xanthine oxidase and superoxide scavengers. J. Nat. Prod. 1998, 61, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Wang, W.; Guo, B.H.; Gao, H.; Liu, Y.; Liu, X.H.; Yao, H.L.; Cheng, K. Chemical evidence for potent xanthine oxidase inhibitory activity of ethyl acetate extract of Citrus aurantium L. dried immature fruits. Molecules 2016, 21, 302. [Google Scholar] [CrossRef] [PubMed]
- Tamta, H.; Kalra, S.; Mukhopadhyay, A.K. Biochemical characterization of some pyrazolopyrimidine-based inhibitors of xanthine oxidase. Biochemistry (Mosc.) 2006, 71, S49–S54. [Google Scholar] [CrossRef] [PubMed]
- Borges, F.; Fernandes, E.; Roleira, F. Progress towards the discovery of xanthine oxidase inhibitors. Curr. Med. Chem. 2002, 9, 195–217. [Google Scholar] [CrossRef]
- Mo, S.F.; Zhou, F.; Lv, Y.Z.; Hu, Q.H.; Zhang, D.M.; Kong, L.D. Hypouricemic action of selected flavonoids in mice: structure-activity relationships. Biol. Pharm. Bull. 2007, 30, 1551–1556. [Google Scholar] [CrossRef]
Sample Availability: Not available. |
| Compound | Xanthine Oxidase | ||
|---|---|---|---|
| IC50 a | Inhibition % b | Kinetic Mode (Ki c, µM) | |
| EtOAc | >500 ppm | 8.4 ± 0.8 | NT d |
| 70% Ethanol | 78.4 ± 2.4 ppm | 74.2 ± 1.2 | NT |
| H2O | 284.7 ± 5.1 ppm | 53.7 ± 1.6 | NT |
| 1 | >100 µM | 42.9 ± 1.4 | NT |
| 2 | >100 µM | 47.8 ± 1.1 | NT |
| 3 | 2.79 ± 0.2 µM | 96.4 ± 0.7 | Noncompetitive (3.1) |
| 4 | >100 µM | 38.9 ± 1.9 | NT |
| 5 | >100 µM | 31.7 ± 1.3 | NT |
| Allopurinol | 2.28 ± 0.3 µM | >98 | Competitive |
| Oxypurinol | 28.4 ± 0.5 µM | 87.6 ± 0.9 | NT |
| Peak | tR | λmax | Dried Leaves (mg/g) a | [M − H]− (m/z) | Molecular Formula | Identification b | ||
|---|---|---|---|---|---|---|---|---|
| (min) | (nm) | EtOAc | 70% EtOH | H2O | (ESI-HRMS) | (ppm Error) | ||
| 1 | 5.90 | 254, 352 | tr | 0.69 | 0.71 | 609.1448 | C27H29O16 (−1.3) | Quer-3-O-Rut |
| 2 | 6.23 | 255, 353 | tr | 1.32 | 0.40 | 463.0893 | C21H19O12 (3.5) | Quer -3-O-β-d-Glc |
| 3 | 7.37 | 279 | tr | 1.34 | 0.25 | 939.1119 | C41H31O26 (1.6) | PGG |
| 4 | 7.79 | 255, 348 | tr | 7.12 | 2.05 | 447.0939 | C21H19O11 (2.7) | Quer-3-O-α-l-Rham |
| 5 | 9.56 | 263, 344 | tr | 1.46 | 0.41 | 431.0974 | C21H19O10 (−0.9) | Kaem-3-O-α-l-Rham |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yuk, H.J.; Lee, Y.-S.; Ryu, H.W.; Kim, S.-H.; Kim, D.-S. Effects of Toona sinensis Leaf Extract and Its Chemical Constituents on Xanthine Oxidase Activity and Serum Uric Acid Levels in Potassium Oxonate-Induced Hyperuricemic Rats. Molecules 2018, 23, 3254. https://doi.org/10.3390/molecules23123254
Yuk HJ, Lee Y-S, Ryu HW, Kim S-H, Kim D-S. Effects of Toona sinensis Leaf Extract and Its Chemical Constituents on Xanthine Oxidase Activity and Serum Uric Acid Levels in Potassium Oxonate-Induced Hyperuricemic Rats. Molecules. 2018; 23(12):3254. https://doi.org/10.3390/molecules23123254
Chicago/Turabian StyleYuk, Heung Joo, Young-Sil Lee, Hyung Won Ryu, Seung-Hyung Kim, and Dong-Seon Kim. 2018. "Effects of Toona sinensis Leaf Extract and Its Chemical Constituents on Xanthine Oxidase Activity and Serum Uric Acid Levels in Potassium Oxonate-Induced Hyperuricemic Rats" Molecules 23, no. 12: 3254. https://doi.org/10.3390/molecules23123254
APA StyleYuk, H. J., Lee, Y.-S., Ryu, H. W., Kim, S.-H., & Kim, D.-S. (2018). Effects of Toona sinensis Leaf Extract and Its Chemical Constituents on Xanthine Oxidase Activity and Serum Uric Acid Levels in Potassium Oxonate-Induced Hyperuricemic Rats. Molecules, 23(12), 3254. https://doi.org/10.3390/molecules23123254






