Deep Eutectic Solvent-Based Ultrahigh Pressure Extraction of Baicalin from Scutellaria baicalensis Georgi
Abstract
:1. Introduction
2. Results and Discussion
2.1. Comparison of the Solubility
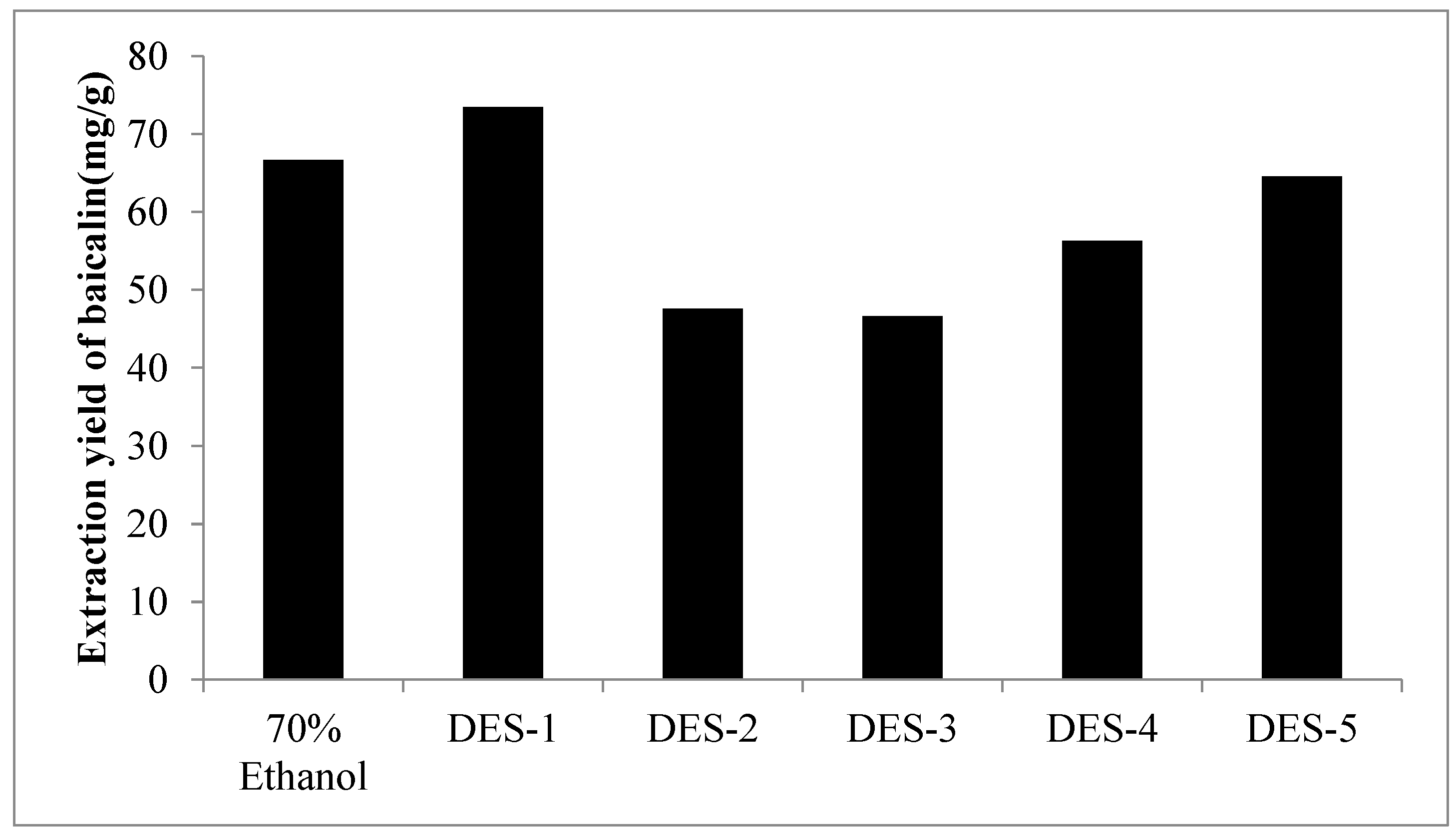
2.2. Comparison of the Extractability of DESs
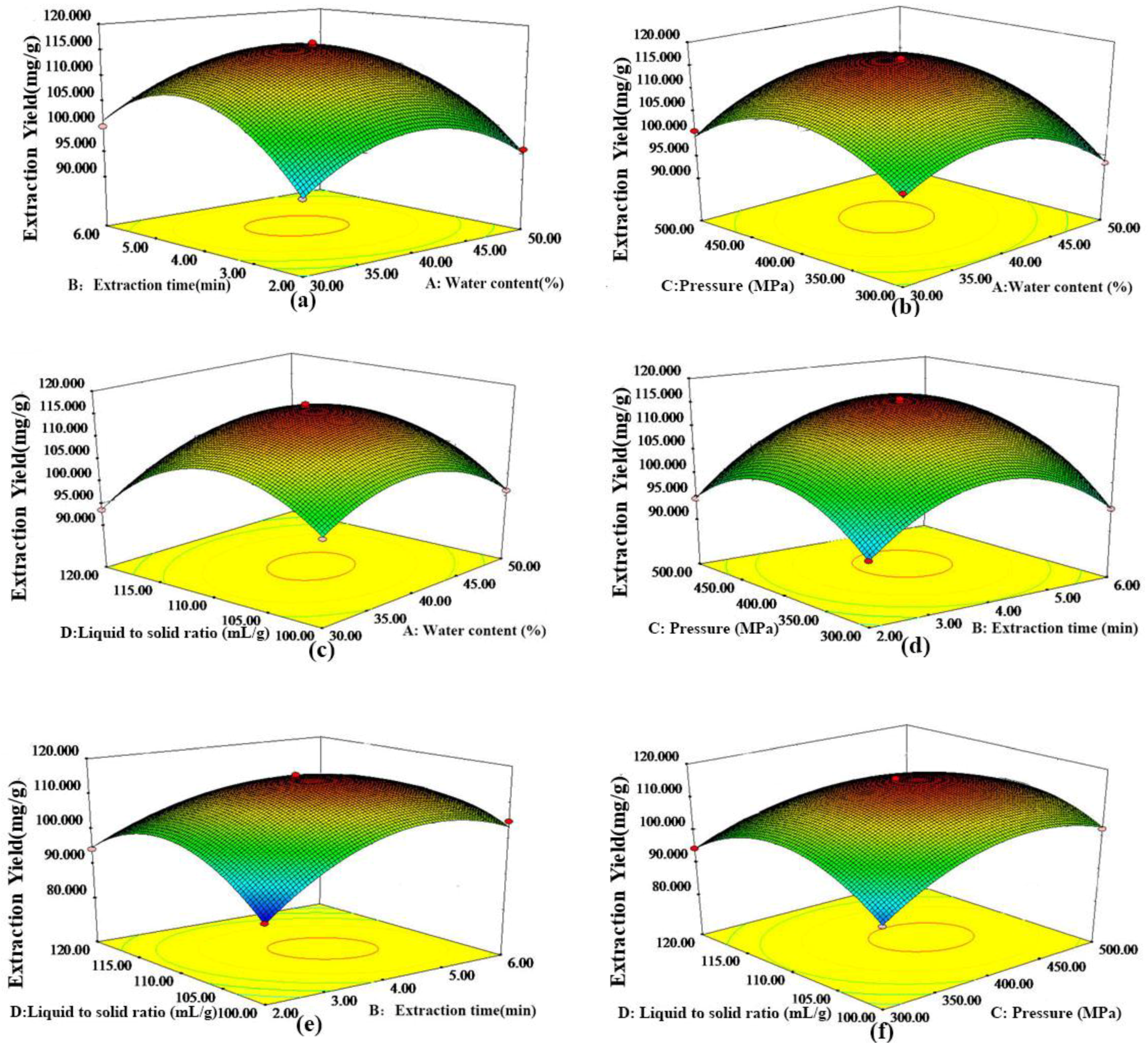
2.3. Optimization of the Extraction Conditions by RSM
2.4. Microstructure Alteration of Different Extraction Procedures
3. Materials and Methods
3.1. Materials and Reagents
3.2. HPLC Analysis
3.3. Preparation of DESs
3.4. Sample Digestion and Analysis
3.5. Extraction with Different Solvents
3.6. Comparison Experiments
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zeng, L.; Luo, L.Y.; Guan, X.L. Process Optimization for Ultra-High-Pressure Extraction of Tea Polyphenols. Food Sci. 2011, 32, 85–88. [Google Scholar]
- Qin, X.; Jin, X.Y.; Liu, H.J. Study on Optimum Process for Ultra High-Pressure Extraction Caffetannic Acid from Chrysanthemum morifolium Ramat. Hubei Agric. Sci. 2010, 49, 2215–2217. [Google Scholar]
- Xi, J.; Zhang, S.; Li, B.; Zhang, R.; Li, Y.; Shen, D.; Zhang, G. Separation of major catechins from green tea by ultrahigh pressure extraction. Int. J. Pharm. 2010, 386, 229–231. [Google Scholar]
- Li, B.; Jia, S.R.; Zhong, C.; Liu, J. Extraction of Stevioside from Stevia by Ultrahigh Pressure Technique. Mod. Food Sci. Technol. 2010, 261, 117–1120. [Google Scholar]
- Zong, W.; Li, X. Study of extract flavonoids from Lonicera japonica Thunb by ultrahigh pressure method. Food Res. Dev. 2009, 30, 65–68. [Google Scholar]
- Ma, T.; Qin, X.; Jia, C. Extraction of soybean lecithin using ultrahigh pressure extraction technology. Chin. Agric. Sci. Bull. 2010, 26, 46–51. [Google Scholar]
- Liu, F.; Wang, D.; Liu, W.; Wang, X.; Bai, A.; Huang, L. Ionic liquid-based ultrahigh pressure extraction of five tanshinones from salvia miltiorrhiza bunge. Sep. Purif. Technol. 2013, 110, 86–92. [Google Scholar] [CrossRef]
- Sun, X.J.; Li, X.X.; Feng, Y.B.; Li, J.R.; Li, Y.C.; Lv, Y.F.; Liu, X.F. Optimization of Extracting Technique Assisted by Ultrahigh Pressure Extraction of β-carotene from Dunaliella salina. J. Chin. Inst. Food Sci. Technol. 2016, 16, 88–94. [Google Scholar]
- Qin, X.; Ren, X.Y.; Li, H. Room temperature ultrahigh pressure extraction in big beautiful crape myrtle leaf 2-alphahydroxyursolic acid. Adv. Eng. Res. 2016, 117, 1–5. [Google Scholar]
- Zhang, Q.; Vigier, K.D.O.; Royer, S.; Jérôme, F. Deep eutectic solvents: syntheses, properties and applications. Chem. Soc. Rev. 2012, 41, 7108–7146. [Google Scholar] [CrossRef]
- Abbott, A.P.; Capper, G.; Davies, D.L.; Rasheed, R.K.; Tambyrajah, V. Novel solvent properties of choline chloride/urea mixtures. Chem. Commun. 2002, 9, 70–71. [Google Scholar]
- Hayyan, M.; Hashim, M.A.; Al-Saadi, M.A.; Hayyan, A.; AlNashef, I.M.; Mirghani, M.E. Assessment of cytotoxicity and toxicity for phosphonium-based deep eutectic solvents. Chemosphere. 2013, 93, 455–459. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Deng, D.; Chen, Y.; Shan, H.; Ai, N. Solubilities and thermodynamic properties of CO2 in choline-chloride based deep eutectic solvents. J. Chem. Thermodyn. 2014, 75, 58–62. [Google Scholar] [CrossRef]
- Radošević, K.; Bubalo, M.C.; Srček, V.G.; Grgas, D.; Dragičević, T.L.; Redovniković, I.R. Evaluation of toxicity and biodegradability of choline chloride based deep eutectic solvents. Ecotoxicol. Environ. Saf. 2015, 112, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.; Rozema, E.; Verpoorte, R.; Choi, Y.H. Application of natural deep eutectic solvents to the extraction of anthocyanins from catharanthus roseus with high extractability and stability replacing conventional organic solvents. J. Chromatogr. A 2016, 1434, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Liu, M.; Wang, Q.; Du, H.; Zhang, L. Deep eutectic solvent-based microwave- assisted method for extraction of hydrophilic and hydrophobic components from radix salvia miltiorrhizae. Molecules 2016, 21, 1383. [Google Scholar] [CrossRef]
- Qi, X.L.; Peng, X.; Huang, Y.Y.; Li, L.; Wei, Z.F.; Zu, Y.G.; Fu, Y.J. Green and efficient extraction of bioactive flavonoids from equisetum palustre, L. by deep eutectic solvents-based negative pressure cavitation method combined with macroporous resin enrichment. Ind. Crop. Prod. 2015, 70, 142–148. [Google Scholar] [CrossRef]
- Bi, W.; Tian, M.; Row, K.H. Evaluation of alcohol-based deep eutectic solvent in extraction and determination of flavonoids with response surface methodology optimization. J. Chromatogr. A 2013, 1285, 22–30. [Google Scholar] [CrossRef]
- Cao, J.; Yang, M.; Cao, F.L.; Wang, J.; Su, E.Z. Well-designed hydrophobic deep eutectic solvents as green and efficient media for the extraction of artemisinin from artemisia annua leaves. ACS Sustain. Chem. Eng. 2017, 5, 3270–3278. [Google Scholar] [CrossRef]
- Zhang, L.J.; Wang, M.S. Optimization of deep eutectic solvent-based ultrasound-assisted extraction of polysaccharides from Dioscorea opposita Thunb. Int. J. Biol. Macromol. 2017, 95, 675–681. [Google Scholar] [CrossRef]
- Cao, J.; Chen, L.Y.; Li, M.H.; Cao, F.L.; Zhao, L.G.; Su, E.Z. Efficient extraction of proanthocyanidin from Ginkgo biloba leaves employing rationally designed deep eutectic solvent-water mixture and evaluation of the antioxidant activity. J. Pharmaceut. Biomed. 2018, 158, 317–326. [Google Scholar] [CrossRef]
- Li, H.B.; Jiang, Y.; Chen, F. Separation methods used for scutellaria baicalensis active components. J. Chromatogr. B 2014, 812, 277–290. [Google Scholar] [CrossRef]
- Guo, Q.; Zhao, L.; You, Q.; Yang, Y.; Gu, H.; Song, G.; Lu, N.; Xin, J. Anti-hepatitis B virus activity of wogonin in vitro and in vivo. Antiviral. Res. 2007, 74, 16–24. [Google Scholar] [CrossRef]
- Kowalczyk, E.; Krzesiński, P.; Kura, M.; Niedworok, J.; Kowalski, J.; Błaszczyk, J. Pharmacological effects of flavonoids from scutellaria baicalensis. Przegl. Lek. 2006, 63, 95–106. [Google Scholar] [PubMed]
- Li, C.; Lin, G.; Zuo, Z. Pharmacological effects and pharmacokinetics properties of radix scutellariae and its bioactive flavones. Biopharm. Drug Dispos. 2011, 32, 427–445. [Google Scholar] [CrossRef] [PubMed]
- Liao, J.F.; Hung, W.Y.; Chen, C.F. Anxiolytic-like effects of baicalein and baicalin in the vogel conflict test in mice. Eur. J. Pharmacol. 2003, 464, 141–146. [Google Scholar] [CrossRef]
- Cheng, C.G.; Jin, W.Y.; Wu, L. Study on microwave-assisted extraction of baicalin from scutellaria baicalensis. Chem. Ind. Forest Prod. 2005, 25, 81–83. [Google Scholar]
- Rostagno, M.A.; Palma, M.; Barroso, C.G. Ultrasound-assisted extraction of soy isoflavone. Anal. Chim. Acta. 2007, 588, 274–282. [Google Scholar] [CrossRef]
- Hao, J.Y.; Han, W.; Huang, S.D.; Xue, B.Y.; Deng, X. Microwave-assisted extraction of artemisinin from Artemisia annua L. Chin. J. Pharm. 2002, 28, 191–196. [Google Scholar] [CrossRef]
- Zhou, H.Y.; Liu, C.Z. Microwave-assisted extraction of solanesol from tobacco leaves. J. Chromatogr. A 2006, 1129, 135–139. [Google Scholar] [CrossRef]
- Li, T.T. Deep eutectic solvents-based Microwave-assisted Extraction the main flavonoids from Scutellaria baicalensis Georgi; Northeast Forestry University: Harbin, China, 2014. [Google Scholar]
- Wang, H.; Ma, X.; Cheng, Q.; Xi, X.; Zhang, L. Deep Eutectic Solvent-based Microwave-Assisted Extraction of Baicalin from Scutellaria Baicalensis Georgi. J. Chem. 2018, 9579872, 1–10. [Google Scholar] [CrossRef]
- Dai, Y.; Spronsen, J.V.; Witkamp, G.J.; Verpoorte, R.; Choi, Y.H. Natural deep eutectic solvents as new potential media for green technology. Anal. Chim. Acta 2013, 766, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Tang, B.K.; Zhang, H.; Row, K.H. Application of Deep Eutectic Solvents in the Extraction and Separation of Target Compounds from Various Samples. J. Sep. Sci. 2015, 38, 1053–1064. [Google Scholar] [CrossRef] [PubMed]
- Jeong, K.M.; Lee, M.S.; Nam, M.W.; Zhao, J.; Jin, Y.; Lee, D.K.; Kwon, S.W.; Jeong, J.H.; Lee, J. Tailoring and Recycling of Deep Eutectic Solvents as Sustainable and Efficient Extraction Media. J. Chromatogr. A 2015, 1424, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Nam, M.W.; Zhao, J.; Lee, M.S.; Jeong, J.H.; Lee, J. Enhanced Extraction of Bioactive Natural Products using Tailor-made Deep Eutectic Solvents. Green Chem. 2015, 17, 1718–1727. [Google Scholar] [CrossRef]
- Liu, W.; Zhang, K.D.; Yu, J.J.; Bi, Y.L. A Green Ultrasonic-Assisted Liquid-Liquid Microextraction Based on Deep Eutectic Solvent for the HPLC-UV Determination of TBHQ in Edible Oils. Food Anal. Methods 2017, 10, 3209–3215. [Google Scholar] [CrossRef]
- Lu, C.; Cao, J.; Wang, N.; Su, E.Z. Significantly Improving the Solubility of Nonsteroidal Anti-inflammatory Drugs in Deep Eutectic Solvents for Potential Non-aqueous Liquid Administration. Med. Chem. Commun. 2016, 7, 955–959. [Google Scholar] [CrossRef]
- Jie, X.; Chen, X.; Zhong, D. Stability of baicalin in biological fluids in vitro. J. Pharmaceut. Biomed. 2015, 39, 593–600. [Google Scholar]
- Lu, Y.; Ma, W.; Hu, R.; Dai, X.; Pan, Y. Ionic liquid-based microwave-assisted extraction of phenolic alkaloids from the medicinal plant nelumbo nucifera gaertn. J. Chromatogr. A 2008, 1208, 42–46. [Google Scholar] [CrossRef]
- Yao, Y.H.; Zhang, L.W. Study on Stability of Baicalein. Chin. J. Spectrosc. 2006, 23, 346–348. [Google Scholar]
- Yu, B.; Zhang, Z.; Liu, W.; Wang, P.; Yang, T. Studies on stability of baicalin. Chin. Tradit. Herbal Drugs 2002, 23, 218–220. [Google Scholar]
- Butz, P.; Koller, W.D.; Tauscher, B.; Wolf, S. Ultra-high-pressure processing of onions: chemical and sensory changes. LWT-Food Sci. Technol. 1994, 27, 463–467. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhao, S.H.; Chen, J.; Zhang, L.W. Application of ionic liquid-based microwave-assisted extraction of flavonoids from Scutellaria baicalensis Georgi. J. Chromatogr. B 2015, 1002, 411–417. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds DES-1, DES-2, DES-3, DES-4 and DES-5 are available from the authors. |

| No. | Type of HBD | Abbreviation | ChCl/HBD Ratio |
|---|---|---|---|
| DES-1 | Lactic acid | LA | 1:1 |
| DES-2 | Glucose | GLU | 1:1 |
| DES-3 | Glycerol | GL | 1:1 |
| DES-4 | 1,4-Butanediol | BDO | 1:1 |
| DES-5 | Ethylene glycol | EG | 1:1 |
| Number | Solvents | Solubility of Baicalin (mg/g) |
|---|---|---|
| 1 | Water | 0.18 |
| 2 | 70% Ethanol | 1.85 |
| 3 | DES-1 | 22.7 |
| 4 | DES-2 | 18.2 |
| 5 | DES-3 | 12.35 |
| 6 | DES-4 | 2.25 |
| 7 | DES-5 | 17.8 |
| Factor | Actual and Coded Levels Used for the Conditions | ||
|---|---|---|---|
| Low (−1) | Medium (0) | High (+1) | |
| A = Water content (%) | 30 | 40 | 50 |
| B = Time (min) | 2 | 4 | 6 |
| C = Pressure | 300 | 400 | 500 |
| D = Liquid to solid ratio | 100 | 110 | 120 |
| Dependent variable | Constrains | ||
| R1 = Baicalin/(mg/g) | Maximize | ||
| Run | Factor A: Water Content % | Factor B: Time min | Factor C: Pressure MPa | Factor D: Liquid-Solid Ratio (mL/g) | Baicalin (mg/g) |
|---|---|---|---|---|---|
| 1 | 40 | 2 | 400 | 100 | 88.08 |
| 2 | 40 | 2 | 300 | 110 | 93.08 |
| 3 | 50 | 6 | 400 | 110 | 98.03 |
| 4 | 40 | 6 | 300 | 110 | 94.79 |
| 5 | 40 | 2 | 500 | 110 | 94.52 |
| 6 | 40 | 4 | 300 | 120 | 94.58 |
| 7 | 30 | 4 | 300 | 110 | 99.60 |
| 8 | 40 | 4 | 400 | 110 | 116.560 |
| 9 | 50 | 4 | 400 | 100 | 96.35 |
| 10 | 50 | 2 | 400 | 110 | 95.989 |
| 11 | 40 | 4 | 500 | 120 | 93.04 |
| 12 | 30 | 4 | 400 | 120 | 93.56 |
| 13 | 40 | 4 | 400 | 110 | 116.05 |
| 14 | 30 | 2 | 400 | 110 | 93.84 |
| 15 | 30 | 4 | 400 | 100 | 99.05 |
| 16 | 40 | 4 | 400 | 110 | 115.88 |
| 17 | 50 | 4 | 500 | 110 | 103.09 |
| 18 | 40 | 4 | 400 | 110 | 115.98 |
| 19 | 40 | 6 | 400 | 120 | 90.22 |
| 20 | 30 | 6 | 400 | 110 | 100.06 |
| 21 | 40 | 6 | 500 | 110 | 102.91 |
| 22 | 50 | 4 | 300 | 110 | 93.43 |
| 23 | 50 | 4 | 400 | 120 | 95.26 |
| 24 | 40 | 4 | 300 | 100 | 91.18 |
| 25 | 40 | 6 | 400 | 100 | 104.95 |
| 26 | 30 | 4 | 500 | 110 | 100.78 |
| 27 | 40 | 4 | 400 | 110 | 115.26 |
| 28 | 40 | 4 | 500 | 100 | 102.51 |
| 29 | 40 | 2 | 400 | 120 | 94.31 |
| Source | Sum of Squares | df | Mean Square | F Value | p-Value Prob > F | |
|---|---|---|---|---|---|---|
| Model | 2022.273 | 14 | 144.4481 | 229.5803 | <0.0001 | significant |
| A-Water content | 1.872498 | 1 | 1.872498 | 2.976078 | 0.1065 | |
| B-Time | 80.8396 | 1 | 80.8396 | 128.4834 | <0.0001 | |
| C-Pressure | 75.90223 | 1 | 75.90223 | 120.6362 | <0.0001 | |
| D-Liquid-solid ratio | 37.33898 | 1 | 37.33898 | 59.34518 | < 0.0001 | |
| AB | 4.378975 | 1 | 4.378975 | 6.959778 | 0.0195 | |
| AC | 17.9536 | 1 | 17.9536 | 28.53477 | 0.0001 | |
| AD | 4.840466 | 1 | 4.840466 | 7.693255 | 0.0149 | |
| BC | 11.18712 | 1 | 11.18712 | 17.78039 | 0.0009 | |
| BD | 109.843 | 1 | 109.843 | 174.5803 | <0.0001 | |
| CD | 41.42853 | 1 | 41.42853 | 65.84495 | <0.0001 | |
| A2 | 439.4122 | 1 | 439.4122 | 698.3853 | <0.0001 | |
| B2 | 716.8069 | 1 | 716.8069 | 1139.266 | <0.0001 | |
| C2 | 516.0715 | 1 | 516.0715 | 820.2247 | <0.0001 | |
| D2 | 853.7272 | 1 | 853.7272 | 1356.882 | <0.0001 | |
| Residual | 8.808564 | 14 | 0.629183 | |||
| Lack of Fit | 7.89223 | 10 | 0.789223 | 3.445136 | 0.1223 | not significant |
| Pure Error | 0.916333 | 4 | 0.229083 | |||
| Cor Total | 2031.081 | 28 | ||||
| R-Squared | 0.995663 | |||||
| Adj R-Squared | 0.991326 | |||||
| Pred R-Squared | 0.976913 | |||||
| Extract Method | Solvent | Baicalin (mg/g) |
|---|---|---|
| UPE | 70% Ethyl alcohol | 108.6 |
| Moisture content 40% of ChCl-LA (1:1) | 116.8 | |
| HRAE | 70% Ethyl alcohol | 110.4 |
| Moisture content 40% of ChCl-LA (1:1) | 84.3 | |
| MAE | 70% Ethyl alcohol | 89.3 |
| Moisture content 40% of ChCl-LA (1:1) | 101.5 | |
| MAE | Moisture content 20% of ChCl-LA (1:2) | 33.1 [31] |
| MAE | Moisture content 33% of DecA-N4444-Cl (1:2) | 106.96 [32] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, H.; Ma, X.; Cheng, Q.; Wang, L.; Zhang, L. Deep Eutectic Solvent-Based Ultrahigh Pressure Extraction of Baicalin from Scutellaria baicalensis Georgi. Molecules 2018, 23, 3233. https://doi.org/10.3390/molecules23123233
Wang H, Ma X, Cheng Q, Wang L, Zhang L. Deep Eutectic Solvent-Based Ultrahigh Pressure Extraction of Baicalin from Scutellaria baicalensis Georgi. Molecules. 2018; 23(12):3233. https://doi.org/10.3390/molecules23123233
Chicago/Turabian StyleWang, Hui, Xiaodi Ma, Qibin Cheng, Li Wang, and Liwei Zhang. 2018. "Deep Eutectic Solvent-Based Ultrahigh Pressure Extraction of Baicalin from Scutellaria baicalensis Georgi" Molecules 23, no. 12: 3233. https://doi.org/10.3390/molecules23123233
APA StyleWang, H., Ma, X., Cheng, Q., Wang, L., & Zhang, L. (2018). Deep Eutectic Solvent-Based Ultrahigh Pressure Extraction of Baicalin from Scutellaria baicalensis Georgi. Molecules, 23(12), 3233. https://doi.org/10.3390/molecules23123233