The Various Roles of Fatty Acids
Abstract
1. Fatty Acid Synthesis
2. Fatty Acids as Modulators of Membrane Properties
2.1. In Prokaryotes
2.2. In Eukaryotes
3. Energy Suppliers and Storage Material
3.1. In Prokaryotes
3.2. In Eukaryotes
3.3. Sequestration of Toxicants in Lipid Droplets
4. The Roles of PUFA
5. Biomarkers of Organisms
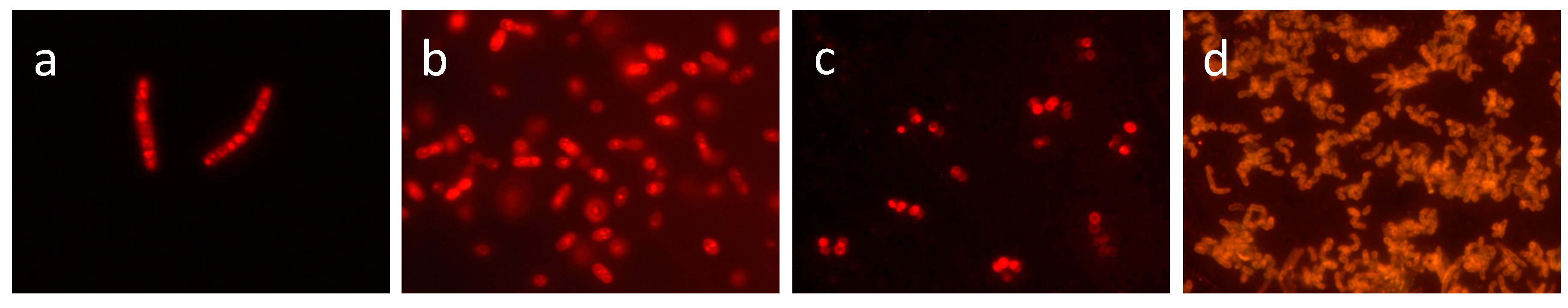
5.1. Identification of Prokaryotes by FA Fingerprint
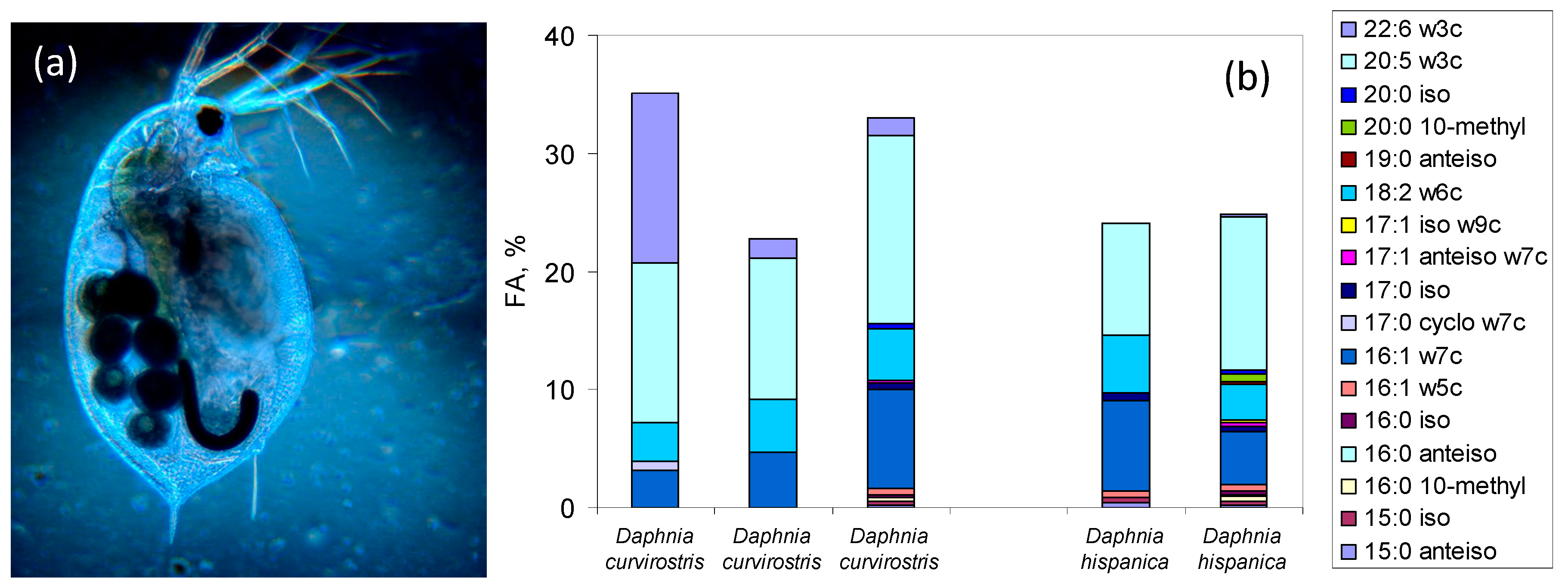
5.2. FA Markers in Eukaryotes and the Use of FA Profiles to Assess Species Composition of Assemblages
5.3. Using FA to Follow Energy Fluxes in Food Webs
6. Conclusions
Funding
Conflicts of Interest
References
- Berg, J.M.; Tymoczko, J.L.; Stryer, L. Section 22.5–Acetyl Coenzyme A carboxylase plays a key role in controlling fatty acid metabolism. In Biochemistry, 5th ed.; Berg, J.M., Tymoczko, J.L., Stryer, L., Eds.; W. H. Freeman: New York, NY, USA, 2002; ISBN 10: 0716730510. [Google Scholar]
- Leibundgut, M.; Maier, T.; Jenni, S.; Ban, N. The multienzyme architecture of eukaryotic fatty acid synthases. Curr. Opin. Struct. Biol. 2008, 18, 714–725. [Google Scholar] [CrossRef] [PubMed]
- Schweizer, E.; Hofmann, J. Microbial type I fatty acid synthases (FAS): Major players in a network of cellular FAS systems. Microbiol. Mol. Biol. Rev. 2004, 68, 501–517. [Google Scholar] [CrossRef] [PubMed]
- White, S.W.; Zheng, J.; Zhang, Y.M.; Rock, C.O. The structural biology of type II fatty acid biosynthesis. Annu. Rev. Biochem. 2005, 74, 791–831. [Google Scholar] [CrossRef] [PubMed]
- Harwood, J.L. Fatty acid metabolism. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1988, 39, 101–138. [Google Scholar] [CrossRef]
- Harwood, J.L. Recent advances in the biosynthesis of plant fatty acids. Biochim. Biophys. Acta (BBA) Lipids Lipid Metab. 1996, 1301, 7–56. [Google Scholar] [CrossRef]
- Bloch, K.; Vance, D. Control mechanisms in the synthesis of saturated fatty acids. Annu. Rev. Biochem. 1977, 46, 263–298. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi, S.; Rainwater, D.L.; Kolattukudy, P.E. Purification and characterization of an unusually large fatty acid synthase from Mycobacterium tuberculosis var. bovis BCG. Arch. Biochem. Biophys. 1992, 295, 318–326. [Google Scholar] [CrossRef]
- Kolattukudy, P.E.; Fernandes, N.D.; Azad, A.K.; Fitzmaurice, A.M.; Sirakova, T.D. Biochemistry and molecular genetics of cell-wall lipid biosynthesis in mycobacteria. Mol. Microbiol. 2003, 24, 263–270. [Google Scholar] [CrossRef]
- Boucher, Y. Lipids: Biosynthesis, function and evolution. In Archaea; Cavicchioli, R., Ed.; ASM Press: Washington, DC, USA, 2007; pp. 341–353. [Google Scholar]
- Jain, S.; Caforio, A.; Driessen, A.J.M. Biosynthesis of archaeal membrane ether lipids. Front. Microbiol. 2014, 5, 641. [Google Scholar] [CrossRef] [PubMed]
- Gattinger, A.; Schloter, M.; Munch, J.C. Phospholipid etherlipid and phospholipid fatty acid fingerprints in selected euryarchaeotal monocultures for taxonomic profiling. FEMS Microbiol. Lett. 2002, 213, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Pugh, E.L.; Wassef, M.K.; Kates, M. Inhibition of fatty acid synthetase in Halobacterium cutirubrum and Escherichia coli by high salt concentrations. Can. J. Biochem. 1971, 49, 953–958. [Google Scholar] [CrossRef] [PubMed]
- Pugh, E.L.; Kates, M. Acylation of proteins of the archaebacteria Halobacterium cutirubrum and Methanobacterium thermoautotrophicum. Biochim. Biophys. Acta (BBA) Biomembr. 1994, 1196, 38–44. [Google Scholar] [CrossRef]
- Lombard, J.; López-García, P.; Moreira, D. An ACP-independent fatty acid synthesis pathway in Archaea: Implications for the origin of phospholipids. Mol. Biol. Evol. 2012, 29, 3261–3265. [Google Scholar] [CrossRef] [PubMed]
- Pereira, S.L.; Leonard, A.E.; Mukerji, P. Recent advances in the study of fatty acid desaturases from animals and lower eukaryotes. Prostaglandins Leukot. Essent. Fat. Acids 2003, 68, 97–106. [Google Scholar] [CrossRef]
- Leonard, A.E.; Pereira, S.L.; Sprecher, H.; Huang, Y.S. Elongation of long-chain fatty acids. Prog. Lipid Res. 2004, 43, 36–54. [Google Scholar] [CrossRef]
- Cook, H.W. Chapter 5 Fatty acid desaturation and chain elongation in eukaryotes. In New Comprehensive Biochemistry; Vance, D.E., Vance, J.E., Eds.; Elsevier: Amsterdam, The Netherlands, 1996; Volume 31, pp. 129–152. [Google Scholar]
- Sperling, P.; Ternes, P.; Zank, T.K.; Heinz, E. The evolution of desaturases. Prostaglandins Leukot. Essent. Fat. Acids 2003, 68, 73–95. [Google Scholar] [CrossRef]
- Hashimoto, K.; Yoshizawa, A.C.; Saito, K.; Yamada, T.; Kanehisa, M. The repertoire of desaturases for unsaturated fatty acid synthesis in 397 genomes. Genome Inform. 2006, 17, 173–183. [Google Scholar] [CrossRef] [PubMed]
- Harwood, J.L.; Guschina, I.A. The versatility of algae and their lipid metabolism. Biochimie 2009, 91, 679–684. [Google Scholar] [CrossRef] [PubMed]
- Monroig, Ó.; Tocher, D.R.; Navarro, J.C. Biosynthesis of polyunsaturated fatty acids in marine invertebrates: Recent advances in molecular mechanisms. Mar. Drugs 2013, 11, 3998–4018. [Google Scholar] [CrossRef] [PubMed]
- Kabeya, N.; Fonseca, M.M.; Ferrier, D.E.K.; Navarro, J.C.; Bay, L.K.; Francis, D.S.; Tocher, D.R.; Castro, L.F.C.; Monroig, Ó. Genes for de novo biosynthesis of omega-3 polyunsaturated fatty acids are widespread in animals. Sci. Adv. 2018, 4, eaar6849. [Google Scholar] [CrossRef] [PubMed]
- Cook, H.W.; McMaster, C.R. Chapter 7 Fatty acid desaturation and chain elongation in eukaryotes. In New Comprehensive Biochemistry; Elsevier: Amsterdam, The Netherlands, 2002; Volume 36, pp. 181–204. [Google Scholar]
- Tocher, D.R. Metabolism and functions of lipids and fatty acids in teleost fish. Rev. Fish. Sci. 2003, 11, 107–184. [Google Scholar] [CrossRef]
- Parrish, C.C. Essential fatty acids in aquatic food webs. In Lipids in Aquatic Ecosystems; Kainz, M., Brett, M.T., Arts, M.T., Eds.; Springer: New York, NY, USA, 2009; pp. 309–326. [Google Scholar]
- Castro, L.F.C.; Tocher, D.R.; Monroig, O. Long-chain polyunsaturated fatty acid biosynthesis in chordates: Insights into the evolution of Fads and Elovl gene repertoire. Prog. Lipid Res. 2016, 62, 25–40. [Google Scholar] [CrossRef] [PubMed]
- Bell, M.V.; Tocher, D.R. Biosynthesis of polyunsaturated fatty acids in aquatic ecosystems: General pathways and new directions. In Lipids in Aquatic Ecosystems; Kainz, M., Brett, M.T., Arts, M.T., Eds.; Springer: New York, NY, USA, 2009; pp. 211–236. [Google Scholar]
- Tocher, D.R. Fatty acid requirements in ontogeny of marine and freshwater fish. Aquacult. Res. 2010, 41, 717–732. [Google Scholar] [CrossRef]
- Brenna, J.T. Efficiency of conversion of α-linolenic acid to long chain n-3 fatty acids in man. Curr. Opin. Clin. Nutr. Metab. Care 2002, 5, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Barceló-Coblijn, G.; Murphy, E.J. Alpha-linolenic acid and its conversion to longer chain n−3 fatty acids: Benefits for human health and a role in maintaining tissue n−3 fatty acid levels. Prog. Lipid Res. 2009, 48, 355–374. [Google Scholar] [CrossRef] [PubMed]
- Scott, B.L.; Bazan, N.G. Membrane docosahexaenoate is supplied to the developing brain and retina by the liver. Proc. Natl. Acad. Sci. USA 1989, 86, 2903–2907. [Google Scholar] [CrossRef] [PubMed]
- Brossard, N.; Croset, M.; Normand, S.; Pousin, J.; Lecerf, J.; Laville, M.; Tayot, J.L.; Lagarde, M. Human plasma albumin transports [13C]docosahexaenoic acid in two lipid forms to blood cells. J. Lipid Res. 1997, 38, 1571–1582. [Google Scholar] [PubMed]
- Rapoport, S.I.; Rao, J.S.; Igarashi, M. Brain metabolism of nutritionally essential polyunsaturated fatty acids depends on both the diet and the liver. Prostaglandins Leukot. Essent. Fat. Acids 2007, 77, 251–261. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.X. The importance of omega-6/omega-3 fatty acid ratio in cell function. In Omega-6/Omega-3 essential Fatty Acid Ratio: The Scientific Evidence; Simopoulos, A.P., Ed.; Karger: Basel, Switzerland, 2003; Volume 92, pp. 23–36. [Google Scholar]
- Simopoulos, A.P. The importance of the omega-6/omega-3 fatty acid ratio in cardiovascular disease and other chronic diseases. Exp. Biol. Med. 2008, 233, 674–688. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. An increase in the omega-6/omega-3 fatty acid ratio increases the risk for obesity. Nutrients 2016, 8, 128. [Google Scholar] [CrossRef] [PubMed]
- Weaver, K.L.; Ivester, P.; Seeds, M.; Case, L.D.; Arm, J.P.; Chilton, F.H. Effect of dietary fatty acids on inflammatory gene expression in healthy humans. J. Biol. Chem. 2009, 284, 15400–15407. [Google Scholar] [CrossRef]
- Donahue, S.M.A.; Rifas-Shiman, S.L.; Gold, D.R.; Jouni, Z.E.; Gillman, M.W.; Oken, E. Prenatal fatty acid status and child adiposity at age 3 y: Results from a US pregnancy cohort. Am. J. Clin. Nutri. 2011, 93, 780–788. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Dietetic Products, Nutrition, and Allergies (NDA). Scientific Opinion on Dietary Reference Values for fats, including saturated fatty acids, polyunsaturated fatty acids, monounsaturated fatty acids, trans fatty acids and cholesterol. EFSA J. 2010, 8, 1461. [Google Scholar] [CrossRef]
- Alberts, B.; Johnson, A.; Lewis, J.; Morgan, D.; Raff, M.; Roberts, K.; Walter, P. Molecular Biology of the Cell; Garland Science: New York, NY, USA; Abingdon, UK, 2015; p. 1464. [Google Scholar]
- Hamerly, T.; Tripet, B.; Wurch, L.; Hettich, R.L.; Podar, M.; Bothner, B.; Copié, V. Characterization of fatty acids in Crenarchaeota by GC-MS and NMR. Archaea 2015, 2015, 9. [Google Scholar] [CrossRef] [PubMed]
- Koga, Y. Thermal adaptation of the archaeal and bacterial lipid membranes. Archaea 2012, 2012, 6. [Google Scholar] [CrossRef] [PubMed]
- Weijers, J.W.H.; Schouten, S.; Hopmans, E.C.; Geenevasen, J.A.J.; David, O.R.P.; Coleman, J.M.; Pancost, R.D.; Sinninghe Damsté, J.S. Membrane lipids of mesophilic anaerobic bacteria thriving in peats have typical archaeal traits. Environ. Microbiol. 2006, 8, 648–657. [Google Scholar] [CrossRef] [PubMed]
- de Carvalho, C.C.C.R.; Fernandes, P. Production of metabolites as bacterial responses to the marine environment. Mar. Drugs 2010, 8, 705–727. [Google Scholar] [CrossRef] [PubMed]
- de Carvalho, C.C.C.R.; Caramujo, M.J. Carotenoids in aquatic ecosystems and aquaculture: A colorful business with implications for human health. Front. Mar. Sci. 2017, 4, 93. [Google Scholar] [CrossRef]
- Hazel, J.R.; Eugene Williams, E. The role of alterations in membrane lipid composition in enabling physiological adaptation of organisms to their physical environment. Prog. Lipid Res. 1990, 29, 167–227. [Google Scholar] [CrossRef]
- Konings, W.N.; Albers, S.-V.; Koning, S.; Driessen, A.J.M. The cell membrane plays a crucial role in survival of bacteria and archaea in extreme environments. Antonie Van Leeuwenhoek 2002, 81, 61–72. [Google Scholar] [CrossRef] [PubMed]
- Yazawa, K. Production of eicosapentaenoic acid from marine bacteria. Lipids 1996, 31, S297–S300. [Google Scholar] [CrossRef] [PubMed]
- Okuyama, H.; Orikasa, Y.; Nishida, T.; Watanabe, K.; Morita, N. Bacterial genes responsible for the biosynthesis of eicosapentaenoic and docosahexaenoic acids and their heterologous expression. Appl. Environ. Microbiol. 2007, 73, 665–670. [Google Scholar] [CrossRef] [PubMed]
- Fang, J.; Barcelona, M.J.; Nogi, Y.; Kato, C. Biochemical implications and geochemical significance of novel phospholipids of the extremely barophilic bacteria from the Marianas Trench at 11,000 m. Deep Sea Res. Part I Oceanogr. Res. Pap. 2000, 47, 1173–1182. [Google Scholar] [CrossRef]
- Yano, Y.; Nakayama, A.; Ishihara, K.; Saito, H. Adaptive changes in membrane lipids of barophilic bacteria in response to changes in growth pressure. Appl. Environ. Microbiol. 1998, 64, 479–485. [Google Scholar] [PubMed]
- de Carvalho, C.C.C.R.; da Fonseca, M.M.R. Preventing biofilm formation: Promoting cell separation with terpenes. FEMS Microbiol. Ecol. 2007, 61, 406–413. [Google Scholar] [CrossRef] [PubMed]
- Kaye, J.Z.; Baross, J.A. Synchronous effects of temperature, hydrostatic pressure and salinity on growth, phospholipid profiles and protein patterns of four Halomonas species isolated from deep-sea hydrothermal-vent and sea surface environments. Appl. Environ. Microbiol. 2004, 70, 6220–6229. [Google Scholar] [CrossRef] [PubMed]
- de Carvalho, C.C.C.R.; Wick, L.Y.; Heipieper, H.J. Cell wall adaptations of planktonic and biofilm Rhodococcus erythropolis cells to growth on C5 to C16 n-alkane hydrocarbons. Appl. Microbiol. Biotechnol. 2009, 82, 311–320. [Google Scholar] [CrossRef] [PubMed]
- Wirsen, C.O.; Jannasch, H.W.; Wakeham, S.G.; Canuel, E.A. Membrane lipids of a psychrophilic and barophilic deep-sea bacterium. Curr. Microbiol. 1986, 14, 319–322. [Google Scholar] [CrossRef]
- Nichols, D.S.; Nichols, P.D.; Russell, N.J.; Davies, N.W.; McMeekin, T.A. Polyunsaturated fatty acids in the psychrophilic bacterium Shewanella gelidimarina ACAM 456T: Molecular species analysis of major phospholipids and biosynthesis of eicosapentaenoic acid. Biochim. Biophys. Acta (BBA) Lipids Lipid Metab. 1997, 1347, 164–176. [Google Scholar] [CrossRef]
- Yoshida, K.; Hashimoto, M.; Hori, R.; Adachi, T.; Okuyama, H.; Orikasa, Y.; Nagamine, T.; Shimizu, S.; Ueno, A.; Morita, N. Bacterial long-chain polyunsaturated fatty acids: Their biosynthetic genes, functions and practical use. Mar. Drugs 2016, 14, 94. [Google Scholar] [CrossRef] [PubMed]
- de Carvalho, C.C.C.R.; Marques, M.P.C.; Hachicho, N.; Heipieper, H.J. Rapid adaptation of Rhodococcus erythropolis cells to salt stress by synthesizing polyunsaturated fatty acids. Appl. Microbiol. Biotechnol. 2014, 98, 5599–5606. [Google Scholar] [CrossRef] [PubMed]
- de Carvalho, C.C.C.R. Adaptation of Rhodococcus erythropolis cells for growth and bioremediation under extreme conditions. Res. Microbiol. 2012, 163, 125–136. [Google Scholar] [CrossRef] [PubMed]
- Heipieper, H.J.; Fischer, J. Bacterial solvent responses and tolerance: Cis-trans isomerization. In Handbook of Hydrocarbon and Lipid Microbiology; Timmis, K.N., Ed.; Springer: Berlin/Heidelberg, Germany, 2010; pp. 4203–4211. [Google Scholar]
- Eberlein, C.; Baumgarten, T.; Starke, S.; Heipieper, H.J. Immediate response mechanisms of Gram-negative solvent-tolerant bacteria to cope with environmental stress: Cis-trans isomerization of unsaturated fatty acids and outer membrane vesicle secretion. Appl. Microbiol. Biotechnol. 2018, 102, 2583–2593. [Google Scholar] [CrossRef] [PubMed]
- Saunders, L.P.; Sen, S.; Wilkinson, B.J.; Gatto, C. Insights into the mechanism of homeoviscous adaptation to low temperature in branched-chain fatty acid-containing bacteria through modeling FabH kinetics from the foodborne pathogen Listeria monocytogenes. Front. Microbiol. 2016, 7, 1386. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, F.D.A.; de Carvalho, C.C.C.R. Phenotypic modifications in Staphylococcus aureus cells exposed to high concentrations of vancomycin and teicoplanin. Front. Microbiol. 2016, 7, 13. [Google Scholar] [CrossRef] [PubMed]
- Unell, M.; Kabelitz, N.; Jansson, J.K.; Heipieper, H.J. Adaptation of the psychrotroph Arthrobacter chlorophenolicus A6 to growth temperature and the presence of phenols by changes in the anteiso/iso ratio of branched fatty acids. FEMS Microbiol. Lett. 2006, 266, 138–143. [Google Scholar] [CrossRef] [PubMed]
- Glickman, M.S.; Cox, J.S.; Jacobs, W.R., Jr. A novel mycolic acid cyclopropane synthetase is required for cording, persistence and virulence of Mycobacterium tuberculosis. Mol. Cell 2000, 5, 717–727. [Google Scholar] [CrossRef]
- Monteoliva-Sanchez, M.; Ramos-Cormenzana, A.; Russell, N.J. The effect of salinity and compatible solutes on the biosynthesis of cyclopropane fatty acids in Pseudomonas halosaccharolytica. Microbiology 1993, 139, 1877–1884. [Google Scholar] [CrossRef]
- Álvarez-Ordóñez, A.; Fernández, A.; López, M.; Arenas, R.; Bernardo, A. Modifications in membrane fatty acid composition of Salmonella typhimurium in response to growth conditions and their effect on heat resistance. Int. J. Food Microbiol. 2008, 123, 212–219. [Google Scholar] [CrossRef] [PubMed]
- Vinçon-Laugier, A.; Cravo-Laureau, C.; Mitteau, I.; Grossi, V. Temperature-dependent alkyl glycerol ether lipid composition of mesophilic and thermophilic sulfate-reducing bacteria. Front. Microbiol. 2017, 8, 1532. [Google Scholar] [CrossRef] [PubMed]
- Ernst, R.; Ejsing, C.S.; Antonny, B. Homeoviscous adaptation and the regulation of membrane lipids. J. Mol. Biol. 2016, 428, 4776–4791. [Google Scholar] [CrossRef] [PubMed]
- Melchior, D.L. Lipid phase transitions and regulation of membrane fluidity in prokaryotes. In Current Topics in Membranes and Transport; Bronner, F., Kleinteller, A., Eds.; Academic Press: Cambridge, MA, USA, 1982; Volume 17, pp. 263–316. [Google Scholar]
- Siliakus, M.F.; van der Oost, J.; Kengen, S.W.M. Adaptations of archaeal and bacterial membranes to variations in temperature, pH and pressure. Extremophiles 2017, 21, 651–670. [Google Scholar] [CrossRef] [PubMed]
- Eze, M.O. Phase transitions in phospholipid bilayers: Lateral phase separations play vital roles in biomembranes. Biochem. Educ. 1991, 19, 204–208. [Google Scholar] [CrossRef]
- Edidin, M. Rotational and translational diffusion in membranes. Annu. Rev. Biophys. Bioeng. 1974, 3, 179–201. [Google Scholar] [CrossRef] [PubMed]
- Wan, C.; Kiessling, V.; Tamm, L.K. Coupling of cholesterol-rich lipid phases in asymmetric bilayers. Biochemistry 2008, 47, 2190–2198. [Google Scholar] [CrossRef] [PubMed]
- Mouritsen, O.G. Phase transitions in biological membranes. Ann. N. Y. Acad. Sci. 1987, 491, 166–169. [Google Scholar] [CrossRef]
- Phadtare, S. Recent developments in bacterial cold-shock response. Curr. Issues Mol. Biol. 2004, 6, 125–136. [Google Scholar] [PubMed]
- Diomandé, S.; Nguyen-The, C.; Guinebretière, M.H.; Broussolle, V.; Brillard, J. Role of fatty acids in Bacillus environmental adaptation. Front. Microbiol. 2015, 6, 813. [Google Scholar] [CrossRef] [PubMed]
- Aguilar, P.S.; Cronan, J.E.; de Mendoza, D. A Bacillus subtilis gene induced by cold shock encodes a membrane phospholipid desaturase. J. Bacteriol. 1998, 180, 2194–2200. [Google Scholar] [PubMed]
- Klein, W.; Weber, M.H.W.; Marahiel, M.A. Cold shock response of Bacillus subtilis: Isoleucine-dependent switch in the fatty acid branching pattern for membrane adaptation to low temperatures. J. Bacteriol. 1999, 181, 5341–5349. [Google Scholar] [PubMed]
- Graumann, P.L.; Marahiel, M.A. Cold shock response in Bacillus subtilis. J. Mol. Microbiol. Biotechnol. 1999, 1, 203–209. [Google Scholar] [PubMed]
- Yamanaka, K. Cold shock response in Escherichia coli. J. Mol. Microbiol. Biotechnol. 1999, 1, 193–202. [Google Scholar] [PubMed]
- Annous, B.A.; Becker, L.A.; Bayles, D.O.; Labeda, D.P.; Wilkinson, B.J. Critical role of anteiso-C15:0 fatty acid in the growth of Listeria monocytogenes at low temperatures. Appl. Environ. Microbiol. 1997, 63, 3887–3894. [Google Scholar] [PubMed]
- Kaneda, T. Iso- and anteiso-fatty acids in bacteria: Biosynthesis, function and taxonomic significance. Microbiol. Rev. 1991, 55, 288–302. [Google Scholar] [PubMed]
- Suutari, M.; Laakso, S. Microbial fatty acids and thermal adaptation. Crit. Rev. Microbiol. 1994, 20, 285–328. [Google Scholar] [CrossRef] [PubMed]
- Mantsch, H.H.; Madec, C.; Lewis, R.N.A.H.; McElhaney, R.N. Thermotropic phase behavior of model membranes composed of phosphatidylcholines containing dl-methyl anteisobranched fatty acids. 2. An infrared spectroscopy study. Biochemistry 1987, 26, 4045–4049. [Google Scholar] [CrossRef] [PubMed]
- Diomandé, S.E.; Guinebretière, M.H.; De Sarrau, B.; Nguyen-the, C.; Broussolle, V.; Brillard, J. Fatty acid profiles and desaturase-encoding genes are different in thermo- and psychrotolerant strains of the Bacillus cereus Group. BMC Res. Notes 2015, 8, 329. [Google Scholar] [CrossRef] [PubMed]
- Suutari, M.; Laakso, S. Unsaturated and branched chain-fatty acids in temperature adaptation of Bacillus subtilis and Bacillus megaterium. Biochim. Biophys. Acta (BBA) Lipids Lipid Metab. 1992, 1126, 119–124. [Google Scholar] [CrossRef]
- Heipieper, H.J.; Meinhardt, F.; Segura, A. The cis-trans isomerase of unsaturated fatty acids in Pseudomonas and Vibrio: Biochemistry, molecular biology and physiological function of a unique stress adaptive mechanism. FEMS Microbiol. Lett. 2003, 229, 1–7. [Google Scholar] [CrossRef]
- Heipieper, H.J.; Fischer, J.; Meinhardt, F. Cis–trans isomerase of unsaturated fatty acids: An immediate bacterial adaptive mechanism to cope with emerging membrane perturbation caused by toxic hydrocarbons. In Handbook of Hydrocarbon and Lipid Microbiology; Timmis, K.N., Ed.; Springer: Berlin/Heidelberg, Germany, 2010; pp. 1605–1614. [Google Scholar]
- Okuyama, H.; Okajima, N.; Sasaki, S.; Higashi, S.; Murata, N. The cis/trans isomerization of the double bond of a fatty acid as a strategy for adaptation to changes in ambient temperature in the psychrophilic bacterium, Vibrio sp. strain ABE-1. Biochim.e Biophys. Acta (BBA) Lipids Lipid Metab. 1991, 1084, 13–20. [Google Scholar] [CrossRef]
- Junker, F.; Ramos, J.L. Involvement of the cis/trans isomerase Cti in solvent resistance of Pseudomonas putida DOT-T1E. J. Bacteriol. 1999, 181, 5693–5700. [Google Scholar] [PubMed]
- Pinkart, H.C.; White, D.C. Phospholipid biosynthesis and solvent tolerance in Pseudomonas putida strains. J. Bacteriol. 1997, 179, 4219–4226. [Google Scholar] [CrossRef] [PubMed]
- Atashgahi, S.; Sánchez-Andrea, I.; Heipieper, H.J.; van der Meer, J.R.; Stams, A.J.M.; Smidt, H. Prospects for harnessing biocide resistance for bioremediation and detoxification. Science 2018, 360, 743–746. [Google Scholar] [CrossRef] [PubMed]
- Kadurugamuwa, J.L.; Beveridge, T.J. Virulence factors are released from Pseudomonas aeruginosa in association with membrane vesicles during normal growth and exposure to gentamicin: A novel mechanism of enzyme secretion. J. Bacteriol. 1995, 177, 3998–4008. [Google Scholar] [CrossRef] [PubMed]
- Baumgarten, T.; Sperling, S.; Seifert, J.; von Bergen, M.; Steiniger, F.; Wick, L.Y.; Heipieper, H.J. Membrane vesicle formation as a multiple-stress response mechanism enhances Pseudomonas putida DOT-T1E cell surface hydrophobicity and biofilm formation. Appl. Environ. Microbiol. 2012, 78, 6217–6224. [Google Scholar] [CrossRef] [PubMed]
- DeLong, E.F.; Yayanos, A.A. Biochemical function and ecological significance of novel bacterial lipids in deep-sea procaryotes. Appl. Environ. Microbiol. 1986, 51, 730–737. [Google Scholar] [PubMed]
- Nichols, D.S. Prokaryotes and the input of polyunsaturated fatty acids to the marine food web. FEMS Microbiol. Lett. 2003, 219, 1–7. [Google Scholar] [CrossRef]
- Marsh, D. Handbook of Lipid Bilayers; CRC Press: Boca Raton, FL, USA, 2013; p. 1174. [Google Scholar]
- Allen, E.E.; Facciotti, D.; Bartlett, D.H. Monounsaturated but not polyunsaturated fatty acids are required for growth of the deep-sea bacterium Photobacterium profundum SS9 at high pressure and low temperature. Appl. Environ. Microbiol. 1999, 65, 1710–1720. [Google Scholar] [PubMed]
- Presentato, A.; Cappelletti, M.; Sansone, A.; Ferreri, C.; Piacenza, E.; Demeter, M.A.; Crognale, S.; Petruccioli, M.; Milazzo, G.; Fedi, S.; et al. Aerobic growth of Rhodococcus aetherivorans BCP1 using selected naphthenic acids as the sole carbon and energy sources. Front. Microbiol. 2018, 9, 672. [Google Scholar] [CrossRef] [PubMed]
- Marsh, D. Structural and thermodynamic determinants of chain-melting transition temperatures for phospholipid and glycolipids membranes. Biochim. Biophys. Acta (BBA)-Biomembr. 2010, 1798, 40–51. [Google Scholar] [CrossRef] [PubMed]
- Poger, D.; Caron, B.; Mark, A.E. Effect of methyl-branched fatty acids on the structure of lipid bilayers. J. Phys. Chem. B 2014, 118, 13838–13848. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.Y.; Cronan, J.E. Membrane cyclopropane fatty acid content is a major factor in acid resistance of Escherichia coli. Mol. Microbiol. 1999, 33, 249–259. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.H.; Kim, S.; Kim, H.G.; Lee, J.; Lee, I.S.; Park, Y.K. The formation of cyclopropane fatty acids in Salmonella enterica serovar Typhimurium. Microbiology 2005, 151, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Grogan, D.W.; Cronan, J.E., Jr. Cyclopropane ring formation in membrane lipids of bacteria. Microbiol. Mol. Biol. Rev. 1997, 61, 429–441. [Google Scholar] [PubMed]
- Desbois, A.P.; Smith, V.J. Antibacterial free fatty acids: Activities, mechanisms of action and biotechnological potential. Appl. Microbiol. Biotechnol. 2010, 85, 1629–1642. [Google Scholar] [CrossRef] [PubMed]
- Yoon, B.; Jackman, J.; Valle-González, E.; Cho, N.J. Antibacterial free fatty acids and monoglycerides: Biological activities, experimental testing and therapeutic applications. Int. J. Mo. Sci. 2018, 19, 1114. [Google Scholar] [CrossRef] [PubMed]
- Harayama, T.; Riezman, H. Understanding the diversity of membrane lipid composition. Nat. Rev. Mol. Cell Biol. 2018, 19, 281. [Google Scholar] [CrossRef] [PubMed]
- Simons, K.; Ikonen, E. Functional rafts in cell membranes. Nature 1997, 387, 569. [Google Scholar] [CrossRef] [PubMed]
- Van Meer, G.; Voelker, D.R.; Feigenson, G.W. Membrane lipids: Where they are and how they behave. Nat. Rev. Mol. Cell Biol. 2008, 9, 112. [Google Scholar] [CrossRef] [PubMed]
- Ibarguren, M.; López, D.J.; Escribá, P.V. The effect of natural and synthetic fatty acids on membrane structure, microdomain organization, cellular functions and human health. Biochim. Biophys. Acta (BBA) Biomembr. 2014, 1838, 1518–1528. [Google Scholar] [CrossRef] [PubMed]
- Guschina, I.A.; Harwood, J.L. Mechanisms of temperature adaptation in poikilotherms. FEBS Lett. 2006, 580, 5477–5483. [Google Scholar] [CrossRef] [PubMed]
- Klose, C.; Surma, M.A.; Gerl, M.J.; Meyenhofer, F.; Shevchenko, A.; Simons, K. Flexibility of a eukaryotic lipidome–insights from yeast lipidomics. PLoS ONE 2012, 7, e35063. [Google Scholar] [CrossRef] [PubMed]
- Suutari, M.; Liukkonen, K.; Laakso, S. Temperature adaptation in yeasts: The role of fatty acids. Microbiology 1990, 136, 1469–1474. [Google Scholar] [CrossRef] [PubMed]
- Pearcy, R.W. Effect of growth temperature on the fatty acid composition of the leaf lipids in Atriplex lentiformis (Torr.) Wats. Plant Physiol. 1978, 61, 484–486. [Google Scholar] [CrossRef] [PubMed]
- Berry, J.; Bjorkman, O. Photosynthetic response and adaptation to temperature in higher plants. Annu. Rev. Plant Physiol. 1980, 31, 491–543. [Google Scholar] [CrossRef]
- Williams, J.P.; Khan, M.U.; Mitchell, K.; Johnson, G. The effect of temperature on the level and biosynthesis of unsaturated fatty acids in diacylglycerols of Brassica napus leaves. Plant Physiol. 1988, 87, 904–910. [Google Scholar] [CrossRef] [PubMed]
- Harwood, J.L. Environmental factors which can alter lipid metabolism. Prog. Lipid Res. 1994, 33, 193–202. [Google Scholar] [CrossRef]
- Falcone, D.L.; Ogas, J.P.; Somerville, C.R. Regulation of membrane fatty acid composition by temperature in mutants of Arabidopsis with alterations in membrane lipid composition. BMC Plant Biol. 2004, 4, 17. [Google Scholar] [CrossRef] [PubMed]
- Mizusawa, N.; Wada, H. The role of lipids in photosystem II. Biochim. Biophys. Acta (BBA) Bioenerg. 2012, 1817, 194–208. [Google Scholar] [CrossRef] [PubMed]
- Farkas, T.; Fodor, E.; Kitajka, K.; Halver, J.E. Response of fish membranes to environmental temperature. Aquacult. Res. 2001, 32, 645–655. [Google Scholar] [CrossRef]
- Hachicho, N.; Reithel, S.; Miltner, A.; Heipieper, H.J.; Küster, E.; Luckenbach, T. Body mass parameters, lipid profiles and protein contents of zebrafish embryos and effects of 2,4-dinitrophenol exposure. PLoS ONE 2015, 10, e0134755. [Google Scholar] [CrossRef] [PubMed]
- Gurr, M.I.; James, A.T. Phospholipids. In Lipid Biochemistry: An Introduction; Gurr, M.I., James, A.T., Eds.; Springer: Dordrecht, The Netherlands, 1980; pp. 129–154. [Google Scholar]
- Gurr, M.I.; James, A.T. Fatty acids. In Lipid Biochemistry: An Introduction; Gurr, M.I., James, A.T., Eds.; Springer: Dordrecht, The Netherlands, 1980; pp. 18–89. [Google Scholar]
- Huster, D.; Jin, A.J.; Arnold, K.; Gawrisch, K. Water permeability of polyunsaturated lipid membranes measured by 17O NMR. Biophys. J. 1997, 73, 855–864. [Google Scholar] [CrossRef]
- Koenig, B.W.; Strey, H.H.; Gawrisch, K. Membrane lateral compressibility determined by NMR and X-ray diffraction: Effect of acyl chain polyunsaturation. Biophys. J. 1997, 73, 1954–1966. [Google Scholar] [CrossRef]
- Smaby, J.M.; Momsen, M.M.; Brockman, H.L.; Brown, R.E. Phosphatidylcholine acyl unsaturation modulates the decrease in interfacial elasticity induced by cholesterol. Biophys. J. 1997, 73, 1492–1505. [Google Scholar] [CrossRef]
- Williams, E.E.; Jenski, L.J.; Stillwell, W. Docosahexaenoic acid (DHA) alters the structure and composition of membranous vesicles exfoliated from the surface of a murine leukemia cell line. Biochim. Biophys. Acta (BBA) Biomembr. 1998, 1371, 351–362. [Google Scholar] [CrossRef]
- Calder, P.C. Immunomodulation by omega-3 fatty acids. Prostaglandins Leukot. Essent. Fat. Acids 2007, 77, 327–335. [Google Scholar] [CrossRef] [PubMed]
- Stillwell, W.; Wassall, S.R. Docosahexaenoic acid: Membrane properties of a unique fatty acid. Chem. Phys. Lipids 2003, 126, 1–27. [Google Scholar] [CrossRef]
- Armstrong, V.T.; Brzustowicz, M.R.; Wassall, S.R.; Jenski, L.J.; Stillwell, W. Rapid flip-flop in polyunsaturated (docosahexaenoate) phospholipid membranes. Arch. Biochem. Biophys. 2003, 414, 74–82. [Google Scholar] [CrossRef]
- Ma, D.W.L.; Seo, J.; Switzer, K.C.; Fan, Y.Y.; McMurray, D.N.; Lupton, J.R.; Chapkin, R.S. n−3 PUFA and membrane microdomains: A new frontier in bioactive lipid research. J. Nutri. Biochem. 2004, 15, 700–706. [Google Scholar] [CrossRef] [PubMed]
- Wassall, S.R.; Stillwell, W. Polyunsaturated fatty acid-cholesterol interactions: Domain formation in membranes. Biochim. Biophys. Acta (BBA) Biomembr. 2009, 1788, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Stulnig, T.M.; Huber, J.; Leitinger, N.; Imre, E.M.; Angelisová, P.; Nowotny, P.; Waldhäusl, W. Polyunsaturated eicosapentaenoic acid displaces proteins from membrane rafts by altering raft lipid composition. J. Biol. Chem. 2001, 276, 37335–37340. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Jump, D.B.; Esselman, W.J.; Busik, J.V. Inhibition of cytokine signaling in human retinal endothelial cells through modification of Caveolae/Lipid Rafts by docosahexaenoic acid. Investig. Ophthalmol. Vis. Sci. 2007, 48, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Ye, S.; Tan, L.; Ma, J.; Shi, Q.; Li, J. Polyunsaturated docosahexaenoic acid suppresses oxidative stress induced endothelial cell calcium influx by altering lipid composition in membrane caveolar rafts. Prostaglandins Leukot. Essent. Fat. Acids 2010, 83, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Schumann, J.; Leichtle, A.; Thiery, J.; Fuhrmann, H. Fatty acid and peptide profiles in plasma membrane and membrane rafts of PUFA supplemented RAW264.7 macrophages. PLoS ONE 2011, 6, e24066. [Google Scholar] [CrossRef] [PubMed]
- Salmon, A.; Dodd, S.W.; Williams, G.D.; Beach, J.M.; Brown, M.F. Configurational statistics of acyl chains in polyunsaturated lipid bilayers from deuterium NMR. J. Am. Chem. Soc. 1987, 109, 2600–2609. [Google Scholar] [CrossRef]
- Tanguy, E.; Kassas, N.; Vitale, N. Protein-phospholipid interaction motifs: A focus on phosphatidic acid. Biomolecules 2018, 8, 20. [Google Scholar] [CrossRef] [PubMed]
- Pinot, M.; Vanni, S.; Pagnotta, S.; Lacas-Gervais, S.; Payet, L.A.; Ferreira, T.; Gautier, R.; Goud, B.; Antonny, B.; Barelli, H. Polyunsaturated phospholipids facilitate membrane deformation and fission by endocytic proteins. Science 2014, 345, 693. [Google Scholar] [CrossRef] [PubMed]
- Barelli, H.; Antonny, B. Lipid unsaturation and organelle dynamics. Curr. Opin. Cell Biol. 2016, 41, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Rawicz, W.; Olbrich, K.C.; McIntosh, T.; Needham, D.; Evans, E. Effect of chain length and unsaturation on elasticity of lipid bilayers. Biophys. J. 2000, 79, 328–339. [Google Scholar] [CrossRef]
- Hashidate-Yoshida, T.; Harayama, T.; Hishikawa, D.; Morimoto, R.; Hamano, F.; Tokuoka, S.M.; Eto, M.; Tamura-Nakano, M.; Yanobu-Takanashi, R.; Mukumoto, Y.; et al. Fatty acid remodeling by LPCAT3 enriches arachidonate in phospholipid membranes and regulates triglyceride transport. eLife 2015, 4, e06328. [Google Scholar] [CrossRef] [PubMed]
- Rustan, A.C.; Drevon, C.A. Fatty acids: Structures and properties. In eLS; John Wiley & Sons Ltd.: Chischester, UK, 2005. [Google Scholar] [CrossRef]
- Weijers, R.N.M. Lipid composition of cell membranes and its relevance in Type 2 diabetes Mellitus. Curr. Diabetes Rev. 2012, 8, 390–400. [Google Scholar] [CrossRef] [PubMed]
- Koehrer, P.; Saab, S.; Berdeaux, O.; Isaïco, R.; Grégoire, S.; Cabaret, S.; Bron, A.M.; Creuzot-Garcher, C.P.; Bretillon, L.; Acar, N. Erythrocyte phospholipid and polyunsaturated fatty acid composition in diabetic retinopathy. PLoS ONE 2014, 9, e106912. [Google Scholar] [CrossRef] [PubMed]
- Weijers, R.N.M. Membrane flexibility, free fatty acids and the onset of vascular and neurological lesions in type 2 diabetes. J. Diabetes Metab. Disord. 2016, 15, 13. [Google Scholar] [CrossRef] [PubMed]
- Olukoshi, E.R.; Packter, N.M. Importance of stored triacylglycerols in Streptomyces: Possible carbon source for antibiotics. Microbiology 1994, 140, 931–943. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, H.; Steinbüchel, A. Triacylglycerols in prokaryotic microorganisms. Appl. Microbiol. Biotechnol. 2002, 60, 367–376. [Google Scholar] [CrossRef] [PubMed]
- Lemoigne, M. Produit de deshydratation et de polymerisation de l’acide β-oxybutyrique. Bull. Soc. Chim. Biol. 1926, 8, 770–782. [Google Scholar]
- Koller, M. Advances in polyhydroxyalkanoate (PHA) production. Bioengineering 2017, 4, 88. [Google Scholar] [CrossRef] [PubMed]
- Anderson, A.J.; Dawes, E.A. Occurrence, metabolism, metabolic role and industrial uses of bacterial polyhydroxyalkanoates. Microbiol. Rev. 1990, 54, 450–472. [Google Scholar] [PubMed]
- Bresan, S.; Sznajder, A.; Hauf, W.; Forchhammer, K.; Pfeiffer, D.; Jendrossek, D. Polyhydroxyalkanoate (PHA) granules have no phospholipids. Sci. Rep. 2016, 6, 26612. [Google Scholar] [CrossRef] [PubMed]
- Cavalheiro, J.M.B.T.; Almeida, M.C.M.D.; da Fonseca, M.M.R.; de Carvalho, C.C.C.R. Adaptation of Cupriavidus necator to conditions favoring polyhydroxyalkanoate production. J. Biotechnol. 2013, 164, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Koller, M. Production of polyhydroxyalkanoate (PHA) biopolyesters by extremophiles. MOJ Polym. Sci. 2017, 1, 69–85. [Google Scholar] [CrossRef]
- Obruca, S.; Sedlacek, P.; Mravec, F.; Samek, O.; Marova, I. Evaluation of 3-hydroxybutyrate as an enzyme-protective agent against heating and oxidative damage and its potential role in stress response of poly(3-hydroxybutyrate) accumulating cells. Appl. Microbiol. Biotechnol. 2016, 100, 1365–1376. [Google Scholar] [CrossRef] [PubMed]
- Wältermann, M.; Hinz, A.; Robenek, H.; Troyer, D.; Reichelt, R.; Malkus, U.; Galla, H.J.; Kalscheuer, R.; Stöveken, T.; Von Landenberg, P.; et al. Mechanism of lipid-body formation in prokaryotes: How bacteria fatten up. Mol. Microbiol. 2005, 55, 750–763. [Google Scholar] [CrossRef] [PubMed]
- Barney, B.M.; Wahlen, B.D.; Garner, E.; Wei, J.; Seefeldt, L.C. Differences in substrate specificities of five bacterial wax ester synthases. Appl. Environ. Microbiol. 2012, 78, 5734–5745. [Google Scholar] [CrossRef] [PubMed]
- Wältermann, M.; Steinbüchel, A. Neutral lipid bodies in prokaryotes: Recent insights into structure, formation and relationship to eukaryotic lipid depots. J. Bacteriol. 2005, 187, 3607–3619. [Google Scholar] [CrossRef] [PubMed]
- Sirakova, T.D.; Deb, C.; Daniel, J.; Singh, H.D.; Maamar, H.; Dubey, V.S.; Kolattukudy, P.E. Wax ester synthesis is required for Mycobacterium tuberculosis to enter in vitro dormancy. PLoS ONE 2012, 7, e51641. [Google Scholar] [CrossRef] [PubMed]
- Daniel, J.; Maamar, H.; Deb, C.; Sirakova, T.D.; Kolattukudy, P.E. Mycobacterium tuberculosis uses host triacylglycerol to accumulate lipid droplets and acquires a dormancy-like phenotype in lipid-loaded macrophages. PLoS Pathog. 2011, 7, e1002093. [Google Scholar] [CrossRef] [PubMed]
- Daniel, J.; Deb, C.; Dubey, V.S.; Sirakova, T.D.; Abomoelak, B.; Morbidoni, H.R.; Kolattukudy, P.E. Induction of a novel class of diacylglycerol acyltransferases and triacylglycerol accumulation in Mycobacterium tuberculosis as it goes into a dormancy-like state in culture. J. Bacteriol. 2004, 186, 5017–5030. [Google Scholar] [CrossRef] [PubMed]
- de Carvalho, C.C.C.R.; da Fonseca, M.M.R. The remarkable Rhodococcus erythropolis. Appl. Microbiol. Biotechnol. 2005, 67, 715–726. [Google Scholar] [CrossRef] [PubMed]
- Cortes, M.A.L.R.M.; de Carvalho, C.C.C.R. Effect of carbon sources on lipid accumulation in Rhodococcus cells. Biochem. Eng. J. 2015, 94, 100–105. [Google Scholar] [CrossRef]
- Wältermann, M.; Luftmann, H.; Baumeister, D.; Kalscheuer, R.; Steinbüchel, A. Rhodococcus opacus strain PD630 as a new source of high-value single-cell oil? Isolation and characterization of triacylglycerols and other storage lipids. Microbiology 2000, 146, 1143–1149. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, H.; Silva, R.; Herrero, M.; Hernández, M.; Villalba, M.S. Metabolism of triacylglycerols in Rhodococcus species: Insights from physiology and molecular genetics. J. Mol. Biochem. 2013, 2, 69–78. [Google Scholar]
- Alvarez, H.M. Relationship between β-oxidation pathway and the hydrocarbon-degrading profile in actinomycetes bacteria. Int. Biodeterior. Biodegrad. 2003, 52, 35–42. [Google Scholar] [CrossRef]
- Alvarez, H.M.; Mayer, F.; Fabritius, D.; Steinbüchel, A. Formation of intracytoplasmic lipid inclusions by Rhodococcus opacus strain PD630. Arch. Microbiol. 1996, 165, 377–386. [Google Scholar] [CrossRef] [PubMed]
- Castro, A.R.; Rocha, I.; Alves, M.M.; Pereira, M.A. Rhodococcus opacus B4: A promising bacterium for production of biofuels and biobased chemicals. AMB Express 2016, 6, 35. [Google Scholar] [CrossRef] [PubMed]
- Farese, R.V.; Walther, T.C. Lipid droplets finally get a little R-E-S-P-E-C-T. Cell 2009, 139, 855–860. [Google Scholar] [CrossRef] [PubMed]
- Walther, T.C.; Farese, R.V. Lipid droplets and cellular lipid metabolism. Annu. Rev. Biochem. 2012, 81, 687–714. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, A.S.; Coleman, R.A.; Kraemer, F.B.; McManaman, J.L.; Obin, M.S.; Puri, V.; Yan, Q.W.; Miyoshi, H.; Mashek, D.G. The role of lipid droplets in metabolic disease in rodents and humans. J. Clin. Investig. 2011, 121, 2102–2110. [Google Scholar] [CrossRef] [PubMed]
- Welte, M.A. Expanding roles for lipid droplets. Curr. Biol. CB 2015, 25, R470–R481. [Google Scholar] [CrossRef] [PubMed]
- Borel, P.; Grolier, P.; Armand, M.; Partier, A.; Lafont, H.; Lairon, D.; Azais-Braesco, V. Carotenoids in biological emulsions: Solubility, surface-to-core distribution and release from lipid droplets. J. Lipid Res. 1996, 37, 250–261. [Google Scholar] [PubMed]
- Goodrich, H.B.; Hill, G.A.; Arrick, M.S. The chemical identification of gene-controlled pigments in Platypoecilus and Xiphophorus and comparisons with other tropical fish. Genetics 1941, 26, 573–586. [Google Scholar] [PubMed]
- Matsumoto, J. Studies on fine structure and cytochemical properties of erythrophores in swordtail, Xiphophorus helleri, with special reference to their pigment granules (pterinosomes). J. Cell Biol. 1965, 27, 493–504. [Google Scholar] [CrossRef] [PubMed]
- Krinsky, N.I. The protective function of carotenoid pigments. In Photophysiology; Giese, A.C., Ed.; Academic Press: New York, NY, USA, 1968; pp. 123–195. [Google Scholar]
- Cogdell, R.J. Carotenoids in photosynthesis. Philos. Transac. R. Soc. Lond. B Biol. Sci. 1978, 284, 569–579. [Google Scholar] [CrossRef]
- Maoka, T. Carotenoids in marine animals. Mar. Drugs 2011, 9, 278. [Google Scholar] [CrossRef] [PubMed]
- Schneider, T.; Grosbois, G.; Vincent, W.F.; Rautio, M. Carotenoid accumulation in copepods is related to lipid metabolism and reproduction rather than to UV-protection. Limnol. Oceanogr. 2016, 61, 1201–1213. [Google Scholar] [CrossRef]
- Schneider, T.; Grosbois, G.; Vincent, W.F.; Rautio, M. Saving for the future: Pre-winter uptake of algal lipids supports copepod egg production in spring. Freshw. Biol. 2017, 62, 1063–1072. [Google Scholar] [CrossRef]
- Goodman, D.S. Overview of current knowledge of metabolism of vitamin A and carotenoids12. JNCI J. Nat. Cancer Inst. 1984, 73, 1375–1379. [Google Scholar] [CrossRef] [PubMed]
- Kainz, M.J.; Fisk, A.T. Integrating lipids and contaminants in aquatic ecology and ecotoxicology. In Lipids in Aquatic Ecosystems; Kainz, M., Brett, M.T., Arts, M.T., Eds.; Springer: New York, NY, USA, 2009; pp. 93–114. [Google Scholar]
- Ansari, G.A.S.; Kaphalia, B.S.; Khan, M.F. Fatty acid conjugates of xenobiotics. Toxicol. Lett. 1995, 75, 1–17. [Google Scholar] [CrossRef]
- D’Adamo, R.; Pelosi, S.; Trotta, P.; Sansone, G. Bioaccumulation and biomagnification of polycyclic aromatic hydrocarbons in aquatic organisms. Mar. Chem. 1997, 56, 45–49. [Google Scholar] [CrossRef]
- Wong, C.S.; Mabury, S.A.; Whittle, D.M.; Backus, S.M.; Teixeira, C.; DeVault, D.S.; Bronte, C.R.; Muir, D.C.G. Organochlorine compounds in Lake Superior: chiral polychlorinated biphenyls and biotransformation in the aquatic food web. Environ. Sci. Technol. 2004, 38, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Murphy, G.; Rouse, R.L.; Polk, W.W.; Henk, W.G.; Barker, S.A.; Boudreaux, M.J.; Floyd, Z.E.; Penn, A.L. Combustion-derived hydrocarbons localize to lipid droplets in respiratory cells. Am. J. Respir. Cell Mol. Biol. 2008, 38, 532–540. [Google Scholar] [CrossRef] [PubMed]
- Rowan-Carroll, A.; Halappanavar, S.; Williams, A.; Somers, C.M.; Yauk, C.L. Mice exposed in situ to urban air pollution exhibit pulmonary alterations in gene expression in the lipid droplet synthesis pathways. Environ. Mol. Mutag. 2013, 54, 240–249. [Google Scholar] [CrossRef] [PubMed]
- Olofsson, S.O.; Boström, P.; Andersson, L.; Rutberg, M.; Perman, J.; Borén, J. Lipid droplets as dynamic organelles connecting storage and efflux of lipids. Biochim. Biophys. Acta (BBA) Mol. Cell Biol. Lipids 2009, 1791, 448–458. [Google Scholar] [CrossRef] [PubMed]
- Henne, W.M.; Reese, M.L.; Goodman, J.M. The assembly of lipid droplets and their roles in challenged cells. EMBO J. 2018. [Google Scholar] [CrossRef] [PubMed]
- Hugo, W.B.; Stretton, R.J. The role of cellular lipid in the resistance of Gram-positive bacteria to penicillins. Microbiology 1966, 42, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Chang, W.; Zhang, M.; Zheng, S.; Li, Y.; Li, X.; Li, W.; Li, G.; Lin, Z.; Xie, Z.; Zhao, Z.; et al. Trapping toxins within lipid droplets is a resistance mechanism in fungi. Sci. Rep. 2015, 5, 15133. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, H.M.; Luftmann, H.; Silva, R.A.; Cesari, A.C.; Viale, A.; Wältermann, M.; Steinbüchel, A. Identification of phenyldecanoic acid as a constituent of triacylglycerols and wax ester produced by Rhodococcus opacus PD630. Microbiology 2002, 148, 1407–1412. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, H.M.; Souto, M.F.; Viale, A.; Pucci, O.H. Biosynthesis of fatty acids and triacylglycerols by 2,6,10,14-tetramethyl pentadecane-grown cells of Nocardia globerula 432. FEMS Microbiol. Lett. 2001, 200, 195–200. [Google Scholar] [CrossRef] [PubMed]
- Uauy, R.; Hoffman, D.R.; Peirano, P.; Birch, D.G.; Birch, E.E. Essential fatty acids in visual and brain development. Lipids 2001, 36, 885–895. [Google Scholar] [CrossRef] [PubMed]
- Glomset, J.A. Role of docosahexaenoic acid in neuronal plasma membranes. Sci. STKE 2006, 2006, pe6. [Google Scholar] [CrossRef] [PubMed]
- Cao, D.; Kevala, K.; Kim, J.; Moon, H.S.; Jun, S.B.; Lovinger, D.; Kim, H.Y. Docosahexaenoic acid promotes hippocampal neuronal development and synaptic function. J. Neurochem. 2009, 111, 510–521. [Google Scholar] [CrossRef] [PubMed]
- Marcheselli, V.L.; Hong, S.; Lukiw, W.J.; Tian, X.H.; Gronert, K.; Musto, A.; Hardy, M.; Gimenez, J.M.; Chiang, N.; Serhan, C.N.; et al. Novel docosanoids inhibit brain ischemia-reperfusion-mediated leukocyte infiltration and pro-inflammatory gene expression. J. Biol. Chem. 2003, 278, 43807–43817. [Google Scholar] [CrossRef] [PubMed]
- Bazan, N.G.; Molina, M.F.; Gordon, W.C. Docosahexaenoic acid signalolipidomics in nutrition: Significance in aging, neuroinflammation, macular degeneration, Alzheimer’s and other neurodegenerative diseases. Annu. Rev. Nutr. 2011, 31, 321–351. [Google Scholar] [CrossRef] [PubMed]
- Orr, S.K.; Trépanier, M.O.; Bazinet, R.P. n-3 Polyunsaturated fatty acids in animal models with neuroinflammation. Prostaglandins Leukot. Essent. Fat. Acids 2013, 88, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Bang, H.O.; Dyerberg, J. Plasma lipids and lipoproteins in Greenlandic west coast Eskimos. Acta Med. Scand. 1972, 192, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Dyerberg, J.; Bang, H.O.; Stoffersen, E.; Moncada, S.; Vane, J.R. Eicosapentaenoic acid and prevention of thrombosis and atheroscloerosis? Lancet 1978, 312, 117–119. [Google Scholar] [CrossRef]
- Dyerberg, J. Linolenate-derived polyunsaturated fatty acids and prevention of atherosclerosis. Nutr. Rev. 1986, 44, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Leaf, A.; Kang, J.X.; Xiao, Y.F.; Billman, G. Clinical prevention of sudden cardiac death by n-3 polyunsaturated fatty acids and mechanism of prevention of arrhythmias by n-3 fish oils. Circulation 2003, 107, 2646–2652. [Google Scholar] [CrossRef] [PubMed]
- Tong, M.; Wang, J.; Ji, Y.; Chen, X.; Wang, J.; Wang, S.; Ruan, L.; Cui, H.; Zhou, Y.; Zhang, Q.; et al. Effect of eicosapentaenoic acid and pitavastatin on electrophysiology and anticoagulant gene expression in mice with rapid atrial pacing. Exp. Ther. Med. 2017, 14, 2310–2316. [Google Scholar] [CrossRef] [PubMed]
- Harris, W.S. n-3 fatty acids and serum lipoproteins: Human studies. Am. J. Clin. Nutr. 1997, 65, 1645S–1654S. [Google Scholar] [CrossRef] [PubMed]
- Gladine, C.; Newman, J.W.; Durand, T.; Pedersen, T.L.; Galano, J.M.; Demougeot, C.; Berdeaux, O.; Pujos-Guillot, E.; Mazur, A.; Comte, B. Lipid profiling following intake of the omega 3 fatty acid DHA identifies the peroxidized metabolites F4-neuroprostanes as the best predictors of atherosclerosis prevention. PLoS ONE 2014, 9, e89393. [Google Scholar] [CrossRef] [PubMed]
- Phillips, M.C. Molecular mechanisms of cellular cholesterol efflux. J. Biol. Chem. 2014, 289, 24020–24029. [Google Scholar] [CrossRef] [PubMed]
- Yamagata, K. Docosahexaenoic acid regulates vascular endothelial cell function and prevents cardiovascular disease. Lipids Health Dis. 2017, 16, 118. [Google Scholar] [CrossRef] [PubMed]
- Knapp, H.R. Dietary fatty acids in human thrombosis and hemostasis. Am. J. Clin. Nutri. 1997, 65, 1687S–1698S. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Polyunsaturated fatty acids, inflammation and immunity. Lipids 2001, 36, 1007–1024. [Google Scholar] [CrossRef] [PubMed]
- Ferreri, C.; Masi, A.; Sansone, A.; Giacometti, G.; Larocca, V.A.; Menounou, G.; Scanferlato, R.; Tortorella, S.; Rota, D.; Conti, M.; et al. Fatty acids in membranes as homeostatic, metabolic and nutritional biomarkers: Recent advancements in analytics and diagnostics. Diagnostics 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Bergström, S.; Danielsson, H.; Samuelsson, B. The enzymatic formation of prostaglandin E2 from arachidonic acid prostaglandins and related factors 32. Biochim. Biophys. Acta (BBA) Gen. Subj. 1964, 90, 207–210. [Google Scholar] [CrossRef]
- Van Dorp, D.A.; Beerthuis, R.K.; Nugteren, D.H.; Vonkeman, H. The biosynthesis of prostaglandins. Biochim. Biophys. Acta (BBA) Gen. Subj. 1964, 90, 204–207. [Google Scholar] [CrossRef]
- Corey, E.J.; Albright, J.O.; Barton, A.E.; Hashimoto, S. Chemical and enzymic syntheses of 5-HPETE, a key biological precursor of slow-reacting substance of anaphylaxis (SRS) and 5-HETE. J. Am. Chem. Soc. 1980, 102, 1435–1436. [Google Scholar] [CrossRef]
- Von Euler, U.S. On the specific vaso-dilating and plain muscle stimulating substances from accessory genital glands in man and certain animals (prostaglandin and vesiglandin). J. Physiol. 1936, 88, 213–234. [Google Scholar] [CrossRef] [PubMed]
- Samuelsson, B. From studies of biochemical mechanism to novel biological mediators: Prostaglandin endoperoxides, thromboxanes and leukotrienes (Nobel Lecture). Angew. Chem. Int. Ed. Engl. 1983, 22, 805–815. [Google Scholar] [CrossRef]
- Ricciotti, E.; FitzGerald, G.A. Prostaglandins and inflammation. Atertio. Thromb. Vasc. Biol. 2011, 31, 986–1000. [Google Scholar] [CrossRef] [PubMed]
- Duvall, M.G.; Levy, B.D. DHA- and EPA-derived resolvins, protectins and maresins in airway inflammation. Eur. J. Pharmacol. 2016, 785, 144–155. [Google Scholar] [CrossRef] [PubMed]
- Levy, B.D.; Clish, C.B.; Schmidt, B.; Gronert, K.; Serhan, C.N. Lipid mediator class switching during acute inflammation: Signals in resolution. Nat. Immunol. 2001, 2, 612. [Google Scholar] [CrossRef] [PubMed]
- Serhan, C.N. Novel pro-resolving lipid mediators in inflammation are leads for resolution physiology. Nature 2014, 510, 92–101. [Google Scholar] [CrossRef] [PubMed]
- Stanley-Samuelson, D.W. The biological significance of prostaglandins and related eicosanoids in invertebrates. Am. Zool. 1994, 34, 589–598. [Google Scholar] [CrossRef]
- Stanley, D.W.; Howard, R.W. The biology of prostaglandins and related eicosanoids in invertebrates: Cellular, organismal and ecological actions. Am. Zool. 1998, 38, 369–381. [Google Scholar] [CrossRef]
- Miralto, G.B.; Romano, G.; Sa, P.; Ianora, A.; Russo, G.L.; Buttino, I.; Mazzarella, G.; Laabir, M.; Cabrini, M.; Mg, G. The insidious effect of diatoms on copepod reproduction. Nature 1999, 402, 173–176. [Google Scholar] [CrossRef]
- Eckardt, N.A. Oxylipin signaling in plant stress responses. Plant Cell 2008, 20, 495–497. [Google Scholar] [CrossRef] [PubMed]
- Okazaki, Y.; Saito, K. Roles of lipids as signaling molecules and mitigators during stress response in plants. Plant J. 2014, 79, 584–596. [Google Scholar] [CrossRef] [PubMed]
- Watson, S.B.; Caldwell, G.; Pohnert, G. Fatty acids and oxylipins as semiochemicals. In Lipids in Aquatic Ecosystems; Kainz, M., Brett, M.T., Arts, M.T., Eds.; Springer: New York, NY, USA, 2009; pp. 65–92. [Google Scholar]
- Pohl, H.C.; Kock, L.J. Oxidized fatty acids as inter-kingdom signaling molecules. Molecules 2014, 19, 1273–1285. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, T.; Katoh, S. Linolenic acid binding by chloroplasts. Plant Cell Physiol. 1977, 18, 539–550. [Google Scholar] [CrossRef]
- Wu, J.T.; Chiang, Y.R.; Huang, W.Y.; Jane, W.N. Cytotoxic effects of free fatty acids on phytoplankton algae and cyanobacteria. Aquat. Toxicol. 2006, 80, 338–345. [Google Scholar] [CrossRef] [PubMed]
- Murakami, M.; Makabe, K.; Yamaguchi, K.; Konosu, S. Cytotoxic polyunsaturated fatty acid from Pediastrum. Phytochemistry 1989, 28, 625–626. [Google Scholar] [CrossRef]
- Rukmini, C. Reproductive toxicology and nutritional studies on mahua oil (Madhuca latifolia). Food Chem. Toxicol. 1990, 28, 601–605. [Google Scholar] [CrossRef]
- Sellem, F.; Pesando, D.; Bodennec, G.; El Abed, A.; Girard, J.P. Toxic effects of Gymnodinium cf. mikimotoi unsaturated fatty acids to gametes and embryos of the sea urchin Paracentrotus lividus. Water Res. 2000, 34, 550–556. [Google Scholar] [CrossRef]
- Reinikainen, M.; Meriluoto, J.A.O.; Spoof, L.; Harada, K.-i. The toxicities of a polyunsaturated fatty acid and a microcystin to Daphnia magna. Environ. Toxicol. 2001, 16, 444–448. [Google Scholar] [CrossRef] [PubMed]
- Hombeck, M.; Pohnert, G.; Boland, W. Biosynthesis of dictyopterene A: Stereoselectivity of a lipoxygenase/hydroperoxide lyase from Gomphonema parvulum (Bacillariophyceae). Chem. Commun. 1999, 243–244. [Google Scholar] [CrossRef]
- Barofsky, A.; Pohnert, G. Biosynthesis of polyunsaturated short chain aldehydes in the diatom Thalassiosira rotula. Org. Lett. 2007, 9, 1017–1020. [Google Scholar] [CrossRef] [PubMed]
- Wichard, T.; Pohnert, G. Formation of halogenated medium chain hydrocarbons by a lipoxygenase/hydroperoxide halolyase-mediated transformation in planktonic microalgae. J. Am. Chem. Soc. 2006, 128, 7114–7115. [Google Scholar] [CrossRef] [PubMed]
- Tosti, E.; Romano, G.; Buttino, I.; Cuomo, A.; Ianora, A.; Miralto, A. Bioactive aldehydes from diatoms block the fertilization current in ascidian oocytes. Mol. Reprod. Dev. 2003, 66, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Adolph, S.; Poulet, S.A.; Pohnert, G. Synthesis and biological activity of α,β,γ,δ-unsaturated aldehydes from diatoms. Tetrahedron 2003, 59, 3003–3008. [Google Scholar] [CrossRef]
- Lewis, C.; Caldwell, G.S.; Bentley, M.G.; Olive, P.J.W. Effects of a bioactive diatom-derived aldehyde on developmental stability in Nereis virens (Sars) larvae: An analysis using fluctuating asymmetry. J. Exp. Mar. Biol. Ecol. 2004, 304, 1–16. [Google Scholar] [CrossRef]
- Caldwell, G.S. The influence of bioactive oxylipins from marine diatoms on invertebrate reproduction and development. Mar. Drugs 2009, 7, 367–400. [Google Scholar] [CrossRef] [PubMed]
- Ianora, A.; Poulet, S.A.; Miralto, A. The effects of diatoms on copepod reproduction: A review. Phycologia 2003, 42, 351–363. [Google Scholar] [CrossRef]
- de Carvalho, C.C.C.R.; Caramujo, M.J. Bacterial diversity assessed by cultivation-based techniques shows predominance of Staphylococccus species on coins collected in Lisbon and Casablanca. FEMS Microbiol. Ecol. 2014, 88, 26–37. [Google Scholar] [CrossRef] [PubMed]
- Dijkman, N.A.; Kromkamp, J.C. Phospholipid-derived fatty acids as chemotaxonomic markers for phytoplankton: Application for inferring phytoplankton composition. Mar. Ecol. Prog. Ser. 2006, 324, 113–125. [Google Scholar] [CrossRef]
- Graeve, M.; Kattner, G.; Hagen, W. Diet-induced changes in the fatty acid composition of Arctic herbivorous copepods: Experimental evidence of trophic markers. J. Exp. Mar. Biol. Ecol. 1994, 182, 97–110. [Google Scholar] [CrossRef]
- Pollierer, M.M.; Scheu, S.; Haubert, D. Taking it to the next level: Trophic transfer of marker fatty acids from basal resource to predators. Soil Biol. Biochem. 2010, 42, 919–925. [Google Scholar] [CrossRef]
- Kunitsky, C.; Osterhout, G.; Sasser, M. Identification of microorganisms using fatty acid methyl ester (FAME) analysis and the MIDI Sherlock Microbial Identification System. In Encyclopedia of Rapid Microbiological Methods; Miller, M.J., Ed.; PDA Bookstore: Baltimore, MD, USA, 2006; Volume 3, pp. 1–18. [Google Scholar]
- Ifkovits, R.W.; Ragheb, H.S. Cellular fatty acid composition and identification of rumen bacteria. Appl. Microbiol. 1968, 16, 1406–1413. [Google Scholar] [PubMed]
- Tang, Y.W.; Ellis, N.M.; Hopkins, M.K.; Smith, D.H.; Dodge, D.E.; Persing, D.H. Comparison of phenotypic and genotypic techniques for identification of unusual aerobic pathogenic Gram-negative bacilli. J. Clin. Microbiol. 1998, 36, 3674–3679. [Google Scholar] [PubMed]
- Abel, K.; deSchmertzing, H.; Peterson, J.I. Classification of microorganisms by analysis of chemical composition I. Feasibility of utilizing gas chromatography. J. Bacteriol. 1963, 85, 1039–1044. [Google Scholar] [PubMed]
- Osterhout, G.J.; Shull, V.H.; Dick, J.D. Identification of clinical isolates of gram-negative nonfermentative bacteria by an automated cellular fatty acid identification system. J. Clin. Microbiol. 1991, 29, 1822–1830. [Google Scholar] [PubMed]
- Sasser, M. Bacterial Identification by Gas Chromatographic Analysis of Fatty Acid Methyl Esters (GC-FAME); Technical Note #101; MIDI, Inc.: Newark, DE, USA, 1990. [Google Scholar]
- Stead, D.E.; Sellwood, J.E.; Wilson, J.; Viney, I. Evaluation of a commercial microbial identification system based on fatty acid profiles for rapid, accurate identification of plant pathogenic bacteria. J. Appl. Bacteriol. 1992, 72, 315–321. [Google Scholar] [CrossRef]
- Leonard, R.B.; Mayer, J.; Sasser, M.; Woods, M.L.; Mooney, B.R.; Brinton, B.G.; Newcomb-Gayman, P.L.; Carroll, K.C. Comparison of MIDI Sherlock system and pulsed-field gel electrophoresis in characterizing strains of methicillin-resistant Staphylococcus aureus from a recent hospital outbreak. J. Clin. Microbiol. 1995, 33, 2723–2727. [Google Scholar] [PubMed]
- Birnbaum, D.; Herwaldt, L.; Low, D.E.; Noble, M.; Pfaller, M.; Sherertz, R.; Chow, A.W. Efficacy of microbial identification system for epidemiologic typing of coagulase-negative staphylococci. J. Clin. Microbiol. 1994, 32, 2113–2119. [Google Scholar] [PubMed]
- Ash, C.; Farrow, J.A.E.; Wallbanks, S.; Collins, M.D. Phylogenetic heterogeneity of the genus Bacillus revealed by comparative analysis of small-subunit-ribosomal RNA sequences. Lett. Appl. Microbiol. 1991, 13, 202–206. [Google Scholar] [CrossRef]
- Priest, F.G.; Kaji, D.A.; Rosato, Y.B.; Canhos, V.P. Characterization of Bacillus thuringiensis and related bacteria by ribosomal RNA gene restriction fragment length polymorphisms. Microbiology 1994, 140, 1015–1022. [Google Scholar] [CrossRef] [PubMed]
- Bourque, S.N.; Valero, J.R.; Lavoie, M.C.; Levesque, R.C. Comparative analysis of the 16S to 23S ribosomal intergenic spacer sequences of Bacillus thuringiensis strains and subspecies and of closely related species. Appl. Environ. Microbiol. 1995, 61, 1623–1626. [Google Scholar] [PubMed]
- Wintzingerode, F.; Rainey, F.A.; Kroppenstedt, R.M.; Stackebrandt, E. Identification of environmental strains of Bacillus mycoides by fatty acid analysis and species-specific 16S rDNA oligonucleotide probe. FEMS Microbiol. Ecol. 1997, 24, 201–209. [Google Scholar] [CrossRef]
- Slabbinck, B.; De Baets, B.; Dawyndt, P.; De Vos, P. Genus-wide Bacillus species identification through proper artificial neural network experiments on fatty acid profiles. Antonie Van Leeuwenhoek 2008, 94, 187–198. [Google Scholar] [CrossRef] [PubMed]
- Abraham, W.R.; Lünsdorf, H.; Vancanneyt, M.; Smit, J. Cauliform bacteria lacking phospholipids from an abyssal hydrothermal vent: Proposal of Glycocaulis abyssi gen. nov., sp. nov., belonging to the family Hyphomonadaceae. Int. J. Syst. Evol. Microbiol. 2013, 63, 2207–2215. [Google Scholar] [CrossRef] [PubMed]
- Torreblanca, M.; Rodriguez-Valera, F.; Juez, G.; Ventosa, A.; Kamekura, M.; Kates, M. Classification of non-alkaliphilic halobacteria based on numerical taxonomy and polar lipid composition and description of Haloarcula gen. nov. and Haloferax gen. nov. Syst. Appl. Microbiol. 1986, 8, 89–99. [Google Scholar] [CrossRef]
- Oren, A.; Ventosa, A. Subcommittee on the taxonomy of Halobacteriaceae and Subcommittee on the taxonomy of Halomonadaceae. Int. J. Syst. Evol. Microbiol. 2013, 63, 3540–3544. [Google Scholar] [CrossRef] [PubMed]
- de la Haba, R.R.; Corral, P.; Sánchez-Porro, C.; Infante-Domínguez, C.; Makkay, A.M.; Amoozegar, M.A.; Ventosa, A.; Papke, R.T. Genotypic and lipid analyses of strains from the Archaeal genus Halorubrum reveal insights into their taxonomy, divergence and population structure. Front. Microbiol. 2018, 9, 512. [Google Scholar] [CrossRef] [PubMed]
- White, D.C.; Davis, W.M.; Nickels, J.S.; King, J.D.; Bobbie, R.J. Determination of the sedimentary microbial biomass by extractible lipid phosphate. Oecologia 1979, 40, 51–62. [Google Scholar] [CrossRef] [PubMed]
- Pinkart, H.; Ringelberg, D.; Piceno, Y.; Macnaughton, S.; White, D.C. Biochemical Approaches to Biomass Measurements and Community Structure; American Society for Microbiology Press: Washington, DC, USA, 2002; pp. 101–113. [Google Scholar]
- Frostegård, Å.; Bååth, E.; Tunlio, A. Shifts in the structure of soil microbial communities in limed forests as revealed by phospholipid fatty acid analysis. Soil Biol. Biochem. 1993, 25, 723–730. [Google Scholar] [CrossRef]
- Feinstein, L.M.; Sul, W.J.; Blackwood, C.B. Assessment of bias associated with incomplete extraction of microbial DNA from soil. Appl. Environ. Microbiol. 2009, 75, 5428–5433. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Qu, Y.; Li, S.; Feng, K.; Wang, S.; Cai, W.; Liang, Y.; Li, H.; Xu, M.; Yin, H.; et al. Soil bacterial quantification approaches coupling with relative abundances reflecting the changes of taxa. Sci. Rep. 2017, 7, 4837. [Google Scholar] [CrossRef] [PubMed]
- Ramsey, P.W.; Rillig, M.C.; Feris, K.P.; Holben, W.E.; Gannon, J.E. Choice of methods for soil microbial community analysis: PLFA maximizes power compared to CLPP and PCR-based approaches. Pedobiologia 2006, 50, 275–280. [Google Scholar] [CrossRef]
- White, D.C.; Ringelberg, D.B. Signature lipid biomarker analysis. In Techniques in Microbial Ecology; Burlage, R.S., Atlas, R., Stahl, D., Geesey, G., Sayler, G., Eds.; Oxford University Press: New York, NY, USA, 1998; pp. 255–272. [Google Scholar]
- Ackman, R.G.; Tocher, C.S.; McLachlan, J. Marine phytoplankter fatty acids. J. Fish. Res. Board Can. 1968, 25, 1603–1620. [Google Scholar] [CrossRef]
- Ahlgren, G.; Gustafsson, I.B.; Boberg, M. Fatty acid content and chemical composition of freshwater microalgae. J. Phycol. 1992, 28, 37–50. [Google Scholar] [CrossRef]
- Bourdier, G.G.; Amblard, C.A. Lipids in Acanthodiaptomus denticomis during starvation and fed on three different algae. J. Plankton Res. 1989, 11, 1201–1212. [Google Scholar] [CrossRef]
- Nichols, P.D.; Jones, G.J.; De Leeuw, J.W.; Johns, R.B. The fatty acid and sterol composition of two marine dinoflagellates. Phytochemistry 1984, 23, 1043–1047. [Google Scholar] [CrossRef]
- Viso, A.-C.; Marty, J.C. Fatty acids from 28 marine microalgae. Phytochemistry 1993, 34, 1521–1533. [Google Scholar] [CrossRef]
- Volkman, J.K.; Jeffrey, S.W.; Nichols, P.D.; Rogers, G.I.; Garland, C.D. Fatty acid and lipid composition of 10 species of microalgae used in mariculture. J. Exp. Mar. Biol. Ecol. 1989, 128, 219–240. [Google Scholar] [CrossRef]
- Dunstan, G.A.; Volkman, J.K.; Barrett, S.M.; Leroi, J.M.; Jeffrey, S.W. Essential polyunsaturated fatty acids from 14 species of diatom (Bacillariophyceae). Phytochemistry 1993, 35, 155–161. [Google Scholar] [CrossRef]
- Zhukova, N.V.; Aizdaicher, N.A. Fatty acid composition of 15 species of marine microalgae. Phytochemistry 1995, 39, 351–356. [Google Scholar] [CrossRef]
- Caramujo, M.J.; Boschker, H.T.S.; Admiraal, W.I.M. Fatty acid profiles of algae mark the development and composition of harpacticoid copepods. Freshw. Biol. 2008, 53, 77–90. [Google Scholar] [CrossRef]
- Napolitano, G.E. The relationship of lipids with light and chlorophyll measurements in freshwater algae and periphyton. J. Phycol. 1994, 30, 943–950. [Google Scholar] [CrossRef]
- Guschina, I.A.; Harwood, J.L. Algal lipids and effect of the environment on their biochemistry. In Lipids in Aquatic Ecosystems; Kainz, M., Brett, M.T., Arts, M.T., Eds.; Springer: New York, NY, USA, 2009; pp. 1–24. [Google Scholar]
- Reuss, N.; Poulsen, L. Evaluation of fatty acids as biomarkers for a natural plankton community. A field study of a spring bloom and a post-bloom period off West Greenland. Mar. Biol. 2002, 141, 423–434. [Google Scholar] [CrossRef]
- Cañavate, J.P. Advancing assessment of marine phytoplankton community structure and nutritional value from fatty acid profiles of cultured microalgae. Rev. Aquacult. 2018. [Google Scholar] [CrossRef]
- Bec, A.; Perga, M.E.; Desvilettes, C.; Bourdier, G. How well can the fatty acid content of Lake Seston be predicted from its taxonomic composition? Freshw. Biol. 2010, 55, 1958–1972. [Google Scholar] [CrossRef]
- Desvilettes, C.; Bec, A. Formation and transfer of fatty acids in aquatic microbial food webs: Role of heterotrophic protists. In Lipids in Aquatic Ecosystems; Kainz, M., Brett, M.T., Arts, M.T., Eds.; Springer: New York, NY, USA, 2009; pp. 25–42. [Google Scholar]
- Copeman, L.A.; Parrish, C.C.; Gregory, R.S.; Jamieson, R.E.; Wells, J.; Whiticar, M.J. Fatty acid biomarkers in coldwater eelgrass meadows: Elevated terrestrial input to the food web of age-0 Atlantic cod Gadus morhua. Mar. Ecol. Prog. Ser. 2009, 386, 237–251. [Google Scholar] [CrossRef]
- McMeans, B.C.; Koussoroplis, A.M.; Kainz, M.J. Effects of seasonal seston and temperature changes on lake zooplankton fatty acids. Limnol. Oceanogr. 2015, 60, 573–583. [Google Scholar] [CrossRef]
- Budge, S.; Springer, A.; Iverson, S.; Sheffield, G. Fatty acid biomarkers reveal niche separation in an Arctic benthic food web. Mar. Ecol. Prog. Ser. 2007, 336, 305–309. [Google Scholar] [CrossRef]
- James Henderson, R.; Tocher, D.R. The lipid composition and biochemistry of freshwater fish. Prog. Lipid Res. 1987, 26, 281–347. [Google Scholar] [CrossRef]
- Dalsgaard, J.; St John, M.; John, G.; Kattner, D.C.; Muller-Navarra, D.C.; Hagen, W. Fatty acid trophic markers in the pelagic marine food environment. Adv. Mar. Biol. 2003, 46, 226–340. [Google Scholar] [CrossRef]
- Iverson, S.J.; Field, C.; Don Bowen, W.; Blanchard, W. Quantitative fatty acid signature analysis: A new method of estimating predator diets. Ecol. Monogr. 2004, 74, 211–235. [Google Scholar] [CrossRef]
- Falk-Petersen, S.; Haug, T.; Nilssen, K.T.; Wold, A.; Dahl, T.M. Lipids and trophic linkages in harp seal (Phoca groenlandica) from the eastern Barents Sea. Polar Res. 2006, 23, 43–50. [Google Scholar] [CrossRef]
- Martin-Creuzburg, D.; Kowarik, C.; Straile, D. Cross-ecosystem fluxes: Export of polyunsaturated fatty acids from aquatic to terrestrial ecosystems via emerging insects. Sci. Total Environ. 2017, 577, 174–182. [Google Scholar] [CrossRef] [PubMed]
- Ludvigsen, L.; Albrechtsen, H.J.; Holst, H.; Christensen, T.H. Correlating phospholipid fatty acids (PLFA) in a landfill leachate polluted aquifer with biogeochemical factors by multivariate statistical methods. FEMS Microbiol. Rev. 1997, 20, 447–460. [Google Scholar] [CrossRef]
- Green, C.T.; Scow, K.M. Analysis of phospholipid fatty acids (PLFA) to characterize microbial communities in aquifers. Hydrogeol. J. 2000, 8, 126–141. [Google Scholar] [CrossRef]
- de Carvalho, C.C.C.R.; Caramujo, M.-J. Fatty acids as a tool to understand microbial diversity and their role in food webs of Mediterranean temporary ponds. Molecules 2014, 19, 5570–5598. [Google Scholar] [CrossRef] [PubMed]
- Sundh, I.; Nilsson, M.; Borga, P. Variation in microbial community structure in two boreal peatlands as determined by analysis of phospholipid fatty acid profiles. Appl. Environ. Microbiol. 1997, 63, 1476–1482. [Google Scholar] [PubMed]
- Quezada, M.; Buitrón, G.; Moreno-Andrade, I.; Moreno, G.; López-Marín, L.M. The use of fatty acid methyl esters as biomarkers to determine aerobic, facultatively aerobic and anaerobic communities in wastewater treatment systems. FEMS Microbiol. Lett. 2007, 266, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Torres-Ruiz, M.; Wehr, J.D.; Perrone, A.A. Trophic relations in a stream food web: Importance of fatty acids for macroinvertebrate consumers. J. N. Am. Benthol. Soc. 2007, 26, 509–522. [Google Scholar] [CrossRef]
- de Carvalho, C.C.C.R.; Caramujo, M. Lipids of prokaryotic origin at the base of marine food webs. Mar. Drugs 2012, 10, 2698–2714. [Google Scholar] [CrossRef] [PubMed]
- Middelburg, J.J. Stable isotopes dissect aquatic food webs from the top to the bottom. Biogeosciences 2014, 11, 2357–2371. [Google Scholar] [CrossRef]
- De Troch, M.; Boeckx, P.; Cnudde, C.; Van Gansbeke, D.; Vanreusel, A.; Vincx, M.; Caramujo, M.J. Bioconversion of fatty acids at the basis of marine food webs: Insights from a compound-specific stable isotope analysis. Mar. Ecol. Prog. Ser. 2012, 465, 53–67. [Google Scholar] [CrossRef]
- Dijkman, N.A.; Boschker, H.T.S.; Stal, L.J.; Kromkamp, J.C. Composition and heterogeneity of the microbial community in a coastal microbial mat as revealed by the analysis of pigments and phospholipid-derived fatty acids. J. Sea Res. 2010, 63, 62–70. [Google Scholar] [CrossRef]
- Olsson, P.A. Signature fatty acids provide tools for determination of the distribution and interactions of mycorrhizal fungi in soil. FEMS Microbiol. Ecol. 1999, 29, 303–310. [Google Scholar] [CrossRef]
- Costello, A.M.; Auman, A.J.; Macalady, J.L.; Scow, K.M.; Lidstrom, M.E. Estimation of methanotroph abundance in a freshwater lake sediment. Environ. Microbiol. 2002, 4, 443–450. [Google Scholar] [CrossRef] [PubMed]
- Bowman, J.P.; Skerratt, J.H.; Nichols, P.D.; Sly, L.I. Phospholipid fatty acid and lipopolysaccharide fatty acid signature lipids in methane-utilizing bacteria. FEMS Microbiol. Lett. 1991, 85, 15–21. [Google Scholar] [CrossRef]
- Fang, J.; Chan, O.; Joeckel, R.M.; Huang, Y.; Wang, Y.; Bazylinski, D.A.; Moorman, T.B.; Ang Clement, B.J. Biomarker analysis of microbial diversity in sediments of a saline groundwater seep of Salt Basin, Nebraska. Org. Geochem. 2006, 37, 912–931. [Google Scholar] [CrossRef]
- Elvert, M.; Boetius, A.; Knittel, K.; Jørgensen, B.B. Characterization of specific membrane fatty acids as chemotaxonomic markers for sulfate-reducing bacteria involved in anaerobic oxidation of methane. Geomicrobiol. J. 2003, 20, 403–419. [Google Scholar] [CrossRef]
- Zelles, L. Identification of single cultured micro-organisms based on their whole-community fatty acid profiles, using an extended extraction procedure. Chemosphere 1999, 39, 665–682. [Google Scholar] [CrossRef]
- Lee, A.K.Y.; Chan, C.K.; Fang, M.; Lau, A.P.S. The 3-hydroxy fatty acids as biomarkers for quantification and characterization of endotoxins and Gram-negative bacteria in atmospheric aerosols in Hong Kong. Atmos. Environ. 2004, 38, 6307–6317. [Google Scholar] [CrossRef]
- Frostegård, A.; Bååth, E. The use of phospholipid fatty acid analysis to estimate bacterial and fungal biomass in soil. Biol. Fertil. Soils 1996, 22, 59–65. [Google Scholar] [CrossRef]
- Fernandes, M.F.; Saxena, J.; Dick, R.P. Comparison of whole-cell fatty acid (MIDI) or phospholipid fatty acid (PLFA) extractants as biomarkers to profile soil microbial communities. Microb. Ecol. 2013, 66, 145–157. [Google Scholar] [CrossRef] [PubMed]
- Rajendran, N.; Matsuda, O.; Imamura, N.; Urushigawa, Y. Microbial community structure analysis of euxinic sediments using phospholipid fatty acid biomarkers. J. Oceanogr. 1995, 51, 21–38. [Google Scholar] [CrossRef]
- Jiang, L.; Cai, C.; Zhang, Y.; Mao, S.; Sun, Y.; Li, K.; Xiang, L.; Zhang, C. Lipids of sulfate-reducing bacteria and sulfur-oxidizing bacteria found in the Dongsheng uranium deposit. Chin. Sci. Bull. 2012, 57, 1311–1319. [Google Scholar] [CrossRef]
- Frostegård, Å.; Tunlid, A.; Bååth, E. Phospholipid fatty acid composition, biomass and activity of microbial communities from two soil types experimentally exposed to different heavy metals. Appl. Environ. Microbiol. 1993, 59, 3605–3617. [Google Scholar] [PubMed]
- White, D.C.; Geyer, R.; Peacock, A.D.; Hedrick, D.B.; Koenigsberg, S.S.; Sung, Y.; He, J.; Löffler, F.E. Phospholipid furan fatty acids and ubiquinone-8: Lipid biomarkers that may protect Dehalococcoides strains from free radicals. Appl. Environ. Microbiol. 2005, 71, 8426–8433. [Google Scholar] [CrossRef] [PubMed]
- Stahl, P.D.; Klug, M.J. Characterization and differentiation of filamentous fungi based on fatty acid composition. Appl. Environ. Microbiol. 1996, 62, 4136–4146. [Google Scholar] [PubMed]
- Sud, M.; Fahy, E.; Cotter, D.; Brown, A.; Dennis, E.; Glass, C.; Murphy, R.; Raetz, C.; Russell, D.; Subramaniam, S. LIPID MAPS structure database. Nucleic Acids Res. 2006, 35, D527–D532. [Google Scholar] [CrossRef] [PubMed]
| Lipids | Example | Role | Specificities |
|---|---|---|---|
| Fatty acids | 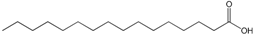 16:0 | Building blocks for numerous lipids, regulation of membrane fluidity | - |
| Glycerophospholipids or phospholipids |  PE (16:0/18:0) | Main constituent of cellular membranes in prokaryotes and eukaryotes | Two FA linked to a glycerol molecule connected to a phosphate head group |
| Glycerophospholipids plasmalogens | 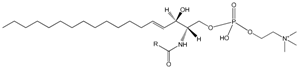 Sphingomyelin | Organization and stability of membranes; cellular signalling | Contain a vinyl-ether and an ester bond at the glycerol backbone |
| Glycerophospholipids sphingolipids | Sphingomyelin | Role in cell division, differentiation and cell death | Long-chain or sphingoid base linked to a FA via an amide bond |
| Glycerolipids | 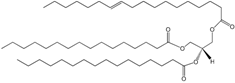 TG (16:0/16:0/18:1) | Storage compounds in prokaryotes | Mono-, di-, or tri-substituted glycerols |
| Ether-linked lipids such as glycerol dibiphytanyl glycerol tetraethers | 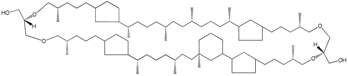 Crenarchaeol | Core cellular membrane lipids in many archaea | Isoprenoid moieties linked by ether bonds to glycerol |
| Wax esters | 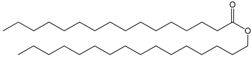 WE(16:0/16:0) | Energy storage and cell structure | Ester of FA and a fatty alcohol |
| Sterol lipids | 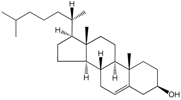 Cholesterol | Maintenance of membrane fluidity | Steroids with a hydroxyl group at the 3-position of the A-ring |
| FA Modification | Effect | Microorganism | Reference |
|---|---|---|---|
| fatty acyl chain length | growth at different temperatures | Micrococcus cryophilus, Shewanella oneidensis, Escherichia coli | [49,50,51] |
| growth in deep-sea | barophilic bacteria | [52] | |
| adaptation to the presence of organic compounds | Rhodococcus erythropolis | [53] | |
| unsaturation | growth at different temperatures, pH, pressure, salinity, in the presence of organic solvents | archaea and bacteria | [43,54,55] |
| polyunsaturation | growth in deep-sea | Alteromonas sp., Shewanella sp. | [56,57,58] |
| rapid adaptation to increased salinity and extreme conditions | Rhodococcus erythropolis | [59,60] | |
| cis-trans isomerization | fast adaptation to environmental conditions when growth is inhibited | Pseudomonas and Vibrio | [61,62] |
| branching at iso or anteiso position | growth at different temperatures | Listeria monocytogenes | [63] |
| persistence to high concentrations of antibiotics | Staphylococcus aureus | [64] | |
| growth temperature and presence of phenols | Arthrobacter chlorophenolicus | [65] | |
| cyclopropanation | persistence and virulence of the cells | Mycobacterium tuberculosis | [66] |
| osmotic tolerance | Pseudomunas halosaccharolytic | [67] | |
| growth at different temperature and pH | Rhodococcus erythropolis, Salmonella typhimurium | [60,68] | |
| polyunsaturated fatty acids associated to phosphatidylglycerol | growth in the Marianas Trench at 11,000 m | Barophilic bacteria DB21MT-2 and DB21MT-5 | [51] |
| composition of the alkyl and acyl chains in glycerol ether lipids | growth at different temperatures | Desulfatibacillum aliphaticivorans, D. alkenivorans, Thermodesulfobacterium commune | [69] |
| Fatty Acid | Category | Reference |
|---|---|---|
| Mono-Unsaturated Fatty Acids (MUFA) | ||
| 16:1ω7c | Bacteria Bacillariophyceae (diatoms) Cyanophyceae (cyanobacteria) Prymnesiophyceae | [245,274,292,305] |
| 16:1ω5c | mycorrhizal fungi | [306] |
| 16:1ω8c | Type I methanotrophs (gamma-proteobacteria) | [307,308] |
| 17:1 | Cyanobacteria | [309] |
| 17:1ω6c (up to 60%) | Desulfobulbus, Desulforhabdus, Desulforhopalus (sulphate reducing bacteria) | [310] |
| 18:1ω9c | Chlorophyceae (green algae) Cyanophyceae Dinophyceae Prymnesiophyceae Gram-positive bacteria | [245,274,292,305,311] |
| 18:1ω7c | Bacillariophyceae (up to 10-fold more 18:1ω7c than 18:1ω9c) Cryptophyceae Cyanophyceae (less amount than 18:1ω9c) Prymnesiophyceae | [245,274,292] |
| 18:1ω7t | Gram-negative bacteria | [311] |
| 18:1ω8c | Type II methanotrophs (alpha-proteobacteria) | [307,308] |
| Hydroxy substituted Fatty Acids (OH FA) | ||
| (e.g., 3-OH 10:0) | Gram-negative bacteria | [312] |
| Cyclopropyl saturated Fatty Acids (cyFA) | ||
| (e.g., cyclo17:0, cyclo19:0) | Gram-negative bacteria, anaerobic bacteria | [313,314] |
| Cyclo17:0ω5,6 | Desulfosarcina/Desulfococcus (sulphate reducing bacteria) | [310] |
| Iso- and anteiso-branched Fatty Acids | ||
| (e.g., iso-15:0, anteiso-17:0) iso-17:1ω7c | Gram-positive bacteria Sulphate reducing bacteria Desulfovibrio sp. (sulphate reducing bacteria) | [313,314,315,316] |
| Methyl-branched Fatty Acids (10-Me FA) | ||
| 10-Me 18:0 10-Me 16:0 2-Me 17:0 | Actinomycetales (Actinobacteria) Desulfobacter sp.- sulphate reducing bacteria Cyanobacteria | [309,315,316,317] |
| Furan Fatty Acids | ||
| Fu18:2ω6, Fu17:2 ω5 and ω6 | Dehalococcoides sp. | [318] |
| Polyunsaturated Fatty Acids (PUFA) | ||
| 16:2ω7 | Bacillariophyceae | [245] |
| 16:2ω6 | Chlorophyta | [245] |
| 16:2ω4 | Bacillariophyceae Prasinophyceae | [245,279] |
| 16:3ω4 | Bacillariophyceae | [245,279] |
| 16:3ω3 | Chlorophyta | [245] |
| 16:4ω3 | Chlorophyceae Prasinophyceae | [245] |
| 16:4ω1 | Bacillariophyceae (diatoms) | [245,279] |
| 18:2ω6 | Chlorophyta Cyanophyceae (freshwater) Dinophyceae Prymnesiophyceae Fungi | [245,274,292,305,313,314] |
| 18:3ω6 | Cyanophyceae (freshwater) Saprophytic fungi | [274,314,319] |
| 18:3ω3 | Chlorophyceae Crypophyceae Cyanophyceae Dinophyceae Prasinophyceae Prymnesiophyceae | [245,274,292] |
| 18:4ω3 | Most groups (both marine and freshwater) | [245,274,292] |
| 18:5ω3 | Dynophyceae | [245] |
| 20:4ω6 | Bacillariophyceae Rhodophyceae | [245] |
| 20:5ω3 | Bacillariophyceae Cryptophyceae Dinophyceae Pavlovophyceae Rhodophyceae | [245,292] |
| 22:5ω3 | Bacillariophyceae Cryptophyceae Prasinophyceae | [245] |
| 22:6ω3 | Bacillariophyceae Cryptophyceae Dinophyceae Haptophyta (Prymnesiophyceae and Pavlovophyceae) | [245,274,292] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Carvalho, C.C.C.R.; Caramujo, M.J. The Various Roles of Fatty Acids. Molecules 2018, 23, 2583. https://doi.org/10.3390/molecules23102583
De Carvalho CCCR, Caramujo MJ. The Various Roles of Fatty Acids. Molecules. 2018; 23(10):2583. https://doi.org/10.3390/molecules23102583
Chicago/Turabian StyleDe Carvalho, Carla C. C. R., and Maria José Caramujo. 2018. "The Various Roles of Fatty Acids" Molecules 23, no. 10: 2583. https://doi.org/10.3390/molecules23102583
APA StyleDe Carvalho, C. C. C. R., & Caramujo, M. J. (2018). The Various Roles of Fatty Acids. Molecules, 23(10), 2583. https://doi.org/10.3390/molecules23102583






