Biotinylated Cyclooligosaccharides for Paclitaxel Solubilization
Abstract
1. Introduction
2. Results and Discussion
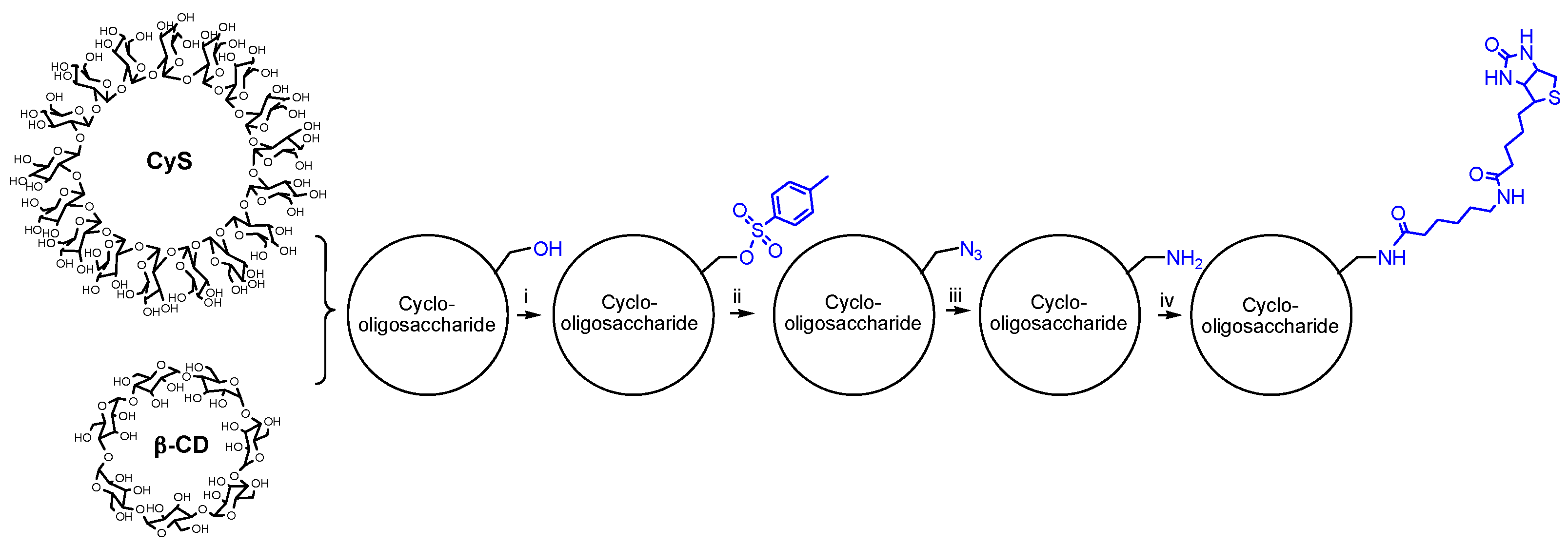
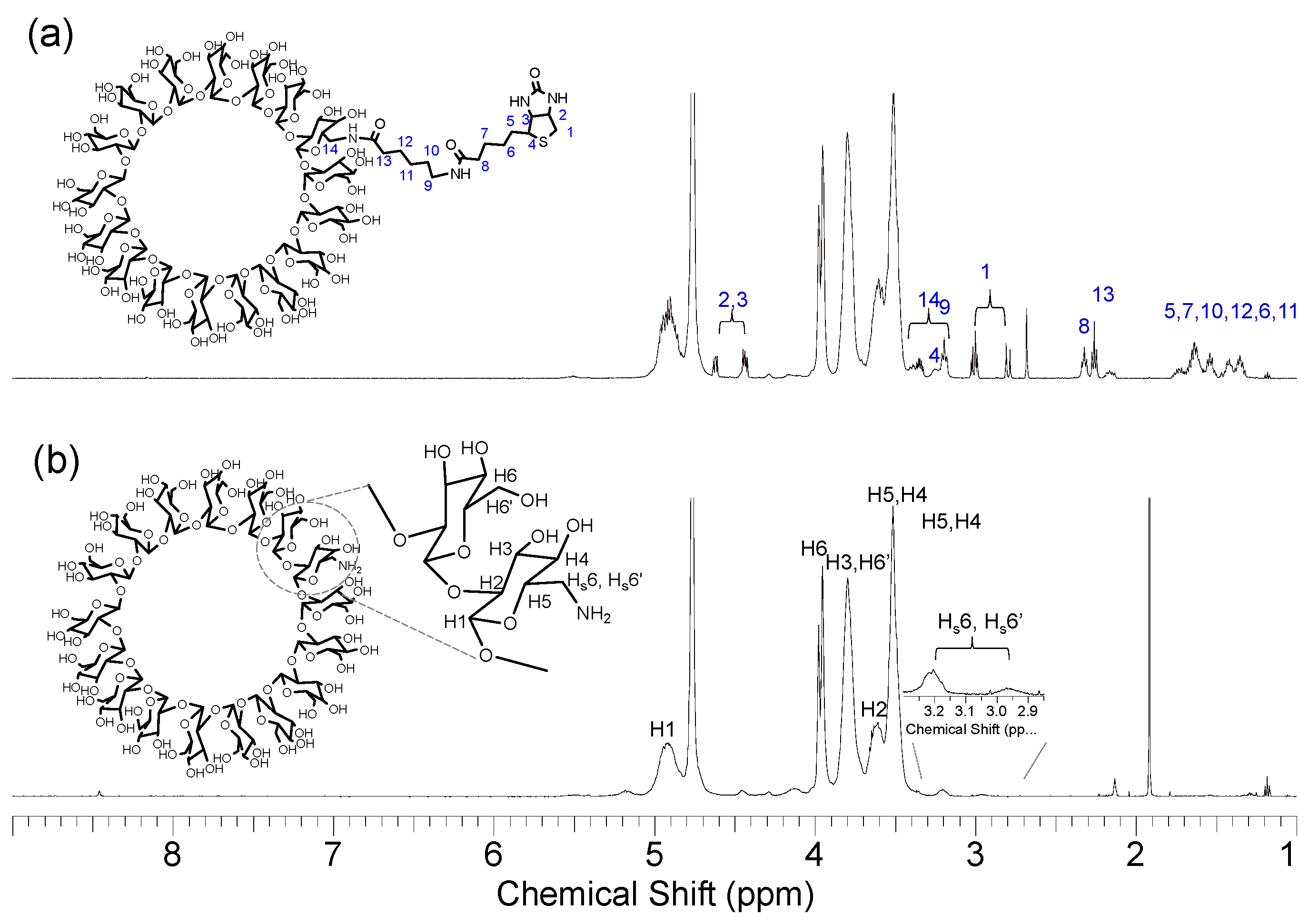
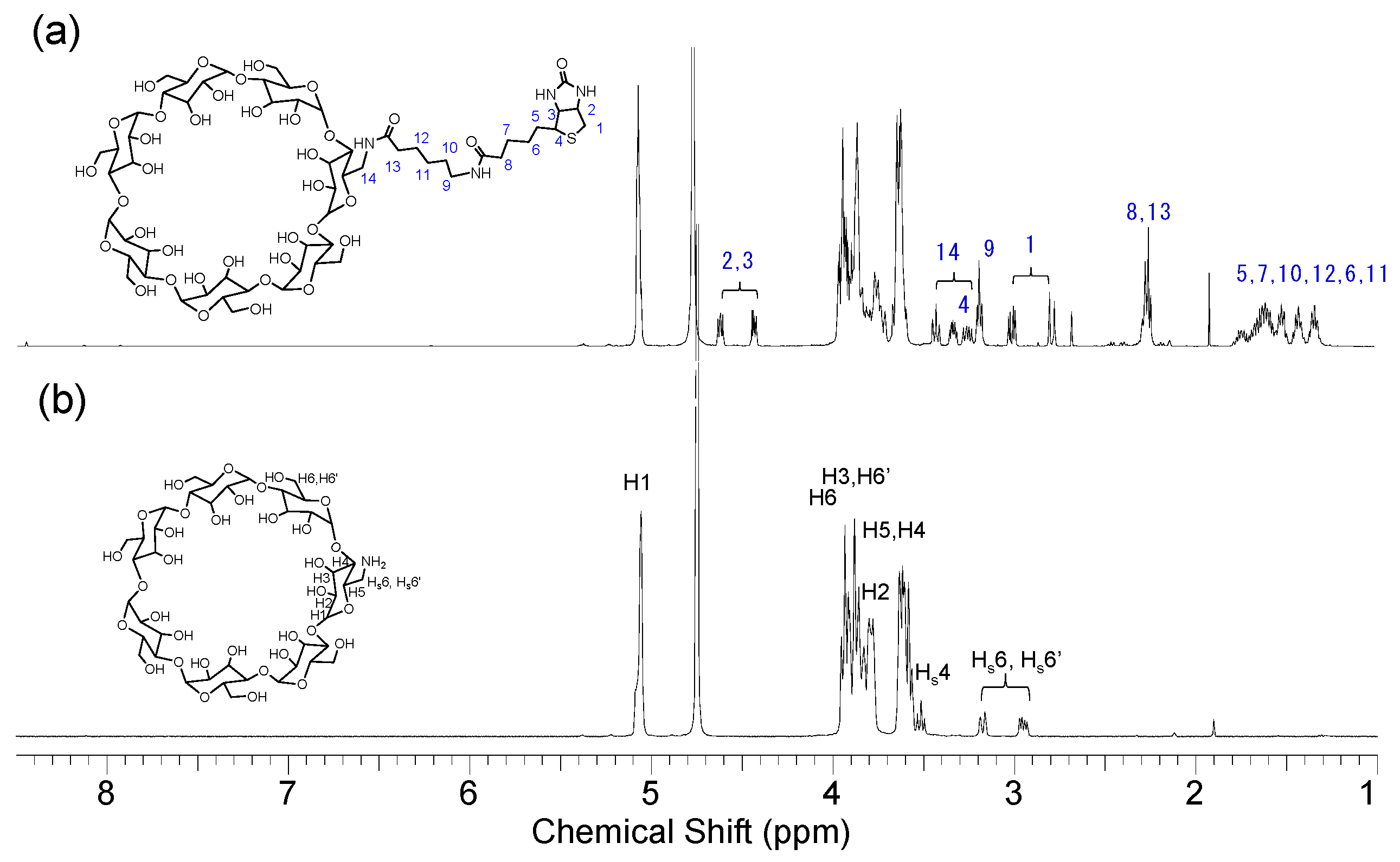
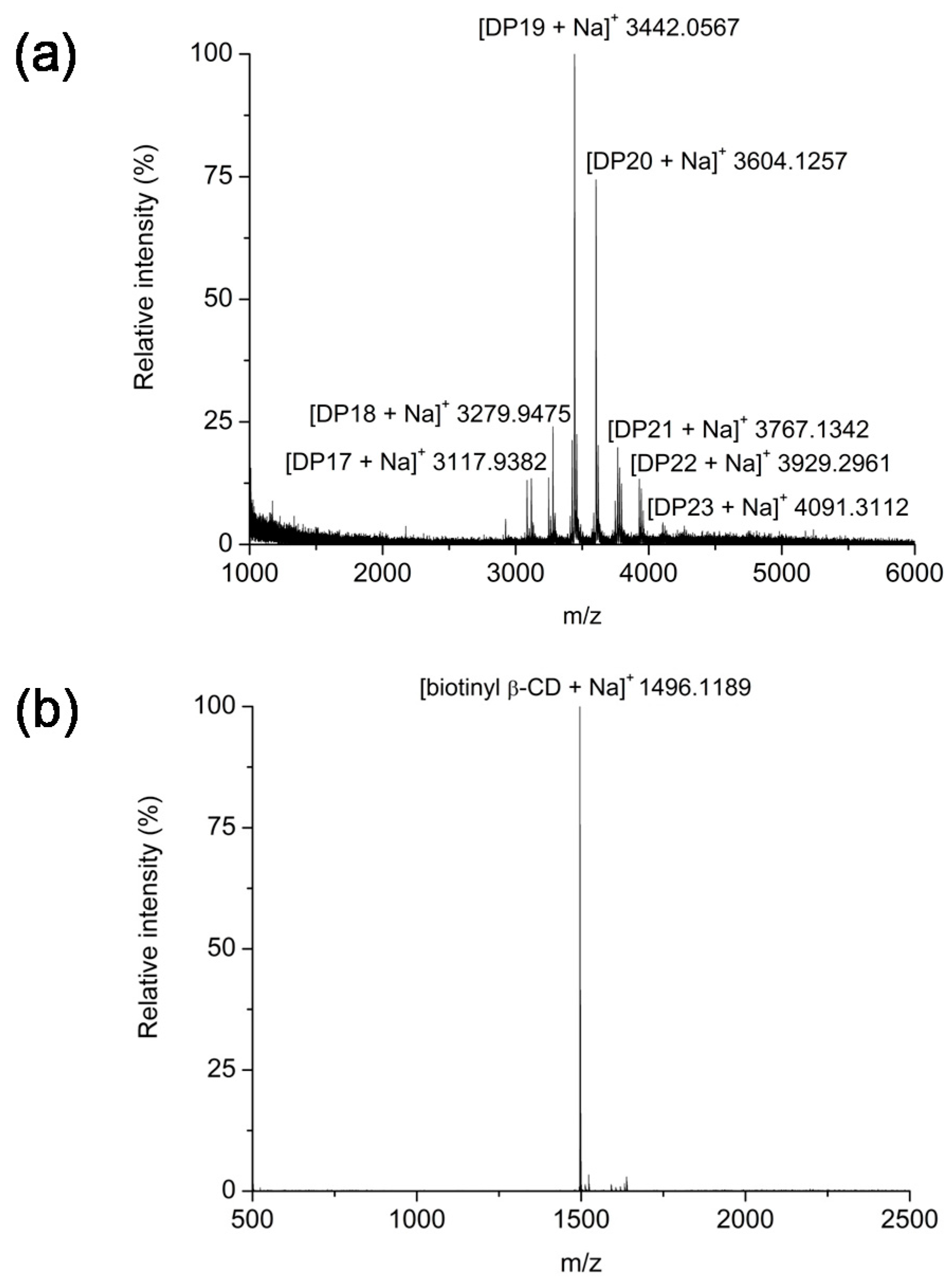
2.1. Synthesis and Structural Analyses of Biotinyl Cyclooligosaccharides
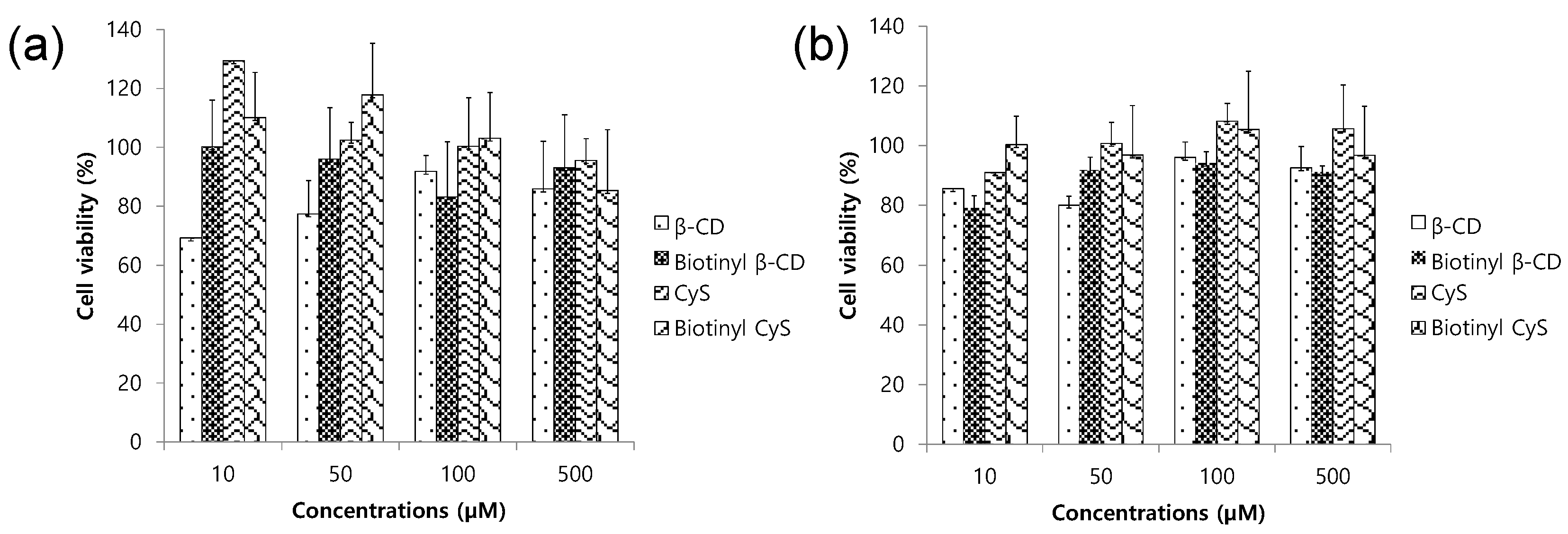
2.2. Biocompatibility of Cyclooligosaccharides and the Biotinyl Derivatives
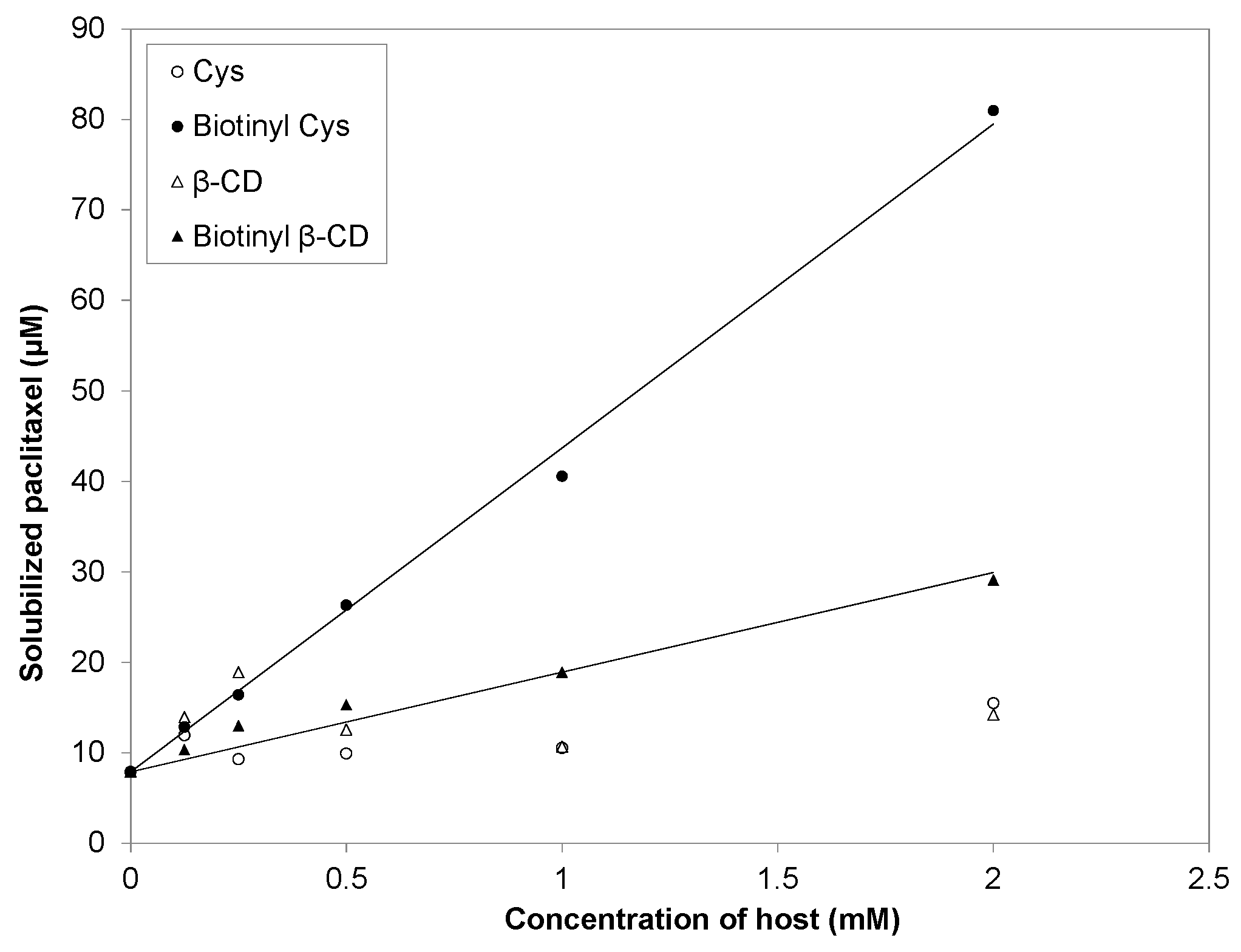
2.3. Phase Solubility Study
2.4. 1H NMR Analysis
3. Materials and Methods
3.1. Materials
3.2. Isolation and Purification of CyS
3.3. Synthesis of Biotinyl Cyclooligosaccharides
3.4. MALDI-TOF MS and NMR Spectroscopic Analysis
3.5. Cytotoxicity Assay
3.6. Phase Solubility Studies
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Nicolaou, K.C.; Dai, W.M.; Guy, R.K. Chemistry and biology of taxol. Angew. Chem. Int. Ed. 1994, 33, 15–44. [Google Scholar] [CrossRef]
- Jordan, M. Mechanism of action of antitumor drugs that interact with microtubules and tubulin. Curr. Med. Chem. AntiCancer Agents 2002, 2, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Suffness, M. Overview of Paclitaxel Research: Progress on Many Fronts; ACS Publications: Washington, DC, USA, 1995. [Google Scholar]
- Goldspiel, B.R. Clinical overview of the taxanes. Pharmacother. J. Hum. Pharmacol. Drug Ther. 1997, 17, 110S–125S. [Google Scholar]
- Lipinski, C. Poor aqueous solubility—An industry wide problem in drug discovery. Am. Pharm. Rev. 2002, 5, 82–85. [Google Scholar]
- Merisko-Liversidge, E.; Liversidge, G.G.; Cooper, E.R. Nanosizing: A formulation approach for poorly-water-soluble compounds. Eur. J. Pharm. Sci. 2003, 18, 113–120. [Google Scholar] [CrossRef]
- Zhang, J.A.; Anyarambhatla, G.; Ma, L.; Ugwu, S.; Xuan, T.; Sardone, T.; Ahmad, I. Development and characterization of a novel cremophor® el free liposome-based paclitaxel (lep-etu) formulation. Eur. J. Pharm. Biopharm. 2005, 59, 177–187. [Google Scholar] [CrossRef] [PubMed]
- Kingston, D.G.; Zhao, Z.-Y. Water Soluble Derivatives of Taxol. U.S. Patent 5,352,805 A, 4 October 1994. [Google Scholar]
- Gelderblom, H.; Verweij, J.; Nooter, K.; Sparreboom, A. Cremophor El: The drawbacks and advantages of vehicle selection for drug formulation. Eur. J. Cancer 2001, 37, 1590–1598. [Google Scholar] [CrossRef]
- Cho, E.; Jung, S. Supramolecular complexation of carbohydrates for the bioavailability enhancement of poorly soluble drugs. Molecules 2015, 20, 19620–19646. [Google Scholar] [CrossRef] [PubMed]
- Davis, M.E.; Brewster, M.E. Cyclodextrin-based pharmaceutics: Past, present and future. Nat. Rev. Drug Discov. 2004, 3, 1023–1035. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Seo, D.-H.; Park, H.-L.; Choi, Y.; Jung, S. Solubility enhancement of a hydrophobic flavonoid, luteolin by the complexation with cyclosophoraoses isolated from rhizobium meliloti. Antonie Van Leeuwenhoek 2003, 84, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Salústio, P.; Cabral-Marques, H.; Costa, P.; Pinto, J. Comparison of ibuprofen release from minitablets and capsules containing ibuprofen: Β-cyclodextrin complex. Eur. J. Pharm. Biopharm. 2011, 78, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Cartledge, J.; Midgley, J.; Youle, M.; Gazzard, B. Itraconazole cyclodextrin solution—Effective treatment for hiv-related candidosis unresponsive to other azole therapy. J. Antimicrob. Chemother. 1994, 33, 1071–1073. [Google Scholar] [CrossRef] [PubMed]
- Ficarra, R.; Ficarra, P.; Di Bella, M.; Raneri, D.; Tommasini, S.; Calabro, M.; Villari, A.; Coppolino, S. Study of the inclusion complex of atenolol with β-cyclodextrins. J. Pharm. Biomed. Anal. 2000, 23, 231–236. [Google Scholar] [CrossRef]
- Amemura, A. Synthesis of (1→2)-β-d-glucan by cell-free extracts of agrobacterium radiobacter ifo 12665b 1 and rhizobium phaseoli ahu 1133. Agric. Biol. Chem. 1984, 48, 1809–1817. [Google Scholar]
- André, I.; Mazeau, K.; Taravel, F.R.; Tvaroska, I. Conformation and dynamics of a cyclic (1→2)-β-d-glucan. Int. J. Biol. Macromol. 1995, 17, 189–198. [Google Scholar] [CrossRef]
- Koizumi, K.; Okada, Y.; Horiyama, S.; Utamura, T.; Higashiura, T.; Ikeda, M. Preparation of cyclosophoraose-a and its complex-forming ability. In Clathrate Compounds, Molecular Inclusion Phenomena, and Cyclodextrins; Springer: Dordrecht, The Netherlands, 1984; pp. 891–899. [Google Scholar]
- Kwon, C.; Choi, Y.-H.; Kim, N.; Yoo, J.S.; Yang, C.-H.; Kim, H.-W.; Jung, S. Complex forming ability of a family of isolated cyclosophoraoses with ergosterol and its monte carlo docking computational analysis. J. Incl. Phenom. Macrocycl. Chem. 2000, 36, 55–64. [Google Scholar] [CrossRef]
- Szente, L.; Szejtli, J. Highly soluble cyclodextrin derivatives: Chemistry, properties, and trends in development. Adv. Drug Deliv. Rev. 1999, 36, 17–28. [Google Scholar] [CrossRef]
- Bender, M.L.; Komiyama, M. Cyclodextrin Chemistry; Springer: Heidelberg/Berlin, Germany, 2012; Volume 6. [Google Scholar]
- Casu, B.; Reggiani, M.; Gallo, G.; Vigevani, A. Conformation of O-methylated amylose and cyclodextrins. Tetrahedron 1968, 24, 803–821. [Google Scholar] [CrossRef]
- Uekama, K.; Minami, K.; Hirayama, F. 6A-O-[(4-biphenylyl) acetyl]-α-,-β-, and-γ-cyclodextrins and 6A-deoxy-6A-[[(4-biphenylyl) acetyl] amino]-α-,-β-, and-γ-cyclodextrins: Potential prodrugs for colon-specific delivery. J. Med. Chem. 1997, 40, 2755–2761. [Google Scholar] [CrossRef] [PubMed]
- Karunaratne, D.N.; Farmer, S.; Hancock, R.E. Synthesis of bulky. Beta.-lactams for inhibition of cell surface. Beta.-lactamase activity. Bioconjug. Chem. 1993, 4, 434–439. [Google Scholar] [CrossRef] [PubMed]
- Russell-Jones, G.; McTavish, K.; McEwan, J.; Rice, J.; Nowotnik, D. Vitamin-mediated targeting as a potential mechanism to increase drug uptake by tumours. J. Inorg. Biochem. 2004, 98, 1625–1633. [Google Scholar] [CrossRef] [PubMed]
- Dordunoo, S.K.; Burt, H.M. Solubility and stability of taxol: Effects of buffers and cyclodextrins. Int. J. Pharm. 1996, 133, 191–201. [Google Scholar] [CrossRef]
- Hamada, H.; Ishihara, K.; Masuoka, N.; Mikuni, K.; Nakajima, N. Enhancement of water-solubility and bioactivity of paclitaxel using modified cyclodextrins. J. Biosci. Bioeng. 2006, 102, 369–371. [Google Scholar] [CrossRef] [PubMed]
- Surapaneni, M.S.; Das, S.K.; Das, N.G. Designing paclitaxel drug delivery systems aimed at improved patient outcomes: Current status and challenges. ISRN Pharmacol. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Jing, J.; Szarpak-Jankowska, A.; Guillot, R.; Pignot-Paintrand, I.; Picart, C.; Auzély-Velty, R. Cyclodextrin/paclitaxel complex in biodegradable capsules for breast cancer treatment. Chem. Mater. 2013, 25, 3867–3873. [Google Scholar] [CrossRef]
- Brunner, H.; Hankofer, P.; Holzinger, U.; Treittinger, B.; Schönenberger, H. Synthesis and antitumor activity of platinum (ii) complexes containing substituted ethylenediamine ligands. Eur. J. Med. Chem. 1990, 25, 35–44. [Google Scholar] [CrossRef]
- Lukin, O.; Dolgonos, G.; Leszczynski, J. A comprehensive test of computational approaches for evaluation of cyclodextrin complexes. Self-inclusion in monosubstituted β-cyclodextrins—A case study. Tetrahedron 2017, 73, 5302–5306. [Google Scholar] [CrossRef]
- Zhang, X.; Scialis, R.J.; Feng, B.; Leach, K. Detection of statin cytotoxicity is increased in cells expressing the oatp1b1 transporter. Toxicol. Sci. 2013, 134, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Samperio, C.; Boyer, R.; Eigel, W.N., III; Holland, K.W.; McKinney, J.S.; O’Keefe, S.F.; Smith, R.; Marcy, J.E. Enhancement of plant essential oils’ aqueous solubility and stability using alpha and beta cyclodextrin. J. Agric. Food. Chem. 2010, 58, 12950–12956. [Google Scholar] [CrossRef] [PubMed]
- International Organization for Standardization. Biological Evaluation of Medical Devices—Part 5: Tests for In Vitro Cytotoxicity; ISO 10993-5: 2009; International Organization for Standardization: Geneva, Sweden, 2009. [Google Scholar]
- Higuchi, T.; Connors, A. Phase-solubility techniques. Adv. Anal. Chem. Instrum. 1965, 4, 117–212. [Google Scholar]
- Loftsson, T.; Magnúsdóttir, A.; Másson, M.; Sigurjónsdóttir, J.F. Self-association and cyclodextrin solubilization of drugs. J. Pharm. Sci. 2002, 91, 2307–2316. [Google Scholar] [CrossRef] [PubMed]
- Dindulkar, S.D.; Jeong, D.; Kim, H.; Jung, S. Functionalized β-cyclodextrin as supramolecular ligand and their pd (OAc)2 complex: Highly efficient and reusable catalyst for mizoroki-heck cross-coupling reactions in aqueous medium. Carbohydr. Res. 2016, 430, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Guo, Q.-X. The driving forces in the inclusion complexation of cyclodextrins. J. Incl. Phenom. Macrocycl. Chem. 2002, 42, 1–14. [Google Scholar] [CrossRef]
- Zhao, L.; Feng, S.-S. Effects of cholesterol component on molecular interactions between paclitaxel and phospholipid within the lipid monolayer at the air—Water interface. J. Colloid Interface Sci. 2006, 300, 314–326. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Kim, H.; Shinde, V.V.; Jeong, D.; Choi, Y.; Cho, E.; Jung, S. Carboxymethyl cyclosophoraoses as a flexible ph-responsive solubilizer for pindolol. Carbohydr. Polym. 2017, 175, 493–501. [Google Scholar] [CrossRef] [PubMed]
- Schneider, H.-J.; Hacket, F.; Rüdiger, V.; Ikeda, H. Nmr studies of cyclodextrins and cyclodextrin complexes. Chem. Rev. 1998, 98, 1755–1786. [Google Scholar] [CrossRef] [PubMed]
- Jeong, D.; Kim, H.K.; Jeong, J.-p.; Dindulkar, S.D.; Cho, E.; Yang, Y.-H.; Jung, S. Cyclosophoraose/cellulose hydrogels as an efficient delivery system for galangin, a hydrophobic antibacterial drug. Cellulose 2016, 23, 2609–2625. [Google Scholar] [CrossRef]
- Kim, H.; Dindulkar, S.D.; Jeong, D.; Park, S.; Jun, B.-H.; Cho, E.; Jung, S. A synthetic encapsulating emulsifier using complex-forming pentacosadiynoyl cyclosophoraoses (cyclic β-(1,2)-d-glucan). J. Ind. Eng. Chem. 2016, 44, 195–203. [Google Scholar] [CrossRef]
- Cho, E.; Kim, H.; Yang, J.E.; Jun, B.-H.; Paik, S.R.; Jung, S. Supramolecular self-assembled aggregates formed by pentacosa-10,12-diynyl amidomethyl-β-cyclodextrin. Carbohydr. Res. 2014, 391, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Scudiero, D.A.; Shoemaker, R.H.; Paull, K.D.; Monks, A.; Tierney, S.; Nofziger, T.H.; Currens, M.J.; Seniff, D.; Boyd, M.R. Evaluation of a soluble tetrazolium/formazan assay for cell growth and drug sensitivity in culture using human and other tumor cell lines. Cancer Res. 1988, 48, 4827–4833. [Google Scholar] [PubMed]
- Kathiravan, G.; Sureban, S.M.; Sree, H.N.; Bhuvaneshwari, V.; Kramony, E. Isolation of anticancer drug taxol from pestalotiopsis breviseta with apoptosis and b-cell lymphoma protein docking studies. J. Basic Clin. Pharm. 2012, 4, 14–19. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Not available. |
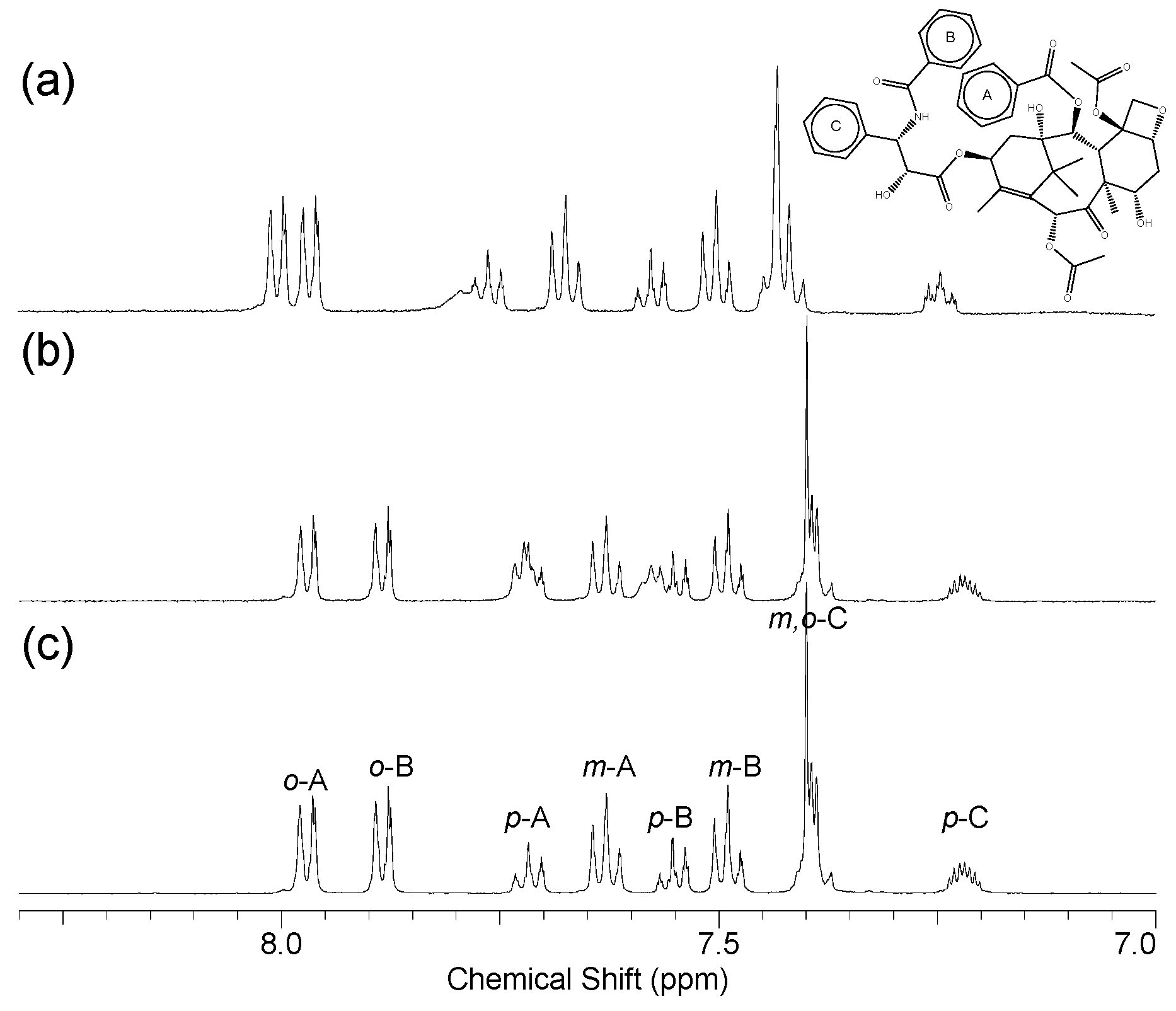
| Paclitaxel | Biotinyl CyS (ppm) | Biotinyl β-CD (ppm) | |||
|---|---|---|---|---|---|
| Protons | δ b | δ a | Δδ | δ a | Δδ |
| o-A | 7.97 | 8.00 | 0.03 | 7.97 | 0 |
| m-A | 7.63 | 7.67 | 0.04 | 7.63 | 0 |
| p-A | 7.72 | 7.76 (7.80) c | 0.04 | 7.72 d | 0 |
| o-B | 7.88 | 7.96 | 0.08 | 7.88 | 0 |
| m-B | 7.49 | 7.50 | 0.01 | 7.49 | 0 |
| p-B | 7.55 | 7.58 | 0.03 | 7.55 (7.57) c | 0 |
| o-C | 7.39 | 7.42 d | 0.03 | 7.39 | 0 |
| m-C | 7.40 | 7.43 d | 0.03 | 7.40 | 0 |
| p-C | 7.22 | 7.25 d | 0.03 | 7.22 | 0 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cho, E.; Jung, S. Biotinylated Cyclooligosaccharides for Paclitaxel Solubilization. Molecules 2018, 23, 90. https://doi.org/10.3390/molecules23010090
Cho E, Jung S. Biotinylated Cyclooligosaccharides for Paclitaxel Solubilization. Molecules. 2018; 23(1):90. https://doi.org/10.3390/molecules23010090
Chicago/Turabian StyleCho, Eunae, and Seunho Jung. 2018. "Biotinylated Cyclooligosaccharides for Paclitaxel Solubilization" Molecules 23, no. 1: 90. https://doi.org/10.3390/molecules23010090
APA StyleCho, E., & Jung, S. (2018). Biotinylated Cyclooligosaccharides for Paclitaxel Solubilization. Molecules, 23(1), 90. https://doi.org/10.3390/molecules23010090






