Amyloid Biomarkers in Conformational Diseases at Face Value: A Systematic Review
Abstract
:1. Introduction
2. Results
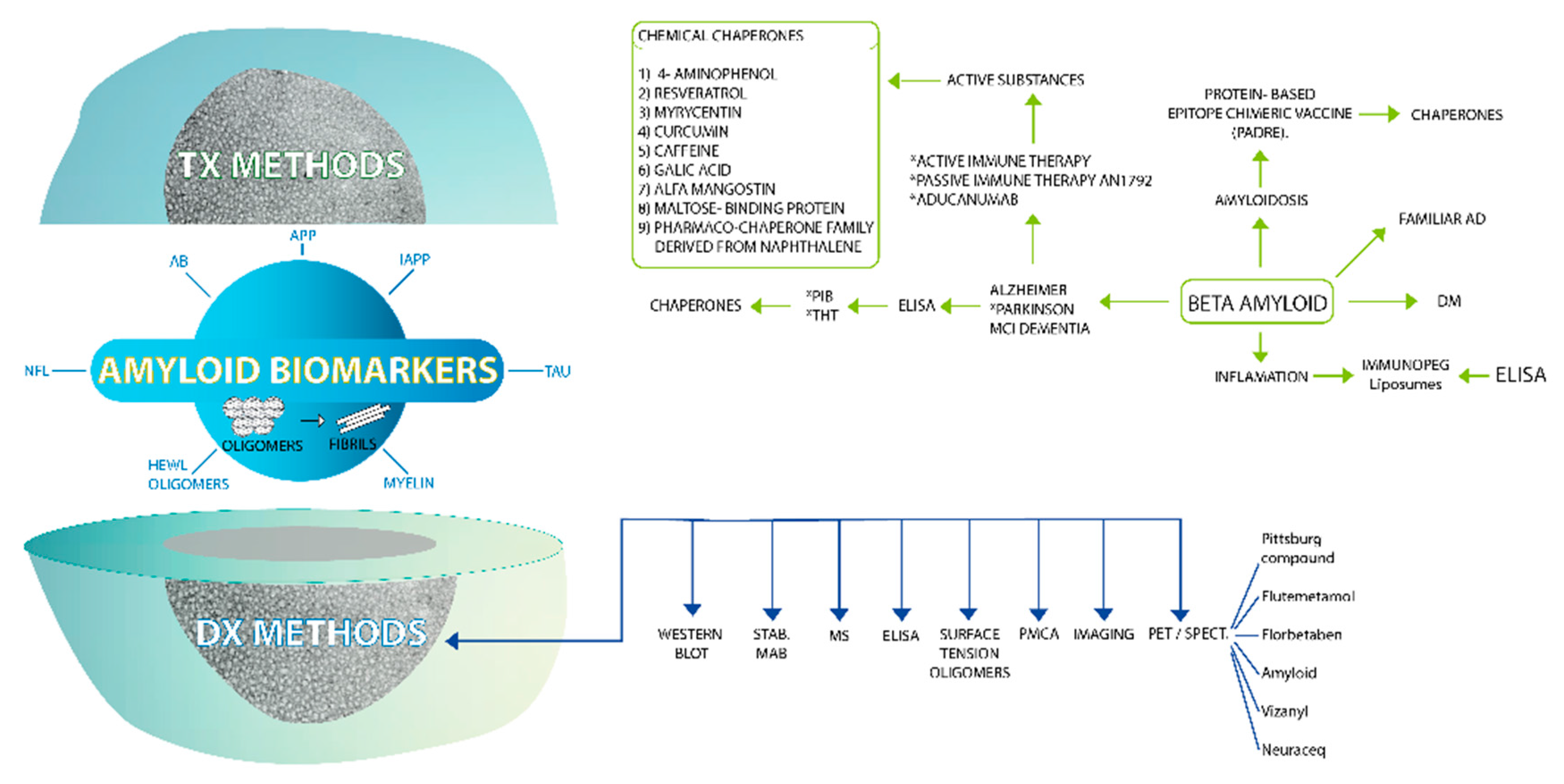
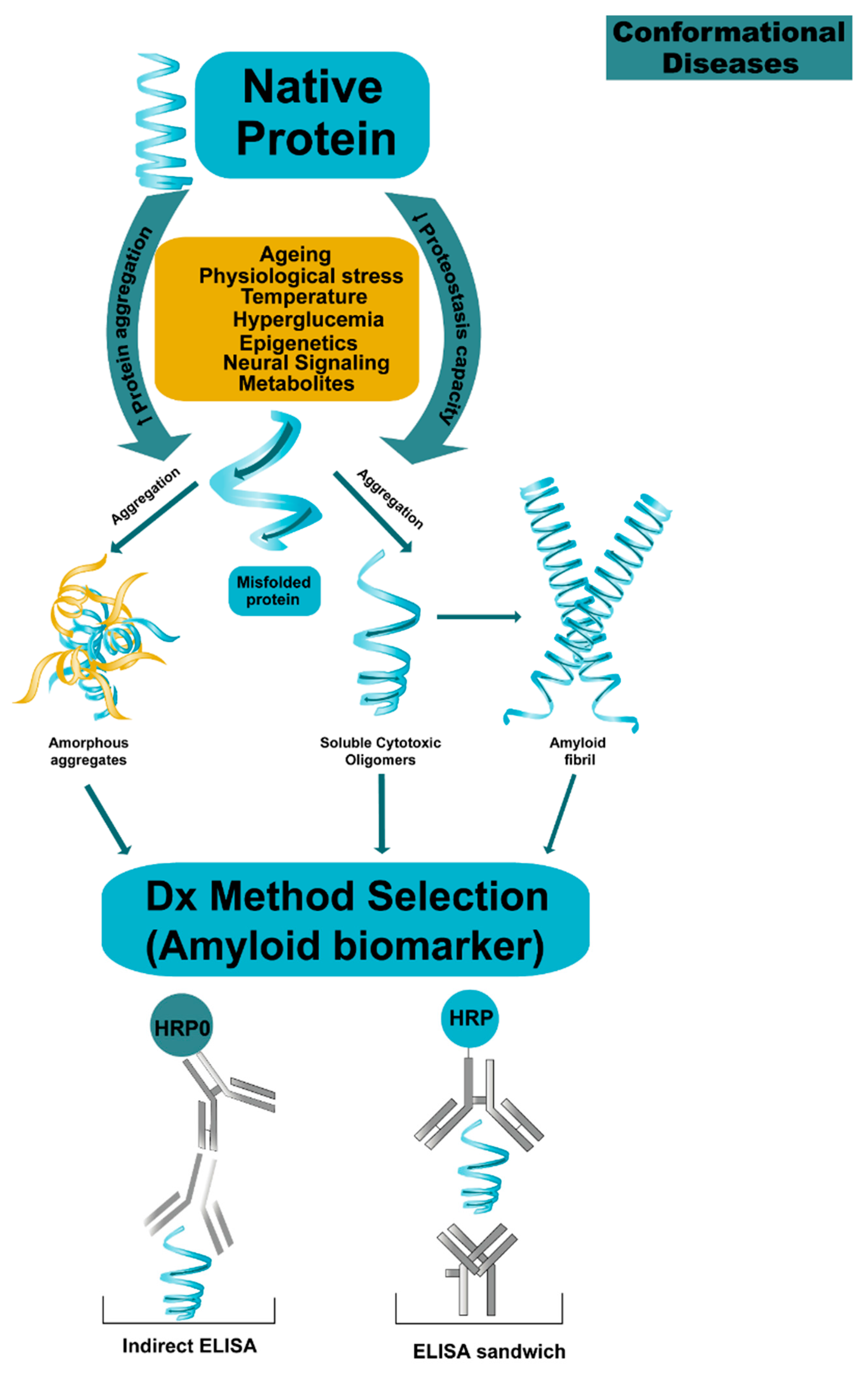
2.1. The Biomarker Dilemma
2.2. Technique Challenges and Their Evolving Solutions
2.3. Polymorphic Amyloid Structures Implications
2.4. Multi-Target Therapeutic Approaches
3. Discussion
4. Materials and Methods
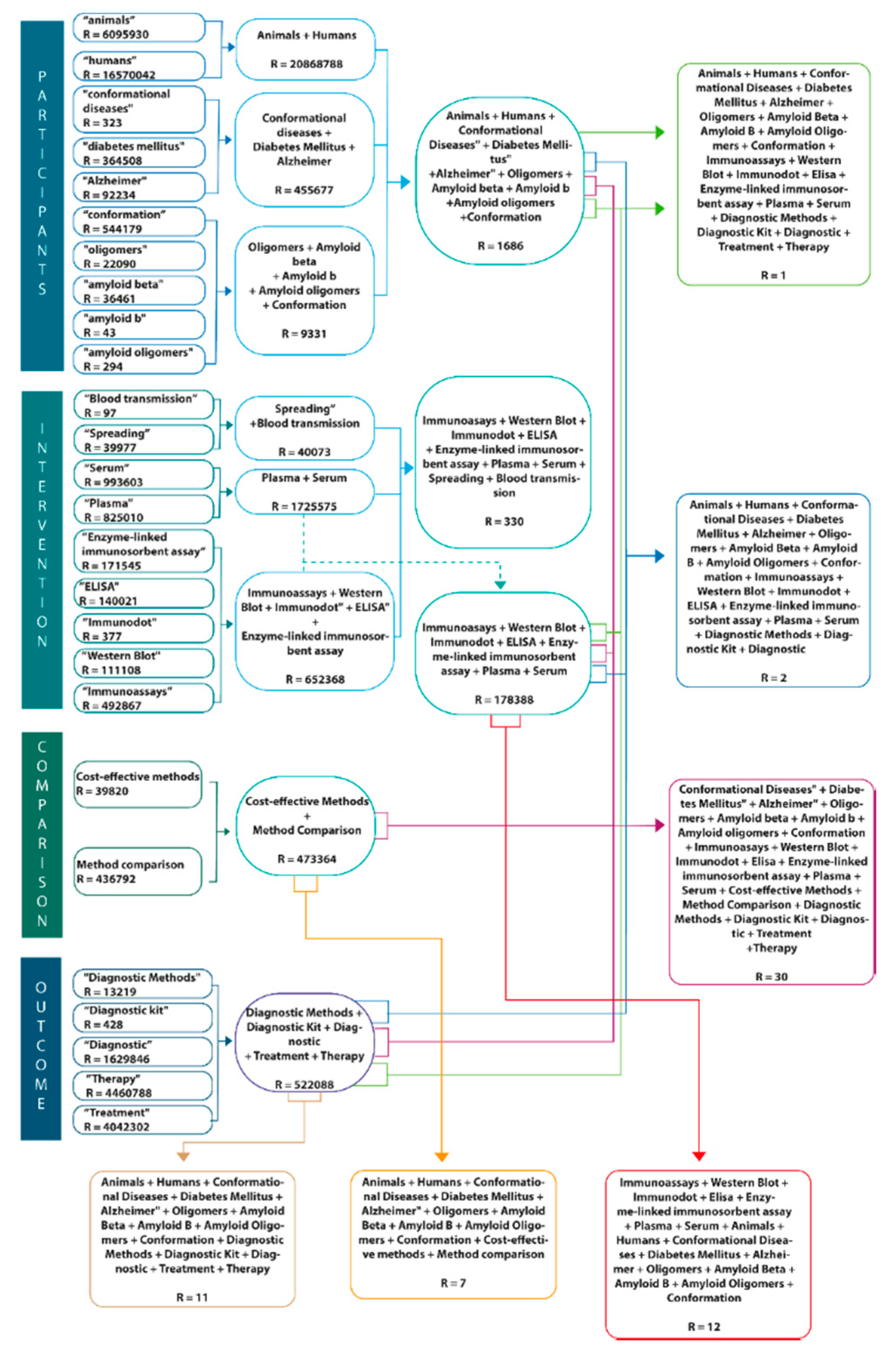
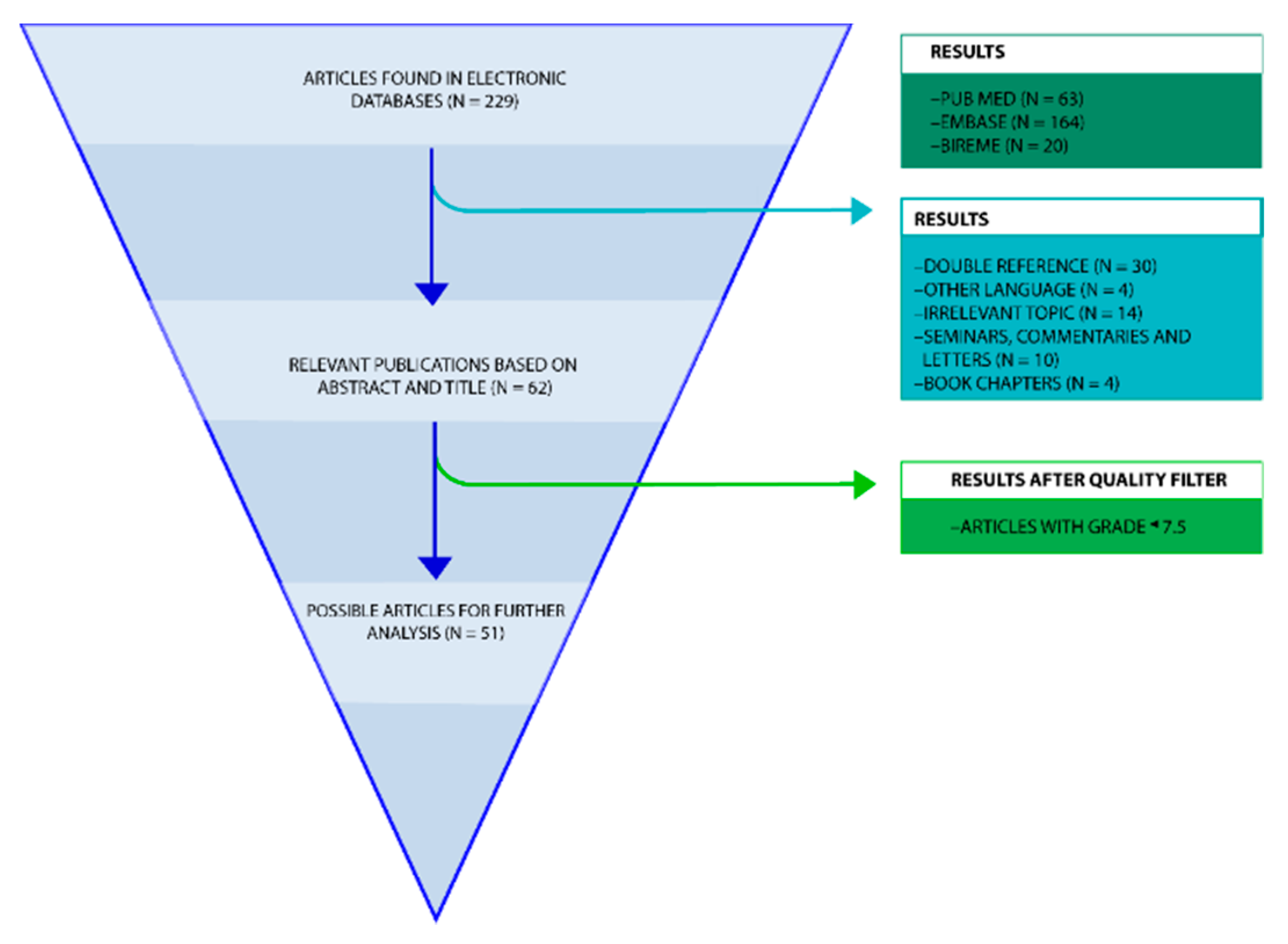
4.1. Data Sources and Searches
- P (participants): Humans, animals, Alzheimer, Diabetes Mellitus, conformational diseases, amyloid oligomers, amyloid-β, oligomers, conformation.
- I (intervention): Immunoassays, Western Blot, Immunodot, ELISA, Enzyme-linked immunosorbent assay, plasma, serum, spreading, blood transmission.
- C (comparison): Method comparison and cost-effectiveness.
- (outcome): Diagnostic methods, diagnostic kit, diagnostic, treatment, therapy.
4.2. Eligibility Criteria
4.3. Data Extraction and Quality Assessment
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Eisenberg, D.; Jucker, M. The amyloid state of proteins in human diseases. Cell 2012, 148, 1188–1203. [Google Scholar] [CrossRef] [PubMed]
- Chiti, F.; Dobson, C.M. Protein Misfolding, Functional Amyloid, and Human Disease. Annu. Rev. Biochem. 2006, 75, 333–366. [Google Scholar] [CrossRef] [PubMed]
- Daggett, V.; Fersht, A.R. Protein folding and binding: Moving into unchartered territory. Curr. Opin. Struct. Biol. 2009, 19, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Chiti, F.; Dobson, C.M. Amyloid formation by globular proteins under native conditions. Nat. Chem. Biol. 2009, 5, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Bemporad, F.; Ramazzotti, M. From the evolution of protein sequences able to resist self-assembly to the prediction of aggregation propensity. Int. Rev. Cell Mol. Biol. 2017, 329, 1–47. [Google Scholar] [CrossRef] [PubMed]
- Riek, R.; Eisenberg, D.S. The activities of amyloids from a structural perspective. Nature 2016, 539, 227–235. [Google Scholar] [CrossRef] [PubMed]
- Cheng, P.-N.; Liu, C.; Zhao, M.; Eisenberg, D.; Nowick, J.S. Amyloid β-sheet mimics that antagonize protein aggregation and reduce amyloid toxicity. Nat. Chem. 2012, 4, 927–933. [Google Scholar] [CrossRef] [PubMed]
- Soriaga, A.B.; Sangwan, S.; Macdonald, R.; Sawaya, M.R.; Eisenberg, D. Crystal structures of IAPP amyloidogenic segments reveal a novel packing motif of out-of-register beta sheets. J. Phys. Chem. B 2016, 120, 5810–5816. [Google Scholar] [CrossRef] [PubMed]
- Bucciantini, M.; Calloni, G.; Chiti, F.; Formigli, L.; Nosi, D.; Dobson, C.M.; Stefani, M. Prefibrillar amyloid protein aggregates share common features of cytotoxicity. J. Biol. Chem. 2004, 279, 31374–31382. [Google Scholar] [CrossRef] [PubMed]
- Chiti, F.; Webster, P.; Taddei, N.; Clark, A.; Stefani, M.; Ramponi, G.; Dobson, C.M. Designing conditions for in vitro formation of amyloid protofilaments and fibrils. Proc. Natl. Acad. Sci. USA 1999, 96, 3590–3594. [Google Scholar] [CrossRef] [PubMed]
- Sengupta, U.; Nilson, A.N.; Kayed, R. The role of amyloid-β oligomers in toxicity, propagation, and immunotherapy. EBioMedicine 2016, 6, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Nicolls, M.R. The clinical and biological relationship between Type II diabetes mellitus and Alzheimer’s disease. Curr. Alzheimer Res. 2004, 1, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Beta Amyloid Hypothesis. Available online: https://www.alz.org/national/documents/topicsheet_betaamyloid.pdf (accessed on 20 July 2017).
- Querfurth, H.W.; LaFerla, F.M. Alzheimer’s disease. N. Engl. J. Med. 2010, 362, 329–344. [Google Scholar] [CrossRef] [PubMed]
- Kayed, R. Common structure of soluble amyloid oligomers implies common mechanism of pathogenesis. Science 2003, 300, 486–489. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, D.; Singh, P.K.; Sahay, S.; Jha, N.N.; Jacob, R.S.; Sen, S.; Kumar, A.; Riek, R.; Maji, S.K. Structure based aggregation studies reveal the presence of helix-rich intermediate during α-Synuclein aggregation. Sci. Rep. 2015, 5, 9228. [Google Scholar] [CrossRef] [PubMed]
- De Matos, A.M.; de Macedo, M.P.; Rauter, A.P. Bridging Type 2 diabetes and Alzheimer’s disease: Assembling the puzzle pieces in the quest for the molecules with therapeutic and preventive potential. Med. Res. Rev. 2017. [Google Scholar] [CrossRef] [PubMed]
- Carrell, R.W.; Lomas, D.A. Conformational disease. Lancet 1997, 350, 134–138. [Google Scholar] [CrossRef]
- Carrell, R.W. Cell toxicity and conformational disease. Trends Cell Biol. 2005, 15, 574–580. [Google Scholar] [CrossRef] [PubMed]
- Leyva-García, E.; Lara-Martínez, R.; Morán-Zanabria, L.; Revilla-Monsalve, C.; Jiménez-García, L.F.; Oviedo, N.; Murata, C.; Garrido-Magaña, E.; Altamirano-Bustamante, N.F.; Altamirano-Bustamante, M.M. Novel insight into streptozotocin-induced diabetic rats from the protein misfolding perspective. Sci. Rep. 2017, 7, 11552. [Google Scholar] [CrossRef] [PubMed]
- Kayed, R.; Canto, I.; Breydo, L.; Rasool, S.; Lukacsovich, T.; Wu, J.; Albay Iii, R.; Pensalfini, A.; Yeung, S.; Head, E.; et al. Conformation dependent monoclonal antibodies distinguish different replicating strains or conformers of prefibrillar Ab oligomers. Mol. Neurodegener. 2010, 5, 57. [Google Scholar] [CrossRef] [PubMed]
- Sablón-Carrazana, M.; Fernández, I.; Bencomo, A.; Lara-Martínez, R.; Rivera-Marrero, S.; Domínguez, G.; Pérez-Perera, R.; Jiménez-García, L.F.; Altamirano-Bustamante, N.F.; Diaz-Delgado, M.; et al. Drug development in conformational diseases: A novel family of chemical chaperones that bind and stabilise several polymorphic amyloid structures. PLoS ONE 2015, 10, e0135292. [Google Scholar] [CrossRef] [PubMed]
- Dobson, C.M. Protein misfolding, evolution and disease. Trends Biochem. 1999, 24, 329–332. [Google Scholar] [CrossRef]
- Breydo, L.; Kurouski, D.; Rasool, S.; Milton, S.; Wu, J.W.; Uversky, V.N.; Lednev, I.K.; Glabe, C.G. Structural differences between amyloid beta oligomers. Biochem. Biophys. Res. Commun. 2016, 477, 700–705. [Google Scholar] [CrossRef] [PubMed]
- Despa, S.; Margulies, K.B.; Chen, L.; Knowlton, A.A.; Havel, P.J.; Taegtmeyer, H.; Bers, D.M.; Despa, F. Hyperamylinemia contributes to cardiac dysfunction in obesity and diabetes: A study in humans and rats. Circ. Res. 2012, 110, 598–608. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.-L.; Sui, Y.; Guan, J.; He, L.; Gu, X.-M.; Wong, H.K.; Baum, L.; Lai, F.M.M.; Tong, P.C.Y.; Chan, J.C.N. Amyloid oligomers in diabetic and nondiabetic human pancreas. Transl. Res. 2009, 153, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Blennow, K.; Dubois, B.; Fagan, A.M.; Lewczuk, P.; de Leon, M.J.; Hampel, H. Clinical utility of cerebrospinal fluid biomarkers in the diagnosis of early Alzheimer’s disease. Alzheimer’s Dement. 2015, 11, 58–69. [Google Scholar] [CrossRef] [PubMed]
- Iwatsubo, T. Amyloid β protein in plasma as a diagnostic marker for Alzheimer’s disease. Neurobiol. Aging 1998, 19, 161–163. [Google Scholar] [CrossRef]
- Kepp, K.P. Alzheimer’s disease: How metal ions define β-amyloid function. Coord. Chem. Rev. 2017. [Google Scholar] [CrossRef]
- Xie, B.; Liu, F.; Dong, X.; Wang, Y.; Liu, X.M.; Sun, Y. Modulation effect of acidulated human serum albumin on Cu2+-mediated amyloid β-protein aggregation and cytotoxicity under a mildly acidic condition. J. Inorg. Biochem. 2017, 171, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Bush, A.I.; Williamson, T.G.; Moir, R.D.; Li, Q.-X.; Currie, J.; Small, D.; Rumble, B.; Mönning, U.; Beyreuther, K.; Masters, C.L. Plasma amyloid precursor protein is a marker for alzheimer’s disease. Pathology 1992, 24. [Google Scholar] [CrossRef]
- Ordóñez-Gutiérrez, L.; Posado-Fernández, A.; Ahmadvand, D.; Lettiero, B.; Wu, L.; Antón, M.; Flores, O.; Moghimi, S.M.; Wandosell, F. ImmunoPEGliposome-mediated reduction of blood and brain amyloid levels in a mouse model of Alzheimer’s disease is restricted to aged animals. Biomaterials 2017, 112, 141–152. [Google Scholar] [CrossRef] [PubMed]
- Giacomelli, C.; Daniele, S.; Martini, C. Potential biomarkers and novel pharmacological targets in protein aggregation-related neurodegenerative diseases. Biochem. Pharmacol. 2017, 131, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Read, S.; Wagner, S.; Vintners, H.; Tomiyasu, U. Amyloid precursor protein activity in early Alzheimer’s disease: Diagnostic and prognostic implications. Neurobiol. Aging 1992, 13. [Google Scholar] [CrossRef]
- Bittner, T.; Zetterberg, H.; Teunissen, C.E.; Ostlund, R.E.; Militello, M.; Andreasson, U.; Hubeek, I.; Gibson, D.; Chu, D.C.; Eichenlaub, U.; et al. Technical performance of a novel, fully automated electrochemiluminescence immunoassay for the quantitation of β-amyloid (1-42) in human cerebrospinal fluid. Alzheimer’s Dement. J. Alzheimer’s Assoc. 2016, 12, 517–526. [Google Scholar] [CrossRef] [PubMed]
- Capule, C.C.; Yang, J. Enzyme-linked immunosorbent assay-based method to quantify the association of small molecules with aggregated amyloid peptides. Anal. Chem. 2012, 84, 1786–1791. [Google Scholar] [CrossRef] [PubMed]
- Van Mol, M.M.C.; Kompanje, E.J.O.; Benoit, D.D.; Bakker, J.; Nijkamp, M.D.; Seedat, S. The prevalence of compassion fatigue and burnout among healthcare professionals in intensive care units: A systematic review. PLoS ONE 2015, 10, e0136955. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, H.-C.; Yu, Y.-Z.; Liu, S.; Zhao, M.; Xu, Q. Peripherally administered sera antibodies recognizing amyloid-β oligomers mitigate Alzheimer’s disease-like pathology and cognitive decline in aged 3× Tg-AD mice. Vaccine 2016, 34, 1758–1766. [Google Scholar] [CrossRef] [PubMed]
- Andreasen, N.; Hesse, C.; Davidsson, P.; Minthon, L.; Wallin, A.; Winblad, B.; Vanderstichele, H.; Vanmechelen, E.; Blennow, K. Cerebrospinal fluid beta-amyloid(1-42) in Alzheimer disease: Differences between early- and late-onset Alzheimer disease and stability during the course of disease. Arch. Neurol. 1999, 56, 673–680. [Google Scholar] [CrossRef] [PubMed]
- Mattsson, N.; Andreasson, U.; Persson, S.; Carrillo, M.C.; Collins, S.; Chalbot, S.; Cutler, N.; Dufour-Rainfray, D.; Fagan, A.M. CSF biomarker variability in the Alzheimer’s Association quality control program. Alzheimer’s Dement. 2013, 9, 251–261. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arai, H.; Terajima, M.; Miura, M.; Higuchi, S.; Takase, S.; Clark, C. Tau in cerebrospinal fluid: A potential diagnostic marker in Alzheimer´s diseases. Ann. Neurol. 1995, 38, 649–652. [Google Scholar] [CrossRef] [PubMed]
- Arai, H.; Terajima, M.; Miura, M.; Higuchi, S.; Takase, S.; Clark, C.; VM, L.; Trojanowski, J.Q.; Sasaki, H. 666 Microtubule-associated protein Tau in CSF: Implications for a potential diagnostic marker in Alzheimer’s Disease. Ann. Neurol. 1996, 17, S166. [Google Scholar]
- Benedet, A.L.; Labbe, A.; Lemay, P.; Zimmer, E.R.; Pascoal, T.A.; Mathotaarachchi, S. CSF biomarkers and incipient Alzheimer disease in patients with mild cognitive impairment. Neurobiol. Aging 2017, 6, 643–648. [Google Scholar] [CrossRef]
- Bouwman, F.H.; Schoonenboom, S.N.M.; van der Flier, W.M.; van Elk, E.J.; Kok, A.; Barkhof, F.; Blankenstein, M.A.; Scheltens, P. CSF biomarkers and medial temporal lobe atrophy predict dementia in mild cognitive impairment. Neurobiol. Aging 2007, 28, 1070–1074. [Google Scholar] [CrossRef] [PubMed]
- Brys, M.; Pirraglia, E.; Rich, K.; Rolstad, S.; Mosconi, L.; Switalski, R.; Glodzik-Sobanska, L.; De Santi, S.; Zinkowski, R.; Mehta, P.; et al. Prediction and longitudinal study of CSF biomarkers in mild cognitive impairment. Neurobiol. Aging 2009, 30, 682–690. [Google Scholar] [CrossRef] [PubMed]
- Chetri, G.; Pandey, T.; Chinta, R.; Kumar, A.; Tripathi, T. An improved method for high-level soluble expression and purification of recombinant amyloid. Protein Expr. Purif. 2015, 114, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Clarke, E.E.; Shearman, M.S. Quantitation of amyloid-beta peptides in biological milieu using a novel homogeneous time-resolved fluorescence (HTRF) assay. J. Neurosci. Methods 2000, 102, 61–68. [Google Scholar] [CrossRef]
- Condello, C.; Stöhr, J. Aβ propagation and strains: Implications for the phenotypic diversity in Alzheimer’s disease. Neurobiol. Dis. 2017. [Google Scholar] [CrossRef] [PubMed]
- Doran, T.; Gao, Y.; Simanski, S.; McEnaney, P.; Kodadek, T. High affinity binding of conformationally constrained synthetic oligomers to an antigen-specific antibody: Discovery of a diagnostically useful synthetic ligand for murine Type 1 diabetes autoantibodies. Bioorg. Med. Chem. Lett. 2015, 25, 4901–4917. [Google Scholar] [CrossRef] [PubMed]
- Gustafson, D.R.; Skoog, I.; Rosengren, L.; Zetterberg, H.; Blennow, K. Cerebrospinal fluid beta-amyloid 1-42 concentration may predict cognitive decline in older women. J. Neurol. Neurosurg. Psychiatry 2007, 78, 461–464. [Google Scholar] [CrossRef] [PubMed]
- Hansson, O.; Zetterberg, H.; Buchhave, P.; Londos, E.; Blennow, K.; Minthon, L. Association between CSF biomarkers and incipient Alzheimer’s disease in patients with mild cognitive impairment: A follow-up study. Lancet Neurol. 2006, 5, 228–234. [Google Scholar] [CrossRef]
- Hansson, O.; Zetterberg, H.; Buchhave, P.; Andreasson, U.; Londos, E.; Minthon, L.; Blennow, K. Prediction of Alzheimer’s disease using the CSF Abeta42/Abeta40 ratio in patients with mild cognitive impairment. Dement. Geriatr. Cogn. Disord. 2007, 23, 316–320. [Google Scholar] [CrossRef] [PubMed]
- Hampel, H.; Teipel, S.; Fuchsberger, T.; Andreasen, N.; Wiltfang, J.; Otto, M.; Shen, Y.; Dodel, R.; Du, Y.; Farlow, M.; et al. Value of CSF β-amyloid 1-42 and tau as predictors of Alzheimer’s disease in patients with mild cognitive impairment. Mol. Psychiatry 2004, 9, 705–710. [Google Scholar] [CrossRef] [PubMed]
- Herukka, S.-K.; Hallikainen, M.; Soininen, H.; Pirttila, T. CSF Abeta 42 and tau or phosphorylated tau and prediction of progressive mild cognitive impairment. Neurology 2005, 64, 1294–1297. [Google Scholar] [CrossRef] [PubMed]
- Herukka, S.-K.; Helisalmi, S.; Hallikainen, M.; Tervo, S.; Soininen, H.; Pirttilä, T. CSF Abeta42, Tau and phosphorylated Tau, APOE epsilon4 allele and MCI type in progressive MCI. Neurobiol. Aging 2007, 28, 507–514. [Google Scholar] [CrossRef] [PubMed]
- Janssen, L.; Sobott, F.; De Deyn, P.P.; Van Dam, D. Signal loss due to oligomerization in ELISA analysis of amyloid-beta can be recovered by a novel sample pre-treatment method. MethodsX 2015, 2, 112–123. [Google Scholar] [CrossRef] [PubMed]
- Jayamani, J.; Shanmugam, G. Gallic acid, one of the components in many plant tissues, is a potential inhibitor for insulin amyloid fibril formation. Eur. J. Med. Chem. 2014, 85, 352–358. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.H.; Kumar, R. Trehalose induced conformational changes in the amyloid-β peptide. Pathology 2017, 213, 643–648. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.S.; Ahn, H.-S.; Cho, S.M.; Lee, J.E.; Kim, Y.; Lee, C. Detection and quantification of plasma amyloid-β by selected reaction monitoring mass spectrometry. Anal. Chim. Acta 2014, 840, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Klaver, A.C.; Coffey, M.P.; Smith, L.M.; Bennett, D.A.; Finke, J.M.; Dang, L.; Loeffler, D.A. ELISA measurement of specific non-antigen-bound antibodies to Aβ1-42 monomer and soluble oligomers in sera from Alzheimer’s disease, mild cognitively impaired, and noncognitively impaired subjects. J. Neuroinflamm. 2011, 8, 93. [Google Scholar] [CrossRef] [PubMed]
- Kuo, C.T.; Chen, Y.L.; Hsu, W.T.; How, S.C.; Cheng, Y.H.; Hsueh, S.S.; Liu, H.S.; Lin, T.H.; Wu, J.W.; Wang, S.S.S. Investigating the effects of erythrosine B on amyloid fibril formation derived from lysozyme. Int. J. Biol. Macromol. 2017, 98, 159–168. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Sokal, I.; Quinn, J.F.; Leverenz, J.B.; Brodey, M.; Schellenberg, G.D.; Kaye, J.A.; Raskind, M.A.; Zhang, J.; Peskind, E.R.; et al. CSF tau/Abeta42 ratio for increased risk of mild cognitive impairment: A follow-up study. Neurology 2007, 69, 631–639. [Google Scholar] [CrossRef] [PubMed]
- Mangione, M.R.; Vilasi, S.; Marino, C.; Librizzi, F.; Canale, C.; Spigolon, D.; Bucchieri, F.; Fucarino, A.; Passantino, R.; Cappello, F.; et al. Biochimica et biophysica Acta (BBA)-general subjects. Biochim. Biophys. Acta (BBA) Gen. Subj. 2017, 1861, 2515–2944. [Google Scholar]
- Mattsson, N.; Ewers, M.; Rich, K.; Kaiser, E.; Mulugeta, E.; Rose, E. CSF Biomarkers and Incipient Alzheimer Disease. JAMA 2009, 302, 385–393. [Google Scholar] [CrossRef] [PubMed]
- Murakami, K. Conformation-specific antibodies to target amyloid β oligomers and their application to immunotherapy for Alzheimer’s disease. Biosci. Biotechnol. Biochem. 2014, 78, 1293–1305. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Niedowicz, D.M.; Beckett, T.L.; Matveev, S.; Weidner, A.M.; Baig, I.; Kryscio, R.J.; Mendiondo, M.S.; LeVine, H.; Keller, J.N.; Murphy, M.P. Pittsburgh compound B and the postmortem diagnosis of alzheimer disease. Ann. Neurol. 2012, 72, 564–570. [Google Scholar] [CrossRef] [PubMed]
- Permanne, B.; Buée, L.; David, J.-P.; Fallet-Bianco, C.; Di Menza, C.; Delacourte, A. Quantitation of Alzheimer’s amyloid peptide and identification of related amyloid proteins by dot-blot immunoassay. Brain Res. 1995, 685, 154–162. [Google Scholar] [CrossRef]
- Plagg, B.; Marksteiner, J.; Kniewallner, K.M.; Humpel, C. Platelet dysfunction in hypercholesterolemia mice, two Alzheimer’s disease mouse models and in human patients with Alzheimer’s disease. Biogerontology 2015, 16, 543–558. [Google Scholar] [CrossRef] [PubMed]
- Riemenschneider, M.; Lautenschlager, N.; Wagenpfeil, S.; Diehl, J.; Drzezga, A.; Kurz, A. Cerebrospinal fluid Tau and β-amyloid 42 proteins identify Alzheimer disease in subjects with mild cognitive impairment. Arch. Neurol. 2002, 59, 1729. [Google Scholar] [CrossRef] [PubMed]
- Ringman, J.M.; Younkin, S.G.; Pratico, D.; Seltzer, W.; Cole, G.M.; Geschwind, D.H.; Rodriguez-Agudelo, Y.; Schaffer, B.; Fein, J.; Sokolow, S.; et al. Biochemical markers in persons with preclinical familial Alzheimer disease. Neurology 2008, 71, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Rogeberg, M.; Almdahl, I.S.; Wettergreen, M.; Nilsson, L.N.G.; Fladby, T. Isobaric quantification of cerebrospinal fluid amyloid-β peptides in Alzheimer’s disease: C-terminal truncation relates to early measures of neurodegeneration. J. Proteome Res. 2015, 14, 4834–4843. [Google Scholar] [CrossRef] [PubMed]
- Roqanian, S.; Meratan, A.A.; Ahmadian, S.; Shafizadeh, M.; Ghasemi, A.; Karami, L. Polyphenols protect mitochondrial membrane against permeabilization induced by HEWL oligomers: Possible mechanism of action. Int. J. Biol. Macromol. 2017, 103, 709–720. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, E.D.; Almada, M.; Burboa, M.G.; Taboada, P.; Mosquera, V.; Valdez, M.A.; Juárez, J. Oligomers, protofibrils and amyloid fibrils from recombinant human lysozyme (rHL): Fibrillation process and cytotoxicity evaluation for ARPE-19 cell line. Coll. Surf. B Biointerfaces 2015, 126, 335–343. [Google Scholar] [CrossRef] [PubMed]
- Salvadores, N.; Shahnawaz, M.; Scarpini, E.; Tagliavini, F.; Soto, C. Detection of misfolded Aβ oligomers for sensitive biochemical diagnosis of Alzheimer’s disease. Cell Rep. 2014, 7, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Sharoar, M.; Shahnawaz, M.; Islam, M.; Ramasamy, V.; Shin, S.; Park, I. The inhibitory effects of Escherichia coli maltose binding protein on β-amyloid aggregation and cytotoxicity. Arch. Biochem. Biophys. 2013, 538, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Shaw, L.M.; Vanderstichele, H.; Knapik-Czajka, M.; Clark, C.M.; Aisen, P.S.; Petersen, R.C.; Blennow, K.; Soares, H.; Simon, A.; Lewczuk, P.; et al. Alzheimer’s disease neuroimaging initiative cerebrospinal fluid biomarker signature in Alzheimer’s disease neuroimaging initiative subjects. Ann. Neurol. 2009, 65, 403–413. [Google Scholar] [CrossRef] [PubMed]
- Skoog, I.; Davidsson, P.; Aevarsson, O.; Vanderstichele, H.; Vanmechelen, E.; Blennow, K. Cerebrospinal fluid beta-amyloid 42 is reduced before the onset of sporadic dementia: A population-based study in 85-year-olds. Dement. Geriatr. Cogn. Disord. 2003, 15, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Spiegel, D.M.; Sprague, S.M. Serum amyloid P component: A predictor of clinical beta 2-microglobulin amyloidosis. Am. J. Kidney Dis. 1992, 19, 427–432. [Google Scholar] [CrossRef]
- Stern, R.; Trojanowski, J.Q.; Lee, V. Antibodies to the beta-amyloid peptide cross-react with conformational epitopes in human fibrinogen subunits from peripheral blood. FEBS Lett. 1990, 264, 43–47. [Google Scholar] [PubMed]
- Stomrud, E.; Hansson, O.; Blennow, K.; Minthon, L.; Londos, E. Cerebrospinal fluid biomarkers predict decline in subjective cognitive function over 3 years in healthy elderly. Dement. Geriatr. Cogn. Disord. 2007, 24, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Tamaoka, A.; Sawamura, N.; Fukushima, T.; Shoji, S.; Matsubara, E.; Shoji, M.; Hirai, S.; Furiya, Y.; Endoh, R.; Mori, H. Amyloid β protein 42(43) in cerebrospinal fluid of patients with Alzheimer’s disease. J. Neurol. Sci. 1997, 148, 41–45. [Google Scholar] [CrossRef]
- Wang, Y.; Xia, Z.; Xu, J.-R.; Wang, Y.-X.; Hou, L.-N.; Qiu, Y.; Chen, H.-Z. A-Mangostin, a polyphenolic xanthone derivative from mangosteen, attenuates β-amyloid oligomers-induced neurotoxicity by inhibiting amyloid aggregation. Neuropharmacology 2012, 871–881. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.-Y.; Lee, K.-C.; Pei, Z.; Khan, A.; Bakshi, K.; Burns, L.H. PTI-125 binds and reverses an altered conformation of filamin A to reduce Alzheimer’s disease pathogenesis. Neurobiol. Aging 2017, 55, 99–114. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; Hong, S.; O’Malley, T.; Sperling, R.A.; Walsh, D.M.; Selkoe, D.J. New ELISAs with high specificity for soluble oligomers of amyloid β-protein detect natural Aβ oligomers in human brain but not CSF. Alzheimer’s Dement. 2013, 9, 99–112. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Chauhan, A.; Mehta, S.; Mehta, P.; Gu, F.; Chauhan, V. Trichostatin A increases the levels of plasma gelsolin and amyloid beta-protein in a transgenic mouse model of Alzheimer’s disease. Life Sci. 2014, 99, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Zetterberg, H.; Wahlund, L.-O.; Blennow, K. Cerebrospinal fluid markers for prediction of Alzheimer’s disease. Neurosci. Lett. 2003, 352, 67–69. [Google Scholar] [CrossRef] [PubMed]
- Kayed, R.; Glabe, C.G. Conformation-dependent anti-amyloid oligomer antibodies. Methods Enzym. 2006, 413, 326–344. [Google Scholar] [CrossRef]
- Nelson, R.; Sawaya, M.R.; Balbirnie, M.; Madsen, A.Ø.; Riekel, C.; Grothe, R.; Eisenberg, D. Structure of the cross-beta spine of amyloid-like fibrils. Nature 2005, 435, 773–778. [Google Scholar] [CrossRef] [PubMed]
- Colegio oficial de enfermeria de málaga. Formulación de preguntas clínicas específicas en formato PICO. Rev. Cuid. Digit. 2013, 5, 2013. Available online: http://revistacuidandote.eu/fileadmin/VOLUMENES/2013/Volumen5/Formacion/FORMULACION_DE_PREGUNTAS.....pdf (accessed on 20 July 2017).
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. PLoS Med. 2009, 6. [Google Scholar] [CrossRef] [PubMed]
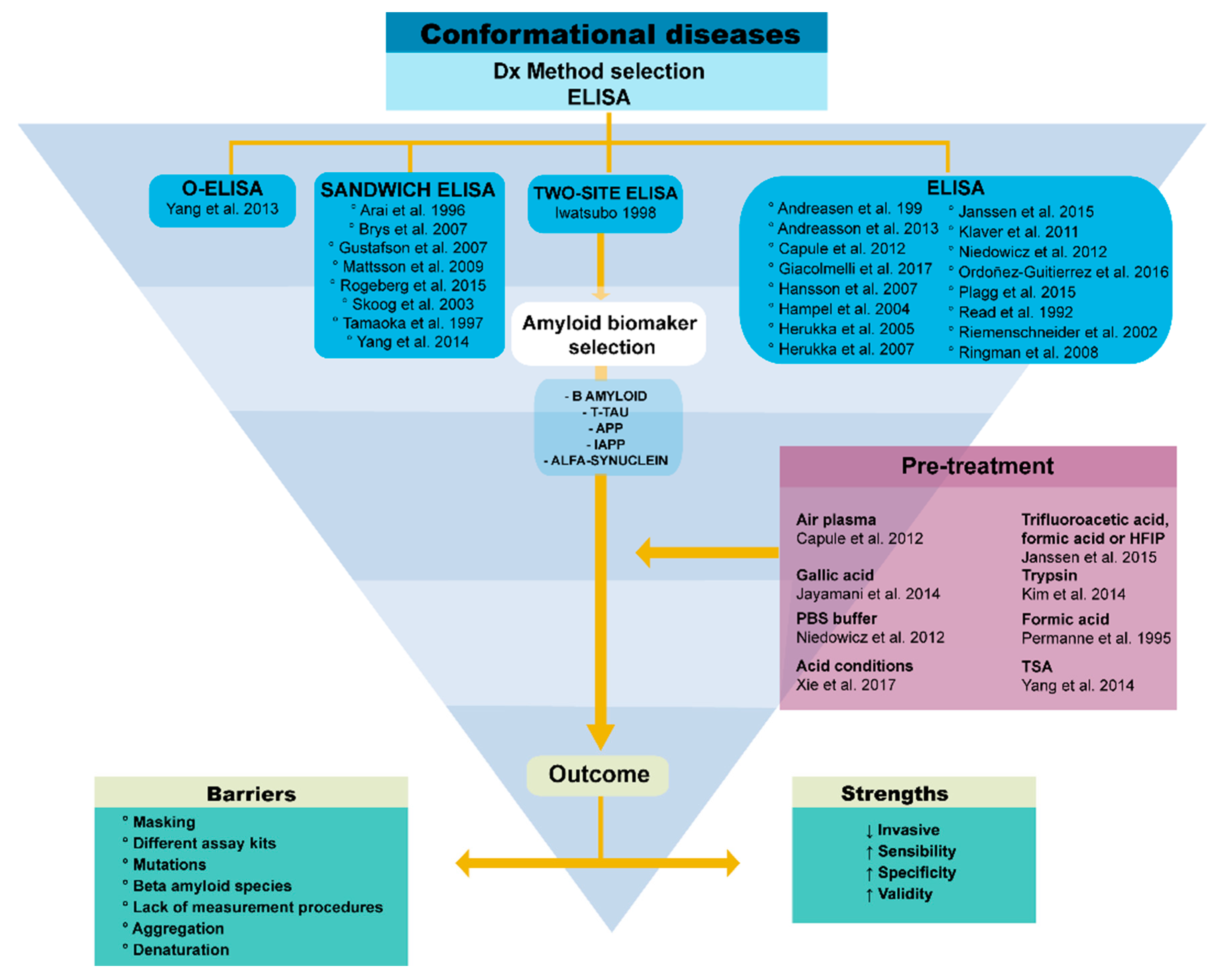
| First Author/Year of Publication | Setting | Target Population and Sample Size | Target Molecule | Method | Quality (%) |
|---|---|---|---|---|---|
| Andreasen, et al. 1999 [39] | Sweden | 16 MCI-AD patients and 15 age-matched controls | CSF Aβ42 and CSF T-tau | Longitudinal MCI-control study/Elisa | 100 |
| Arai, et al. 1995 [41,42] | Japan | 201 CSF samples, 87 patients with Alzheimer’s disease (AD) and 114 patients without neurological diseases | Microtubule-associated protein tau in CSF | Elisa | 75 |
| Benedett, et al. 2015 [43] | Canada | 417 participants and 174 additional patients with samples of CSF and P-Tau | Aβ | SUVR and PET | 87.5 |
| Bittner, et al. 2015 [35] | Roche Diagnostics | 372 Human CSF with diagnosed AD | Aβ | Elecsys β-amyloid assay (electro chemiluminescence immunoassay) | 100 |
| Blennow, et al. 2014 [27] | United States | Humans | Cerebrospinal fluid biomarkers such as B-amyloid, Tau and Tau phosphorylated for Alzheimer’s detection | Article review | 75 |
| Bouwman, et al. 2007 [44] | The Netherlands | 59 MCI patients, 30 of them developed AD | CSF Aβ42 and CSF T-tau | Longitudinal MCI study | 100 |
| Breydo, et al. 2016 [24] | United States | Synthetic Aβ | Aβ40 oligomers FOs and PFOs | Raman, FTIR, CD spectroscopy and Western Blot | 62.5 |
| Brys, et al. 2007 [45] | United States | 65 MCI patients, 22 of who later developed AD and 21 controls | CSF Aβ42, Aβ42/Aβ40 ratio, CSF T-tau, CSF P-tau231 | Elisa and Innotest hTAU antigen kit | 100 |
| Bush, et al. 1992 [31] | Germany | Human platelets and plasma samples of patients diagnosed with AD | APP | Western Blot and Immunodot | 87.5 |
| Capule, et al. 2012 [36] | United States | Synthetic β-amyloid proteins and AD Aβ samples | Binding molecules to Aβ | Elisa | 87.5 |
| Chetri et al. 2015 [46] | India | Cultivated Aβ | Cultivated Aβ | Cloning of Aβ with PCR | 62.5 |
| Clarke, et al. 2000 [47] | United Kingdom | Antibodies and Aβ from humans and rodents | Aβ | HTRF immunoassay | 87.5 |
| Condello, et al. 2017 [48] | United States | Humans samples diagnosed with AD | Aβ and Tau aggregates | Therapy and diagnostic options revision for Alzheimer’s | 87.5 |
| Despa, et al. 2012 [25] | United States | Left ventricular homogenates of obese, non-obese and diabetic patients and rodents | Amylin | Western Blot/Dot Blot | 100 |
| Doran, et al. 2015 [49] | United States | Antigen substitutes | Antigens capable of identifying sites of antibodies. Types: OBOC of animals or humans control and with disease of interest. | ELISA measurement | 75 |
| Giacomelli, et al. 2017 [33] | Italy | Human post-mortem brain, plasma, platelets, CSF, RBC, samples and AD mice | Aβ,tau and a-syn | Bibliographic analysis | 75 |
| Gustafson, et al. 2007 [50] | Sweden | 55 cognitively healthy women | CSF Aβ42 | Longitudinal cohort study and Sandwich ELISA (Innotest hTAU-Ag) | 100 |
| Hansson, et al. 2006 [51] | Sweden | 137 MCI patients, 57 of whom developed AD | CSF Aβ42, high CSF T-tau, and CSF P-tau181 | Luminex xMAP technology | 100 |
| Hansson, et al. 2007 [52] | Sweden | 137 MCI patients, 57 of whom developed AD | Aβ42/Aβ40 ratio | Elisa | 100 |
| Hampel, et al. 2004 [53] | Germany | 52 MCI patients, 93 AD patients and 10 controls | CSF Aβ42 and CSF T-tau | Elisa | 100 |
| Herukka, et al. 2005 [54] | Finland | 78 MCI patients, 23 of whom developed AD, 46 controls | CSF Aβ42, CSF T-tau, and CSF P-tau181 | Elisa | 100 |
| Herukka, et al. 2007 [55] | Finland | 79 MCI patients, 33 of whom developed AD, 60 controls | CSF Aβ42, CSF T-tau, and CSF P-tau181 | PCR and Elisa | 100 |
| Iwatsubo. 1998 [28] | Japan | 6 patients with Beta APP 717, 44 patients with sporadic AD, 22 controls of neurological diseases and 15 controls without neurological disease | C-terminus of β-amyloid 42 and β-amyloid 40 | Two-site Elisa | 87.5 |
| Janssen, et al. 2015 [56] | Belgium | Aggregated β-amyloid oligomers from mouse brain and human CSF | β-amyloid | Samples exposed to a pretreatment with TFA, FA and HFIP with ELISA/Western Blot (SDS-PAGE) | 75 |
| Jayamani, et al. 2014 [57] | India | Synthetic insulin and Gallic acid | Insulin in vitro and Aβ fibril formation | UV-Visible spectroscopy, ThT fluorescence spectroscopy, CD, Fourier-transformed infrared (FTIR) spectroscopy, and fibril morphology using atomic force microscopy (AFM) | 87.5 |
| Kepp, et al. 2017 [29] | Denmark | Patients with Alzheimer’s disease | Metal ions and β-amyloid | Bibliographic review | 75 |
| Khan, et al. 2017 [58] | United States | Human Aβ40 synthetic peptides | Aβ40 | Dichroism and Fluorescence emission spectroscopy | 62.5 |
| Kim, et al. 2014 [59] | Korea | Human plasma | Synthetic Aβ40 | SRM-MS and antibody-free spectrometry | 87.5 |
| Klaver, et al. 2011 [60] | United States | 328 Serum antibodies with AD, MCI or ICU patients | β-amyloid and antibodies | Western Blot and Elisa | 87.5 |
| Kuo, et al. 2017 [61] | Taiwan | Hen lysozyme | Amyloid fibrils and erythrosine B | Molecular docking and molecular dynamics simulations | 62.5 |
| Li, et al. 2007 [62] | United States | 43 controls, 4 of whom developed MCI | T-tau/Aβ42 ratio | Luminex reagents | 100 |
| Mangione, et al. 2016 [63] | Italy | Chaperone Hsp60 | Aβ40 | CD, TEM, AFM and SEC | 75 |
| Mattsson, et al. 2009 [40,64] | Sweden | 750 MCI patients, 271 of whom developed AD | CSF Aβ42, CSF T-tau, and CSF P-tau181 | Sandwich Elisa | 100 |
| Murakami. 2014 [65] | Japan | Alzheimer’s disease patients | Aβ oligomers | Immunotherapy | 87.5 |
| Niedowicz, et al. 2012 [66] | United States | 23 controls age 87 years average, 22 patients with Alzheimer's average age 85 | Aβ | Elisa | 75 |
| Ordóñez-Gutiérrez, et al. 2016 [32] | Spain | ImmunoPEGliposomes with β-amyloid in brain cells and in vitro phagocytes in mice aged 16 months and 10 months of age | β-amyloid peptides | Elisa and Western Blot | 87.5 |
| Permanne, et al. 1995 [67] | France | 17 elderly with AD and no AD | Aβ | Dot-blot and Western Blot | 100 |
| Plagg, et al. 2015 [68] | Austria | Platelets of mice with hypercholesterolemia and 43 humans with and 30 controls | APP | Elisa and Western Blot | 87.5 |
| Read, et al. 1992 [34] | The Netherlands | 4 CSF samples from patients with dementia | APP | Elisa | 75 |
| Riemenschneider, et al. 2002 [69] | Germany | 4 ventricular and lumbar CSF samples ventricular with AD | APP | Elisa | 100 |
| Ringman, et al. 2008 [70] | United States | CSF biomarker results were compared in 7 asymptomatic carriers of familial AD (FAD)-associated mutations and four non-carriers | CSF Aβ42, Aβ42/Aβ40 ratio, CSF T-tau, and CSF P-tau181 | Elisa | 100 |
| Rogeberg, et al. 2015 [71] | Norway | 19 of the Alzheimer’s CSF samples and 19 CSF healthy controls | Aβ | Mass spectrometry (MS), sandwich Elisa, Immunoprecipitation | 87.5 |
| Roqanian, et al. 2017 [72] | Iran | Cerebral rat samples | HEWL Oligomers | ThT fluorescent assay, and Nile red binding assay | 100 |
| Ruiz, et al. 2015 [73] | Mexico | Aggregated β-amyloid fibrils | Aβ fibrils | Surface tension and DLS | 75 |
| Salvadores, et al. 2014 [74] | United States | 50 samples of CSF patients with AD + 30 healthy control patients and 39 other neurological diseases | Aβ oligomers/PrP | PMCA | 87.5 |
| Sharoar, et al. 2013 [75] | Bangladesh | Samples of human neuroblastoma cells in vitro | Aβ | Th-T assay, CD spectroscopy, Immunoblot and Dot blot | 75 |
| Shaw, et al. 2009 [76] | United States | 196 MCI patients, 37 of whom developed AD | CSF Aβ42, CSF T-tau, and CSF P-tau181 | Multiplex immunoassay; xMAP Luminex | 100 |
| Sengupta, et al. 2016 [11] | United States | Alzheimer’s disease | Aβ | Bibliographic Review | 87.5 |
| Skoog, et al. 2003 [77] | Sweden | 57 cognitively normal controls underwent LP and were followed for 3 years | CSF Aβ42 | Sandwich Elisa | 100 |
| Spiegel, et al. 1992 [78] | United States | 22 blood samples from control patients aged 50 years on average and 59 patients with chronic hemodialysis | Aβ | 'Rocket' immunodiffusion test | 100 |
| Stern, et al. 1990 [79] | United States | Blood samples and blood antibodies | Aβ | Western Blot | 75 |
| Stomrud, et al. 2007 [80] | Sweden | 57 cognitively normal controls underwent LP and were followed for 3 years | CSF Aβ42 | xMAP technology and the INNO-BIA AlzBio3 kit | 100 |
| Tamaoka, et al. 1997 [81] | Japan | CSF of AD patient samples and 34 without AD | Aβ | Elisa and sandwich Elisa | 100 |
| Wang, et al. 2012 [82] | China | Primary rat cerebral cortical neurons | β-amyloid with alfa-M | Dot blot assay, Western Blot and ThT fluorescence | 75 |
| Wang, et al. 2016 [38] | China | Brain samples of passively immunized 3× Tg-AD mice and rabbits control and wild-type | Dynamin 1 | Western Blot, Immunoblots, Dot Blot and Elisa | 75 |
| Wang, et al. 2017 [83] | United States | Triple-transgenic (3× Tg) AD mice and humans samples diagnosed with AD | Solubilized immunoprecipitates | Western Blot | 87.5 |
| Xie, et al. 2017 [30] | China | Human neuroblastoma SH-SY5Y cells | Aβ aggregates | Thioflavin T fluorescent assay | 87.5 |
| Yang, et al. 2013 [84] | United States | 90 samples of human brain and CSF samples with AD and no AD and transgenic mouse brains | Aβ oligomers | Sandwich Elisa, o-Elisa, Immunoprecipitation and Western Blot | 87.5 |
| Yang, et al. 2014 [85] | United States | 26 APPswe/PS1 transgenic mice blood samples | Gelsolin | Western Blot and sandwich Elisa | 75 |
| Zetterberg, et al. 2003 [86] | Sweden | 53 MCI patients, 22 of whom developed AD | CSF Aβ42, CSF T-tau, and CSF P-tau181 | Electroencephalogram, magnetic resonance imaging, single photon emission, computed tomography and MMSE | 100 |
| Zhao, et al. 2009 [26] | China | 8 nondiabetic control subjects, 8 type 2 diabetic cases without islet amyloid, and 8 type 2 diabetic patients with islet amyloid | Aβ oligomers | Immunofluorescent microscopy and autopsy | 100 |
| Reference | Setting | Target Molecule | Method of Analysis | Target Population | Clinical Data | Sensibility and Specificity | Pretreatment | Antibodies |
|---|---|---|---|---|---|---|---|---|
| Andreasen, et al. 1999 [39] | Sweden | CSF Aβ42 and CSF T-tau | Elisa | 16 MCI-AD patients, 15 age-matched controls | Low CSF Aβ42, high CSF T-tau associated with AD | Sensibility: 88% | IA | -Antibodies: 21F12 and 3D6 -Kit: Innotest β-amyloid (1–40) Innogenetics |
| Arai, et al. 1996 [41,42] | Japan | Microtubule-associated protein tau in CSF | Sandwich Elisa and Western Blot | 114 patients non-AD neurological diseases, 22 normal subjects | CSF tau increased in AD patients compared with non-AD neurological disease | Sensitivity and specificity missing | IA | -Not mentioned |
| Bittner, et al. 2015 [35] | Roche Diagnostics | Aβ | Elecsys β-amyloid assay | 372 Human CSF | Elecsys β-amyloid 42 has high analytical performance that improves biomarker-based AD diagnosis | Sensitivity and specificity missing | IA | |
| Bouwman, et al. 2007 [44] | The Netherlands | CSF Aβ42 and CSF T-tau | Innotest β-amyloid1-42 and Innotest hTau-Ag | 59 MCI patients, 30 of whom developed AD | Patients with abnormal values at baseline had higher risk of developing AD. | Sensitivity and specificity missing. | Pretreatment missing. | |
| Brys, et al. 2007 [45] | United States | CSF Aβ42, Aβ42/Aβ40 ratio, CSF T-tau, CSF P-tau231 | Innotest hTAU antigen kit; sandwich Elisa for P-tau231 | 65 MCI patients, 22 of whom developed AD, 21 controls | All biomarkers were statistically significant predictors of the decline from MCI to AD with P-tau231 and T-tau the strongest univariate predictors. | Sensitivity 68–86%, specificity 60–91% | Pretreatment missing. | -Antibody: Monoclonal antibody 6E10 -Kit: INNOTEST hTAU antigen kit (Innogenetics®, Gent, Belgium). |
| Bush, et al. 1992 [31] | Germany | APP | Western Blot; Immunodot | Human platelets, human plasma of AD cases | β-amyloid deposition may result in failure of APP | Sensitivity and specificity missing | Pretreatment missing | |
| Capule, et al. 2012 [36] | United States | Binding molecules to Aβ | Elisa | 96 ELISA plates of Synthetic beta amyloid proteins and AD Aβ samples | Protocol overcomes many limitations of previously reported spectroscopic or radioactivity assays and facilitate the screening and evaluation of a more structurally diverse set of amyloid-targeting agents | Sensitivity and specificity missing. | Air plasma | -Antibody: 6E10 -Kit: not mentioned |
| Clarke, et al. 2000 [47] | United Kingdom | Aβ | Homogeneous time-resolved fluorescence (HTRF) immunoassay | Synthetic β-amyloid proteins and antibodies from humans and rodents | This assay allows specific, direct quantitation of Aβ peptides in cell culture medium, plasma, cerebrospinal fluid and brain tissue extracts. | Sensitivity and specificity missing | Pretreatment missing | |
| Despa, et al. 2012 [25] | United States | Amylin | Western Blot/Dot Blot | Left ventricular homogenates of humans and rodents with DM2 and controls | Hyperamylinemia promotes amylin deposition in the heart, causing alterations of cardiac myocyte structure and function. | Sensitivity and specificity missing. | Pretreatment missing. | |
| Giacomelli, et al. 2017 [33] | Italy | Aβ,tau and a-syn | Congo red Fluorescent microscopy, Thioflavin-T, Elisa, SPECT, PET and Western Blot | Human post-mortem brain, plasma, platelets, CSF, RBC, samples and AD mice | Biomarkers establishment and assessment is important for diagnosis and therapeutic options | IA | IA | |
| Gustafson, et al. 2007 [50] | Sweden | CSF Aβ42 | Sandwich ELISA and Innotest hTAU-Ag | 55 cognitively healthy women | Low levels of CSF Aβ42 predicted cognitive decline. | Sensitivity and specificity missing. | Pretreatment missing. | -Antibody: not mentioned -Kit: Innotest β-amyloid 1-42; Innogenetics, Zwijndrecht, Belgium |
| Hansson, et al. 2006 [51] | Sweden | CSF Aβ42, high CSF T-tau, and CSF P-tau181 | Luminex xMAP technology | 137 MCI patients, 57 of whom developed AD | Concentrations of T-tau, P-tau181, and Aβ42 in CSF are strongly associated with future development of Alzheimer’s disease in patients with MCI. | Sensitivity 95%, specificity 83%, PPV 81%, NPV 96% | IA | |
| Hansson, et al. 2007 [52] | Sweden | Aβ42/Aβ40 ratio | Elisa | 137 MCI patients, 57 of whom developed AD | Amyloid precursor protein metabolism is disturbed in early sporadic AD and points to the usefulness of the Aβ42/Aβ40 ratio as a predictive biomarker for AD. | Sensitivity 87%, specificity 78% | Pretreatment missing. | -Antibodies: W02 (epitope AB 5–8), detection anti-bodies G2–10 for Aβ40 and G2–13 for Aβ42 -Kit: The Genetics Company |
| Hampel, et al. 2004 [53] | Germany | CSF Aβ42 and CSF T-tau | Elisa | 52 MCI patients, 93 AD patients and 10 controls | CSF tau and Aβ1-42 may be useful biomarkers in the early identification of AD in MCI subjects. | Sensitivity 59–83%, specificity 90–100% | Pretreatment missing. | -Antibody: not mentioned -Kit: Innotest β-amyloid 1-42 and Innotest hTAU-Ag, Innogenetics, Zwjindrecht, Belgium |
| Herukka, et al. 2005 [54] | Finland | CSF Aβ42, CSF T-tau, and CSF P-tau181 | Elisa | 78 MCI patients, 23 of whom developed AD, 46 controls | The most predictive assay for AD among the patients with MCI was the combination of Aβ42 and P-tau. | Sensitivity 91%, specificity 56% | Pretreatment missing. | -Antibody: not mentioned -Kit: Innogenetics, Ghent, Bel-gium |
| Herukka, et al. 2007 [55] | Finland | CSF Aβ42, CSF T-tau, and CSF P-tau181 | PCR and Elisa | 79 MCI patients, 33 of whom developed AD, 60 controls | Low levels of CSF Aβ42 predicted progression to AD. | Sensitivity and specificity missing. | Pretreatment missing. | -Antibody: not mentioned -Kit: Innogenetics, Ghent, Belgium |
| Iwatsubo, 1998 [28] | Japan | C-terminus of Aβ 42 and β-amyloid 40 | Two-site Elisa | 6 patients with Beta APP 717, 44 patients with sporadic AD, 22 controls of neurological diseases and 15 controls without neurological disease | Levels of Aβ, especially those of Aβ42 are altered in the plasma of patients with AD, including carriers of APP717 mutation that is linked to familial AD. | Sensitivity and specificity missing. | Pretreatment missing. | |
| Janssen, et al. 2015 [56] | Belgium | β-amyloid | Elisa and Western Blot | Aggregated β-amyloid oligomers from mouse brain and human CSF | Chemically pre-treating samples to disaggregate oligomers can (partially) recover the signal loss. | 7.81 and 500 pg/mL | Trifluoroacetic acid, formic acid or HFIP | -Antibody: 6E10 -Kit: Human Amyloid β (1-x) Assay kit (IBL International) |
| Jayamani, et al. 2014 [57] | India | Insulin in vitro | UV-Visible spectroscopy, ThT fluorescence spectroscopy, CD, Fourier-transformed infrared (FTIR) spectroscopy, and fibril morphology using atomic force microscopy (AFM) | Synthetic insulin and Gallic acid | Gallic acid can inhibit insulin Aβ fibril formation in vitro | Sensitivity and specificity missing. | Gallic acid | |
| Kepp, et al. 2017 [29] | Denmark | Metal ions and β-amyloid | IA | Patients with Alzheimer’s disease | The metal-Aβ interactions have elements of both gain of toxic function. Possible treatments for β-Amyloid accumulation: metal chelation, treatment with anti-oxidant and anti-inflammatory molecules. | IA | IA | |
| Kim, et al. 2014 [59] | Korea | β-amyloid | Mass spectrometry (MS)-based quantification | Human plasma | β-amyloid can be measured regardless of the conformational status of the biomarker | Sensitivity and specificity missing. | Trypsin | |
| Klaver, et al. 2011 [60] | United States | β-amyloid and antibodies | Western Blot and Elisa | 328 Serum antibodies with AD patients, subjects with mild cognitive impairment, and aged non-cognitively impaired individuals | Hypothesis that reduced levels of anti- Aβ antibodies might contribute to AD’s pathogenesis not proven. | Sensitivity and specificity missing. | Pretreatment missing | -Antibody: 6E10 anti-Aβ antibody -Kit: not mentioned |
| Li, et al. 2007 [62] | United States | T-tau/Aβ42 ratio | Luminex reagents | 43 controls, 4 of whom developed MCI | Individuals with high ratio had higher APOE ε4 allele frequency and higher risk of progression to MCI | High sensitivity and specificity | IA | |
| Mangione, et al. 2016 [63] | Italy | Aβ40 | CD, TEM, AFM and SEC | Chaperone Hsp60 | Hsp60 inhibits Aβ | Sensitivity and specificity missing | IA | |
| Mattsson, et al. 2009 [40,64] | Sweden | CSF Aβ42, CSF T-tau, and CSF P-tau181 | Sandwich Elisa | 750 MCI patients, 271 of whom developed AD | CSFA 42, T-tau, and P-tauidentify incipient AD with good accuracy | Sensitivity 83%, specificity 88% for MCI-AD versus controls; sensitivity83%, specificity 72% for MCI-AD versus all MCI cases | Pretreatment missing | -Innotest Phospho-Tau[181P] -Innotest-amyloid (1-42) |
| Murakami. 2014 [65] | Japan | Aβ oligomers | Immunotherapy | Alzheimer’s disease patients | Immunotherapy using anti-Aβ anti- body is a possible approach for AD treatment | IA | IA | |
| Niedowicz, et al. 2012 [66] | United States | Aβ | Elisa | Samples of patients with AD (22) and controls (23) and four different brain regions | Postmortem PiB binding is useful in distinguishing AD from control cases, SDS-soluble Ab measured by standard immunoassay was better. | Sensitivity of 100% and a specificity of 95.7% | PBS buffer | -Antibody: Ab9 (human sequence Ab1–16), Ab13.1.1, 12F4 (Covance, Princeton, NJ) y 4G8 -Kit: not mentioned |
| Ordóñez-Gutiérrez, et al. 2016 [32] | Spain | Aβ | Elisa and Western Blot | ImmunoPEGliposomes with β-amyloid in brain cells and in vitro phagocytes in mice aged 16 months and 10 months of age | Treatment lowered the ratio of phosphorylated Tau to total Tau. Therapeutic efficacy of immunoliposome treatment was superior to free monoclonal antibody administration. | Sensitivity and specificity missing. | Pretreatment missing | -Antibody: 6E10 anti-Aβ -Kit: Qubits Protein Assay Kit. |
| Permanne, et al. 1995 [67] | France | Aβ | Dot-blot and Western Blot | 17 elderly with AD and no AD | Method detects amyloid-associated components such as apolipoprotein E. | High sensitivity | Formic acid | |
| Plagg, et al. 2015 [68] | Austria | APP | Elisa and Western Blot | 73 patients with AD (43) and controls (30) | Decreased platelet APP isoforms in AD patients, APP beta altered in humans and mice with AD, lower EGF levels in human AD patients. | Highly sensitive. | Pretreatment missing | -Antibody: biotinylated antibody CD62P -Kit: sAPPb-w (highly sensitive) Assay Kit—IBL |
| Read, et al. 1992 [34] | The Netherlands | APP | Elisa | 4 ventricular and lumbar CSF samples ventricular with AD | Low APP levels as a diagnostic marker for AD | Sensitivity and specificity missing. | Pretreatment missing | -Antibody: not mentioned -Kit: not mentioned |
| Riemenschneider, et al. 2002 [69] | Germany | CSF Aβ42 and CSF T-tau | Elisa | 28 MCI patients, 10 of whom developed AD | Results indicate that altered tau and Aβ42 concentration can be detected in MCI patients but with pathological changes of AD | Sensitivity 90%, specificity 90% | Pretreatment missing | -Antibody: not metioned -Kit: Innogenetics, Zwjindrecht, Belgium |
| Ringman, et al. 2008 [70] | United States | CSF Aβ42, Aβ42/Aβ40 ratio, CSF T-tau, and CSF P-tau181 | Elisa | CSF biomarker results were compared in 7 asymptomatic carriers of familial AD (FAD)-associated mutations and four non-carriers | Asymptomatic FAD mutation carriers had abnormal CSF biomarkers already in their 30 s | Sensitivity and specificity missing | Pretreatment missing | -Antibody: Takeda BAN50/BA27 and BNT77/BC05 antibodies -Kit: not mentioned |
| Rogeberg, et al. 2015 [71] | Norway | Aβ | Mass spectrometry (MS), sandwich Elisa, Immunoprecipitation | 19 samples of CSF of AD patients and 9 control healthy patients | Method could be used to assess disease-modifying therapies directed at Aβ production or degradation. | Sensitivity and specificity missing | Pretreatment missing | -Antibodies: 4G8, 6E10, and a 12EF325 mid-domain antibody -Kit: Innotest hTau Ag, Phospho-Tau 181P, β-amyloid (1-42) |
| Roqanian, et al. 2017 [72] | Iran | HEWL Oligomers | ThT fluorescent assay, Nile red binding assay | Cerebral rat samples | Polyphenols frequently interacting with amyloid aggregates may serve as a therapeutic approach for amyloid-related diseases. | Sensitivity and specificity missing. | IA | |
| Ruiz, et al. 2015 [73] | Mexico | Aβ fibrils | DLS | Aggregated β-amyloid fibrils | IA | Sensitivity and specificity missing. | IA | |
| Salvadores, et al. 2014 [74] | Italy | Aβ oligomers/PrP | PMCA | 50 samples of CSF patients with AD + 30 healthy control patients + 39 other neurological diseases | AD patients from control individuals affected by a variety of other neurodegenerative disorders or nondegenerative neurological diseases | Sensitivity of 90% and specificity of 92% | IA | |
| Shaw, et al. 2009 [76] | United States | CSF Aβ42, CSF T-tau, and CSF P-tau181 | Multiplex immunoassay and xMAP Luminex | 196 MCI patients, 37 of whom developed AD | CSF Aβ1-42 was the most sensitive biomarker for AD in the autopsy cohort of CSF | CSF T-tau/Aβ42 had a sensitivity of 89% for MCI cases with progression to AD | Pretreatment missing. | |
| Skoog, et al. 2003 [77] | Sweden | CSF Aβ42 | Sandwich Elisa | 35 non-demented 85 year olds underwent LP and were followed for 3 years | Low levels of CSF Aβ42 predicted progression to dementia | Sensitivity and specificity missing. | Pretreatment missing. | -Antibody: 3D6 -Kit: not mentioned |
| Stomrud, et al. 2007 [80] | Sweden | CSF Aβ42 | xMAP technology | 57 cognitively normal controls underwent LP and were followed for 3 years | Low levels of CSF Aβ42 predicted cognitive decline | Sensitivity of 71.4% and a specificity of 75.7% | IA | |
| Tamaoka, et al. 1997 [81] | Japan | Aβ | Elisa and sandwich Elisa | CSF of AD patient samples and 34 without AD | CSF-Aβ42(43) could reflect increased amino terminal truncations and/or modifications of Aβ42(43) in AD brains | Sensitivity and specificity missing. | Pretreatment missing. | -Antibodies: BNT77 (anti-Aβ11-28) and BAN50 (anti-Aβ1-16) -Kit: not mentioned |
| Wang, et al. 2012 [82] | China | β-amyloid with alfa-M | Dot blot assay, Western Blot and ThT fluorescence | Primary rat cerebral cortical neurons | a-M could be a great potential candidate for AD treatment | Sensitivity and specificity missing. | Pretreatment missing. | |
| Wang, et al. 2016 [38] | China | Dynamin 1 | Western Blot, Immunoblot, Dot Blot and Elisa | Brain samples of passively immunized 3× Tg-AD mice control and wild-type | Passive immunization with Aβ42 possible treatment | Sensitivity and specificity missing. | Pretreatment missing. | -Antibody: not mentioned -Kit: Biosource ELISA kits (Invitrogen, Carlsbad, CA, USA) |
| Wang, et al. 2017 [83] | United States | Solubilized immunoprecipitates | Western Blot | Triple-transgenic (3× Tg) AD mice | PTI-125 is the first therapeutic candidate for AD | Sensitivity and specificity missing. | Pretreatment missing. | |
| Xie, et al. 2017 [30] | China | Aβ aggregates | Thioflavin T fluorescent assay | Human neuroblastoma SH-SY5Y cells | A-HSA worked as a bifunctional inhibitor against Cu2+-mediated Aβ42 aggregation and cytotoxicity under a mildly acidic condition | Sensitivity and specificity missing. | Acid conditions | |
| Yang, et al. 2013 [84] | United States | Aβ oligomers | Sandwich Elisa, Immunoprecipitation and Western Blot | Human brain samples with AD and no AD and transgenic mouse brains | New o-ELISAs method for biomarker AD | Sensitivity and specificity missing. | IA | -Antibodies: (MAb) 266 to the Aßmidregion (residues 13–28) or MAb, 3D6 (to residues 1–5), Mab 4G8 (to residues 18–22; D6 or MAbNAB61, MAbs266 and 3D6 were kindly provided by Elan, plc -Kit: not mentioned |
| Yang, et al. 2014 [85] | United States | Gelsolin | Western Blot and sandwich Elisa | 26 APPswe/PS1 transgenic mice blood samples | Gelsolin decreases Aβ | Sensitivity and specificity missing. | TSA | -Antibody: 6E10 -Kit: not mentioned |
| Zetterberg, et al. 2003 [86] | Sweden | CSF Aβ42, CSF T-tau, and CSF P-tau181 | Electroencephalogram, MRI, single photon emission, computed tomography and MMSE | 53 MCI patients, 22 of whom developed AD | Missing information. | Sensitivity 68%, specificity 97%, PPV 94%, NPV 81% | IA | |
| Zhao, et al. 2009 [26] | China | Aβ oligomers | Immunofluorescent microscopy | 8 nondiabetic control subjects, 8 type 2 diabetic cases without islet amyloid, and 8 type 2 diabetic patients with islet amyloid | Large oligomers were spatially localized adjacent to amyloid fibrils and were associated with apoptosis | Sensitivity and specificity missing. | IA |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Avila-Vazquez, M.F.; Altamirano-Bustamante, N.F.; Altamirano-Bustamante, M.M. Amyloid Biomarkers in Conformational Diseases at Face Value: A Systematic Review. Molecules 2018, 23, 79. https://doi.org/10.3390/molecules23010079
Avila-Vazquez MF, Altamirano-Bustamante NF, Altamirano-Bustamante MM. Amyloid Biomarkers in Conformational Diseases at Face Value: A Systematic Review. Molecules. 2018; 23(1):79. https://doi.org/10.3390/molecules23010079
Chicago/Turabian StyleAvila-Vazquez, Maria Fernanda, Nelly F. Altamirano-Bustamante, and Myriam M. Altamirano-Bustamante. 2018. "Amyloid Biomarkers in Conformational Diseases at Face Value: A Systematic Review" Molecules 23, no. 1: 79. https://doi.org/10.3390/molecules23010079
APA StyleAvila-Vazquez, M. F., Altamirano-Bustamante, N. F., & Altamirano-Bustamante, M. M. (2018). Amyloid Biomarkers in Conformational Diseases at Face Value: A Systematic Review. Molecules, 23(1), 79. https://doi.org/10.3390/molecules23010079





