Curcumin Attenuates on Carbon Tetrachloride-Induced Acute Liver Injury in Mice via Modulation of the Nrf2/HO-1 and TGF-β1/Smad3 Pathway
Abstract
:1. Introduction
2. Results
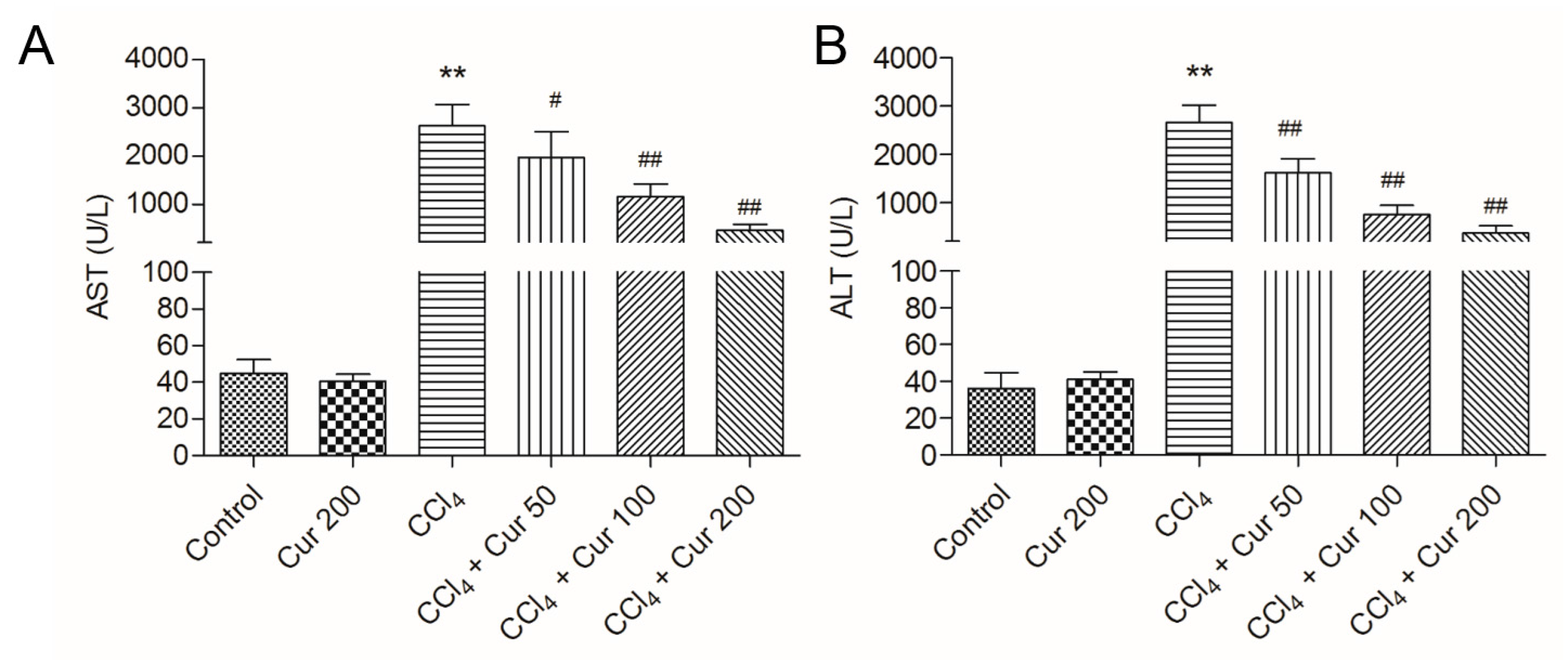
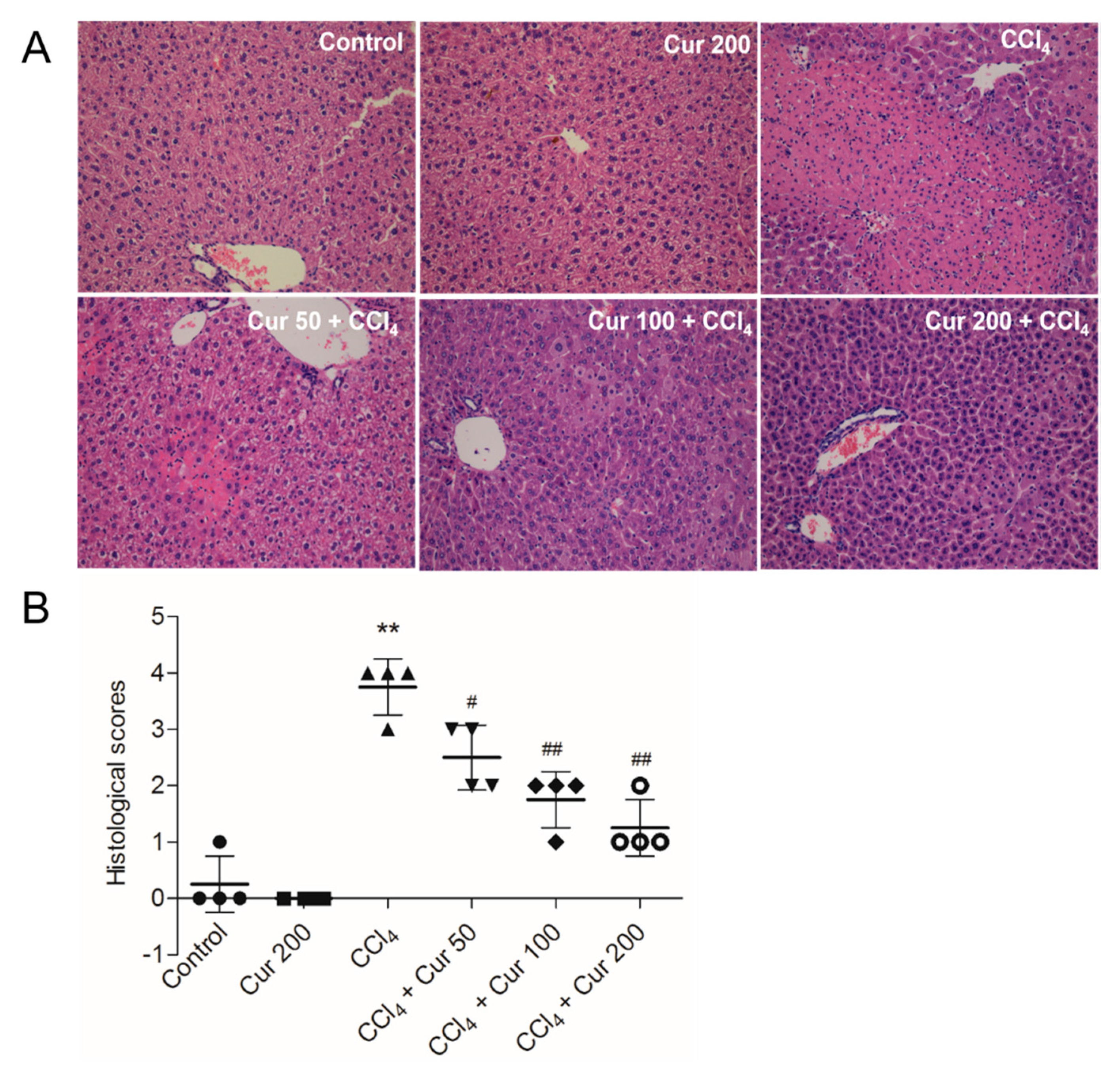
2.1. Curcumin Ameliorates CCl4-Induced Acute Liver Injury in Mice
2.2. Curcumin Ameliorates CCl4-Induced Oxidative Stress in Liver Tissue
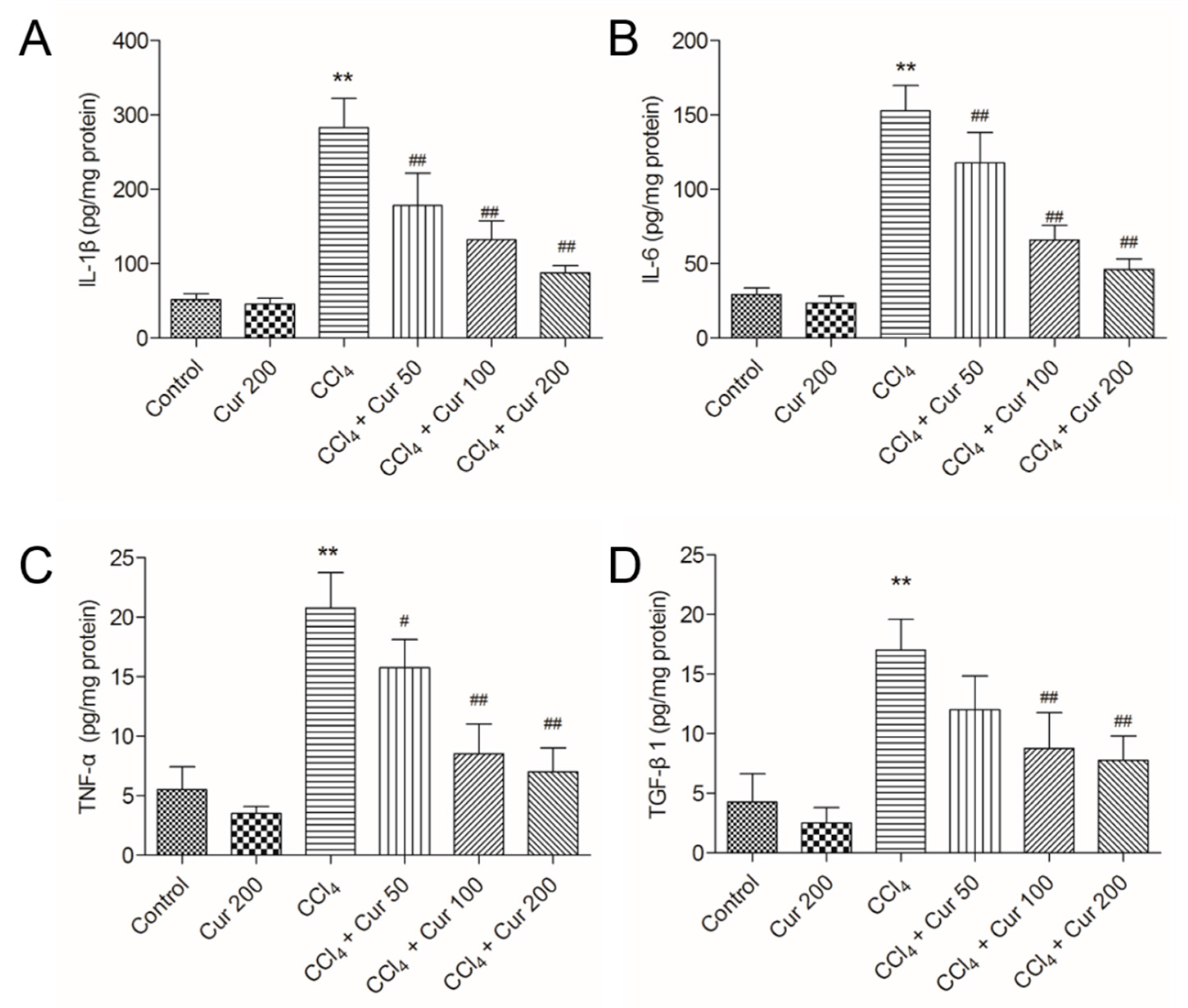
2.3. Curcumin Ameliorates CCl4-Induced Inflammatory Response in Livers of Mice
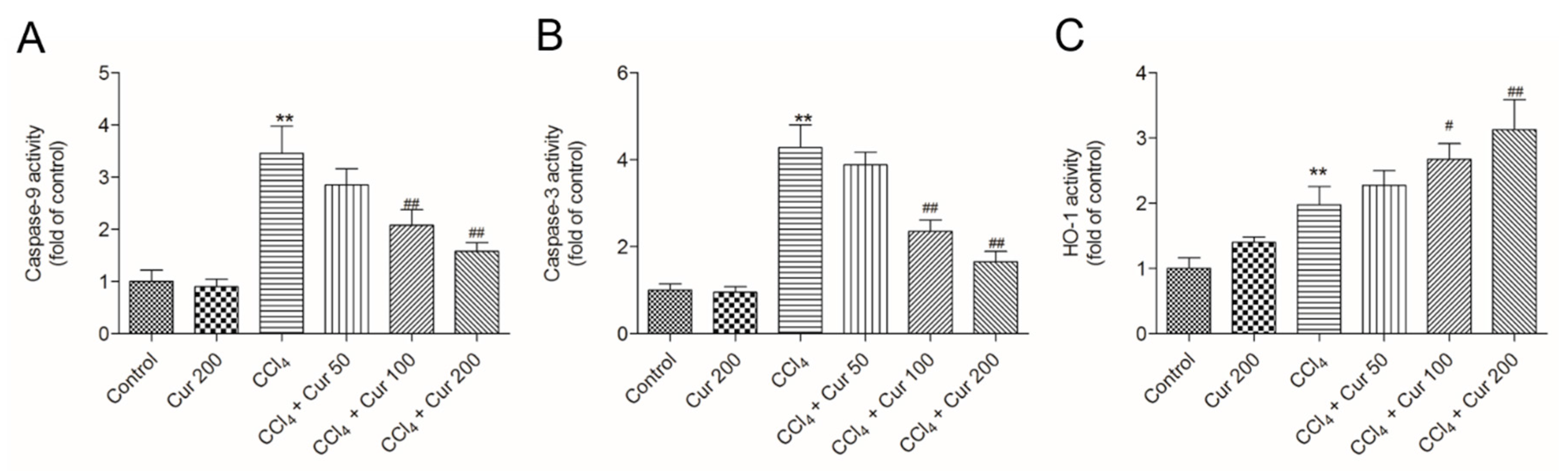
2.4. Curcumin Down-Regulates the Activities of Caspase-9 and -3
2.5. Curcumin Up-Regulates Nrf2 Expression and Down-Regulates the Expressions of TGF-β1 and Smad3 Proteins
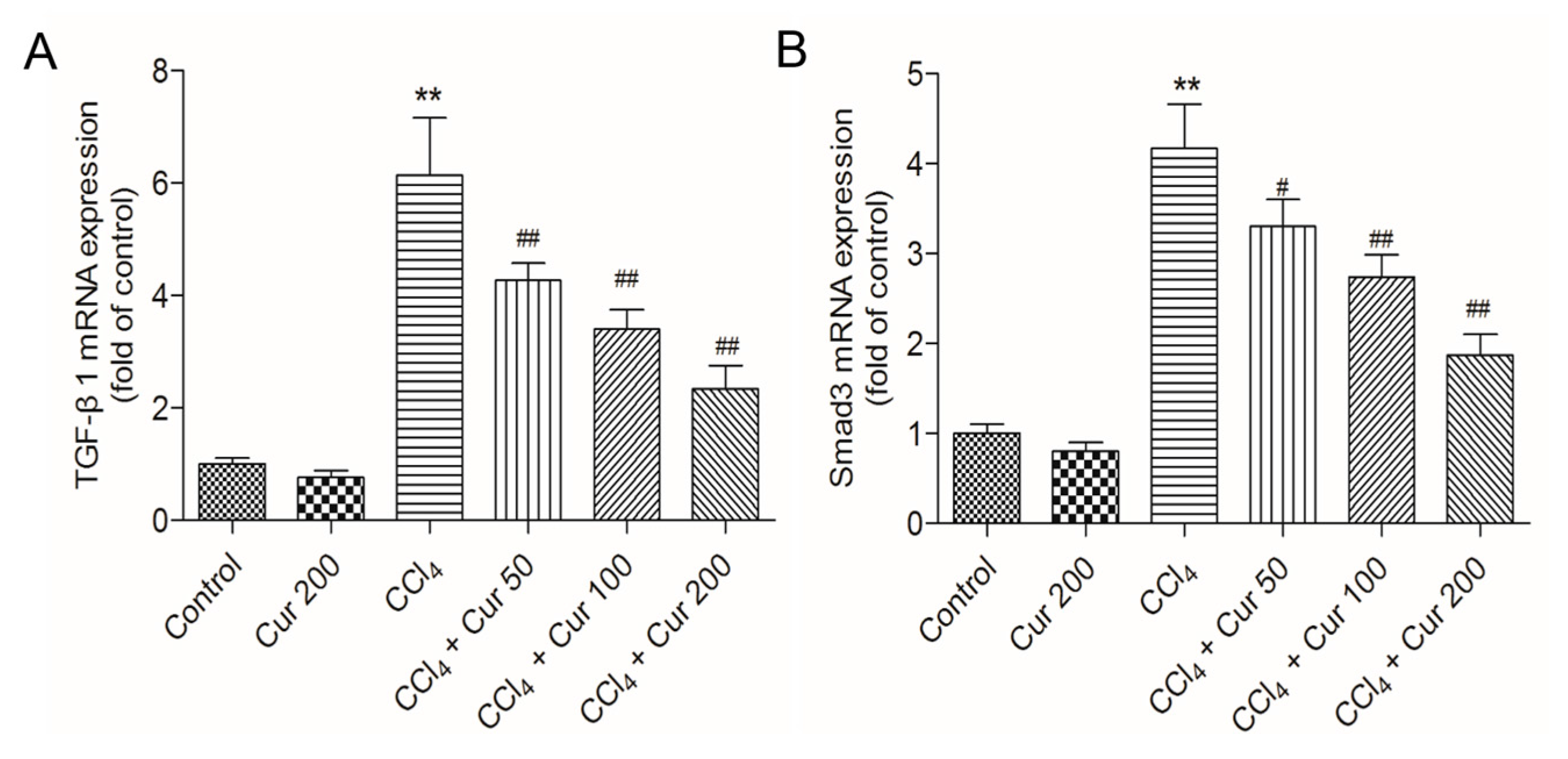
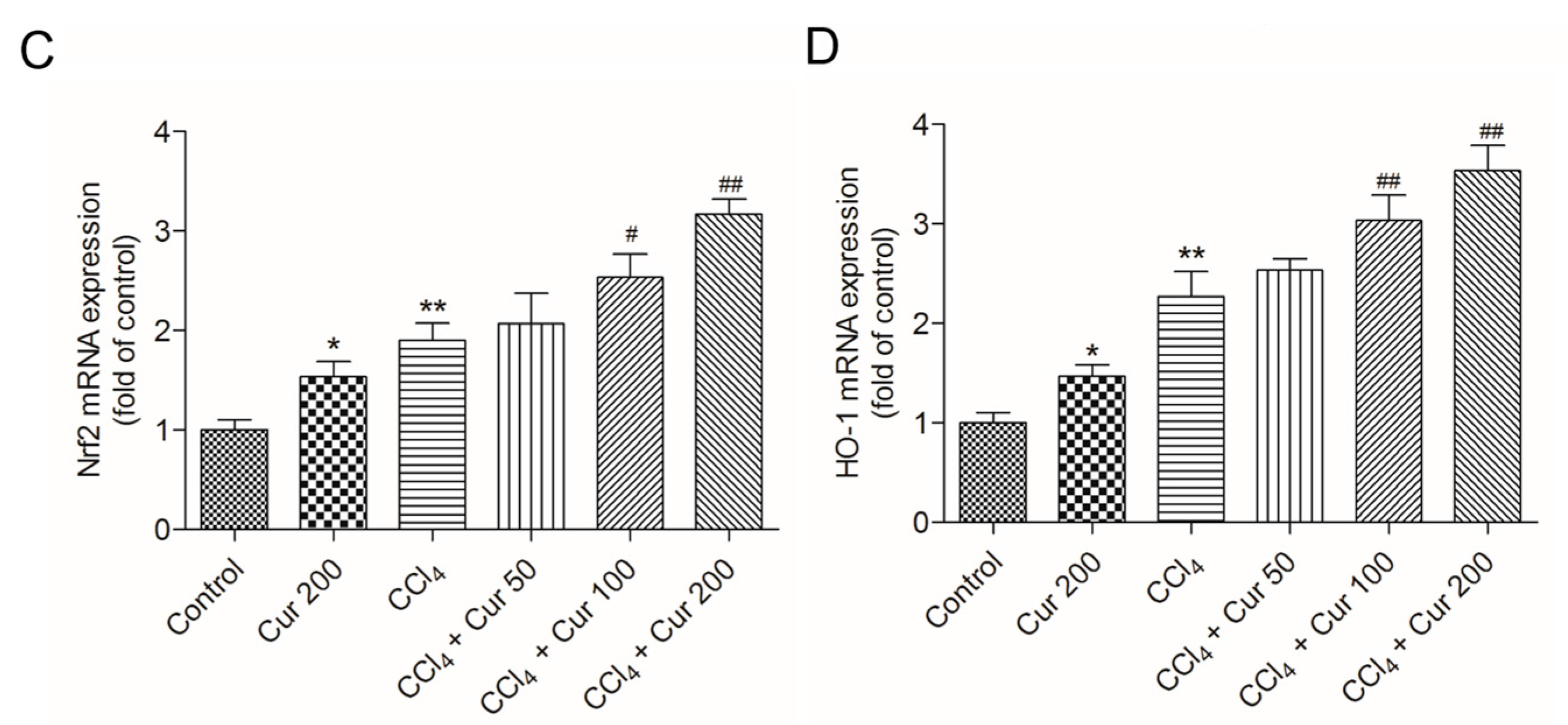
2.6. Curcumin Down-Regulates the Expressions of TGF-β and Smad3 mRNAs and Up-Regulates the Expressions of Nrf2 and HO-1 mRNAs
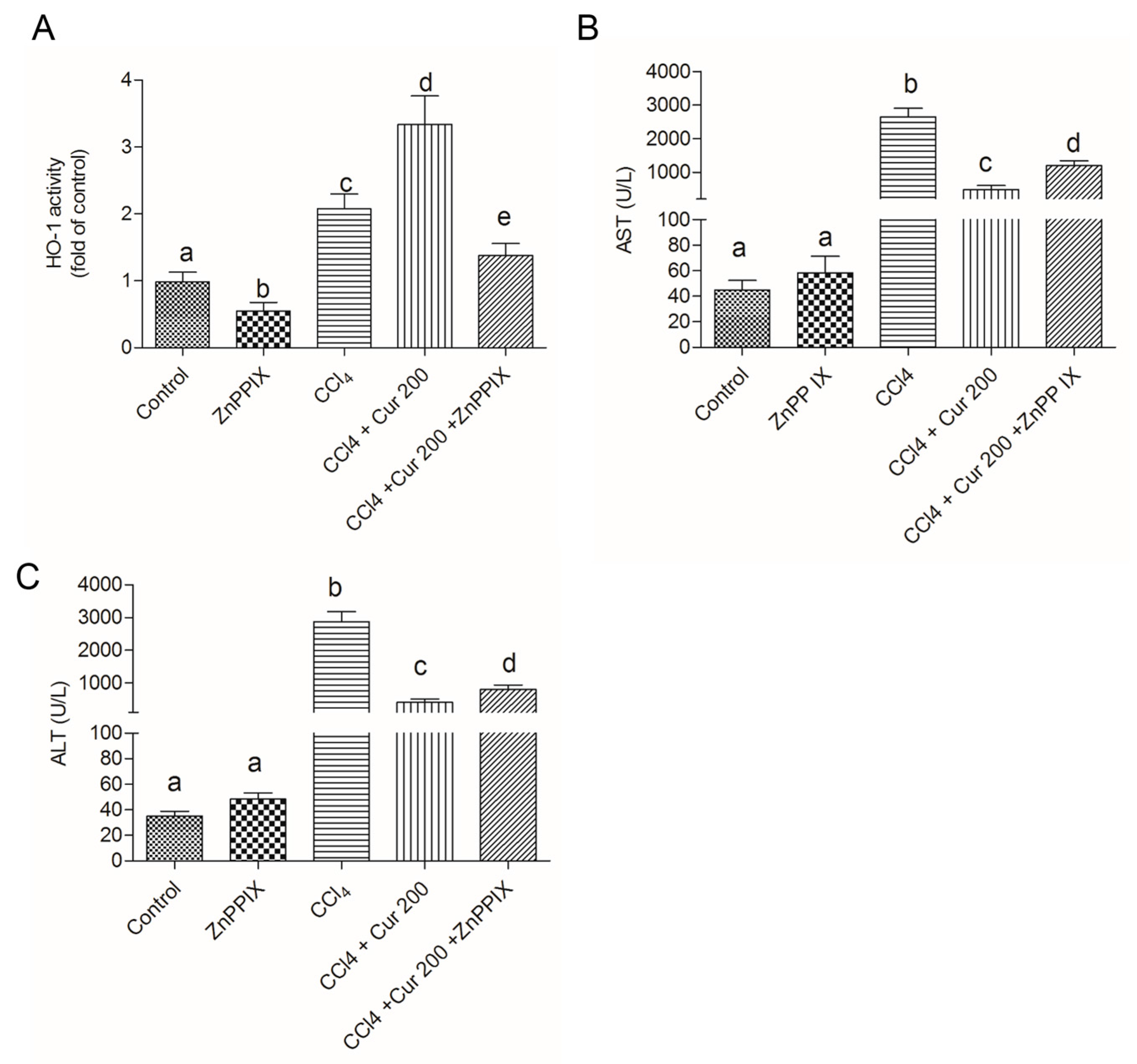
2.7. Inhibition of HO-1 Abolished the Protective Effect of Curcumin on CCl4-Induced Liver Injury
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Animal Treatment
4.3. Measurement of Serum Alanine Transaminase (ALT) and Aspartate Transaminase (AST)
4.4. Histological Examination of Liver Damage
4.5. Measurement of Activities of Superoxide Dismutase (SOD), Catalase (CAT) and Levels of Malondialdehyde (MDA) and Glutathione (GSH)
4.6. Measurement of Caspase-9, -3 and HO-1 Activities
4.7. Measurement of TNF-α, IL-1β, IL-6 and TGF-β1 Levels
4.8. Western Blotting
4.9. RNA Extraction and Real-Time Quantitative Polymerase Chain Reaction (PCR)
4.10. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Taub, R. Liver regeneration: From myth to mechanism. Nat. Rev. Mol. Cell Biol. 2004, 5, 836–847. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Han, M.; Chen, T.; Yan, W.; Ning, Q. Acute liver failure: Mechanisms of immune-mediated liver injury. Liver Int. 2010, 30, 782–794. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Han, W.; Shi, H.; Ren, F.; Chen, D.; Chen, Y.; Duan, Z. Augmenter of liver regeneration protects against carbon tetrachloride-induced liver injury by promoting autophagy in mice. Oncotarget 2017, 8, 12637–12648. [Google Scholar] [PubMed]
- Jaeschke, H.; McGill, M.R.; Ramachandran, A. Oxidant stress, mitochondria, and cell death mechanisms in drug-induced liver injury: Lessons learned from acetaminophen hepatotoxicity. Drug Metab. Rev. 2012, 44, 88–106. [Google Scholar] [CrossRef] [PubMed]
- Niu, L.; Cui, X.; Qi, Y.; Xie, D.; Wu, Q.; Chen, X.; Ge, J.; Liu, Z. Involvement of TGF-β1/Smad3 signaling in carbon tetrachloride-induced acute liver injury in mice. PLoS ONE 2016, 11, e0156090. [Google Scholar] [CrossRef] [PubMed]
- Cong, M.; Zhao, W.; Liu, T.; Wang, P.; Fan, X.; Zhai, Q.; Bao, X.; Zhang, D.; You, H.; Kisseleva, T.; et al. Protective effect of human serum amyloid P on CCl4-induced acute liver injury in mice. Int. J. Mol. Med. 2017, 40, 454–464. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.G.; Zhang, C.; Wang, J.X.; Wang, B.W.; Wang, H.; Zhang, Z.H.; Chen, Y.H.; Lu, Y.; Tao, L.; Wang, J.Q.; et al. Obeticholic acid protects against carbon tetrachloride-induced acute liver injury and inflammation. Toxicol. Appl. Pharmacol. 2017, 314, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.; Liong, E.C.; Huang, H.; On Tse, W.; Lau, K.S.; Pan, J.; Nanji, A.A.; Fung, M.L.; Xing, F.; Tipoe, G.L. Cyclooxygenase-1 serves a vital hepato-protective function in chemically induced acute liver injury. Toxicol. Sci. 2015, 143, 430–440. [Google Scholar] [CrossRef] [PubMed]
- Ding, J.; Cui, X.; Liu, Q. Emerging role of HMGB1 in lung diseases: Friend or foe. J. Cell. Mol. Med. 2016, 21, 1046–1057. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Xiong, J.B.; Ma, K.; Wang, A.Z.; Qian, K.J. Rac2 deficiency attenuates CCl4-induced liver injury through suppressing inflammation and oxidative stress. Biomed. Pharmacother. 2017, 94, 140–149. [Google Scholar] [CrossRef] [PubMed]
- Zeng, B.; Su, M.; Chen, Q.; Chang, Q.; Wang, W.; Li, H. Protective effect of a polysaccharide from anoectochilus roxburghii against carbon tetrachloride-induced acute liver injury in mice. J. Ethnopharmacol. 2017, 200, 124–135. [Google Scholar] [CrossRef] [PubMed]
- Torres, L.R.; Santana, F.C.; Torres-Leal, F.L.; Melo, I.L.; Yoshime, L.T.; Matos-Neto, E.M.; Seelaender, M.C.; Araujo, C.M.; Cogliati, B.; Mancini-Filho, J. Pequi (Caryocar brasiliense camb.) almond oil attenuates carbon tetrachloride-induced acute hepatic injury in rats: Antioxidant and anti-inflammatory effects. Food Chem. Toxicol. 2016, 97, 205–216. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Pan, H.; Chang, R.C.; So, K.F.; Brecha, N.C.; Pu, M. Activation of the Nrf2/HO-1 antioxidant pathway contributes to the protective effects of lycium barbarum polysaccharides in the rodent retina after ischemia-reperfusion-induced damage. PLoS ONE 2014, 9, e84800. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zoja, C.; Benigni, A.; Remuzzi, G. The Nrf2 pathway in the progression of renal disease. Nephrol. Dial. Transplant. 2014, 29 (Suppl. S1), i19–i24. [Google Scholar] [CrossRef] [PubMed]
- Cao, M.; Wang, H.; Guo, L.; Yang, S.; Liu, C.; Khor, T.O.; Yu, S.; Kong, A.N. Dibenzoylmethane protects against CCl4-induced acute liver injury by activating Nrf2 via jnk, ampk, and calcium signaling. AAPS J. 2017, 19, 1703–1714. [Google Scholar] [CrossRef] [PubMed]
- Pan, C.W.; Yang, S.X.; Pan, Z.Z.; Zheng, B.; Wang, J.Z.; Lu, G.R.; Xue, Z.X.; Xu, C.L. Andrographolide ameliorates d-galactosamine/lipopolysaccharide-induced acute liver injury by activating Nrf2 signaling pathway. Oncotarget 2017, 8, 41202–41210. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.L.; Chu, J.G.; Jian, X.M.; Dong, J.Z.; Wang, L.P.; Li, G.X.; Yang, N.B. Curcumin attenuates lipopolysaccharide/d-galactosamine-induced acute liver injury by activating Nrf2 nuclear translocation and inhibiting NF-kB activation. Biomed. Pharmacother. 2017, 91, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Bellezza, I.; Tucci, A.; Galli, F.; Grottelli, S.; Mierla, A.L.; Pilolli, F.; Minelli, A. Inhibition of NF-kB nuclear translocation via HO-1 activation underlies α-tocopheryl succinate toxicity. J. Nutr. Biochem. 2012, 23, 1583–1591. [Google Scholar] [CrossRef] [PubMed]
- Oh, C.J.; Kim, J.Y.; Choi, Y.K.; Kim, H.J.; Jeong, J.Y.; Bae, K.H.; Park, K.G.; Lee, I.K. Dimethylfumarate attenuates renal fibrosis via NF-E2-related factor 2-mediated inhibition of transforming growth factor-beta/smad signaling. PLoS ONE 2012, 7, e45870. [Google Scholar] [CrossRef] [PubMed]
- Yodkeeree, S.; Chaiwangyen, W.; Garbisa, S.; Limtrakul, P. Curcumin, demethoxycurcumin and bisdemethoxycurcumin differentially inhibit cancer cell invasion through the down-regulation of MMPs and uPA. J. Nutr. Biochem. 2009, 20, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Sandur, S.K.; Pandey, M.K.; Sung, B.; Ahn, K.S.; Murakami, A.; Sethi, G.; Limtrakul, P.; Badmaev, V.; Aggarwal, B.B. Curcumin, demethoxycurcumin, bisdemethoxycurcumin, tetrahydrocurcumin and turmerones differentially regulate anti-inflammatory and anti-proliferative responses through a ros-independent mechanism. Carcinogenesis 2007, 28, 1765–1773. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.T.; Ma, W.; Lu, Y.P.; Chang, R.L.; Fisher, C.; Manchand, P.S.; Newmark, H.L.; Conney, A.H. Effects of curcumin, demethoxycurcumin, bisdemethoxycurcumin and tetrahydrocurcumin on 12-O-tetradecanoylphorbol-13-acetate-induced tumor promotion. Carcinogenesis 1995, 16, 2493–2497. [Google Scholar] [CrossRef] [PubMed]
- Dai, C.; Lei, L.; Li, B.; Lin, Y.; Xiao, X.; Tang, S. Involvement of the activation of Nrf2/HO-1, p38 MAPK signaling pathways and endoplasmic reticulum stress in furazolidone induced cytotoxicity and S phase arrest in human hepatocyte L02 cells: Modulation of curcumin. Toxicol. Mech. Methods 2017, 27, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Dai, C.; Ciccotosto, G.D.; Cappai, R.; Tang, S.; Li, D.; Xie, S.; Xiao, X.; Velkov, T. Curcumin attenuates colistin-induced neurotoxicity in N2a cells via anti-inflammatory activity, suppression of oxidative stress, and apoptosis. Mol. Neurobiol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Dai, C.; Li, B.; Zhou, Y.; Li, D.; Zhang, S.; Li, H.; Xiao, X.; Tang, S. Curcumin attenuates quinocetone induced apoptosis and inflammation via the opposite modulation of Nrf2/HO-1 and NF-kB pathway in human hepatocyte L02 cells. Food Chem. Toxicol. 2016, 95, 52–63. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.; Lin, S.H.; Yang, Y.Z.; Guo, R.; Cao, J.; Liu, Q. The effect of curcumin on sepsis-induced acute lung injury in a rat model through the inhibition of the TGF-β1/Smad3 pathway. Int. Immunopharmacol. 2013, 16, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Zhu, R.Z.; Xiang, D.; Xie, C.; Li, J.J.; Hu, J.J.; He, H.L.; Yuan, Y.S.; Gao, J.; Han, W.; Yu, Y. Protective effect of recombinant human IL-1Ra on CCl4-induced acute liver injury in mice. World J. Gastroenterol. 2010, 16, 2771–2779. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.Y.; Zhang, X.Y.; Guan, S.W.; Hua, Z.C. Protective effect of procyanidin B2 against CCl4-induced acute liver injury in mice. Molecules 2015, 20, 12250–12265. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.C.; Zhang, L.; Yu, H.X.; Sun, Z.; Lin, X.F.; Tan, C.; Lu, R.R. Curcumin protects mouse neuroblastoma Neuro-2A cells against hydrogen-peroxide-induced oxidative stress. Food Chem. 2011, 129, 387–394. [Google Scholar] [CrossRef]
- Sahin, K.; Orhan, C.; Tuzcu, Z.; Tuzcu, M.; Sahin, N. Curcumin ameloriates heat stress via inhibition of oxidative stress and modulation of Nrf2/HO-1 pathway in quail. Food Chem. Toxicol. 2012, 50, 4035–4041. [Google Scholar] [CrossRef] [PubMed]
- Tokac, M.; Taner, G.; Aydin, S.; Ozkardes, A.B.; Dundar, H.Z.; Taslipinar, M.Y.; Arikok, A.T.; Kilic, M.; Basaran, A.A.; Basaran, N. Protective effects of curcumin against oxidative stress parameters and DNA damage in the livers and kidneys of rats with biliary obstruction. Food Chem. Toxicol. 2013, 61, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Pan, X.; Fu, H.; Zheng, Y.; Dai, Y.; Yin, Y.; Chen, Q.; Hao, Q.; Bao, D.; Hou, D. Effect of curcumin on glycerol-induced acute kidney injury in rats. Sci. Rep. 2017, 7, 10114. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Xu, L.; Zhang, L.; Ying, Z.; Su, W.; Wang, T. Curcumin attenuates d-galactosamine/lipopolysaccharide-induced liver injury and mitochondrial dysfunction in mice. J. Nutr. 2014, 144, 1211–1218. [Google Scholar] [CrossRef] [PubMed]
- Lee, G.H.; Lee, H.Y.; Choi, M.K.; Chung, H.W.; Kim, S.W.; Chae, H.J. Protective effect of Curcuma longa L. Extract on CCl4-induced acute hepatic stress. BMC Res. Notes 2017, 10, 77. [Google Scholar] [CrossRef] [PubMed]
- Dai, C.; Li, D.; Gong, L.; Xiao, X.; Tang, S. Curcumin ameliorates furazolidone-induced DNA damage and apoptosis in human hepatocyte L02 cells by inhibiting ros production and mitochondrial pathway. Molecules 2016, 21, 1061. [Google Scholar] [CrossRef] [PubMed]
- Qiu, P.; Sun, J.; Man, S.; Yang, H.; Ma, L.; Yu, P.; Gao, W. Curcumin attenuates N-Nitrosodiethylamine-induced liver injury in mice by utilizing the method of metabonomics. J. Agric. Food Chem. 2017, 65, 2000–2007. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama, T.; Nagata, J.; Yamagishi, A.; Endoh, K.; Saito, M.; Yamada, K.; Yamada, S.; Umegaki, K. Selective protection of curcumin against carbon tetrachloride-induced inactivation of hepatic cytochrome P450 isozymes in rats. Life Sci. 2006, 78, 2188–2193. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Aisaki, K.; Ikawa, Y.; Wake, K. Evidence of hepatocyte apoptosis in rat liver after the administration of carbon tetrachloride. Am. J. Pathol. 1998, 153, 515–525. [Google Scholar] [CrossRef]
- Dai, C.; Tang, S.; Deng, S.; Zhang, S.; Zhou, Y.; Velkov, T.; Li, J.; Xiao, X. Lycopene attenuates colistin-induced nephrotoxicity in mice via activation of the Nrf2/HO-1 pathway. Antimicrob. Agents Chemother. 2015, 59, 579–585. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Liu, J.; Chen, T.M.; Lan, Q.; Zhang, Q.Y.; Liu, B.; Dai, D.; Zhang, W.D.; Hu, L.P.; Zhu, R.Z. Dihydromyricetin alleviates carbon tetrachloride-induced acute liver injury via JNK-dependent mechanism in mice. World J. Gastroenterol. 2015, 21, 5473–5481. [Google Scholar] [CrossRef] [PubMed]
- Sung, B.; Pandey, M.K.; Ahn, K.S.; Yi, T.; Chaturvedi, M.M.; Liu, M.; Aggarwal, B.B. Anacardic acid (6-nonadecyl salicylic acid), an inhibitor of histone acetyltransferase, suppresses expression of nuclear factor-kappaB-regulated gene products involved in cell survival, proliferation, invasion, and inflammation through inhibition of the inhibitory subunit of nuclear factor-kappaBalpha kinase, leading to potentiation of apoptosis. Blood 2008, 111, 4880–4891. [Google Scholar] [PubMed]
- Bansal, M.B.; Kovalovich, K.; Gupta, R.; Li, W.; Agarwal, A.; Radbill, B.; Alvarez, C.E.; Safadi, R.; Fiel, M.I.; Friedman, S.L.; et al. Interleukin-6 protects hepatocytes from CCl4-mediated necrosis and apoptosis in mice by reducing MMP-2 expression. J. Hepatol. 2005, 42, 548–556. [Google Scholar] [CrossRef] [PubMed]
- Wielockx, B.; Lannoy, K.; Shapiro, S.D.; Itoh, T.; Itohara, S.; Vandekerckhove, J.; Libert, C. Inhibition of matrix metalloproteinases blocks lethal hepatitis and apoptosis induced by tumor necrosis factor and allows safe antitumor therapy. Nat. Med. 2001, 7, 1202–1208. [Google Scholar] [CrossRef] [PubMed]
- Prakash, M.; Kale, S.; Ghosh, I.; Kundu, G.C.; Datta, K. Hyaluronan-binding protein 1 (HABP1/p32/gC1qR) induces melanoma cell migration and tumor growth by NF-kappa B dependent MMP-2 activation through integrin α(v)β(3) interaction. Cell. Signal. 2011, 23, 1563–1577. [Google Scholar] [CrossRef] [PubMed]
- Sodhi, K.; Puri, N.; Kim, D.H.; Hinds, T.D.; Stechschulte, L.A.; Favero, G.; Rodella, L.; Shapiro, J.I.; Jude, D.; Abraham, N.G. PPARδ binding to heme oxygenase 1 promoter prevents angiotensin II-induced adipocyte dysfunction in goldblatt hypertensive rats. Int. J. Obes. 2014, 38, 456–465. [Google Scholar] [CrossRef] [PubMed]
- Randle, L.E.; Goldring, C.E.; Benson, C.A.; Metcalfe, P.N.; Kitteringham, N.R.; Park, B.K.; Williams, D.P. Investigation of the effect of a panel of model hepatotoxins on the Nrf2-Keap1 defence response pathway in CD-1 mice. Toxicology 2008, 243, 249–260. [Google Scholar] [CrossRef] [PubMed]
- Su, C.; Xia, X.; Shi, Q.; Song, X.; Fu, J.; Xiao, C.; Chen, H.; Lu, B.; Sun, Z.; Wu, S.; et al. Neohesperidin dihydrochalcone versus CCl(4)-induced hepatic injury through different mechanisms: The implication of free radical scavenging and Nrf2 activation. J. Agric. Food Chem. 2015, 63, 5468–5475. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.H.; Kim, D.W.; Yun, N.; Choi, J.S.; Islam, M.N.; Kim, Y.S.; Lee, S.M. Protective effects of hyperoside against carbon tetrachloride-induced liver damage in mice. J. Nat. Prod. 2011, 74, 1055–1060. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.C.; Kim, S.H.; Baek, H.S.; Moon, C.; Kim, S.H.; Kim, Y.B.; Yun, W.K.; Kim, H.C.; Kim, J.C. Protective effects of diallyl disulfide on carbon tetrachloride-induced hepatotoxicity through activation of Nrf2. Environ. Toxicol. 2015, 30, 538–548. [Google Scholar] [CrossRef] [PubMed]
- Cai, Z.; Lou, Q.; Wang, F.; Li, E.; Sun, J.; Fang, H.; Xi, J.; Ju, L. N-acetylcysteine protects against liver injure induced by carbon tetrachloride via activation of the Nrf2/HO-1 pathway. Int. J. Clin. Exp. Pathol. 2015, 8, 8655–8662. [Google Scholar] [PubMed]
- Magesh, S.; Chen, Y.; Hu, L. Small molecule modulators of keap1-Nrf2-ARE pathway as potential preventive and therapeutic agents. Med. Res. Rev. 2012, 32, 687–726. [Google Scholar] [CrossRef] [PubMed]
- Kong, D.; Zhang, F.; Shao, J.; Wu, L.; Zhang, X.; Chen, L.; Lu, Y.; Zheng, S. Curcumin inhibits cobalt chloride-induced epithelial-to-mesenchymal transition associated with interference with TGF-β/smad signaling in hepatocytes. Lab. Investig. 2015, 95, 1234–1245. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Z.; Yue, L.; Zhao, W.; Yang, X.; Shu, G. Protective effects of protostemonine on LPS/GalN-induced acute liver failure: Roles of increased hepatic expression of heme oxygenase-1. Int. Immunopharmacol. 2015, 29, 798–807. [Google Scholar] [CrossRef] [PubMed]
- Li, S.Q.; Wang, D.M.; Shu, Y.J.; Wan, X.D.; Xu, Z.S.; Li, E.Z. Proper heat shock pretreatment reduces acute liver injury induced by carbon tetrachloride and accelerates liver repair in mice. J. Toxicol. Pathol. 2013, 26, 365–373. [Google Scholar] [CrossRef] [PubMed]
- Taye, A.; Ibrahim, B.M. Activation of renal haeme oxygenase-1 alleviates gentamicin-induced acute nephrotoxicity in rats. J. Pharm. Pharmacol. 2013, 65, 995–1004. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zhang, L.; Wang, F.; Shi, Y.; Ren, Y.; Liu, Q.; Cao, Y.; Duan, H. Attenuation of glomerular injury in diabetic mice with tert-butylhydroquinone through nuclear factor erythroid 2-related factor 2-dependent antioxidant gene activation. Am. J. Nephrol. 2011, 33, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Dai, C.; Tang, S.; Velkov, T.; Xiao, X. Colistin-induced apoptosis of neuroblastoma-2a cells involves the generation of reactive oxygen species, mitochondrial dysfunction, and autophagy. Mol. Neurobiol. 2016, 53, 4685–4700. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |

| Treatment Group | ||||||
|---|---|---|---|---|---|---|
| Biomarker | Control | Cur 200 | CCl4 | Cur 50 + CCl4 | Cur 100 + CCl4 | Cur 200 + CCl4 |
| MDA (mmol/mg protein) | 1.84 ± 0.22 | 1.77 ± 0.25 | 2.79 ± 0.31 ** | 2.54 ± 0.37 # | 2.41 ± 0.18 # | 2.14 ± 0.39 ## |
| SOD (U/mg protein) | 110.2 ± 24.20 | 116.4 ± 13.94 | 78.4 ± 10.5 ** | 84.6 ± 15.1 | 94.3 ± 12.1 ## | 99.7 ± 17.2 ## |
| CAT (U/mgprotein) | 130.7 ± 15.8 | 138.4 ± 21.4 | 86.5 ± 15.1 ** | 98.1 ± 10.6 # | 106.7 ± 20.1 # | 118.2 ± 17.8 ## |
| GSH (mmol/mg protein) | 57.1 ± 6.7 | 62.6 ± 8.1 | 38.2 ± 9.4 ** | 43.3 ± 7.8 | 46.8 ± 10.3 # | 50.4 ± 9.6 # |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peng, X.; Dai, C.; Liu, Q.; Li, J.; Qiu, J. Curcumin Attenuates on Carbon Tetrachloride-Induced Acute Liver Injury in Mice via Modulation of the Nrf2/HO-1 and TGF-β1/Smad3 Pathway. Molecules 2018, 23, 215. https://doi.org/10.3390/molecules23010215
Peng X, Dai C, Liu Q, Li J, Qiu J. Curcumin Attenuates on Carbon Tetrachloride-Induced Acute Liver Injury in Mice via Modulation of the Nrf2/HO-1 and TGF-β1/Smad3 Pathway. Molecules. 2018; 23(1):215. https://doi.org/10.3390/molecules23010215
Chicago/Turabian StylePeng, Xinyan, Chongshan Dai, Quanwen Liu, Junke Li, and Jingru Qiu. 2018. "Curcumin Attenuates on Carbon Tetrachloride-Induced Acute Liver Injury in Mice via Modulation of the Nrf2/HO-1 and TGF-β1/Smad3 Pathway" Molecules 23, no. 1: 215. https://doi.org/10.3390/molecules23010215
APA StylePeng, X., Dai, C., Liu, Q., Li, J., & Qiu, J. (2018). Curcumin Attenuates on Carbon Tetrachloride-Induced Acute Liver Injury in Mice via Modulation of the Nrf2/HO-1 and TGF-β1/Smad3 Pathway. Molecules, 23(1), 215. https://doi.org/10.3390/molecules23010215





