Mechanochemical Synthesis and Biological Evaluation of Novel Isoniazid Derivatives with Potent Antitubercular Activity
Abstract
:1. Introduction
2. Results and Discussion
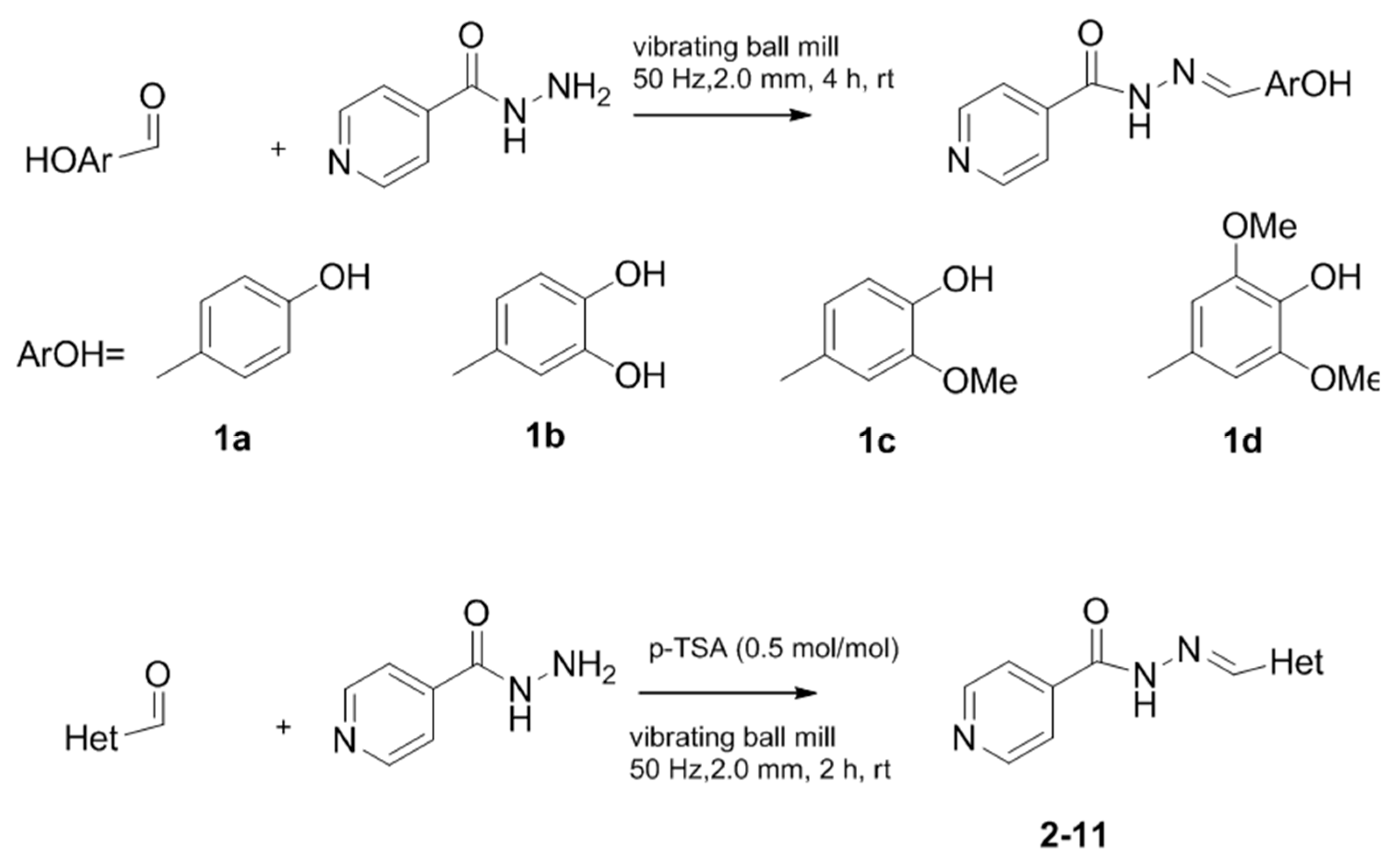
2.1. Chemistry
Mechanosynthesis of Isonicotinoyl Hydrazones
2.2. Structural Analysis of Isonicotinoyl Hydrazones by DFT and NMR: Determination of the Free Activation Energy (ΔG≠) between Conformers of Selected Compounds 1a and 5
- kexch = πΔν/√2 = 2.22·Δν (Hz)
- kexch = (kB·Tc/h)exp(−ΔG≠/RTc) (Eyringss equation)
- ΔG≠ = −RTcLn(kexch·h/kB·Tc)
2.3. Physicochemical Studies of Some Isonicotinoyl Hydrazones
2.3.1. Hydrolytic Stability
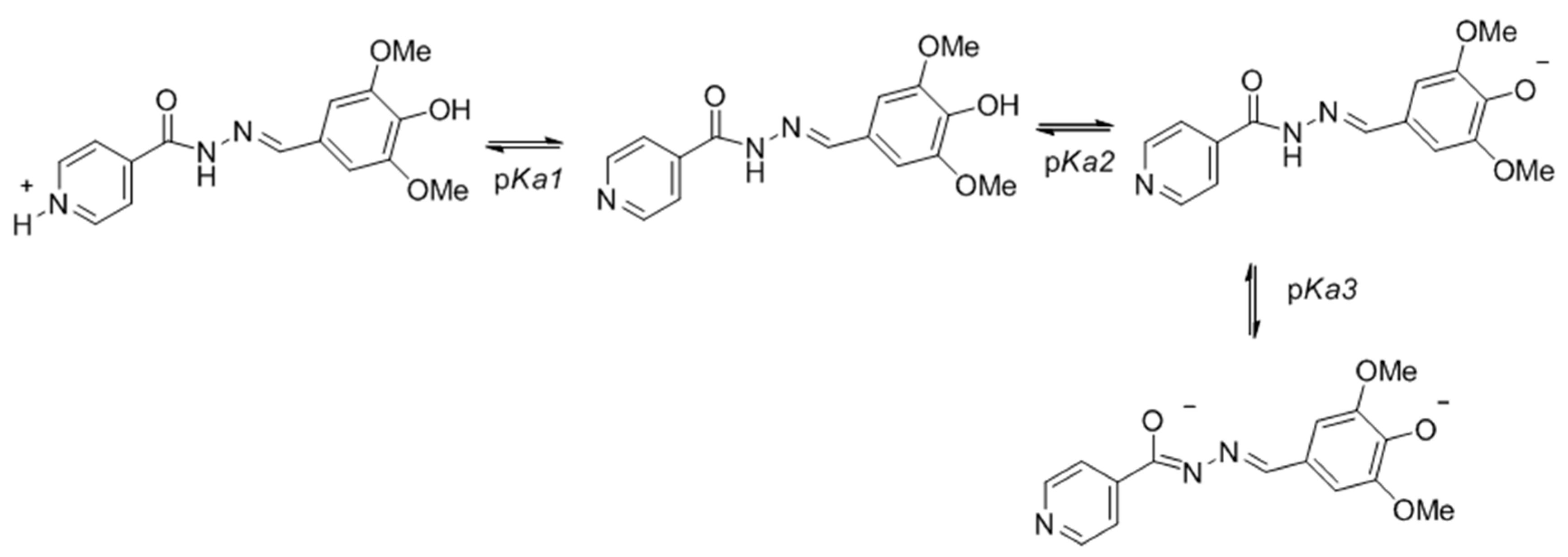
2.3.2. pKa Determination
2.4. Biology of Mechanochemically Synthesized Hydrazones
2.4.1. InhA Inhibition Assay
2.4.2. Activity of Phenolic Isonicotinoyl Hydrazones (1a–d) against M.tb Cell Growth
2.4.3. M.tb H37Rv Growth Inhibition Assays of Nitrogen Heterocyclic Hydrazones (2–11)
2.4.4. Cytotoxicity and Selectivity Index Determination
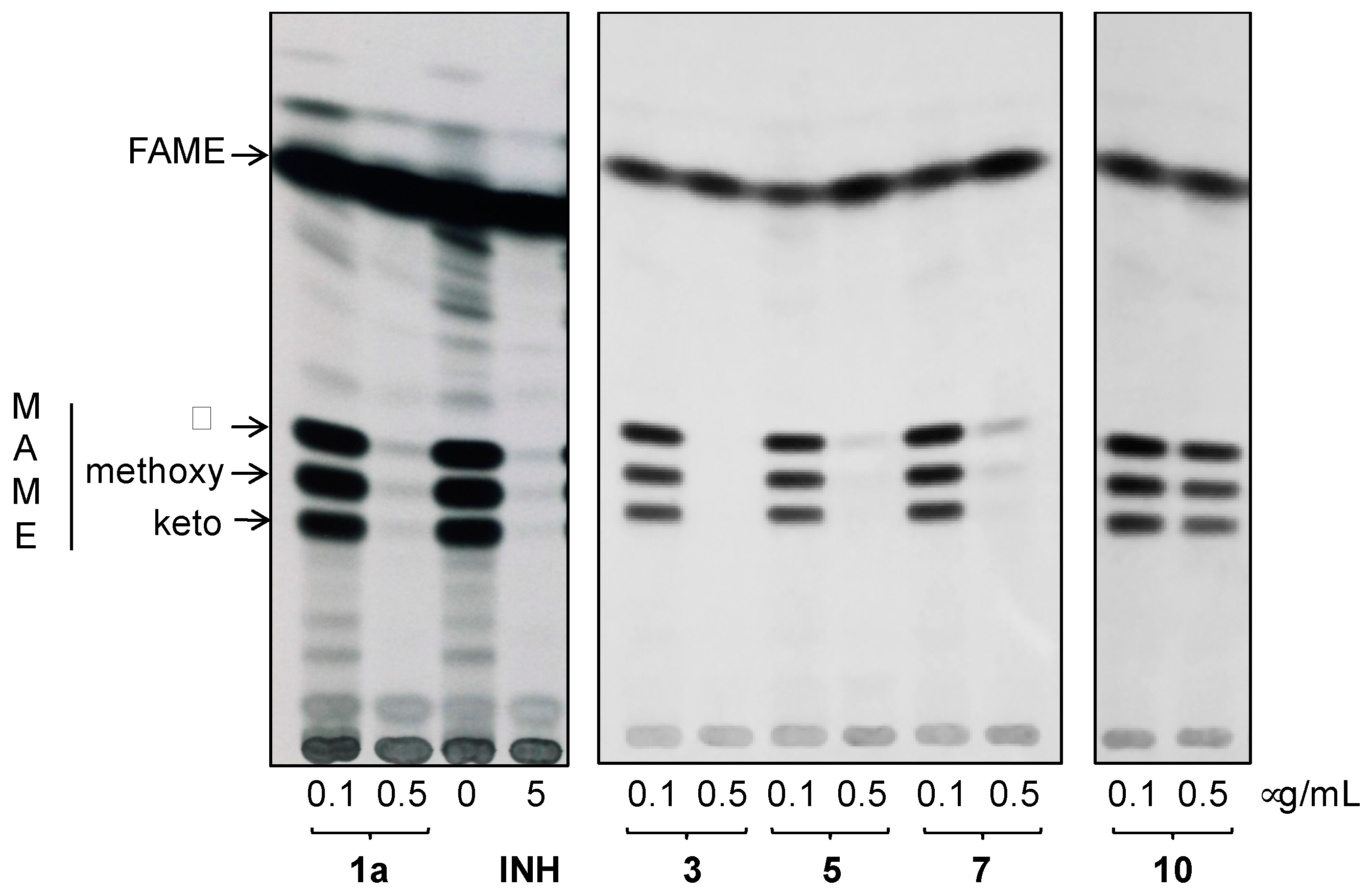
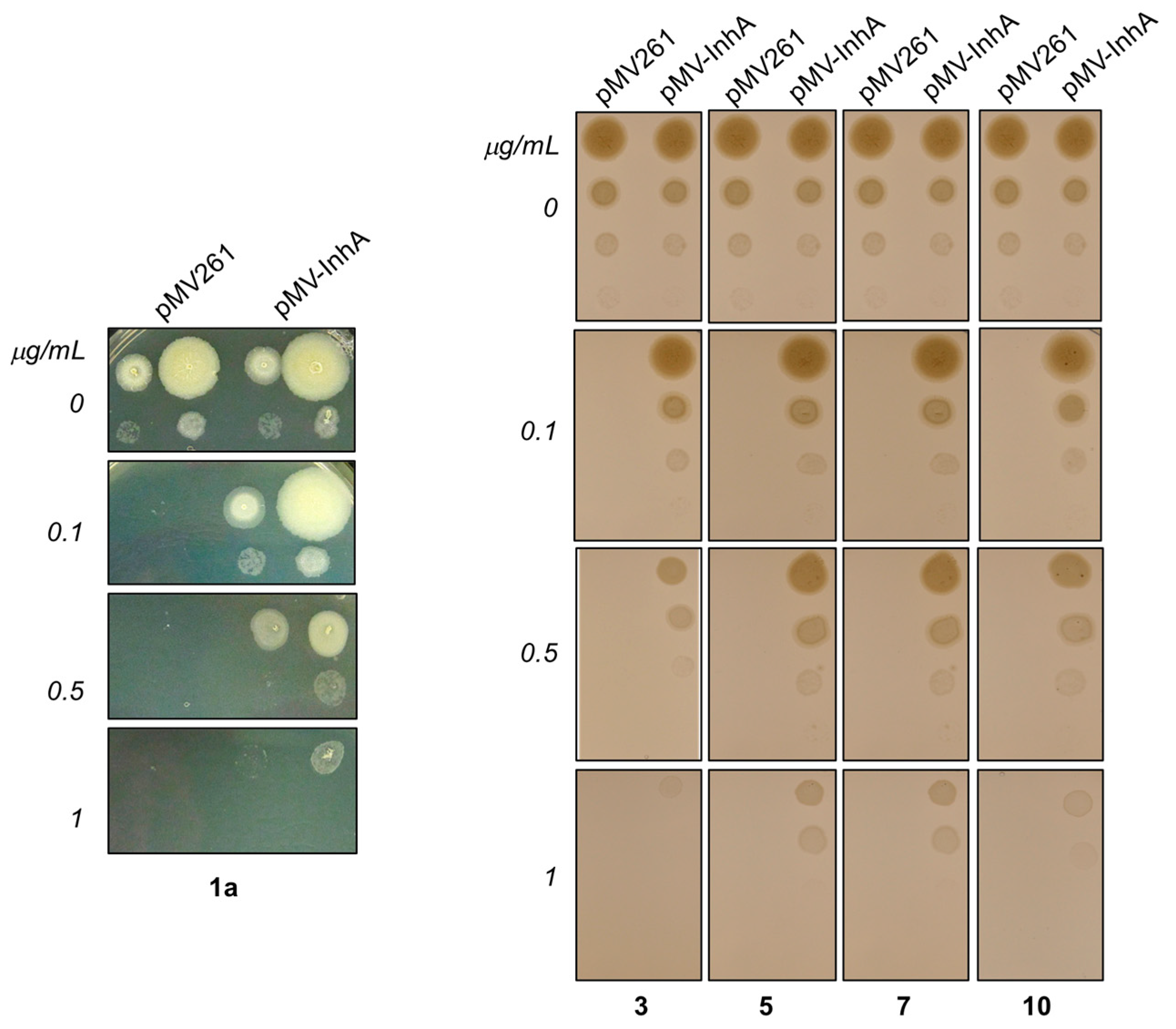
2.4.5. In Search for the Molecular Target of Prepared Hydrazones in Mycobacteria
3. Materials and Methods
3.1. Material
3.2. Chemistry
3.2.1. General Procedure for Phenolic Isonicotinoyl Hydrazones Synthesis
3.2.2. General Procedure for Isoniazid Nitrogen-Containing Heterocycles Derivatives 2–11
(E)-N′-((1H-imidazol-4-yl)methylene)isonicotinohydrazide (2)

- m.p.: 296.7 °C (dec.). Rf: 0.1 EtOAc/MeOH (4:1 v/v).
- 1H-NMR (300 MHz, DMSO-d6) δ ppm: 8.15 (d, J = 1.4 Hz, 1H, H5′), 8.36 (dd, J = 6.6, 1.5 Hz, 2H, H2,6), 8.52 (s, 1H, H-C=N), 9.10 (dd, J = 6.6, 1.5 Hz, 2H, H3,5), 9.21 (d, J = 1.3 Hz, 1H, H3′), 15.67 (s, 2H, N-H).
- 13C-NMR (75 MHz, DMSO) δ ppm: 122.02 (1C, C5′), 125.10 (2C, C2,6), 128.25 (1C, C1′), 138.37 (1C, C=N), 136,80 (1C, C3′), 144.77 (2C, C3,5), 146.91 (1C, C1), 160.14 (1C, C=O). 126.78 (2C, C2,6), 134,36 (1C, C5′), 143.08 (2C, C3,5).
- FTIR (KBr) ν cm−1: 3193.59 (N-H), 3038.08 (C-Har), 1648.96 (C=O), 1626.02 (C=N-N), 1596.86 (C=Car), 1551.04 (Car
N), 1506.46 (C=N).
- UV (EtOH, 182 μM, 25 °C): λ = 309 nm, ε = 5495.05 dm3·mol−1·cm−1 (very poorly soluble).
- MS (ES, TOF, MeOH) m/z: 238.0708 [M + Na+]; 216.0887 [M + H+].
- HRMS (ES, TOF) m/z: M + H+ calc. for C10H10N5O: 216.0885. Found: 216.0887.
(E)-N′-((4-methyl-1H-imidazol-5-yl)methylene)isonicotinohydrazide (3)

- m.p.: 299 °C (dec.). Rf: 0.1 EtOAc/MeOH (4:1 v/v).
- 1H-NMR (300 MHz, DMSO-d6) δ ppm: 2.43 (s, 3H, CH3), 8.42 (dd, J = 5.5, 1.8 Hz, 2H, H2,6), 8.53 (s, 1H, H-C=N), 9.11 (s, 1H, H3′), 9.14 (dd, J = 5.5, 1.8 Hz, 2H, H3,5), 15.98 (br, 2H, N-H).
- 13C-NMR (75 MHz, DMSO) δ ppm: 9.38 (1C, CH3), 123.60 (1C, C1′) 125.38 (2C, C2,6), 131.78 (1C, C5′), 138.44 (1C, C=N), 142.84 (1C, C3′), 144.03 (2C, C3,5), 147.78 (1C, C1), 159.52 (1C, C=O).
- FTIR (KBr) ν cm−1: 3194.75 (N-H), 3097.19 (Car-H), 1660.58 (C=O), 1621.45 (C=N-N), 1602.24 (Car=Car), 1551.49 (Car
N).
- UV (EtOH, 161.58 μM, 25 °C): λ = 316 nm, ε = 5551.73 dm3·mol−1·cm−1 (very poorly soluble).
- MS (ES, TOF, MeOH) m/z: 252.0866 [M + Na+]; 230.1049 [M + H+].
- HRMS (ES, TOF) m/z: [M + H+] calc. for C11H12N5O: 230.1042. Found: 230.1049.
(E)-N′-((3a,7a-dihydro-1H-indazol-5-yl)methylene)isonicotinohydrazide (4)

- m.p.: 302.5 °C. Rf: 0.45 PE/EtOAc/MeOH (5:5:3 v/v/v).
- 1H-NMR (300 MHz, DMSO-d6) δ ppm: 7.63 (d, J = 8.7 Hz, 1H, H3′), 7.84 (dd, J = 4.4, 1.6 Hz, 2H, H2,6), 7.90 (dd, J = 8.8, 1.5 Hz, 1H, H2′), 8.03–8.10 (m, 1H, H7′), 8.17 (t, J = 1.2 Hz, 1H, H6′), 8.57 (s, 1H, H-C=N), 8.79 (s, 2H, H3,5), 12.01 (s, 1H, N-H), 13.31 (s, 1H, N-Hind).
- 13C-NMR (75 MHz, DMSO) δ ppm: 111.39 (1C, C3′), 121.98 (2C, C2,6), 122.73 (1C, C7′), 123.35 (1C, C9′), 124.19 (1C, C2′), 127.23 (1C, C1′), 134.99 (1C, C6′), 141.07 (1C, C1), 141.14 (1C, C8′), 150.31 (1C, C=N), 150.76 (2C, C3,5), 161.87 (1C, C=O).
- FTIR (KBr) ν cm−1: 3188.96 (N-H), 3027.37 (Car-H), 1652 (C=O), 1622.47 (C=N-N), 1607.84 (Car=Car), 1549.40 (Car
N).
- UV (EtOH, 38.37 μM, 25 °C): λ = 234 nm, ε = 22,666.92 dm3·mol−1·cm−1.
- MS (ES, TOF, MeOH) m/z: 266.1046 [M + H+].
- HRMS (ES, TOF) m/z: [M + H+] calc. for C14H12N5O: 266.1042. Found: 266.1046.
(E)-N′-((2H-indazol-6-yl)methylene)isonicotinohydrazide (5)

- m.p.: 295.2 °C. Rf: 0.45 PE/EtOAc/MeOH (5:5:3 v/v/v).
- 1H-NMR (300 MHz, DMSO-d6) δ ppm: 7.61 (dd, J = 8.5, 1.3 Hz, 1H, H7′), 7.84 (m, 4H, H2,6, H2′, H6′), 8.13 (d, J = 1.3 Hz, 1H, H5′), 8.60 (s, 1H, H-C=N), 8.80 (br, 2H, H3,5), 12.12 (s, 1H, N-H), 13.28 (s, 1H, N-Hind).
- 13C-NMR (75 MHz, DMSO) δ ppm: 110.75 (1C, C2′), 119.03 (1C, C7′), 121.51 (2C, C2,6), 122.01 (1C, C6′), 124.39 (1C, C8′), 132.43 (1C, C1′), 134.21 (1C, C5′), 140.33 (1C, C1), 140.95 (1C, C9′), 150.03 (1C, C=N), 150.79 (2C, C3,5), 162.08 (1C, C=O).
- FTIR (KBr) ν cm−1: 3193.59 (N-H), 3038.08 (C-Har), 1648.96 (C=O), 1626.02 (C=N-N), 1596.86 (C=Car), 1551.04 (Car
N), 1506.46 (C=N).
- UV (EtOH, 37.39 μM, 25 °C): λ = 313 nm, ε = 25,055.1 dm3·mol−1·cm−1.
- MS (ES, TOF, MeOH) m/z: 266.1047 [M + H+].
- HRMS (ES, TOF) m/z: [M + H+] calc. for C14H12N5O: 266.1042. Found: 266.1047.
(E)-N′-((2-methyl-1H-indol-3-yl)methylene)isonicotinohydrazide (6)

- m.p.: 281.1 °C. Rf: 0.55 PE/EtOAc/MeOH (5:5:3 v/v/v).
- 1H-NMR (300 MHz, DMSO-d6) δ ppm: 2.54 (s, 3H, CH3), 7.12 (ddt, J = 24.4, 9.3, 1.9, 1.9 Hz, 2H, H5′, H6′), 7.35 (tt, J = 2.4, 1.7, 0.9, 0.8 Hz, 1H, H4′), 7.85 (dd, J = 4.5, 1.8 Hz, 2H, H2,6), 8.16–8.32 (m, 1H, H7′), 8.71 (s, 1H, H-C=N), 8.78 (dd, J = 4.4, 1.6 Hz, 2H, H3,5), 11.55 (d, J = 7.4 Hz, 1H, N-H), 11.67 (s, 1H, N-Hind).
- 13C-NMR (75 MHz, DMSO) δ ppm: 11.97 (1C, CH3), 107.84 (1C, C1′), 111.34 (1C, C4′), 120.82 (1C, C7′), 121.63(1C, C5′), 121.87 (2C, C2,6), 122.34 (1C, C6′), 125.85 (1C, C9′), 136.19 (1C, C8′), 141.05 (1C, C2′), 141.56 (1C, C1), 146.10 (1C, C=N), 150.69 (2C, C3,5), 161.00 (1C, C=O).
- FTIR (KBr) ν cm−1: 3385.07 (N-H), 3209.03 (N-H), 3049.08 (Car-H), 1655.09 (C=O), 1626.02 (C=N-N), 1599.50 (C=Car), 1550.60 (Car
N), 1506.46 (C=N).
- UV (EtOH, 57.29 μM, 25 °C): λ = 224 nm, ε = 21,909.58 dm3·mol−1·cm−1.
- MS (ES, TOF, MeOH) m/z: 279.1246 [M + H+].
- HRMS (ES, TOF) m/z: [M + H+] calc. for C16H15N4O: 279.1246. Found: 279.1246.
- Compound 6 can also be found (commercial source; CAS No.: 113143-57-0).
(E)-N′-((1H-pyrrolo[2,3-b]pyridin-3-yl)methylene)isonicotinohydrazide (7)

- m.p.: 323.2 °C (dec.). Rf: 0.34 PE/EtAc/MeOH (5:5:3 v/v).
- 1H-NMR (300 MHz, DMSO-d6) δ ppm: 77.24 (tt, J = 7.8, 4.7, 4.7 Hz, 1H, H6′), 7.84 (dd, J = 4.2, 1.7 Hz, 2H, H2,6), 8.03 (d, J = 2.3 Hz, 1H, H2′), 8.33 (dd, J = 4.7, 1.7 Hz, 1H, H7′), 8.58 (d, J = 1.6 Hz, 1H, H5′), 8.62 (s, 1H, H-C=N), 8.78 (dd, J = 4.4, 1.7 Hz, 2H, H3,5), 11.86 (s, 1H, N-H), 12.17 (s, 1H, N-Hind).
- 13C-NMR (75 MHz, DMSO)δ ppm: 110.85 (1C, C1), 117,13 (1C, C9′), 117.44 (1C, C6′), 121.95 (2C, C2,6), 130.56 (1C, C5′), 131.51 (1C, C2′), 141.39 (1C, C1), 144.56 (1C, C7′), 146.09 (1C, C=N), 149.84 (1C, C8′), 150.71 (2C, C3,5), 161.49 (1C, C=O).
- FTIR (KBr) ν cm−1: 3454.03 (N-H), 3199.51 (N-H), 3031.09 (Car-H), 1662.68 (C=O), 1611.72 (C=N-N), 1600.48 (C=Car), 1551.26 (Car
N), 1284.78 (C-N).
- UV (EtOH, 58.08 μM, 25 °C): λ = 200 nm, ε = 18,839.92 dm3·mol−1·cm−1, λ = 218 nm, ε = 17,446.62 dm3·mol−1·cm−1, λ = 322 nm, ε = 17,193.69 dm3·mol−1·cm−1.
- MS (ES, TOF, MeOH) m/z: 266.1045 [M + H+].
- HRMS (ES, TOF) m/z: [M + H+] calc. for C14H12N5O: 266.1042. Found: 266.1045.
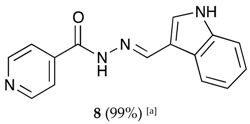
(E)-N′-((1H-indol-3-yl)methylene)isonicotinohydrazide (8)

- m.p.: 242 °C. Rf: 0.55 PE/EtAc/MeOH (5:5:3 v/v/v).
- 1H-NMR (300 MHz, DMSO-d6) δ ppm: δ 7.11–7.28 (m, 2H, H5′,6′), 7.47 (dt, J = 7.9, 0.9 Hz, 1H, H4′), 7.85 (dd, J = 4.4, 1.6 Hz, 2H, H2,6), 7.88 (d, J = 2.8 Hz, 1H, H2′), 8.31 (dd, J = 6.8, 1.5 Hz, 1H, H9′), 8.65 (s, 1H, H-C=N), 8.78 (dd, J = 4.4, 1.6 Hz, 2H, H3, 5), 11.65 (s, 1H, N-H), 11.76 (s, 1H, N-Hind).
- 13C-NMR (75 MHz, DMSO) δ ppm: 111.96 (1C, C1′), 112.34 (1C, C4′), 121.00 (1C, C5′), 121.95 (2C, C2,6), 122.44 (1C, C7′), 123.17 (1C, C6′), 123.35 (1C, C7′), 124.77 (1C, C9′), 131.36 (1C, C2′), 137.53 (1C, C8′), 141.54 (1C, C1), 146.63 (1C, C=N), 150.69 (2C, C3,5), 161.33 (1C, C=O).
- FTIR (KBr) ν cm−1: 3543.66 (N-H), 3395.82 (N-H), 2886.55 (C-Har), 1656.52 (C=O), 1626.02 (C=N-N), 1598.83 (C=Car), 1550.54 (Car
N), 1496.83 (C=N).
- UV (EtOH, 40.01 μM, 25 °C): λ = 221 nm, ε = 22,519 dm3·mol−1·cm−1.
- MS (ES, TOF, MeOH) m/z: 265.1092 [M + H+].
- HRMS (ES, TOF) m/z: [M + H+] calc. for C15H13N4O: 265.1089. Found: 265.1092.
- Compound 8 can also be found (commercial source; CAS No.: 10245-44-0).
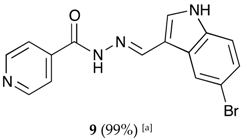
(E)-N′-((5-bromo-1H-indol-3-yl)methylene)isonicotinohydrazide (9)

- m.p.: 309.3 °C (dec.). Rf: 0.61 PE/EtAc/MeOH (5:5:3 v/v).
- 1H-NMR (300 MHz, DMSO-d6) δ ppm: 7.35 (dd, J = 8.7, 2.1 Hz, 1H5′), 7.44 (d, J = 8.5 Hz, 1H, H4′), 7.84 (dd, J = 4.5, 1.9 Hz, 2H, H2,6), 7.95 (d, J = 2.7 Hz, 1H, H9′), 8.48 (s, 1H, H2′), 8.62 (s, 1H, H-C=N), 8.73–8.89 (m, 2H, H3,5), 11.83 (d, J = 6.0 Hz, 2H, N-H).
- 13C-NMR (75 MHz, DMSO) δ ppm: 111.59 (1C, C1′), 113,70 (1C, C6′), 114.42 (1C, C4′), 121.93 (2C, C2,6), 124.60 (1C, C2′), 125.71 (1C, C5′), 126.42 (1C, C9′), 132.71 (1C, C7′), 136.27 (1C, C8′), 141.38 (1C, C1), 146.13 (1C, C=N), 150.72 (2C, C3,5), 161.40 (1C, C=O).
- FTIR (KBr) ν cm−1: 3127.58 (N-Hind), 2891.39 (C-Har), 1662.69 (C=O), 1618.34 (C=N-N), 1538 (C=Car), 1552.13 (Car
N), 1041.12 (Car-Br).
- UV (EtOH, 52.65 μM, 25 °C): λ = 201 nm, ε = 29,890 dm3·mol−1·cm−1, λ = 226 nm, ε = 25,981 dm3·mol−1·cm−1, λ = 330 nm, ε = 18,196 dm3·mol−1·cm−1.
- MS (ES, TOF, MeOH) m/z: 343.0194 [M + H+].
- HRMS (ES, TOF) m/z: [M + H+] calc. for C15H12BrN4O: 343.0193. Found: 343.0194.
- Compound 9 can also be found (CAS No.: 113143-44-5).
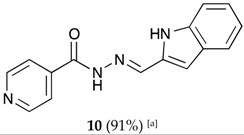
(E)-N′-((1H-indol-2-yl)methylene)isonicotinohydrazide (10)

- m.p.: 231 °C (dec.). Rf: 0.82 PE/EtAc/MeOH (5:5:3 v/v/v).
- 1H-NMR (300 MHz, DMSO-d6) δ ppm: 76.90 (dd, J = 2.1, 0.9 Hz, 1H), 7.02 (ddd, J = 8.0, 7.0, 1.1 Hz, 1H), 7.18 (ddd, J = 8.3, 7.0, 1.2 Hz, 1H), 7.47 (dq, J = 8.2, 0.9 Hz, 1H), 7.58 (dd, J = 7.9, 1.1 Hz, 1H), 7.86 (dd, J = 4.4, 1.6 Hz, 2H), 8.51 (s, 1H), 8.81 (dd, J = 4.4, 1.7 Hz, 2H), 11.65 (s, 1H), 12.05 (s, 1H).
- 13C-NMR (75 MHz, DMSO) δ ppm: 107.98 (1C, C2′), 112.57 (1C, C3′), 120.06 (1C, C4′), 121.31 (1C, C6′), 122.01 (2C, C2,6), 128.05 (1C, C9′), 133.28 (1C, C1′), 138.47 (1C, C8′), 142.16 (1C, C=N), 150.79 (2C, C3, 5), 161.84 (1C, C=O).
- FTIR (KBr) ν cm−1: 3250.19 (N-H), 3032.16 (C-Har), 1689.09 (C=O), 1621.50 (C=N-N), 1599.80 (C=Car), 1548.50 (Car
N).
- UV (EtOH, 52.65 μM, 25 °C): λ = 206 nm, ε = 27,480 dm3·mol−1·cm−1, λ = 350 nm, ε = 33,825 dm3·mol−1·cm−1.
- MS (ES, TOF, MeOH) m/z: 265.1090 [M + H+].
- HRMS (ES, TOF) m/z: [M + H+] calc. for C15H13N4O: 265.1089. Found: 265.1090.
- X-Ray structure of this compound has been recently reported [83].
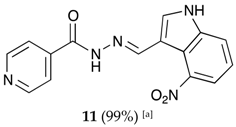
(E)-N′-((4-nitro-1H-indol-3-yl)methylene)isonicotinohydrazide (11)

- m.p.: 317.1 °C (dec.). Rf: 0.70 PE/EtOAc/MeOH (5:5:3 v/v/v).
- 1H-NMR (300 MHz, DMSO-d6) δ ppm: 7.36 (t, J = 8.0 Hz, 1H, H5′), 7.84 (dd, J = 4.4, 1.7 Hz, 2H, H2,6), 7.94 (dd, J = 11.8, 7.9 Hz, 2H, H4′,6′), 8.28 (d, J = 2.9 Hz, 1H, H2′), 8.78 (dd, J = 4.4, 1.7 Hz, 2H, H3,5), 8.91 (s, 1H, H-C=N), 12.00 (s, 1H, N-H), 12.55 (s, 1H, N-H).
- 13C-NMR (75 MHz, DMSO) δ ppm: 110.29 (1C, C9′), 117.59 (1C, C1′), 118.94 (1C, C4′), 119.92 (1C, C6′), 121.50 (1C, C5′), 122.01 (2C, C2, 6), 131.31 (1C, C2′), 139.55 (1C, C8′), 141.22 (1C, C1), 141.90 (1C, C-NO2), 146.43 (1C, C=N), 150.66 (2C, C3,5), 161.56 (1C, C=O).
- FTIR (KBr) ν cm−1: 3156.34 (N-H), 3137.68 (N-H), 3053.25 (C-Har), 1664.06 (C=O), 1628.34 (C=N-N), 1590.99 (C=Car), 1554.10 (Car
N), 1513.15 (C=N-NO2).
- UV (EtOH, 52.64 μM, 25 °C): λ = 214 nm, ε = 26,971.88 dm3·mol−1·cm−1, λ = 331 nm, ε = 17,046.35 dm3·mol−1·cm−1.
- MS (ES, TOF, MeOH) m/z: 332.0760 [M + Na+]; 310.0940 [M + H+].
- HRMS (ES, TOF) m/z: [M + H+] calc. for C15H12N5O3: 310.0937. Found: 310.0940.
3.3. Physicochemical Studies: Hydrolytic Stability and pKa Determination
3.3.1. Hydrolytic Stability
3.3.2. pKa Determination
3.3.3. DMSO stability of Compound 1d
3.4. Biological Assays
3.4.1. Inhibition Kinetics in the Presence of InhA
3.4.2. MIC Determination in M.tb
3.4.3. Determination of LC50
3.4.4. Analysis of the Effect of Hydrazones on Lipids and Mycolic Acids of M.tb H37Ra
3.4.5. Determination of Sensitivity of M.tb H37Ra Strain Overproducing InhA to Hydrazones
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- WHO. Gobal Tuberculosis Report 2016. 2016. Available online: http://www.who.int/tb/publications/global_report/en/ (accessed on 14 October 2016).
- Cynamon, M.H.; Zhang, Y.; Harpster, T.; Cheng, S.; DeStefano, M.S. High-dose isoniazid therapy for isoniazid-resistant murine Mycobacterium tuberculosis infection. Antimicrob. Agents Chemother. 1999, 43, 2922–2924. [Google Scholar] [PubMed]
- Bemer-Melchior, P.; Bryskier, A.; Drugeon, H.B. Comparison of the in vitro activities of rifapentine and rifampicin against Mycobacterium tuberculosis complex. J. Antimicrob. Chemother. 2000, 46, 571–576. [Google Scholar] [CrossRef] [PubMed]
- Jain, A.; Mondal, R. Extensively drug-resistant tuberculosis: Current challenges and threats. FEMS Immunol. Med. Microbiol. 2008, 53, 145–150. [Google Scholar] [CrossRef] [PubMed]
- Parida, S.K.; Axelsson-Robertson, R.; Rao, M.V.; Singh, N.; Master, I.; Lutckii, A.; Keshavjee, S.; Andersson, J.; Zumla, A.; Maeurer, M. Totally drug-resistant tuberculosis and adjunct therapies. J. Intern. Med. 2015, 277, 388–405. [Google Scholar] [CrossRef] [PubMed]
- Food and Drug Administration. Sirturo (bedaquiline) Product Insert. Silver Spring MD: Food and Drug Administration, 2015. Available online: http://www.accessdata.fda.gov/drugsatfda_docs/label/2012/204384s000lbl.pdf (accessed on 11 July 2015).
- European Medicines Agency C for MP for HU. European Medicines Agency, Assessment Report, Deltyba. European Medicines Agency C for MP for HU: London, England, 2013. Procedure No EMEA/H/C/002552. 2015. Available online: http://www.ema.europa.eu/ema/index.jsp?curl=pages/medicines/human/medicines/002552/smops/Positive/human_smop_000572.jsp&mid=WC0b01ac058001d127 (accessed on 11 July 2015).
- Banerjee, D.R.; Biswas, R.; Das, A.K.; Basak, A. Design, synthesis and characterization of dual inhibitors against new targets FabG4 and HtdX of Mycobacterium tuberculosis. Eur. J. Med. Chem. 2015, 100, 223–234. [Google Scholar] [CrossRef] [PubMed]
- Rollas, S.; Küçükgüzel, S.G. Biological Activities of Hydrazone Derivatives. Molecules 2007, 12, 1910–1939. [Google Scholar] [CrossRef] [PubMed]
- Rasras, A.J.M.; Al-Tel, T.H.; Al-Aboudi, A.F.; Al-Qawasmeh, R.A. Synthesis and antimicrobial activity of cholic acid hydrazone analogues. Eur. J. Med. Chem. 2010, 45, 2307–2313. [Google Scholar] [CrossRef] [PubMed]
- Mohareb, R.M.; Fleita, D.H.; Sakka, O.K. Novel Synthesis of Hydrazide-Hydrazone Derivatives and Their Utilization in the Synthesis of Coumarin, Pyridine, Thiazole and Thiophene Derivatives with Antitumor Activity. Molecules 2011, 16, 16–27. [Google Scholar] [CrossRef] [PubMed]
- Asif, M. Pharmacologically potentials of hydrazonone containing compounds: A promising scaffold. Int. J. Adv. Chem. 2014, 2, 85–103. [Google Scholar] [CrossRef]
- Bairwa, R.; Kakwani, M.; Tawari, N.R.; Lalchandani, J.; Ray, M.K.; Rajan, M.G.R.; Degani, M.S. Novel molecular hybrids of cinnamic acids and guanylhydrazones as potential antitubercular agents. Bioorg. Med. Chem. Lett. 2010, 20, 1623–1625. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, S.A.; da Silva, E.F.; de Souza, M.V.N.; Lourenço, M.C.S.; Vicente, F.R. Synthesis and antimycobacterial evaluation of new trans-cinnamic acid hydrazide derivatives. Bioorg. Med. Chem. Lett. 2008, 18, 538–541. [Google Scholar] [CrossRef] [PubMed]
- Vavříková, E.; Polanc, S.; Kočevar, M.; Horváti, K.; Bősze, S.; Stolaříková, J.; Vávrová, K.; Vinšová, J. New fluorine-containing hydrazones active against MDR-tuberculosis. Eur. J. Med. Chem. 2011, 46, 4937–4945. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, K.N.; Chiaradia, L.D.; Martins, P.G.A.; Mascarello, A.; Cordeiro, M.N.S.; Guido, R.V.C.; Andricopulo, A.D.; Yunes, R.A.; Nunes, R.J.; Terenzi, J.H. Sulfonyl-hydrazones of cyclic imides derivatives as potent inhibitors of the Mycobacterium tuberculosis protein tyrosine phosphatase B (PtpB). Med. Chem. Comm. 2011, 2, 500–504. [Google Scholar] [CrossRef]
- Reddy, K.S.; Ramesh, M.; Srimai, V.; Chandra, K.S.; Satyender, A. Synthesis, antimycobacterial activity and docking studies of L-proline derived hydrazones. Der Pharma Chemica 2014, 6, 267–271. [Google Scholar]
- Maccari, R.; Ottanà, R.; Vigorita, M.G. In vitro advanced antimycobacterial screening of isoniazid-related hydrazones, hydrazides and cyanoboranes: Part 14. Bioorg. Med. Chem. Lett. 2005, 15, 2509–2513. [Google Scholar] [CrossRef] [PubMed]
- Naveen Kumar, H.S.; Parumasivam, T.; Jumaat, F.; Ibrahim, P.; Asmawi, M.Z.; Sadikun, A. Synthesis and evaluation of isonicotinoyl hydrazone derivatives as antimycobacterial and anticancer agents. Med. Chem. Res. 2014, 23, 269–279. [Google Scholar] [CrossRef]
- Vavříková, E.; Polanc, S.; Kočevar, M.; Košmrlj, J.; Horváti, K.; Bősze, S.; Stolaříková, J.; Imramovský, A.; Vinšová, J. New series of isoniazid hydrazones linked with electron-withdrawing substituents. Eur. J. Med. Chem. 2011, 46, 5902–5909. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.-L.; Zhang, Y.-B.; Tang, J.-F.; Yang, Y.-S.; Chen, R.-Q.; Zhang, F.; Zhu, H.-L. Design, synthesis and antibacterial activities of vanillic acylhydrazone derivatives as potential β-ketoacyl-acyl carrier protein synthase III (FabH) inhibitors. Eur. J. Med. Chem. 2012, 57, 373–382. [Google Scholar] [CrossRef] [PubMed]
- Hearn, M.J.; Cynamon, M.H.; Chen, M.F.; Coppins, R.; Davis, J.; Joo-On Kang, H.; Noble, A.; Tu-Sekine, B.; Terrot, M.S.; Trombino, D.; et al. Preparation andantitubercular activities in vitro and in vivo of novel Schiff bases of isoniazid. Eur. J. Med. Chem. 2009, 44, 4169–4178. [Google Scholar] [CrossRef] [PubMed]
- Hearn, M.J.; Cynamon, M.H. Desing and synthesis of antituberculars: Preparation and evaluation against Mycobacterium tuberculosis of an isoniazid Schiff base. J. Antimicrob. Chemother. 2004, 53, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Ventura, C.; Martins, F. Application of quantitative structure−activity relationships to the modeling of antitubercular compounds 1. The hydrazide family. J. Med. Chem. 2008, 51, 612–624. [Google Scholar] [CrossRef] [PubMed]
- Martins, F.; Santos, S.; Ventura, C.; Elvas-Leitão, R.; Santos, L.; Vitorino, S.; Reis, M.; Miranda, V.; Correia, H.F.; et al. Design, synthesis and biological evaluation of novel isoniazid derivatives with potent antitubercular activity. Eur. J. Med. Chem. 2014, 81, 119–138. [Google Scholar] [CrossRef] [PubMed]
- De, P.; Baltas, M.; Bedos-Belval, F. Cinnamic acid derivatives as anticancer agents—A review. Curr. Med. Chem. 2011, 18, 1672–1703. [Google Scholar] [CrossRef] [PubMed]
- De, P.; Bedos-Belval, F.; Vanucci-Bacqué, C.; Baltas, M. Cinnamic acid derivatives in tuberculosis, malaria and cardiovascular diseases—A review. Curr. Org. Chem. 2012, 16, 747–768. [Google Scholar] [CrossRef]
- De, P.; De, K.; Veau, D.; Bedos-Belval, F.; Chassaing, S.; Baltas, M. Recent advances in the development of cinnamic-like derivatives as antituberculosis agents. Expert Opin. Ther. Pat. 2012, 22, 155–168. [Google Scholar] [CrossRef] [PubMed]
- De, P.; Veau, D.; Bedos-Belval, F.; Chassaing, S.; Baltas, M. Cinnamic Derivatives in Tuberculosis. In Understanding Tuberculosis—New Approaches to Fighting Against Drug Resistance; Cardona, P.-J., Ed.; InTech: Rijeka, Croatia, 2012; pp. 337–362. [Google Scholar] [CrossRef]
- Menendez, C.; Chollet, A.; Rodriguez, F.; Inard, C.; Pasca, M.R.; Lherbet, C.; Baltas, M. Chemical synthesis and biological evaluation of triazole derivatives as inhibitors of InhA and antituberculosis agents. Eur. J. Med. Chem. 2012, 52, 275–283. [Google Scholar] [CrossRef] [PubMed]
- Menendez, C.; Rodriguez, F.; Ribeiro, A.L.; Zara, F.; Frongia, C.; Lobjois, V.; Saffon, N.; Pasca, M.R.; Lherbet, C.; Baltas, M. Synthesis and evaluation of α-ketotriazoles and α,β-diketotriazoles as inhibitors of Mycobacterium tuberculosis. Eur. J. Med. Chem. 2013, 69, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Menendez, C.; Mori, G.; Maillot, M.; Fabing, I. Synthesis and evaluation of β-hydroxytriazoles and related compounds as antitubercular agents. French-Ukrainian J. Chem. 2015, 3, 82–96. [Google Scholar] [CrossRef]
- Veau, D.; Krykun, S.; Mori, G.; Orena, B.S.; Pasca, M.R.; Frongia, C.; Lobjois, V.; Chassaing, S.; Lherbet, C.; Baltas, M. Triazolophthalazines: Easily accessible compounds with potent antitubercular activity. Chem. Med. Chem. 2016, 11, 1078–1089. [Google Scholar] [CrossRef] [PubMed]
- Matviiuk, T.; Madacki, J.; Mori, G.; Orena, B.S.; Menendez, C.; Kysil, A.; André-Barrès, C.; Rodriguez, F.; Korduláková, J.; Mallet-Ladeira, S.; et al. Pyrrolidinone and pyrrolidine derivatives: Evaluation as inhibitors of InhA and Mycobacterium tuberculosis. Eur. J. Med. Chem. 2016, 123, 462–475. [Google Scholar] [CrossRef] [PubMed]
- Matviiuk, T.; Mori, G.; Lherbet, C.; Rodriguez, F.; Pasca, M.R.; Gorichko, M.; Guidetti, B.; Voitenko, Z.; Baltas, M. Synthesis of 3-heteryl substituted pyrrolidine-2,5-diones via catalytic Michael reaction and evaluation of their inhibitory activity against InhA and Mycobacterium tuberculosis. Eur. J. Med. Chem. 2014, 71, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Matviiuk, T.; Rodriguez, F.; Saffon, N.; Mallet-Ladeira, S.; Gorichko, M.; de Jesus Lopes Ribeiro, A.L.; Pasca, M.R.; Lherbet, C.; Voitenko, Z.; Baltas, M. Design, chemical synthesis of 3-(9H-fluoren-9-yl)pyrrolidine-2,5-dione derivatives and biological activity against enoyl-ACP reductase (InhA) and Mycobacterium tuberculosis. Eur. J. Med. Chem. 2013, 70, 37–48. [Google Scholar] [CrossRef] [PubMed]
- Pavan, F.R.; da S Maia, P.I.; Leite, S.R.A.; Deflon, V.M.; Batista, A.A.; Sato, D.N.; Franzblau, S.G.; Leite, C.Q.F. Thiosemicarbazones, semicarbazones, dithiocarbazates and hydrazide/hydrazones: Anti- Mycobacterium tuberculosis activity and cytotoxicity. Eur. J. Med. Chem. 2010, 45, 1898–1905. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, P.F.M.; Baron, M.; Chamayou, A.; André-Barrès, C.; Guidetti, B.; Baltas, M. Solvent-free mechanochemical route for green synthesis of pharmaceutically attractive phenol-hydrazones. RSC Adv. 2014, 4, 56736–56742. [Google Scholar] [CrossRef]
- Marques de Oliveira, P.F. Investigation of Mechanochemical Synthesis of Condensed 1,4-diazines and Pharmaceutically Attractive Hydrazones. Ph.D. Thesis, Ecole des mines d’Albi-Carmaux, Albi, France, October 2015. [Google Scholar]
- Baláž, P. Mechanical activation in hydrometallurgy. Int. J. Miner. Process. 2003, 72, 341–354. [Google Scholar] [CrossRef]
- Balema, V.P.; Wiench, J.W.; Pruski, M.; Pecharsky, V.K. Mechanically induced solid-state generation of phosphorus ylides and the solvent-free wittig reaction. J. Am. Chem. Soc. 2002, 124, 6244–6245. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, H.; Hiraoka, R.; Senna, M. A Diels-Alder reaction catalyzed by eutectic complexes autogenously formed from solid state phenols and quinones. Tetrahedron Lett. 2006, 47, 4481–4484. [Google Scholar] [CrossRef]
- Zhang, Z.; Peng, Z.-W.; Hao, M.-F.; Gao, J.-G. Mechanochemical diels-alder cycloaddition reactions for straightforward synthesis of endo-norbornene derivatives. Synlett 2010, 2010, 2895–2898. [Google Scholar] [CrossRef]
- Zhang, Z.; Dong, Y.-W.; Wang, G.-W.; Komatsu, K. Highly efficient mechanochemical reactions of 1,3-dicarbonyl compounds with chalcones and azachalcones catalyzed by potassium carbonate. Synlett 2004, 61–64. [Google Scholar] [CrossRef]
- Zhang, Z.; Dong, Y.-W.; Wang, G.-W.; Komatsu, K. Mechanochemical Michael reactions of chalcones and azachalcones with ethyl acetoacetate catalyzed by K2CO3 under solvent-free conditions. Chem. Lett. 2004, 33, 168–169. [Google Scholar] [CrossRef]
- Kaupp, G.; Naimi-Jamal, M.R.; Schmeyers, J. Quantitative reaction cascades of Ninhydrin in the solid state. Chem. Eur. J. 2002, 8, 594–600. [Google Scholar] [CrossRef]
- Heintz, A.S.; Gonzales, J.E.; Fink, M.J.; Mitchell, B.S. Catalyzed self-aldol reaction of valeraldehyde via a mechanochemical method. J. Mol. Catal. A Chem. 2009, 304, 117–120. [Google Scholar] [CrossRef]
- Burmeister, C.F.; Stolle, A.; Schmidt, R.; Jacob, K.; Breitung-Faes, S.; Kwade, A. Experimental and Computational Investigation of Knoevenagel Condensation in Planetary Ball Mills. Chem. Eng. Technol. 2014, 37, 857–864. [Google Scholar] [CrossRef]
- Fulmer, D.A.; Shearouse, W.C.; Medonza, S.T.; Mack, J. Solvent-free Sonogashira coupling reaction via high speed ball milling. Green Chem. 2009, 11, 1821. [Google Scholar] [CrossRef]
- Thorwirth, R.; Stolle, A.; Ondruschka, B. Fast copper-, ligand- and solvent-free Sonogashira coupling in a ball mill. Green Chem. 2010, 12, 985. [Google Scholar] [CrossRef]
- Schneider, F.; Ondruschka, B. Mechanochemical solid-state Suzuki reactions using an in situ generated base. ChemSusChem 2008, 1, 622–625. [Google Scholar] [CrossRef] [PubMed]
- Bernhardt, F.; Trotzki, R.; Szuppa, T.; Stolle, A.; Ondruschka, B. Solvent-free and time-efficient Suzuki-Miyaura reaction in a ball mill: The solid reagent system KF-Al2O3 under inspection. Beilstein J. Org. Chem. 2010, 6, 30–34. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.; Loots, L.; Friščić, T. Towards medicinal mechanochemistry: Evolution of milling from pharmaceutical solid form screening to the synthesis of active pharmaceutical ingredients (APIs). Chem. Commun. 2016. [Google Scholar] [CrossRef] [PubMed]
- Mikhailenko, M.A.; Shakhtshneider, T.P.; Boldyrev, V.V. Acylation of sulfathiazole with maleic anhydride under mechanochemical activation. Mendeleev Commun. 2007, 17, 315–317. [Google Scholar] [CrossRef]
- Carlier, L.; Baron, M.; Chamayou, A.; Couarraze, G. Use of co-grinding as a solvent-free solid state method to synthesize dibenzophenazines. Tetrahedron Lett. 2011, 52, 4686–4689. [Google Scholar] [CrossRef]
- Lee, B.; Kang, P.; Lee, K.H.; Cho, J.; Nam, W.; Lee, W.K.; Hur, N.H. Solid-state and solvent-free synthesis of azines, pyrazoles, and pyridazinones using solid hydrazine. Tetrahedron Lett. 2013, 54, 1384–1388. [Google Scholar] [CrossRef]
- Estévez, V.; Villacampa, M.; Menéndez, J.C. Three-component access to pyrroles promoted by the CAN–silver nitrate system under high-speed vibration milling conditions: A generalization of the Hantzsch pyrrole synthesis. Chem. Commun. 2013, 49, 591–593. [Google Scholar] [CrossRef] [PubMed]
- Schmeyers, J.; Toda, F.; Boy, J.; Kaupp, G. Quantitative solid–solid synthesis of azomethines. J. Chem. Soc. Perk. Trans. 2 1998, 4, 989–994. [Google Scholar] [CrossRef]
- Dolotko, O.; Wiench, J.W.; Dennis, K.W.; Pecharsky, V.K.; Balema, V.P. Mechanically induced reactions in organic solids: Liquid eutectics or solid-state processes? New J. Chem. 2010, 34, 25–28. [Google Scholar] [CrossRef]
- Kaupp, G.; Schmeyers, J.; Boy, J. Iminium Salts in Solid-State Syntheses Giving 100% Yield. J. Für Prakt. Chem. 2000, 342, 269–280. [Google Scholar] [CrossRef]
- Mokhtari, J.; Naimi-Jamal, M.R.; Hamzeali, H.; Dekamin, M.G.; Kaupp, G. Kneading Ball-milling and stoichiometric melts for the quantitative derivatization of carbonyl compounds with gas-solid recovery. ChemSusChem 2009, 2, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Nun, P.; Martin, C.; Martinez, J.; Lamaty, F. Solvent-free synthesis of hydrazones and their subsequent N-alkylation in a Ball-mill. Tetrahedron 2011, 67, 8187–8194. [Google Scholar] [CrossRef]
- Shalini, K.; Sharma, P.; Kumar, N. Imidazole and its biological activities: A review. Chem. Sin. 2010, 1, 36–47. [Google Scholar]
- Verma, A.; Joshi, S.; Singh, D. Imidazole: Having versatile biological activities. J. Chem. 2013, 2013, 1–12. [Google Scholar] [CrossRef]
- Thangadurai, A.; Minu, M.; Wakode, S.; Agrawal, S.; Narasimhan, B. Indazole: A medicinally important heterocyclic moiety. Med. Chem. Res. 2012, 21, 1509–1523. [Google Scholar] [CrossRef]
- De Luca, L. Naturally occurring and synthetic imidazoles: Their chemistry and their biological activities. Curr. Med. Chem. 2006, 13, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Sharma, V.; Kumar, P.; Pathak, D. Biological importance of the indole nucleus in recent years: A comprehensive review. J. Heterocycl. Chem. 2010, 47, 491–502. [Google Scholar] [CrossRef]
- Oliveira, P.F.M.; Haruta, N.; Chamayou, A.; Guidetti, B.; Baltas, M.; Tanaka, K.; Sato, T.; Baron, M. Comprehensive experimental investigation of mechanically induced 1,4-diazines synthesis in solid state. Tetrahedron 2017, 73, 2305–2310. [Google Scholar] [CrossRef]
- Palla, G.; Predieri, G.; Domiano, P.; Vignali, C.; Turner, W. Conformational behaviour and/isomerization of -acyl and -aroylhydrazones. Tetrahedron 1986, 42, 3649–3654. [Google Scholar] [CrossRef]
- Syakaev, V.V.; Podyachev, S.N.; Buzykin, B.I.; Latypov, S.K.; Habicher, W.D.; Konovalov, A.I. NMR study of conformation and isomerization of aryl- and heteroarylaldehyde 4-tert-butylphenoxyacetylhydrazones. J. Mol. Struct. 2006, 788, 55–62. [Google Scholar] [CrossRef]
- Ünsal-Tan, O.; Özden, K.; Rauk, A.; Balkan, A. Synthesis and cyclooxygenase inhibitory activities of some N-acylhydrazone derivatives of isoxazolo[4,5-d]pyridazin-4(5H)-ones. Eur. J. Med. Chem. 2010, 45, 2345–2352. [Google Scholar] [CrossRef]
- Patorski, P.; Wyrzykiewicz, E.; Bartkowiak, G. Synthesis and conformational assignment of N-(E)-stilbenyloxymethylenecarbonyl-substituted hydrazones of acetone and o- (m-and p-) chloro- (nitro-) benzaldehydes by means of and NMR spectroscopy. J. Spectrosc. 2013, 1–12. [Google Scholar] [CrossRef]
- Lodewyk, M.W.; Siebert, M.R.; Tantillo, D.J. Computational prediction of 1 H and 13 C chemical shifts: A useful tool for natural product, mechanistic, and synthetic organic chemistry. Chem. Rev. 2012, 112, 1839–1862. [Google Scholar] [CrossRef] [PubMed]
- Kalia, J.; Raines, R.T. Hydrolytic stability of hydrazones and oximes. Angew. Chemie Int. Ed. 2008, 47, 7523–7526. [Google Scholar] [CrossRef] [PubMed]
- Wiberg, K.B.; Glaser, R. Resonance interactions in acyclic systems. 4. stereochemistry, energetics, and electron distributions in 3-center-four-.pi.-electron systems A:BC. J. Am. Chem. Soc. 1992, 114, 841–850. [Google Scholar] [CrossRef]
- Doungdee, P.; Sarel, S.; Wongvisetsirikul, N.; Avramovici-Grisaru, S. Iron chelators of the pyridoxal 2-pyridyl hydrazone class. Part 4. pKa values of the chelators and their relevance to biological properties. J. Chem. Soc. Perkin Trans. 1995, 2, 319. [Google Scholar] [CrossRef]
- Vilchèze, C.; Jacobs, W.R., Jr. The mechanism of isoniazid killing: Clarity through the scope of genetics. Annu. Rev. Microbiol. 2007, 61, 35–50. [Google Scholar] [CrossRef] [PubMed]
- Johnsson, K.; Schultz, P.G. Mechanistic studies of the oxidation of isoniazid by the catalase peroxidase from Mycobacterium tuberculosis. J. Am. Chem. Soc. 1994, 116, 7425–7426. [Google Scholar] [CrossRef]
- Rawat, R.; Whitty, A.; Tonge, P.J. The isoniazid-NAD adduct is a slow, tight-binding inhibitor of InhA, the Mycobacterium tuberculosis enoyl reductase: Adduct affinity and drug resistance. Proc. Natl. Acad. Sci. USA 2003, 100, 13881–13886. [Google Scholar] [CrossRef] [PubMed]
- Lei, B.; Wei, C.J.; Tu, S.C. Action mechanism of antitubercular isoniazid-activation by Mycobacterium tuberculosis KatG, isolation, and characterization of InhA inhibitor. J. Biol. Chem. 2000, 275, 2520–2526. [Google Scholar] [CrossRef] [PubMed]
- Menendez, C.; Gau, S.; Lherbet, C.; Rodriguez, F.; Inard, C.; Pasca, M.R.; Baltas, M. Synthesis and biological activities of triazole derivatives as inhibitors of InhA and antituberculosis agents. Eur. J. Med. Chem. 2011, 46, 5524–5531. [Google Scholar] [CrossRef] [PubMed]
- Campbell, P.J.; Morlock, G.P.; Sikes, R.D.; Dalton, T.L.; Metchock, B.; Starks, A.M.; Hooks, D.P.; Cowan, L.S.; Plikaytis, B.B.; Posey, J.E. Molecular detection of mutations associated with first- and second-line drug resistance compared with conventional drug susceptibility testing of Mycobacterium tuberculosis. Antimicrob. Agents Chemother. 2011, 55, 2032–2041. [Google Scholar] [CrossRef] [PubMed]
- Saranaya, S.; Haribabu, J.; Bhuvanesh, N.S.P.; Karvembu, R.; Gayathri, D. Crystal structures of the Schiff base derivatives (E)-N[(1H-indol-3-yl)methylidene]isonicotino-hydrazide ethanol monosolvate and (E)-N-methyl-2-[1-(2-oxo-2H-chromen-3-yl)ethyl-idene]hydrazinecarbothioamide. Acta Crystallogr. E Crystallogr. Commun. 2017, 73 Pt 4, 594–597. [Google Scholar] [CrossRef] [PubMed]
- Mochon, M. Salicylaldehyde-1-phthalazinohydrazone as an analytical reagent. Talanta 1986, 33, 627–630. [Google Scholar] [CrossRef]
- Chollet, A.; Mori, G.; Menendez, C.; Rodriguez, F.; Fabing, I.; Pasca, M.R.; Madacki, J.; Korduláková, J.; Constant, P.; Quémard, A.; et al. Design, synthesis and evaluation of new GEQ derivatives as inhibitors of InhA enzyme and Mycobacterium tuberculosis growth. Eur. J. Med. Chem. 2015, 101, 218–235. [Google Scholar] [CrossRef] [PubMed]
- Stadthagen, G.; Korduláková, J.; Griffin, R.; Constant, P.; Bottová, I.; Barilone, N.; Gicquel, B.; Daffé, M.; Jackson, M. p-Hydroxybenzoic acid synthesis in Mycobacterium tuberculosis. J. Biol. Chem. 2005, 280, 40699–40706. [Google Scholar] [CrossRef] [PubMed]
- Phetsuksiri, B.; Baulard, A.R.; Cooper, A.M.; Minnikin, D.E.; Douglas, J.D.; Besra, G.S.; Brennan, P.J. Antimycobacterial activities of isoxyl and new derivatives through the inhibition of mycolic acid synthesis. Antimicrob. Agents Chemother. 1999, 43, 1042–1051. [Google Scholar] [PubMed]
- Larsen, M.H.; Vilchèze, C.; Kremer, L.; Besra, G.S.; Parsons, L.; Salfinger, M.; Heifets, L.; Hazbon, M.H.; Alland, D.; Sacchettini, J.C.; et al. Overexpression of inhA, but not kasA, confers resistance to isoniazid and ethionamide in Mycobacterium smegmatis, M. bovis BCG and M. tuberculosis. Mol. Microbiol. 2002, 46, 453–466. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds 1a–d, 2–11 are available from the authors. |




| Aldehydes | Heterocyclic Hydrazones (6) Derived From Isoniazid | |
|---|---|---|
| Imidazole derivatives | 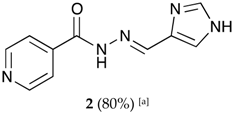 | 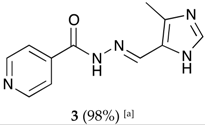 |
| Indazole derivatives | 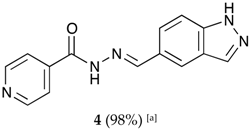 | 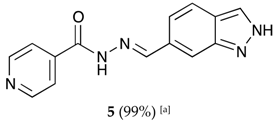 |
| Indole derivatives |  |  |
 |  | |
 |  | |
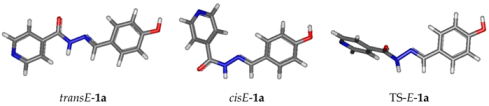
| In the Gas Phase | E (ua) | G (ua) | ΔG (kcal/mol) | % |
| transE_1a | −816.730569 | −816.553707 | 1.47 | 8 |
| cisE_1a | −816.733307 | −816.55605 | 0 | 92 |
| In DMSO | E (ua) | G (ua) | ΔG (kcal/mol) | % |
| transE_1a | −816.763427 | −816.585816 | −1.68 | 94 |
| cisE_1a | −816.761415 | −816.583145 | 0 | 6 |
| TS_1a | −816.765271 | −816.557724 | 17.63 | - |
| Isomer | Geometry (In Gas Phase) | G (In Gas Phase) | % | G (In DMSO) | % |
|---|---|---|---|---|---|
| transE_5_1 | 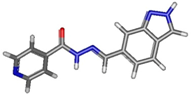 | −888.90736 | 0.34 | −888.94471 | 28.22 |
| transE_5_2 | 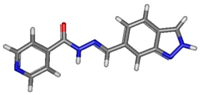 | −888.909926 | 5.09 | −888.94528 | 51.50 |
| cisE_5_1 |  | −888.910465 | 9.0 | −888.941968 | 1.54 |
| cisE_5_2 | 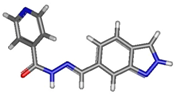 | −888.912591 | 85.6 | −888.944326 | 18.72 |
| Compound | Medium | Conc. (mol/L) | pH | Time | Stability |
|---|---|---|---|---|---|
| 1a | 28% EtOH/H2O | 6.0 × 10−5 | 6.1 | 22 h | stable |
| 1b | 6.3 × 10−5 | 6.5 | 22 h | stable | |
| 1c | 6.9 × 10−5 | 6.3 | 15 h | stable | |
| 1d | 5.9 × 10−5 | 6.2 | 21 h | stable | |
| 5 | 5% EtOH/PIPES buffer (50 mM) | 4.1 × 10−5 | 6.8 | 20 h | 3% Abs. reduction |
| 7 | 3.1 × 10−5 | 6.8 | 20 h | stable | |
| 7 days | 5% Abs. reduction |
| Compound | 1a | 1b | 1c | 1d | 5 | 7 |
|---|---|---|---|---|---|---|
| pKa1 | nd a | nd a | 3.4 | 3.0 | 3.4 | 3.6 |
| pKa2 | 9.2 ± 0.1 | 7.4 ± 0.1 | 9.1 ± 0.1 | 9.5 ± 0.1 | 10.4 ± 0.1 | 11 |
| pKa3 | - | 8.9 ± 0.1 | - | >11 | - | - |
| Compound | % Inhibition at 50 μM (Inhibitor) | Compound | % Inhibition at 50 μM (Inhibitor) |
|---|---|---|---|
| 1a | 45 | 5 | 19 |
| 1b | 54 | 6 | 43 |
| 1c | 48 | 7 | 39 |
| 1d | 64 | 8 | 42 |
| 2 | 54 | 9 | 32 |
| 3 | 3 | 10 | 79 |
| 4 | 33 | 11 | not soluble |
| TCL | >99 |
| Compound | MW (g/mol) | MIC (μg/mL)/(μM) | LogP | Cpd | MW (g/mol) | MIC (μg/mL)/(μM) | LogP |
|---|---|---|---|---|---|---|---|
| 1a | 241.25 | 0.0125/0.05 | 1.64 | 1c | 271.27 | 0.125/0.46 | 1.51 |
| 1b | 257.24 | 0.125/0.49 | 1.25 | 1d | 301.30 | 0.125/0.41 | 1.38 |
| INH | 137.14 | 0.025/0.18 | −0.64 |
| Compound | MW (g/mol) | MIC (μg/mL)/(μM) | LogP | Cpd | MW (g/mol) | MIC (μg/mL)/(μM) | LogP |
|---|---|---|---|---|---|---|---|
| 2 | 215.21 | 0.03/0.14 | −1.00 | 7 | 265.27 | 0.015/0.056 | 0.24 |
| 3 | 229.24 | 0.03/0.13 | −1.37 | 8 | 264.28 | 0.06/0.23 | 0.86 |
| 4 | 265.27 | 0.06/0.23 | 1.38 | 9 | 343.18 | 0.125/0.36 | 1.69 |
| 5 | 265.27 | 0.03/0.11 | −0.52 | 10 | 264.28 | 0.06/0.23 | 0.52 |
| 6 | 278.31 | 0.25/0.90 | 0.49 | 11 | 309.28 | 0.25/0.81 | 1.39 |
| INH | 137.14 | 0.05/0.36 | −0.64 |
| Compound | MIC (μg/mL)/(μM) | |
|---|---|---|
| H37Rv | IC2 | |
| 1a | 0.0125/0.05 | 2.5/10.36 |
| 1b | 0.125/0.49 | 1/3.89 |
| 1c | 0.125/0.46 | >2.5/>9.22 |
| 1d | 0.125/0.41 | >2.5/>8.30 |
| 2 | 0.03/0.14 | 5/23.2 |
| 3 | 0.03/0.13 | 5–10/21.8–43.6 |
| 4 | 0.06/0.23 | >10 |
| 5 | 0.03/0.11 | >10 |
| 6 | 0.25/0.90 | >10 |
| 7 | 0.015/0.056 | >10 |
| 8 | 0.06/0.23 | >10 |
| 9 | 0.125/0.36 | >10 |
| 10 | 0.06/0.23 | >10 |
| 11 | 0.25/0.81 | 5–10/18.9–37.8 |
| INH | 0.025/0.18 | >2/>14.58 |
| Compound | LD50 (μM) | SI | Compound | LD50 (μM) | SI |
|---|---|---|---|---|---|
| 1a | >80 | >1600 | 5 | >80 | >727 |
| 1b | 36.3 | 74 | 6 | 129 | 143 |
| 1c | >80 | >173 | 7 | >80 | >1429 |
| 1d | >80 | >195 | 8 | >80 | >364 |
| 2 | >80 | >571 | 9 | >80 | >222 |
| 3 | >80 | >615 | 10 | 71.4 | 310 |
| 4 | >80 | >364 | 11 | 156 | 193 |
| INH | - | - |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oliveira, P.F.M.; Guidetti, B.; Chamayou, A.; André-Barrès, C.; Madacki, J.; Korduláková, J.; Mori, G.; Orena, B.S.; Chiarelli, L.R.; Pasca, M.R.; et al. Mechanochemical Synthesis and Biological Evaluation of Novel Isoniazid Derivatives with Potent Antitubercular Activity. Molecules 2017, 22, 1457. https://doi.org/10.3390/molecules22091457
Oliveira PFM, Guidetti B, Chamayou A, André-Barrès C, Madacki J, Korduláková J, Mori G, Orena BS, Chiarelli LR, Pasca MR, et al. Mechanochemical Synthesis and Biological Evaluation of Novel Isoniazid Derivatives with Potent Antitubercular Activity. Molecules. 2017; 22(9):1457. https://doi.org/10.3390/molecules22091457
Chicago/Turabian StyleOliveira, Paulo F. M., Brigitte Guidetti, Alain Chamayou, Christiane André-Barrès, Jan Madacki, Jana Korduláková, Giorgia Mori, Beatrice Silvia Orena, Laurent Roberto Chiarelli, Maria Rosalia Pasca, and et al. 2017. "Mechanochemical Synthesis and Biological Evaluation of Novel Isoniazid Derivatives with Potent Antitubercular Activity" Molecules 22, no. 9: 1457. https://doi.org/10.3390/molecules22091457
APA StyleOliveira, P. F. M., Guidetti, B., Chamayou, A., André-Barrès, C., Madacki, J., Korduláková, J., Mori, G., Orena, B. S., Chiarelli, L. R., Pasca, M. R., Lherbet, C., Carayon, C., Massou, S., Baron, M., & Baltas, M. (2017). Mechanochemical Synthesis and Biological Evaluation of Novel Isoniazid Derivatives with Potent Antitubercular Activity. Molecules, 22(9), 1457. https://doi.org/10.3390/molecules22091457








