Human Adenocarcinoma Cell Line Sensitivity to Essential Oil Phytocomplexes from Pistacia Species: a Multivariate Approach †
Abstract
:1. Introduction
2. Results and Discussion
2.1. Chemical Analysis of Essential Oils
2.2. Cytotoxic Activity of Essential Oils on Human Tumor Cell Lines
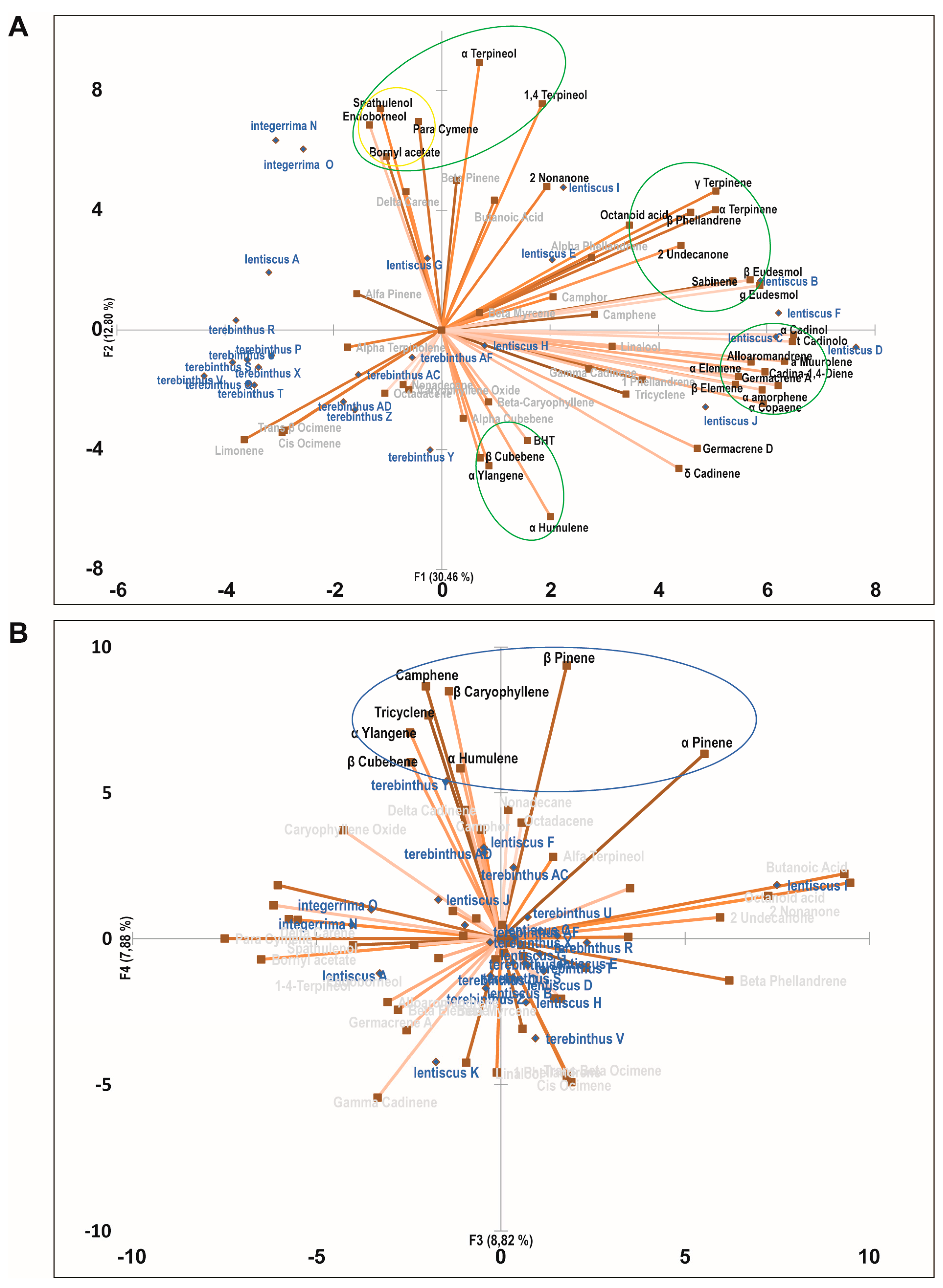
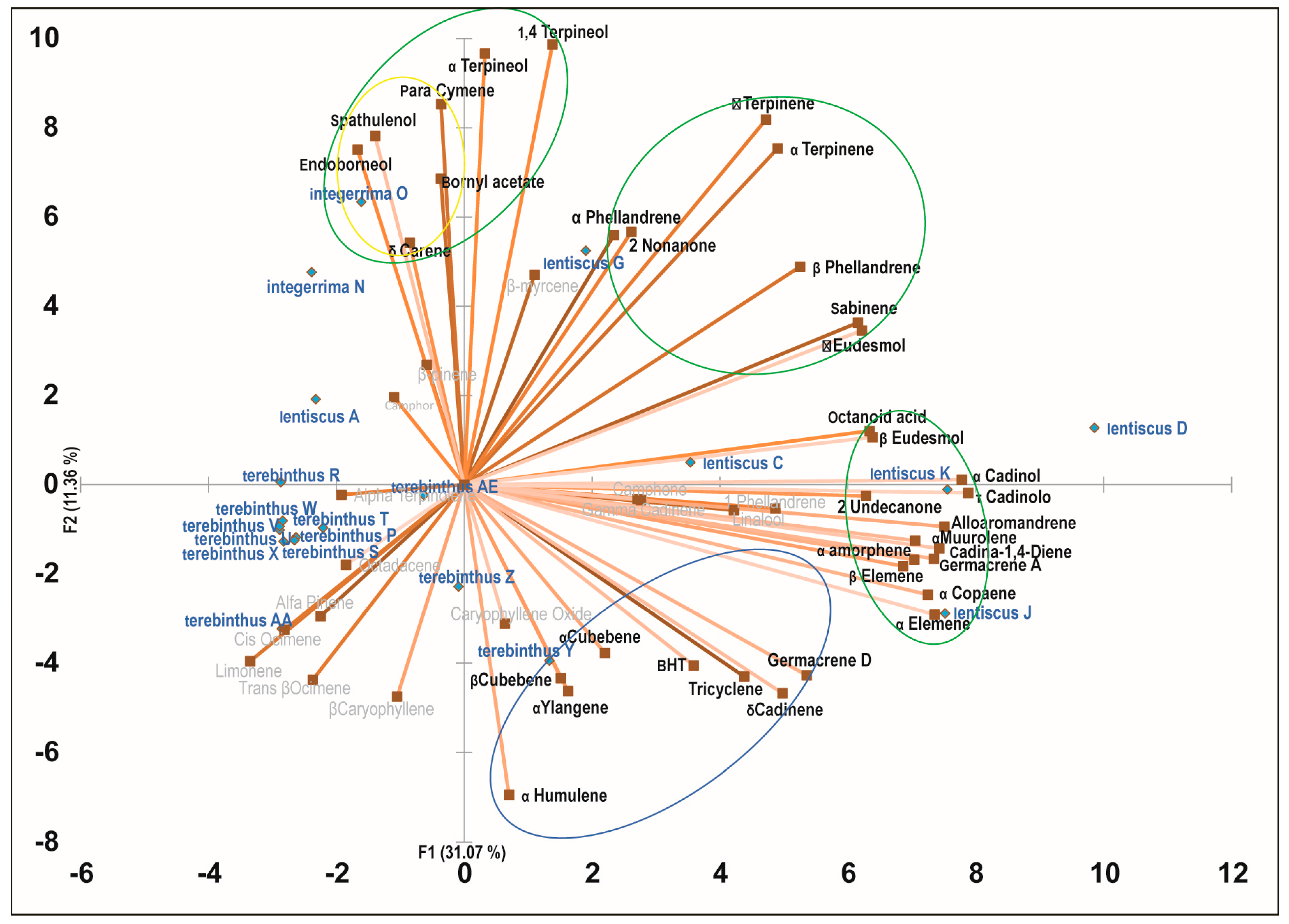
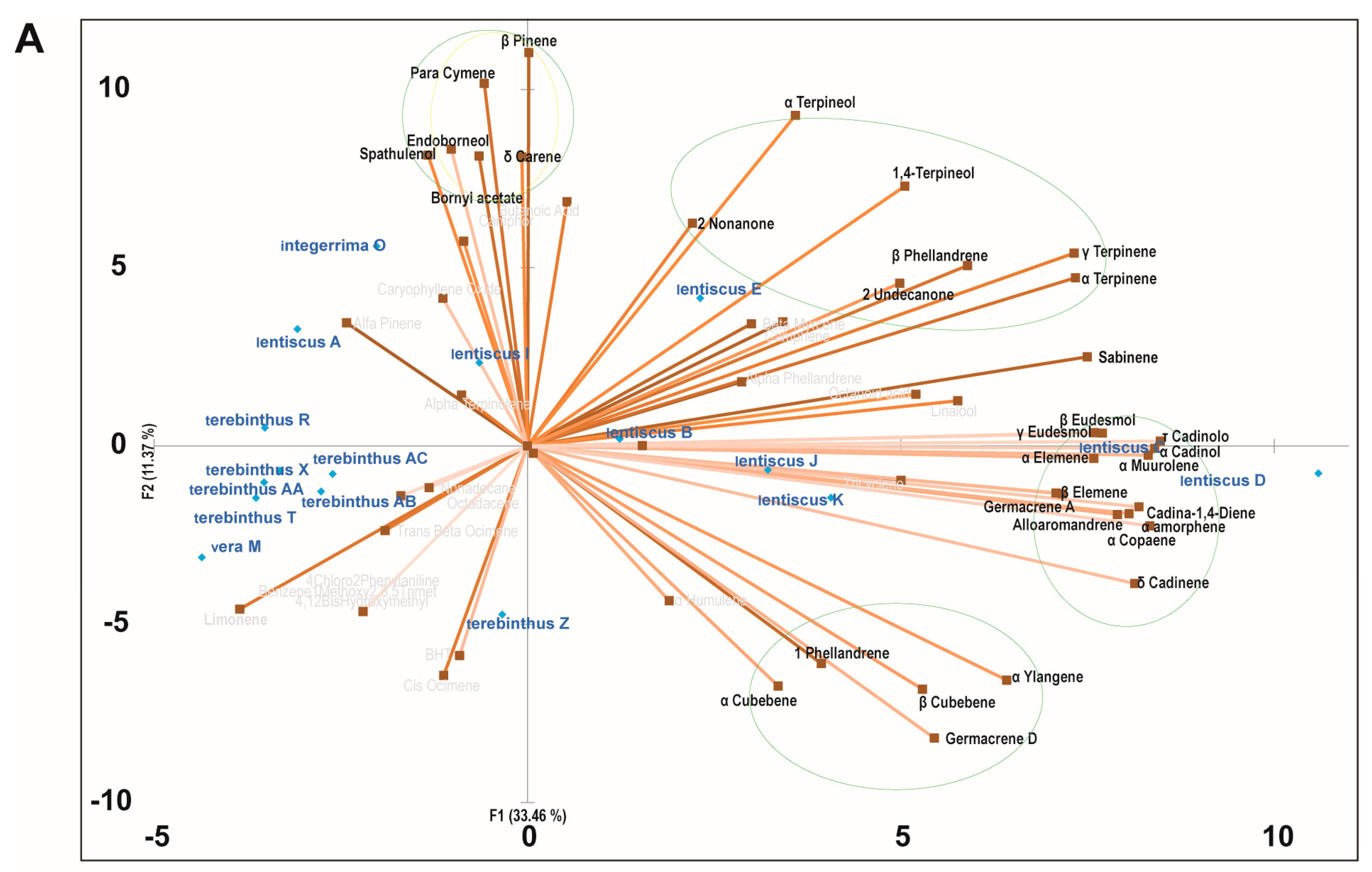
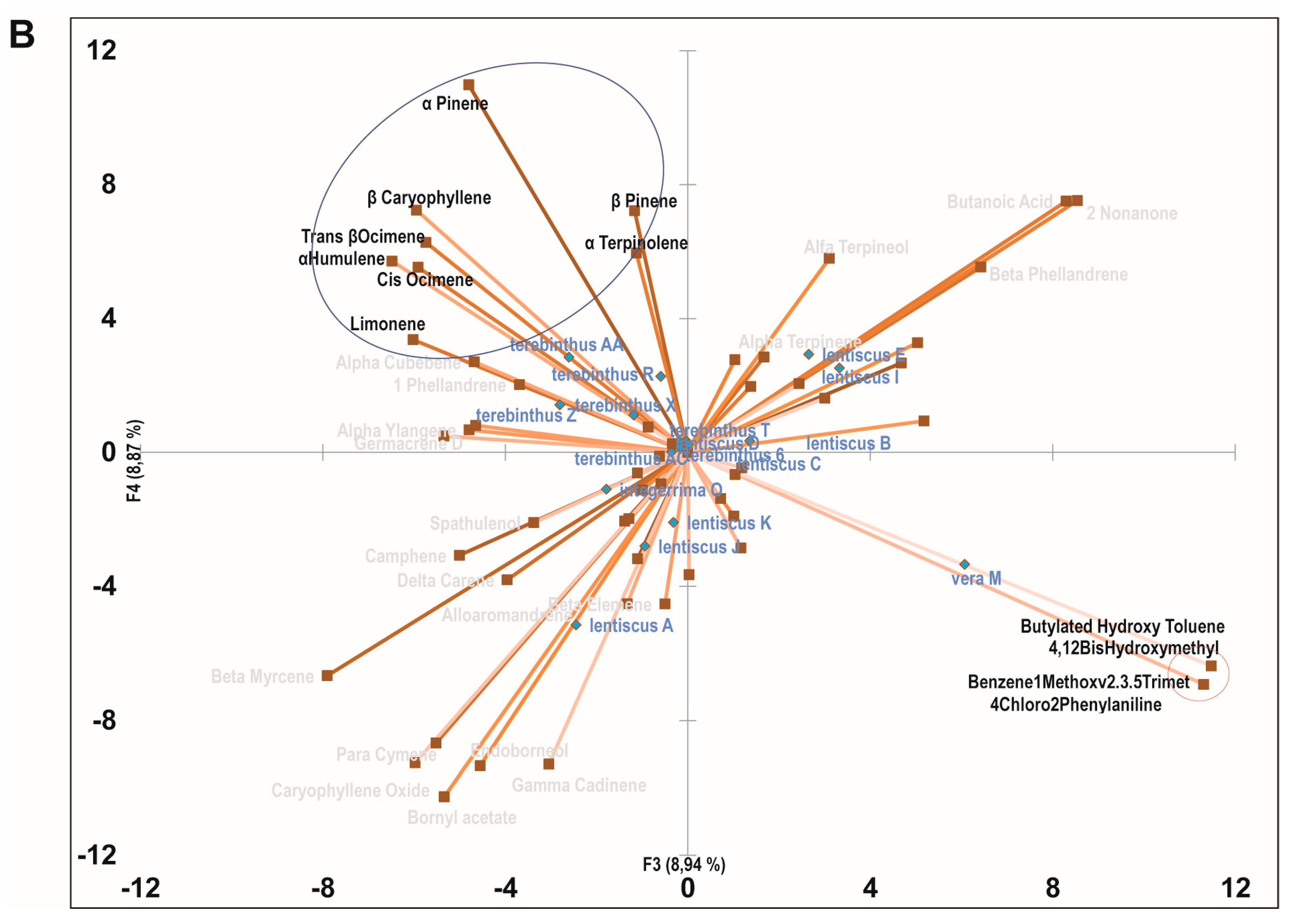
2.3. Principal Component Analysis of the Bioactive Essential Oils
3. Materials and Methods
3.1. Chemicals
3.2. Plant Collection
3.3. Extraction of the Essential Oil
3.4. Phytochemical Determination of Essential Oil Constituents
3.5. Sample Preparation for Cell Assays
3.6. Cell Lines
3.7. Cytotoxicity
3.8. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Buriani, A.; Fortinguerra, S.; Carrara, M.; Pelkonen, O. Systems Network Pharmaco-Toxicology in the Study of Herbal Medicines. In Toxicology of Herbal Products; Pelkonen, O., Duez, P., Vuorela, P.M., Vuorela, H., Eds.; Springer: Cham, Switzerland, 2017; pp. 129–164. [Google Scholar]
- Buriani, A.; Fortinguerra, S. The systems biology oriented, holistic vision of personalized medicine and the emerging concept of proactive herbal medicine. Jacobs J. Intern Med. 2015, 1, 007. [Google Scholar]
- Buriani, A.; Garcia-Bermejo, M.L.; Bosisio, E.; Xu, Q.; Li, H.; Dong, X.; Simmonds, M.S.; Carrara, M.; Tejedor, N.; Lucio-Cazana, J.; et al. Omic techniques in systems biology approaches to traditional Chinese medicine research: present and future. J. Ethnopharmacol. 2012, 140, 535–544. [Google Scholar] [CrossRef] [PubMed]
- Bilecová-Rabajdová, M.; Birková, A.; Urban, P.; Gregová, K.; Durovcová, E.; Mareková, M. Naturally occurring substances and their role in chemo-protective effects. Cent. Eur. J. Public Health 2013, 21, 213–219. [Google Scholar] [PubMed]
- Crowell, P.L. Prevention and therapy of cancer by dietary monoterpenes. J. Nutr. 1999, 129, 775S–778S. [Google Scholar] [PubMed]
- Morgan-Meadows, S.; Dubey, S.; Gould, M.; Tutsch, K.; Marnocha, R.; Arzoomanin, R.; Alberti, D.; Binger, K.; Feierabend, C.; Volkman, J.; et al. Phase I trial of perillyl alcohol administered four times daily continuously. Cancer Chemother. Pharmacol. 2003, 52, 361–366. [Google Scholar] [CrossRef] [PubMed]
- Bozorgi, M.; Memariani, Z.; Mobli, M.; Salehi Surmaghi, M.H.; Shams-Ardekani, M.R.; Rahimi, R. Five Pistacia. species (P. vera, P. atlantica, P. terebinthus, P. khinjuk, and P. lentiscus): A review of their traditional uses, phytochemistry, and pharmacology. Sci. World J. 2013, 15, e219815. [Google Scholar]
- Lesgards, J.F.; Baldovini, N.; Vidal, N.; Pietri, S. Anticancer activities of essential oils constituents and synergy with conventional therapies: A review. Phytother. Res. 2014, 28, 1423–1446. [Google Scholar] [CrossRef] [PubMed]
- Bayala, B.; Bassole, I.H.N.; Scifo, R.; Gnoula, C.; Morel, L.; Lobaccaro, J.M.A.; Simpore, J. Anticancer activity of essential oils and their chemical components—A review. Am. J. Cancer Res. 2014, 4, 591–607. [Google Scholar] [PubMed]
- Homburger, F.; Treger, A.; Boger, E. Inhibition of murine subcutaneous and intravenous benzo (rst) pentaphene. Carcinogenesis by sweet orange oils and d-limonene. Oncology 1971, 25, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Wattenberg, L.W.; Coccia, J.B. Inhibition of 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone carcinogenesis in mice by D-limonene and citrus fruit oils. Carcinogenesis 1991, 12, 115–117. [Google Scholar] [CrossRef] [PubMed]
- Haag, J.D.; Lindstrom, M.J.; Gould, M.N. Limonene-induced regression of mammary carcinomas. Cancer Res. 1992, 15, 4021–4026. [Google Scholar]
- Gould, M.N.; Moore, C.J.; Zhang, R.; Wang, B.; Kennan, W.S.; Haag, J.D. Limonene chemoprevention of mammary carcinoma induction following direct in situ transfer of v-Ha-ras. Cancer Res. 1994, 54, 3540–3543. [Google Scholar] [PubMed]
- Lu, X.G.; Zhan, L.B.; Feng, B.A.; Qu, M.Y.; Yu, L.H.; Xie, J.H. Inhibition of growth and metastasis of human gastric cancer implanted in nude mice by d-limonene. World J. Gastroenterol. 2004, 10, 2140–2144. [Google Scholar] [CrossRef] [PubMed]
- Sobral, M.V.; Xavier, A.L.; Lima, T.C.; de Sousa, D.; Pergentino, O. Antitumor activity of monoterpenes found in essential oils. Sci. World J. 2014, 2014, e953451. [Google Scholar] [CrossRef] [PubMed]
- Carnesecchi, S.; Bras-Gonçalves, R.; Bradaia, A.; Zeisel, M.; Gossé, F.; Poupon, M.F.; Raul, F. Geraniol, a component of plant essential oils, modulates DNA synthesis and potentiates 5-fluorouracil efficacy on human colon tumor xenografts. Cancer Lett. 2004, 215, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Lampronti, I.; Saab, A.M.; Gambari, R. Antiproliferative activity of essential oils derived from plants belonging to the Magnoliophyta division. Int. J. Oncol. 2006, 29, 989–995. [Google Scholar] [CrossRef] [PubMed]
- Legault, J.; Pichette, A. Potentiating effect of beta-caryophyllene on anticancer activity of alpha-humulene, isocaryophyllene and paclitaxel. J. Pharm. Pharmacol. 2007, 59, 1643–1647. [Google Scholar] [CrossRef] [PubMed]
- Pal, D.; Banerjee, S.; Mukherjee, S.; Roy, A.; Panda, C.K.; Das, S. Eugenol restricts DMBA croton oil induced skin carcinogenesis in mice: Downregulation of c-Myc and H-ras, and activation of p53 dependent apoptotic pathway. J. Dermatol. Sci. 2010, 59, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Cardozo, M.T.; de Conti, A.; Ong, T.P.; Scolastici, C.; Purgatto, E.; Horst, M.A.; Bassoli, B.K.; Moreno, F.S. Chemopreventive effects of β-ionone and geraniol during rat hepatocarcinogenesis promotion: Distinct actions on cell proliferation, apoptosis, HMGCoA reductase, and RhoA. J. Nutr. Biochem. 2011, 22, 130–135. [Google Scholar] [CrossRef] [PubMed]
- Satooka, H.; Kubo, I. Effects of thymol on B16-F10 melanoma cells. J. Agric. Food Chem. 2012, 60, 2746–2752. [Google Scholar] [CrossRef] [PubMed]
- Al-Saghir, M.G.; Porter, D.M. Taxonomic Revision of the Genus Pistacia L. (Anacardiaceae). AJPS 2012, 3, 12–32. [Google Scholar] [CrossRef]
- Zohara, Y.; Nativ, D. Medicinal and Aromatic Plants of the Middle-East; Springer: Cham, Switzerland, 2014. [Google Scholar]
- Loutrari, H.; Magkouta, S.; Pyriochou, A.; Koika, V.; Kolisis, F.N.; Papapetropoulos, A.; Roussos, C.; Roussos, C. Mastic oil from Pistacia lentiscus var. chia inhibits growth and survival of human K562 leukemia cells and attenuates angiogenesis. Nutr. Cancer 2006, 55, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Balan, K.V.; Prince, J.; Han, Z.; Dimas, K.; Cladaras, M.; Wyche, J.H.; Sitaras, N.M.; Pantazis, P.; Pantazis, P. Antiproliferative activity and induction of apoptosis in human colon cancer cells treated in vitro with constituents of a product derived from Pistacia lentiscus L. var. chia. Phytomedicine 2007, 14, 263–272. [Google Scholar] [CrossRef] [PubMed]
- Loutrari, H.; Magkouta, S.; Papapetropoulos, A.; Roussos, C. Mastic oil inhibits the metastatic phenotype of mouse lung adenocarcinoma cells. Cancers 2011, 3, 789–801. [Google Scholar] [CrossRef] [PubMed]
- Giaginis, C.; Theocharis, S. Current evidence on the anticancer potential of Chios mastic gum. Nutr. Cancer 2011, 63, 1174–1184. [Google Scholar] [CrossRef] [PubMed]
- Morad, S.A.; Schmidt, C.; Büchele, B.; Schneider, B.; Wenzler, M.; Syrovets, T.; Simmet, T.; Simmet, T. (8R)-3β,8-Dihydroxypolypoda-13E,17E,21-triene induces cell cycle arrest and apoptosis in treatment-resistant prostate cancer cells. J. Nat. Prod. 2011, 74, 1731–1736. [Google Scholar] [CrossRef] [PubMed]
- Dimas, K.; Hatziantoniou, S.; Wyche, J.H.; Pantazis, P. A mastic gum extract induces suppression of growth of human colorectal tumor xenografts in immunodeficient mice. In Vivo 2009, 23, 63–68. [Google Scholar] [PubMed]
- Catalani, S.; Palma, F.; Battistelli, S.; Benedetti, S. Oxidative stress and apoptosis induction in human thyroid carcinoma cells exposed to the essential oil from Pistacia lentiscus aerial parts. PLoS ONE 2017, 12, e0172138. [Google Scholar] [CrossRef] [PubMed]
- Spyridopoulou, K.; Tiptiri-Kourpeti, A.; Lampri, E.; Fitsiou, E.; Vasileiadis, S.; Vamvakias, M.; Bardouki, H.; Goussia, A.; Malamou-Mitsi, V.; Panayiotidis, M.I.; et al. Dietary mastic oil extracted from Pistacia lentiscus var. chia suppresses tumor growth in experimental colon cancer models. Sci. Rep. 2017, 7, 3782. [Google Scholar] [CrossRef] [PubMed]
- Almehdar, H.; Abdallah, H.M.; Osman, A.M.M.; Abdel-Sattar, E.A. In vitro cytotoxic screening of selected Saudi medicinal plants. J. Nat. Med. 2012, 66, 406–412. [Google Scholar] [CrossRef] [PubMed]
- Bibi, Y.; Nisa, S.; Zia, M.; Waheed, A.; Ahmed, S.; Chaudhary, M.F. The study of anticancer and antifungal activities of Pistacia integerrima extract in vitro. Indian J. Pharm. Sci. 2012, 74, 375–379. [Google Scholar] [PubMed]
- Rezaei, P.F.; Fouladdel, S.; Hassani, S.; Yousefbeyk, F.; Ghaffari, S.M.; Amin, G.; Ebrahim, A.; Ebrahim, A. Induction of apoptosis and cell cycle arrest by pericarp polyphenol-rich extract of Baneh in human colon carcinoma HT29 cells. Food Chem. Toxicol. 2012, 50, 1054–1059. [Google Scholar] [CrossRef] [PubMed]
- Fathalizadeh, J.; Bagheri, V.; Khorramdelazad, H.; Kazemi Arababadi, M.; Jafarzadeh, A.; Mirzaei, M.R.; Shamsizadeh, A.; Hajizadeh, M.R. Induction of apoptosis by pistachio (Pistacia vera L.) hull extract and its molecular mechanisms of action in human hepatoma cell line HepG2. Cell. Mol. Biol. 2015, 61, 128–134. [Google Scholar] [PubMed]
- Mirian, M.; Behrooeian, M.; Ghanadian, M.; Dana, N.; Sadeghi-Aliabadi, H. Cytotoxicity and antiangiogenic effects of Rhus coriaria, Pistacia vera and Pistacia khinjuk oleoresin methanol extracts. Res. Pharm. Sci. 2015, 10, 233–240. [Google Scholar] [PubMed]
- Mezni, F.; Shili, S.; Ben Ali, N.; Larbi Khouja, M.; Khaldi, A.; Maaroufi, A. Evaluation of Pistacia lentiscus seed oil and phenolic compounds for in vitro antiproliferative effects against BHK21 cells. Pharm. Biol. 2016, 54, 747–751. [Google Scholar] [CrossRef] [PubMed]
- Piccolella, S.; Nocera, P.; Carillo, P.; Woodrow, P.; Greco, V.; Manti, L.; Fiorentino, A.; Pacifico, S. An apolar Pistacia lentiscus L. leaf extract: GC-MS metabolic profiling and evaluation of cytotoxicity and apoptosis inducing effects on SH-SY5Y and SK-N-BE(2)C cell lines. Food Chem. Toxicol. 2016, 95, 64–74. [Google Scholar] [CrossRef] [PubMed]
- Loizzo, M.R.; Tundis, R.; Menichini, F.; Saab, A.M.; Statti, G.A.; Menichini, F. Cytotoxic activity of essential oils from labiatae and lauraceae families against in vitro human tumor models. Anticancer Res. 2007, 27, 3293–3299. [Google Scholar] [PubMed]
- Da Silva, J.K.; Da Trindade, R.; Moreira, E.C.; Maia, J.G.S.; Dosoky, N.S.; Miller, R.S.; Cseke, L.J.; Setzer, W.N. Chemical Diversity, Biological Activity, and GeneticAspects of Three Ocotea Species from the Amazon. Nat. Prod. Commun. 2013, 8, 1481–1486. [Google Scholar]
- Paudel, P.; Satyal, P.; Dosoky, N.S.; Maharjan, S.; Setzer, W.N. Juglans regia and J. nigra, two trees important in traditional medicine: A comparison of leaf essential oil compositions and biological activities. Int. J. Mol. Sci. 2017, 18, 1081. [Google Scholar]
- Kaneda, N.; Pezzuto, J.M.; Kinghorn, A.D.; Farnsworth, N.R.; Santisuk, T.; Tuchinda, P.; Udchachon, J.; Reutrakul, V. Plant anticancer agents, L. cytotoxic triterpenes from Sandoricum koetjape stems. J. Nat. Prod. 1992, 55, 654–659. [Google Scholar] [PubMed]
- Graidist, P.; Martla, M.; Sukpondma, Y. Cytotoxic activity of Piper cubeba extract in breast cancer cell lines. Nutrients 2015, 10, 2707–2718. [Google Scholar] [CrossRef] [PubMed]
- Russo, R.; Corasaniti, M.T.; Bagetta, G.; Morrone, L.A. Exploitation of cytotoxicity of some essential oils for translation in cancer therapy. Evid. Based Complement. Altern. Med. 2015, 2015, 397821. [Google Scholar] [CrossRef] [PubMed]
- Owolabi, M.S.; Ogundajo, A.L.; Dosoky, N.S.; Setzer, W.N. The cytotoxic activity of annona muricata leaf oil from badagary, Nigeria. Am. J. Essent. Oil Nat. Prod. 2013, 1, 1–3. [Google Scholar]
- Shapira, S.; Pleban, S.; Kazanov, D.; Tirosh, P.; Arber, N. Terpinen-4-ol: A novel and promising therapeutic agent for human gastrointestinal cancers. PLoS ONE 2016, 11, e0156540. [Google Scholar] [CrossRef] [PubMed]
- Boelens, M.H.; Jimenez, R. Chemical composition of the essential oils from the gum and from various parts of Pistacia lentiscus L. (mastic gum tree). Flavour Fragr. J. 1991, 6, 271–275. [Google Scholar] [CrossRef]
- De Pooter, H.L.; Schamp, N.M.; Aboutabl, E.A.; El Tohamy, S.F.; Doss, S.L. Essential oils from the leaves of three Pistacia species grown in Egypt. Flavour Fragr. J. 1991, 6, 229–232. [Google Scholar] [CrossRef]
- Magiatis, P.; Melliou, E.; Skaltsounis, A.L.; Chinou, I.B.; Mitaku, S. Chemical composition and antimicrobial activity of the essential oils of Pistacia lentiscus var. chia. Planta Med. 1999, 65, 749–752. [Google Scholar] [CrossRef] [PubMed]
- Papageorgiou, V.P.; Argyriadou, N. Trace constituents in the essential oil of Thymus capitatus. Phytochemistry 1981, 20, 2295–2297. [Google Scholar] [CrossRef]
- Gardeli, C.; Vassiliki, P.; Athanasios, M.; Kibouris, T.; Komaitis, M. Essential oil composition of Pistacia lentiscus L. and Myrtus communis L.: Evaluation of antioxidant capacity of methanolic extracts. Food Chem. 2008, 107, 1120–1130. [Google Scholar] [CrossRef]
Sample Availability: Samples of essential oils can be available from the authors. |
| RT | Oil Composition | P. lentiscus | var. chia | P. vera | P. integerrima | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| A (l) | B (l) | C (l) | D (l) | E (l) | F (l) | G (be) | H (be) | I (be) | J (br) | K (br) | L (r) | M (n) | N (g) | O (g) | ||
| 6.08 | Tricyclene | - | 0.33 | 0.58 | 0.14 | 0.06 | 1.20 | - | - | - | 0.90 | - | - | - | - | - |
| 6.22 | α-Phellandrene | - | 1.51 | 0.12 | 0.14 | 0.20 | 0.15 | 0.61 | - | 0.20 | - | 0.14 | - | - | - | - |
| 6.48 | α-Pinene | 12.95 | 11.45 | 12.97 | 9.17 | 24.68 | 12.86 | 20.13 | 6.11 | 40.68 | 18.15 | 13.73 | 72.93 | 0.99 | 14.41 | 22.37 |
| 6.94 | Camphene | 2.05 | 1.46 | 2.49 | 0.52 | 0.58 | 4.96 | 1.06 | - | 0.98 | 3.90 | 0.22 | 0.58 | - | 0.94 | 1.32 |
| 7.78 | Sabinene | 1.16 | 4.31 | 4.56 | 2.87 | 2.32 | 4.47 | 0.99 | 1.21 | 0.86 | 2.83 | 6.77 | 0.30 | - | 2.51 | 2.46 |
| 7.88 | β-Pinene | 4.33 | 3.07 | 4.38 | 1.15 | 8.64 | 4.25 | 4.13 | 0.48 | 7.60 | 4.19 | 0.94 | 2.58 | - | 5.62 | 7.63 |
| 8.40 | β-Myrcene | 8.42 | 0.97 | 0.67 | 4.45 | 0.91 | 9.23 | 23.80 | 57.79 | 1.08 | 4.08 | 5.47 | 13.57 | - | 2.81 | 3.20 |
| 8.86 | 1-Phellandrene | 0.69 | 4.64 | 0.80 | 4.60 | 2.12 | 2.07 | 3.84 | 3.79 | 0.92 | 0.81 | 8.35 | - | - | 0.56 | 0.77 |
| 9.02 | Methylanisol | - | - | - | - | - | - | - | - | - | - | - | 0.58 | - | - | - |
| 9.07 | δ-Carene | 0.67 | - | - | - | - | - | - | - | - | 3.14 | - | - | - | 4.06 | 8.36 |
| 9.33 | α-Terpinene | 0.56 | 4.66 | 3.60 | 3.76 | 3.11 | 2.82 | 2.71 | 0.90 | 1.13 | 0.72 | 1.73 | - | - | 0.80 | 1.80 |
| 9.69 | para-Cymene | 6.43 | 0.64 | 0.22 | 0.50 | 0.35 | 0.30 | 0.48 | 0.34 | 0.16 | 0.42 | 0.98 | - | - | 2.09 | 3.72 |
| 9.81 | β-Phellandrene | - | 6.48 | 4.93 | 5.79 | 11.35 | 4.73 | 6.48 | 3.44 | 8.03 | - | 6.61 | - | - | - | - |
| 9.89 | Limonene | 17.55 | - | - | - | - | - | - | - | - | - | - | 0.89 | 1.54 | 5.94 | 7.81 |
| 10.21 | cis-Ocimene | - | 0.60 | 0.35 | 1.00 | 0.72 | 0.52 | - | 0.29 | 0.22 | 1.08 | 0.61 | - | - | - | - |
| 10.62 | Trans-β-Ocimene | 0.14 | 0.41 | 0.23 | 0.53 | 0.46 | 0.35 | - | 0.57 | - | 0.32 | 0.23 | - | - | - | - |
| 10.96 | Butanoic Acid | - | - | - | - | 0.55 | - | - | - | 1.07 | - | - | - | - | - | - |
| 11.03 | γ-Terpinene | 1.24 | 7.42 | 5.90 | 6.11 | 4.89 | 4.94 | 4.93 | 1.85 | 2.24 | 1.51 | 2.96 | - | - | 1.52 | 3.74 |
| 12.26 | α-Terpinolene | 0.61 | 2.48 | 2.08 | 2.35 | 2.59 | 1.75 | 2.08 | 0.63 | 2.27 | 0.64 | 1.04 | - | - | 0.57 | 1.61 |
| 12.49 | 2-Nonanone | - | 0.34 | 0.17 | 0.19 | 0.63 | 0.30 | 0.44 | 0.31 | 1.65 | - | - | - | - | - | - |
| 12.60 | α-Pinene Oxide | - | - | - | - | - | - | - | - | - | - | - | 0.56 | - | - | - |
| 12.74 | Linalool | 0.11 | - | 0.15 | 0.22 | 0.39 | - | - | - | - | - | 0.36 | 0.73 | - | - | - |
| 12.96 | Pinocarveol | - | - | - | - | - | - | - | - | - | - | - | 0.21 | - | - | - |
| 14.64 | Pinocarvone | - | - | - | - | - | - | - | - | - | - | - | 0.10 | - | - | - |
| 14.70 | Camphor | 0.13 | - | - | - | 0.08 | 0.31 | - | - | - | - | - | - | - | - | - |
| 15.74 | Endoborneol | 6.77 | 0.12 | 0.34 | - | 0.24 | 0.43 | 0.32 | - | 0.65 | - | - | - | - | 3.12 | 1.02 |
| 16.20 | 1-4 Terpineol | 3.37 | 14.89 | 12.26 | 13.11 | 7.12 | 10.43 | 8.78 | 4.09 | 2.63 | 3.03 | 6.98 | - | 2.13 | 29.52 | 20.69 |
| 16.79 | α-Terpineol | 0.78 | 4.92 | 5.36 | 6.42 | 7.41 | 4.96 | 7.97 | 1.11 | 9.83 | 1.41 | 1.13 | - | 2.56 | 14.08 | 8.74 |
| 17.48 | Myrtenol | - | - | - | - | - | - | - | - | - | - | - | 0.18 | - | - | - |
| 19.44 | Octanoid Acid | - | 0.18 | 0.13 | 0.22 | - | 0.19 | - | - | 0.54 | - | 0.17 | - | - | - | - |
| 21.04 | Bornyl Acetate | 24.48 | 2.12 | 1.77 | 0.17 | - | 0.52 | 1.94 | - | - | 0.98 | 0.28 | - | - | 4.55 | 1.55 |
| 21.39 | Verbenone | - | - | - | - | - | - | - | - | - | - | - | 0.26 | - | - | - |
| 21.42 | 2-Undecanone | 0.30 | 0.69 | 0.19 | 0.55 | 0.99 | 0.67 | 0.41 | 0.59 | 1.38 | 1.47 | 0.65 | - | - | - | - |
| 24.77 | α-Ylangene | - | 0.42 | 0.87 | 0.70 | - | 0.30 | - | 0.32 | - | 0.46 | 0.60 | - | - | - | - |
| 25.39 | β-Cubebene | - | - | 0.25 | 0.21 | - | - | - | - | - | - | 0.34 | - | - | - | - |
| 25.47 | β-Elemene | - | 0.15 | 0.19 | 0.23 | 0.11 | 0.16 | - | - | - | 0.52 | 0.51 | - | - | - | - |
| 26.61 | β-Caryophyllene | 2.09 | 2.79 | 3.21 | 3.24 | 4.02 | 4.92 | 0.44 | 0.81 | 1.18 | 3.91 | 1.70 | 0.30 | 0.60 | 2.81 | 1.96 |
| 27.85 | α-Cubebene | - | - | 0.05 | 0.24 | - | - | - | - | - | - | - | - | - | - | - |
| 27.96 | α-Humulene | 0.17 | 1.02 | 0.88 | 1.13 | 0.48 | 1.09 | 0.25 | 0.37 | - | 1.10 | 0.60 | - | - | - | - |
| 28.25 | Alloaromandrene | 0.10 | 0.30 | 0.34 | 0.39 | - | 0.24 | - | - | - | 0.44 | 0.48 | - | - | - | - |
| 28.99 | α-Amorphene | 0.37 | 0.85 | 1.51 | 1.30 | 0.17 | 0.64 | 0.35 | 0.80 | 0.39 | 1.16 | 1.13 | - | - | - | - |
| 29.10 | Germacrene D | 0.25 | 4.71 | 6.82 | 5.00 | 1.37 | 4.34 | 2.06 | 2.76 | 1.23 | 8.36 | 8.32 | - | 0.62 | - | - |
| 29.66 | α-Elemene | - | 0.33 | 0.41 | 0.51 | 0.26 | 0.34 | - | - | 0.35 | 0.90 | 0.99 | - | - | - | - |
| 29.89 | α-Muurolene | 0.13 | 0.71 | 1.21 | 1.07 | 0.33 | 0.68 | - | 0.68 | 0.44 | 0.98 | 0.93 | - | - | - | - |
| 30.05 | Germacrene A | - | 0.14 | 0.18 | 0.30 | 0.16 | 0.23 | - | - | - | 0.44 | 0.68 | - | - | - | - |
| 30.42 | Butylated Hydroxy Toluene | - | 0.60 | 0.81 | 1.36 | 0.44 | 1.14 | 0.88 | 1.23 | 1.72 | 4.33 | 1.25 | - | 15.74 | - | - |
| 30.81 | δ-Cadinene | 0.20 | 2.82 | 4.80 | 4.40 | 1.11 | 2.52 | 1.32 | 3.28 | 1.74 | 4.44 | 3.99 | - | 0.66 | - | - |
| 32.26 | Spathulenol | - | - | - | - | - | - | - | - | - | - | - | - | - | 1.26 | 0.53 |
| 32.78 | γ-Cadinene | 0.54 | - | - | - | - | - | - | - | - | 0.19 | 0.87 | - | - | - | - |
| 33.20 | Caryophyllene Oxide | 2.53 | - | - | - | 0.12 | - | - | - | - | 0.83 | - | - | - | - | - |
| 34.79 | Cadina-1,4-Diene | - | 0.83 | 1.26 | 1.50 | 0.20 | 0.61 | - | 0.46 | - | 0.86 | 0.71 | - | - | - | - |
| 34.92 | γ-Eudesmol | - | 1.21 | 0.96 | 1.07 | 0.48 | 0.77 | 0.41 | 0.63 | 0.28 | - | 0.61 | - | - | - | - |
| 35.31 | α-Cadinol | - | 2.50 | 3.29 | 4.13 | 1.63 | 2.52 | 0.76 | 1.96 | 0.97 | 3.18 | 2.70 | - | - | - | - |
| 35.45 | α-Copaene | - | 0.75 | 1.03 | 1.29 | 0.49 | 0.76 | - | 0.53 | 0.31 | 1.22 | 0.81 | - | - | - | - |
| 35.58 | β-Eudesmol | - | 0.47 | 0.31 | 0.41 | 0.19 | 0.28 | - | - | 0.20 | - | 0.46 | - | - | - | - |
| 35.80 | T-Cadinolo | 0.85 | 3.16 | 3.90 | 4.73 | 2.61 | 3.55 | 1.15 | 2.79 | 1.68 | 4.53 | 3.77 | - | - | - | - |
| 56.28 | Nonadecane | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| 61.56 | Octadecane | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| 67.79 | CPA 1 | - | - | - | - | - | - | - | - | - | - | - | - | 32.88 | - | - |
| 67.96 | Benzene1Methox 2 | - | - | - | - | - | - | - | - | - | - | - | - | 25.63 | - | - |
| 69.78 | 4,12BisHydroxy 3 | - | - | - | - | - | - | - | - | - | - | - | - | 4.29 | - | - |
| RT | Oil Composition | P (l) | Q (l) | R (l) | S (l) | T (l) | U (l) | V (l) | W (l) | X (l) | Y (be) | Z (be) | AA (g) | AB (g) | AC (f) | AD (f) | AE (f) | AF (br) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 6.08 | Tricyclene | - | - | - | - | - | 0.25 | - | - | - | 0.30 | - | - | - | 0.48 | 0.45 | - | 0.18 |
| 6.22 | α-Phellandrene | - | - | - | - | - | - | - | - | - | - | - | - | - | 0.21 | - | - | 0.36 |
| 6.48 | α-Pinene | 35.6 | 12.17 | 41.03 | 11.61 | 14.1 | 60.9 | 10.0 | 10.99 | 18.7 | 31.63 | 8.69 | 54.19 | 42.16 | 28.90 | 26.43 | 11.74 | 28.72 |
| 6.94 | Camphene | 0.55 | 0.26 | 1.02 | - | - | 1.56 | 0.24 | 0.37 | 0.46 | 1.50 | 0.48 | 0.69 | 0.67 | 2.12 | 1.83 | - | 1.06 |
| 7.78 | Sabinene | - | 0.62 | - | 0.39 | - | 0.22 | 0.15 | 0.28 | - | 1.20 | 0.85 | 0.65 | 0.18 | 2.00 | 0.91 | 1.41 | 4.53 |
| 7.88 | β-Pinene | 1.12 | 1.14 | 6.17 | 1.39 | 0.71 | 1.87 | 0.32 | 1.48 | 3.27 | 7.44 | 1.39 | 3.68 | 1.81 | 6.40 | 5.85 | 2.12 | 4.07 |
| 8.40 | β-Myrcene | 1.57 | 1.68 | 1.37 | 1.82 | 2.03 | 1.98 | 1.41 | 1.60 | 2.90 | 1.21 | 1.11 | 1.05 | 1.42 | 1.68 | 0.97 | 31.56 | 1.67 |
| 8.86 | 1-Phellandrene | 0.62 | 0.35 | 0.49 | 0.75 | 0.62 | 0.47 | 0.19 | 0.29 | 0.27 | 3.34 | 7.66 | 0.49 | 6.61 | 0.32 | 0.37 | 0.61 | 0.59 |
| 9.02 | Methylanisol | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| 9.07 | δ-Carene | - | - | - | 1.23 | - | 0.76 | - | - | - | - | - | - | - | - | - | - | - |
| 9.33 | α-Terpinene | - | 0.43 | 0.83 | 0.71 | - | 0.44 | - | 0.21 | 0.34 | 0.25 | 0.34 | - | - | 0.65 | 0.51 | - | 1.46 |
| 9.69 | Para Cymene | - | 0.11 | - | - | - | 0.17 | - | - | - | 0.38 | - | - | 0.18 | 0.20 | - | 0.79 | 0.49 |
| 9.81 | β-Phellandrene | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| 9.89 | Limonene | 27.4 | 50.70 | 6.09 | 21.04 | 60.4 | 6.16 | 1.23 | 8.03 | 60.2 | 12.59 | 32.85 | 13.97 | 31.07 | 28.76 | 23.03 | 5.58 | 6.56 |
| 10.21 | Cis-Ocimene | 19.6 | 15.64 | 10.93 | 20.97 | 3.86 | 6.31 | 63.6 | 54.19 | 0.96 | 6.79 | 17.99 | 1.89 | 0.37 | - | 3.88 | - | 13.44 |
| 10.62 | Transβ-Ocimene | 5.49 | 4.45 | 3.80 | 6.04 | 1.17 | 1.86 | 18.3 | 16.46 | 0.38 | 1.66 | 5.01 | 0.40 | - | - | 1.22 | - | 3.33 |
| 10.96 | Butanoic Acid | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| 11.03 | γ-Terpinene | 0.22 | 0.69 | 0.48 | 0.62 | - | 0.49 | 0.17 | 0.34 | 0.60 | 0.33 | 0.42 | 0.12 | - | 1.08 | 0.77 | 0.46 | 2.46 |
| 12.26 | α-Terpinolene | 0.92 | 0.93 | 17.07 | 16.59 | 4.90 | 7.13 | 0.38 | 0.46 | 1.76 | 0.44 | 1.27 | 0.26 | 0.28 | 0.57 | 2.99 | - | 1.08 |
| 12.49 | 2-Nonanone | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| 12.60 | α-Pinene Oxide | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| 12.80 | Linalool | - | - | - | - | - | 0.18 | - | - | - | - | 0.17 | - | - | - | - | - | - |
| 12.96 | Pinocarveol | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| 14.64 | Pinocarvone | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| 14.70 | Camphor | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| 15.74 | Endoborneol | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| 16.20 | 1-4 Terpineol | 0.31 | 1.63 | 0.29 | 1.86 | 1.21 | 0.29 | 0.35 | 0.50 | 0.46 | 1.00 | 0.87 | 0.18 | 0.13 | 2.86 | 1.55 | - | 8.91 |
| 16.79 | α-Terpineol | 1.63 | 4.05 | 5.22 | 1.92 | 1.18 | 2.23 | 1.35 | 1.22 | 2.87 | 2.43 | 0.93 | 0.35 | 1.36 | 1.69 | 1.45 | - | 2.26 |
| 17.48 | Myrtenol | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| 19.44 | Octanoid Acid | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| 21.04 | Bornyl Acetate | - | - | 0.47 | - | - | - | - | - | - | 0.64 | 0.35 | - | 0.18 | - | - | - | 0.23 |
| 21.39 | Verbenone | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| 21.42 | 2-Undecanone | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| 24.77 | α-Ylangene | 0.23 | 0.16 | - | - | - | - | - | - | - | 4.51 | 0.96 | 0.15 | 0.19 | - | 2.14 | - | 0.51 |
| 25.39 | β-Cubebene | - | - | - | - | - | - | - | - | - | 1.60 | 0.40 | - | - | - | 0.37 | - | 0.19 |
| 25.47 | β-Elemene | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| 26.61 | β-Caryophyllene | 3.23 | 1.38 | 2.75 | 4.72 | 2.00 | 3.86 | 0.62 | 1.29 | 3.43 | 4.77 | 1.64 | 14.33 | 0.94 | 0.97 | 8.05 | 0.63 | 1.06 |
| 27.85 | α-Cubebene | - | - | - | - | - | - | - | - | - | 0.26 | 0.42 | - | - | - | - | - | 0.74 |
| 27.96 | α-Humulene | 0.47 | 0.33 | - | 0.86 | 1.07 | 1.21 | - | - | 1.46 | 1.47 | 0.55 | 2.15 | 0.33 | 0.65 | 1.24 | - | 0.22 |
| 28.25 | Alloaromandrene | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| 28.99 | α-Amorphene | - | 0.24 | - | - | - | - | 0.17 | - | 0.30 | 0.55 | 0.24 | - | 0.15 | 0.43 | 0.48 | 1.27 | 0.77 |
| 29.10 | Germacrene D | 0.23 | 0.62 | - | 1.56 | 1.40 | - | 0.14 | 0.45 | - | 2.24 | 9.42 | 2.91 | 7.08 | 5.87 | 2.44 | 4.45 | 2.91 |
| 29.66 | α-Elemene | - | - | - | - | - | - | - | - | - | 0.18 | - | - | - | - | - | - | 0.46 |
| 29.89 | α-Muurolene | - | - | - | - | - | - | - | - | - | 0.25 | - | - | - | 0.33 | 0.27 | 0.76 | 0.47 |
| 30.05 | Germacrene A | - | - | - | - | - | - | - | - | - | - | - | - | 0.14 | - | - | - | - |
| 30.42 | Butylated Hydroxy Toluene | 0.32 | 0.77 | 0.37 | 3.34 | 2.36 | 0.62 | 0.46 | 0.45 | 0.55 | 0.58 | 0.46 | 0.30 | 0.45 | 1.56 | 1.58 | - | 1.82 |
| 30.81 | δ-Cadinene | 0.44 | 0.67 | - | 1.32 | 0.92 | 0.31 | 0.31 | 0.63 | 0.61 | 6.42 | 2.33 | 0.31 | 0.55 | 1.22 | 3.61 | 3.06 | 3.51 |
| 32.26 | Spathulenol | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| 32.78 | γ-Cadinene | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 0.84 | - |
| 33.20 | Caryophyllene Oxide | - | 0.23 | - | - | - | 0.27 | - | - | - | 0.40 | - | 0.39 | - | - | 0.75 | - | - |
| 34.79 | Cadina-1,4-Diene | - | - | - | - | - | - | - | - | - | 0.31 | 0.17 | - | - | - | 0.36 | - | 0.31 |
| 34.92 | γ-Eudesmol | - | - | - | - | - | - | - | - | - | - | - | - | 0.70 | - | - | - | - |
| 35.31 | α-Cadinol | - | - | - | - | - | - | - | - | - | 0.26 | - | - | 0.21 | 0.81 | 0.73 | - | 0.53 |
| 35.45 | α-Copaene | - | - | - | - | - | - | - | - | - | 0.39 | 0.55 | - | - | 0.25 | 0.19 | - | - |
| 35.58 | β-Eudesmol | - | - | - | - | - | - | - | - | - | - | - | - | 0.21 | - | - | - | - |
| 35.80 | T-Cadinolo | - | 0.17 | - | - | - | - | - | - | - | 0.31 | 0.65 | - | 0.30 | 1.14 | 0.87 | 1.26 | 0.36 |
| 56.28 | Nonadecane | - | - | - | - | - | - | - | - | - | - | - | - | - | 2.20 | 1.04 | - | - |
| 61.56 | Octadecane | - | - | - | 0.47 | - | - | - | - | 0.25 | - | - | - | - | 1.99 | 0.71 | - | - |
| 67.79 | CPA 1 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| 67.96 | Benzene1Methox 2 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| 69.78 | 4,12BisHydroxy 3 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Samples | LoVo Cells | 2008 Cells | MCF-7 Cells |
|---|---|---|---|
| A | 173.9 (151.0–200.3) µg/mL | 181.5 (166.5–197.7) µg/mL | 249.4 (243.5–255.4) µg/mL |
| n = 3 | n = 7 | n = 3 | |
| B | 410.8 (302.4–558.1) µg/mL | >600 µg/mL | 239.6 (189.3–303.3) µg/mL |
| n = 2 | n = 2 | n = 2 | |
| C | 399.3 (349.6–456.1) µg/mL | 220.4 (107.0–454.2) µg/mL | 543.1 (503.6–585.7) µg/mL |
| n = 2 | n = 3 | n = 1 | |
| D | 420.0 (335.7–525.6) µg/mL | 395.0 (310.8–502.1) µg/mL | 591.4 (272.7–1283) µg/mL |
| n = 5 | n = 4 | n = 2 | |
| E | 401.3 (323.5–497.8) µg/mL | >600 µg/mL | 499.4 (447.9–556.9) µg/mL |
| n = 4 | n = 5 | n = 2 | |
| F | 461.6 (273.1–780.2) µg/mL | N.A. | >600 µg/mL |
| n = 3 | n = 2 | n = 1 | |
| G | 438.3 (309.0–621.6) µg/mL | 515.9 (435.1–611.6) µg/mL | N.A. |
| n = 2 | n = 3 | n = 2 | |
| H | 414.4 (362.9–473.3) µg/mL | N.A. | N.A. |
| n = 2 | n = 2 | n = 3 | |
| I | 539.3 (475.0–612.3) µg/mL | N.A. | 299.9 (260.4–345.4) µg/mL |
| n = 3 | n = 1 | n = 2 | |
| J | 369.1 (334.1–407.7) µg/mL | 388.0 (334.5–450.1) µg/mL | 356.1 (295.1–429.7) µg/mL |
| n = 2 | n = 2 | n = 3 | |
| K | 417.7 (392.1–445.1) µg/mL | 312.1 (140.6–692.7) µg/mL | 311.6 (260.4–373.0) µg/mL |
| n = 2 | n = 2 | n = 2 | |
| L | N.A. | N.A. | N.A. |
| n = 3 | n = 3 | n = 3 | |
| M | N.A. | N.A. | 290.6 (210.2–401.8) µg/mL |
| n = 2 | n = 1 | n = 3 | |
| N | 444.7 (418.3–472.8) µg/mL | 359.0 (328.1–392.9) µg/mL | N.A. |
| n = 3 | n = 2 | n = 3 | |
| O | 511.2 (427.6–611.2) µg/mL | 556.1 (512.2–603.7) µg/mL | 515.0 (407.2–651.5) µg/mL |
| n = 3 | n = 2 | n = 2 | |
| P | 403.1 (255.9–635.0) µg/mL | 401.5 (334.7–481.5) µg/mL | N.A. |
| n = 2 | n = 3 | n = 4 | |
| Q | 473.4 (418.8–535.1) µg/mL | N.A. | N.A. |
| n = 2 | n = 1 | n = 1 | |
| R | 464.4 (421.9–511.3) µg/mL | 464.1 (384.0–560.9) µg/mL | 501.2 (429.6–584.7) µg/mL |
| n = 2 | n = 2 | n = 2 | |
| S | 427.6 (411.7–444.2) µg/mL | 337.6 (243.8–467.4) µg/mL | N.A. |
| n = 2 | n = 1 | n = 3 | |
| T | 425.9 (395.2–458.9) µg/mL | 325.0 (238.9–442.1) µg/mL | 392.0 (357.1–430.4) µg/mL |
| n = 2 | n = 2 | n = 3 | |
| U | 406.7 (367.2–450.4) µg/mL | 348.1 (263.7–459.5) µg/mL | N.A. |
| n = 3 | n = 2 | n = 3 | |
| V | 400.4 (270.0–593.6) µg/mL | 398.0 (367.0–431.6) µg/mL | N.A. |
| n = 2 | n = 2 | n = 3 | |
| W | N.A. | 371.6 (291.9–472.9) µg/mL | N.A. |
| n = 2 | n = 2 | n = 3 | |
| X | 392.1 (310.7–494.8) µg/mL | 484.0 (387.2–604.9) µg/mL | 572.6 (424.8–771.8) µg/mL |
| n = 2 | n = 3 | n = 2 | |
| Y | 509.8 (377.6–688.3) µg/mL | 598.6 (557.5–642.8) µg/mL | N.A. |
| n = 2 | n = 6 | n = 4 | |
| Z | 411.8 (276.4–613.6) µg/mL | 453.2 (403.7–508.7) µg/mL | 512.9 (323.6–812.9) µg/mL |
| n = 2 | n = 2 | n = 2 | |
| AA | >600 µg/mL | 591.9 (509.2–688.0) µg/mL | 503.3 (470.9–537.9) µg/mL |
| n = 2 | n = 3 | n = 1 | |
| AB | N.A. | N.A. | 254.9 (243.3–267.1) µg/mL |
| n = 2 | n = 1 | n = 2 | |
| AC | 441.0 (404.4–481.0) µg/mL | N.A. | 297.7 (277.6–319.2) µg/mL |
| n = 3 | n = 2 | n = 3 | |
| AD | 345.2 (297.8–400.1) µg/mL | N.A. | N.A. |
| n = 2 | n = 1 | n = 1 | |
| AE | >600 µg/mL | 331.4 (266.9–411.5) µg/mL | >600 µg/mL |
| n = 1 | n = 1 | n = 1 | |
| AF | 405.6 (355.5–462.7) µg/mL | N.A. | N.A. |
| n = 4 | n = 1 | n = 1 | |
| CisPlatin | 10.7 (7.1–17.9) µg/mL | 4.1 (2.6–7.2) µg/mL | 45.0 (25.9–71.7) µg/mL |
| n = 3 | n = 3 | n = 3 |
| Principal Component | LoVo | 2008 | MCF7 |
|---|---|---|---|
| PC1 | 30.4% | 31.1% | 33.5% |
| PC2 | 12.8% | 11.4% | 11.4% |
| PC3 | 8.8% | 10.6% | 8.9% |
| PC4 | 8.0% | 8.9% | 8.9% |
| Total per cell line | 60.0% | 62.0% | 62.7% |
| Compound | LoVo | 2008 | MCF7 | Reported Cytotoxic Activity | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| I1 | L1 | L2 | L3 | L4 | T1 | S | I1a | L1a | L2a | L3a | T2 | I1b | L1b | L2b | L3b | L4a | T3 | V1 | S | ||
| spathulenol | [42] | ||||||||||||||||||||
| endoborneol | |||||||||||||||||||||
| p-cymene | [15] | ||||||||||||||||||||
| bornyl acetate | |||||||||||||||||||||
| δ-carene | |||||||||||||||||||||
| α-pinene | |||||||||||||||||||||
| β-pinene | |||||||||||||||||||||
| α-terpineol | [17] | ||||||||||||||||||||
| 1-4 terpineol | [15,46] | ||||||||||||||||||||
| cterpinene | |||||||||||||||||||||
| γ-terpinene | [15,17] | ||||||||||||||||||||
| β-phellandrene | |||||||||||||||||||||
| γ-eudesmol | |||||||||||||||||||||
| sabinene | |||||||||||||||||||||
| 2-nonanone | |||||||||||||||||||||
| α-phellandrene | |||||||||||||||||||||
| 2-undecanone | |||||||||||||||||||||
| β-eudesmol | |||||||||||||||||||||
| α-copaene | [17] | ||||||||||||||||||||
| α-cadinol | |||||||||||||||||||||
| T-cadinol | |||||||||||||||||||||
| cadina-1,4-diene | |||||||||||||||||||||
| β-elemene | [8] | ||||||||||||||||||||
| α-amorphene | |||||||||||||||||||||
| germacrene A | |||||||||||||||||||||
| α-muurolene | |||||||||||||||||||||
| α-elemene | |||||||||||||||||||||
| alloaromandrene | |||||||||||||||||||||
| Octanoid acid | |||||||||||||||||||||
| germacrene D | |||||||||||||||||||||
| α-cubebene | |||||||||||||||||||||
| β-cubebene | |||||||||||||||||||||
| α-ylangene | |||||||||||||||||||||
| 1-phellandrene | |||||||||||||||||||||
| BHT 1 | |||||||||||||||||||||
| α-cadinene | |||||||||||||||||||||
| tricyclene | |||||||||||||||||||||
| α-humulene | [18] | ||||||||||||||||||||
| trans β-ocimene | |||||||||||||||||||||
| cis ocimene | |||||||||||||||||||||
| d-limonene | [8,15] | ||||||||||||||||||||
| β-caryophyllene | [8] | ||||||||||||||||||||
| BMTTHU 2 | |||||||||||||||||||||
| HMHMOH 3 | |||||||||||||||||||||
| CPA 4 | |||||||||||||||||||||
| camphene | |||||||||||||||||||||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Buriani, A.; Fortinguerra, S.; Sorrenti, V.; Dall’Acqua, S.; Innocenti, G.; Montopoli, M.; Gabbia, D.; Carrara, M. Human Adenocarcinoma Cell Line Sensitivity to Essential Oil Phytocomplexes from Pistacia Species: a Multivariate Approach. Molecules 2017, 22, 1336. https://doi.org/10.3390/molecules22081336
Buriani A, Fortinguerra S, Sorrenti V, Dall’Acqua S, Innocenti G, Montopoli M, Gabbia D, Carrara M. Human Adenocarcinoma Cell Line Sensitivity to Essential Oil Phytocomplexes from Pistacia Species: a Multivariate Approach. Molecules. 2017; 22(8):1336. https://doi.org/10.3390/molecules22081336
Chicago/Turabian StyleBuriani, Alessandro, Stefano Fortinguerra, Vincenzo Sorrenti, Stefano Dall’Acqua, Gabbriella Innocenti, Monica Montopoli, Daniela Gabbia, and Maria Carrara. 2017. "Human Adenocarcinoma Cell Line Sensitivity to Essential Oil Phytocomplexes from Pistacia Species: a Multivariate Approach" Molecules 22, no. 8: 1336. https://doi.org/10.3390/molecules22081336
APA StyleBuriani, A., Fortinguerra, S., Sorrenti, V., Dall’Acqua, S., Innocenti, G., Montopoli, M., Gabbia, D., & Carrara, M. (2017). Human Adenocarcinoma Cell Line Sensitivity to Essential Oil Phytocomplexes from Pistacia Species: a Multivariate Approach. Molecules, 22(8), 1336. https://doi.org/10.3390/molecules22081336