Phenolic Compounds Contained in Little-known Wild Fruits as Antiadhesive Agents Against the Beverage-Spoiling Bacteria Asaia spp.
Abstract
1. Introduction
2. Results and Discussion
2.1. Carbohydrate Content
2.2. Antioxidant Capacity and Total Phenolic Content
2.3. Phenolic Profiles
2.4. Bacterial Adhesion
3. Materials and Methods
3.1. Plant Material
3.2. Bacterial Cultures
3.3. Carriers
3.4. Chemical Analysis of Juices
3.4.1. Carbohydrates
3.4.2. Total Phenolic Content (TPC)
3.4.3. Total Antioxidant Capacity (DPPH)
3.4.4. Ferric-Reducing Antioxidant Power (FRAP)
3.4.5. Phenolic Compounds
3.5. Microbiological Analysis
3.5.1. Culture Media and Growth Conditions
3.5.2. Bacterial Adhesion
3.5.3. Fluorescent Microscopy
3.6. Statistics
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lia, R.; Kudab, T.; Yanoa, T. Effect of food residues on efficiency of surfactant disinfectants against food related pathogens adhered on polystyrene and ceramic surfaces. LWT Food Sci. Technol. 2014, 57, 200–206. [Google Scholar] [CrossRef]
- Kręgiel, D. Attachment of Asaia lannensis to materials commonly used in beverage industry. Food Control 2013, 32, 537–542. [Google Scholar] [CrossRef]
- Antolak, H.; Czyżowska, A.; Kręgiel, D. Black currant (Ribes nigrum L.) and bilberry (Vaccinium myrtillus L.) fruit juices inhibit adhesion of Asaia spp. BioMed Res. Int. 2016, 2016, 3671306. [Google Scholar] [CrossRef] [PubMed]
- Rushton, L.; Sass, A.; Baldwin, A.; Dowson, C.G.; Donoghue, D.; Mahenthiralingam, E. Key role for efflux in the preservative susceptibility and adaptive resistance of Burkholderia cepacia complex bacteria. Antimicrob. Agents Chemother. 2013, 57, 2972–2980. [Google Scholar] [CrossRef] [PubMed]
- Charlebois, A.; Jacques, M.; Boulianne, M.; Archambault, M. Tolerance of Clostridium perfringens biofilms to disinfectants commonly used in the food industry. Food Microbiol. 2017, 62, 32–38. [Google Scholar] [CrossRef] [PubMed]
- Antolak, H.; Czyżowska, A.; Kręgiel, D. Antibacterial and antiadhesive activities of extracts from edible plants against soft drink spoilage by Asaia spp. J. Food Prot. 2017, 80, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Borlinghaus, J.; Albrecht, F.; Gruhlke, M.C.H.; Nwachukwu, I.D.; Slusarenko, A.J. Allicin: Chemistry and biological properties. Molecules 2014, 9, 12591–12618. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs from 1981 to 2014. J. Nat. Prod. 2016, 79, 629–661. [Google Scholar] [CrossRef] [PubMed]
- Atanasov, A.G.; Waltenberger, B.; Pferschy-Wenzig, E.M.; Linder, T.; Wawrosch, C.; Uhrin, P.; Temml, V.; Wang, L.; Schwaiger, S.; Heiss, E.H.; et al. Discovery and resupply of pharmacologically active plant-derived natural products: A review. Biotechnol. Adv. 2015, 33, 1582–1614. [Google Scholar] [CrossRef] [PubMed]
- Lipińska, L.; Klewicka, E.; Sójka, M. Structure, occurrence and biological activity of ellagitannins: A general review. Acta Sci. Pol. Technol. Aliment. 2014, 13, 289–299. [Google Scholar] [CrossRef] [PubMed]
- Nohynek, L.J.; Alakomi, H.L.; Kähkönen, M.P.; Heinonen, M.; Helander, I.M.; Oksman-Caldentey, K.M.; Puupponen-Pimiä, R.H. Berry phenolics: Antimicrobial properties and mechanisms of action against severe human pathogens. Nutr. Cancer 2006, 54, 18–32. [Google Scholar] [CrossRef] [PubMed]
- Howell, A.; Souza, D.; Roller, M.; Fromentin, E. Comparison of the anti-adhesion activity of three different cranberry extracts on uropathogenic P-fimbriated Escherichia coli: A randomized, double-blind, placebo controlled, ex vivo, acute study. Nat. Prod. Commun. 2015, 10, 1215–1218. [Google Scholar] [PubMed]
- Gasparrini, M.; Forbes-Hernandez, T.Y.; Giampieri, F.; Afrin, S.; Alvarez-Suarez, J.M.; Mazzoni, L.; Mezzetti, B.; Quiles, J.L.; Battino, M. Anti-inflammatory effect of strawberry extract against LPS-induced stress in RAW 264.7 macrophages. Food Chem. Toxicol. 2017, 102, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Afrin, S.; Gasparrini, M.; Forbes-Hernandez, T.Y.; Reboredo-Rodriguez, P.; Mezzetti, B.; Varela-López, A.; Giampieri, F.; Battino, M. Promising health benefits of the strawberry: A focus on clinical studies. J. Agric. Food Chem. 2016, 64, 4435–4449. [Google Scholar] [CrossRef] [PubMed]
- Veberic, R.; Slatnar, A.; Bizjak, J.; Stampar, F.; Mikulic-Petkovsek, M. Anthocyanin composition of different wild and cultivated berry species. LWT Food Sci. Technol. 2015, 60, 509–517. [Google Scholar] [CrossRef]
- Szajdek, A.; Borowska, E.J. Bioactive compounds and health-promoting properties of berry fruits: A review. Plant. Foods Hum. Nutr. 2008, 63, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Forbes-Hernandez, T.Y.; Gasparrini, M.; Afrin, S.; Bompadre, S.; Mezzetti, B.; Quiles, J.L.; Giampieri, F.; Battino, M. The healthy effects of strawberry polyphenols: Which strategy behind antioxidant capacity? Crit. Rev. Food Sci. Nutr. 2016, 56, 46–59. [Google Scholar] [CrossRef] [PubMed]
- Sidor, A.; Gramza-Michałowska, A. Advanced research on the antioxidant and health benefit of elderberry (Sambucus nigra) in food—A review. J. Funct. Foods 2015, 18, 941–958. [Google Scholar] [CrossRef]
- Drkenda, P.; Spahic, A.; Begic-Akagic, A.; Gasi, F.; Vranac, A.; Hudina, M.; Blanke, M. Pomological characteristics of some autochthonous genotypes of cornelian cherry (Cornus mas L.) in Bosnia and Herzegovina. Erwerbs-Obstbau 2014, 56, 59–66. [Google Scholar] [CrossRef]
- Tolmacheva, A.A.; Rogozhin, E.A.; Deryabin, D.G. Antibacterial and quorum sensing regulatory activities of some traditional Eastern-European medicinal plants. Acta Pharm. 2014, 64, 173–186. [Google Scholar] [CrossRef] [PubMed]
- Veberic, R.; Jakopic, J.; Stampar, F.; Schmitzer, V. European elderberry (Sambucus nigra L.) rich in sugars, organic acids, anthocyanins and selected polyphenols. Food Chem. 2009, 114, 511–515. [Google Scholar] [CrossRef]
- Visti, A.; Viljakainen, S.; Laakso, S. Preparation of fermentable lingonberry juice through removal of benzoic acid by Saccharomyces cerevisiae yeast. Food Res. Int. 2003, 36, 597–602. [Google Scholar] [CrossRef]
- Cheng, G.; He, Y.-N.; Yue, T.-X.; Wang, J.; Zhang, Z.-W. Effects of climatic conditions and soil properties on cabernet sauvignon berry growth and anthocyanin profiles. Molecules 2014, 19, 13683–13703. [Google Scholar] [CrossRef] [PubMed]
- Prior, R.L.; Sintara, M.; Chang, T. Multi-radical (ORACMR5) antioxidant capacity of selected berries and effects of food processing. J. Berry Res. 2016, 6, 159–173. [Google Scholar] [CrossRef]
- Viskelis, P.; Rubinskienė, M.; Bobinaitė, R.; Dambrauskienė, E. Bioactive compounds and antioxidant activity of small fruits in Lithuania. J. Food Agric. Environ. 2010, 8, 259–263. [Google Scholar]
- Jakobek, L.; Šeruga, M.; Medvidović-Kosamović, M.; Novak, I. Anthocyanin content and antioxidant activity of various red fruit juices. Deutsch. Lebensm. Rundshau 2007, 103, 58–64. [Google Scholar]
- Ersoy, N.; Bagci, Y.; Gok, V. Antioxidant properties of 12 cornelian cherry fruit types (Cornus mas L.) selected from Turkey. Sci. Res. Essays 2011, 6, 98–102. [Google Scholar]
- Georgieva, M.; Badjakov, I.; Dincheva, I.; Yancheva, S.; Kondakova, V. In vitro propagation of wild Bulgarian small berry fruits (bilberry, lingonberry, raspberry and strawberry). Bulg. J. Agric. Sci. 2016, 22, 46–51. [Google Scholar]
- Tarko, T.; Duda-Chodak, A.; Semik, D.; Nycz, M. The use of fruit extracts for production of beverages with high antioxidative activity. Potravin. Slovak J. Food Sci. 2015, 9, 280–283. [Google Scholar] [CrossRef]
- Šamec, D.; Piljac-Žegarac, J. Postharvest stability of antioxidant compounds in hawthorn and cornelian cherries at room and refrigerator temperatures—Comparison with blackberries, white and red grapes. Sci. Hort. 2011, 131, 15–21. [Google Scholar] [CrossRef]
- Moldovan, B.; Filip, A.; Clichici, S.; Suharoschi, R.; Bolfa, P.; David, L. Antioxidant activity of Cornelian cherry (Cornus mas L.) and the in vivo evaluation of its anti-inflammatory effects. J. Funct. Foods 2016, 26, 77–87. [Google Scholar] [CrossRef]
- Nile, S.H.; Park, S.W. Edible berries: Bioactive components and their effect on human health. Nutrition 2014, 30, 134–144. [Google Scholar] [CrossRef] [PubMed]
- Silva, P.; Ferreira, S.; Nunes, F.M. Elderberry (Sambucus nigra L.) by-products a source of anthocyanins and antioxidant polyphenols. Ind. Crops Prod. 2017, 95, 227–234. [Google Scholar] [CrossRef]
- Kucharska, A.Z.; Sokół-Łętowska, A.; Oszmiański, J.; Piórecki, N.; Fecka, I. Iridoids, phenolic compounds and antioxidant activity of edible honeysuckle berries (Lonicera caerulea var. kamtschatica Sevast.). Molecules 2017, 22. [Google Scholar] [CrossRef] [PubMed]
- Hosseini-Behesht, E.; Lund, S.T.; Kitts, D.D. Characterization of antioxidant capacity from fruits with distinct anthocyanin biosynthetic pathways. J. Nutr. Food Sci. 2012, 2. [Google Scholar] [CrossRef]
- Deng, S.; West, B.J.; Jensen, C.J. UPLC-TOF-MS characterization and identification of bioactive iridoids in Cornus mas fruit. J. Anal. Methods Chem. 2013, Article ID 710972. [Google Scholar] [CrossRef] [PubMed]
- Radovanović, B.C.; Anđelković, A.S.M.; Radovanović, A.B.; Anđelković, M.Z. Antioxidant and antimicrobial activity of polyphenol extracts from wild berry fruits grown in southeast Serbia. Trop. J. Pharm. Res. 2013, 12, 813–819. [Google Scholar] [CrossRef]
- Häkkinen, S.; Heinonen, M.; Kärenlampi, S.; Mykkänen, H.; Ruuskanen, J.; Törrönen, R. Screening of selected flavonoids and phenolic acids in 19 berries. Food Res. Int. 1999, 32, 345–353. [Google Scholar] [CrossRef]
- Mattila, P.; Hellström, J.; Törrönen, R. Phenolic acids in berries, fruits, and beverages. J. Agric. Food Chem. 2006, 54, 7193–7199. [Google Scholar] [CrossRef] [PubMed]
- Tarko, T.; Duda-Chodak, A.; Wajda, Ł.; Satora, P.; Sroka, P.; Emik-Szczurak, D. Application of principal component analysis for optimization of polyphenol extraction from alternative plant sources. J. Food Nutr. Res. 2017, 56, 61–72. [Google Scholar]
- Lee, J.; Finn, E.F. Anthocyanins and other polyphenolics in American elderberry (Sambucus canadensis) and European elderberry (S. nigra) cultivars. J. Sci. Food Agric. 2007, 8, 2665–2675. [Google Scholar] [CrossRef] [PubMed]
- Mikulic-Petkovsek, M.; Ivancic, A.; Todorovic, B.; Veberic, R.; Stampar, F. Fruit phenolic composition of different elderberry species and hybrids. J. Food Sci. 2015, 80, 2180–2190. [Google Scholar] [CrossRef] [PubMed]
- Milenković-Andjelković, A.S.; Andjelković, M.Z.; Radovanović, A.N.; Radovanović, B.C.; Nikolić, V. Phenol composition, DPPH radical scavenging and antimicrobial activity of Cornelian cherry (Cornus mas) fruit and leaf extracts. Hem. Ind. 2015, 69, 331–337. [Google Scholar] [CrossRef]
- He, Q.; Lv, Y.; Yao, K. Effects of tea polyphenols on the activities of α-amylase, pepsin, trypsin and lipase. Food Chem. 2007, 101, 1178–1182. [Google Scholar] [CrossRef]
- Riihinen, K.R.; Ou, Z.M.; Gödecke, T.; Lankin, D.C.; Pauli, G.F.; Wu, C.D. The antibiofilm activity of lingonberry flavonoids against oral pathogens is a case connected to residual complexity. Fitoterapia 2014, 97, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Krawitz, C.; Mraheil, M.A.; Stein, M.; Imirzalioglu, C.; Domann, E.; Pleschka, S.; Hain, T. Inhibitory activity of a standardized elderberry liquid extract against clinically-relevant human respiratory bacterial pathogens and influenza A and B viruses. BMC Complement. Altern. Med. 2011, 11, 16. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, A.; Yasmin, T.; Bagchi, D.; Stohs, S.J. Inhibition of Helicobacter pylori in vitro by various berry extracts, with enhanced susceptibility to clarithromycin. Mol. Cell. Biochem. 2004, 265, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Kyriakopoulos, A.M.; Dinda, B. Cornus mas (Linnaeus) novel devised medicinal preparations: bactericidal effect against Staphylococcus aureus and Pseudomonas aeruginosa. Molecules 2015, 20, 11202–11218. [Google Scholar] [CrossRef] [PubMed]
- Ho, K.Y.; Tsai, C.C.; Huang, J.S.; Chen, C.P.; Lin, T.C.; Lin, C.C. Antimicrobial activity of tannin components from Vaccinium vitis-idaea L. J. Pharm Pharmacol. 2001, 53, 187–191. [Google Scholar] [CrossRef] [PubMed]
- Kręgiel, D.; Otlewska, A.; Antolak, H. Attachment of Asaia bogorensis originating in fruit-flavored water to packaging materials. BioMed Res. Int. 2014, Article ID 514190. [Google Scholar] [CrossRef]
- Berłowska, J.; Cieciura, W.; Borowski, S.; Dudkiewicz, M.; Binczarski, M.; Witonska, I.; Otlewska, A.; Kregiel, D. Simultaneous saccharification and fermentation of sugar beet pulp with mixed bacterial cultures for lactic acid and propylene glycol production. Molecules 2016, 21, 1380. [Google Scholar] [CrossRef] [PubMed]
- Mišan, A.C.; Sakač, M.; Medić, Đ.; Tadić, V.; Marković, G.; Gyura, J.; Pagano, E.; Izzo, A.A.; Borrelli, F.; Šarić, B.; et al. Antioxidant and physicochemical properties of hydrogen peroxide-treated sugar beet dietary fibre. Phytother. Res. 2016, 30, 855–860. [Google Scholar] [CrossRef] [PubMed]
- Oyaizu, M. Studies on products of browning reaction: antioxidative activities of products of browning reaction prepared from glucosamine. Jpn. J. Nutr. 1986, 44, 307–315. [Google Scholar] [CrossRef]
- Mišan, A.C.; Mimica-Dukić, N.M.; Mandić, A.I.; Sakač, M.B.; Milovanović, I.J.; Sedej, I.J. Development of a rapid resolution HPLC method for the separation and determination of 17 phenolic compounds in crude plant extracts. Cent. Eur. J. Chem. 2011, 9, 133–142. [Google Scholar] [CrossRef]
- Brodowska, A.J.; Śmigielski, K.; Nowak, A.; Czyżowska, A.; Otlewska, A. The impact of ozone treatment in dynamic bed parameters on changes in biologically active substances of juniper berries. PLoS ONE 2015, 10, e0144855. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Bacterial strains are available from the authors. |

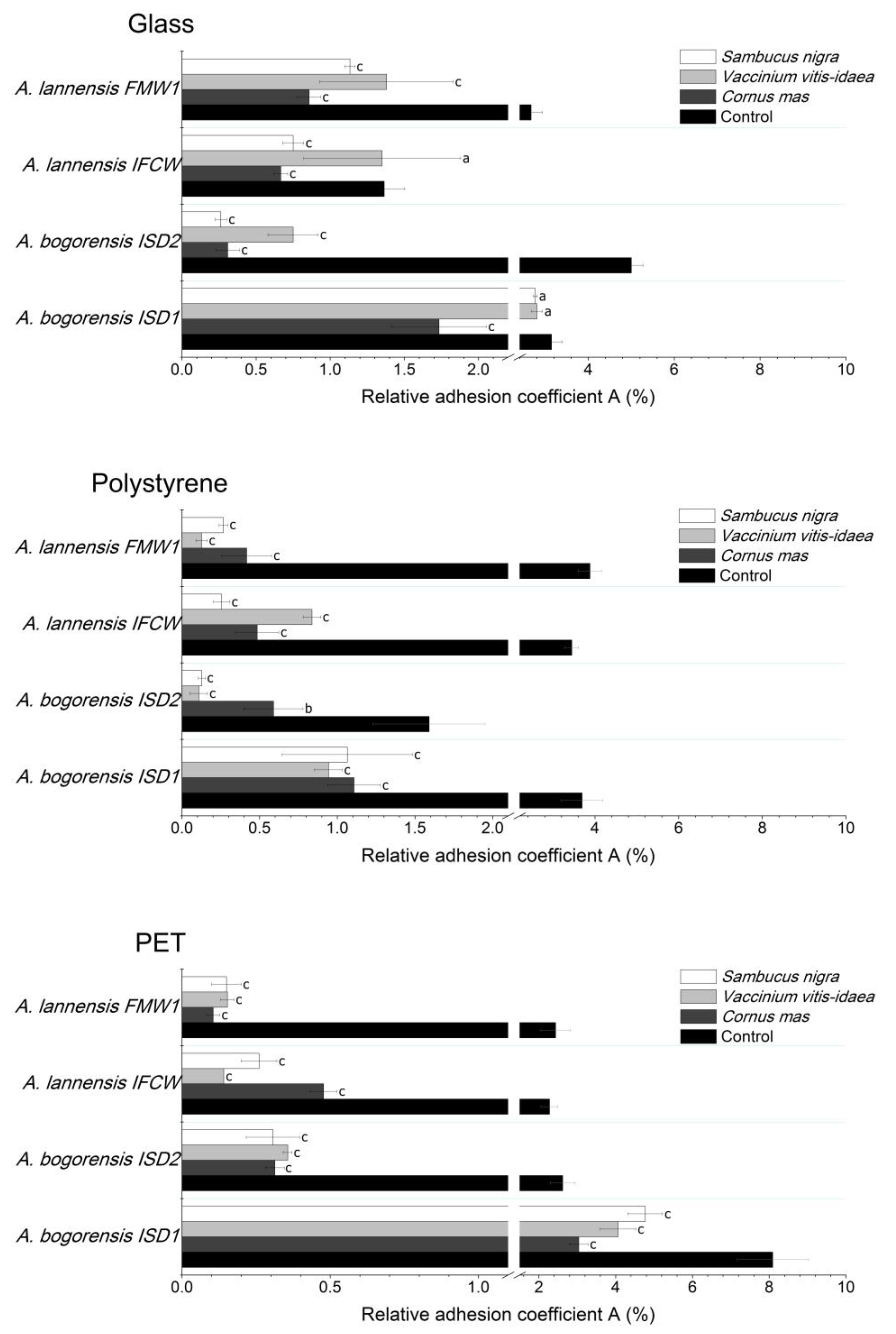
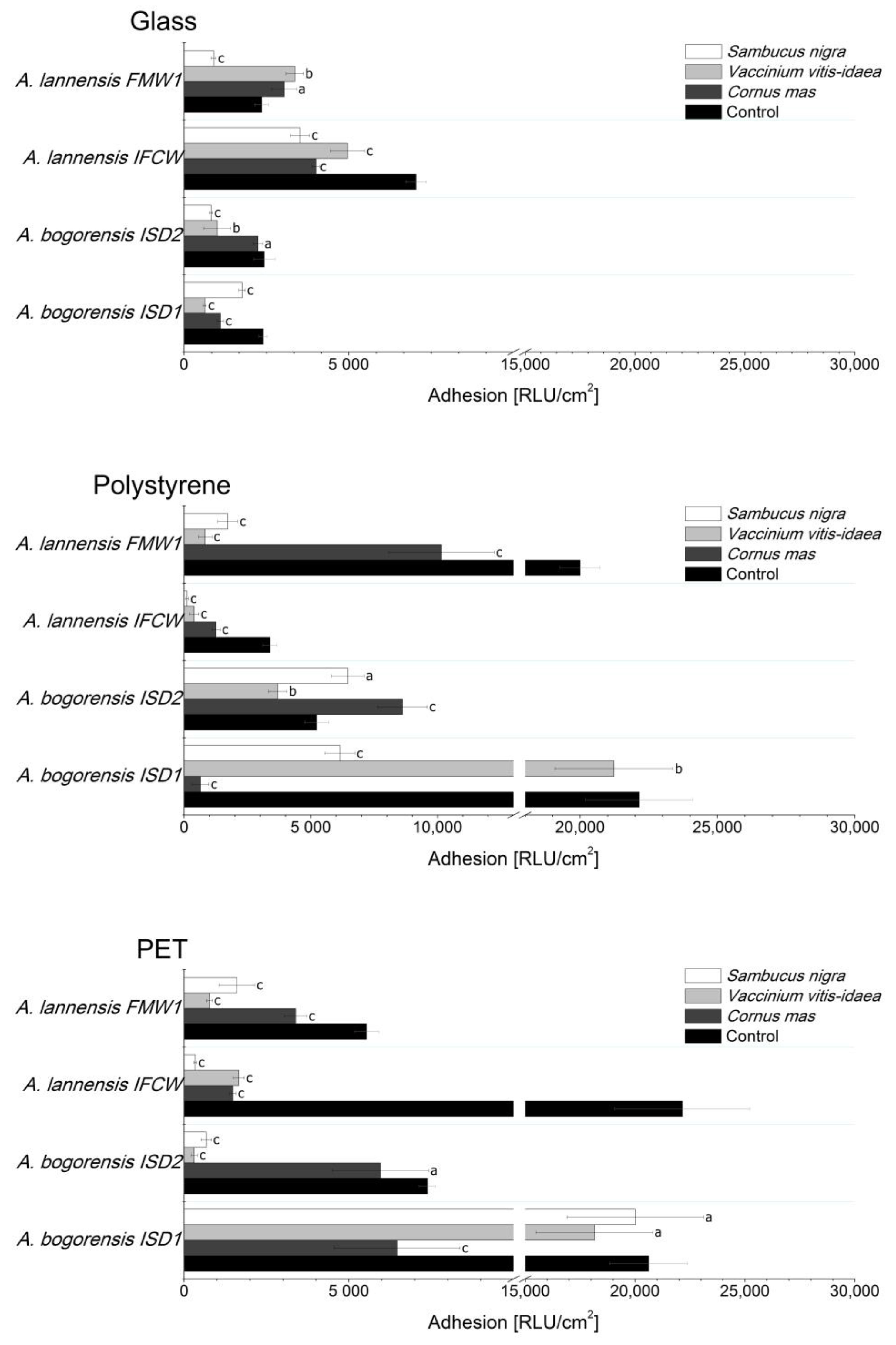
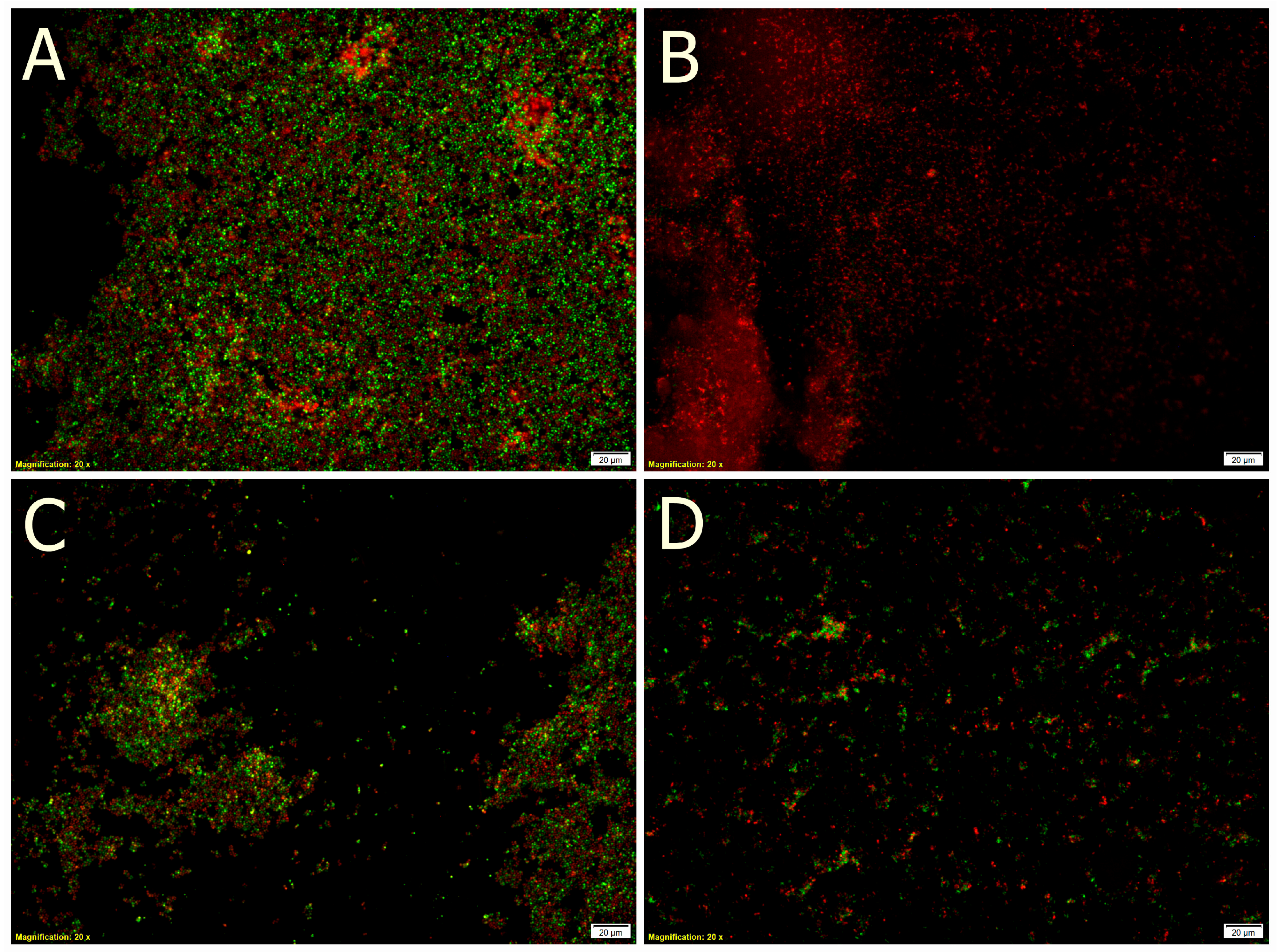
| Berry Juice | Sugar Content (g/100 mL) | TPC (mg GAE/mL) | Antioxidant Activity | ||
|---|---|---|---|---|---|
| Fructose | Glucose | DPPH IC50 (g/mL) | FRAP IC50 (g/mL) | ||
| Cornelian cherry (Cornus mas) | 5.56 ± 0.061 | 2.97 ± 0.046 | 2.33 ± 0.013 | 0.045 ± 0.001 | 0.042 ± 0.001 |
| Lingonberry (Vaccinium vitis-idaea) | 3.89 ± 0.043 c | 4.54 ± 0.071 c | 4.87 ± 0.044 c | 0.054 ± 0.002 c | 0.030 ± 0.002 c |
| Elderberry (Sambucus nigra) | 3.29 ± 0.015 c | 3.19 ± 0.022 b | 8.02 ± 0.027 c | 0.072 ± 0.001 c | 0.021 ± 0.001 c |
| Proposed Molecule | Concentration (µg/mL) | ||
|---|---|---|---|
| Cornus mas | Vaccinium vitis-idaea | Sambucus nigra | |
| Caffeic acid | nd | nd | 2.603 ± 0.313 d |
| Cinnamic acid | 0.143 ± 0.011 | 0.191 ± 0.014 b | nd |
| Gallic acid | 2.025 ± 0.314 | 0.071 ± 0.009 c | 0.286 ± 0.082 c |
| Protocatechuic acid | 0.379 ± 0.271 | 0.497 ± 0.087 a | 0.550 ± 0.057 a |
| p-coumaric acid | 0.108 ± 0.048 | 0.179 ± 0.052 a | nd |
| Rosmarinic acid | 0.128 ± 0.062 | 0.128 ± 0.037 a | 0.128 ± 0.019 a |
| 4-hydroxybenzoic acid | nd | 0.150 ± 0.074 a | 0.265 ± 0.096 a |
| Catechin | nd | 0.662 ± 0.121 a | 0.918 ± 0.107 a |
| Epicatechin | nd | 0.304 ± 0.082 d | nd |
| Rutin | nd | nd | 1.321 ± 0.307 d |
| Delphinidin-3-glucoside | nd | nd | 2.057 ± 0.371 d |
| Cyanidin-3-sambubioside-5-glucoside | nd | nd | 2.260 ± 0.219 d |
| Cyanidin-3-glucoside | 0.280 ± 0.039 | 0.605 ± 0.054 c | 3.738 ± 0.147 c |
| Cyanidin-3-sambubioside | nd | nd | 3.143 ± 0.262 d |
| Cyanidin-3-robinobioside | 0.321 ± 0.041 | nd | nd |
| Petunidin-3-galactoside | nd | 0.320 ± 0.057 d | nd |
| Petunidin-3-glucoside | nd | 0.528 ± 0.052 d | nd |
| Pelargonidin-3-glucoside | 0.380 ± 0.052 | 0.359 ± 0.063 a | nd |
| Pelargonidin-3-rutinoside | nd | 0.344 ± 0.074 d | nd |
| Pelargonidin-3-robinobioside | 0.302 ± 0.022 | nd | nd |
| Aglycone Class | Proposed Molecule | λmax (nm) | [M − H]− | MS2 | Cornus mas | Vaccinium vitis-idaea | Sambucus nigra |
|---|---|---|---|---|---|---|---|
| Phenolic acids | Caffeic | 279 | 179 | 135 | − | − | + |
| Caffeic acid derivative | 234, 279 | 341 | 177, 195 | − | − | + | |
| Caffeoyl hexoside | 231, 282 | 341 | 179 | − | + | − | |
| Chlorogenic | 295, 323 | 353 | 191 | − | + | + | |
| Neochlorogenic | 323 | 353 | 179, 191 | − | + | + | |
| Ferulic | 237, 323 | 193 | 149, 173 | + | − | − | |
| Gallic | 237, 276 | 205 | 111, 125, 173 | + | + | + | |
| Quinic | 235, 284 | 191 | 111, 173 | − | + | − | |
| Flavonols | Kaempferol | 239, 279, 325 | 285 | 213, 257 | − | − | + |
| Kaempferol-3-glucoside | 263, 344 | 447 | 255, 284, 327 | − | + | + | |
| Kaempferol-3-rutinoside | 265, 342 | 593 | 285 | − | − | + | |
| Quercetin | 235, 279, 341 | 301 | 229, 255 | + | + | + | |
| Quercetin-3-glucoside | 257, 353 | 463 | 301 | + | + | + | |
| Quercetin-3-rhamnoside | 257, 349 | 447 | 301 | − | + | − | |
| Quercetin-3-O-rutinoside (Rutin) | 256, 350 | 609 | 301 | - | - | + | |
| Myricetin-3-galactoside | 238, 278 | 491 | 317 | + | − | − | |
| Anthocyanins | Delphinidin-3-glucoside | 276 | 463 | 301 | − | + | + |
| Cyanidin-3-glucoside | 282 | 449 | 287 | + | + | + | |
| Petunidin-3-glucoside | 236, 269 | 479 | 317 | + | + | − | |
| Peonidin-3-galactoside | 235, 280 | 465 | 301 | − | − | + | |
| Pelargonidin-3-robinobioside | 271 | 577 | 431, 269 | + | − | − | |
| Cyanidin-3-samburoside | 279 | 581 | 449, 287 | − | − | + | |
| Cyanidin-3-robinobioside | 280 | 593 | 447, 285 | + | − | − | |
| Flavanols | Catechin | 233, 280 | 289 | 205, 245 | − | + | − |
| Epicatechin | 231, 281 | 289 | 205, 245 | − | + | − | |
| Proantho-cyanidins | Procyanidin dimer | 281 | 575 | 425, 407 | - | + | − |
| Procyanidin trimer | 277 | 863 | 575 | − | + | − | |
| Others | Coumaroyl iridoid | 238, 282 | 366 | 309 | − | + | − |
| Cornuside | 242, 274 | 541 | 169, 347 | + | − | − | |
| Loganic | 239, 279 | 375 | 213, 169 | + | − | − | |
| Loganic acid | 239, 279 | 375 | 213, 169 | + | − | − |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Antolak, H.; Czyzowska, A.; Sakač, M.; Mišan, A.; Đuragić, O.; Kregiel, D. Phenolic Compounds Contained in Little-known Wild Fruits as Antiadhesive Agents Against the Beverage-Spoiling Bacteria Asaia spp. Molecules 2017, 22, 1256. https://doi.org/10.3390/molecules22081256
Antolak H, Czyzowska A, Sakač M, Mišan A, Đuragić O, Kregiel D. Phenolic Compounds Contained in Little-known Wild Fruits as Antiadhesive Agents Against the Beverage-Spoiling Bacteria Asaia spp. Molecules. 2017; 22(8):1256. https://doi.org/10.3390/molecules22081256
Chicago/Turabian StyleAntolak, Hubert, Agata Czyzowska, Marijana Sakač, Aleksandra Mišan, Olivera Đuragić, and Dorota Kregiel. 2017. "Phenolic Compounds Contained in Little-known Wild Fruits as Antiadhesive Agents Against the Beverage-Spoiling Bacteria Asaia spp." Molecules 22, no. 8: 1256. https://doi.org/10.3390/molecules22081256
APA StyleAntolak, H., Czyzowska, A., Sakač, M., Mišan, A., Đuragić, O., & Kregiel, D. (2017). Phenolic Compounds Contained in Little-known Wild Fruits as Antiadhesive Agents Against the Beverage-Spoiling Bacteria Asaia spp. Molecules, 22(8), 1256. https://doi.org/10.3390/molecules22081256









