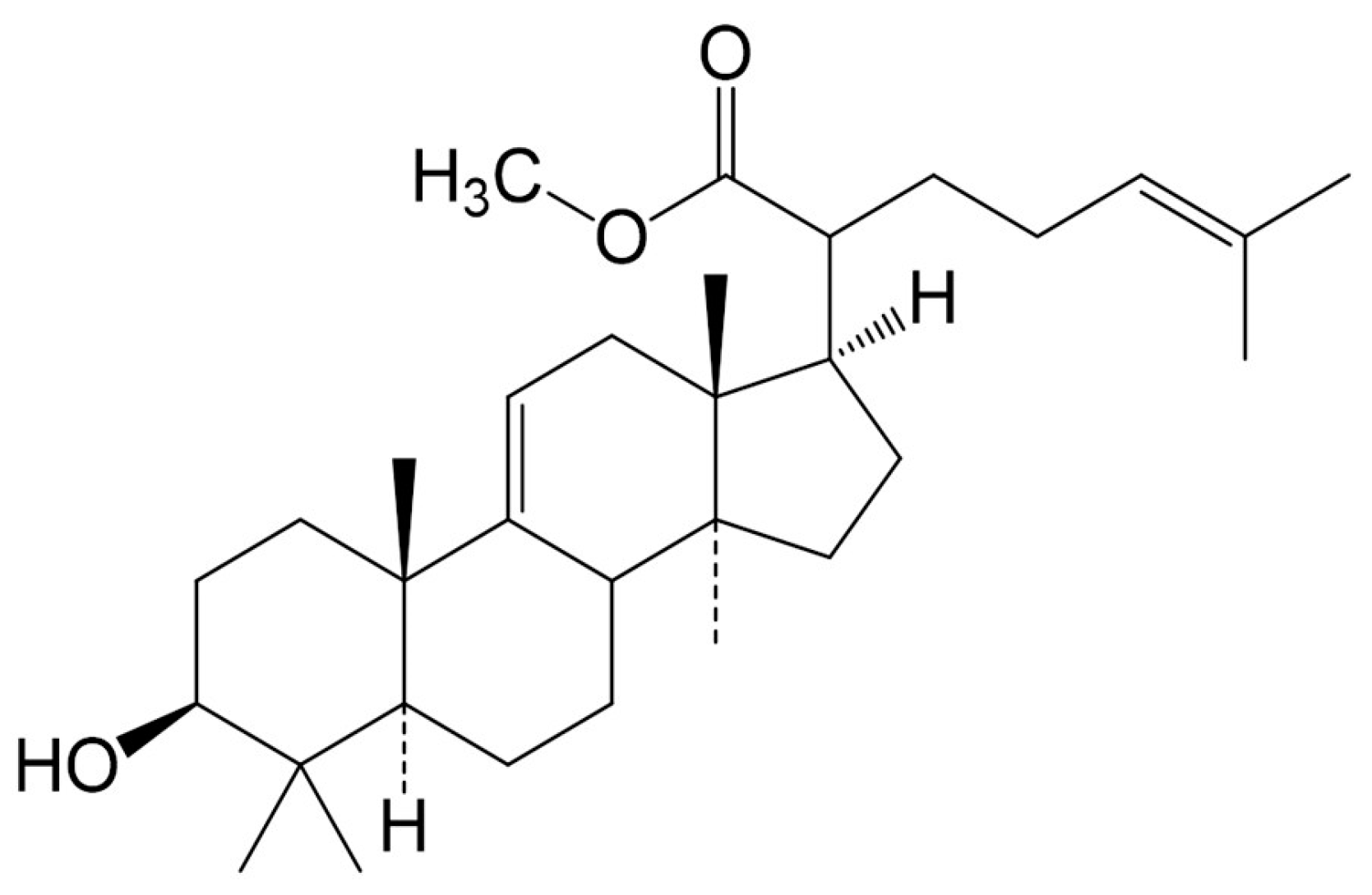
A Lanosteryl Triterpene from Protorhus longifolia Improves Glucose Tolerance and Pancreatic Beta Cell Ultrastructure in Type 2 Diabetic Rats
Abstract
:1. Introduction
2. Results
2.1. RA-3 Displayed Hypoglycemic Effect in the Type 2 Diabetic Rats
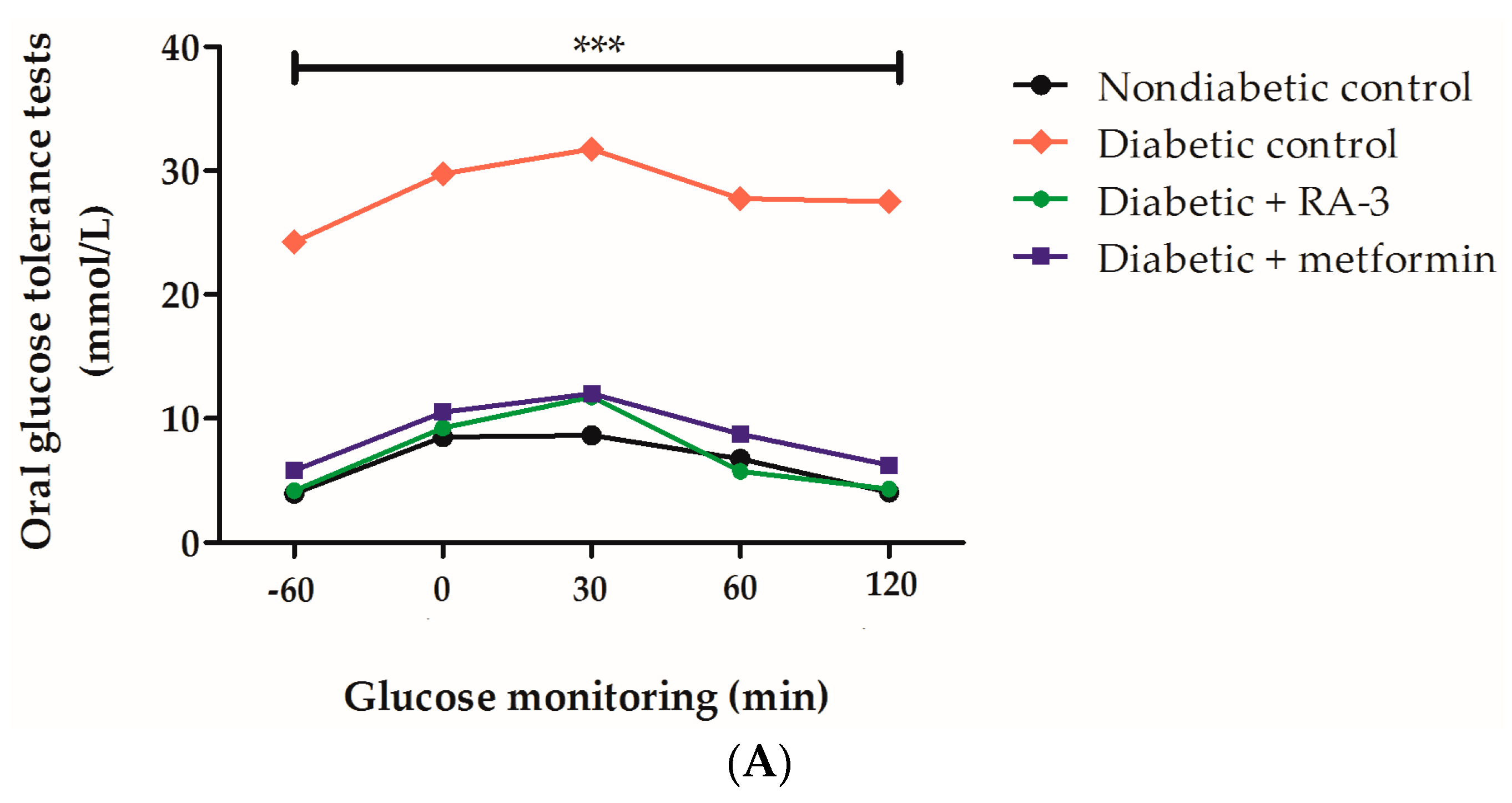
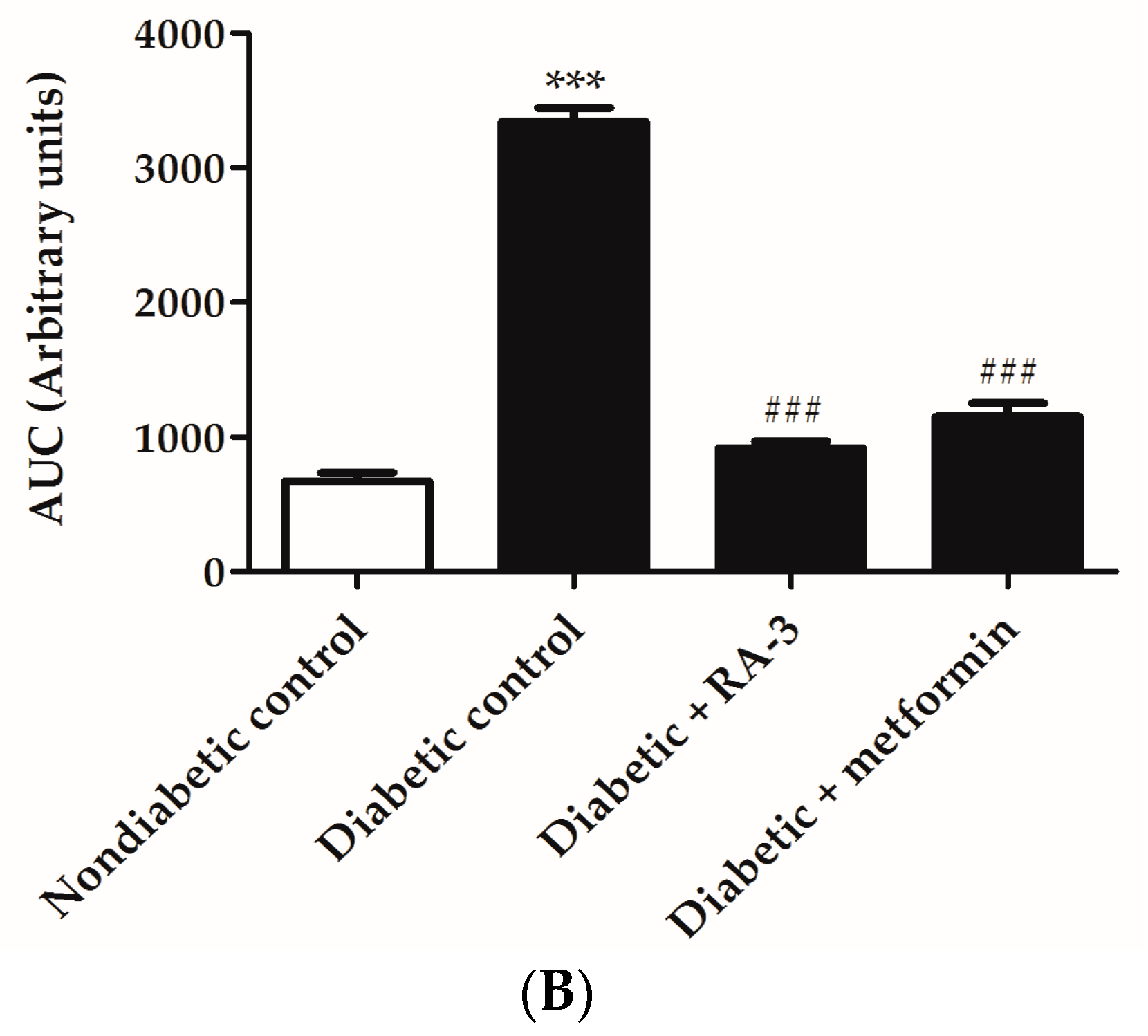
2.2. RA-3 Improved Glucose Tolerance in Type 2 Diabetic Rats
2.3. RA-3 Prevented Lipid Peroxidation through Enhancement of Endogenous Antioxidant Status in the Type 2 Diabetic Rats
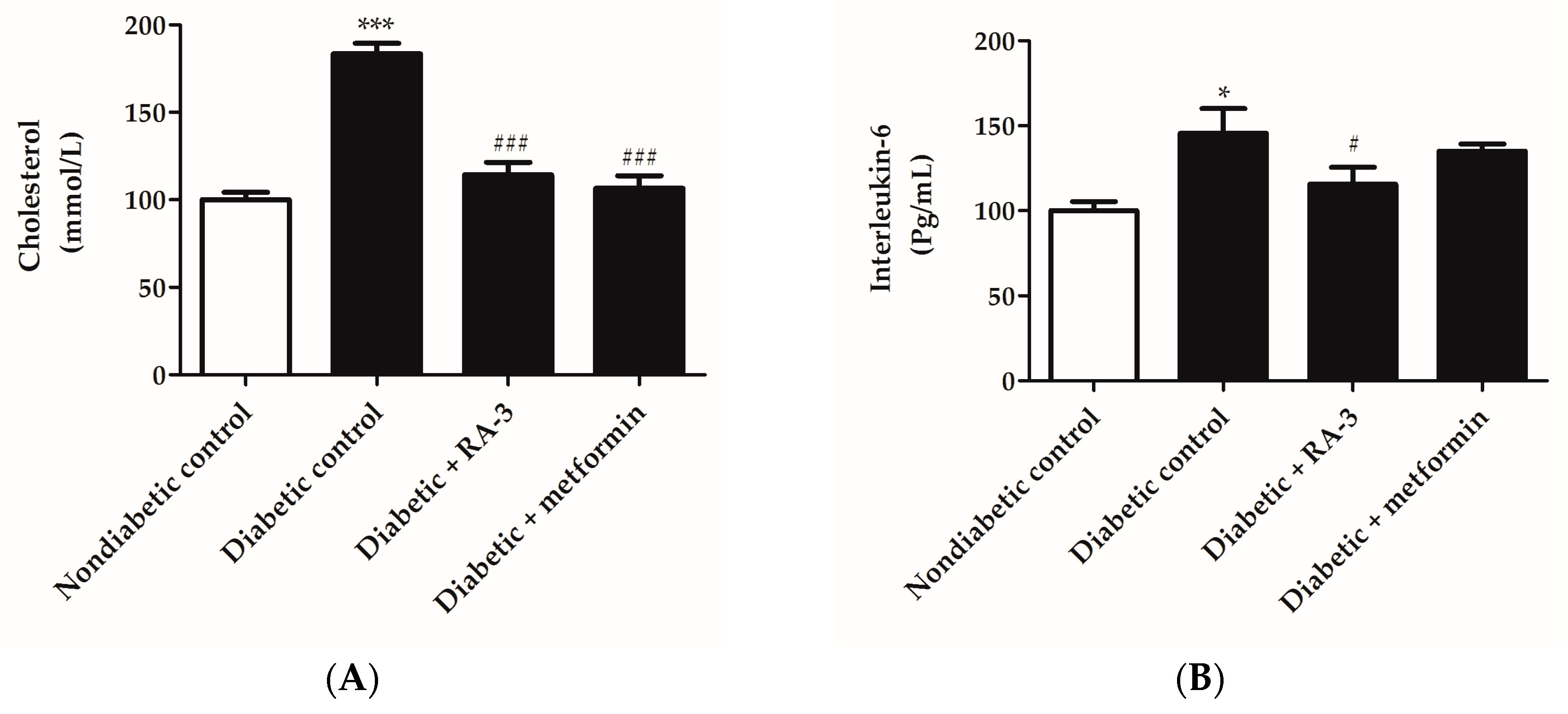
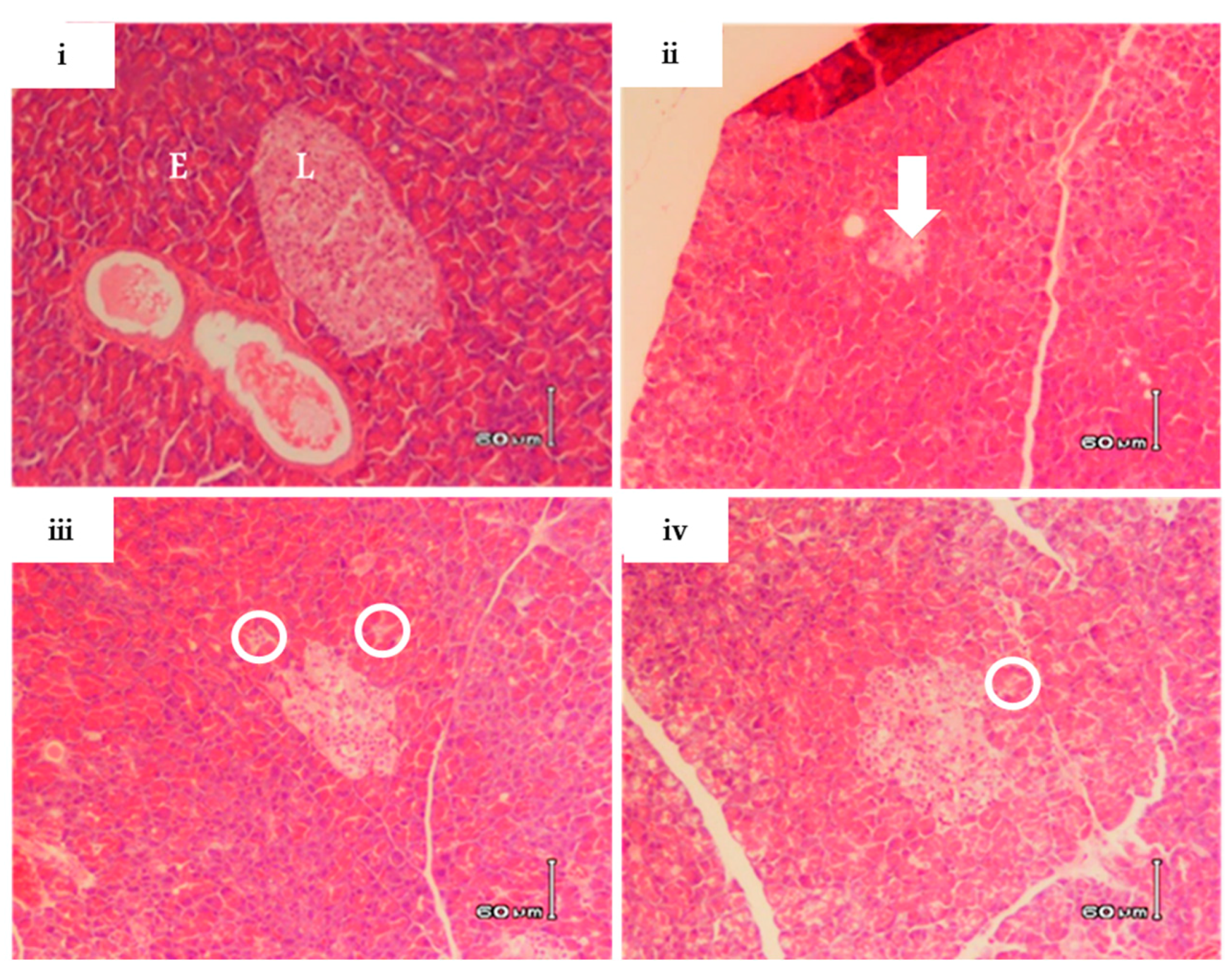
2.4. RA-3 Reduced Cholesterol and Interleukin-6 Levels While it Improved the Pancreatic Beta Cell Ultrastructure of the Type 2 Diabetic Rats
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Extraction and Compound Isolation
4.3. Animals
4.4. Establishment of a Type 2 Diabetic Rat Model
4.5. Treatment of High Fat Diet- Induced Diabetic Rats with RA-3
4.6. Oral Glucose Tolerance Test
4.7. Determination of Fasting Plasma Glucose Levels
4.8. Biochemical Analysis
4.9. Histopathological Studies
4.10. Data Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- International Diabetes Federation (IDF). IDF Diabetes Atlas, 7th ed. Available online: http://www.diabetesatlas.org/ (accessed on 29 May 2017).
- World Health Organization (WHO). World Health Statistics 2012. Available online: http://apps.who.int/iris/bitstream/10665/44844/1/9789241564441_eng.pdf?ua=1 (accessed on 29 May 2017).
- Drews, G.; Krippeit-Drews, P.; Dufer, M. Oxidative stress and beta-cell dysfunction. Pflug. Arch. 2010, 460, 703–718. [Google Scholar] [CrossRef] [PubMed]
- Wright, E.; Scism-Bacon, J.L.; Glass, L.C. Oxidative stress in type 2 diabetes: The role of fasting and postprandial glycaemia. Int. J. Clin. Pract. 2006, 60, 308–314. [Google Scholar] [CrossRef] [PubMed]
- Elmarakby, A.A.; Sullivan, J.C. Relationship between oxidative stress and inflammatory cytokines in diabetic nephropathy. Cardiovasc. Ther. 2012, 30, 49–59. [Google Scholar] [CrossRef] [PubMed]
- Montane, J.; Cadavez, L.; Novials, A. Novials, Stress and the inflammatory process: A major cause of pancreatic cell death in type 2 diabetes. Diabetes Metab. Syndr. Obes. 2014, 7, 25–34. [Google Scholar] [PubMed]
- Bonora, E. Protection of pancreatic beta-cells: Is it feasible? Nutr. Metab. Cardiovas. 2008, 18, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Olokoba, A.B.; Obateru, O.A.; Olokoba, L.B. Type 2 diabetes mellitus: A review of current trends. Oman Med. J. 2012, 27, 269–273. [Google Scholar] [CrossRef] [PubMed]
- Atanasov, A.G.; Waltenberger, B.; Pferschy-Wenzig, E.M.; Linder, T.; Wawrosch, C.; Uhrin, P.; Temml, V.; Wang, L.; Schwaiger, S.; Heiss, E.H.; et al. Discovery and resupply of pharmacologically active plant-derived natural products: A review. Biotechnol. Adv. 2015, 33, 1582–1614. [Google Scholar] [CrossRef] [PubMed]
- Ji, H.F.; Li, X.J.; Zhang, H.Y. Natural products and drug discovery. Can thousands of years of ancient medical knowledge lead us to new and powerful drug combinations in the fight against cancer and dementia? EMBO Rep. 2009, 10, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Rasoanaivo, P.; Wright, C.W.; Willcox, M.L.; Gilbert, B. Whole plant extracts versus single compounds for the treatment of malaria: Synergy and positive interactions. Malar. J. 2011, 10, S4. [Google Scholar] [CrossRef] [PubMed]
- Kuang, Q.T.; Zhao, J.J.; Ye, C.L.; Wang, J.R.; Ye, K.H.; Zhang, X.Q.; Wang, Y.; Ye, W.C. Nephro-protective effects of total triterpenoids from Psidium guajava leaves on type 2 diabetic rats. J. Med. Chin. Mater. 2012, 35, 94–97. [Google Scholar]
- Nazaruk, J.; Borzym-Kluczyk, M. The role of triterpenes in the management of diabetes mellitus and its complications. Phytochem. Rev. 2015, 14, 675–690. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Jin, S.; Song, C.; Chen, C.; Zhang, Y.; Xiang, Y.; Xu, Y.; Feng, Y.; Wan, Q.; Jiang, H. The metabolic change of serum lysophosphatidylcholines involved in the lipid lowering effect of triterpenes from Alismatis rhizoma on high-fat diet induced hyperlipidemia mice. J. Ethnopharmacol. 2016, 177, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Cui, W.X.; Yang, J.; Chen, X.Q.; Mao, Q.; Wei, X.L.; Wen, X.D.; Wang, Q. Triterpenoid-rich fraction from Ilex hainanensis Merr. attenuates non-alcoholic fatty liver disease induced by high fat diet in rats. Am. J. Chin. Med. 2013, 41, 487–502. [Google Scholar] [CrossRef] [PubMed]
- Leite, P.E.; Lima-Araujo, K.G.; Franca, G.R.; Lagrota-Candido, J.; Santos, W.C.; Quirico-Santos, T. Implant of polymer containing pentacyclic triterpenes from Eugenia punicifolia inhibits inflammation and activates skeletal muscle remodeling. Arch. Immunol. Ther. Exp. (Warsz). 2014, 62, 483–491. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Wang, Y.; Li, C.; Wang, X.; He, X. Anti-inflammatory oleanolic triterpenes from Chinese Acorns. Molecules 2016, 21, 669. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.; Seo, J.; Kim, J.; Kim, H.; Youn, U.; Lee, J.; Jung, H.; Na, M.; Hattori, M.; Min, B.; et al. Lanostane triterpenes from the fruiting bodies of Ganoderma lucidum and their inhibitory effects on adipocyte differentiation in 3T3-L1 Cells. J. Nat. Prod. 2010, 73, 172–176. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zhu, L.; Tan, J.; Zhou, X.; Xiao, L.; Yang, X.; Wang, B. Glucosidase inhibitory activity and antioxidant activity of flavonoid compound and triterpenoid compound from Agrimonia Pilosa Ledeb. BMC Complement. Altern. Med. 2014, 14, 12. [Google Scholar] [CrossRef] [PubMed]
- Mosa, R.A.; Cele, N.D.; Mabhida, S.E.; Shabalala, S.C.; Penduka, D.; Opoku, A.R. In vivo antihyperglycemic activity of a lanosteryl triterpene from Protorhus longifolia. Molecules 2015, 20, 13374–13383. [Google Scholar] [CrossRef] [PubMed]
- Oh, Y.S. Plant-derived compounds targeting pancreatic beta cells for the treatment of diabetes. Evid. Based Complement. Altern. Med. 2015, 2015, 629863. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.J.; Ye, J.M.; Turner, N.; Hohnen-Behrens, C.; Ke, C.Q.; Tang, C.P.; Chen, T.; Weiss, H.C.; Gesing, E.R.; Rowland, A.; et al. Antidiabetic activities of triterpenoids isolated from bitter melon associated with activation of the AMPK pathway. Chem. Biol. 2008, 15, 263–273. [Google Scholar] [CrossRef] [PubMed]
- Machaba, K.E.; Cobongela, S.Z.; Mosa, R.A.; Oladipupo, L.A.; Djarova, T.G.; Opoku, A.R. In vivo anti-hyperlipidemic activity of the triterpene from the stem bark of Protorhus longifolia (Benrh) Engl. Lipids Health Dis. 2014, 13, 131. [Google Scholar] [CrossRef] [PubMed]
- Mosa, R.A.; Hlophe, N.B.; Ngema, N.T.; Penduka, D.; Lawal, O.A.; Opoku, A.R. Cardioprotective potential of a lanosteryl triterpene from Protorhus longifolia. Pharm Biol. 2016, 54, 3244–3248. [Google Scholar] [CrossRef] [PubMed]
- Mosa, R.A.; Naidoo, J.J.; Nkomo, F.S.; Mazibuko, S.E.; Muller, C.J.; Opoku, A.R. In vitro antihyperlipidemic potential of triterpenes from stem bark of Protorhus longifolia. Planta Med. 2014, 80, 1685–1691. [Google Scholar] [CrossRef] [PubMed]
- Mosa, R.A.; Lazarus, G.; Gwala, P.E.; Oyedeji, A.O.; Opoku, A.R. In vitro anti-platelet aggregation, antioxidant and cytotoxic activity of extracts of some Zulu medicinal plants. J. Nat. Prod. 2011, 4, 136–146. [Google Scholar]
- Mosa, R.A.; Oyedeji, O.A.; Shode, F.O.; Singh, M.; Opoku, A.R. Triterpenes from the stem bark of Protorhus longifolia exhibit anti-platelet aggregation activity. Afr. J. Pharm. Pharmacol. 2011, 5, 2698–2714. [Google Scholar]
- Himpe, E.; Cunha, D.A.; Song, I.; Bugliani, M.; Marchetti, P.; Cnop, M.; Bouwens, L. Phenylpropenoic acid glucoside from rooibos protects pancreatic beta cells against cell death induced by acute injury. PLoS ONE 2016, 11, e0157604. [Google Scholar] [CrossRef] [PubMed]
- Dludla, P.V.; Muller, C.J.; Joubert, E.; Louw, J.; Essop, M.F.; Gabuza, K.B.; Ghoor, S.; Huisamen, B.; Johnson, R. Aspalathin protects the heart against hyperglycemia-induced oxidative damage by up-regulating nrf2 expression. Molecules 2017, 22, 129. [Google Scholar] [CrossRef] [PubMed]
- Alexandraki, K.; Piperi, C.; Kalofoutis, C.; Singh, J.; Alaveras, A.; Kalofoutis, A. Inflammatory process in type 2 diabetes: The role of cytokines. Ann. N. Y. Acad. Sci. 2006, 1084, 89–117. [Google Scholar] [CrossRef] [PubMed]
- Dudhgaonkar, S.; Thyagarajan, A.; Sliva, D. Suppression of the inflammatory response by triterpenes isolated from the mushroom Ganoderma lucidum. Int. Immunopharmacol. 2009, 9, 1272–1280. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.E.; Kim, J.W.; Lee, E.M.; Ahn, Y.B.; Song, K.H.; Yoon, K.H.; Kim, H.W.; Park, C.W.; Li, G.; Liu, Z.; et al. Chronic resveratrol treatment protects pancreatic islets against oxidative stress in db/db mice. PLoS ONE 2012, 7, e50412. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.; Dludla, P.V.; Muller, C.J.; Huisamen, B.; Essop, M.F.; Louw, J. The transcription profile unveils the cardioprotectiveeffect of aspalathin against lipid toxicity in an in vitro H9c2 Model. Molecules 2017, 22, 219. [Google Scholar] [CrossRef] [PubMed]
- Reisman, S.A.; Aleksunes, L.M.; Klaassen, C.D. Oleanolic acid activates Nrf2 and protects from acetaminophen hepatotoxicity via Nrf2-dependent and Nrf2-independent processes. GG Biochem. Pharmacol. 2009, 77, 1273–1282. [Google Scholar] [CrossRef]
- Loboda, A.; Rojczyk-Golebiewska, E.; Bednarczyk-Cwynar, B.; Lucjusz, Z.; Jozkowicz, A.; Dulak, J. Targeting nrf2-mediated gene transcription by triterpenoids and their derivatives. Biomol. Ther. (Seoul) 2012, 20, 499–505. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, E.H.; Suzuki, T.; Funayama, R.; Nagashima, T.; Hayashi, M.; Sekine, H.; Tanaka, N.; Moriguchi, T.; Motohashi, H.; Nakayama, K.; et al. Nrf2 suppresses macrophage inflammatory response by blocking proinflammatory cytokine transcription. Nat. Commun. 2016, 7, 11624. [Google Scholar] [CrossRef] [PubMed]
- Iseli, T.J.; Turner, N.; Zeng, X.Y.; Cooney, G.J.; Kraegen, E.W.; Yao, S.; Ye, Y.; James, D.E.; Ye, J.M. Activation of AMPK by bitter melon triterpenoids involves CaMKKbeta. PLoS ONE 2013, 8, e62309. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compound (RA-3) are available fromauthors. Metformin is available through commercial sources. |
| Experimental Group | FPG Day 0 (mmol/L) | FPG Day 28 (mmol/L) | C-peptide Day 28 (µg/L) |
|---|---|---|---|
| Non-diabetic control | 4.1 ± 0.22 | 3.9 ± 0.04 | 0.8 ± 0.01 |
| Diabetic control | 18.4 ± 0.78 *** | 29.0 ± 1.09 *** | 0.2 ± 2.41 *** |
| Diabetic + RA-3 | 11.5 ± 0.38 ***,### | 4.3 ± 0.11 ### | 0.4 ± 0.14 *,# |
| Diabetic + metformin | 15.7 ± 0.66 ***,# | 4.5 ± 0.22 ### | 0.4 ± 0.12 *,# |
| Experimental Group | GSH Content (nmol/mL) | SOD Activity (Inhibition Rate %) | CAT Activity (Units/mL) | MDA Levels (nmol/µL) |
|---|---|---|---|---|
| Nondiabetic control | 7.33 ± 0.01 | 56 ± 0.005 | 0.12 ± 0.005 | 0.37 ± 0.004 |
| Diabetic control | 2.38 ± 0.01 *** | 30 ± 0.012 ** | 0.08 ± 0.004 * | 1.31 ± 0.008 *** |
| Diabetic + RA-3 | 4.40 ± 0.006 *,# | 41 ± 0.004 * | 0.13 ± 0.001 # | 0.75 ± 0.005 *,## |
| Diabetic + metformin | 4.10 ± 0.003 *,# | 39 ± 0.004 * | 0.11 ± 0.005 # | 0.53 ± 0.003 *,## |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mabhida, S.E.; Mosa, R.A.; Penduka, D.; Osunsanmi, F.O.; Dludla, P.V.; Djarova, T.G.; Opoku, A.R. A Lanosteryl Triterpene from Protorhus longifolia Improves Glucose Tolerance and Pancreatic Beta Cell Ultrastructure in Type 2 Diabetic Rats. Molecules 2017, 22, 1252. https://doi.org/10.3390/molecules22081252
Mabhida SE, Mosa RA, Penduka D, Osunsanmi FO, Dludla PV, Djarova TG, Opoku AR. A Lanosteryl Triterpene from Protorhus longifolia Improves Glucose Tolerance and Pancreatic Beta Cell Ultrastructure in Type 2 Diabetic Rats. Molecules. 2017; 22(8):1252. https://doi.org/10.3390/molecules22081252
Chicago/Turabian StyleMabhida, Sihle E., Rebamang A. Mosa, Dambudzo Penduka, Foluso O. Osunsanmi, Phiwayinkosi V. Dludla, Tryana G. Djarova, and Andy R. Opoku. 2017. "A Lanosteryl Triterpene from Protorhus longifolia Improves Glucose Tolerance and Pancreatic Beta Cell Ultrastructure in Type 2 Diabetic Rats" Molecules 22, no. 8: 1252. https://doi.org/10.3390/molecules22081252
APA StyleMabhida, S. E., Mosa, R. A., Penduka, D., Osunsanmi, F. O., Dludla, P. V., Djarova, T. G., & Opoku, A. R. (2017). A Lanosteryl Triterpene from Protorhus longifolia Improves Glucose Tolerance and Pancreatic Beta Cell Ultrastructure in Type 2 Diabetic Rats. Molecules, 22(8), 1252. https://doi.org/10.3390/molecules22081252






