Magnolol, a Natural Polyphenol, Attenuates Dextran Sulfate Sodium-Induced Colitis in Mice
Abstract
:1. Introduction
2. Results
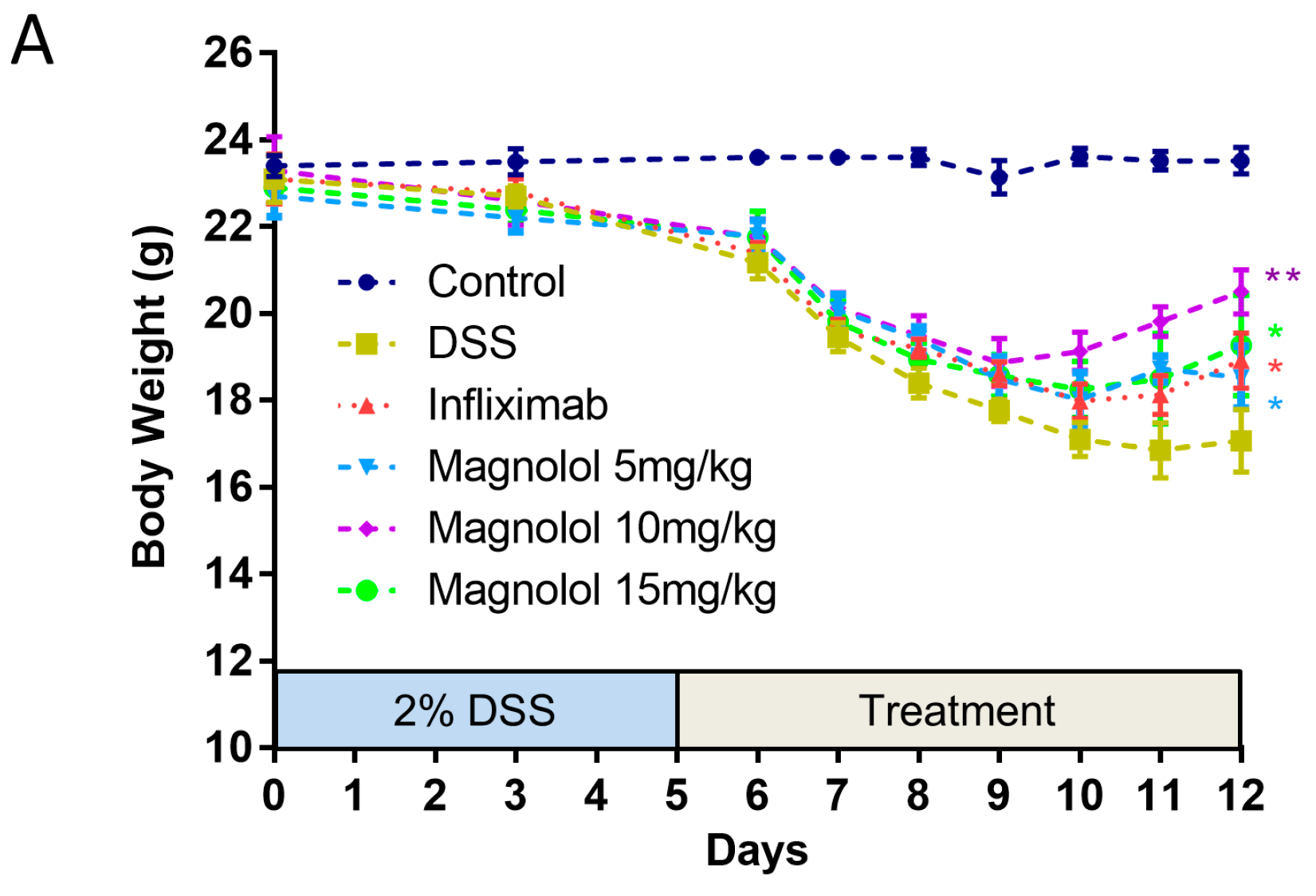
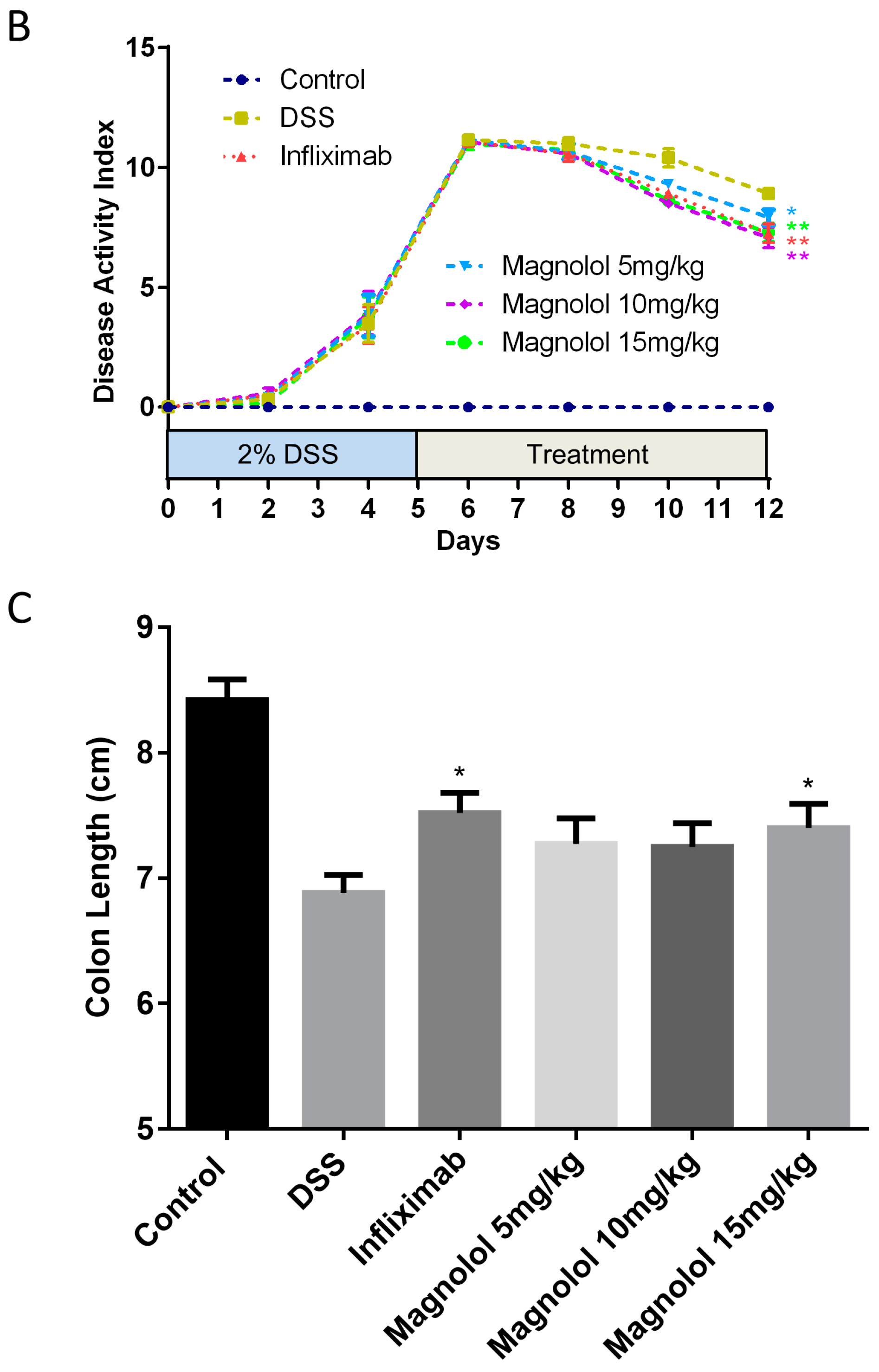
2.1. Magnolol Impact the Severity of DSS-Induced Colitis in Mice
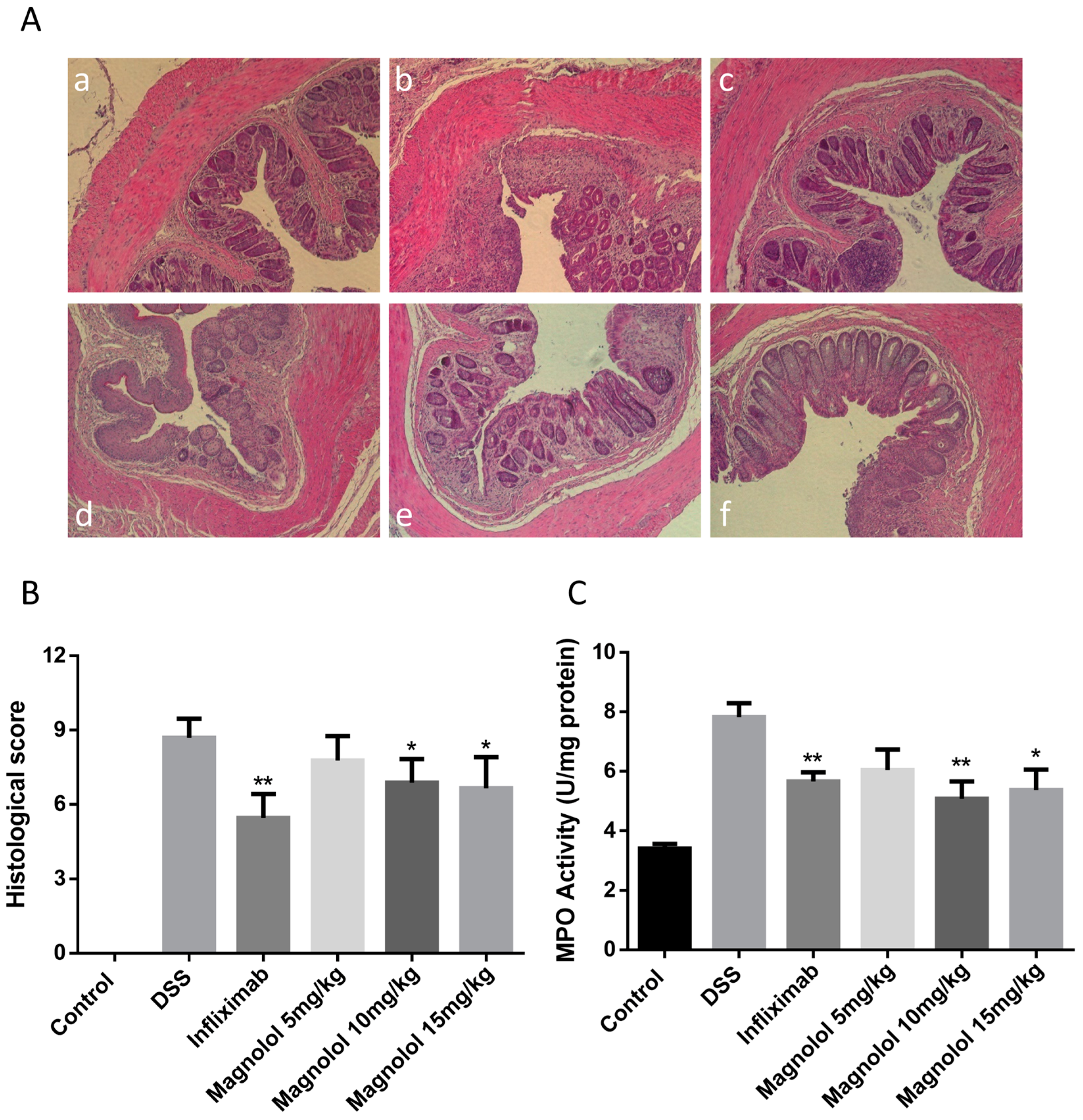
2.2. Effects of Magnolol on Histopathological Changes and Myeloperoxidase Activity in the Colon of DSS-Treated Mice
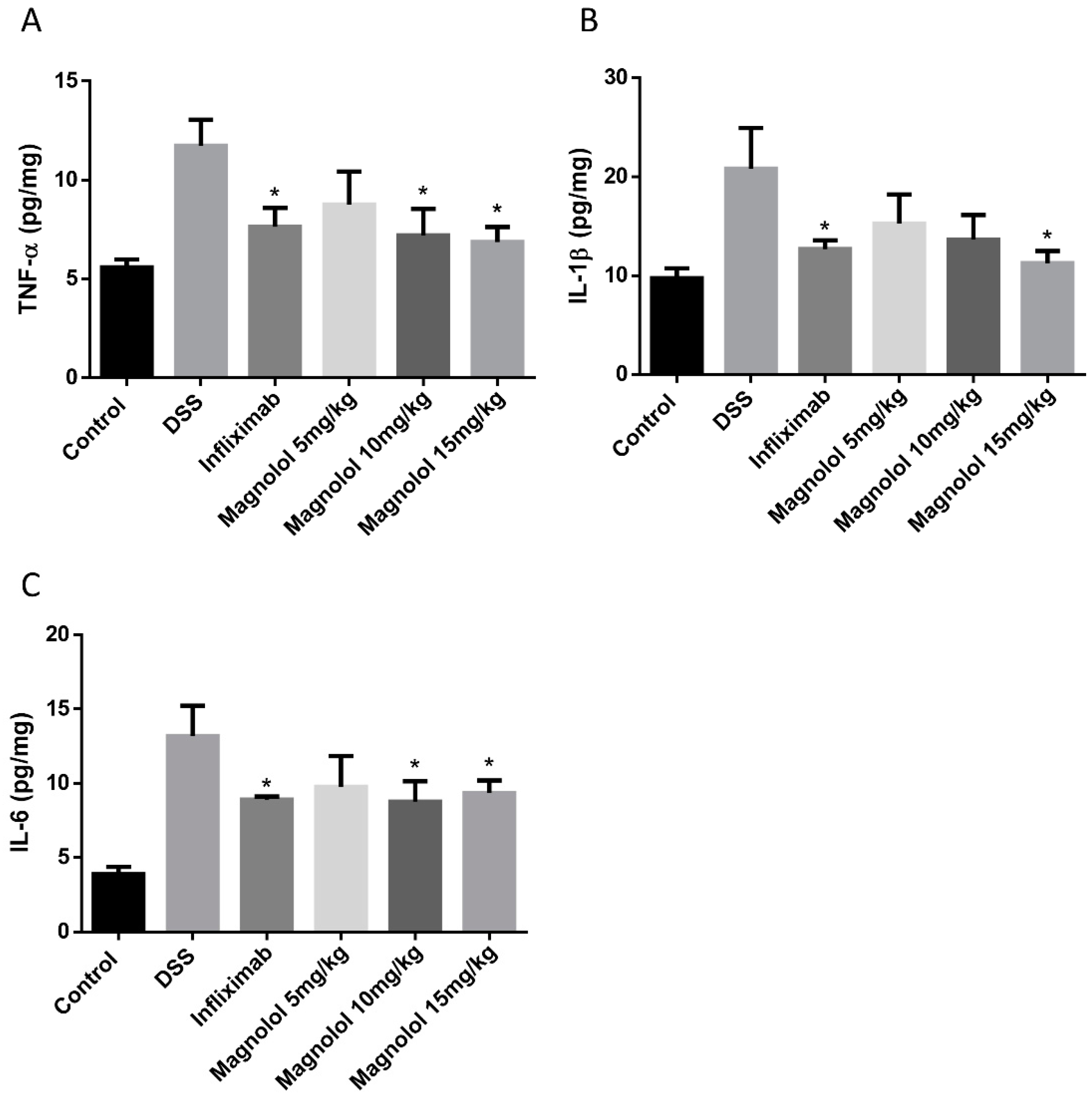
2.3. Effects of Magnolol on Colonic Pro-Inflammatory Cytokines in DSS-Treated Mice
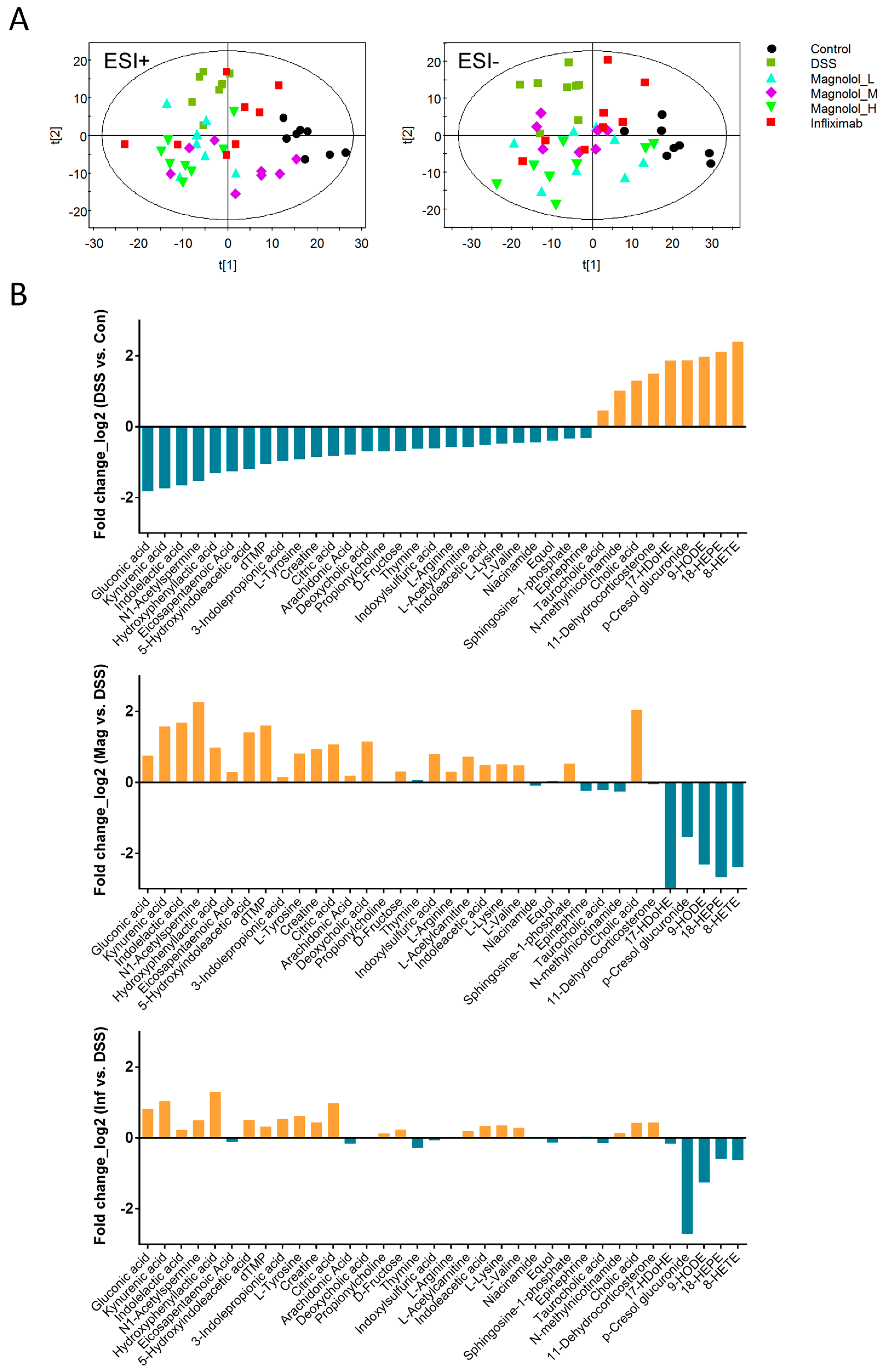
2.4. Effect of Magnolol on Alterations of Serum Metabolome in DSS-Treated Mice
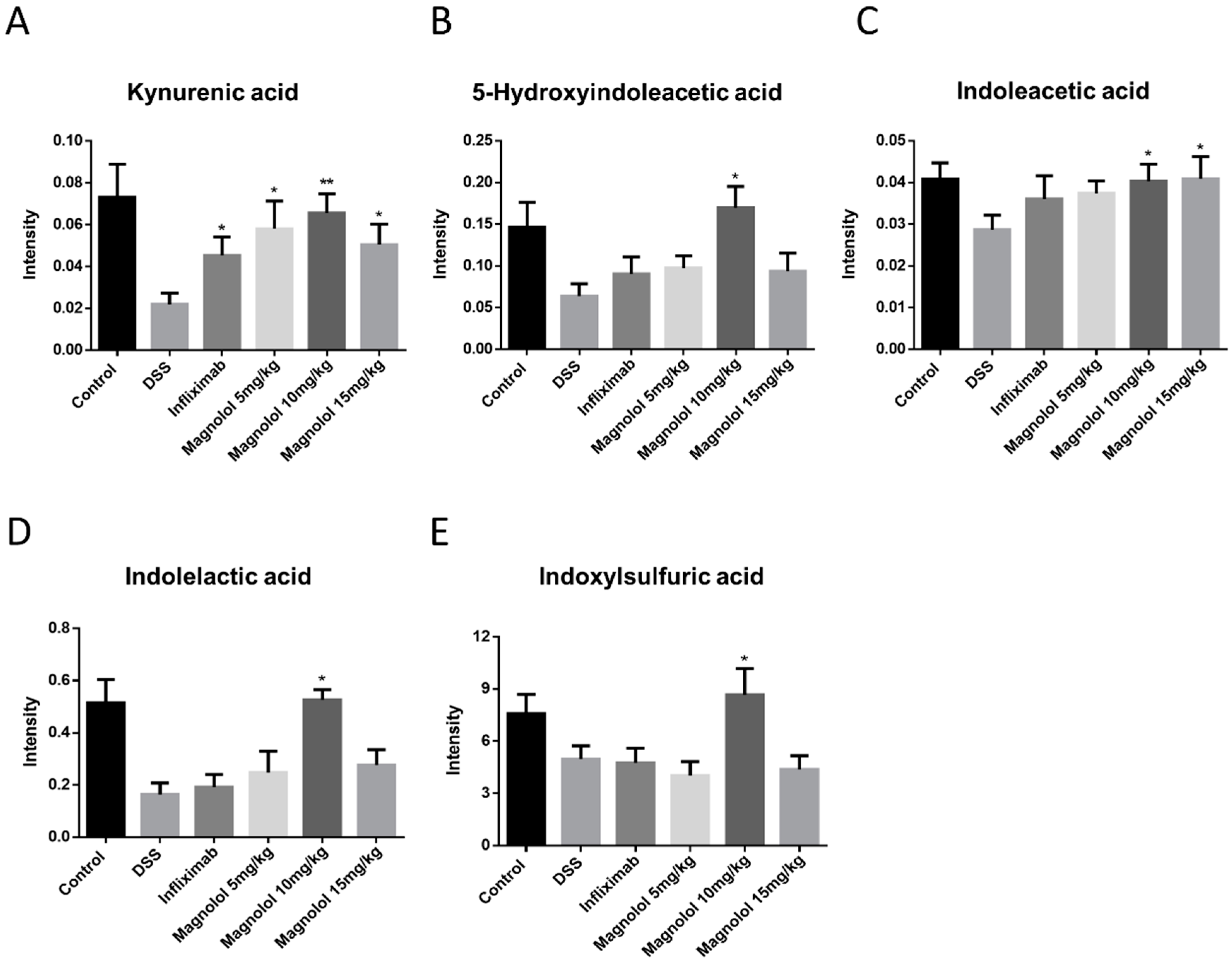
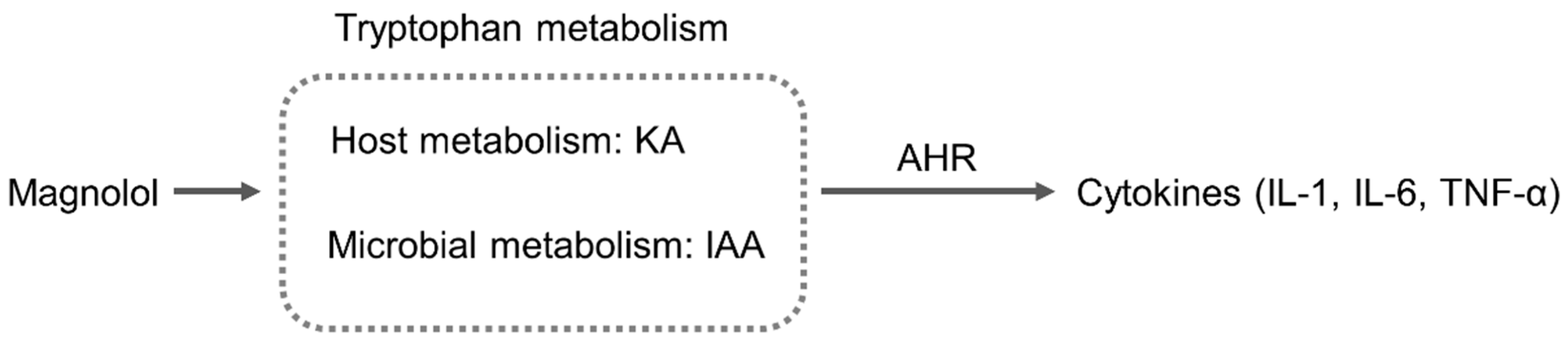
2.5. Magnolol Regulated Tryptophan Metabolic Pathway in Mice
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Chemicals and Reagents
4.3. Colitis Model and Drug Treatment
4.4. Disease Activity Index Evaluation
4.5. Hematoxylin/Eosin Staining
4.6. Myeloperoxidase Activity Assay
4.7. Measurement of Colonic Levels of Cytokines
4.8. UPLC Separation of Serum Metabolites
4.9. QTOF-MS Analysis and Metabolite Identification
4.10. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Xavier, R.J.; Podolsky, D.K. Unravelling the pathogenesis of inflammatory bowel disease. Nature 2007, 448, 427–434. [Google Scholar] [CrossRef] [PubMed]
- Ananthakrishnan, A.N. Epidemiology and risk factors for IBD. Nat. Rev. Gastroenterol. Hepatol. 2015, 12, 205–217. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Wang, F.; Zhang, H.J.; Sheng, J.Q.; Yan, W.F.; Ma, M.X.; Fan, R.Y.; Gu, F.; Li, C.F.; Chen, D.F.; et al. Corticosteroid therapy in ulcerative colitis: Clinical response and predictors. World J. Gastroenterol. 2015, 21, 3005–3015. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Li, N.; Ren, Y.; Ma, T.; Wang, C.; Wang, J.; You, S. Efficacy and safety of beclomethasone dipropionate versus 5-aminosalicylic acid in the treatment of ulcerative colitis: A systematic review and meta-analysis. PLoS ONE 2016, 11, e0160500. [Google Scholar] [CrossRef] [PubMed]
- Timmer, A.; Patton, P.H.; Chande, N.; McDonald, J.W.; MacDonald, J.K. Azathioprine and 6-mercaptopurine for maintenance of remission in ulcerative colitis. Cochrane Database Syst. Rev. 2016. [Google Scholar] [CrossRef]
- Wang, Y.; MacDonald, J.K.; Vandermeer, B.; Griffiths, A.M.; El-Matary, W. Methotrexate for maintenance of remission in ulcerative colitis. Cochrane Database Syst. Rev. 2015, CD007560. [Google Scholar] [CrossRef]
- Rutgeerts, P.; Sandborn, W.J.; Feagan, B.G.; Reinisch, W.; Olson, A.; Johanns, J.; Travers, S.; Rachmilewitz, D.; Hanauer, S.B.; Lichtenstein, G.R.; et al. Infliximab for induction and maintenance therapy for ulcerative colitis. N. Engl. J. Med. 2005, 353, 2462–2476. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.J.; Lee, Y.M.; Lee, C.K.; Jung, J.K.; Han, S.B.; Hong, J.T. Therapeutic applications of compounds in the Magnolia family. Pharmacol. Ther. 2011, 130, 157–176. [Google Scholar] [CrossRef] [PubMed]
- Kou, D.Q.; Jiang, Y.L.; Qin, J.H.; Huang, Y.H. Magnolol attenuates the inflammation and apoptosis through the activation of SIRT1 in experimental stroke rats. Pharmacol. Rep. 2017, 69, 642–647. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Xu, Y.; Yu, S.; Huang, Y.; Lu, L.; Liang, X. Anti-angiogenic and anti-inflammatory effect of Magnolol in the oxygen-induced retinopathy model. Inflamm. Res. 2016, 65, 81–93. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.H.; Chen, M.C.; Chen, T.H.; Chang, H.Y.; Chou, T.C. Magnolol ameliorates lipopolysaccharide-induced acute lung injury in rats through PPAR-gamma-dependent inhibition of NF-kB activation. Int. Immunopharmacol. 2015, 28, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Ni, Y.F.; Jiang, T.; Cheng, Q.S.; Gu, Z.P.; Zhu, Y.F.; Zhang, Z.P.; Wang, J.; Yan, X.L.; Wang, W.P.; Ke, C.K.; et al. Protective effect of magnolol on lipopolysaccharide-induced acute lung injury in mice. Inflammation 2012, 35, 1860–1866. [Google Scholar] [CrossRef] [PubMed]
- Wei, W.; Liang, D.; Song, X.; Wang, T.; Cao, Y.; Yang, Z.; Zhang, N. Magnolol inhibits the inflammatory response in mouse mammary epithelial cells and a mouse mastitis model. Inflammation 2015, 38, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.H.; Shih, K.S.; Liou, J.P.; Wu, Y.W.; Chang, A.S.; Wang, K.L.; Tsai, C.L.; Yang, C.R. Anti-arthritic effects of magnolol in human interleukin 1β-stimulated fibroblast-like synoviocytes and in a rat arthritis model. PLoS ONE 2012, 7, e31368. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Liu, B.; Zhang, N.; Liu, Z.; Liang, D.; Li, F.; Cao, Y.; Feng, X.; Zhang, X.; Yang, Z. Magnolol inhibits lipopolysaccharide-induced inflammatory response by interfering with TLR4 mediated NF-kappaB and MAPKs signaling pathways. J. Ethnopharmacol. 2013, 145, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Murakami, Y.; Kawata, A.; Seki, Y.; Koh, T.; Yuhara, K.; Maruyama, T.; Machino, M.; Ito, S.; Kadoma, Y.; Fujisawa, S. Comparative inhibitory effects of magnolol, honokiol, eugenol and bis-eugenol on cyclooxygenase-2 expression and nuclear factor-kappa B activation in RAW264.7 macrophage-like cells stimulated with fimbriae of Porphyromonas gingivalis. In Vivo 2012, 26, 941–950. [Google Scholar] [PubMed]
- Yunhe, F.; Bo, L.; Xiaosheng, F.; Fengyang, L.; Dejie, L.; Zhicheng, L.; Depeng, L.; Yongguo, C.; Xichen, Z.; Naisheng, Z.; et al. The effect of magnolol on the Toll-like receptor 4/nuclear factor kappa B signaling pathway in lipopolysaccharide-induced acute lung injury in mice. Eur. J. Pharmacol. 2012, 689, 255–261. [Google Scholar] [CrossRef] [PubMed]
- Liang, C.J.; Lee, C.W.; Sung, H.C.; Chen, Y.H.; Wang, S.H.; Wu, P.J.; Chiang, Y.C.; Tsai, J.S.; Wu, C.C.; Li, C.Y.; et al. Magnolol reduced TNF-alpha-induced vascular cell adhesion molecule-1 expression in endothelial cells via JNK/p38 and NF-kappaB signaling pathways. Am. J. Chin. Med. 2014, 42, 619–637. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Zang, K.H.; Luo, J.L.; Leung, F.P.; Huang, Y.; Lin, C.Y.; Yang, Z.J.; Lu, A.P.; Tang, X.D.; Xu, H.X.; et al. Magnolol inhibits colonic motility through down-regulation of voltage-sensitive L-type Ca2+ channels of colonic smooth muscle cells in rats. Phytomedicine 2013, 20, 1272–1279. [Google Scholar] [CrossRef] [PubMed]
- Bian, Z.X.; Zhang, G.S.; Wong, K.L.; Hu, X.G.; Liu, L.; Yang, Z.; Li, M. Inhibitory effects of magnolol on distal colon of guinea pig in vitro. Biol. Pharm. Bull. 2006, 29, 790–795. [Google Scholar] [CrossRef] [PubMed]
- Miao, B.; Zhang, S.; Wang, H.; Yang, T.; Zhou, D.; Wang, B.E. Magnolol pretreatment prevents sepsis-induced intestinal dysmotility by maintaining functional interstitial cells of Cajal. Inflammation 2013, 36, 897–906. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.C.; Zhang, S.W.; Sun, L.N.; Wang, H.; Ren, A.M. Magnolol attenuates sepsis-induced gastrointestinal dysmotility in rats by modulating inflammatory mediators. World J. Gastroenterol. 2008, 14, 7353–7360. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Xu, Y.; Zhang, M.; Gao, L.; Fang, C.; Zhou, C. Magnolol inhibits LPS-induced inflammatory response in uterine epithelial cells: Magnolol inhibits LPS-induced inflammatory response. Inflammation 2013, 36, 997–1003. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.H.; Chen, T.H.; Chou, T.C. Magnolol Inhibits RANKL-induced osteoclast differentiation of raw 264.7 macrophages through heme oxygenase-1-dependent inhibition of NFATc1 expression. J. Nat. Prod. 2015, 78, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Karki, R.; Ho, O.M.; Kim, D.W. Magnolol attenuates neointima formation by inducing cell cycle arrest via inhibition of ERK1/2 and NF-kappaB activation in vascular smooth muscle cells. Biochim. Biophys. Acta 2013, 1830, 2619–2628. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.H.; Huang, R.Y.; Chou, T.C. Magnolol ameliorates ligature-induced periodontitis in rats and osteoclastogenesis: In vivo and in vitro study. Evid. Based Complement. Alternat. Med. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.W.; Li, Y.; Wang, X.Q.; Tian, F.; Cao, H.; Wang, M.W.; Sun, Q.S. Effects of magnolol and honokiol derived from traditional Chinese herbal remedies on gastrointestinal movement. World J. Gastroenterol. 2005, 11, 4414–4418. [Google Scholar] [CrossRef] [PubMed]
- Jeong, S.I.; Kim, Y.S.; Lee, M.Y.; Kang, J.K.; Lee, S.; Choi, B.K.; Jung, K.Y. Regulation of contractile activity by magnolol in the rat isolated gastrointestinal tracts. Pharmacol. Res. 2009, 59, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Han, X.; Tang, S.; Xiao, W.; Tan, Z.; Zhou, C.; Wang, M.; Kang, J. Magnolol and honokiol regulate the calcium-activated potassium channels signaling pathway in Enterotoxigenic Escherichia coli-induced diarrhea mice. Eur. J. Pharmacol. 2015, 755, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Poritz, L.S.; Garver, K.I.; Green, C.; Fitzpatrick, L.; Ruggiero, F.; Koltun, W.A. Loss of the tight junction protein ZO-1 in dextran sulfate sodium induced colitis. J. Surg. Res. 2007, 140, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Storr, M.; Vogel, H.J.; Schicho, R. Metabolomics: Is it useful for inflammatory bowel diseases? Curr. Opin. Gastroenterol. 2013, 29, 378–383. [Google Scholar] [CrossRef] [PubMed]
- Schicho, R.; Nazyrova, A.; Shaykhutdinov, R.; Duggan, G.; Vogel, H.J.; Storr, M. Quantitative Metabolomic Profiling of Serum and Urine in DSS-Induced Ulcerative Colitis of Mice by 1H NMR Spectroscopy. J. Proteome Res. 2010, 9, 6265–6273. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Xiao, H.T.; Wang, H.S.; Mu, H.X.; Zhao, L.; Du, J.; Yang, D.P.; Wang, D.M.; Bian, Z.X.; Lin, S.H. Halofuginone reduces the inflammatory responses of DSS-induced colitis through metabolic reprogramming. Mol. Biosyst. 2016, 12, 2296–2303. [Google Scholar] [CrossRef] [PubMed]
- Williams, H.R.T.; Willsmore, J.D.; Cox, I.J.; Walker, D.G.; Cobbold, J.F.L.; Taylor-Robinson, S.D.; Orchard, T.R. Serum Metabolic Profiling in Inflammatory Bowel Disease. Dig. Dis. Sci. 2012, 57, 2157–2165. [Google Scholar] [CrossRef] [PubMed]
- Dawiskiba, T.; Deja, S.; Mulak, A.; Zabek, A.; Jawien, E.; Pawelka, D.; Banasik, M.; Mastalerz-Migas, A.; Balcerzak, W.; Kaliszewski, K.; et al. Serum and urine metabolomic fingerprinting in diagnostics of inflammatory bowel diseases. World J. Gastroenterol. 2014, 20, 163–174. [Google Scholar] [CrossRef] [PubMed]
- Dong, F.C.; Zhang, L.L.; Hao, F.H.; Tang, H.R.; Wang, Y.L. Systemic responses of mice to dextran sulfate sodium-induced acute ulcerative colitis using 1H NMR spectroscopy. J. Proteome Res. 2013, 12, 2958–2966. [Google Scholar] [CrossRef] [PubMed]
- Etienne-Mesmin, L.; Chassaing, B.; Gewirtz, A.T. Tryptophan: A gut microbiota-derived metabolites regulating inflammation. World J. Gastrointest. Pharmacol. Ther. 2017, 8, 7–9. [Google Scholar] [CrossRef] [PubMed]
- Richard, D.M.; Dawes, M.A.; Mathias, C.W.; Acheson, A.; Hill-Kapturczak, N.; Dougherty, D.M. L-Tryptophan: Basic metabolic functions, behavioral research and therapeutic indications. Int. J. Tryptophan Res. 2009, 2, 45–60. [Google Scholar] [PubMed]
- Berger, M.; Gray, J.A.; Roth, B.L. The expanded biology of serotonin. Annu. Rev. Med. 2009, 60, 355–366. [Google Scholar] [CrossRef] [PubMed]
- Qin, H.Y.; Luo, J.L.; Qi, S.D.; Xu, H.X.; Sung, J.J.Y.; Bian, Z.X. Visceral hypersensitivity induced by activation of transient receptor potential vanilloid type 1 is mediated through the serotonin pathway in rat colon. Eur. J. Pharmacol. 2010, 647, 75–83. [Google Scholar] [CrossRef] [PubMed]
- O’Mahony, S.M.; Clarke, G.; Borre, Y.E.; Dinan, T.G.; Cryan, J.F. Serotonin, tryptophan metabolism and the brain-gut-microbiome axis. Behav. Brain Res. 2015, 277, 32–48. [Google Scholar] [CrossRef] [PubMed]
- Hubbard, T.D.; Murray, I.A.; Perdew, G.H. Indole and tryptophan metabolism: Endogenous and dietary routes to ah receptor activation. Drug Metab. Dispos. 2015, 43, 1522–1535. [Google Scholar] [CrossRef] [PubMed]
- Elizondo, G.; Rodriguez-Sosa, M.; Estrada-Muniz, E.; Gonzalez, F.J.; Vega, L. Deletion of the aryl hydrocarbon receptor enhances the inflammatory response to leishmania major infection. Int. J. Biol. Sci. 2011, 7, 1220–1229. [Google Scholar] [CrossRef] [PubMed]
- Lamas, B.; Richard, M.L.; Leducq, V.; Pham, H.P.; Michel, M.L.; Da Costa, G.; Bridonneau, C.; Jegou, S.; Hoffmann, T.W.; Natividad, J.M.; et al. CARD9 impacts colitis by altering gut microbiota metabolism of tryptophan into aryl hydrocarbon receptor ligands. Nat. Med. 2016, 22, 598–605. [Google Scholar] [CrossRef] [PubMed]
- Lamas, B.; Richard, M.L.; Sokol, H. Caspase recruitment domain 9, microbiota, and tryptophan metabolism: Dangerous liaisons in inflammatory bowel diseases. Curr. Opin. Clin. Nutr. Metab. Care 2017, 20, 243–247. [Google Scholar] [CrossRef] [PubMed]
- Jensen, B.A.; Leeman, R.J.; Schlezinger, J.J.; Sherr, D.H. Aryl hydrocarbon receptor (AhR) agonists suppress interleukin-6 expression by bone marrow stromal cells: An immunotoxicology study. Environ. Health 2003, 2, 16. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Pu, A.; Sheng, B.; Zhang, Z.; Li, L.; Liu, Z.; Wang, Q.; Li, X.; Ma, Y.; Yu, M.; et al. Aryl hydrocarbon receptor activation modulates CD8alphaalpha+TCRalphabeta+ IELs and suppression of colitis manifestations in mice. Biomed. Pharmacother. 2017, 87, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Wirtz, S.; Neufert, C.; Weigmann, B.; Neurath, M.F. Chemically induced mouse models of intestinal inflammation. Nat. Protoc. 2007, 2, 541–546. [Google Scholar] [CrossRef] [PubMed]
- Xiao, H.T.; Lin, C.Y.; Ho, D.H.; Peng, J.; Chen, Y.; Tsang, S.W.; Wong, M.; Zhang, X.J.; Zhang, M.; Bian, Z.X. Inhibitory effect of the gallotannin corilagin on dextran sulfate sodium-induced murine ulcerative colitis. J. Nat. Prod. 2013, 76, 2120–2125. [Google Scholar] [CrossRef] [PubMed]
- Mu, H.X.; Liu, J.; Fatima, S.; Lin, C.Y.; Shi, X.K.; Du, B.; Xiao, H.T.; Fan, B.M.; Bian, Z.X. Anti-inflammatory Actions of (+)-3’alpha-Angeloxy-4’-keto-3’,4’-dihydroseselin (Pd-Ib) against Dextran Sulfate Sodium-Induced Colitis in C57BL/6 Mice. J. Nat. Prod. 2016, 79, 1056–1062. [Google Scholar] [CrossRef] [PubMed]
- Qiu, Y.; Cai, G.; Su, M.; Chen, T.; Zheng, X.; Xu, Y.; Ni, Y.; Zhao, A.; Xu, L.X.; Cai, S.; et al. Serum metabolite profiling of human colorectal cancer using GC-TOFMS and UPLC-QTOFMS. J. Proteome Res. 2009, 8, 4844–4850. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.A.; Want, E.J.; O’Maille, G.; Abagyan, R.; Siuzdak, G. XCMS: Processing mass spectrometry data for metabolite profiling using nonlinear peak alignment, matching, and identification. Anal. Chem. 2006, 78, 779–787. [Google Scholar] [CrossRef] [PubMed]
- Storey, J.D.; Tibshirani, R. Statistical significance for genomewide studies. Proc. Natl. Acad. Sci. USA 2003, 100, 9440–9445. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Not available. |
| N. | ESI | R.T. | m/z | Metabolite | Pathway Classification |
|---|---|---|---|---|---|
| 1 | - | 8.97 | 319.2285 | 8-HETE | Arachidonic acid metabolism |
| 2 | - | 10.89 | 303.2334 | Arachidonic Acid | Arachidonic acid metabolism |
| 3 | - | 6.40 | 407.2810 | Cholic acid | Bile acid biosynthesis |
| 4 | - | 7.72 | 391.2861 | Deoxycholic acid | Bile acid biosynthesis |
| 5 | - | 8.40 | 317.2126 | 18-HEPE | Biosynthesis of unsaturated fatty acid |
| 6 | - | 8.87 | 343.2285 | 17-HDoHE | Biosynthesis of unsaturated fatty acid |
| 7 | - | 10.30 | 301.2178 | Eicosapentaenoic Acid | Biosynthesis of unsaturated fatty acid |
| 8 | - | 0.82 | 191.0199 | Citric acid | Citrate cycle |
| 9 | - | 0.56 | 179.0558 | d-Fructose | Fructose and mannose metabolism |
| 10 | - | 8.57 | 295.2282 | 9-HODE | Linoleic acid metabolism |
| 11 | - | 1.70 | 241.0831 | Equol | Microbial isoflavones biotransformation |
| 12 | - | 2.46 | 181.0509 | Hydroxyphenyllactic acid | Microbial tyrosine biotransformation |
| 13 | - | 3.55 | 283.0826 | p-Cresol glucuronide | Microbial tyrosine biotransformation |
| 14 | - | 0.59 | 195.0506 | Gluconic acid | Pentose phosphate pathway |
| 15 | - | 5.02 | 321.0445 | dTMP | Pyrimidine metabolism |
| 16 | - | 7.77 | 378.2422 | Sphingosine-1-phosphate | Sphingolipid metabolism |
| 17 | - | 2.62 | 190.0543 | 5-Hydroxyindoleacetic acid | Tryptophan metabolism |
| 18 | - | 3.71 | 212.0027 | Indoxylsulfuric acid | Tryptophan metabolism |
| 19 | - | 4.05 | 204.0669 | Indolelactic acid | Tryptophan metabolism |
| 20 | + | 0.61 | 132.0775 | Creatine | Arginine and proline metabolism |
| 21 | + | 0.56 | 175.1212 | l-Arginine | Arginine and proline metabolism |
| 22 | + | 10.46 | 245.2275 | N1-Acetylspermine | Arginine and proline metabolism |
| 23 | + | 8.11 | 516.3067 | Taurocholic acid | Bile acid biosynthesis |
| 24 | + | 0.60 | 161.1364 | Propionylcholine | Glycerophospholipid metabolism |
| 25 | + | 0.50 | 147.1104 | l-Lysine | Lysine degradation |
| 26 | + | 0.79 | 123.0548 | Niacinamide | Nicotinate and nicotinamide metabolism |
| 27 | + | 0.59 | 137.0726 | N-methylnicotinamide | Nicotinate and nicotinamide metabolism |
| 28 | + | 1.69 | 127.0508 | Thymine | Pyrimidine metabolism |
| 29 | + | 5.18 | 345.2066 | 11-Dehydrocorticosterone | Steroid hormone biosynthesis |
| 30 | + | 4.77 | 190.0861 | 3-Indolepropionic acid | Tryptophan metabolism |
| 31 | + | 0.57 | 176.0668 | Indoleacetic acid | Tryptophan metabolism |
| 32 | + | 2.81 | 190.0501 | Kynurenic acid | Tryptophan metabolism |
| 33 | + | 0.97 | 182.0759 | l-Tyrosine | Tyrosine metabolism |
| 34 | + | 0.60 | 118.0875 | l-Valine | Valine, leucine and isoleucine metabolism |
| Metabolite Name | CON | DSS | 5 mg/kg | 10 mg/kg | 15 mg/kg | POS |
|---|---|---|---|---|---|---|
| 8-HETE | 0.433 ± 0.092 | 2.283 ± 0.623 | 1.234 ± 0.415 | 0.568 ± 0.096 * | 1.218 ± 0.332 | 1.496 ± 0.621 |
| Arachidonic Acid | 1.223 ± 0.196 | 0.708 ± 0.079 | 0.972 ± 0.184 | 0.809 ± 0.130 | 0.814 ± 0.117 | 0.633 ± 0.116 |
| Cholic acid | 0.498 ± 0.094 | 0.898 ± 0.107 | 0.673 ± 0.125 | 2.087 ± 0.507 * | 2.192 ± 0.547 * | 0.424 ± 0.164 * |
| Deoxycholic acid | 0.325 ± 0.039 | 0.200 ± 0.027 | 0.145 ± 0.039 | 0.447 ± 0.190 | 0.272 ± 0.079 | 0.197 ± 0.070 |
| 18-HEPE | 0.435 ± 0.115 | 1.883 ± 0.498 | 0.759 ± 0.237 | 0.381 ± 0.085 * | 0.704 ± 0.197 * | 1.249 ± 0.581 |
| 17-HDoHE | 0.559 ± 0.142 | 2.045 ± 0.553 | 0.782 ± 0.353 | 0.195 ± 0.072 * | 0.613 ± 0.167 * | 0.898 ± 0.449 |
| Eicosapentaenoic Acid | 1.421 ± 0.272 | 0.594 ± 0.081 | 0.817 ± 0.164 | 0.729 ± 0.148 | 0.623 ± 0.115 | 0.551 ± 0.109 |
| Citric acid | 5.263 ± 0.925 | 2.982 ± 0.545 | 6.155 ± 1.213 * | 6.280 ± 0.640 * | 5.226 ± 0.726 * | 5.886 ± 0.700 * |
| d-Fructose | 0.326 ± 0.019 | 0.203 ± 0.017 | 0.290 ± 0.034 * | 0.252 ± 0.022 | 0.251 ± 0.048 | 0.240 ± 0.025 |
| 9-HODE | 0.255 ± 0.054 | 1.001 ± 0.281 | 0.354 ± 0.108 * | 0.202 ± 0.041 * | 0.233 ± 0.058 * | 0.309 ± 0.107 * |
| Equol | 0.245 ± 0.018 | 0.186 ± 0.024 | 0.139 ± 0.020 | 0.191 ± 0.023 | 0.140 ± 0.017 | 0.171 ± 0.022 |
| Hydroxyphenyllactic acid | 0.484 ± 0.078 | 0.195 ± 0.048 | 0.496 ± 0.134 | 0.386 ± 0.072 * | 0.444 ± 0.078 * | 0.479 ± 0.094 * |
| p-Cresol glucuronide | 0.221 ± 0.059 | 0.810 ± 0.300 | 0.285 ± 0.069 | 0.280 ± 0.087 | 0.311 ± 0.094 | 0.124 ± 0.024 * |
| Gluconic acid | 0.253 ± 0.034 | 0.072 ± 0.009 | 0.161 ± 0.039 * | 0.121 ± 0.014 * | 0.161 ± 0.031 * | 0.127 ± 0.019 * |
| dTMP | 3.155 ± 0.363 | 1.516 ± 0.384 | 1.543 ± 0.413 | 4.627 ± 0.579 * | 2.014 ± 0.500 | 1.894 ± 0.486 |
| Sphingosine-1-phosphate | 0.645 ± 0.046 | 0.513 ± 0.032 | 0.676 ± 0.083 | 0.744 ± 0.064 * | 0.710 ± 0.060 * | 0.512 ± 0.040 |
| 5-Hydroxyindoleacetic acid | 0.146 ± 0.030 | 0.064 ± 0.015 | 0.098 ± 0.014 | 0.170 ± 0.025 * | 0.094 ± 0.022 | 0.090 ± 0.020 |
| Indoxylsulfuric acid | 7.599 ± 1.098 | 4.970 ± 0.765 | 4.024 ± 0.806 | 8.662 ± 1.511 * | 4.378 ± 0.787 | 4.752 ± 0.856 |
| Indolelactic acid | 0.515 ± 0.091 | 0.164 ± 0.044 | 0.248 ± 0.082 | 0.526 ± 0.038 * | 0.276 ± 0.059 | 0.191 ± 0.048 |
| Creatine | 1.277 ± 0.239 | 0.710 ± 0.147 | 1.061 ± 0.187 | 1.365 ± 0.217 * | 1.159 ± 0.357 | 0.960 ± 0.193 |
| l-Arginine | 0.242 ± 0.035 | 0.162 ± 0.009 | 0.154 ± 0.014 | 0.200 ± 0.023 | 0.153 ± 0.030 | 0.160 ± 0.018 |
| N1-Acetylspermine | 0.046 ± 0.014 | 0.016 ± 0.004 | 0.056 ± 0.019 | 0.076 ± 0.016 * | 0.045 ± 0.009 * | 0.022 ± 0.008 |
| Taurocholic acid | 0.538 ± 0.043 | 0.742 ± 0.108 | 0.453 ± 0.073 * | 0.640 ± 0.088 | 0.532 ± 0.104 | 0.673 ± 0.151 |
| Propionylcholine | 0.148 ± 0.013 | 0.091 ± 0.006 | 0.091 ± 0.009 | 0.090 ± 0.008 | 0.098 ± 0.009 | 0.100 ± 0.008 |
| l-Lysine | 0.086 ± 0.007 | 0.061 ± 0.005 | 0.097 ± 0.011 * | 0.088 ± 0.006 * | 0.089 ± 0.011 * | 0.079 ± 0.005 |
| Niacinamide | 0.642 ± 0.074 | 0.472 ± 0.063 | 0.581 ± 0.055 | 0.444 ± 0.035 | 0.533 ± 0.052 | 0.482 ± 0.043 |
| N-methylnicotinamide | 0.068 ± 0.010 | 0.138 ± 0.031 | 0.221 ± 0.047 | 0.116 ± 0.016 | 0.187 ± 0.028 | 0.152 ± 0.019 |
| Thymine | 0.069 ± 0.005 | 0.044 ± 0.006 | 0.034 ± 0.005 | 0.047 ± 0.008 | 0.034 ± 0.005 | 0.037 ± 0.006 |
| 11-Dehydrocorticosterone | 0.012 ± 0.001 | 0.035 ± 0.005 | 0.058 ± 0.012 | 0.034 ± 0.004 | 0.079 ± 0.016 * | 0.047 ± 0.010 |
| 3-Indolepropionic acid | 0.064 ± 0.007 | 0.032 ± 0.003 | 0.033 ± 0.006 | 0.036 ± 0.008 | 0.021 ± 0.002 * | 0.047 ± 0.007 |
| Indoleacetic acid | 0.041 ± 0.004 | 0.028 ± 0.003 | 0.037 ± 0.003 | 0.040 ± 0.004 * | 0.041 ± 0.005 * | 0.036 ± 0.005 |
| Kynurenic acid | 0.073 ± 0.016 | 0.022 ± 0.005 | 0.058 ± 0.013 * | 0.065 ± 0.009 * | 0.050 ± 0.009 * | 0.065 ± 0.009 * |
| l-Tyrosine | 3.308 ± 0.338 | 1.742 ± 1.344 | 2.024 ± 0.302 | 3.071 ± 0.326 * | 2.057 ± 0.238 | 2.665 ± 0.495 |
| l-Valine | 1.305 ± 0.107 | 0.951 ± 0.082 | 1.072 ± 0.176 | 1.329 ± 0.096 * | 1.184 ± 0.221 | 1.157 ± 0.196 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, L.; Xiao, H.-t.; Mu, H.-x.; Huang, T.; Lin, Z.-s.; Zhong, L.L.D.; Zeng, G.-z.; Fan, B.-m.; Lin, C.-y.; Bian, Z.-x. Magnolol, a Natural Polyphenol, Attenuates Dextran Sulfate Sodium-Induced Colitis in Mice. Molecules 2017, 22, 1218. https://doi.org/10.3390/molecules22071218
Zhao L, Xiao H-t, Mu H-x, Huang T, Lin Z-s, Zhong LLD, Zeng G-z, Fan B-m, Lin C-y, Bian Z-x. Magnolol, a Natural Polyphenol, Attenuates Dextran Sulfate Sodium-Induced Colitis in Mice. Molecules. 2017; 22(7):1218. https://doi.org/10.3390/molecules22071218
Chicago/Turabian StyleZhao, Ling, Hai-tao Xiao, Huai-xue Mu, Tao Huang, Ze-si Lin, Linda L. D. Zhong, Guang-zhi Zeng, Bao-min Fan, Cheng-yuan Lin, and Zhao-xiang Bian. 2017. "Magnolol, a Natural Polyphenol, Attenuates Dextran Sulfate Sodium-Induced Colitis in Mice" Molecules 22, no. 7: 1218. https://doi.org/10.3390/molecules22071218
APA StyleZhao, L., Xiao, H.-t., Mu, H.-x., Huang, T., Lin, Z.-s., Zhong, L. L. D., Zeng, G.-z., Fan, B.-m., Lin, C.-y., & Bian, Z.-x. (2017). Magnolol, a Natural Polyphenol, Attenuates Dextran Sulfate Sodium-Induced Colitis in Mice. Molecules, 22(7), 1218. https://doi.org/10.3390/molecules22071218





