Correction: Zielinski, W., et al. Ionic Liquids as Solvents for Rhodium and Platinum Catalysts Used in Hydrosilylation Reaction. Molecules 2016, 21, 1115
1. Former Table 1
| Ionic Liquid | Catalyst | Yields in Subsequent Cycles (%) 1 | |||||
|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | |||
| [P44414][NTf2] 1 | K2PtCl4 | 98.45 | 99.60 | 98.68 | 98.71 | 98.71 | |
| K2PtCl6 | 70.26 | 58.65 | 13.09 | 10.87 | 9.56 | ||
| Pt(PPh2)2Cl2 | 100.00 | 100.00 | 100.00 | 95.25 | 87.74 | ||
| Pt(PPh3)4 | 99.82 | 99.22 | 99.13 | 22.40 | 4.77 | ||
| [RhCl(PPh3)3] | 98.34 | 11.73 | 6.56 | 4.53 | 3.57 | ||
| Karstedt | 99.27 | 99.12 | 98.95 | 99.19 | 55.41 | ||
| [BuPy][NTf2] 2 | K2PtCl4 | 85.68 | 2.25 | 0.39 | 0.75 | 3.92 | |
| K2PtCl6 | 86.05 | 3.42 | 5.47 | 10.74 | 13.58 | ||
| Pt(PPh2)2Cl2 | 84.22 | 2.30 | 5.23 | 5.68 | 6.38 | ||
| Pt(PPh3)4 | 16.12 | 14.79 | 13.14 | 11.46 | 11.52 | ||
| [RhCl(PPh3)3] | 100.00 | 12.22 | 9.56 | 2.58 | 8.91 | ||
| Karstedt | 100.00 | 80.09 | 16.02 | 17.37 | 9.44 | ||
| [BMMIM][NTf2] 3 | K2PtCl4 | 60.26 | 55.68 | 15.15 | 16.75 | 17.40 | |
| K2PtCl6 | 29.35 | 28.69 | 27.45 | 30.26 | 33.48 | ||
| Pt(PPh2)2Cl2 | 100.00 | 100.00 | 86.24 | 85.84 | 59.07 | ||
| Pt(PPh3)4 | 86.39 | 85.46 | 84.81 | 84.52 | 85.18 | ||
| [RhCl(PPh3)3] | 98.99 | 97.82 | 98.71 | 99.27 | 5.44 | ||
| Karstedt | 98.30 | 45.89 | 35.15 | 34.05 | 34.12 | ||
| [S222][NTf2] 4 | K2PtCl4 | 86.03 | 85.17 | 84.93 | 86.35 | 84.41 | |
| K2PtCl6 | 87.80 | 83.62 | 86.47 | 85.03 | 84.57 | ||
| Pt(PPh2)2Cl2 | 85.52 | 85.43 | 85.66 | 85.77 | 86.30 | ||
| Pt(PPh3)4 | 90.00 | 90.00 | 85.00 | 72.00 | 67.00 | ||
| [RhCl(PPh3)3] | 94.00 | 97.00 | 89.00 | 62.00 | 7.00 | ||
| Karstedt | 86.03 | 85.17 | 0.00 | 0.00 | 0.00 | ||
| [AllPy][NTf2] 5 | K2PtCl4 | 58.00 | 47.00 | 14.00 | 3.00 | 1.00 | |
| K2PtCl6 | 91.00 | 66.00 | 31.00 | 10.00 | 1.00 | ||
| Pt(PPh2)2Cl2 | 47.00 | 32.00 | 23.00 | 14.00 | 9.00 | ||
| Pt(PPh3)4 | 39.00 | 22.00 | 13.00 | 3.00 | 1.00 | ||
| [RhCl(PPh3)3] | 88.00 | 0.00 | 0.00 | x | x | ||
| Karstedt | 97.15 | 93.70 | 57.58 | 6.08 | 0.00 | ||
| [diAllMIM][NTf2] 6 | K2PtCl4 | 1.00 | 0.00 | 0.00 | x | x | |
| K2PtCl6 | 0.00 | 0.00 | 0.00 | x | x | ||
| Pt(PPh2)2Cl2 | 15.00 | 11.00 | 8.00 | 9.00 | 2.00 | ||
| Pt(PPh3)4 | 3.00 | 3.00 | 3.00 | 0.00 | 0.00 | ||
| [RhCl(PPh3)3] | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | ||
| Karstedt | 16.00 | 9.00 | 6.00 | 2.00 | 0.00 | ||
| [AlldiMIM][NTf2] 7 | K2PtCl4 | 10.00 | 8.00 | 2.00 | 0.00 | 0.00 | |
| K2PtCl6 | 2.00 | 5.00 | 3.00 | 0.00 | 0.00 | ||
| Pt(PPh2)2Cl2 | 35.00 | 27.00 | 20.00 | 14.00 | 11.00 | ||
| Pt(PPh3)4 | 30.00 | 25.00 | 19.00 | 8.00 | 0.00 | ||
| [RhCl(PPh3)3] | 78.00 | 18.00 | 2.00 | 0.00 | 0.00 | ||
| Karstedt | 30.00 | 15.00 | 8.00 | 3.00 | 0.00 | ||
2. New Table 1
| Ionic Liquid | Catalyst | Yields in Subsequent Cycles (%) 1 | ||||
|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | ||
| [P44414][NTf2] 1 | K2PtCl4 | 98.34 | 11.73 | 6.56 | 4.53 | 3.57 |
| K2PtCl6 | 70.26 | 58.65 | 13.09 | 10.87 | 9.56 | |
| Pt(PPh3)2Cl2 | 100.00 | 100.00 | 100.00 | 95.25 | 87.74 | |
| Pt(PPh3)4 | 99.82 | 99.22 | 99.13 | 22.40 | 4.77 | |
| [RhCl(PPh3)3] | 98.45 | 99.60 | 98.68 | 98.71 | 98.71 | |
| Karstedt | 99.27 | 99.12 | 98.95 | 99.19 | 55.41 | |
| [BuPy][NTf2] 2 | K2PtCl4 | 85.68 | 2.25 | 0.39 | 0.75 | 3.92 |
| K2PtCl6 | 86.05 | 3.42 | 5.47 | 10.74 | 13.58 | |
| Pt(PPh3)2Cl2 | 84.22 | 2.30 | 5.23 | 5.68 | 6.38 | |
| Pt(PPh3)4 | 16.12 | 14.79 | 13.14 | 11.46 | 11.52 | |
| [RhCl(PPh3)3] | 100.00 | 12.22 | 9.56 | 2.58 | 8.91 | |
| Karstedt | 100.00 | 80.09 | 16.02 | 17.37 | 9.44 | |
| [BMMIM][NTf2] 3 | K2PtCl4 | 60.26 | 55.68 | 15.15 | 16.75 | 17.40 |
| K2PtCl6 | 29.35 | 28.69 | 27.45 | 30.26 | 33.48 | |
| Pt(PPh3)2Cl2 | 100.00 | 100.00 | 86.24 | 85.84 | 59.07 | |
| Pt(PPh3)4 | 86.39 | 85.46 | 84.81 | 84.52 | 85.18 | |
| [RhCl(PPh3)3] | 98.99 | 97.82 | 98.71 | 99.27 | 5.44 | |
| Karstedt | 98.30 | 45.89 | 35.15 | 34.05 | 34.12 | |
| [S222][NTf2] 4 | K2PtCl4 | 86.03 | 85.17 | 84.93 | 86.35 | 84.41 |
| K2PtCl6 | 87.80 | 83.62 | 86.47 | 85.03 | 84.57 | |
| Pt(PPh3)2Cl2 | 85.52 | 85.43 | 85.66 | 85.77 | 86.30 | |
| Pt(PPh3)4 | 90.00 | 90.00 | 85.00 | 72.00 | 67.00 | |
| [RhCl(PPh3)3] | 94.00 | 97.00 | 89.00 | 62.00 | 7.00 | |
| Karstedt | 86.03 | 85.17 | 0.00 | 0.00 | 0.00 | |
| [AllPy][NTf2] 5 | K2PtCl4 | 58.00 | 47.00 | 14.00 | 3.00 | 1.00 |
| K2PtCl6 | 91.00 | 66.00 | 31.00 | 10.00 | 1.00 | |
| Pt(PPh2)2Cl2 | 47.00 | 32.00 | 23.00 | 14.00 | 9.00 | |
| Pt(PPh3)4 | 39.00 | 22.00 | 13.00 | 3.00 | 1.00 | |
| [RhCl(PPh3)3] | 88.00 | 0.00 | 0.00 | x | x | |
| Karstedt | 97.15 | 93.70 | 57.58 | 6.08 | 0.00 | |
| [diAllMIM][NTf2] 6 | K2PtCl4 | 1.00 | 0.00 | 0.00 | x | x |
| K2PtCl6 | 0.00 | 0.00 | 0.00 | x | x | |
| Pt(PPh3)2Cl2 | 15.00 | 11.00 | 8.00 | 9.00 | 2.00 | |
| Pt(PPh3)4 | 3.00 | 3.00 | 3.00 | 0.00 | 0.00 | |
| [RhCl(PPh3)3] | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | |
| Karstedt | 16.00 | 9.00 | 6.00 | 2.00 | 0.00 | |
| [AlldiMIM][NTf2] 7 | K2PtCl4 | 10.00 | 8.00 | 2.00 | 0.00 | 0.00 |
| K2PtCl6 | 2.00 | 5.00 | 3.00 | 0.00 | 0.00 | |
| Pt(PPh3)2Cl2 | 35.00 | 27.00 | 20.00 | 14.00 | 11.00 | |
| Pt(PPh3)4 | 30.00 | 25.00 | 19.00 | 8.00 | 0.00 | |
| [RhCl(PPh3)3] | 78.00 | 18.00 | 2.00 | 0.00 | 0.00 | |
| Karstedt | 30.00 | 15.00 | 8.00 | 3.00 | 0.00 | |
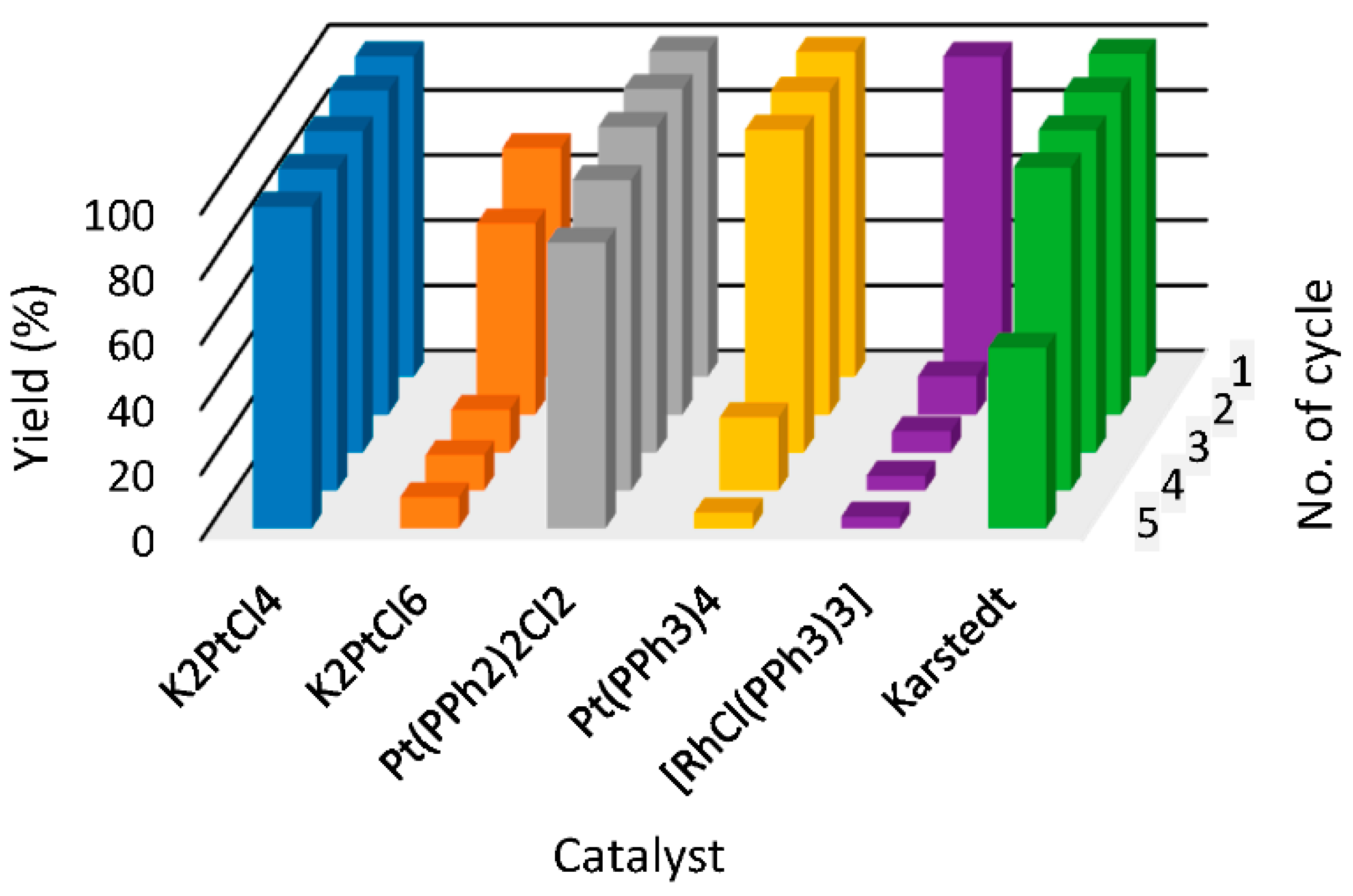
3. Former Figure 3
4. New Figure 3
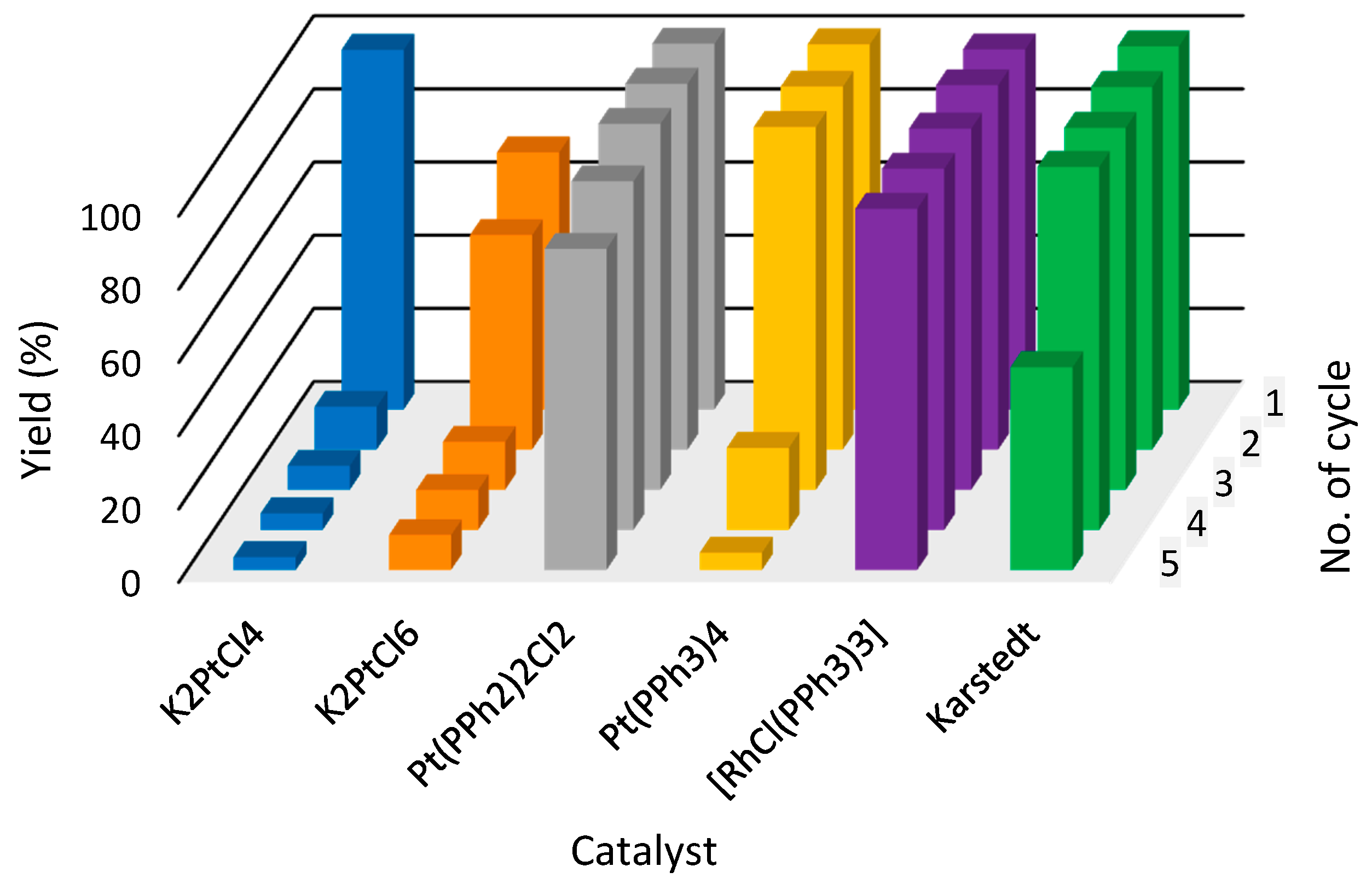
- On page 3, lines 32–34, the sentence “[P44414][NTf2] (1) IL system shows satisfying yields close to 100% throughout the whole five reaction cycles for catalysts such as K2PtCl4, Pt(PPh2)2Cl2 and Karstedt catalyst (100% yield up to fourth cycle)” should be replaced with “[P44414][NTf2] (1) IL system shows satisfying yields close to 100% throughout the whole five reaction cycles for catalysts such as [RhCl(PPh3)3], Pt(PPh3)2Cl2 and Karstedt catalyst (100% yield up to fourth cycle)”.
- On page 3, lines 38 and 39, the sentence “The least effective catalyst for IL (1) is rhodium catalyst for which a major drop in yield was observed after the first reaction cycle” should be replaced with “The least effective catalyst for IL (1) is platinum catalyst, K2PtCl4, for which a major drop in yield was observed after the first reaction cycle”.
- On page 7, lines 10–12, the sentence “[the] most efficient IL systems for hydrosilylation reaction were [P44414][[NTf2] (1)/K2PtCl4 and [P44414][NTf2] (1)/Pt(PPh2)2 for which yields after a fifth cycle were maintained at a level of more than 80%.” should be replaced with “The most efficient IL systems for hydrosilylation reaction were [P44414][[NTf2] (1)/[RhCl(PPh3)3] and [P44414][NTf2] (1)/Pt(PPh3)4, for which yields after a fifth cycle were maintained at a level of more than 80%”.
- On page 7, lines 15 and 16, the statement “which in four out of seven tested IL systems shows [an] immediate drop of the catalyst activity when being recycled for the first time.” should be replaced with “which in three out of seven tested IL systems shows an immediate drop of the catalyst activity when being recycled for the first time”.
Reference
- Zielinski, W.; Kukawka, R.; Maciejewski, H.; Smiglak, M. Ionic Liquids as Solvents for Rhodium and Platinum Catalysts Used in Hydrosilylation Reaction. Molecules 2016, 21, 1115. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zielinski, W.; Kukawka, R.; Maciejewski, H.; Smiglak, M. Correction: Zielinski, W., et al. Ionic Liquids as Solvents for Rhodium and Platinum Catalysts Used in Hydrosilylation Reaction. Molecules 2016, 21, 1115. Molecules 2017, 22, 1203. https://doi.org/10.3390/molecules22071203
Zielinski W, Kukawka R, Maciejewski H, Smiglak M. Correction: Zielinski, W., et al. Ionic Liquids as Solvents for Rhodium and Platinum Catalysts Used in Hydrosilylation Reaction. Molecules 2016, 21, 1115. Molecules. 2017; 22(7):1203. https://doi.org/10.3390/molecules22071203
Chicago/Turabian StyleZielinski, Witold, Rafal Kukawka, Hieronim Maciejewski, and Marcin Smiglak. 2017. "Correction: Zielinski, W., et al. Ionic Liquids as Solvents for Rhodium and Platinum Catalysts Used in Hydrosilylation Reaction. Molecules 2016, 21, 1115" Molecules 22, no. 7: 1203. https://doi.org/10.3390/molecules22071203
APA StyleZielinski, W., Kukawka, R., Maciejewski, H., & Smiglak, M. (2017). Correction: Zielinski, W., et al. Ionic Liquids as Solvents for Rhodium and Platinum Catalysts Used in Hydrosilylation Reaction. Molecules 2016, 21, 1115. Molecules, 22(7), 1203. https://doi.org/10.3390/molecules22071203







