Abstract
A phytochemical study on the aerial parts of Mikania micrantha led to the isolation of two new phenolic compounds, benzyl 5-O-β-d-glucopyranosyl-2,5-dihydroxybenzoate (1) and (7S,8R)-threo-dihydroxydehydrodiconiferyl alcohol 9-acetate (2), together with twelve known compounds, benzyl 2-O-β-d-glucopyranosyl-2,6-dihydroxybenzoate (3), 4-allyl-2,6-dimethoxyphenol glucoside (4), (+)-isolariciresinol (5), icariol A2 (6), 9,10-dihydroxythymol (7), 8,9,10-trihydroxythymol (8), caffeic acid (9), p-coumaric acid (10), ethyl protocatechuate (11), procatechuic aldehyde (12), 4-hydroxybenzoic acid (13), and hydroquinone (14). Their structures were elucidated on the basis of extensive spectroscopic analysis. Except 8 and 9, all the other compounds were isolated from this plant species for the first time. The antioxidant activity of those isolated compounds were evaluated using three different assays. Compounds 1, 2, 3, 9, 10, 13, and 14 demonstrated significant 2,2′-azinobis-(3-ethylbenzthiazoline-6-sulphonic acid) (ABTS) free radical cation scavenging activity ranging from SC50 0.31 to 4.86 µM, which were more potent than l-ascorbic acid (SC50 = 10.48 µM). Compounds 5, 9, 11, and 12 exhibited more potent 1,1-diphenyl-2-picrylhydrazyl (DPPH) radical scavenging activity (SC50 = 16.24–21.67 µM) than l-ascorbic acid (39.48 µM). Moreover, the ferric reducing antioxidant power (FRAP) of compounds 2, 5, 9, and 11 were discovered to be also comparable to or even more potent than l-ascorbic acid.
1. Introduction
Mikania micrantha H. B. K., belonging to the Asteraceae family, is a fast-growing perennial creeping vine indigenous to Central and South America. This plant is known as an invasive species in Southeast Asia and the Pacific region, including southern China [1]. The rapid spread of M. micrantha in South China has caused great damage to agriculture and forestry, as well as the ecological balance in invaded areas [1]. M. micrantha has long been used as a traditional herbal medicine in Jamaica to treat skin itches and athlete’s foot [2]. Previous phytochemical studies have reported some structurally diverse chemicals from this plant, including sesquiterpene lactones, flavonoids, steroids, diterpene glucosides, and phenolic compounds, some of which have shown significant bioactivities [3,4,5,6,7,8,9,10,11,12]. Our recent study on this plant also revealed a group of rare C-9 hydroxylated ent-kaurene diterpene glucosides [13]. During our ongoing phytochemical research on invasive plants in China [14,15,16,17,18,19], two new (1 and 2) and twelve known (3–14) phenolic compounds (Figure 1), were further isolated from the aerial parts of M. micrantha. Herein, we report the isolation and structure elucidation of these compounds, as well as their antioxidant activity.
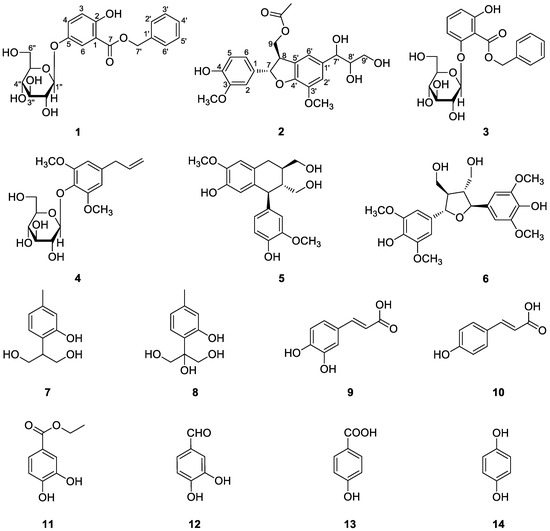
Figure 1.
Chemical structures of compounds 1−14.
2. Results and Discussion
Compound 1 was obtained as a colorless syrup and deduced to have the molecular formula C20H22O9 from its HR-ESI-MS data, m/z 441.0971 [M + Cl]− (calcd for C20H22O9Cl−, 441.0958), which required ten degrees of unsaturation. The 1H and 13C-NMR spectra of 1 (Table 1), coupled with HSQC analysis, indicated twenty carbons, including two methylenes, thirteen methines, and five quaternary carbons including one carboxyl group at δC 168.1 (C-7). The signals of δH 4.69 (1H, d, J = 7.6 Hz, H-1′′) and δC 60.6 (C-6′′), 69.6 (C-4′′), 73.2 (C-2′′), 77.0 (C-3′′), 76.5 (C-5′′), and 101.9 (C-1′′) indicated the presence of a β-d-glucopyranosyl moiety in the molecule. The presence of a benzyl moiety in the molecule was suggested by the proton signals at δH 7.49 (2H, m, H-2′, 6′), 7.42 (2H, m, H-3′, 5′), 7.37 (1H, m, H-4′), and 5.37 (2H, m, H-7′), and further supported by the carbon signals at δC 128.1 (C-2′, 6′), 128.6 (C-3′, 5′), 128.3 (C-4′), and 66.6 (C-7′). Besides, the presence of an ABX spin system was ascertained by the characteristic proton signals of two doublets at δH 6.94 (1H, d, J = 9.0 Hz, H-3) and 7.44 (1H, d, J = 3.1 Hz, H-6), a double doublet at δH 7.28 (1H, dd, J = 9.0, 3.1 Hz, H-4), and further confirmed by the 13C-NMR and HSQC spectra, which revealed three aromatic quaternary carbons at δC 112.9 (C-1), 155.3 (C-2), and 149.9 (C-5), and three aromatic methine carbons at δC 118.3 (C-3), 125.2 (C-4), and 117.0 (C-6). After a more carefully study of the aforementioned NMR data and taking into account the proposed molecular formula, it could be deduced that a free hydroxyl (OH) group would exist in the molecule. In the HMBC spectrum (Figure 2), correlation signals from δH 10.14 (OH-2) to δC 112.9 (C-1), 118.3 (C-3), and 155.3 (C-2) was observed, which revealed the position of the hydroxyl group at C-2. The observation of significant HMBC correlation from δH 4.69 (H-1′′) to δC 149.9 (C-5) verified the location of the glucopyranosyl moiety at C-5. The HMBC correlations from δH 6.94 (H-3) and 7.44 (H-6) to δC 168.1 (C-7) proved the connection of the carboxyl group to C-1. In addition, the ester bond linkage of C-7 with C-7′ was supported by the observation of significant HMBC correlation from δH 5.37 (H-7′) to δC 168.1 (C-7). Therefore, compound 1 was established as benzyl 5-O-β-d-glucopyranosyl-2,5-dihydroxybenzoate.

Table 1.
The 1H (500 MHz) and 13C (125 MHz) nuclear magnetic resonance (NMR) data of compounds 1 and 2.
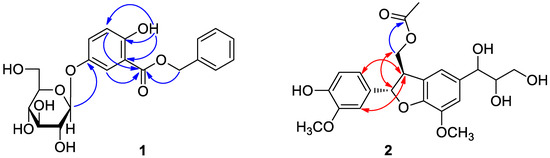
Figure 2.
Key HMBC (blue arrows) and NOE (red arrows) correlations of 1 and 2.
Compound 2, obtained as a colorless oil, was determined to have the molecular formula C22H26O9 according to its HR-ESI-MS data, m/z 457.1467 [M + Na]+ (calcd for C22H26O9Na+, 457.1469), which required ten degrees of unsaturation. The 1H-NMR spectrum (Table 1) showed signals of an acetoxy methyl at δH 2.02 (3H, s), two methoxy groups at δH 3.83 (3H, s) and 3.89 (3H, s), and five aromatic protons. The 13C-NMR spectrum (Table 1), coupled with HSQC analysis, exhibited the signals of twenty-two carbons in total, comprising three methyls, two oxymethylenes [δC 64.3 (C-9′) and 66.7 (C-9)], nine methines (including five aromatic methanes), and eight quaternary carbons [including a carboxyl group at δC 172.7 and seven aromatic quaternary carbons]. Detailed analysis of the NMR data indicated that compound 2 closely resembled threo-dihydroxydehydrodiconiferyl alcohol [20,21,22], with the only difference of the hydroxyl group at C-9 being replaced by an acetoxy group in 2. This deduction was consistent with the molecular formula and in accord with the significant HMBC correlations (Figure 2) from δH 4.33 and 4.44 (C-9) to δC 172.7. The relative configuration of H-7 and H-8 was determined to be trans, as supported by the NOE correlations of H-7/H-9 and H-8/H-6, H-2 (Figure 2). The absolute configuration of C-7 and C-8 in 2 was assigned to be S and R based on the positive CD Cotton effects at 292 nm and 240 nm and the negative Cotton effect at 224 nm [23,24]. Furthermore, according to literature reports about that the chemical shifts of the C-7′ and C-8′ and the value of ∆δC8′–C7′ are characteristically different for the threo and erythro isomers, the relative configuration of C-7′ and C-8′ in 2 could then be determined to be threo based on its 13C-NMR data at δC 75.3 (C-7′) and 77.6 (C-8′) with the value of ∆δC8′–C7′ > 2.0 ppm [25,26,27]. Hence, compound 2 was elucidated as (7S,8R)-threo-dihydroxydehydrodiconiferyl alcohol 9-acetate.
The twelve known compounds were identified as benzyl 2-O-β-d-glucopyranosyl-2,6-dihydroxybenzoate (3) [28], 4-allyl-2,6-dimethoxyphenol glucoside (4) [29], (+)-isolariciresinol (5) [30], icariol A2 (6) [31], 9,10-dihydroxythymol (7) [32], 8,9,10-trihydroxythymol (8) [11], caffeic acid (9) [33], p-coumaric acid (10) [34], ethyl protocatechuate (11) [35], procatechuic aldehyde (12) [33], 4-hydroxybenzoic acid (13) [36], and hydroquinone (14) [37] by comparison of their NMR and ESI-MS data with those reported in the literature. All the known compounds, except 8 and 9, were isolated from this plant species for the first time.
All of the isolated compounds were measured for their antioxidant activity by using three different in vitro assays, i.e., ABTS radical cation (ABTS•+) scavenging assay, DPPH radical (DPPH•) scavenging assay, and FRAP assay, with l-ascorbic acid as a reference compound. As shown in Table 2, new compounds 1 and 2, and known compounds 3, 9, 10, 13, and 14 demonstrated ABTS radical cation scavenging activity with SC50 values ranging from 0.31 to 4.86 µM, which were more potent than l-ascorbic acid (SC50 = 10.48 µM). Compounds 5, 9, 11, and 12 exhibited more potent DPPH radical scavenging activity (SC50 = 16.24–21.67 µM) than l-ascorbic acid (SC50 = 39.48 µM). Moreover, the revealed ferric reducing antioxidant power (FRAP) of compounds 2, 5, 9, and 11 were also comparable to or even more potent than the reference compound.

Table 2.
Antioxidant activity of compounds 1–14.
Generally, free radicals and reactive oxygen species (ROS) are formed unceasingly in human body and the normal presence of free radicals can produce beneficial oxidation during physiological events. However, excessive generation of free radicals in human body will bring harmful oxidation to organisms, which is recognized as a leading cause of a variety of chronic diseases such as atherosclerosis, angiocardiopathy and cancer [38,39]. It is well known that natural antioxidants can help prevent oxidation and help regulate immune function. This study, to some extent, indicate that the invasive plant M. micrantha is rich in structurally diverse natural antioxidants, at least in antioxidant phenolic compounds, which are potential functional chemicals beneficial for human health worthy of further investigation.
3. Materials and Methods
3.1. General Experimental Procedures
Nuclear magnetic resonance (NMR) spectra were recorded on a Bruker DRX-500 NMR spectrometer (Bruker Biospin Gmbh, Rheistetten, Germany). Electrospray ionization mass spectrometry (ESI-MS) was measured on a MDS SCIEX API 2000 LC/MS/MS apparatus (Applied Biosystems Inc., Forster, CA, USA). High-resolution (HR) ESI-MS was measured on a Bruker Bio TOF IIIQ spectrometer (Bruker Daltonics, Billerica, MA, USA). Optical rotations were obtained on a Perkin-Elmer Model 341 polarimeter (Perkin-Elmer, Inc., Waltham, MA). UV spectra were acquired on a Perkin-Elmer Lambda 650 UV-Vis spectrometer (Perkin-Elmer, Inc., Waltham, MA, USA). Preparative HPLC was performed with an HPLC system epuipped with a Shimadzu LC-6AD pump and a Shimadzu RID-10A refractive index detector using a Shim-pack PRC-ODS C-18 column (5 µm, 20 mm × 250 mm). Medium pressure liquid chromatography (MPLC) was carried out on a CXTH P3000 instrument (Beijing Chuang Xin Tong Heng Science and Technology Co., Ltd, Beijing, China) equipped with a UV 3000 UV-Vis Detector and a C-18 column (50 µm, 50 mm × 500 mm).
Silica gel (80–100 and 200–300 mesh, Qingdao Haiyang Chemical Co., Qingdao, China), and Sephadex LH-20 (Pharmacia Fine Chemical Co., Ltd., Oppsala, Sweden) were used for open column chromatography (CC). Thin-layer chromatography (TLC) was conducted on precoated silica gel plates (HSGF254, Yantai Jiangyou Silica Gel Development Co., Ltd., Yantai, China) and spot detection was performed by spraying 10% H2SO4 in ethanol, followed by heating. DPPH, ABTS and 2,4,6-Tripyridy-s-triazine (TPTZ) were purchased from Sigma-Aldrich (Shanghai) Trading Co. (Shanghai, China). Phosphate buffered saline (PBS) and l-ascorbic acid were obtained from Life technologies (Thermo Fisher Scientific, Shanghai, China) and Shanghai Boao Biotech Co. (Shanghai, China), respectively.
3.2. Plant Material
The aerial parts of M. micrantha were collected from Guangzhou, China, in July 2014, and identified by Prof. Hong-Feng Chen, South China Botanical Garden, Chinese Academy of Sciences (CAS). A voucher specimen (No. 20140705) was deposited at the Laboratory of Bioorganic Chemistry of the South China Botanical Garden, CAS.
3.3. Extraction and Isolation
Powdered air-dried aerial parts of M. micrantha (18.5 kg) were extracted three times (each time for three days) with 95% EtOH (50 L) at room temperature. The EtOH extract, after concentration in vacuo, was suspended in water (3 L) and then sequentially extracted three times each with petroleum ether (3 L) and EtOAc (3 L) to yield a petroleum ether-soluble fraction (520 g), and an EtOAc-soluble fraction (260 g) after condensation to dryness under vacuum. The EtOAc-soluble fraction was subjected to silica gel column chromatography, eluted with an increasing polarity of CHCl3/MeOH (from 98:2 to 70:30, v/v, each 18 L) to afford fractions E1–E6 after pooling according to their TLC profiles. Fraction E3 (20 g), obtained on elution with CHCl3/MeOH (95:5), was separated by MPLC using a decreasing polarity of MeOH/H2O (35:65–100:0, v/v, each 1 L) system at the flow rate of 8 mL/min to give fractions E3-1–E3-20. Fraction E3-2, obtained from the elution with MeOH/H2O (35:65), was applied on Sephadex LH-20 column chromatography with the elution of CHCl3/MeOH (1:4, v/v), to provide fractions E3-2-1–E3-2-4. E3-2-4 was purified by preparative HPLC with a Shim-pack PRC-ODS C-18 column (5 µm, 20 mm × 250 mm) using 10% acetonitrile in water (v/v) as a mobile phase at the flow rate of 8 mL/min to obtain 12 (9 mg, tR = 78 min) and 6 (3 mg, tR = 110 min). Fraction E3-8 was applied on LH-20 CC with the elution of CHCl3/MeOH (1:4, v/v) to furnish 5 (5 mg), and 10 (9 mg). Fraction E4 (18.8 g), obtained on elution with CHCl3/MeOH (95:5), was separated by MPLC using a decreasing polarity of MeOH/H2O (35:65–100:0, v/v, each 1 L) system at the flow rate of 8 mL/min to give fractions E4-1–E4-18. Fraction E4-11, obtained from the elution with MeOH/H2O (45:55), was applied on Sephadex LH-20 column chromatography with the elution of CHCl3/MeOH (1:4, v/v), to provide fractions E4-11-1–E4-11-6. The fraction E4-11-3 was further purified by preparative HPLC with a Shim-pack PRC-ODS C-18 column (5 µm, 20 mm × 250 mm) using 16% acetonitrile in water (v/v) as a mobile phase at the flow rate of 8 mL/min to obtain 2 (2 mg, tR = 78 min). Fraction E4-6 was applied on LH-20 CC with the elution of CHCl3/MeOH (1:4, v/v) to furnish 7 (4 mg), 8 (18 mg), and 13 (27 mg). Fraction E4-13 was applied on LH-20 CC with the elution of CHCl3/MeOH (1:4, v/v) and then purified by HPLC using 16% acetonitrile in water (v/v) as mobile phase at 8 mL/min to furnish 4 (10 mg, tR = 90 min) and 3 (13 mg, tR = 100 min). Fraction E5 (18.6 g), obtained on elution with CHCl3/MeOH (90:10), was separated by MPLC using a decreasing polarity of MeOH/H2O (10:90–100:0, v/v , each 1 L) system at the flow rate of 8 mL/min to give fractions E5-1–E5-30. Fraction E5-19, obtained from the elution with MeOH/H2O (65:35), was applied on Sephadex LH-20 column chromatography with the elution of CHCl3/MeOH (1:4, v/v), to provide fractions E5-19-1–E5-19-6. The fraction E5-19-6 was further purified by preparative HPLC with a Shim-pack PRC-ODS C-18 column (5 µm, 20 mm × 250 mm) using 25% acetonitrile in water (v/v) as a mobile phase at the flow rate of 8 mL/min to obtain 1 (10 mg, tR = 65 min). Fraction E5-2, was applied on LH-20 CC with the elution of CHCl3/MeOH (1:4, v/v) to furnish 9 (10 mg), 11 (5 mg), and 14 (3 mg).
Benzyl 5-O-β-d-glucopyranosyl-2,5-dihydroxybenzoate (1): Colorless syrup; [α −22.50 (c 0.68, C5H5N); UV (C5H5N) λmax nm (log ε) 251 (4.16), 257 (4.19); HR-ESI-MS m/z 441.0971 [M + Cl]− (calcd for C20H22ClO9−, 441.0958); 1H-NMR (500 MHz) and 13C-NMR (125 MHz) data in DMSO-d6, see Table 1.
(7S,8R)-threo-Dihydroxydehydrodiconiferyl alcohol 9-acetate (2): Colorless oil; [α + 45.71 (c 0.07, MeOH); UV (MeOH) λmax nm (log ε) 282 (3.70); HR-ESI-MS m/z 457.1467 [M + Na]+ (calcd for C22H26NaO9+, 457.1469); 1H-NMR (500 MHz) and 13C-NMR (125 MHz) data in CD3OD, see Table 1.
3.4. Antioxidant Activity Evaluation
3.4.1. ABTS Radical Cation Scavenging Assay
The ABTS radical cation (ABTS•+) scavenging activity of the isolated compounds was evaluated following the procedures as previously described [40]. Briefly, potassium persulfate was added to 7 mM of ABTS•+, and the mixture was allowed to stand in the dark at room temperature for 12–16 h before use. The ABTS•+ solution was diluted with phosphate-buffered saline (PBS, pH 7.4) to provide an absorbance of 0.70 ± 0.02 at 734 nm. The diluted ABTS•+ solution (190 μL) was added to sample fractions (10 μL) in DMSO at different concentrations. Each treatment was conducted in triplicate. After a mixing time of 10 s and an incubation period of 6 min at 37 °C in the dark, the absorbance in each well was read at 415 nm on a Genios microplate reader (Tecan). l-Ascorbic acid was used as a positive control. The inhibitory rates of ABTS•+ were calculated according to the following formula: ABTS scavenging rate (%) = [1 − (absorbance of compound − absorbance of blank)/absorbance of control] × 100. SC50 values were calculated and expressed as means ± SD in micromolar.
3.4.2. DPPH Radical Scavenging Assay
Scavenging activity of the compounds towards DPPH radicals was carried out by the method as previously described [40]. DPPH radical solution was freshly prepared with MeOH to 0.1 mM. Test compounds were dissolved in DMSO and diluted two-fold to six serial concentrations. The DPPH solution (180 μL) and sample solution (20 μL) were mixed in 96-well plates. l-Ascorbic acid was dissolved in methanol and used as a positive reference. The control group contained DMSO instead of the compound solution, and the blank group contained methanol in place of the DPPH solution. Each treatment was performed in quadruplicate. The plates were incubated at 37 °C for 30 min in the dark. The absorbance in each well was read at 515 nm on a Genios microplate reader (Tecan). The inhibitory rates of DPPH radicals were calculated according to the formula: DPPH scavenging rate (%) = [1 − (absorbance of compound − absorbance of blank)/absorbance of control] × 100. SC50 values (the concentrations required to scavenge 50% DPPH radicals present in the test solution) were calculated and expressed as means ± SD in micromolar.
3.4.3. FRAP Assay
Ferric reducing ability of the compounds was conducted according to the procedures as previously described [40]. FRAP reagent was made freshly by mixing 300 mM acetate buffer (pH 3.6), 10 mM TPTZ solution in 40 mM hydrochloric acid, and 20 mM aqueous ferric chloride (FeCl3) solution in a 10:1:1 (v/v) ratio. The TPTZ solution was prepared on the same day. Test compounds were dissolved in methanol and diluted 2-fold to six concentrations. Twenty microliters of the compound solution and 180 μL of FRAP reagent were mixed in 96-well plates. l-Ascorbic acid was dissolved in methanol and used as a positive reference. Each treatment was conducted in quadruplicate. The plates were incubated at 37 °C for 30 min in the dark. The absorbance of the product (ferrous TPTZ complex) in each well was read at 595 nm using a Genios microplate reader (Tecan Group, Mannedorf, Switzerland). One milliliter of ferrous sulfate (FeSO4) at six different concentrations and 1 mL of 10 mM TPTZ and 10 mL of 300 mM acetate buffer (pH 3.6) were used for a calibration curve. FRAP values were calculated and expressed as means ± the standard deviation (SD) in millimoles of Fe (II) per gram.
4. Conclusions
Fourteen phenolic compounds, including two new ones—1 and 2, were isolated from the aerial parts of M. micrantha. Their structures were identified by analysis of their spectroscopic data. Except 8 and 9, all the other compounds were isolated from this plant species for the first time. Bioassays revealed that seven compounds demonstrated good ABTS radical cation scavenging activity more potent than l-ascorbic acid, and four compounds exhibited more potent DPPH radical scavenging activity than l-ascorbic acid. Moreover, the ferric-reducing antioxidant power (FRAP) of four compounds were comparable to or even more potent than L-ascorbic acid. This study indicates that the invasive plant M. micrantha is rich in structurally diverse phenolic compounds with functional potential beneficial for human health and is worthy of further investigation.
Supplementary Materials
The following are available online: HR-ESI-MS and NMR spectra data of compounds 1 and 2 as supporting information.
Acknowledgments
This research was supported by the National Natural Science Foundation of China (31270406, 31470422 and 31500291), the Natural Science Foundation of Guangdong Province (2014A030313742), and the Science and Technology Project of Guangdong Province (2014B020206003).
Author Contributions
L.-M. Dong and X.-C. Jia performed the isolation and structural elucidation of the chemicals and the bioassay experiments. Q.-W. Luo, Q. Zhang, B. Luo, W.-B. Liu, and X. Zhang contributed the isolation of the chemicals and the bioactive assay. Q.-L. Xu and J.-W. Tan designed and organized the study. All authors approved the final version manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Zhang, L.Y.; Ye, W.H.; Cao, H.L.; Feng, H.L. Mikania micrantha H.B.K. in China-an overview. Weed Res. 2004, 44, 42–49. [Google Scholar] [CrossRef]
- Facey, P.C.; Pascoe, K.O.; Porter, R.B.; Jones, A.D. Investigation of plants used in Jamaican folk medicine for anti-bacterial activity. J. Pharm. Pharmacol. 1999, 51, 1455–1460. [Google Scholar] [CrossRef] [PubMed]
- Herz, W.; Srinivasan, A.; Kalyanaraman, P.S. Mikanokryptin, a new guianolide from Mikania. Phytochemistry 1975, 14, 233–237. [Google Scholar] [CrossRef]
- Nicollier, G.; Thompson, A.C. Essential oil and terpenoids of Mikania micrantha. Phytochemistry 1981, 20, 2587–2588. [Google Scholar] [CrossRef]
- Boeker, R.; Jakupovic, J.; Bohlmann, F.; Schmeda-Hirschmann, G. Germacra-1,10Z,4E-dien-12,8α-olides from Mikania micrantha. Planta Med. 1987, 53, 105–106. [Google Scholar] [CrossRef] [PubMed]
- Cuenca, M.D.R.; Bardon, A.; Catalan, C.A.N. Sesquiterpene lactones from Mikania micrantha. J. Nat. Prod. 1988, 51, 625–626. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.J.; Ye, W.H.; Wu, P.; Lin, L.D.; Wei, X.Y. New sesquiterpene lactones from Mikania micrantha. J. Nat. Prod. 2004, 67, 734–736. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.Y.; Huang, H.J.; Wu, P.; Cao, H.L.; Ye, W.H. Phenolic constituents from Mikania micrantha. Biochem. Syst. Ecol. 2004, 32, 1091–1096. [Google Scholar] [CrossRef]
- But, P.P.H.; He, Z.D.; Ma, S.C.; Chan, Y.M.; Shaw, P.C.; Ye, W.C.; Jiang, R.W. Antiviral constituents against respiratory viruses from Mikania micrantha. J. Nat. Prod. 2009, 72, 925–928. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.L.; Xie, H.H.; Xiao, H.L.; Lin, L.D.; Wei, X.Y. Two new ent-kaurene diterpene glucosides from the roots of Mikania micrantha. Phytochem. Lett. 2013, 6, 425–428. [Google Scholar] [CrossRef]
- Xu, Q.L.; Xie, H.H.; Xiao, H.L.; Wei, X.Y. Phenolic constituents from the roots of Mikania micrantha and their allelopathic effects. J. Agric. Food Chem. 2013, 61, 7309–7314. [Google Scholar] [CrossRef] [PubMed]
- Ríos, E.V.; León, A.; Chávez, M.I.; Torres, Y.; Ramírez-Apan, M.T.; Toscano, R.A.; Bravo-Monzón, Á.E.; Espinosa-García, F.J.; Delgado, G. Sesquiterpene lactones from Mikania micrantha and Mikania cordifolia and their cytotoxic and anti-inflammatory evaluation. Fitoterapia 2014, 94, 155–163. [Google Scholar]
- Dong, L.M.; Jia, X.C.; Luo, Q.W.; Peng, Y.M.; Zhang, Q.; Luo, B.; Tan, J.W. Four new ent-kaurene diterpene glucosides from Mikania micrantha. Phytochem. Lett. 2017, 20, 155–159. [Google Scholar] [CrossRef]
- Yan, J.; Bi, H.H.; Liu, Y.Z.; Zhang, M.; Zhou, Z.Y.; Tan, J.W. Phenolic compounds from Merremia umbellate subsp. orientalis and their allelopathic effects on arabidopsis seed germination. Molecules 2010, 15, 8241–8250. [Google Scholar] [PubMed]
- Zhang, M.; Liu, W.X.; Zheng, M.F.; Xu, Q.L.; Wan, F.H.; Wang, J.; Lei, T.; Zhou, Z.Y.; Tan, J.W. Bioactive quinic acid derivatives from Ageratina adenophora. Molecules 2013, 18, 14096–14104. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.Y.; Liu, W.X.; Pei, G.; Ren, H.; Wang, J.; Xu, Q.L.; Xie, H.H.; Wan, F.H.; Tan, J.W. Phenolics from Ageratina adenophora roots and their phytotoxic effects on Arabidopsis thaliana seed germination and seedling growth. J. Agric. Food Chem. 2013, 61, 11792–11799. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Xu, Q.L.; Dong, L.M.; Zhou, Z.Y.; Chen, Y.C.; Zhang, W.M.; Tan, J.W. A new ursane and a new oleanane triterpene acids from the whole plant of Spermacoce latifolia. Phytochem. Lett. 2015, 11, 127–131. [Google Scholar] [CrossRef]
- Ren, H.; Xu, Q.L.; Luo, Y.; Zhang, M.; Zhou, Z.Y.; Dong, L.M.; Tan, J.W. Two new ent-kaurane diterpenoids from Wedelia trilobata (L.) Hitchc. Phytochem. Lett. 2015, 11, 260–263. [Google Scholar] [CrossRef]
- Ren, H.; Xu, Q.L.; Zhang, M.; Dong, L.M.; Zhang, Q.; Luo, B.; Luo, Q.W.; Tan, JW. Bioactive caffeic acid derivatives from Wedelia trilobata. Phytochem. Lett. 2017, 19, 18–22. [Google Scholar] [CrossRef]
- Deyama, T.; Ikawa, T.; Kitagawa, S. The constituents of Eucommia ulmoides Oliv. V. isolation of dihydroxydehydrodiconiferyl alcohol isomers and phenolic compounds. Chem. Pharm. Bull. 1987, 35, 1785–1789. [Google Scholar] [CrossRef]
- Du, Q.Z.; Jerz, G.; Shen, L.Q.; Xiu, L.; Winterhalter, P. Isolation and structure determination of a lignan from the bark of Salix alba. Nat. Prod. Res. 2007, 21, 451–454. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Li, F.; Yang, C.Y.; Khan, A.A.; Liu, X.; Wang, M.K. Neolignans, lignans and glycoside from the fruits of Melia toosendan. Fitoterapia 2014, 99, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Nakanishi, T.; Iida, N.; Inatomi, Y.; Murata, H.; Inada, A.; Murata, J.; Lang, F.A.; Linuma, M.; Tanaka, T. Neolignan and flavonoid glycosides in Juniperus communis var. Depressa. Phytochemistry 2004, 65, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.C.; Hsieh, P.W.; Kuo, Y.H. Neolignan glucosides from Jasminum urophyllum. Phytochemistry 1998, 48, 719–723. [Google Scholar] [CrossRef]
- Ishikawa, T.; Fujimatu, E.; Kitajima, J. Water-soluble constituents of anise: New glucosides of anethole glycol and its related compounds. Chem. Pharm. Bull. 2002, 50, 1460–1466. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.; Wang, S.J.; Liu, M.T.; Gan, M.; Li, S.; Yang, Y.; Wang, Y.; He, W.; Shi, J. Glycosides from the stem bark of Fraxinus sieboldiana. J. Nat. Prod. 2007, 70, 817–823. [Google Scholar] [CrossRef] [PubMed]
- Gan, M.L.; Zhang, Y.L.; Lin, S.; Liu, M.T.; Song, W.X.; Zi, J.C.; Yang, Y.C.; Fan, X.N.; Shi, J.G.; Hu, J.F.; et al. Glycosides from the root of Iodes cirrhosa. J. Nat. Prod. 2008, 71, 647–654. [Google Scholar] [CrossRef] [PubMed]
- D’Abrosca, B.; DellaGreca, M.; Fiorentino, A.; Monaco, P.; Previtera, L.; Simonet, A.M.; Zarrelli, A. Potential allelochemicals from Sambucus nigra. Phytochemistry 2001, 58, 1073–1081. [Google Scholar] [CrossRef]
- Miyase, T.; Kuroyanagi, M.; Noro, T.; Ueno, A.; Fukushima, S. Studies on sesquiterpenes from Macroclinidium trilobum Makino. II. Chem. Pharm. Bull. 1985, 33, 4445–4450. [Google Scholar] [CrossRef]
- Jutiviboonsuk, A.; Zhang, H.; Tan, G.T.; Cuiying, M.; Hung, N.V.; Cuong, N.M.; Bunyapraphatsara, N.; Soejarto, D.D.; Fong, H.H.S. Bioactive constituents from roots of Bursera tonkinensis. Phytochemistry 2005, 66, 2745–2751. [Google Scholar] [CrossRef] [PubMed]
- Matsushita, H.; Miyase, T.; Ueno, A. Lignan and terpene glycosides from Epzmedium sagittatum. Phytochemistry 1991, 30, 2025–2027. [Google Scholar] [CrossRef]
- Zdero, C.; Bohlmann, F.; Niemeyer, H.M. Sesquiterpene lactones from Perityle emoryi. Phytochemistry 1990, 29, 891–894. [Google Scholar] [CrossRef]
- Song, C.Z.; Wang, Y.H.; Hua, Y.; Yan, H.; Wu, Z.K.; Du, Z.Z. Chemical constituents of Clematis montana. Chin. J. Nat. Med. 2008, 6, 116–119. [Google Scholar]
- Wang, Z.J.; Zhao, Y.Y.; Wang, B.; Min, A.T.; Chen, Y.Y. Depsides from Prunella vulgaris. Chin. Chem. Lett. 2000, 11, 997–1000. [Google Scholar]
- Baderschneider, B.; Winterhalter, P. Isolation and characterization of novel benzoates, cinnamates, flavonoids, and lignans from riesling wine and screening for antioxidant activity. J. Agric. Food Chem. 2001, 49, 2788–2798. [Google Scholar] [CrossRef] [PubMed]
- Xin, X.L.; Aisa, H.A.; Wang, H.Q. Flavonoids and phenolic compounds from seeds of the Chinese plant Nigella glandulifera. Chem. Nat. Comp. 2008, 44, 368–369. [Google Scholar] [CrossRef]
- Erenler, R.; Sen, O.; Aksit, H.; Demirtas, I.; Yaglioglu, A.S.; Elmastas, M.; Telci, İ. Isolation and identification of chemical constituents from Origanum majorana and investigation of antiproliferative and antioxidant activities. J. Sci. Food Agric. 2016, 96, 822–836. [Google Scholar] [CrossRef] [PubMed]
- Willcox, J.K.; Ash, S.L.; Catignani, G.L. Antioxidants and prevention of chronic disease Crit. Rev. Food Sci. Nutr. 2004, 44, 275–295. [Google Scholar] [CrossRef] [PubMed]
- Rai, S.; Wahile, A.; Mukherjee, K.; Saha, B.P.; Mukherjee, P.K. Antioxidant activity of Nelumbo nucifera (sacred lotus) seeds. J. Ethnopharmacol. 2006, 104, 322–327. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.; Xie, H.H.; Li, S.; Zhang, R.F.; Zhang, M.W.; Wei, X.Y. Flavonoids from the pericarps of Litchi chinensis. J. Agric. Food Chem. 2014, 62, 1073–1078. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Not available. |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).