Analytical Techniques and Pharmacokinetics of Gastrodia elata Blume and Its Constituents
Abstract
:1. Introduction
2. Sample Pretreatment Method
2.1 Organic Solvent Precipitation
2.2. Acid Pretreatment
2.3. Solid Phase Extraction
3. Analytical Methods
3.1. Liquid Chromatography (LC)
3.2. Ultraviolet (UV) Spectroscopy
3.3. Mass Spectrometry (MS)
3.4. MS/MS
3.5. Fluorescence Detection (FLD)
4. Pharmacokinetic Studies
4.1. Absorption
4.1.1. GAS
4.1.2. p-Hydroxybenzyl Alcohol
4.1.3. Parishin
4.1.4. N6-(4-Hydroxybenzyl) Adenine Riboside
4.2. Distribution
4.2.1. GAS
4.2.2. p-Hydroxybenzyl Alcohol
4.3. Metabolism and Biotransformation
4.3.1. Phenols
4.3.2. N6-(4-Hydroxybenzyl) Adenine Riboside
4.4. Excretion
4.5. Influencing Factors
4.5.1. Routes of Administration and Dosage
4.5.2. Compatibility
4.5.3. Food
5. Summary and Perspectives
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hsieh, C.L.; Chiang, S.Y.; Cheng, K.S.; Lin, Y.H.; Tang, N.Y.; Lee, C.J.; Pon, C.Z.; Hsieh, C.T. Anticonvulsive and free radical scavenging activities of Gastrodia elata Bl. in kainic acid-treated rats. Am. J. Chin. Med. 2001, 29, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Jung, T.Y.; Suh, S.I.; Lee, H.; Kim, I.S.; Kim, H.J.; Yoo, H.S.; Lee, S.R. Protective effects of several components of Gastrodia elata on lipid peroxidation in gerbil brain homogenates. Phytother. Res. 2007, 21, 960–964. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Mori, A. Antioxidant and free radical scavenging activities of Gastrodia elata Bl. and Uncaria rhynchophylla (Miq.) Jacks. Neuropharmacology 1992, 31, 1287–1298. [Google Scholar] [CrossRef]
- Liu, J.; Mori, A. Antioxidant and pro-oxidant activities of p-hydroxybenzyl alcohol and vanillin: Effects on free radicals, brain peroxidation and degradation of benzoate, deoxyribose, amino acids and DNA. Neuropharmacology 1993, 32, 659–669. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.J.; Hsieh, C.L.; Su, K.P.; Hou, Y.C.; Chiang, H.M.; Lin, I.H.; Sheen, L.Y. The antidepressant effect of Gastrodia elata B1. on the forced-swimming test in rats. Am. J. Chin. Med. 2008, 36, 95–106. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.C.; Lai, Y.S.; Lin, S.H.; Lu, K.H.; Lin, Y.E.; Panyod, S.; Ho, C.T.; Sheen, L.Y. Anti-depressant effects of Gastrodia elata Blume and its compounds gastrodin and 4-hydroxybenzyl alcohol, via the monoaminergic system and neuronal cytoskeletal remodeling. J. Ethnopharmacol. 2016, 182, 190–199. [Google Scholar] [CrossRef] [PubMed]
- Qiu, F.; Liu, T.T.; Qu, Z.W.; Qiu, C.Y.; Yang, Z.; Hu, W.P. Gastrodin inhibits the activity of acid-sensing ion channels in rat primary sensory neurons. Eur. J. Pharmacol. 2014, 731, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Li, M.; Kang, R.X.; Shi, J.G.; Liu, G.T.; Zhang, J.J. NHBA isolated from Gastrodia elata exerts sedative and hypnotic effects in sodium pentobarbital-treated mice. Pharmacol. Biochem. Behav. 2012, 102, 450–457. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y. The Study of the Sedative-Hypnotic Effects of NHBA and Its Analogue B2 and Their Mechanisms. Ph.D. Thesis, Peking Union Medical College, Beijing, China, 2010. [Google Scholar]
- Ojemann, L.M.; Nelson, W.L.; Shin, D.S.; Rowe, A.O.; Buchanan, R.A. Tian ma, an ancient Chinese herb, offers new options for the treatment of epilepsy and other conditions. Epilepsy Behav. 2006, 8, 376–383. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Miao, B.; Wang, X.C.; Duan, J.H.; Ye, X.; Han, W.J.; Wang, W.T.; Luo, C.; Hu, S.J. Gastrodin Inhibits Allodynia and Hyperalgesia in Painful Diabetic Neuropathy Rats by Decreasing Excitability of Nociceptive Primary Sensory Neurons. PLoS ONE 2012, 7, e396476. [Google Scholar] [CrossRef] [PubMed]
- Jang, Y.W.; Lee, J.Y.; Kim, C.J. Anti-asthmatic activity of phenolic compounds from the roots of Gastrodia elata Bl. Int. Immunopharmacol. 2010, 10, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.N.; Zhang, R.G.; Qiao, Y.T.; Xue, F.; Nie, H.; Zhang, Z.J.; Wang, Y.; Peng, Z.W.; Tan, Q.R. Gastrodin ameliorates depression-like behaviors and up-regulates proliferation of hippocampal-derived neural stem cells in rats: Involvement of its anti-inflammatory action. Behav. Brain Res. 2014, 266, 153–160. [Google Scholar] [CrossRef] [PubMed]
- An, S.J.; Park, S.K.; Hwang, I.K.; Choi, S.Y.; Kim, S.K.; Kwon, O.S.; Jung, S.J.; Baek, N.I.; Lee, H.Y.; Won, M.H.; et al. Gastrodin decreases immunoreactivities of gamma-aminobutyric acid shunt enzymes in the hippocampus of seizure-sensitive gerbils. J. Neurosci. Res. 2003, 71, 534–543. [Google Scholar] [CrossRef] [PubMed]
- Hwang, S.M.; Lee, Y.J.; Kang, D.G.; Lee, H.S. Anti-Inflammatory Effect of Gastrodia elata Rhizome in Human Umbilical Vein Endothelial Cells. Am. J. Chin. Med. 2009, 37, 395–406. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, M.T.; Wu, C.R.; Chen, C.F. Gastrodin and p-hydroxybenzyl alcohol facilitate memory consolidation and retrieval, but not acquisition, on the passive avoidance task in rats. J. Ethnopharmacol. 1997, 56, 45–54. [Google Scholar] [CrossRef]
- Wu, C.R.; Hsieh, M.T.; Huang, S.C.; Peng, W.H.; Chang, Y.S.; Chen, C.F. Effects of Gastrodia elata and its active constituents on scopolamine-induced amnesia in rats. Planta Med. 1996, 62, 317–321. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.R.; Hsieh, M.T.; Liao, J. p-Hydroxybenzyl alcohol attenuates learning deficits in the inhibitory avoidance task: Involvement of serotonergic and dopaminergic systems. Chin. J. Physiol. 1996, 39, 265–273. [Google Scholar] [PubMed]
- Zeng, X.; Zhang, S.M.; Zhang, L.; Zhang, K.P.; Zheng, X.X. A study of the neuroprotective effect of the phenolic glucoside gastrodin during cerebral ischemia in vivo and in vitro. Planta Med. 2006, 72, 1359–1365. [Google Scholar] [CrossRef] [PubMed]
- Kumar, H.; Kim, I.S.; More, S.V.; Kim, B.W.; Bahk, Y.Y.; Choi, D.K. Gastrodin Protects Apoptotic Dopaminergic Neurons in a Toxin-Induced Parkinson’s Disease Model. Evid.-Based Complement. Altern. 2013, 514095. [Google Scholar] [CrossRef] [PubMed]
- Zeng, X.H.; Zhang, Y.; Zhang, S.M.; Zheng, X.X. A microdialysis study of effects of gastrodin on neurochemical changes in the ischemic/reperfused rat cerebral hippocampus. Biol. Pharm. Bull. 2007, 30, 801–804. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.L.; Yang, J.M.; Wang, K.C.; Lee, Y.C.; Lin, Y.; Yang, Y. Gastrodia elata prevents huntingtin aggregations through activation of the adenosine A(2A) receptor and ubiquitin proteasome system. J. Ethnopharmacol. 2011, 138, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.S.; Tian, Z.; Zhang, N.; Han, J.; Guo, H.L.; Zhao, M.G. Protective Effects of Gastrodin Against Autophagy-Mediated Astrocyte Death. Phytother. Res. 2016, 30, 386–396. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.F.; Yin, H.; Bi, H.P.; Zhuang, Y.B.; Liu, T.; Ma, Y.H. De novo biosynthesis of Gastrodin in Escherichia coli. Metab. Eng. 2016, 35, 138–147. [Google Scholar] [CrossRef] [PubMed]
- Sate Pharmacopoeia Committee. Pharmacopoeia of the People’s Republic of China; Sate Pharmacopoeia Committee: Beijing, China, 2015; ISBN 978-7-5067-7337-9. [Google Scholar]
- Chen, P.J.; Sheen, L.Y. Gastrodiae Rhizoma (tiān má): A review of biological activity and antidepressant mechanisms. J. Tradit. Complement. Med. 2011, 1, 31–40. [Google Scholar] [CrossRef]
- Jang, J.H.; Son, Y.; Kang, S.S.; Bae, C.S.; Kim, J.C.; Kim, S.H.; Shin, T.; Moon, C. Neuropharmacological Potential of Gastrodia elata Blume and Its Components. Evid.-Based Complement. Altern. 2015, 309261. [Google Scholar] [CrossRef]
- Zhan, H.D.; Zhou, H.Y.; Sui, Y.P.; Du, X.L.; Wang, W.H.; Dai, L.; Sui, F.; Huo, H.R.; Jiang, T.L. The rhizome of Gastrodia elata Blume—An ethnopharmacological review. J. Ethnopharmacol. 2016, 189, 361–385. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.X.; Han, G.Z.; Chang, Y.L.; Su, C.Y.; Tang, N.Y.; Chen, Y.R. Simultaneous determination of gastrodin and its metabolite by HPLC. Biomed. Chromatogr. 1987, 2, 17–19. [Google Scholar] [CrossRef] [PubMed]
- Ju, X.H.; Shi, Y.; Liu, N.; Guo, D.M.; Cui, X. Determination and pharmacokinetics of gastrodin in human plasma by HPLC coupled with photodiode array detector. J. Chromatogr. B 2010, 878, 1982–1986. [Google Scholar] [CrossRef] [PubMed]
- Cheng, G.; Hao, X.H.; Liu, G.L.; Zou, M.J.; Cui, F.D. Pharmacokinetics of gastrodin in rats. Chin. Pharm. J. 2003, 38, 49–51. [Google Scholar]
- Zhang, W.; Sheng, Y.X.; Zhang, J.L. Determination and pharmacokinetics of gastrodin and p-hydroxybenzylalcohol after oral administration of Gastrodia elata Bl. extract in rats by high-performance liquid chromatography–electrospray ionization mass spectrometric method. Phytomedicine 2008, 15, 844–850. [Google Scholar] [CrossRef] [PubMed]
- Cai, Z.; Hou, S.X.; Li, Y.B.; Zhao, B.B.; Yang, Z.X.; Xu, S.G.; Pu, J.X. Effect of borneol on the distribution of gastrodin to the brain in mice via oral administration. J. Drug Target. 2008, 16, 178–184. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Gong, X.J.; Zhou, X.; Kang, Z.J. Relative bioavailability of gastrodin and parishin from extract and powder of Gastrodiae rhizoma in rat. J. Pharm. Biomed. Anal. 2014, 100, 309–315. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Q.; Yue, P.F.; Wu, B.; Hu, P.Y.; Wu, Z.F.; Yang, M. Pharmacokinetics comparative study of a novel Chinese traditional herbal formula and its compatibility. J. Ethnopharmacol. 2011, 137, 221–225. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.W.; Shen, J.; Li, X.; Xie, H.T.; Wang, J.S.; Luo, J.; Wang, K.D.G.; Liu, Q.W.; Kong, L.Y. Identification and analysis of gastrodin and its five metabolites using ultra fast liquid chromatography electrospray ionization tandem mass spectrometry to investigate influence of multiple-dose and food. J. Chromatogr. A 2014, 1358, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Hu, P.Y.; Yue, P.F.; Zheng, Q.; Yang, M.; Zhang, G.S.; Wu, B.; Liu, D. Pharmacokinetic comparative study of gastrodin after oral administration of Gastrodia elata Bl. extract and its compatibility with the different indigents of Ligusticum chuanxiong Hort. to rats. J. Ethnopharmacol. 2016, 191, 82–86. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.W.; Xie, H.T.; Shen, J.; Chen, A.D.; Sun, H.; Li, H.G.; Pan, X.S. Quantitative analysis of gastrodin in rat plasma using electrospray liquid chromatography/mass spectrometry. Chin. J. Mod. Drug Appl. 2010, 4, 1–3. [Google Scholar]
- Yang, Y.; Du, P.; Chen, Y.; Han, F.M. Study on pharmacokinetics of gastrodin after Tianma extract in rats. Acta Chin. Med. Pharm. 2010, 38, 94–97. [Google Scholar]
- Yang, Y.; Han, F.M.; Du, P.; Chen, Y. Pharmacokinetics of gastrodin from compound Tianma granule in rats. Acta Pharm. Sin. 2010, 45, 484–488. [Google Scholar]
- Ge, Z.H.; Xie, Y.Y.; Liang, Q.L.; Wang, Y.M.; Luo, G.G. Pharmacokinetic Comparative Study of Gastrodin and Rhynchophylline after Oral Administration of Different Prescriptions of Yizhi Tablets in Rats by an HPLC-ESI/MS Method. Evid.-Based Complement. Altern. 2014, 167253. [Google Scholar] [CrossRef]
- Sun, C.C.; Dong, Y.B.; Sun, J.; Zhang, W.M.; Lu, W.W.; Ye, M.L.; Kang, C.Q. Determination of human relative bioavailability of gastrodin using HPLC. Chin. Hosp. Pharm. J. 2008, 28, 1804–1806. [Google Scholar]
- Li, L.L.; Zhang, Z.R.; Gong, T.; He, L.L.; Deng, L. Simultaneous determination of Gastrodin and Ligustrazine hydrochloride in dog plasma by gradient high-performance liquid chromatography. J. Pharm. Biomed. 2006, 41, 1083–1087. [Google Scholar] [CrossRef] [PubMed]
- Lu, G.W.; Zou, Y.J.; Mou, Q.Z. Kinetic aspects of absorption, distribution, metabolism and excretion of 3H-gastrodin in rats. Acta Pharm. Sin. 1985, 20, 167–172. [Google Scholar]
- Cai, Z.; Hou, S.X.; Liu, Z.Q.; Chen, Q.H.; Yang, Z.X. Pharmacokinetics of gastrodigenin in brain tissue of mice after intragastric administration of gastrodin. Chin. Tradit. Herb. Drugs 2009, 40, 389–391. [Google Scholar]
- Wang, Q.; Chen, G.S.; Zeng, S. Pharmacokinetics of Gastrodin in rat plasma and CSF after i.n. and i.v. Int. J. Pharm. 2007, 341, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Chen, G.S.; Zeng, S. Distribution and metabolism of gastrodin in rat brain. J. Pharm. Biomed. 2008, 46, 399–404. [Google Scholar] [CrossRef] [PubMed]
- Lv, Y.F.; Hu, X.; Cheng, W.M.; Nie, Y.L.; Bi, K.S. Determination of gastrodin and ligustrazine hydrochloride in plasma and brain dialysate by LC-tandem MS. Chromatographia 2008, 68, 105–110. [Google Scholar] [CrossRef]
- Tang, C.L.; Wang, L.; Li, J.J.; Liu, X.X.; Cheng, M.C.; Xiao, H.B. Analysis of the metabolic profile of parishin by ultra-performance liquid chromatography/quadrupole-time of flight mass spectrometry. Biomed. Chromatogr. 2015, 29, 1913–1920. [Google Scholar] [CrossRef] [PubMed]
- Peng, D.C.; Wang, H.S.; Qu, C.L.; Xie, L.H.; Wicks, S.M.; Xie, J.H. Ginsenoside Re: Its chemistry, metabolism and pharmacokinetics. Chin. Med. 2012, 7. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.F.; Yang, J.L.; Du, F.F.; Gao, X.M.; Ma, X.T.; Huang, Y.H.; Xu, F.; Niu, W.; Wang, F.Q.; Mao, Y.; et al. Absorption and Disposition of Ginsenosides after Oral Administration of Panax notoginseng Extract to Rats. Drug Metab. Dispos. 2009, 37, 2290–2298. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Yu, L.B.; Xu, G.L.; Zhang, Q.Y.; Yan, X.J.; Wang, Y.R. Pharmacokinetic study on combined application of gastrodin and puerarin in rats. China J. Chin. Mater. Med. 2015, 40, 1179–1184. [Google Scholar]
- Lin, L.C.; Chen, Y.F.; Tsai, T.R.; Tsai, T.H. Analysis of brain distribution and biliary excretion of a nutrient supplement, gastrodin, in rat. Anal. Chim. Acta 2007, 590, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Luo, H.; Gong, Y.; Liu, Z.Q.; Cai, Z. Intestinal absorption characteristics of gastrodigenin in rats. China J. Chin. Mater. Med. 2012, 37, 858–861. [Google Scholar]
- Wang, Q.; Chen, G.S.; Zeng, S. Study on the metabolism of gastrodin in rat brain, liver, kidney and different brain regions homogenate. Chin. J. Mod. Appl. Pharm. 2009, 26, 614–619. [Google Scholar]
- Yang, L.; Wang, X.; Xin, X.; Wang, M.Z. The determination method research of gastrodin in liver and kidney of rats. Lishizhen Med. Mater. Med. Res. 2011, 22, 295–298. [Google Scholar]
- Wang, Q. Study on the Gastrodin and Gastrodigenin Metabolism In Vitro and In Vivo, and Brain Target. Ph.D. Thesis, Zhejiang University, Hangzhou, China, 2007. [Google Scholar]
- Luo, J.; Wu, Z.L.; Wang, H.X. Pharmacokinetic study of gastrodin injection. Pharm. J. Chin. People’s Lib. Army 2006, 22, 391–393. [Google Scholar]
- Han, G.Z.; Liu, K.X.; Zhang, Y.L.; Su, C.Y.; Chen, Y.R.; Tang, N.Y. Separation and determination of gastrodin and p-hydroxybenzyl alcohol in rabbit plasma using HPLC. Chin. Pharm. J. 1985, 20, 697. [Google Scholar]
- Tang, C.L.; Wang, L.; Cheng, M.C.; Zhang, X.Z.; Liu, X.Y.; Xiao, H.B. Rapid and sensitive analysis of parishin and its metabolites in rat plasma using ultra high performance liquid chromatography-fluorescence detection. J. Chromatogr. B 2014, 973, 104–109. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.F.; Sun, L.X.; Chen, X.H.; Huo, Y.S.; Yan, B.Q.; Bi, K.S. Simultaneous Quantification of 6,7-Di-Hydroxyligustilide and Gastrodin in Rat Plasma by LC-MS: Application to Pharmacokinetic Study of Tianshu Capsule. Lat. Am. J. Pharm. 2012, 31, 112–119. [Google Scholar]
- Tang, C.L.; Wang, L.; Liu, X.X.; Cheng, M.C.; Xiao, H.B. Chemical fingerprint and metabolic profile analysis of ethyl acetate fraction of Gastrodia elata by ultra performance liquid chromatography/quadrupole-time of flight mass spectrometry. J. Chromatogr. B 2016, 1011, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.L.; Wang, L.; Cheng, M.C.; Liu, X.X.; Xiao, H.B. Determination of N6-(4-hydroxybenzyl) adenine riboside in rat plasma by ultra performance liquid chromatography-quadrupole time of flight mass spectrometry. Chin. J. Chromatogr. 2015, 33, 699–703. [Google Scholar] [CrossRef]
- Ni, S.M.; Qian, D.W.; Duan, J.A.; Guo, J.M.; Wang, Z.Z.; Shang, E.X.; Sun, G.L. Metablites of ligustilide and gastrodin from Dachuanxiong Decoction in rabbit plasma. Chin. Tradit. Pat. Med. 2010, 32, 1115–1120. [Google Scholar]
- Lin, L.C.; Chen, Y.F.; Lee, W.C.; Wu, Y.T.; Tsai, T.H. Pharmacokinetics of gastrodin and its metabolite p-hydroxybenzyl alcohol in rat blood, brain and bile by microdialysis coupled to LC-MS/MS. J. Pharm. Biomed. 2008, 48, 909–917. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.W.; Li, X.; Xie, H.T.; Shen, J.; Luo, J.; Wang, J.S.; Wang, K.D.G.; Liu, Q.W.; Kong, L.Y. Analysis and pharmacokinetics studies of gastrodin and p-hydroxybenzyl alcohol in dogs using ultra fast liquid chromatography–tandem mass spectrometry method. J. Pharm. Biomed. 2014, 99, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Lei, Y.J.; Wang, L.; Cheng, M.C.; Xiao, H.B. Identification of major metabolites in rat urine and plasma of N-6-(4-hydroxybenzyl) adenine riboside by LC/MS/MS. Biomed. Chromatogr. 2011, 25, 344–352. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.H.; Sheng, L.; Li, Y. Determination of YZG-331 in mouse plasma using liquid chromatography-tandem mass spectrometry. J. Chromatogr. B 2014, 944, 6–10. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.L.; Wang, L.; Liu, X.X.; Cheng, M.C.; Xiao, H.B. Pharmacokinetic study of Gastrodia elata in rats. Anal. Bioanal. Chem. 2015, 407, 8903–8910. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.L.; Wang, L.; Liu, X.X.; Cheng, M.C.; Qu, Y.; Xiao, H.B. Comparative pharmacokinetics of gastrodin in rats after intragastric administration of free gastrodin, parishin and Gastrodia elata extract. J. Ethnopharmacol. 2015, 176, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Zhang, Z.X.; Ma, J.G.; Zhang, Z.J.; Tian, Y. Studies on pharmacokinetics and tissue distribution of gastrodin in rats. Chin. J. Pharm. Anal. 2015, 35, 1369–1376. [Google Scholar]
- Zheng, X.Y. Studies on the Quality Control and Pharmacokinetics of Gastrodia elata Bl. in Rats. Master Thesis, Guizhou Normal University, Guiyang, China, 2015. [Google Scholar]
- Liu, K.X.; Zhang, Y.L.; Sun, H.; Han, G.Z.; Su, C.Y.; Tang, N.Y.; Chen, Y.R. Pharmacokinetic study of gastrodin in rabbits. Pharm. Clin. Chin. Mater. Med. 1985, 202–203. [Google Scholar]
- Liu, K.X.; Zhang, Y.L.; Han, G.Z.; Su, C.Y.; Tang, N.Y.; Chen, Y.R. Determination of gastrodin in plasma and its pharmacokinetic study. J. Dalian Med. Univ. 1986, 8, 36–42. [Google Scholar]
- Lu, G.W.; Zou, Y.J.; Chu, D.Q. In vivo metabolism of 3H-gastrodin in rats. Chin. Pharmacol. Bull. 1986, 2, 23–25. [Google Scholar]
- Tang, C.L.; Wang, J.L.; Yua, J.; Wang, L.; Cheng, M.C.; Cui, W.; Zhao, J.S.; Xiao, H.B. Identification, characterization and in vitro neuroprotection of N6-(4-hydroxybenzyl) adenine riboside and its metabolites. Phytochem. Lett. 2017, 20, 146–150. [Google Scholar] [CrossRef]
- He, W.; Liu, G.H.; Cai, H.; Sun, X.M.; Hou, W.E.; Zhang, P.T.; Xie, Z.Y.; Liao, Q.F. Integrated pharmacokinetics of five protoberberine-type alkaloids in normal and insomniac rats after single and multiple oral administration of Jiao-Tai-Wan. J. Ethnopharmacol. 2014, 154, 635–644. [Google Scholar] [CrossRef] [PubMed]
- Xiong, F.; Wang, H.; Jiang, Z.Z.; Huo, M.R.; Yan, C.Y.; Zheng, C.L.; Gu, N. Integrated pharmacokinetics and biodistribution of multiple flavonoid C-glycosides components in rat after oral administration of, Abrus mollis extract and correlations with bio-effects. J. Ethnopharmacol. 2015, 163, 290–296. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wang, J.Y.; Jia, X.B.; Tan, X.B.; Hu, M. Role of Intestinal Hydrolase in the Absorption of Prenylated Flavonoids Present in Yinyanghuo. Molecules 2011, 16, 1336–1348. [Google Scholar] [CrossRef] [PubMed]
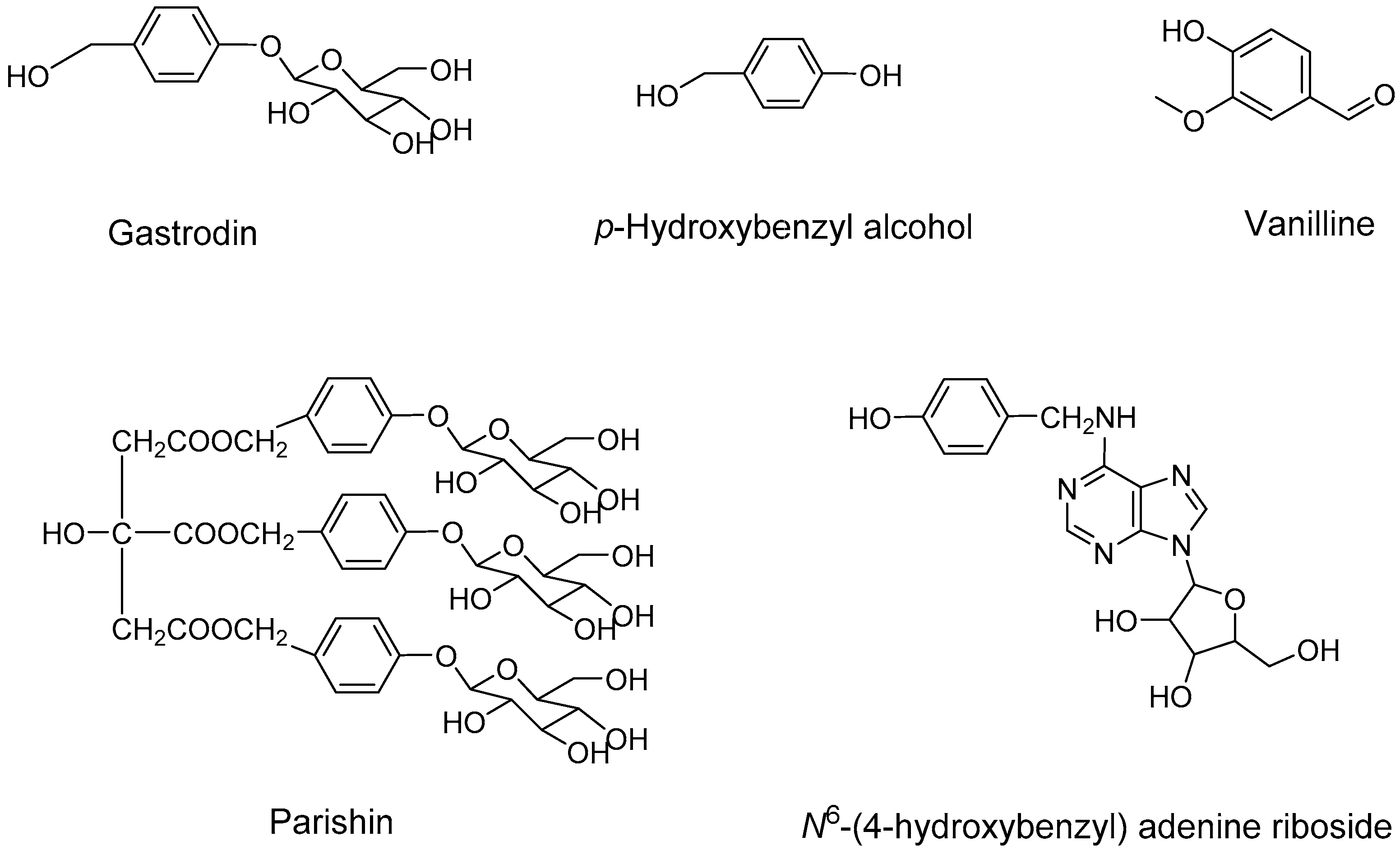
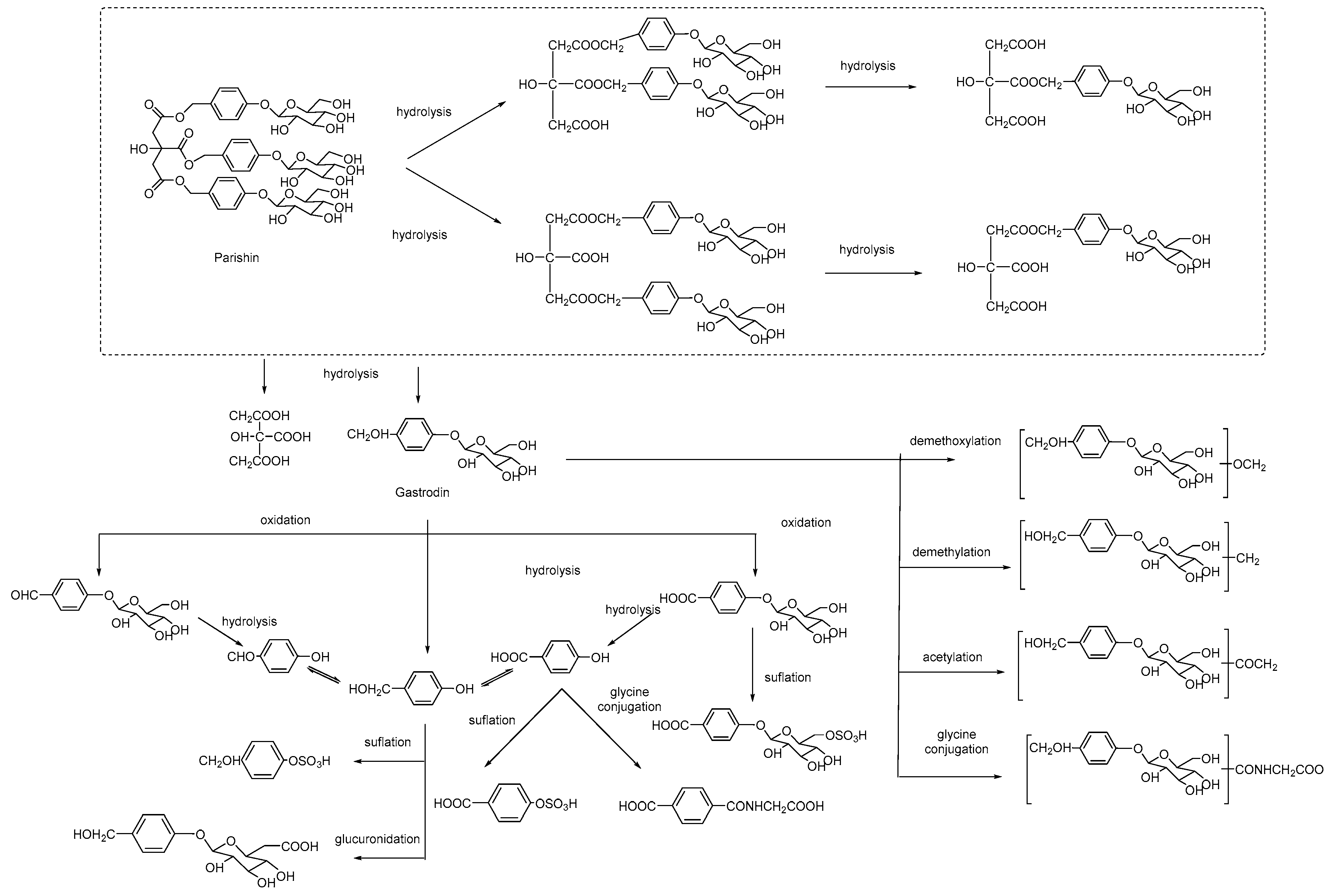
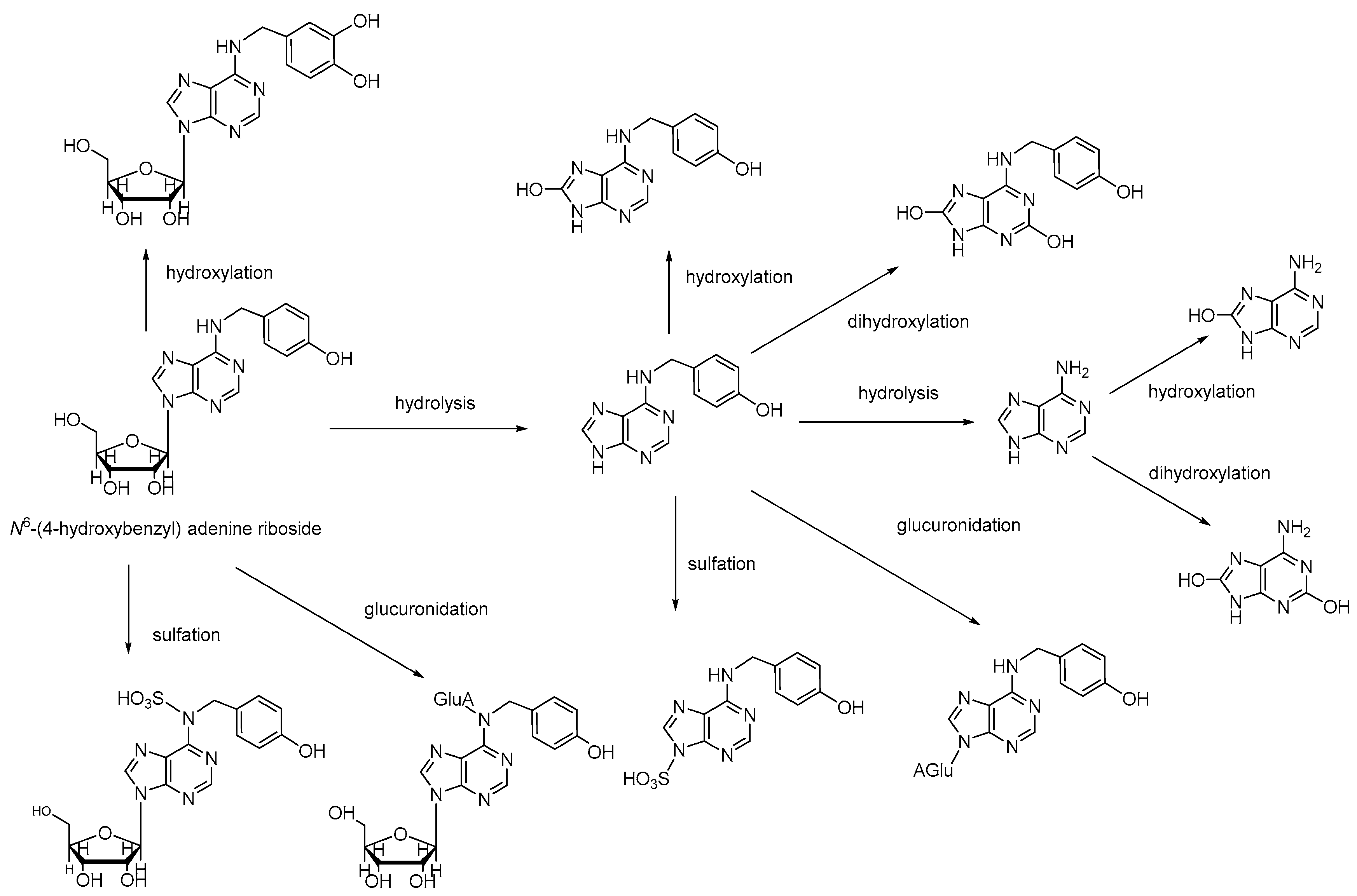
| Number | Method | Analytes | Type of Biological Sample | Time (min) | LOD/LOQ (ng/mL) | λ (nm/ESI) | Ref. |
|---|---|---|---|---|---|---|---|
| 1 | HPLC-UV | GAS and HBA | Plasma, CSF and Brain microdialysate | <30 | 280, 160 and 70/150, 70 and 40 | 221 | [47] |
| 2 | UPLC-UV | HBA | Duodenum, jejunum, ileum and colon | <2 | - | 220 | [47] |
| 3 | HPLC-ESI/APCI-MS | GAS and HBA | Plasma | <10 | 2000 and 830 | (−) | [32] |
| 4 | HPLC-ESI-MS | GAS and HBA | Plasma | <30 | 500 | (−) | [41] |
| 5 | HPLC-ESI-MS/MS | GAS | Plasma and dialysates | <15 | 0.01 | (+) | [48] |
| 6 | HPLC-ESI-MS/MS | GAS | blood, brain and bile | <10 | 0.5 | (−) | [65] |
| 7 | HPLC-ESI-MS/MS | HBA | blood, brain and bile | <10 | 0.2 | (−) | [65] |
| 8 | UFLC-ESI–MS/MS | GAS and its five metabolites | Plasma | <11 | 1 | (−) | [36,66] |
| 9 | HPLC-ESI-TSQ/MS | YZG-331 | Plasma | <7 | 25 | (+) | [68] |
| 10 | UHPLC-ESI-TSQ/MS | Parishin | Plasma | <10 | 0.083 | (−) | [34] |
| 11 | UPLC-QTOF/MS | NHBA | Plasma | <5 | 0.1 | (+) | [63] |
| 12 | UPLC-FLD | GAS, HBA, PB, PC, PA | Plasma | <10 | 0.6, 0.8, 1, 1, 1 | λex: 225 λem:295 | [60] |
| Target | Subject | Administered Route/Dosage (mg/kg)/Tissue | Tmax (min) | Cmax (μg/mL) | t1/2 (min) | AUC (μg·min/mL) | CL, CL/F (mL/min/kg) | V, V/F (mL/kg) | MRT (min) | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|
| GAS | Rabbit | iv/100, iv/200 | - | - | t1/2α: 5.2, t1/2β: 38.1 | 14,356 | 6.98 | 387 | - | [73] |
| Rat | iv/100 | - | - | t1/2α: 0.49, t1/2β: 8.41 | 3200 | 38 | 450 | - | [74] | |
| iv/200 | - | - | t1/2α: 1.9, t1/2β: 12.2 | 7900 | 25.3 | 442 | - | [74] | ||
| iv/200 | - | - | t1/2α: 4.6, t1/2β: 35 | 21,000 | - | - | - | [31] | ||
| ig/200 | 48 | 140 | t1/2α: 22, t1/2β: 52 | 18,000 | - | - | - | [31] | ||
| ig/200 bile duct ligation | 64 | 110 | t1/2α: 27, t1/2β: 60 | 15,000 | - | - | - | [31] | ||
| iv/100 | - | 533 | 38.8 | 8601 | 13.3 | 215 | 17.4 | [53] | ||
| iv/300 | - | 1033 | 38.2 | 29,882 | 102 | 237 | 23.2 | [53] | ||
| in/50 | - | - | - | 222 | - | - | - | [46] | ||
| iv/50 | - | - | - | 2597 | - | - | - | [46] | ||
| iv/200 | - | 350.9 | 41.2 | 19,019 | - | - | 57.5 | [47] | ||
| ig/20,000 | 70 | 13.48 | - | 1870 | 4.28 mL/min | 0.27 L | - | [32] | ||
| ig/200 | 5–30 | 128.4–209.8 | - | 16,297–20,174 | - | - | 104.8–115.2 | [33] | ||
| ig/50 | 21.00 | 29.75 | 172.64 | 3245 | 15.68 | 3710.74 | - | [39] | ||
| ig/100 | 34.50 | 42.80 | 181.16 | 5139 | 19.22 | 4872.31 | - | [39] | ||
| ig/200 | 108.50 | 30.85 | 109.67 | 8593 | 23.59 | 3776.81 | - | [39] | ||
| ig/50 granule | 26.88 | 18.56 | 239.34 | 3454 | 14.93 | 4974.58 | 278.78 | [40] | ||
| ig/100 granule | 42.13 | 28.40 | 225.60 | 5901 | 18.07 | 5808.72 | 337.13 | [40] | ||
| ig/200 granule | 36.00 | 25.45 | 205.71 | 6698 | 31.22 | 9235.03 | 377.37 | [40] | ||
| ig/100 | 34.50 | 42.80 | 181.16 | 5441 | 19.22 | 4872.31 | 171.85 | [40] | ||
| ig/20 | 13.4–18.5 | - | t1/2α: 2.1–9.1, t1/2β: 24.8–167.9 | 228–280 | - | - | - | [35] | ||
| ig/9 g/kg | 45 | 140.72 | - | 13,816 | 3.50 | - | 161.69 | [41] | ||
| ig/5.4 g/kg | 21.67 | 188.45 | - | 21,550 | 2.32 | - | 121.74 | [41] | ||
| ig/315 extract | 18.33 | 1.05 | 326.35 | 96 | 2570 | 1,196,580 | - | [34] | ||
| ig/630 extract | 12.50 | 2.97 | 176.51 | 216 | 2720 | 701,130 | - | [34] | ||
| ig/1260 extract | 14.17 | 4.66 | 158.82 | 293 | 4030 | 920,850 | - | [34] | ||
| ig/315 powder | 18.33 | 1.25 | 177.70 | 131 | 2180 | 574,840 | - | [34] | ||
| ig/630 powder | 25.00 | 1.25 | 345.07 | 113 | 4380 | 2,152,710 | - | [34] | ||
| ig/1260 powder | 40.83 | 2.29 | 260.50 | 229 | 5280 | 2,164,030 | - | [34] | ||
| ig/100 | 25.2 | 44.84 | 67.8 | 3475 | 29.67 | 2920 | 60.18 | [70] | ||
| ig/116 | 49.8 | 3.47 | 185.4 | 298 | 393.33 | 105,020 | 88.2 | [70] | ||
| ig/2300 | 60 | 14.18 | 451.2 | 2183 | 2147.67 | 1,415,580 | 128.4 | [70] | ||
| iv/72.5 | - | - | 67.2 | 1560 | 48.33 | 3720 | - | [69] | ||
| iv/116 | - | - | 70.2 | 1850 | 64 | 6450 | - | [69] | ||
| iv/220 | - | - | 73.2 | 5108 | 43 | 4450 | - | [69] | ||
| iv/64.5 | - | - | 78.6 | 3181 | 20.33 | 2370 | - | [69] | ||
| ig/1 g/kg | 40.2 | 0.26 | 60.6 | 18.6 | 56,293.17 | 4,139,720 | 90 | [69] | ||
| ig/4 g/kg | 42 | 0.21 | 69.6 | 69 | 58,714.17 | 7,270,960 | 109.8 | [69] | ||
| ig/40 | 15–33 | 18.43–21.74 | 58.2–168.6 | 1039–1630 | 24.54–40.43 | - | 49.8–82.2 | [52] | ||
| iv/20 | - | 69.97–75.65 | 68.4–102 | 1324–1634 | 15.13–12.20 | - | 21.6–27.6 | [52] | ||
| ig/21 | 22.8 | 10.78 | - | 557 | - | - | 71.4 | [71] | ||
| iv/21 | 78 | - | - | 1651 | - | - | 64.8 | [71] | ||
| ig/0.322 g | 12–15 | 1.67–4.34 | t1/2α: 9.3–29.4, t1/2β: 51.3–235.9 | 233–372 | 0.01–0.02 L/min | 0.348–1.424 L | 122.3–290.9 | [37] | ||
| iv/100/Brain | - | 1.4 | 14.6 | 42 | - | - | 24.7 | [53] | ||
| iv/300/Brain | - | 5.2 | 58.4 | 353 | - | - | 82.5 | [53] | ||
| ig/200/Brain | 5–15 | 0.45–0.79 | - | 50–105 | - | - | 77.56–139.68 | [33] | ||
| iv/200/CSF | 15.0 | 16.1 | 64.7 | 848 | - | - | 66.4 | [47] | ||
| iv/200/Frontal cortex | - | 21.6 | 29.6 | 588 | - | - | 26.0 | [47] | ||
| iv/200/Hippocampus | - | 24.3 | 25.6 | 580 | - | - | 22.7 | [47] | ||
| iv/200/Thalamus | - | 22.0 | 27.8 | 570 | - | - | 27.9 | [47] | ||
| iv/200/Cerebellum | - | 35.8 | 25.2 | 1042 | - | - | 26.8 | [47] | ||
| iv/100/Bile | - | 37.1 | 27.6 | 1048 | - | - | 38.1 | [53] | ||
| iv/300/Bile | - | 214 | 34.6 | 5352 | - | - | 30.8 | [53] | ||
| Dog | ig | 90 | 51.0 | t1/2α: 2.49, t1/2β: 6.43 | 40,494 | - | - | - | [43] | |
| ig/40 | 109.8 | 23.00 | t1/2α: 111.6, t1/2β: 288 | 7230 | 7.16 | 0.0016 | - | [66] | ||
| iv/50 | - | - | t1/2α: 4.64, t1/2β: 105 | 3800 | 1.4 | 210 | - | [74] | ||
| Human | ig/200 | 48.6 | 1.48 | t1/2α: 226.8, t1/2β: 363.6 | 337 | - | - | - | [30] | |
| ig/20/experiment | 48.6 | 1.48 | 330 | 344 | - | - | - | [42] | ||
| HBA | Rat | ig/9 g/kg | 55 | 10.45 | - | 2267 | - | - | 266.67 | [41] |
| ig/5.4 g/kg | 60 | 9.12 | - | 1656 | - | - | 154.12 | [41] | ||
| ig/200/Brain | Tmax1:15 Tmax2: 90 | Cmax1: 0.55 Cmax2: 0.71 | 54.8 | 52,823 ng min/g | - | - | - | [45] | ||
| iv/50 | 15 | 0.11 | - | - | - | - | - | [65] | ||
| Parishin | Rat | ig/315 extract | 38.33 | 0.02 | 53.24 | 1.84 | 0.18 | 13.44 | - | [34] |
| ig/630 extract | 56.67 | 0.030 | 62.42 | 4.64 | 0.14 | 12.32 | - | [34] | ||
| ig/1260 extract | 113.33 | 0.031 | 38.49 | 4.59 | 0.27 | 15.29 | - | [34] | ||
| ig/315 powder | 26.67 | 0.017 | 62.48 | 2.42 | 0.16 | 16.35 | - | [34] | ||
| ig/630 powder | 75.00 | 0.021 | 54.25 | 3.22 | 0.24 | 21.28 | - | [34] | ||
| ig/1260 powder | 83.33 | 0.021 | 54.28 | 3.60 | 0.15 | 31.2 | - | [34] | ||
| iv/72.5/ | - | - | 21.6 | 268 | 278.83 | 8680 | - | [69] | ||
| iv/116/ | - | - | 17.4 | 358 | 337.5 | 8840 | - | [69] | ||
| iv/220/ | - | - | 24.6 | 1057 | 295 | 12,630 | - | [69] | ||
| Integrated parishin | Rat | iv/72.5 | - | - | 72.6 | 1118 | 64.83 | 6930 | - | [69] |
| iv/116 | - | - | 76.8 | 1387 | 83.5 | 9290 | - | [69] | ||
| iv/220 | - | - | 66.6 | 3629 | 60.5 | 5830 | - | [69] | ||
| ig/116 | - | - | 70.2 | 192 | 599.17 | 60,990 | 89.4 | [69] | ||
| Integrated Gastrodiae capsule | Rat | ig/1 g/kg | - | - | 80.4 | 15.6 | 63,881.17 | 7,411,560 | 91.2 | [69] |
| ig/4 g/kg | - | - | 91.8 | 54.6 | 70,464.67 | 93,335,990 | 160.2 | [69] | ||
| NHBA | Rat | ig/200 | 69 | 0.11 | 465 | 26 | 6619.33 | 5,340,000 | 486 | [63] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, J.; Wu, B.; Tang, C.; Zhao, J. Analytical Techniques and Pharmacokinetics of Gastrodia elata Blume and Its Constituents. Molecules 2017, 22, 1137. https://doi.org/10.3390/molecules22071137
Wu J, Wu B, Tang C, Zhao J. Analytical Techniques and Pharmacokinetics of Gastrodia elata Blume and Its Constituents. Molecules. 2017; 22(7):1137. https://doi.org/10.3390/molecules22071137
Chicago/Turabian StyleWu, Jinyi, Bingchu Wu, Chunlan Tang, and Jinshun Zhao. 2017. "Analytical Techniques and Pharmacokinetics of Gastrodia elata Blume and Its Constituents" Molecules 22, no. 7: 1137. https://doi.org/10.3390/molecules22071137
APA StyleWu, J., Wu, B., Tang, C., & Zhao, J. (2017). Analytical Techniques and Pharmacokinetics of Gastrodia elata Blume and Its Constituents. Molecules, 22(7), 1137. https://doi.org/10.3390/molecules22071137






