Newly Synthesized Doxorubicin Complexes with Selected Metals—Synthesis, Structure and Anti-Breast Cancer Activity
Abstract
:1. Introduction
2. Results
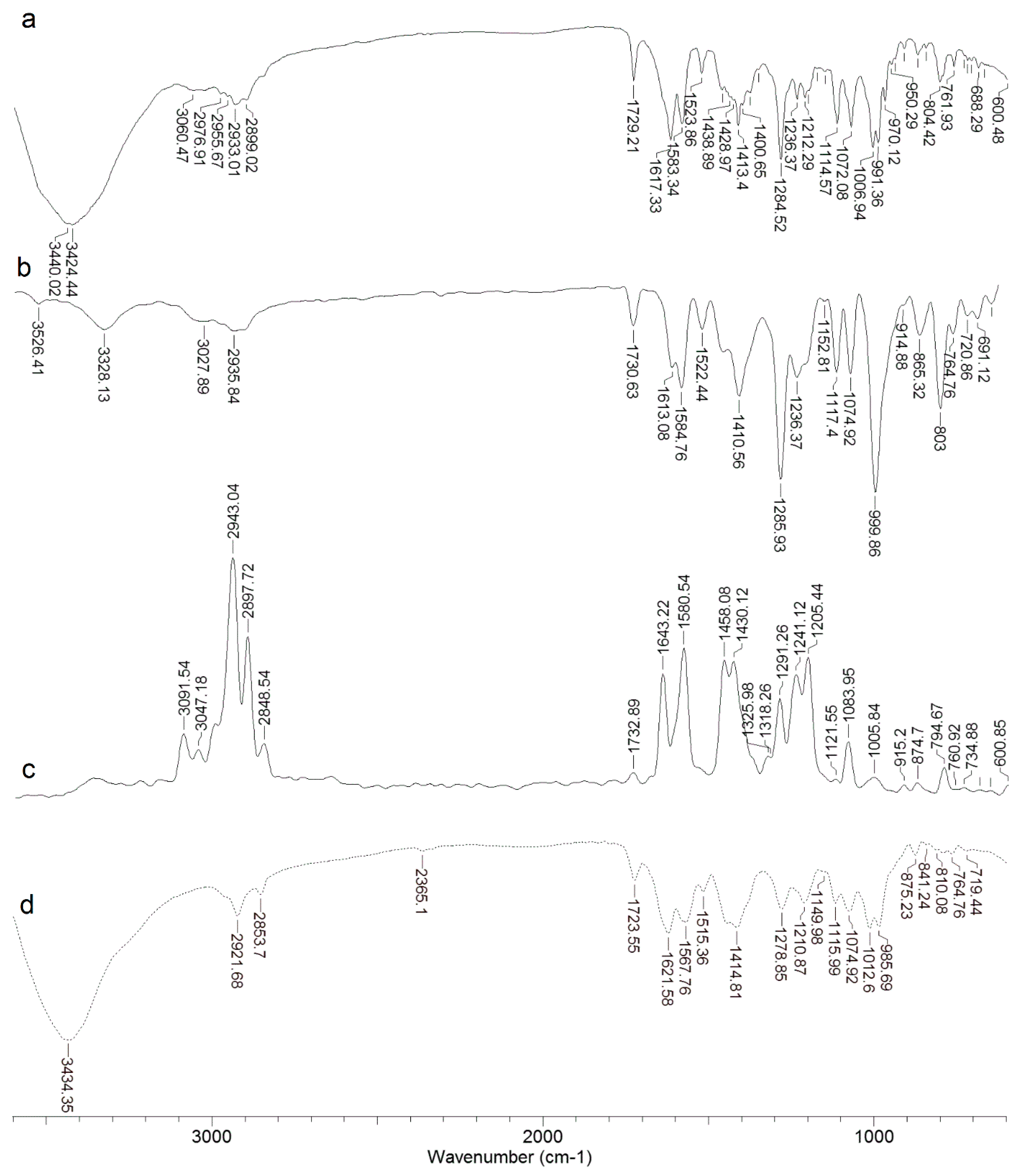
2.1. IR and Raman Spectra of DOX
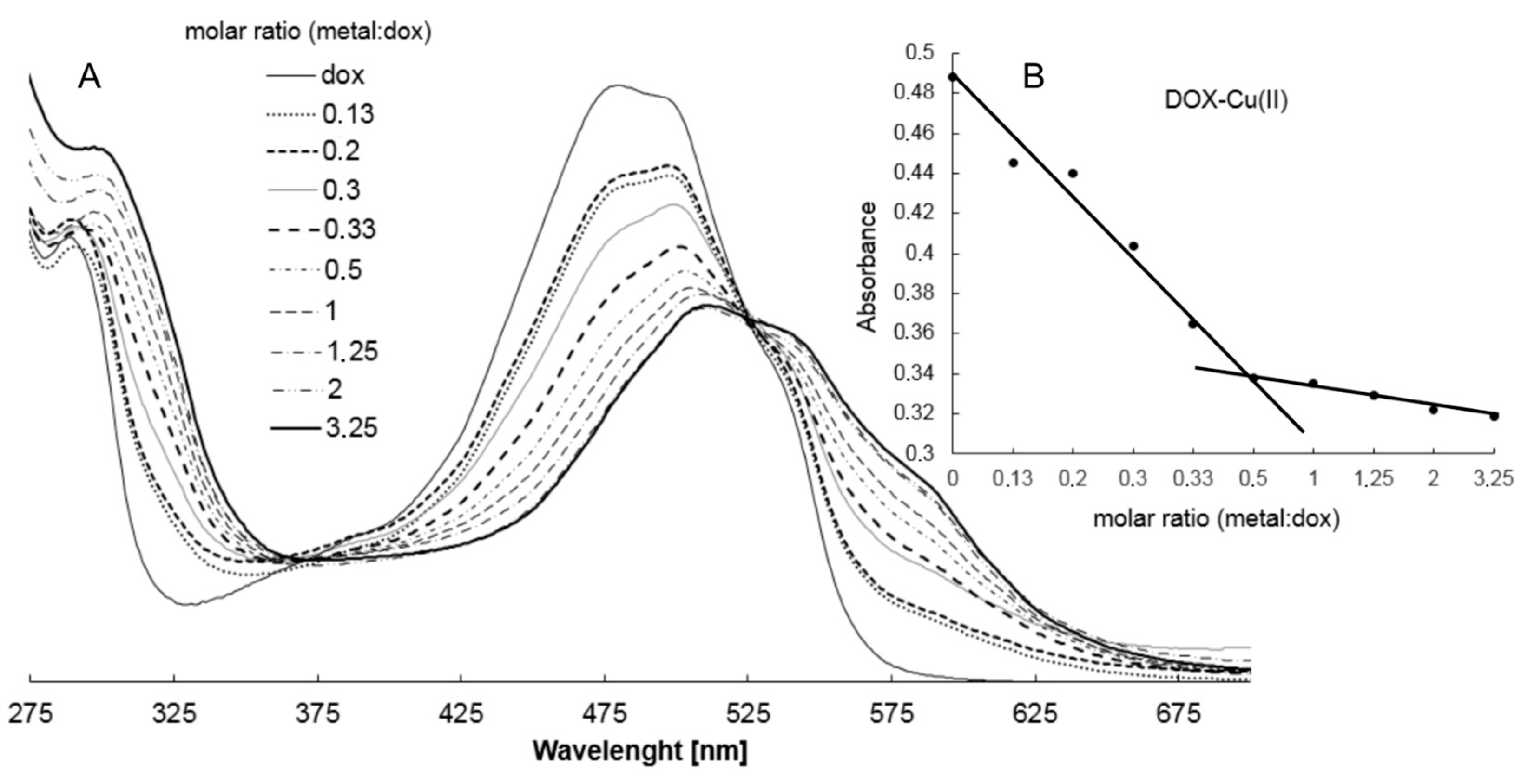
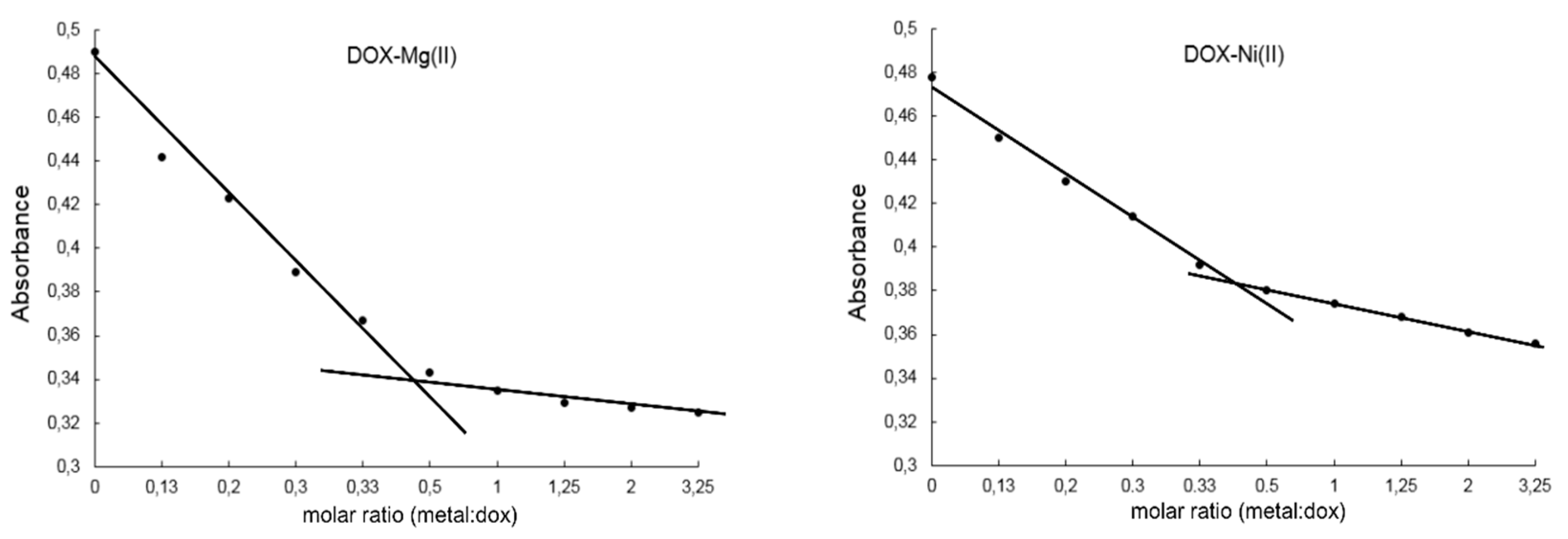
2.2. Study on the Composition of Metal-DOX Complexes in Aqueous Solutions and Solid Phase
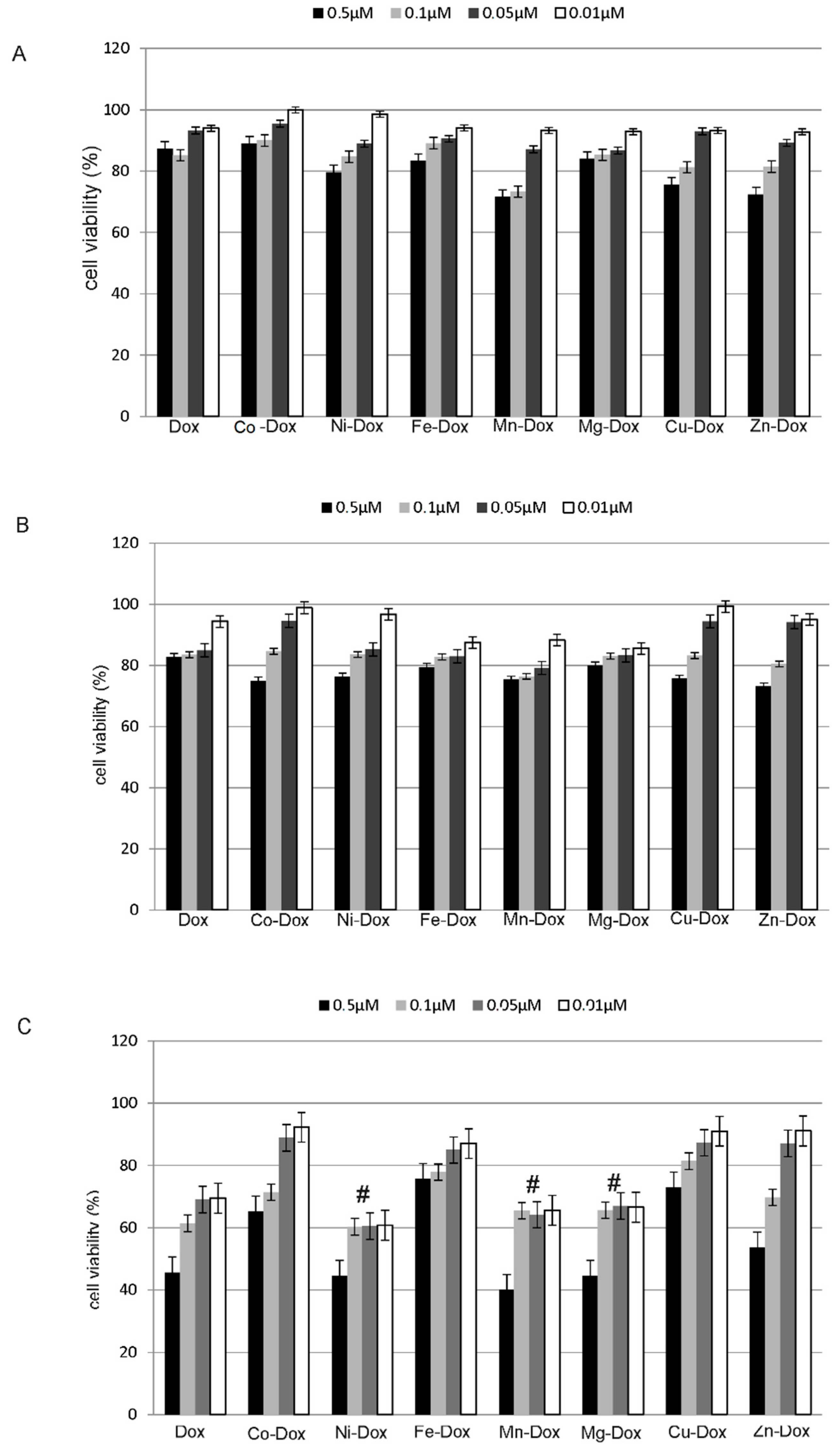
2.3. Estimation of Cells Proliferation
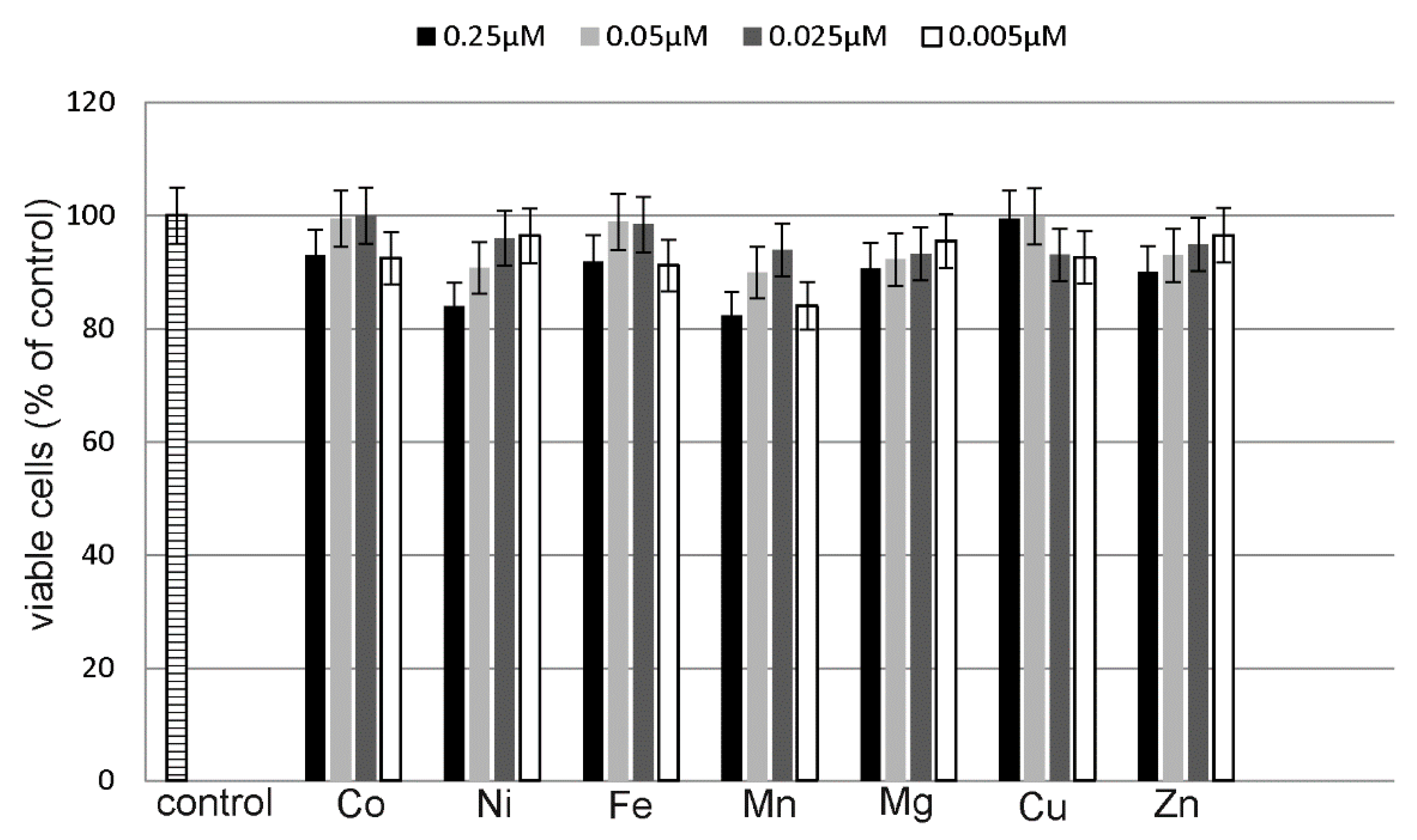
2.4. Estimation of Metals Cytotoxicity
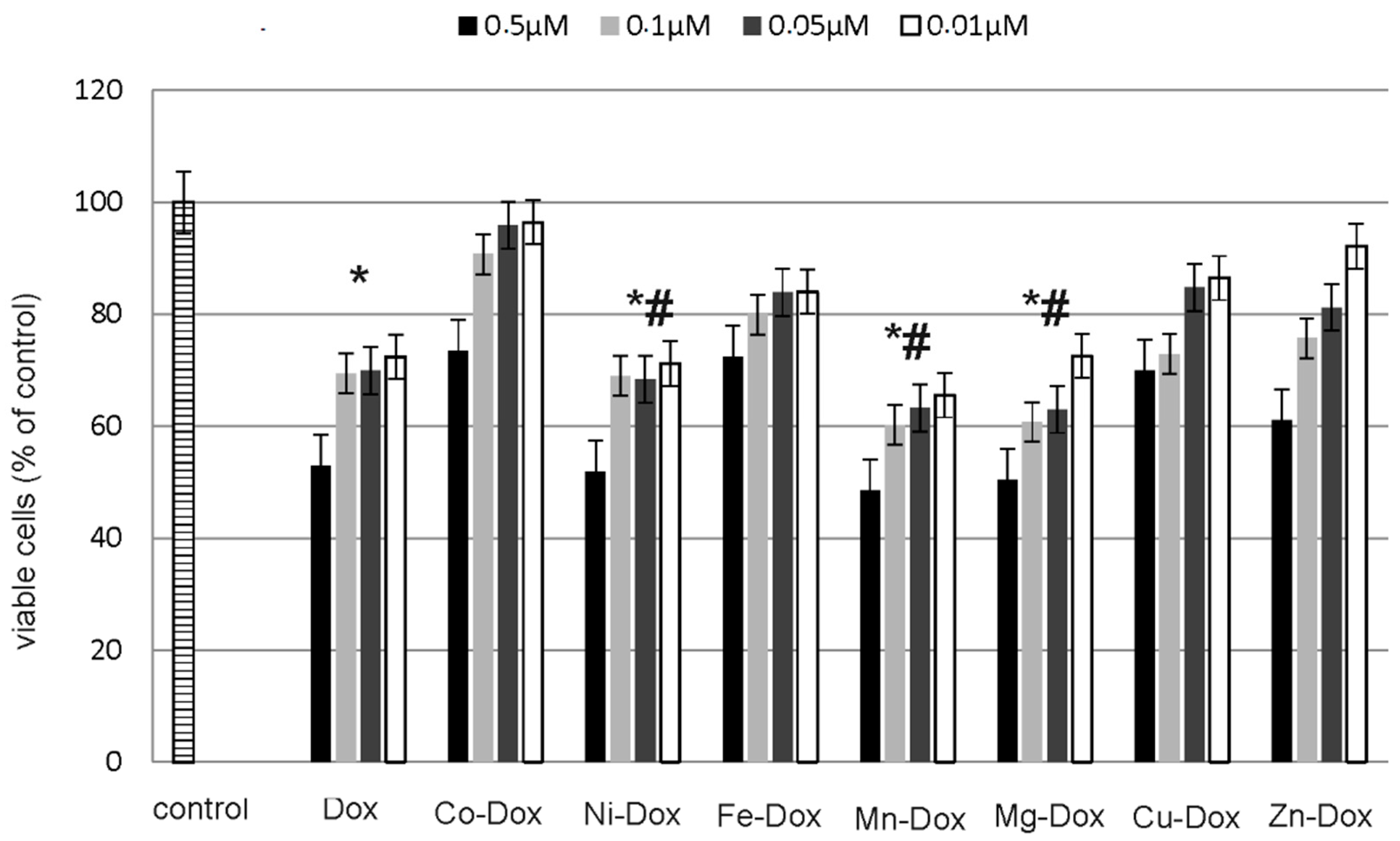
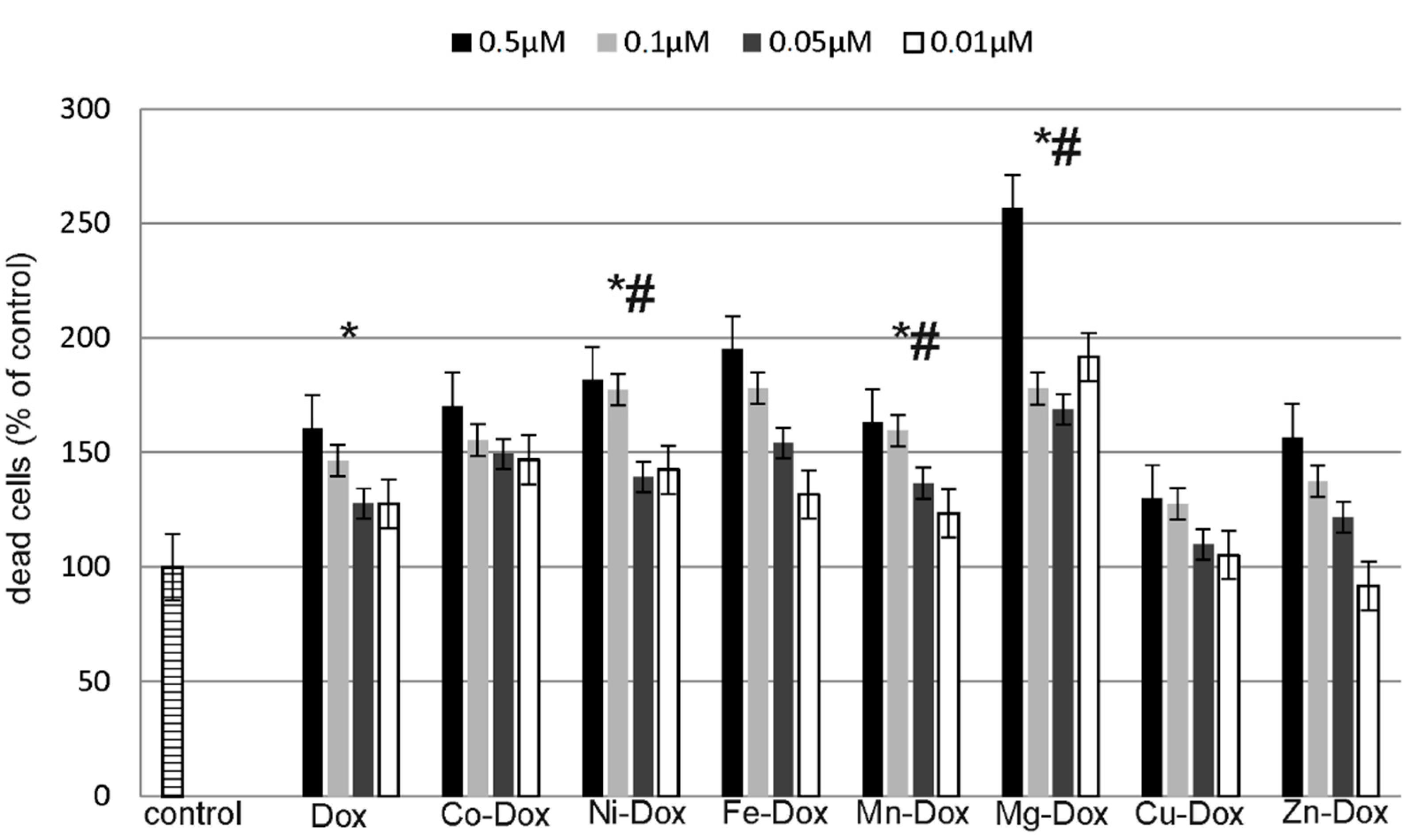
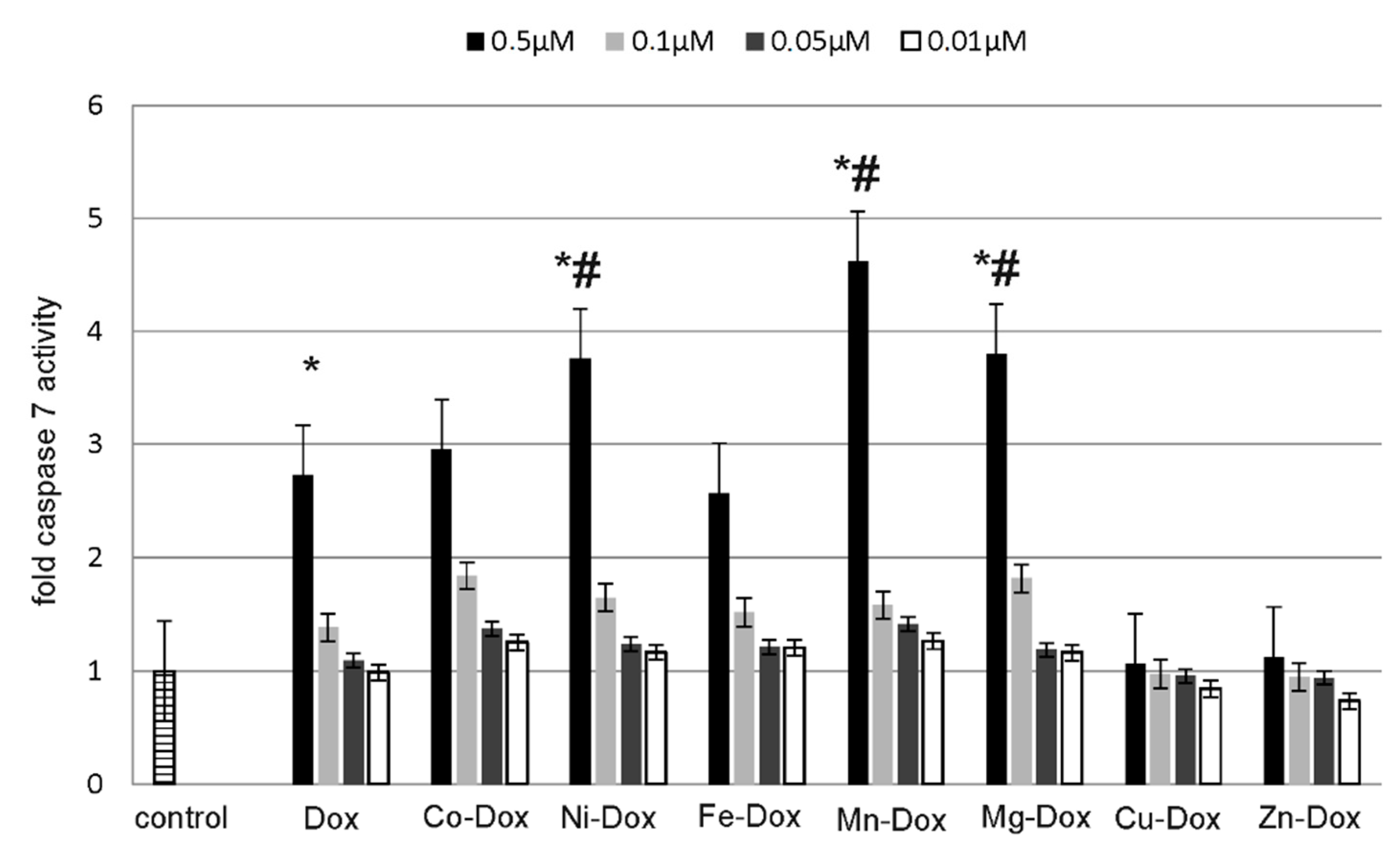
2.5. Estimation of Cells Viability, Cytotoxicity and Apoptosis
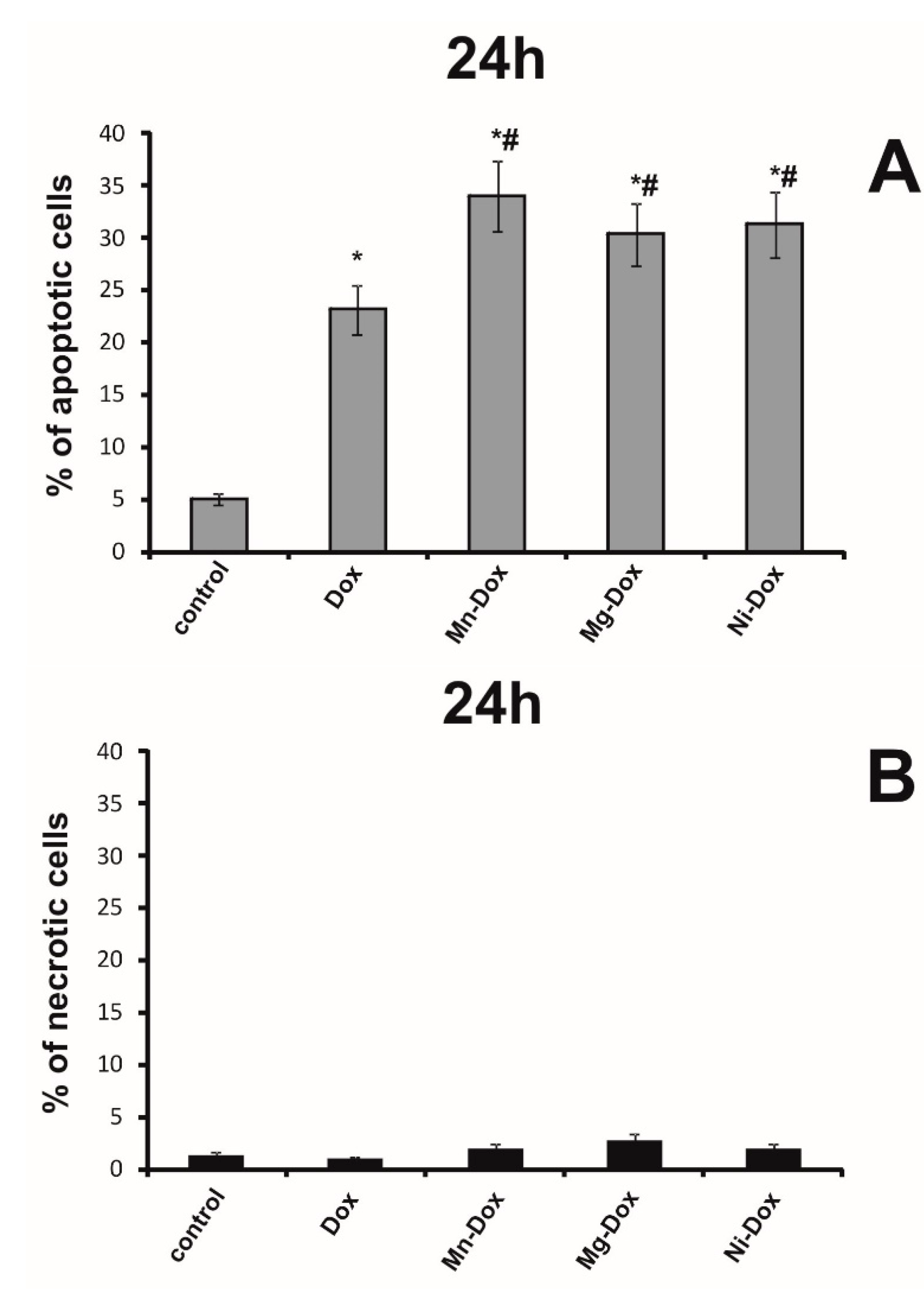
2.6. Detection of Apoptosis and Necrosis
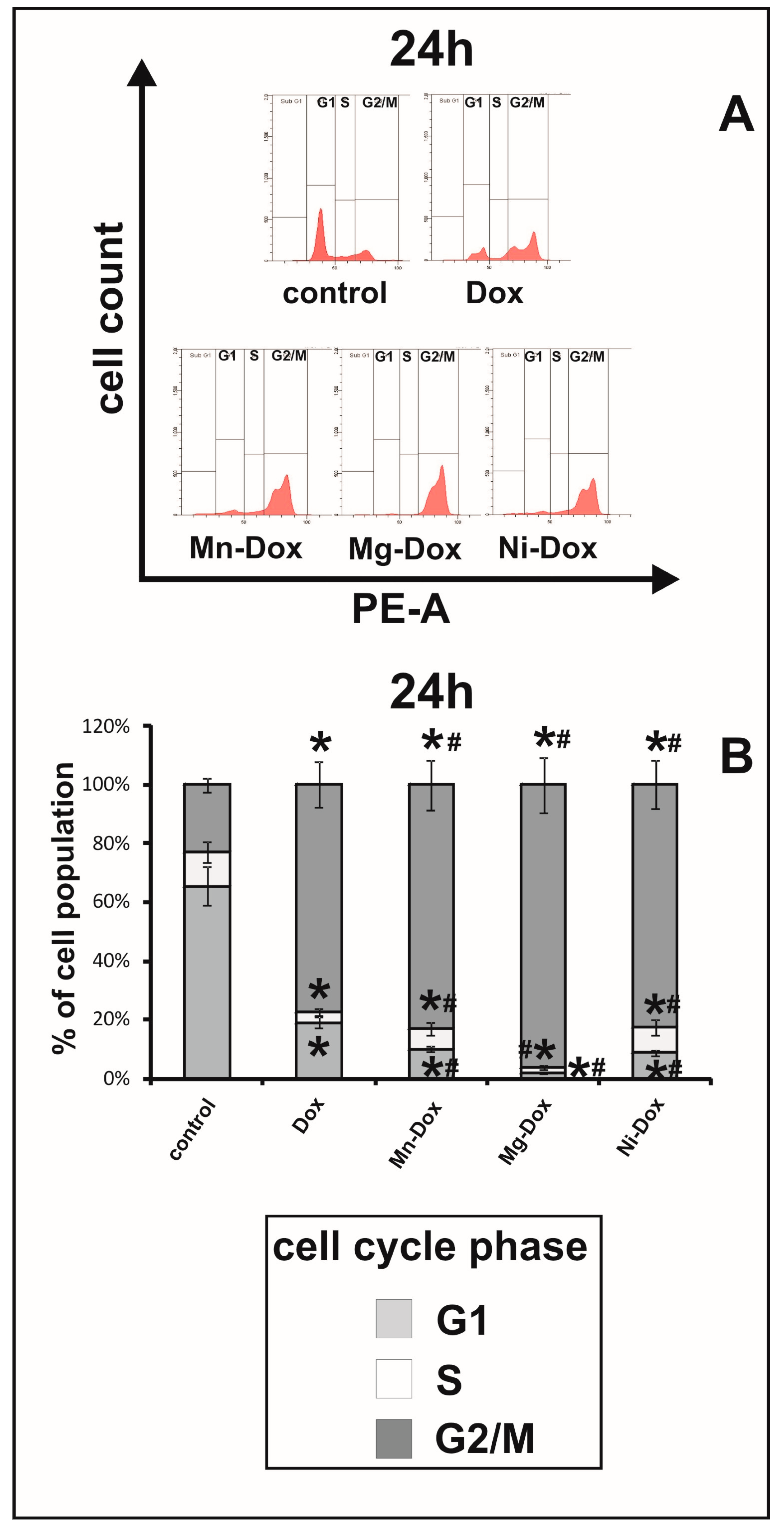
2.7. Cell Cycle Analysis
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Complexes Preparation
4.3. Infrared Spectrum (FT-IR, ATR) and Raman Spectrum of DOX and Metal Complexes
4.4. Study on the Composition of Metal-DOX Complexes in Aqueous Solutions
4.5. Cell Culture
4.6. Cell Exposure to DOX and Its Metal Complexes
4.7. Estimation of Cells Proliferation
4.8. Estimation of Metals Cytotoxicity
4.9. Estimation of Cells Viability, Cytotoxicity and Apoptosis
4.10. Detection of Apoptosis and Necrosis
4.11. Cell Cycle Assay
4.12. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
Compliance with Ethical Standards
Abbreviations
| DOX | Doxorubicin |
| Cu-DOX | Complex of doxorubicin with Cu (molar ratio 1:2) |
| Zn-DOX | Complex of doxorubicin with Zn (molar ratio 1:2) |
| Co-DOX | Complex of doxorubicin with Co (molar ratio 1:2) |
| Ni-DOX | Complex of doxorubicin with Ni (molar ratio 1:2) |
| Fe-DOX | Complex of doxorubicin with Fe (molar ratio 1:3) |
| Mn-DOX | Complex of doxorubicin with Mn (molar ratio 1:2) |
| Mg-DOX | Complex of doxorubicin with Mg (molar ratio 1:2) |
References
- Pilco-Ferreto, N.; Calaf, G.M. Influence of doxorubicin on apoptosis and oxidative stress in breast cancer cell lines. Int. J. Oncol. 2016, 49, 753–762. [Google Scholar] [CrossRef] [PubMed]
- Turner, N.; Biganzoli, L.; Di, L.A. Continued value of adjuvant anthracyclines as treatment for early breast cancer. Lancet Oncol. 2015, 16, e362–e369. [Google Scholar] [CrossRef]
- Szuławska, A.; Czyż, M. Molekularne mechanizmy działania antracyklin. Post. Hig. Med. Dośw. 2006, 60, 78–100. [Google Scholar]
- Missailidis, S. Anticancer Therapeutics; John Wiley & Sons: Chichester, UK, 2008; pp. 53–54. [Google Scholar]
- Mitry, M.A.; Edwards, J.G. Doxorubicin induced heart failure: Phenotype and molecular mechanisms. IJC Heart Vasc. 2016, 10, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Komiyama, T.; Oki, T.; Inui, T. Interaction of new anthracycline antibiotics with DNA. Effects on nucleic acid synthesis and binding to DNA. Biochim. Biophys. Acta 1983, 740, 80–87. [Google Scholar] [CrossRef]
- Gruber, B.M.; Anuszewska, E.L.; Bubko, I.; Goździk, A.; Fokt, I.; Priebe, W. Effect of structural modification at the 4, 3’, and 2’ positions of doxorubicin on topoisomerase II poisoning, apoptosis, and cytotoxicity in human melanoma cells. Arch. Immunol. Ther. Exp. 2007, 55, 193–198. [Google Scholar] [CrossRef] [PubMed]
- Savatier, J.; Rharass, T.; Canal, C.; Gbankoto, A.; Vigo, J.; Salmon, J.M.; Ribou, A.C. Adriamycin dose and time effects on cell cycle, cell death, and reactive oxygen species generation in leukaemia cells. Leuk. Res. 2012, 36, 791–798. [Google Scholar] [CrossRef] [PubMed]
- Feng, M.; Yang, Y.; He, P.; Fang, Y. Spectroscopic studies of copper(II) and iron(II) complexes of Adriamycin. Spectr. Acta A 2000, 56, 581–587. [Google Scholar] [CrossRef]
- Sugioka, K.; Nakano, M. Mechanism of phospholipid peroxidation induced by ferric ion-ADP-adriamycin-coordination complex. Bioch. Biophys. Acta 1982, 713, 333–343. [Google Scholar]
- Greenaway, F.T.; Dabrowiak, J.C. The Binding of Copper Ions to Daunomycin and Adriamycin. J. Inorg. Biochem. 1982, 16, 91–107. [Google Scholar] [CrossRef]
- Akman, S.A.; Doroshow, J.H.; Bruke, T.G.; Dizdaroglu, M. DNA base modifications induced in isolated human chromatin by NADH dehydrogenase- catalyzed reduction of doxorubicin. Biochemistry 1992, 31, 3500–3506. [Google Scholar] [CrossRef] [PubMed]
- Beraldo, H.; Garnier-Suillerot, A.; Tosi, L.; Lavelle, F. Iron(III)-adriamycin and iron(III)-daunorubicin complexes: Physicochemical characteristics, interaction with DNA, and antitumor activity. Biochemistry 1985, 24, 284–289. [Google Scholar] [CrossRef] [PubMed]
- Pereira, E.; Fiallo, M.; Garnier-Suillerot, A.; Kiss, T.; Kozłowski, H. Impact of aluminium ions on adriamycin-type ligands. J. Chem. Soc. 1993, 3, 455–459. [Google Scholar] [CrossRef]
- Fiallo, M.M.L.; Garnier-Suillerot, A. Physicochemical studies of the iron(III)carminomycin complex and evidence of the lack of stimulated superoxide production by NADH dehydrogenase. Biochim. Biophys. Acta 1985, 840, 91–98. [Google Scholar] [CrossRef]
- Fantine, E.; Garnier-Suillerot, A. Interaction of 5’-iminodaunorubicin with Fe(III) and with cardiolipin-containing vesicles. Biochim. Biophys. Acta 1986, 856, 130–136. [Google Scholar] [CrossRef]
- Muindi, J.; Sinha, B.; Gianni, L.; Myers, C. Hydroxyl production and DNA damage induced by anthracycline-iron complex. FEBS Lett. 1984, 2, 226–230. [Google Scholar] [CrossRef]
- Fiallo, M.M.L.; Garnier-Suillerot, A. Metal anthracycline complexes as a new class of anthracycline derivatives. Palladium(II)-adriamycin and palladium(II)-daunorubicin complexes: Physicochemical characteristics and antitumor activity. Biochemistry 1986, 25, 924–930. [Google Scholar] [CrossRef] [PubMed]
- McLennan, I.J.; Lenkinski, R.E. The Binding of Yb(III) to Adriamycin—A H-1-NMR Relaxation Study. J. Am. Chem. Soc. 1984, 106, 6905–6909. [Google Scholar] [CrossRef]
- Minotti, G.; Menna, P.; Salvatorelli, E.; Cairo, G.; Gianni, L. Anthracyclines: Molecular advances and pharmacologic developments in antitumor activity and cardiotoxicity. Pharm. Rev. 2004, 56, 185–229. [Google Scholar] [CrossRef] [PubMed]
- Berlin, V.; Haseltine, W.A. Reduction of adriamycin to a semiquinone-free radical by NADPH cytochrome P-450 reductase produces DNA cleavage in a reaction mediated by molecular oxygen. J. Biol. Chem. 1981, 256, 4747–4756. [Google Scholar] [PubMed]
- Jacobson, M.D.; Weil, M.; Raff, M.C. Programmed cell death in animal development. Cell 1997, 88, 347–354. [Google Scholar] [CrossRef]
- Wong, R.S. Apoptosis in cancer: From pathogenesis to treatment. J. Exp. Clin. Cancer Res. 2011, 30, 87. [Google Scholar] [CrossRef] [PubMed]
- Lewandowski, W.; Kalinowska, M.; Lewandowska, H. The influence of metals on the electronic system of biologically important ligands. Spectroscopic study of benzoates, salicylates, nicotinates and isoorates. J. Inorg. Biochem. 2005, 99, 1407–1423. [Google Scholar] [CrossRef] [PubMed]
- Koczoń, P.; Piekut, J.; Borawska, M.; Lewandowski, W. Vibrational structure and antimicrobial activity of selected isonicotinates, potassium picolinate and nicotinate. J. Mol. Struct. 2003, 651–653, 651–656. [Google Scholar] [CrossRef]
- Kalinowska, M.; Piekut, J.; Bruss, A.; Follet, C.; Sienkiewicz-Gromiuk, J.; Świsłocka, R.; Rzączyńska, Z.; Lewandowski, W. Spectroscopic (FT-IR, FT-Raman, 1H-, 13C-NMR, UV/VIS), thermogravimetric and antimicrobial studies of Ca(II), Mn(II), Cu(II), Zn(II0 and Cd(II) complexes of ferulic acid. Spectrochim. Acta A 2014, 122, 631–638. [Google Scholar] [CrossRef] [PubMed]
- Kowczyk-Sadowy, M.; Świsłocka, R.; Lewandowska, H.; Piekut, J.; Lewandowski, W. Spectroscopic (FT-IR, FT-Raman, 1H- and 13C-NMR), theoretical and microbiological study of trans o-coumaric acid and alkali metal o-coumarates. Molecules 2015, 20, 3146–3169. [Google Scholar] [CrossRef] [PubMed]
- Cheung, B.C.; Sun, T.H.; Leenhouts, J.M.; Cullis, P.R. Loading of doxorubicin into liposomes by forming Mn2+-drug complexes. Biochim. Biophys. Acta 1998, 1414, 205–216. [Google Scholar] [CrossRef]
- Orvig, C.; Abrams, M.J. Medicinal inorganic chemistry: Introduction. Chem. Rev. 1999, 99, 2201–2204. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Milacic, V.; Frezza, M.; Dou, Q.P. Metal complexes, their cellular targets and potential for cancer therapy. Curr. Pharm. Des. 2009, 15, 777–791. [Google Scholar] [CrossRef] [PubMed]
- Haas, K.L.; Franz, K.J. Application of metal coordination chemistry to explore and manipulate cell biology. Chem. Rev. 2009, 109, 4921–4960. [Google Scholar] [CrossRef] [PubMed]
- Fricker, S.P. Metal based drugs: From serendipity to design. Dalton Trans. 2007, 2007, 4903–4917. [Google Scholar] [CrossRef] [PubMed]
- Meggers, E. Targeting proteins with metal complexes. Chem. Commun. 2009, 9, 1001–1010. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.M. New approaches for medicinal applications of bioinorganic chemistry. Curr. Opin. Chem. Biol. 2007, 11, 115–120. [Google Scholar] [CrossRef] [PubMed]
- Bouma, J.; Beijnen, J.H.; Bult, A.; Underberg, W.J.M. Anthracycline antitumour agents: A review of physicochemical, analytical and stability properties. Pharm. Weekbl. Sci. Ed. 1986, 8, 109–133. [Google Scholar] [CrossRef]
- Chiu, G.N.C.; Abraham, S.A.; Ickenstein, L.M.; Ng, R.; Karlsson, G.; Edwards, K.; Wasan, E.K.; Bally, M.B. Encapsulation of doxorubicin into thermosensitive liposomes via complexation with the transition metal manganese. J. Cont. Release. 2005, 104, 271–288. [Google Scholar] [CrossRef] [PubMed]
- Abraham, S.A.; Edwards, K.; Karlsson, G.; MacIntosh, S.; Mayer, L.D.; McKenzie, C.; Bally, M.B. Formation of transition metal–doxorubicin complexes inside liposomes. Biochim. Biophys. Acta 2002, 1565, 41–54. [Google Scholar] [CrossRef]
- Fiallo, M.M.L.; Garnier-Suillerot, A. Copper(II)-anthracycline systems. Evidence of a dihydroxo-bridged dicopper(II) aclacinomycin complex. J. Inorg. Biochem. 1987, 31, 43–55. [Google Scholar] [CrossRef]
- Lange, T.S.; Kim, K.K.; Singh, R.K.; Strongin, R.M.; McCourt, C.K.; Brard, L. Iron(III)-salophene: An organometallic compound with selective cytotoxic and anti-proliferative properties in platinum-resistant ovarian cancer cells. PLoS ONE 2008, 3, e2303. [Google Scholar] [CrossRef]
- Ray, S.; Mohan, R.; Singh, J.K.; Samantaray, M.K.; Shaikh, M.M.; Panda, D.; Ghosh, P. Anticancer and antimicrobial metallopharmaceutical agents based on palladium, gold, and silver N-heterocyclic carbine complexes. J. Am. Chem. Soc. 2007, 129, 15042–15053. [Google Scholar] [CrossRef] [PubMed]
- Sun, R.W.; Ma, D.L.; Wong, E.L.; Che, C.M. Some uses of transition metal complexes as anti-cancer and anti-HIV agents. Dalton Trans. 2007, 43, 4884–4892. [Google Scholar] [CrossRef]
- Baile, M.B.; Kolhe, N.S.; Deotarse, P.P.; Jain, A.S.; Kulkarni, A.A. Metal Ion Complex-Potential Anticancer Drug—A Review. IJPRR 2015, 4, 59–66. [Google Scholar]
- Milacic, V.; Chen, D.; Giovagnini, L.; Diez, A.; Fregona, D.; Dou, Q.P. Pyrrolidine dithiocarbamatezinc(II) and -copper(II) complexes induce apoptosis in tumor cells by inhibiting the proteasomal activity. Toxicol. Appl. Pharmacol. 2008, 231, 24–33. [Google Scholar] [CrossRef] [PubMed]
- Sharifi, S.; Barar, J.; Hejazi, M.S.; Samadi, N. Doxorubicin Changes Bax /Bcl-xL Ratio, Caspase-8 and 9 in Breast Cancer Cells. Adv. Pharm. Bull. 2015, 5, 351–359. [Google Scholar] [CrossRef] [PubMed]
- Krętowski, R.; Borzym-Kluczyk, M.; Stypułkowska, A.; Brańska-Januszewska, J.; Ostrowska, H.; Cechowska-Pasko, M. Low glucose dependent decrease of apoptosis and induction of autophagy in breast cancer MCF-7 cells. Mol. Cell. Biochem. 2016, 417, 35–47. [Google Scholar] [CrossRef] [PubMed]
- McGee, M.M.; Hyland, E.; Campiani, G.; Ramunno, A.; Nacci, V.; Zisterer, D.M. Caspase-3 is not essential for DNA fragmentation in MCF-7 cells during apoptosis induced by the pyrrolo-1,5-benzoxazepine, PBOX-6. FEBS Lett. 2002, 515, 66–70. [Google Scholar] [CrossRef]
- Cuvillier, O.; Nava, V.E.; Murthy, S.K.; Edsall, L.C.; Levade, T.; Milstien, S.; Spiegel, S. Sphingosine generation, cytochrome c release, and activation of caspase-7 in doxorubicin-induced apoptosis of MCF7 breast adenocarcinoma cells. Cell Death Differ. 2001, 8, 162–171. [Google Scholar] [CrossRef] [PubMed]
- Chitambar, C.R.; Wereley, J.P.; Matsuyama, S. Gallium-induced cell death in lymphoma: Role of transferrin receptor cycling, involvement of Bax and the mitochondria, and effects of proteasome inhibition. Mol. Cancer Ther. 2006, 5, 2834–2843. [Google Scholar] [CrossRef] [PubMed]
- Green, M.A.; Welch, M.J. Gallium radiopharmaceutical chemistry. Int. J. Radiat. Appl. Instrum. B 1989, 16, 435–448. [Google Scholar] [CrossRef]
- Yan, Y.; Hein, A.L.; Greer, P.M.; Wang, Z.; Kolb, R.H.; Batra, S.K.; Cowan, K.H. A novel function of HER2/Neu in the activation of G2/M checkpoint in response to γ-irradiation. Oncogene 2015, 34, 2215–2226. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, A.K.; Singh, R.P.; Agarwal, C.; Chan, D.C.; Agarwal, R. Silibinin strongly synergizes human prostate cancer DU145 cells to doxorubicin-induced growth inhibition, G2-M arrest, and apoptosis. Clin. Cancer Res. 2002, 8, 3512–3519. [Google Scholar] [PubMed]
- Ling, Y.H.; el-Naggar, A.K.; Priebe, W.; Perez-Soler, R. Cell cycle dependent cytotoxicity, G2-M phase arrest, and disruption of p34cdc2/cyclin B1 activity induced by doxorubicin in synchronized p388 cells. Mol. Pharmacol. 1996, 49, 832–841. [Google Scholar] [PubMed]
- Wadler, S.; Green, M.D.; Basch, R.; Muggia, F.M. Lethal and sublethal effects of the combination of doxorubicin and the bis-dioxopiperazine(+)-1,2-bis(3,5-diozopeperazinyl-l-yl) propane (ICRF 187) on murine sarcoma S180 in vitro. Biochem. Pharmacol. 1987, 9, 1495–1501. [Google Scholar] [CrossRef]
- Das, G.; Nicastri, A.; Coluccio, M.L.; Gentile, F.; Candeloro, P.; Cojoc, G.; Liberale, C.; De Angelis, F.; Di Fabrizio, E. FT-IR, Raman, RRS measurements and DFT calculation for doxorubicin. Microscop. Res. Tech. 2010, 73, 991–995. [Google Scholar] [CrossRef] [PubMed]
- Fornari, F.A.; Randolph, J.K.; Yalowich, J.C.; Ritke, M.K.; Gewirtz, D.A. Interference by doxorubicin with DNA unwinding in MCF-7 breast tumor cells. Mol. Pharmacol. 1994, 45, 649–656. [Google Scholar] [PubMed]
Sample Availability: Samples of compounds are not available from authors. |
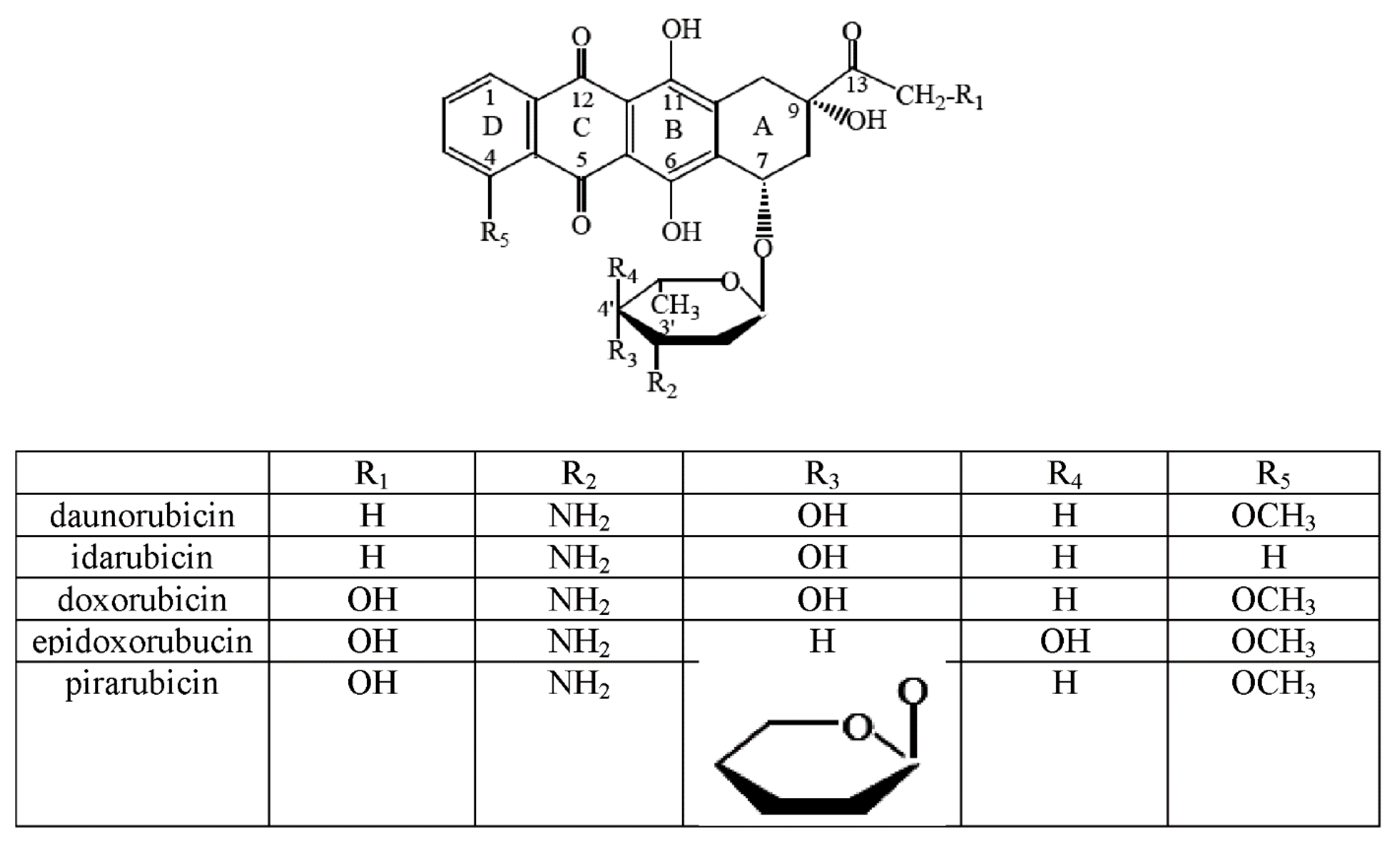
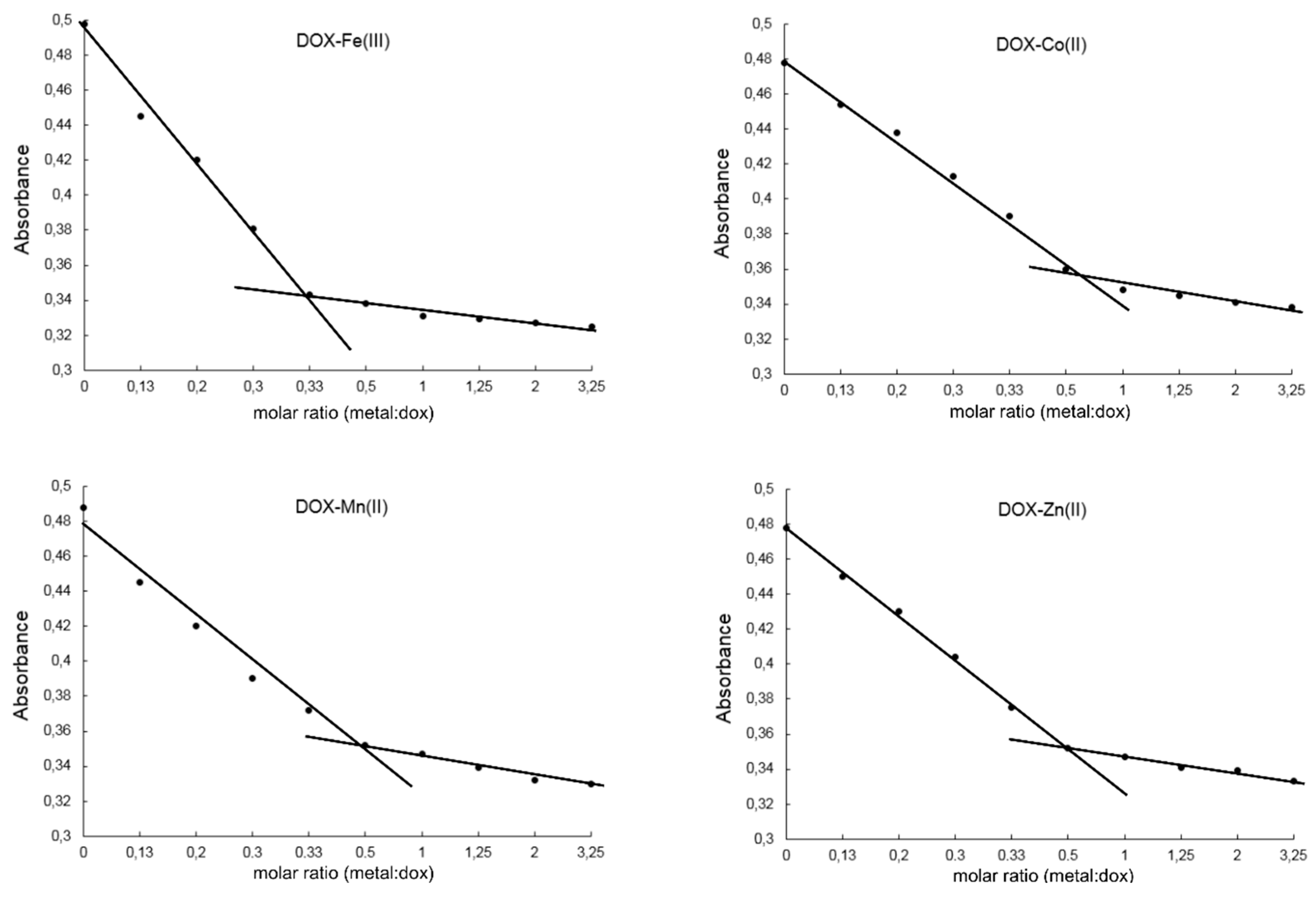
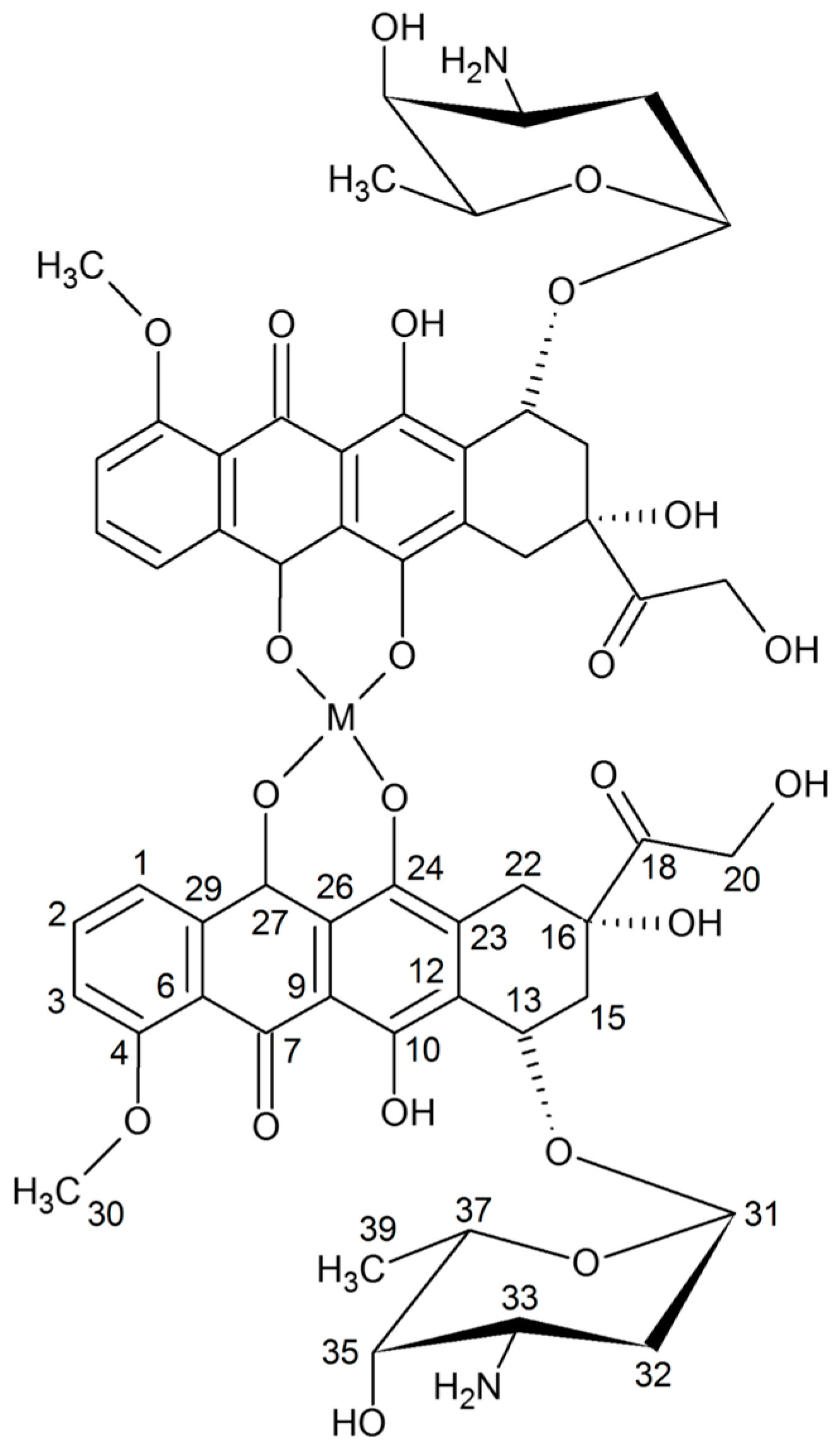
| Metal Ion | Possible Points of Anthracycline Attachment |
|---|---|
| Fe(II) | Carbonyl group (at C-12′) |
| Phenolic group (at C-11′) | |
| Cu(II) | Carbonyl group (at C-5′) |
| Phenolic group (at C-6′) | |
| Carbonyl group (at C-12′) | |
| Phenolic group (at C-11′) | |
| Yb(II) | Carbonyl group (at C-12′) |
| Phenolic group (at C-11′) |
| Doxorubicin | Cu Complex | Assingments (Numbering Atoms in Figure 5) | ||
|---|---|---|---|---|
| IR KBr | IR ATR | Raman | IR KBr | |
| 3423 vs | 3434 vs | νHO-H | ||
| 3026 w | 3047 w | |||
| 2932 m | 2940 vs | 2922 m | νC(33,35,37)-H 33/35/37 H | |
| 2899 m | 2898 s | 2854w | νC(33,35)-H | |
| 1729 m | 1731 w | 1732 w | 1724 w | δN-H |
| 1643 s | νRing, δCO(25)-H | |||
| 1617 m | 1615 m | 1607 m | 1622 m | ring |
| 1583 m | 1583 m | 1581 m | 1568 m | ring |
| 1524 w | 1524 w | 1515 w | sδC(20,22)-H2, sδCO(11,25)-H 20/22 | |
| 1462 m | 1463 m | 1461 s | 1443 m | Ring, δC(30)-H |
| 1428 s | Ring-O, Ring=O, δC(22,20,15)-H2, δC(13,31,33,37)-H, δC(32)-H2 | |||
| 1413 m | 1413 m | 1404 m | 1415 m | Ring, C-Hx(ar) |
| 1379 m | 1374 w | δC(13)-H, δO-H…O, δC-C, C(16)-OH | ||
| 1329 w | δO-H…O, Ring, C(20,15)-H2, δC(16)-OH, C(13)-H | |||
| 1285 s | 1286 vs | 1290 m | 1279 m | δO-H…O, Ring, ouC(32)-H2, δC(11)-OH, C(13)-H |
| 1236 m | 1236 m | 1247 m | C(32)-H2, C(33,35,37)-H, O(36)-H, C-O(38)-C | |
| 1211 m | 1209 m | 1205 s | 1211 m | δO-H…O, Ring, ouC(20)-H2, δC(11)-OH, N-H2 |
| 1153 w | 1149 vw | 1158 vw | 1150 w | Ring external, C(33)-NH+, HC(37)-C(35)H, C(39)-H3 |
| 1116 m | 1116 m | 1119 vw | 1116 m | Ring breathing, δC(16)-C(18)=O, C(30)-H3, |
| 1074 m | 1074 m | 1085 w | 1075 m | δC(16)-C(15)-H, C(4)-O(6)-C(30), δN-H2, δC-Hx (ali) |
| 1007 m | 1009 vw | 1013 m | νC(13)-O(14)-C(31), νC(16)-O(17)H, δC-Hx (ali) | |
| 991 m | 997 vs | 989 vw | 986 m | C(20)-H2 (ali), δC=O, δC(16)-OH, δC(16)C(18)-C(22) |
| 970 m | 970 m | δC-C(27)=O, C(24)-OH, δC-Hx (ali) | ||
| 950 w | 945 w | δC-Hx, CO(14)-C(31), O-C(31)-O | ||
| 912 vw | 913 w | 915 vw | δC(24)-OH, δC(22)-H2, O(28)…H-O(25) | |
| 804 w | 804 m | 795 w | 809 w | ouRing |
| 762 w | 765 w | 764 vw | 765 w | δRing(C-H), ωC(31,35)-H, ωC(39)-H3, |
| Sample Number | Molar Ratio Metal:DOX | DOX (mL) | Metal Chloride (μL) |
|---|---|---|---|
| 1 | 0 | 3 | 0 |
| 2 | 0.13 | 3 | 2 |
| 3 | 0.2 | 3 | 3 |
| 4 | 0.3 | 3 | 4 |
| 5 | 0.33 | 3 | 5 |
| 6 | 0.5 | 3 | 7.5 |
| 7 | 1 | 3 | 15 |
| 8 | 1.25 | 3 | 20 |
| 9 | 2 | 3 | 30 |
| 10 | 3.25 | 3 | 50 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jabłońska-Trypuć, A.; Świderski, G.; Krętowski, R.; Lewandowski, W. Newly Synthesized Doxorubicin Complexes with Selected Metals—Synthesis, Structure and Anti-Breast Cancer Activity. Molecules 2017, 22, 1106. https://doi.org/10.3390/molecules22071106
Jabłońska-Trypuć A, Świderski G, Krętowski R, Lewandowski W. Newly Synthesized Doxorubicin Complexes with Selected Metals—Synthesis, Structure and Anti-Breast Cancer Activity. Molecules. 2017; 22(7):1106. https://doi.org/10.3390/molecules22071106
Chicago/Turabian StyleJabłońska-Trypuć, Agata, Grzegorz Świderski, Rafał Krętowski, and Włodzimierz Lewandowski. 2017. "Newly Synthesized Doxorubicin Complexes with Selected Metals—Synthesis, Structure and Anti-Breast Cancer Activity" Molecules 22, no. 7: 1106. https://doi.org/10.3390/molecules22071106
APA StyleJabłońska-Trypuć, A., Świderski, G., Krętowski, R., & Lewandowski, W. (2017). Newly Synthesized Doxorubicin Complexes with Selected Metals—Synthesis, Structure and Anti-Breast Cancer Activity. Molecules, 22(7), 1106. https://doi.org/10.3390/molecules22071106








