Origins, Phytochemistry, Pharmacology, Analytical Methods and Safety of Cortex Moutan (Paeonia suffruticosa Andrew): A Systematic Review
Abstract
:1. Introduction
2. Origins of CM
3. Chemistry of CM
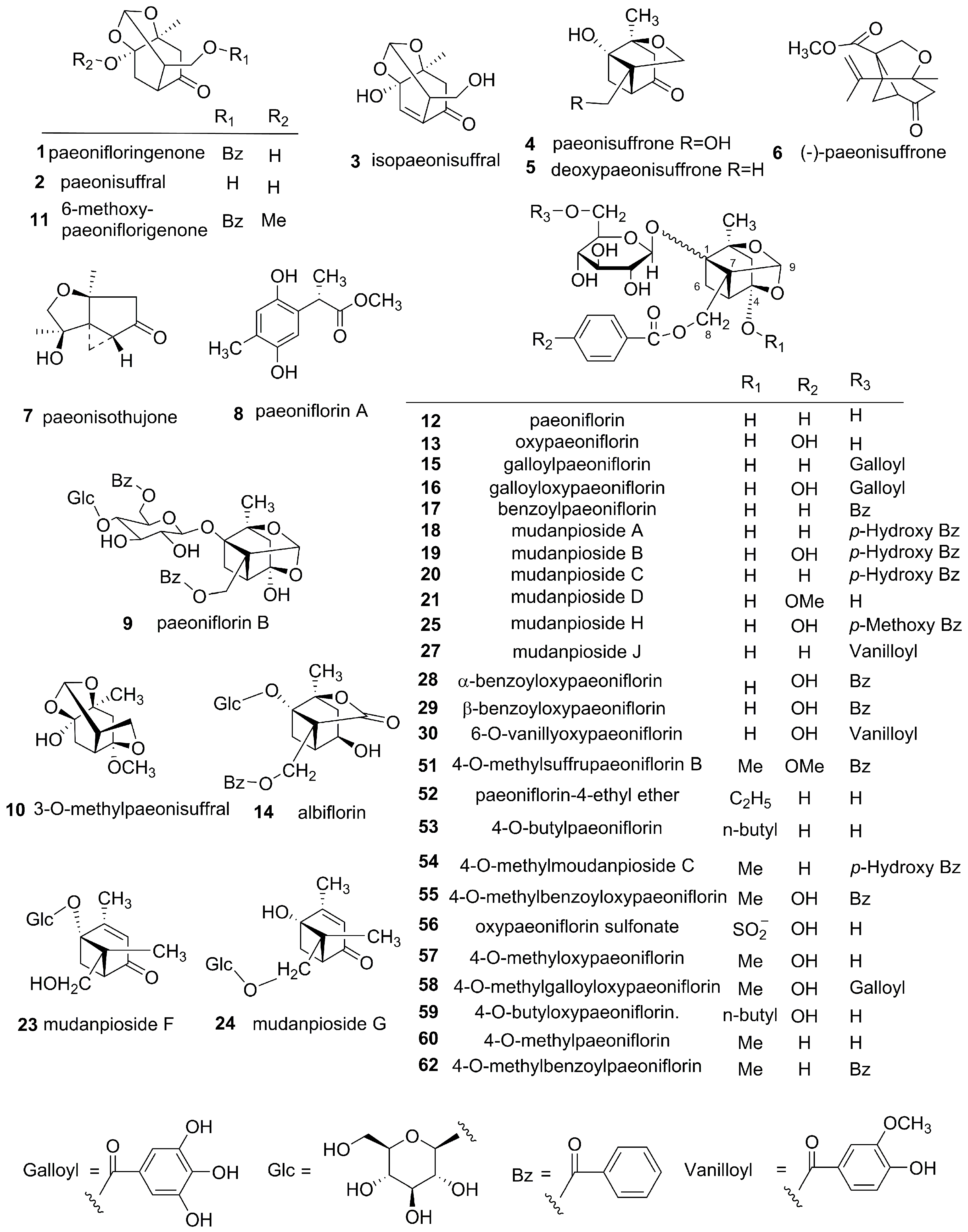
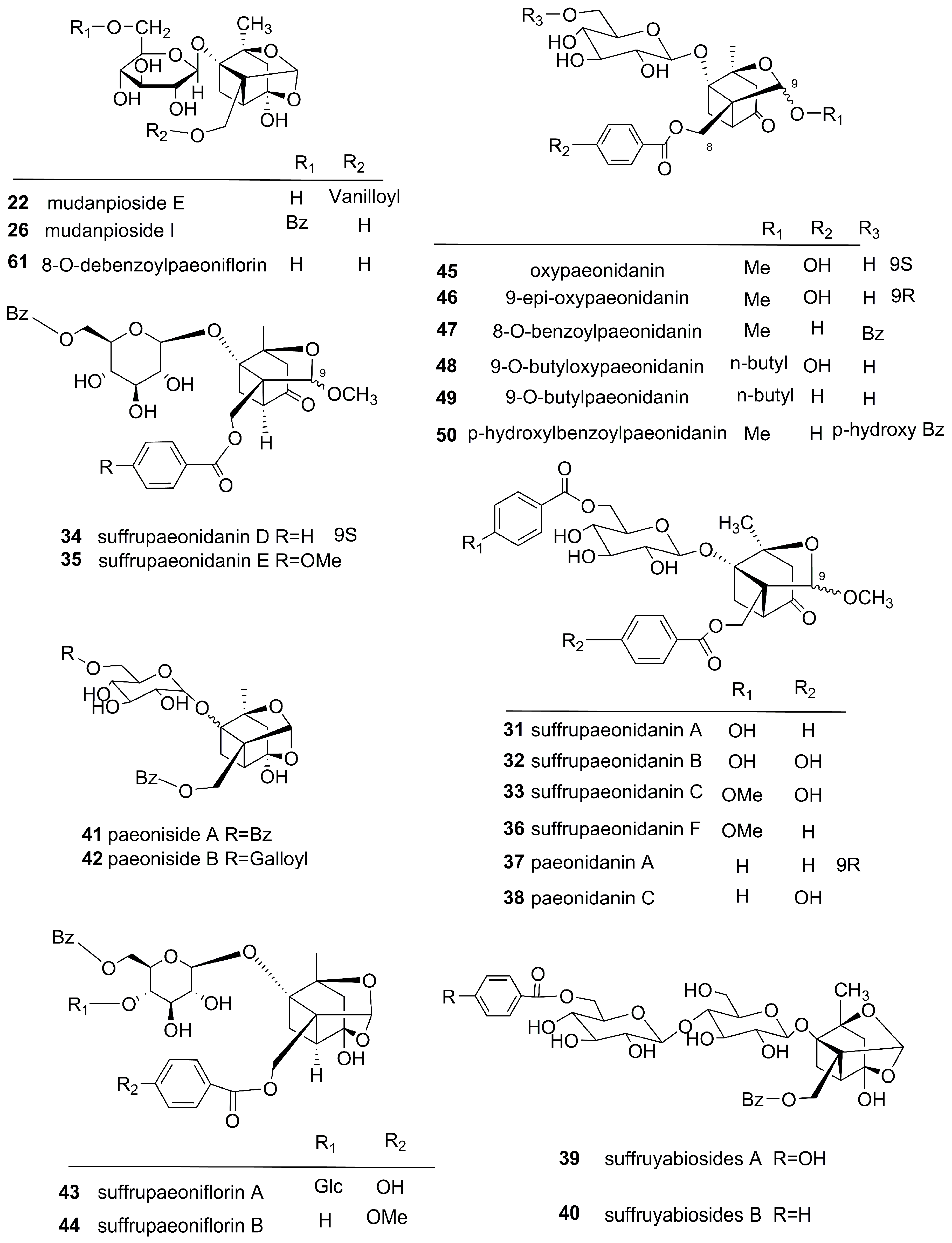
3.1. Monoterpenes and Monoterpene Glycosides
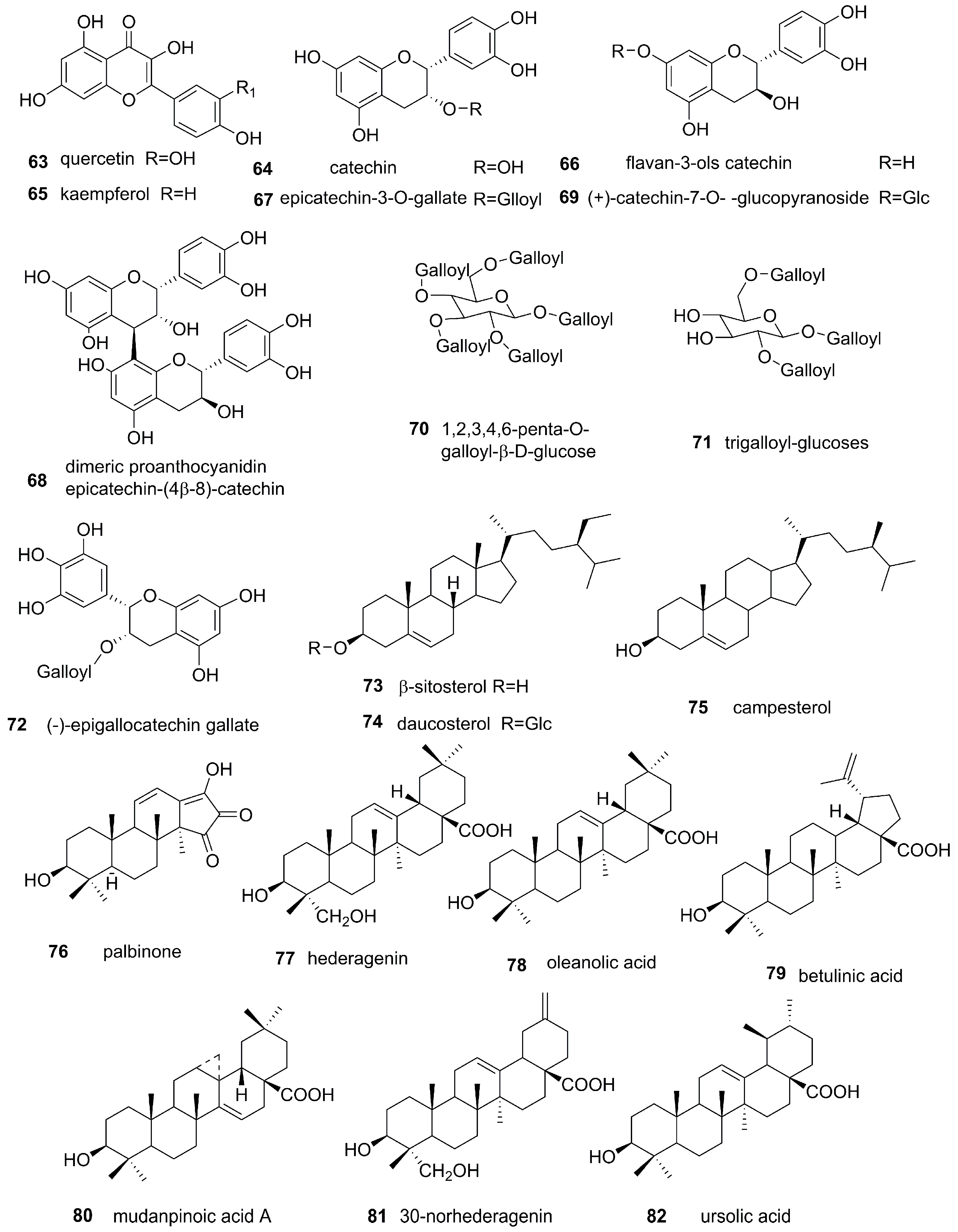
3.2. Flavonoids and Tannins
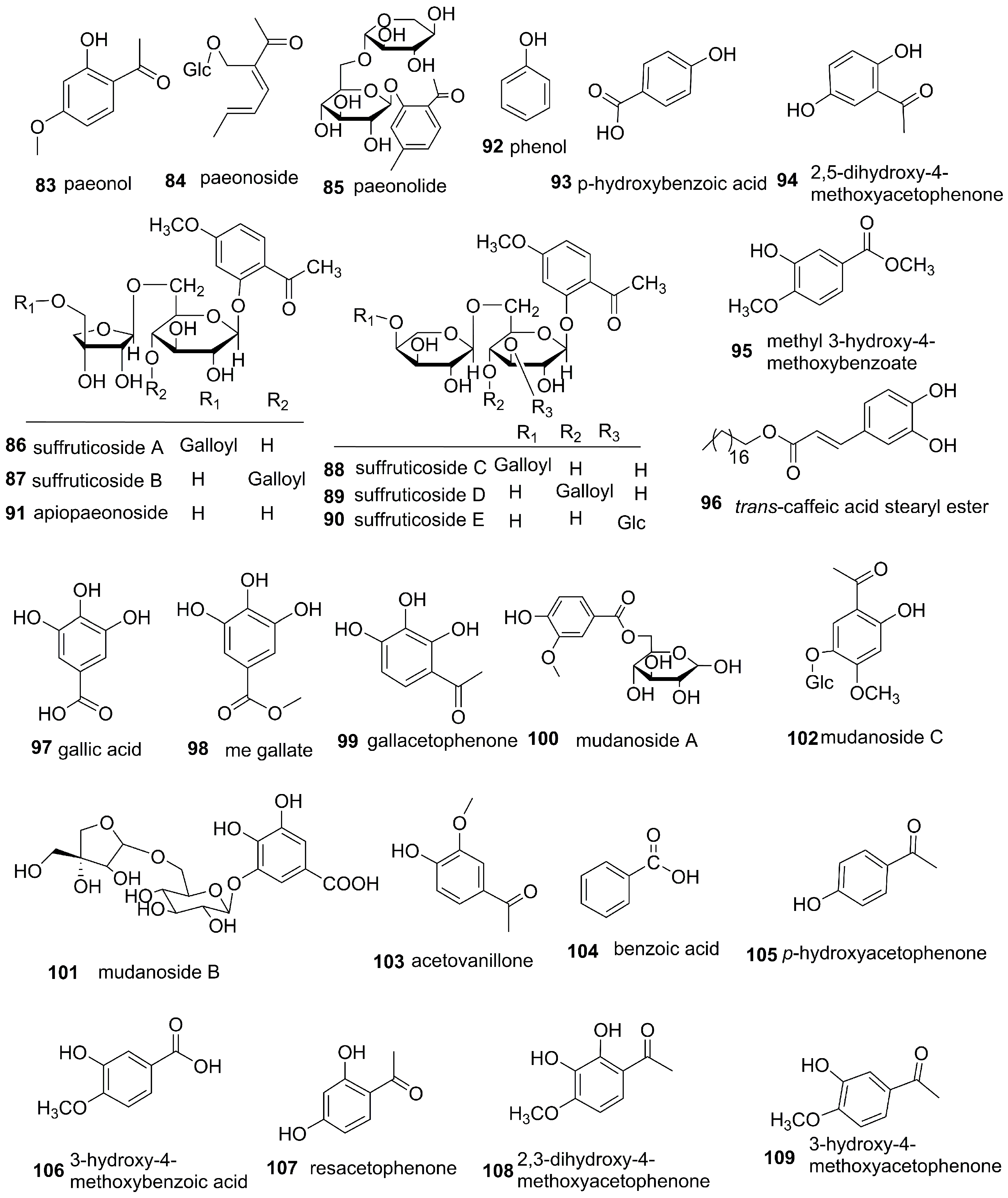
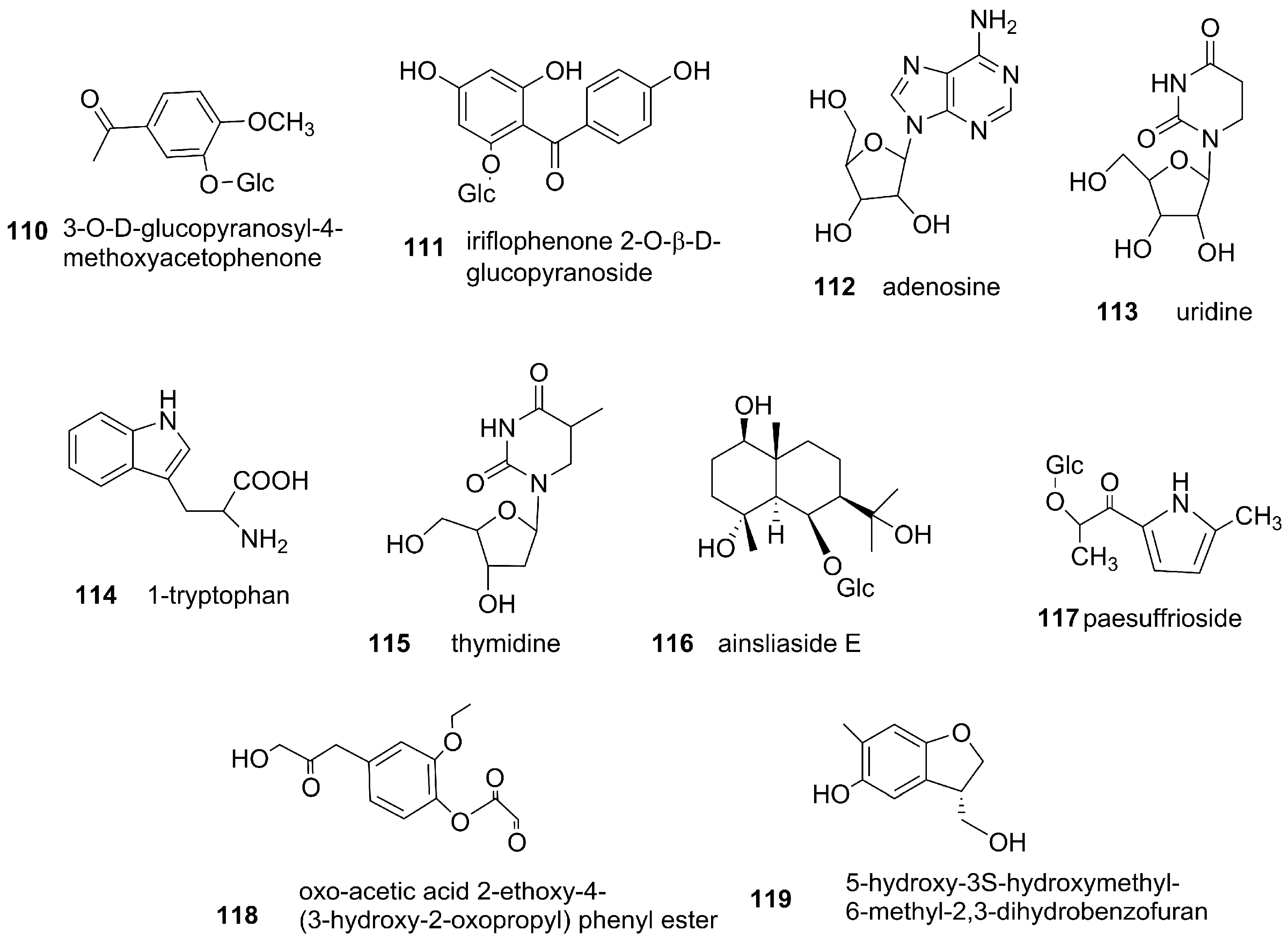
3.3. Phenols
3.4. Triterpenoids and Others
4. Pharmacological Activities of CM
4.1. Anti-Oxidative Effects
4.2. Anti-Inflammatory Effects
4.3. Anti-Tummor Effects
4.4. Cardiovascular System Protective Effects
4.5. Anti-Diabetic Activity
4.6. Neuroprotective Activity
4.7. Hepatoprotective Activity
4.8. Others
5. Analytical Methods for Quality Evaluation of CM
5.1. Quality Criteria of CM in Different Countries
5.2. Qualitative and Quantitative Analysis of CM
5.2.1. Thin-Layer Chromatography (TLC) Analysis
5.2.2. HPLC Analysis
5.2.3. LC-MS Analysis
5.2.4. GC Analysis
5.2.5. CE Analysis
5.2.6. Spectrometric Methods and Others
6. Safety
7. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Chinese Pharmacopeia Commission. Pharmacopoeia of the People’s Republic of China, English Edition; People’s Medical Publishing House: Beijing, China, 2015; Volume I. [Google Scholar]
- Fu, P.K.; Yang, C.Y.; Tsai, T.H.; Hsieh, C.L. Moutan cortex radicis improves lipopolysaccharide-induced acute lung injury in rats through anti-inflammation. Phytomedicine 2012, 19, 1206–1215. [Google Scholar] [CrossRef] [PubMed]
- Lim, T.K. Edible Medicinal and Non-Medicinal Plants; Springer: New York, NY, USA, 2012; Volume 8. [Google Scholar]
- List of Kampo Herbs. Available online: http://en.wikipedia.org/wiki/List_of_kampo_herbs (accessed on 5 June 2017).
- Matsuda, H.; Ohta, T.; Kawaguchi, A.; Yoshikawa, M. Bioactive constituents of chinese natural medicines. VI. Moutan cortex. (2): Structures and radical scavenging effects of suffruticosides A, B, C, D, and E and galloyl-oxypaeoniflorin. Chem. Pharm. Bull. 2001, 49, 69–72. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, M.; Uchida, E.; Kawaguchi, A.; Kitagawa, I.; Yamahara, J. Galloyl-oxypaeoniflorin, suffruticosides A, B, C, and D, five new antioxidative glycosides, and suffruticoside e, a paeonol glycoside, from chinese moutan cortex. Chem. Pharm. Bull. 1992, 40, 2248–2250. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Gu, Z. Screening of bioactive compounds from moutan cortex and their anti-inflammatory activities in rat synoviocytes. Evid.-Based Complement. Altern. Med. 2009, 6, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Yun, C.S.; Choi, Y.G.; Jeong, M.Y.; Lee, J.H.; Lim, S. Moutan cortex radicis inhibits inflammatory changes of gene expression in lipopolysaccharide-stimulated gingival fibroblasts. J. Nat. Med. 2013, 67, 576–589. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.; Nakano, Y.; Yatsuzuka, R.; Ono, R.; Kamei, C. Inhibitory effects of moutan cortex on immediate allergic reactions. Biol. Pharm. Bull. 2007, 30, 1707–1710. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.Y.; Hu, S.; Chan, B.C.; Wat, E.C.; Lau, C.; Hon, K.L.; Fung, K.P.; Leung, P.C.; Hui, P.C.; Lam, C.W. Anti-inflammatory and anti-allergic activities of pentaherb formula, moutan cortex (danpi) and gallic acid. Molecules (Basel Switzerland) 2013, 18, 2483–2500. [Google Scholar] [CrossRef] [PubMed]
- Rho, S.; Chung, H.S.; Kang, M.; Lee, E.; Cho, C.; Kim, H.; Park, S.; Kim, H.Y.; Hong, M.; Shin, M. Inhibition of production of reactive oxygen species and gene expression profile by treatment of ethanol extract of moutan cortex radicis in oxidative stressed pc12 cells. Biol. Pharm. Bull. 2005, 28, 661–666. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Feng, L.; Gu, J.; Ma, L.; Qin, D.; Wu, C.; Jia, X. The attenuation of moutan cortex on oxidative stress for renal injury in ages-induced mesangial cell dysfunction and streptozotocin-induced diabetic nephropathy rats. Oxid. Med. Cell. Longev. 2014, 2014, 463815. [Google Scholar] [CrossRef] [PubMed]
- Lan, B.; Li, J.; Duan, Q. An Encyclopedia of the Tree Peonies in China; China Science and Technology Press: Beijing, China, 2002. [Google Scholar]
- Zhang, J.; Wang, J.; Xia, T.; Zhou, S. DNA barcoding: Species delimitation in tree peonies. Sci. China Ser. C Life Sci. 2009, 52, 568–578. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Peng, B.; Dan, Y.; Peng, Y.; Xiao, P. Chemical taxonomy of tree peony species from china based on root cortex metabolic fingerprinting. Phytochemistry 2014, 107, 69–79. [Google Scholar] [CrossRef] [PubMed]
- Ha, D.T.; Ngoc, T.M.; Lee, I.; Lee, Y.M.; Kim, J.S.; Jung, H.; Lee, S.; Na, M.; Bae, K. Inhibitors of aldose reductase and formation of advanced glycation end-products in moutan cortex (Paeonia suffruticosa). J. Nat. Prod. 2009, 72, 1465–1470. [Google Scholar] [CrossRef] [PubMed]
- Ding, L.; Jiang, Z.; Liu, Y.; Chen, L.; Zhao, Q.; Yao, X.; Zhao, F.; Qiu, F. Monoterpenoid inhibitors of no production from Paeonia suffruticosa. Fitoterapia 2012, 83, 1598–1603. [Google Scholar] [CrossRef] [PubMed]
- Ding, L.; Zhao, F.; Chen, L.; Jiang, Z.; Liu, Y.; Li, Z.; Qiu, F.; Yao, X. New monoterpene glycosides from Paeonia suffruticosa andrews and their inhibition on NO production in LPS-induced RAW 264.7 cells. Bioorg. Med. Chem. Lett. 2012, 22, 7243–7247. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.H.; Wu, D.G.; Chen, Y.W. Chemical constituents and bioactivities of plants from the genus paeonia. Chem. Biodivers. 2010, 7, 90–104. [Google Scholar] [CrossRef] [PubMed]
- Ding, H.Y.; Wu, Y.C.; Lin, H.C.; Chan, Y.Y.; Wu, P.L.; Wu, T.S. Glycosides from Paeonia suffruticosa. Chem. Pharm. Bull. 1999, 47, 652–655. [Google Scholar] [CrossRef]
- Zhou, S.L.; Zou, X.H.; Zhou, Z.Q.; Liu, J.; Xu, C.; Yu, J.; Wang, Q.; Zhang, D.M.; Wang, X.Q.; Ge, S. Multiple species of wild tree peonies gave rise to the ‘king of flowers’, Paeonia suffruticosa andrews. Proc. R. Soc. Lond. B Biol. Sci. 2014, 281, 20141687. [Google Scholar] [CrossRef] [PubMed]
- An, R.B.; Kim, H.C.; Lee, S.H.; Jeong, G.S.; Sohn, D.H.; Park, H.; Kwon, D.Y.; Lee, J.H.; Kim, Y.C. A new monoterpene glycoside and antibacterial monoterpene glycosides from Paeonia suffruticosa. Arch. Pharm. Res. 2006, 29, 815. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Hu, H.Y.; Yu, N.J.; Zhang, Y.; Zhao, Y.M. Three new paeonidanin—Type monoterpene glycosides from Paeonia suffruticosa andr. Helv. Chim. Acta 2010, 93, 1622–1627. [Google Scholar] [CrossRef]
- Song, W.H.; Cheng, Z.H.; Chen, D.F. Anticomplement monoterpenoid glucosides from the root bark of Paeonia suffruticosa. J. Nat. Prod. 2013, 77, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Seo, C.S.; Lee, K.S.; Kim, H.J.; Chang, H.W.; Jung, J.S.; Song, D.K.; Son, J.K. Protective constituents against sepsis in mice from the root cortex of Paeonia suffruticosa. Arch. Pharm. Res. 2004, 27, 1123–1126. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.C.; Ding, H.Y.; Wu, Y.C. Two novel compounds from Paeonia suffruticosa. J. Nat. Prod. 1998, 61, 343–346. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.H.; Yoo, H.J.; Noh, I.C.; Lee, J.M.; Park, J.W.; Choi, W.S.; Choi, J.H. Bioassay-guided isolation of novel compound from Paeonia suffruticosa andrews roots as an IL-1β inhibitor. Arch. Pharm. Res. 2012, 35, 801–805. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Yang, Y.; Li, S.; Shi, J. A new paeoniflorin derivative isolated from the root bark ethanol extract of Paeonia suffruticosa. Zhongguo Zhong Yao Za Zhi 2005, 30, 759–761. [Google Scholar] [PubMed]
- Yoshikawa, M.; Harada, E.; Minematsu, T.; Muraoka, O.; Yamahara, J.; Murakami, N.; Kitagawa, I. Absolute stereostructures of paeonisothujone, a novel skeletal monoterpene ketone, and deoxypaeonisuffrone, and isopaeonisuffral, two new monoterpenes, from moutan cortex. Chem. Pharm. Bull. 1994, 42, 736–738. [Google Scholar] [CrossRef]
- Yoshikawa, M.; Ohta, T.; Kawaguchi, A.; Matsuda, H. Bioactive constituents of chinese natural medicines. V. Radical scavenging effect of moutan cortex. (1): Absolute stereostructures of two monoterpenes, paeonisuffrone and paeonisuffral. Chem. Pharm. Bull. 2000, 48, 1327–1331. [Google Scholar] [CrossRef] [PubMed]
- He, C.N.; Peng, Y.; Zhang, Y.C.; Xu, L.J.; Gu, J.; Xiao, P.G. Phytochemical and biological studies of paeoniaceae. Chem. Biodivers. 2010, 7, 805–838. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.C.; Ding, H.Y.; Wu, T.S.; Wu, P.L. Monoterpene glycosides from Paeonia suffruticosa. Phytochemistry 1996, 41, 237–242. [Google Scholar]
- Zhu, X.; Fang, Z.H. New monoterpene glycosides from the root cortex of Paeonia suffruticosa and their potential anti-inflammatory activity. Nat. Prod. Res. 2014, 28, 301–305. [Google Scholar] [CrossRef] [PubMed]
- Furuya, R.; Hu, H.; Zhang, Z.; Shigemori, H. Suffruyabiosides a and b, two new monoterpene diglycosides from moutan cortex. Molecules (Basel, Switzerland) 2012, 17, 4915–4923. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.J.; Lee, I.S.; Mar, W. Inhibition of inducible nitric oxide synthase and cyclooxygenase-2 activity by 1,2,3,4,6-penta-O-galloyl-β-d-glucose in murine macrophage cells. Arch. Pharm. Res. 2003, 26, 832–839. [Google Scholar] [CrossRef] [PubMed]
- Satoh, K.; Nagai, F.; Ushiyama, K.; Yasuda, I.; Seto, T.; Kano, I. Inhibition of Na+, K+-atpase by 1,2,3,4,6-penta-O-galloyl-β-d-glucose, a major constituent of both moutan cortex and paeoniae radix. Biochem. Pharmacol. 1997, 53, 611–614. [Google Scholar] [CrossRef]
- Wang, R.; Lechtenberg, M.; Sendker, J.; Petereit, F.; Deters, A.; Hensel, A. Wound-healing plants from tcm: In vitro investigations on selected TCM plants and their influence on human dermal fibroblasts and keratinocytes. Fitoterapia 2013, 84, 308–317. [Google Scholar] [CrossRef] [PubMed]
- Ding, L.; Zuo, Q.; Li, D.; Feng, X.; Gao, X.; Zhao, F.; Qiu, F. A new phenone from the roots of Paeonia suffruticosa andrews. Nat. Prod. Res. 2017, 31, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Xiao, K.; Song, Q.H.; Zhang, S.-W.; Xuan, L.J. A pyrrole derivative from Paeonia suffruticosa. Nat. Prod. Res. 2008, 22, 1614–1619. [Google Scholar] [CrossRef] [PubMed]
- Li, H.B.; Wong, C.C.; Cheng, K.W.; Chen, F. Antioxidant properties in vitro and total phenolic contents in methanol extracts from medicinal plants. LWT-Food Sci. Technol. 2008, 41, 385–390. [Google Scholar] [CrossRef]
- Li, H.; Xie, Y.H.; Yang, Q.; Wang, S.W.; Zhang, B.L.; Wang, J.B.; Cao, W.; Bi, L.L.; Sun, J.Y.; Miao, S. Cardioprotective effect of paeonol and danshensu combination on isoproterenol-induced myocardial injury in rats. PLoS ONE 2012, 7, e48872. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.H.; Lin, A.H.; Lee, H.F.; Ko, H.K.; Lee, T.S.; Kou, Y.R. Paeonol attenuates cigarette smoke-induced lung inflammation by inhibiting ros-sensitive inflammatory signaling. Mediat. Inflamm. 2014, 2014, 651890. [Google Scholar] [CrossRef] [PubMed]
- Li, C.R.; Zhou, Z.; Zhu, D.; Sun, Y.N.; Dai, J.M.; Wang, S.Q. Protective effect of paeoniflorin on irradiation-induced cell damage involved in modulation of reactive oxygen species and the mitogen-activated protein kinases. Int. J. Biochem. Cell Biol. 2007, 39, 426–438. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Zhu, X.; Qi, X.; Che, J.; Cao, B. Paeoniflorin protects human ea. Hy926 endothelial cells against gamma-radiation induced oxidative injury by activating the NF-E2-related factor 2/heme oxygenase-1 pathway. Toxicol. Lett. 2013, 218, 224–234. [Google Scholar] [CrossRef] [PubMed]
- Xie, W.; Yu, W.; Zhao, M.; Zhou, W.; Chen, H.; Du, W.; Huang, L.; Xu, Y.; Li, X. Protective effect of paeoniflorin against oxidative stress in human retinal pigment epithelium in vitro. Mol. Vis. 2011, 17, 3512–3533. [Google Scholar]
- Yao, C.W.; Piao, M.J.; Kim, K.C.; Zheng, J.; Cha, J.W.; Hyun, J.W. 6′-O-galloylpaeoniflorin protects human keratinocytes against oxidative stress-induced cell damage. Biomol. Ther. 2013, 21, 349. [Google Scholar] [CrossRef] [PubMed]
- Chun, S.C.; Jee, S.Y.; Lee, S.G.; Park, S.J.; Lee, J.R.; Kim, S.C. Anti-inflammatory activity of the methanol extract of moutan cortex in LPS-activated RAW264. 7 cells. Evid.-Based Complement. Altern. Med. 2007, 4, 327–333. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Chang, E.; Lee, Y.; Kim, J.; Kang, S.; Kim, H. A genome-wide microarray analysis reveals anti-inflammatory target genes of paeonol in macrophages. Inflamm. Res. 2008, 57, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Li, J.Z.; Wu, J.H.; Yu, S.Y.; Shao, Q.R.; Dong, X.M. Inhibitory effects of paeoniflorin on lysophosphatidylcholine-induced inflammatory factor production in human umbilical vein endothelial cells. Int. J. Mol. Med. 2013, 31, 493–497. [Google Scholar] [CrossRef] [PubMed]
- Oh, G.; Pae, H.; Choi, B.; Jeong, S.; Oh, H.; Oh, C.; Rho, Y.; Kim, D.; Shin, M.; Chung, H.-T. Inhibitory effects of the root cortex of Paeonia suffruticosa on interleukin-8 and macrophage chemoattractant protein-1 secretions in U937 cells. J. Ethnopharmacol. 2003, 84, 85–89. [Google Scholar] [CrossRef]
- Chen, N.; Liu, D.; Soromou, L.W.; Sun, J.; Zhong, W.; Guo, W.; Huo, M.; Li, H.; Guan, S.; Chen, Z. Paeonol suppresses lipopolysaccharide—Induced inflammatory cytokines in macrophage cells and protects mice from lethal endotoxin shock. Fundam. Clin. Pharmacol. 2014, 28, 268–276. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Fan, L.L.; Sun, G.-P.; Wan, X.A.; Wang, Z.G.; Wu, Q.; Wang, H. Paeonol inhibits tumor growth in gastric cancer in vitro and in vivo. World J. Gastroenterol. 2010, 16, 4483–4490. [Google Scholar] [CrossRef] [PubMed]
- Sun, G.P.; Wan, X.; Xu, S.P.; Wang, H.; Liu, S.H.; Wang, Z.G. Antiproliferation and apoptosis induction of paeonol in human esophageal cancer cell lines. Dis. Esophagus 2008, 21, 723–729. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Lee, H. Inhibitory effects of Paeonia suffruticosa andrews extracts on VEGF binding to vegf receptor. Nat. Prod. Sci. 2007, 13, 128. [Google Scholar]
- Lin, M.Y.; Shen, C.H.; Chiang, S.Y.; Chen, S.Y.; Lin, Y.S.; Hsu, C.D. Cortex moutan inhibits bladder cancer cell proliferation and expression of angiogenic factors. Pharmacol. Pharm. 2014, 5, 846–858. [Google Scholar] [CrossRef]
- Horng, C.T.; Shieh, P.C.; Tan, T.W.; Yang, W.H.; Tang, C.H. Paeonol suppresses chondrosarcoma metastasis through up-regulation of miR-141 by modulating PKCδ and c-Src signaling pathway. Int. J. Mol. Sci. 2014, 15, 11760–11772. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Tao, L.; Shi, T.; Zhang, F.; Sheng, X.; Cao, Y.; Zheng, S.; Wang, A.; Qian, W.; Jiang, L. Paeonol inhibits B16F10 melanoma metastasis in vitro and in vivo via disrupting proinflammatory cytokines-mediated NF-κB and STAT3 pathways. IUBMB Life 2015, 67, 778–788. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.P.; Sun, G.P.; Shen, Y.X.; Wei, W.; Peng, W.R.; Wang, H. Antiproliferation and apoptosis induction of paeonol in HepG2 cells. World J. Gastroenterol. 2007, 13, 250. [Google Scholar] [CrossRef] [PubMed]
- Ou, Y.; Li, Q.; Wang, J.; Li, K.; Zhou, S. Antitumor and apoptosis induction effects of paeonol on mice bearing EMT6 breast carcinoma. Biomol. Ther. 2014, 22, 341–346. [Google Scholar] [CrossRef] [PubMed]
- Sun, G.P.; Wang, H.; Xu, S.P.; Shen, Y.X.; Wu, Q.; Chen, Z.D.; Wei, W. Anti-tumor effects of paeonol in a hepa-hepatoma bearing mouse model via induction of tumor cell apoptosis and stimulation of IL-2 and TNF-α production. Eur. J. Pharmacol. 2008, 584, 246–252. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.; Chen, S.; Zhang, W.; Hu, S.; Lu, J.; Xing, J.; Dong, Y. Paeonol reverses paclitaxel resistance in human breast cancer cells by regulating the expression of transgelin 2. Phytomedicine 2014, 21, 984–991. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Tan, S.-Y.; Wang, X.-F. Paeonol exerts an anticancer effect on human colorectal cancer cells through inhibition of PGE2 synthesis and cox-2 expression. Oncol. Rep. 2014, 32, 2845–2853. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Yuan, Y.; Cui, J.; Xiao, T.; Jiang, D. Paeoniflorin inhibits proliferation and invasion of breast cancer cells through suppressing Notch-1 signaling pathway. Biomed. Pharmacother. 2016, 78, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Chen, G.-L.; Li, Y.-J.; Chen, Y.; Lin, F.-Z. Paeoniflorin inhibits macrophage-mediated lung cancer metastasis. Chin. J. Nat. Med. 2015, 13, 925–932. [Google Scholar] [CrossRef]
- Li, W.; Qi, Z.; Wei, Z.; Liu, S.; Wang, P.; Chen, Y.; Zhao, Y. Paeoniflorin inhibits proliferation and induces apoptosis of human glioma cells via microrna-16 upregulation and matrix metalloproteinase-9 downregulation. Mol. Med. Rep. 2015, 12, 2735–2740. [Google Scholar] [CrossRef] [PubMed]
- Oh, G.S.; Pae, H.O.; Oh, H.; Hong, S.G.; Kim, I.K.; Chai, K.Y.; Yun, Y.G.; Kwon, T.O.; Chung, H.T. In vitro anti-proliferative effect of 1,2,3,4,6-penta-O-galloyl-β-d-glucose on human hepatocellular carcinoma cell line, sk-hep-1 cells. Cancer Lett. 2001, 174, 17–24. [Google Scholar] [CrossRef]
- Pan, J.Y.; Cheng, Y.Y. Identification and analysis of absorbed and metabolic components in rat plasma after oral administration of ‘shuangdan’ granule by HPLC–DAD–ESI-MS/MS. J. Pharm. Biomed. Anal. 2006, 42, 565–572. [Google Scholar] [CrossRef] [PubMed]
- Dan, H.; Zhang, L.; Qin, X.; Peng, X.; Wong, M.; Tan, X.; Yu, S.; Fang, N. Moutan cortex extract exerts protective effects in a rat model of cardiac ischemia/reperfusion. Can. J. Physiol. Pharmacol. 2015, 94, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Chuang, C.C.; Weng, W.; Zhao, L.; Zheng, Y.; Zhang, J.; Zuo, L. Paeonol protects rat heart by improving regional blood perfusion during no-reflow. Front. Physiol. 2016, 7, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.j.; Bao, J.X.; Xu, J.w.; Murad, F.; Bian, K. Vascular dilation by paeonol—A mechanism study. Vasc. Pharmacol. 2010, 53, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Ye, S.; Liu, X.; Mao, B.; Yang, L.; Liu, N. Paeonol enhances thrombus recanalization by inducing vascular endothelial growth factor 165 via ERK1/2 MAPK signaling pathway. Mol. Med. Rep. 2016, 13, 4853–4858. [Google Scholar] [PubMed]
- Ye, S.; Mao, B.; Yang, L.; Fu, W.; Hou, J. Thrombosis recanalization by paeoniflorin through the upregulation of urokinase-type plasminogen activator via the MAPK signaling pathway. Mol. Med. Rep. 2016, 13, 4593–4598. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.Q.; Dai, M.; Zhong, J.C.; Yin, D.K. Paeonol inhibits oxidized low density lipoprotein-induced monocyte adhesion to vascular endothelial cells by inhibiting the mitogen activated protein kinase pathway. Biol. Pharm. Bull. 2012, 35, 767–772. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Du, P.; Wang, J. Paeoniflorin ameliorates acute myocardial infarction of rats by inhibiting inflammation and inducible nitric oxide synthase signaling pathways. Mol. Med. Rep. 2015, 12, 3937–3943. [Google Scholar] [PubMed]
- Chen, T.; Guo, Z.P.; Wang, L.; Qin, S.; Cao, N.; Li, M.m.; Jia, R.Z.; Wang, T.T. Paeoniflorin suppresses vascular damage and the expression of E-selectin and ICAM-1 in a mouse model of cutaneous arthus reaction. Exp. Dermatol. 2013, 22, 453–457. [Google Scholar] [CrossRef] [PubMed]
- Koo, Y.K.; Kim, J.M.; Koo, J.Y.; Kang, S.S.; Bae, K.; Kim, Y.S.; Chung, J.-H.; Yun-Choi, H.S. Platelet anti-aggregatory and blood anti-coagulant effects of compounds isolated from Paeonia lactiflora and Paeonia suffruticosa. Die Pharm. Int. J. Pharm. Sci. 2010, 65, 624–628. [Google Scholar]
- Hsu, P.C.; Tsai, Y.T.; Lai, J.N.; Wu, C.T.; Lin, S.K.; Huang, C.Y. Integrating traditional chinese medicine healthcare into diabetes care by reducing the risk of developing kidney failure among type 2 diabetic patients: A population-based case control study. J. Ethnopharmacol. 2014, 156, 358–364. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.H.; Feng, L.; Zhu, M.M.; Gu, J.F.; Jiang, J.; Cheng, X.D.; Ding, S.M.; Wu, C.; Jia, X.B. The anti-inflammation effect of moutan cortex on advanced glycation end products-induced rat mesangial cells dysfunction and high-glucose-fat diet and streptozotocin-induced diabetic nephropathy rats. J. Ethnopharmacol. 2014, 151, 591–600. [Google Scholar] [CrossRef] [PubMed]
- Lau, C.; Chan, C.; Chan, Y.; Lau, K.; Lau, T.; Lam, F.; Law, W.; Che, C.; Leung, P.; Fung, K. Pharmacological investigations of the anti-diabetic effect of cortex moutan and its active component paeonol. Phytomedicine 2007, 14, 778–784. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wang, S.; Feng, L.; Ma, D.; Fu, Q.; Song, Y.; Jia, X.; Ma, S. Hypoglycemic and antioxidant activities of paeonol and its beneficial effect on diabetic encephalopathy in streptozotocin-induced diabetic rats. J. Med. Food 2013, 16, 577–586. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; Li, Y.; Wang, L.; Gao, B.; Zhang, N.; Ji, Q. Paeoniflorin prevents diabetic nephropathy in rats. Comp. Med. 2009, 59, 557–566. [Google Scholar] [PubMed]
- Tuan, D.T.; Thu, N.B.; Nhiem, N.X.; Ngoc, T.M.; Yim, N.; Bae, K. Palbinone and triterpenes from moutan cortex (Paeonia suffruticosa, paeoniaceae) stimulate glucose uptake and glycogen synthesis via activation of AMPK in insulin-resistant human HepG2 cells. Bioorg. Med. Chem. Lett. 2009, 19, 5556–5559. [Google Scholar]
- Trung, T.N.; Hien, T.T.; Dao, T.T.; Yim, N.; Ngoc, T.M.; Oh, W.K.; Bae, K. Selected compounds derived from moutan cortex stimulated glucose uptake and glycogen synthesis via AMPK activation in human HepG2 cells. J. Ethnopharmacol. 2010, 131, 417–424. [Google Scholar]
- Zhao, G.; Shen, Y.; Ma, J.; Li, F.; Shi, X. Protection of polysaccharides-2b from mudan cortex of Paeonia suffruticosa andr on diabetic cataract in rats. Zhongguo Zhong Yao Za Zhi 2007, 32, 2036–2039. [Google Scholar] [PubMed]
- Tatsumi, S.; Mabuchi, T.; Abe, T.; Xu, L.; Minami, T.; Ito, S. Analgesic effect of extracts of chinese medicinal herbs moutan cortex and coicis semen on neuropathic pain in mice. Neurosci. Lett. 2004, 370, 130–134. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.G.; Park, G.; Piao, Y.; Kang, M.S.; Pak, Y.K.; Hong, S.P.; Oh, M.S. Effects of the root bark of Paeonia suffruticosa on mitochondria-mediated neuroprotection in an MPTP-induced model of parkinson’s disease. Food Chem. Toxicol. 2014, 65, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.B.; Song, N.N.; Wei, X.B.; Guan, H.S.; Zhang, X.M. Protective effects of paeonol on cultured rat hippocampal neurons against oxygen–glucose deprivation-induced injury. J. Neurol. Sci. 2008, 264, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Su, S.-Y.; Cheng, C.Y.; Tsai, T.H.; Hsiang, C.Y.; Ho, T.Y.; Hsieh, C.L. Paeonol attenuates H2O2-induced NF-κB-associated amyloid precursor protein expression. Am. J. Chin. Med. 2010, 38, 1171–1192. [Google Scholar] [CrossRef] [PubMed]
- Himaya, S.; Ryu, B.; Qian, Z.J.; Kim, S.K. Paeonol from hippocampus kuda bleeler suppressed the neuro-inflammatory responses in vitro via NF-κB and MAPK signaling pathways. Toxicol. In Vitro 2012, 26, 878–887. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.; Lin, H.Y.; Chen, J.H.; Tseng, W.P.; Ko, P.Y.; Liu, Y.S.; Yeh, W.L.; Lu, D.Y. Effects of paeonol on anti-neuroinflammatory responses in microglial cells. Int. J. Mol. Sci. 2015, 16, 8844–8860. [Google Scholar] [CrossRef] [PubMed]
- Zhong, S.Z.; Ge, Q.H.; Qu, R.; Li, Q.; Ma, S.P. Paeonol attenuates neurotoxicity and ameliorates cognitive impairment induced by d-galactose in icr mice. J. Neurol. Sci. 2009, 277, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Tseng, Y.T.; Hsu, Y.Y.; Shih, Y.T.; Lo, Y.C. Paeonol attenuates microglia-mediated inflammation and oxidative stress–induced neurotoxicity in rat primary microglia and cortical neurons. Shock (Augusta Ga.) 2012, 37, 312–318. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Zhou, L.; Hou, D.; Tang, J.; Sun, J.; Bondy, S.C. Paeonol increases levels of cortical cytochrome oxidase and vascular actin and improves behavior in a rat model of alzheimer’s disease. Brain Res. 2011, 1388, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Guo, R.B.; Wang, G.F.; Zhao, A.P.; Gu, J.; Sun, X.L.; Hu, G. Paeoniflorin protects against ischemia-induced brain damages in rats via inhibiting MAPKs/NF-κB-mediated inflammatory responses. PLoS ONE 2012, 7, e49701. [Google Scholar] [CrossRef] [PubMed]
- Nam, K.N.; Yae, C.G.; Hong, J.W.; Cho, D.H.; Lee, J.H.; Lee, E.H. Paeoniflorin, a monoterpene glycoside, attenuates lipopolysaccharide-induced neuronal injury and brain microglial inflammatory response. Biotechnol. Lett. 2013, 35, 1183–1189. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.Z.; Zhu, J.; Jin, D.Z.; Zhang, L.M.; Ji, X.Q.; Ye, Y.; Tang, C.P.; Zhu, X.Z. Behavioral recovery following sub-chronic paeoniflorin administration in the striatal 6-OHDA lesion rodent model of parkinson’s disease. J. Ethnopharmacol. 2007, 112, 327–332. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.Q.; Zhang, W.Y.; Luo, X.T.; Ye, Y.; Zhu, X.Z. Paeoniflorin attenuates neuroinflammation and dopaminergic neurodegeneration in the MPTP model of parkinson’s disease by activation of adenosine a1 receptor. Br. J. Pharmacol. 2006, 148, 314–325. [Google Scholar] [CrossRef] [PubMed]
- Sun, R.; Wang, K.; Wu, D.; Li, X.; Ou, Y. Protective effect of paeoniflorin against glutamate-induced neurotoxicity in PC12 cells via Bcl-2/bax signal pathway. Folia Neuropathol. 2012, 50, 270–276. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Cao, Y.B.; Hu, L.F.; Yang, Y.P.; Li, J.; Wang, F.; Liu, C.F. Asics mediate the modulatory effect by paeoniflorin on alpha-synuclein autophagic degradation. Brain Res. 2011, 1396, 77–87. [Google Scholar] [CrossRef] [PubMed]
- Dong, H.; Li, R.; Yu, C.; Xu, T.; Zhang, X.; Dong, M. Paeoniflorin inhibition of 6-hydroxydopamine-induced apoptosis in PC12 cells via suppressing reactive oxygen species-mediated pkcδ/NF-κB pathway. Neuroscience 2015, 285, 70–80. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Tan, Q.R.; Zhang, Z.J. Neuroprotective effects of paeoniflorin, but not the isomer albiflorin, are associated with the suppression of intracellular calcium and calcium/calmodulin protein kinase ii in PC12 cells. J. Mol. Neurosci. 2013, 51, 581–590. [Google Scholar] [CrossRef] [PubMed]
- Fujiwara, H.; Tabuchi, M.; Yamaguchi, T.; Iwasaki, K.; Furukawa, K.; Sekiguchi, K.; Ikarashi, Y.; Kudo, Y.; Higuchi, M.; Saido, T.C. A traditional medicinal herb Paeonia suffruticosa and its active constituent 1,2,3,4,6-penta-O-galloyl-β-d-glucopyranose have potent anti-aggregation effects on alzheimer’s amyloid β proteins in vitro and in vivo. J. Neurochem. 2009, 109, 1648–1657. [Google Scholar] [CrossRef] [PubMed]
- Choi, B.M.; Kim, H.J.; Oh, G.S.; Pae, H.O.; Oh, H.; Jeong, S.; Kwon, T.O.; Kim, Y.M.; Chung, H.-T. 1,2,3,4,6-penta-O-galloyl-β-d-glucose protects rat neuronal cells (neuro 2a) from hydrogen peroxide-mediated cell death via the induction of heme oxygenase-1. Neurosci. Lett. 2002, 328, 185–189. [Google Scholar] [CrossRef]
- Shon, Y.H.; Nam, K.S. Protective effect of moutan cortex extract on acetaminophen-induced hepatotoxicity in mice. J. Ethnopharmacol. 2004, 90, 415–419. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Xue, X.; Zhang, B.; Jiang, W.; Cao, H.; Wang, R.; Sun, D.; Guo, R. The protective effects of paeonol against epirubicin-induced hepatotoxicity in 4t1-tumor bearing mice via inhibition of the PI3K/Akt/NF-κB pathway. Chem.-Biol. Interact. 2016, 244, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Shen, G.; Zhao, W.; Wang, F.; Jiang, X.; Huang, D. Paeonol, the main active principles of Paeonia moutan, ameliorates alcoholic steatohepatitis in mice. J. Ethnopharmacol. 2010, 128, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Cao, L.; Luo, Y.; Feng, X.; Sun, L.; Wen, M.; Peng, S. Paeoniflorin protects against concanavalin a-induced hepatitis in mice. Int. Immunopharmacol. 2015, 24, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Ma, X.; Wang, J.; Zhu, Y.; Li, R.; Wang, J.; He, X.; Shan, L.; Wang, R.; Wang, L. Paeoniflorin alleviates liver fibrosis by inhibiting HIF-1α through mtor-dependent pathway. Fitoterapia 2014, 99, 318–327. [Google Scholar] [CrossRef] [PubMed]
- Espiritu, A.G.; Doma, B.T., Jr.; Wang, Y.F.; Camille, F. Efficacy of methanol extracts of mentha haplocalyx briq. And Paeonia suffruticosa andr. For potential antibacterial activity. Sustain. Environ. Res. 2014, 24, 319–324. [Google Scholar]
- Yang, J.F.; Yang, C.H.; Chang, H.W.; Yang, C.S.; Lin, C.W.; Chuang, L.Y. Antioxidant and antibacterial properties of pericarpium trichosanthis against nosocomial drug resistant strains of acinetobacter baumannii in taiwan. J. Med. Plants Res. 2009, 3, 982–991. [Google Scholar]
- Hong, M.H.; Kim, J.H.; Na, S.H.; Bae, H.; Shin, Y.C.; Kim, S.H.; Ko, S.G. Inhibitory effects of Paeonia suffruticosa on allergic reactions by inhibiting the nf-kappab/ikappab-alpha signaling pathway and phosphorylation of erk in an animal model and human mast cells. Biosci. Biotechnol. Biochem. 2010, 74, 1152–1156. [Google Scholar] [CrossRef] [PubMed]
- Zhai, T.; Sun, Y.; Li, H.; Zhang, J.; Huo, R.; Li, H.; Shen, B.; Li, N. Unique immunomodulatory effect of paeoniflorin on type i and ii macrophages activities. J. Pharmacol. Sci. 2016, 130, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Wang, B.E.; Zhang, S.W.; Yang, S.M.; Wang, H.; Ren, A.M.; Yi, E.T. Isolation of antifungal compound from Paeonia suffruticosa and its antifungal mechanism. Chin. J. Integr. Med. 2015, 21, 211–216. [Google Scholar] [CrossRef] [PubMed]
- Ishiguro, K.; Ando, T.; Maeda, O.; Hasegawa, M.; Kadomatsu, K.; Ohmiya, N.; Niwa, Y.; Xavier, R.; Goto, H. Paeonol attenuates tnbs-induced colitis by inhibiting NF-κB and stat1 transactivation. Toxicol. Appl. Pharmacol. 2006, 217, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.R.; Sun, Y.; Luo, Y.J.; Zhao, X.; Chen, J.F.; Yanagawa, Y.; Qu, W.M.; Huang, Z.L. Paeoniflorin promotes non-rapid eye movement sleep via adenosine a1 receptors. J. Pharmacol. Exp. Therap. 2016, 356, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Huang, L.; Zhu, J.; Bu, W.; Sun, J.; Fang, Z. Screening nephroprotective compounds from cortex moutan by mesangial cell extraction and uplc. Arch. Pharma. Res. 2015, 38, 1044–1053. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.L.; Huang, L.M.; Wen-Jie, B.U.; Kai, G.Q.; Fan, H.W.; Xian-Hua, L.I.; Sun, M. Analysis of bioactive components in moutan cortex by mesangial cell membrane immobilized chromatography. J. Anhui Univ. 2013, 37, 104–108. [Google Scholar]
- Comparative Studies on Pharmacopoeial Definitions, Requirements and Information for Crude Drugs among FHH Member Countries in 2007. Available online: http://www.nihs.go.jp/dpp/FHH/FHH.htm (accessed on 5 June 2017).
- El Babili, F.; El Babili, M.; Fouraste, I.; Chatelain, C. Peonies: Comparative study by anatomy and tlc of three traditional chinese medicinal plants. Chin. Med. 2013, 4, 166–172. [Google Scholar] [CrossRef]
- Zhang, R. Application of tlc characterastic chromatography in the quality control of cortex moutan. CJTCM 2013, 25, 539–541. [Google Scholar]
- Xu, S.J.; Yang, L.; Zeng, X.; Zhang, M.; Wang, Z.T. Characterization of compounds in the chinese herbal drug Mu-Dan-Pi by liquid chromatography coupled to electrospray ionization mass spectrometry. Rapid Commun. Mass Spectrom. 2006, 20, 3275–3288. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.-Q. HPLC with uv switch determination of 8 indicative components in combination extracts of persicae ramulus and paeoniae radix alba. Chin. J. Pharm. Anal. 2013, 33, 1899–1903. [Google Scholar]
- Wang, S.; Huang, J.; Mao, H.; Wang, Y.; Kasimu, R.; Xiao, W.; Wang, J. A novel method HPLC-DAD analysis of the contentsof moutan cortexand paeoniae radix alba with similar constituents-monoterpene glycosides in guizhi fuling wan. Molecules (Basel Switzerland) 2014, 19, 17957–17967. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Wu, E.; Chen, J.; Nguyen, H.T.; Do, T.H.; Park, K.L.; Bae, K.H.; Kim, Y.H.; Kang, J.S. Quality evaluation of moutan cortex radicis using multiple component analysisby high performance liquid chromatography. Bull. Korean Chem. Soc. 2009, 30, 2240–2244. [Google Scholar]
- Chunnian, H.; Yong, P.; Yuxiong, F.; Bing, P.; Zhe, W.; Peigen, X. Quick comparison of radix paeonia alba, radix paeonia rubra, and cortex moutan by high performance liquid chromatography coupled with monolithic columns and their chemical pattern recognition. Pharmacogn. Mag. 2012, 8, 237. [Google Scholar] [PubMed]
- Wu, M.; He, Y.; Tang, L.; Wang, Z. Clustering analysis of HPLC chromatographic fingerprints of Paeonia suffruticosa. Zhongguo Zhong Yao Za Zhi 2007, 32, 1054–1056. [Google Scholar] [PubMed]
- Hu, Y.-F.; Xu, Q.; Xu, G.-B.; Jiang, L.; Wu, H. Comparison of UPLC fingerprint of cortex moutan before and after stir-frying. China Pharm. 2015, 26, 800–802. [Google Scholar]
- Fan, X.; Wang, Z.; Li, Q.; Ma, T.; Bi, K.; Jia, Y. UPLC characteristic chromatographic profile of moutan cortex. Zhongguo Zhong Yao Za Zhi 2011, 36, 715–717. [Google Scholar] [PubMed]
- Ye, J.; Zhang, X.; Dai, W.; Yan, S.; Huang, H.; Liang, X.; Li, Y.; Zhang, W. Chemical fingerprinting of liuwei dihuang pill and simultaneous determination of its major bioactive constituents by HPLC coupled with multiple detections of DAD, ELSD and ESI-MS. J. Pharm. Biomed. Anal. 2009, 49, 638–645. [Google Scholar] [CrossRef] [PubMed]
- Luo, N.; Ding, W.; Wu, J.; Qian, D.; Li, Z.; Qian, Y.; Guo, J.; Duan, J. UPLC-Q-TOF/MS coupled with multivariate statistical analysis as a powerful technique for rapidly exploring potential chemical markers to differentiate between radix paeoniae alba and radix paeoniae rubra. Nat. Prod. Commun. 2013, 8, 487–491. [Google Scholar] [PubMed]
- He, Q.; Ge, Z.W.; Song, Y.; Cheng, Y.Y. Quality evaluation of cortex moutan by high performance liquid chromatography coupled with diode array detector and electrospary ionization tandem mass spectrometry. Chem. Pharm. Bull. 2006, 54, 1271–1275. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; He, Q.; Li, P.; Cheng, Y.Y. Capillary high performance liquid chromatography coupled with electrospray ionization mass spectrometry for rapid analysis of pinane monoterpene glycosides in cortex moutan. J. Sep. Sci. 2008, 31, 64–70. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.M.; Yu, J.Y.; Ding, M.J.; Zhao, M.; Xue, X.Y.; Che, C.T.; Wang, S.M.; Zhao, B.; Meng, J. Liquid chromatography-diode array detector-electrospray mass spectrometry and principal components analyses of raw and processed moutan cortex. Pharmacog. Mag. 2016, 12, 50. [Google Scholar]
- Li, X.Y.; Duan, S.M.; Liu, H.H.; XU, J.-D.; Kong, M.; Liu, H.Q.; Li, S.L. Sulfur-containing derivatives as characteristic chemical markers in control of sulfur-fumigated moutan cortex. Acta Pharm. Sin. 2016, 51, 972–978. [Google Scholar]
- Gao, X.; Sun, L.; Qiao, S.; Gao, S.; Che, Y.; Zhang, K. HPLC fingerprint of liuwei dihuang condensed pills. Zhongguo Zhong Yao Za Zhi 2012, 37, 3411–3415. [Google Scholar] [PubMed]
- Lu, X.; Zhao, X.; Bai, C.; Zhao, C.; Lu, G.; Xu, G. LC-MS-based metabonomics analysis. J. Chromatogr. B 2008, 866, 64–76. [Google Scholar] [CrossRef] [PubMed]
- Li, X.Y.; Xu, J.D.; Xu, J.; Kong, M.; Zhou, S.S.; Mao, Q.; Brand, E.; Chen, H.B.; Liu, H.Q.; Li, S.L. Uplc-qtof-ms based metabolomics coupled with the diagnostic ion exploration strategy for rapidly evaluating sulfur-fumigation caused holistic quality variation in medicinal herbs, moutan cortex as an example. Anal. Methods 2016, 8, 1034–1043. [Google Scholar] [CrossRef]
- Xiao, C.; Wu, M.; Chen, Y.; Zhang, Y.; Zhao, X.; Zheng, X. Revealing metabolomic variations in cortex moutan from different root parts using HPLC-MS method. Phytochem. Anal. 2015, 26, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.H.; Sun, H.; Zhang, A.H.; Yan, G.L.; Han, Y.; Xue, C.S.; Zhou, X.H.; Shi, H.; Wang, X.J. Serum pharmacochemistry combined with multiple data processing approach to screen the bioactive components and their metabolites in mutan cortex by ultra-performance liquid chromatography tandem mass spectrometry. Biomed. Chromatogr. 2014, 28, 500–510. [Google Scholar] [CrossRef]
- Jiang, Y.; David, B.; Tu, P.; Barbin, Y. Recent analytical approaches in quality control of traditional chinese medicines—A review. Anal. Chim. Acta 2010, 657, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.F.; Wu, D.L.; Xu, G.B. Comparative analysis of moutan cortex extract collected from five regions by GC-MS and chemometrics. Chin. Tradit. Patent Med. 2015, 37, 2003–2007. [Google Scholar]
- Ganzera, M. Quality control of herbal medicines by capillary electrophoresis: Potential, requirements and applications. Electrophoresis 2008, 29, 3489–3503. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.T.; Huang, W.Y.; Lin, T.C.; Sheu, S.J. Determination of moutan tannins by high-performance liquid chromatography and capillary electrophoresis. J. Sep. Sci. 2003, 26, 1629–1634. [Google Scholar] [CrossRef]
- Yu, K.; Wang, Y.; Cheng, Y. Determination of the active components in chinese herb cortex moutan by MEKC and LC. Chromatographia 2006, 63, 359–364. [Google Scholar] [CrossRef]
- Holland, L.A.; Leigh, A.M. Amperometric and voltammetric detection for capillary electrophoresis. Electrophoresis 2002, 23, 3649–3658. [Google Scholar] [CrossRef]
- Chen, G.; Zhang, L.; Zhu, Y. Determination of glycosides and sugars in moutan cortex by capillary electrophoresis with electrochemical detection. J. Pharm. Biomed. Anal. 2006, 41, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Gang, C.; Zhang, L.; Pengyuan, Y. Determination of three bioactive constituents in moutan cortex by capillary electrophoresis with electrochemical detection. Anal. Sci. 2005, 21, 1161–1165. [Google Scholar]
- Chen, G.; Zhang, L.; Wu, X.; Ye, J. Determination of mannitol and three sugars in ligustrum lucidum ait. By capillary electrophoresis with electrochemical detection. Anal. Chim. Acta 2005, 530, 15–21. [Google Scholar] [CrossRef]
- Tanaka, R.; Shibata, H.; Sugimoto, N.; Akiyama, H.; Nagatsu, A. Application of a quantitative 1h-nmr method for the determination of paeonol in moutan cortex, hachimijiogan and keishibukuryogan. J. Nat. Med. 2016, 70, 797–802. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Rong, Z.M.; Ma, C.X.; Zhao, X.F.; Xiao, C.N.; Zheng, X.H. Distribution of metabolites in root barks of seven tree peony cultivars for quality assessment using nmr-based metabolomics. Chin. Herb. Med. 2017, 9, 31–41. [Google Scholar] [CrossRef]
- Zhao, S.Q. Fourier Transform Infrared(ftir) Spectroscopy Investigation of Tree Peony Flowers and Cortex Moutan. Master’s Thesis, Yunnan Normal University, Yunnan, China, 2015. [Google Scholar]
- Yang, S.; Qu, L.; Yang, R.; Li, J.; Yu, L. Modified glassy carbon electrode with nafion/mwnts as a sensitive voltammetric sensor for the determination of paeonol in pharmaceutical and biological samples. J. Appl. Electrochem. 2010, 40, 1371–1378. [Google Scholar] [CrossRef]
- Dong, W.; Shen, H.B.; Liu, X.H.; Li, M.J.; Li, L.S. Cdse/zns quantum dots based fluorescence quenching method for determination of paeonol. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2011, 78, 537–542. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Chen, J.; Liang, Q.; Wu, D.; Zeng, Y.; Jiang, B. Znse quantum dots based fluorescence quenching method for determination of paeoniflorin. J. Lumin. 2014, 145, 569–574. [Google Scholar] [CrossRef]
- Jiang, L.; Yunfei, H.U.; Pei, Y.; Guobing, X.U.; Deling, W.U.; Qian, X.U.; Pharmacy, S.O. Comparation of inorganic elements in paeonol saffuruticosa from different origins by ICP-MS/ICP-OES combined with chemometrics technology. China Pharmacy 2016, 27, 1249–1253. [Google Scholar]
- Liu, W.; Wang, Z.; Hu, J.; Li, J.; Zhang, Z.; Xiao, W. Morphological analysis and dissolution characteristics analysis of 24 trace elements in Paeonia suffruticosa. Chin. J. Exp. Tradit. Med. Formulae 2016, 22, 60–65. [Google Scholar]
- Zhuang, H.; Ni, Y.; Kokot, S. Combining HPLC-DAD and ICP-MS data for improved analysis of complex samples: Classification of the root samples from cortex moutan. Chemom. Intell. Lab. Syst. 2014, 135, 183–191. [Google Scholar] [CrossRef]
- Guo, M.; Chen, W.; Xu, Y. The implication of trace elements on the quality of cortex moutan. China J. Chin. Mater. Med. 2008, 33, 1083–1085. [Google Scholar]
- Hon, K.L.; Chan, B.C.-L.; Leung, P.C. Chinese herbal medicine research in eczema treatment. Chin. Med. 2011, 6, 17. [Google Scholar] [CrossRef] [PubMed]
- Sheng, Z.H.; Yu, C.H.; Wu, Q.F. Determination of peoniflorin and benzoic acid in duration of cultivation of plant in radix paeoniae rubra. Chin. Arch. Tradit. Chin. Med. 2008, 5, 103. [Google Scholar]
- Baye, H.; Hymete, A. Lead and cadmium accumulation in medicinal plants collected from environmentally different sites. Bull. Environ. Contam. Toxicol. 2010, 84, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Qian, G.; Rimao, H.; Xingqin, W.; Xin, M. Simultaneous determination of organochlorines and pyrethroid pesticide residues in cotex moutan based on matrix solid phase dispersion and gas chromatography. Chin. Agric. Sci. Bull. 2012, 21, 242–247. [Google Scholar]
- Liu, Y.; Xia, Y.; Guo, P.; Wang, G.; Shen, Z.; Xu, Y.; Chen, Y. Copper and bacterial diversity in soil enhance paeonol accumulation in cortex moutan of Paeonia suffruticosa ‘fengdan’. Hortic. Environ. Biotechnol. 2013, 54, 331–337. [Google Scholar] [CrossRef]
- Tokalıoğlu, Ş. Determination of trace elements in commonly consumed medicinal herbs by ICP-MS and multivariate analysis. Food Chem. 2012, 134, 2504–2508. [Google Scholar] [CrossRef] [PubMed]
- Deng, B. Determination of trace metals Fe, Zn, Cu and mn in cortex moutan by atomic absorption spectrometry. Chin. J. Spectrosc. Lab. 2008, 25, 630–632. [Google Scholar]
- Guan, D.P.; Wan, Q.; Wu, L. Cortex moutan harmful heavy metal element detection. Chin. J. Ethnomed. Ethnopharm. 2015, 24, 20–21. [Google Scholar]
- Liu, X.H. Analysis of heavy metal elements lead and cadmium in cortex moutan and its planting soil. Chin. J. Spectrosc. Lab. 2013, 30, 821–824. [Google Scholar]
- Liang, X.; Li, H.; Li, S. A novel network pharmacology approach to analyse traditional herbal formulae: The Liu-Wei-Di-Huang pill as a case study. Mol. BioSyst. 2014, 10, 1014–1022. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.Q.; A-Li, M.A.; Yang, Y.S.; Huang, C.H.; Zhou, N. Content determination of trihydroxybenzoic acid and paeonol in Paeonia delavayi from different producing area by HPLC. Chin. J. Exp. Tradit. Med. Formulae 2014, 20, 57–60. [Google Scholar]
- Zhang, Y.; Liu, X.; Zhang, Y. Determination of paeonol in cortex moutan by HPLC. Heilongjiang Med. J. 2010, 23, 151–153. [Google Scholar]
- Li, Y.G.; Song, L.; Liu, M.; Wang, Z.T. Advancement in analysis of salviae miltiorrhizae radix et rhizoma (danshen). J. Chromatogr. A 2009, 1216, 1941–1953. [Google Scholar] [CrossRef] [PubMed]

© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Z.; He, C.; Peng, Y.; Chen, F.; Xiao, P. Origins, Phytochemistry, Pharmacology, Analytical Methods and Safety of Cortex Moutan (Paeonia suffruticosa Andrew): A Systematic Review. Molecules 2017, 22, 946. https://doi.org/10.3390/molecules22060946
Wang Z, He C, Peng Y, Chen F, Xiao P. Origins, Phytochemistry, Pharmacology, Analytical Methods and Safety of Cortex Moutan (Paeonia suffruticosa Andrew): A Systematic Review. Molecules. 2017; 22(6):946. https://doi.org/10.3390/molecules22060946
Chicago/Turabian StyleWang, Zhiqiang, Chunnian He, Yong Peng, Feihu Chen, and Peigen Xiao. 2017. "Origins, Phytochemistry, Pharmacology, Analytical Methods and Safety of Cortex Moutan (Paeonia suffruticosa Andrew): A Systematic Review" Molecules 22, no. 6: 946. https://doi.org/10.3390/molecules22060946
APA StyleWang, Z., He, C., Peng, Y., Chen, F., & Xiao, P. (2017). Origins, Phytochemistry, Pharmacology, Analytical Methods and Safety of Cortex Moutan (Paeonia suffruticosa Andrew): A Systematic Review. Molecules, 22(6), 946. https://doi.org/10.3390/molecules22060946





