Structure Modification of an Active Azo-Compound as a Route to New Antimicrobial Compounds
Abstract
:1. Introduction
2. Results and Discussion
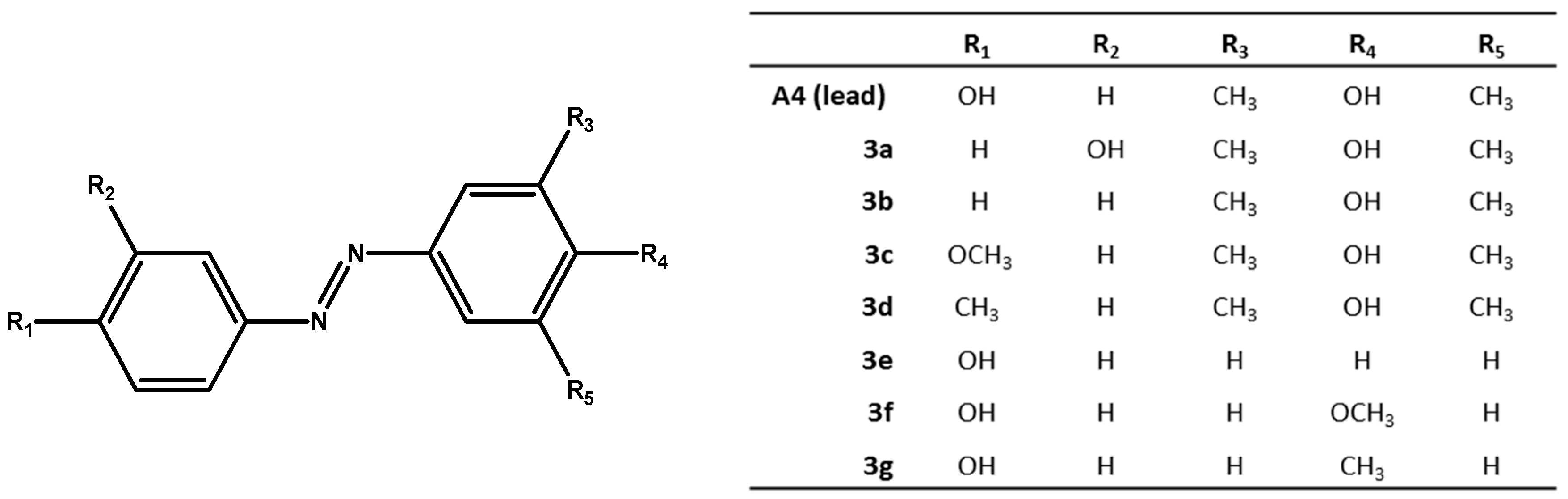
2.1. Chemistry
2.2. Thermal and Optical Properties
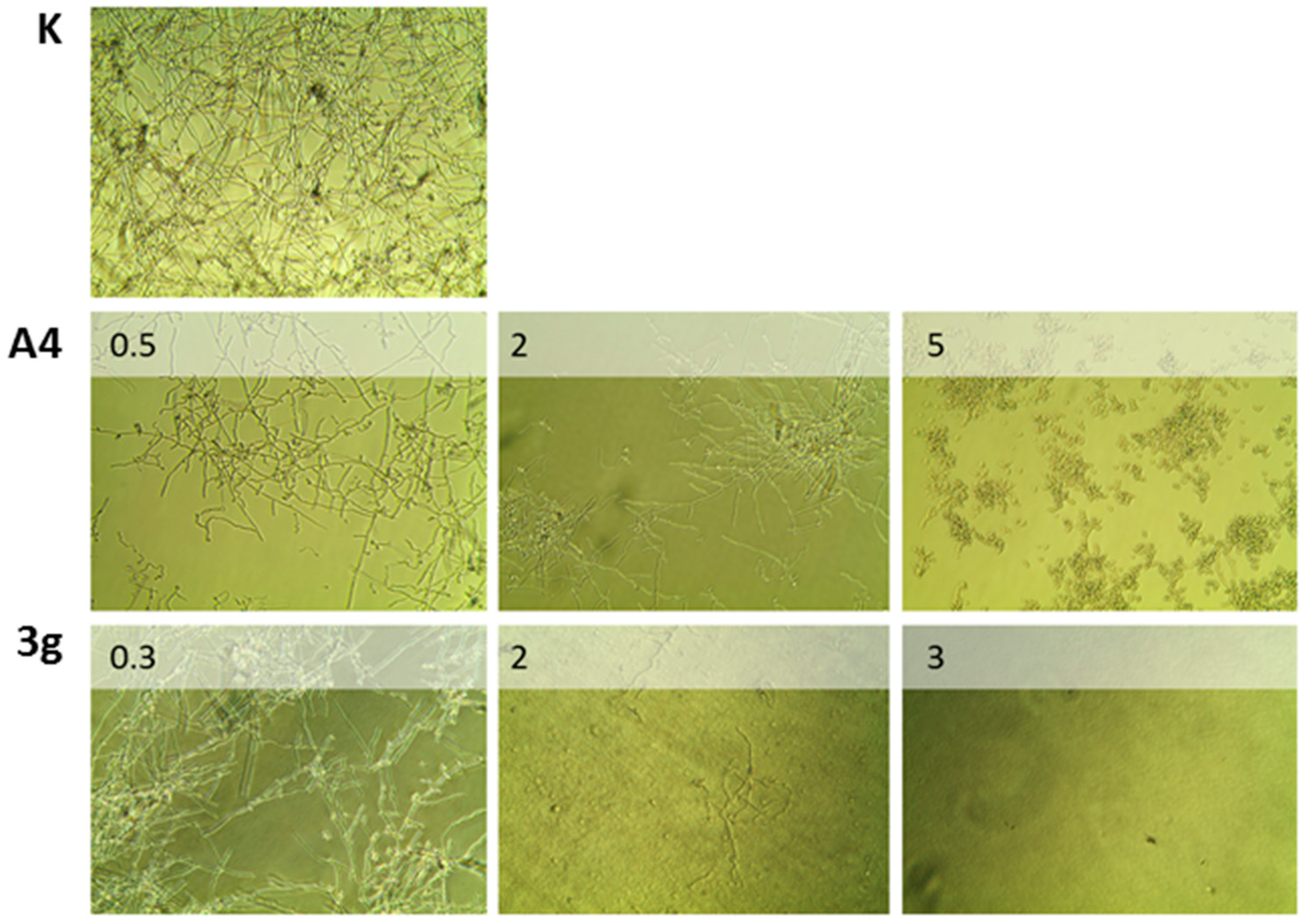
2.3. Antimicrobial Activity
2.4. QSAR Models
3. Materials and Methods
3.1. General Information
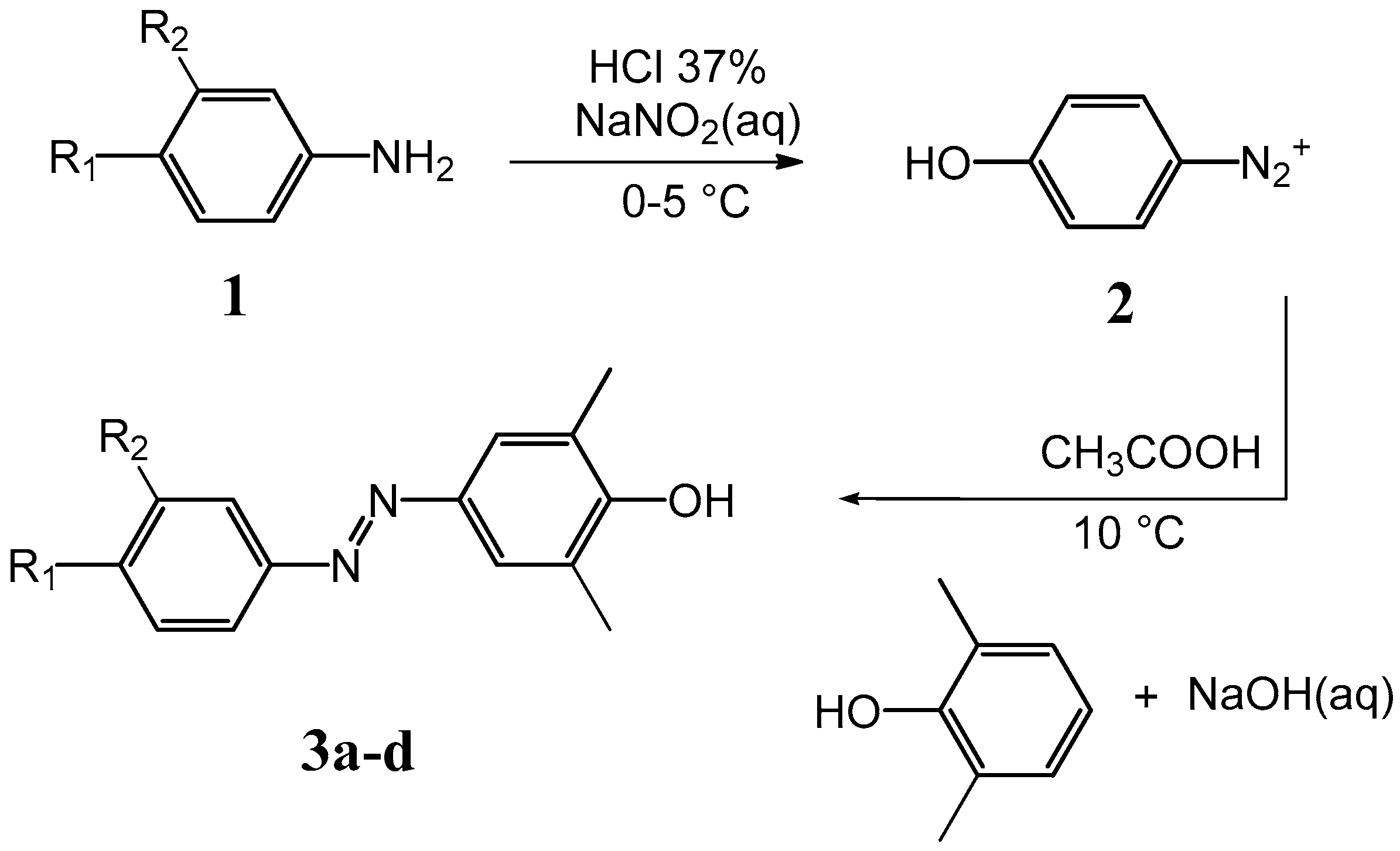
3.2. General Method of the Synthesis of (Phenyl-Diazenyl)Phenols Derivatives 3a–d
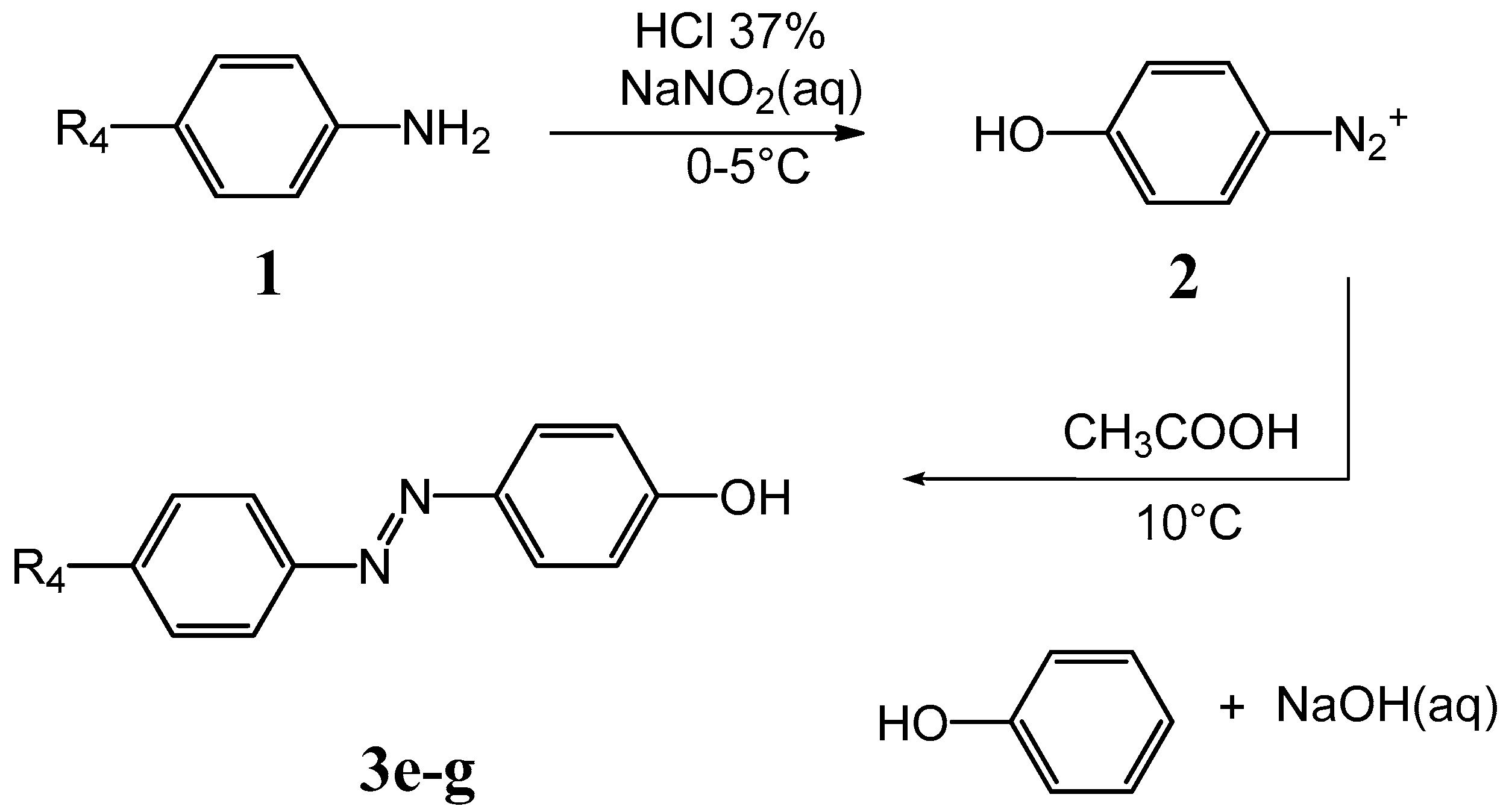
3.3. General Method of the Synthesis of (Phenyl-Diazenyl)Phenols Derivatives 3e–g
3.4. Antimicrobial Tests
3.4.1. Bacterial Strains and Minimum Inhibitory Concentrations
3.4.2. Candida Albicans Morphological Analysis
3.5. QSAR Analysis
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ball, A.P.; Bartlett, J.G.; Craig, W.A.; Drusano, G.L.; Felmingham, D.; Garau, J.A.; Klugman, K.P.; Low, D.E.; Mandell, L.A.; Rubinstein, E.; et al. Future trends in antimicrobial chemotherapy: Expert opinion on the 43rd icaac. J. Chemother. 2004, 16, 419–436. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Bayer, A.; Cosgrove, S.E.; Daum, R.S.; Fridkin, S.K.; Gorwitz, R.J.; Kaplan, S.L.; Karchmer, A.W.; Levine, D.P.; Murray, B.E. Clinical practice guidelines by the infectious diseases society of america for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children. Clin. Infect. Dis. 2011, 52, e18–e55. [Google Scholar] [CrossRef] [PubMed]
- Smith, A. Bacterial resistance to antibiotics. In Hugo and Russell’s Pharmaceutical Microbiology; Blackwell, Science: 350 Main Street, Malden, MA, USA, 2004; pp. 220–223. [Google Scholar]
- Wright, G.D. Bacterial resistance to antibiotics: Enzymatic degradation and modification. Adv. Drug Deliv. Rev. 2005, 57, 1451–1470. [Google Scholar] [CrossRef] [PubMed]
- Stewart, P.S.; Costerton, J.W. Antibiotic resistance of bacteria in biofilms. Lancet 2001, 358, 135–138. [Google Scholar] [CrossRef]
- Tenover, F.C. Development and spread of bacterial resistance to antimicrobial agents: An overview. Clin. Infect. Dis. 2001, 33, S108–S115. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.W. Biofilms and antibiotic therapy: Is there a role for combating bacterial resistance by the use of novel drug delivery systems? Adv. Drug Deliv. Rev. 2005, 57, 1539–1550. [Google Scholar] [CrossRef] [PubMed]
- Jenssen, H.; Hamill, P.; Hancock, R.E. Peptide antimicrobial agents. Clin. Microbiol. Rev. 2006, 19, 491–511. [Google Scholar] [CrossRef] [PubMed]
- Piotto, S.; Concilio, S.; Sessa, L.; Porta, A.; Calabrese, E.C.; Zanfardino, A.; Varcamonti, M.; Iannelli, P. Small azobenzene derivatives active against bacteria and fungi. Eur. J. Med. Chem. 2013, 68, 178–184. [Google Scholar] [CrossRef] [PubMed]
- Concilio, S.; Iannelli, P.; Sessa, L.; Olivieri, R.; Porta, A.; De Santis, F.; Pantani, R.; Piotto, S. Biodegradable antimicrobial films based on poly (lactic acid) matrices and active azo compounds. J. Appl. Polym. Sci. 2015, 132. [Google Scholar] [CrossRef]
- Piotto, S.; Concilio, S.; Sessa, L.; Iannelli, P.; Porta, A.; Calabrese, E.C.; Galdi, M.R.; Incarnato, L. Novel antimicrobial polymer films active against bacteria and fungi. Polym. Compos. 2013, 34, 1489–1492. [Google Scholar] [CrossRef]
- Sessa, L.; Concilio, S.; Iannelli, P.; De Santis, F.; Porta, A.; Piotto, S. Antimicrobial azobenzene compounds and their potential use in biomaterials. AIP Conf. Proc. 2016, 1727, 020018. [Google Scholar]
- Cariati, F.; Caruso, U.; Centore, R.; De Maria, A.; Fusco, M.; Panunzi, B.; Roviello, A.; Tuzi, A. Optical second order nonlinearities in new chromophores obtained by selective mono-reduction of dinitro precursors. Opt. Mater. 2004, 27, 91–97. [Google Scholar] [CrossRef]
- Piotto, S.; Trapani, A.; Bianchino, E.; Ibarguren, M.; López, D.J.; Busquets, X.; Concilio, S. The effect of hydroxylated fatty acid-containing phospholipids in the remodeling of lipid membranes. Biochim. Biophys. Acta Biomembr. 2014, 1838, 1509–1517. [Google Scholar] [CrossRef] [PubMed]
- Scrima, M.; Di Marino, S.; Grimaldi, M.; Campana, F.; Vitiello, G.; Piotto, S.P.; D'Errico, G.; D'Ursi, A.M. Structural features of the c8 antiviral peptide in a membrane-mimicking environment. Biochim. Biophys. Acta Biomembr. 2014, 1838, 1010–1018. [Google Scholar] [CrossRef] [PubMed]
- Concilio, S.; Bugatti, V.; Iannelli, P.; Piotto, S.P. Synthesis and characterization of new photoluminescent oxadiazole/carbazole-containing polymers. Int. J. Polym. Sci. 2010, 2010. [Google Scholar] [CrossRef]
- Concilio, S.; Bugatti, V.; Neitzert, H.C.; Landi, G.; De Sio, A.; Parisi, J.; Piotto, S.; Iannelli, P. Zn-complex based on oxadiazole/carbazole structure: Synthesis, optical and electric properties. Thin Solid Films 2014, 556, 419–424. [Google Scholar] [CrossRef]
- Acierno, D.; Amendola, E.; Bugatti, V.; Concilio, S.; Giorgini, L.; Iannelli, P.; Piotto, S.P. Synthesis and characterization of segmented liquid crystalline polymers with the azo group in the main chain. Macromolecules 2004, 37, 6418–6423. [Google Scholar] [CrossRef]
- Ghose, A.K.; Viswanadhan, V.N.; Wendoloski, J.J. Prediction of hydrophobic (lipophilic) properties of small organic molecules using fragmental methods: An analysis of alogp and clogp methods. J. Phys. Chem. A 1998, 102, 3762–3772. [Google Scholar] [CrossRef]
- CLSI, Clinical and Laboratory Standards Institute. Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeasts; Approved Standard—Third Edition; CLSI: Wayne, PA, USA, 2008; document m27–a3. [Google Scholar]
- Patton, T.; Barrett, J.; Brennan, J.; Moran, N. Use of a spectrophotometric bioassay for determination of microbial sensitivity to manuka honey. J. Microbiol. Methods 2006, 64, 84–95. [Google Scholar] [CrossRef] [PubMed]
- Yap, C.W. Padel-descriptor: An open source software to calculate molecular descriptors and fingerprints. J. Comput. Chem. 2011, 32, 1466–1474. [Google Scholar] [CrossRef] [PubMed]
- Accelrys. Accelrys Materials Studio; Accelrys Inc.: San Diego, CA, USA, 2014. [Google Scholar]
- Todeschini, R.; Consonni, V. Molecular Descriptors for Chemoinformatics, Volume 41 (2 Volume Set); John Wiley & Sons: Hoboken, NJ, USA, 2009. [Google Scholar]
Sample Availability: Samples of the compounds are not available from the authors. |
| Molecule | Thermal Characterization | Optical Characterization | ||||
|---|---|---|---|---|---|---|
| Tm (°C) | ∆Hm (J/g) | Tc (°C) | ∆Hc (J/g) | λmax (nm) | εmax (L mol−1 cm−1) | |
| 3a | 114.4 | 5.7 | - | - | 358 | 9300 |
| 3b | 87.7 | 78.0 | - | - | 344 | 18,300 |
| 3c | 128.3 | 98.1 | 94.6 | 92.1 | 360 | 14,000 |
| 3d | 89.3 | 86.1 | - | - | 354 | 11,400 |
| 3e | 162.0 | 149.8 | - | - | 344 | 14,470 |
| 3f | 147.3 | 123.3 | 110.6 | 104.5 | 355 | 14,200 |
| 3g | 158.7 | 139.4 | 90.7 | 83.7 | 358 | 14,350 |
| Molecule | S. aureus | P. aeruginosa | C. albicans |
|---|---|---|---|
| MIC50 | MIC2 | ||
| A4 (lead) | 15 (MIC100 20 *) | >35 | 15 (MIC0 17 *) |
| 3a | 30 | >35 | 25 |
| 3b | 7 (MIC100 10) | >35 | 10 |
| 3c | >35 | >35 | >35 |
| 3d | >35 | >35 | >35 |
| 3e | 25 | >35 | 20 |
| 3f | 25 | >35 | 25 |
| 3g | 7 (MIC100 20) | >35 | MIC0 3 |
| Molecule | MIC50 Experimental (μg/mL) | Actual Values for C:1/MIC50 | ALogP | Equation (1): Predicted Values | Equation (1): Residual Values | MIC50 Predicted |
|---|---|---|---|---|---|---|
| A4 | 15 | 0.067 | 2.8 | 0.0513 | 0.0154 | 19 |
| 3a | 30 | 0.033 | 2.8 | 0.0520 | −0.0187 | 19 |
| 3b | 7 | 0.143 | 5.0 | 0.1460 | −0.0030 | 7 |
| 3c | 80 * | 0.013 | 3.0 | 0.0214 | −0.0089 | 50 |
| 3d | 80 * | 0.013 | 7.2 | 0.0357 | −0.0232 | 30 |
| 3e | 25 | 0.040 | 1.8 | 0.0452 | −0.0052 | 22 |
| 3f | 25 | 0.040 | 0.7 | 0.0909 | −0.0509 | 11 |
| 3g | 7 | 0.143 | 3.2 | 0.0950 | 0.0479 | 10 |
| A1 | 17 | 0.059 | 0.8 | 0.0756 | −0.0168 | 13 |
| A2 | 15 | 0.067 | 2.6 | 0.0822 | 0.0155 | 12 |
| A3 | 14 | 0.071 | 0.2 | 0.0699 | 0.0015 | 14 |
| A5 | 18 | 0.056 | 4.3 | 0.0625 | −0.0069 | 16 |
| B10 | 80 * | 0.013 | 0.3 | 0.0142 | −0.0017 | 70 |
| B11 | 80 * | 0.013 | 1.1 | 0.0142 | −0.0017 | 70 |
| Parameters | Value |
|---|---|
| Friedman LOF | 0.00302 |
| R-squared | 0.802 |
| Adjusted R-squared | 0.766 |
| Cross validated R-squared | 0.654 |
| Significant Regression | Yes |
| Significance-of-regression F-value | 22.321 |
| Critical SOR F-value (95%) | 5.2341 |
| Replicate points | 0 |
| Computed experimental error | 0.000 |
| Number or points for Lack-of-fit test | 11 |
| Min expt. error for non-significant LOF (95%) | 0.0201 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Concilio, S.; Sessa, L.; Petrone, A.M.; Porta, A.; Diana, R.; Iannelli, P.; Piotto, S. Structure Modification of an Active Azo-Compound as a Route to New Antimicrobial Compounds. Molecules 2017, 22, 875. https://doi.org/10.3390/molecules22060875
Concilio S, Sessa L, Petrone AM, Porta A, Diana R, Iannelli P, Piotto S. Structure Modification of an Active Azo-Compound as a Route to New Antimicrobial Compounds. Molecules. 2017; 22(6):875. https://doi.org/10.3390/molecules22060875
Chicago/Turabian StyleConcilio, Simona, Lucia Sessa, Anna Maria Petrone, Amalia Porta, Rosita Diana, Pio Iannelli, and Stefano Piotto. 2017. "Structure Modification of an Active Azo-Compound as a Route to New Antimicrobial Compounds" Molecules 22, no. 6: 875. https://doi.org/10.3390/molecules22060875
APA StyleConcilio, S., Sessa, L., Petrone, A. M., Porta, A., Diana, R., Iannelli, P., & Piotto, S. (2017). Structure Modification of an Active Azo-Compound as a Route to New Antimicrobial Compounds. Molecules, 22(6), 875. https://doi.org/10.3390/molecules22060875











