Systemic Induction of the Defensin and Phytoalexin Pisatin Pathways in Pea (Pisum sativum) against Aphanomyces euteiches by Acetylated and Nonacetylated Oligogalacturonides
Abstract
:1. Introduction
2. Results
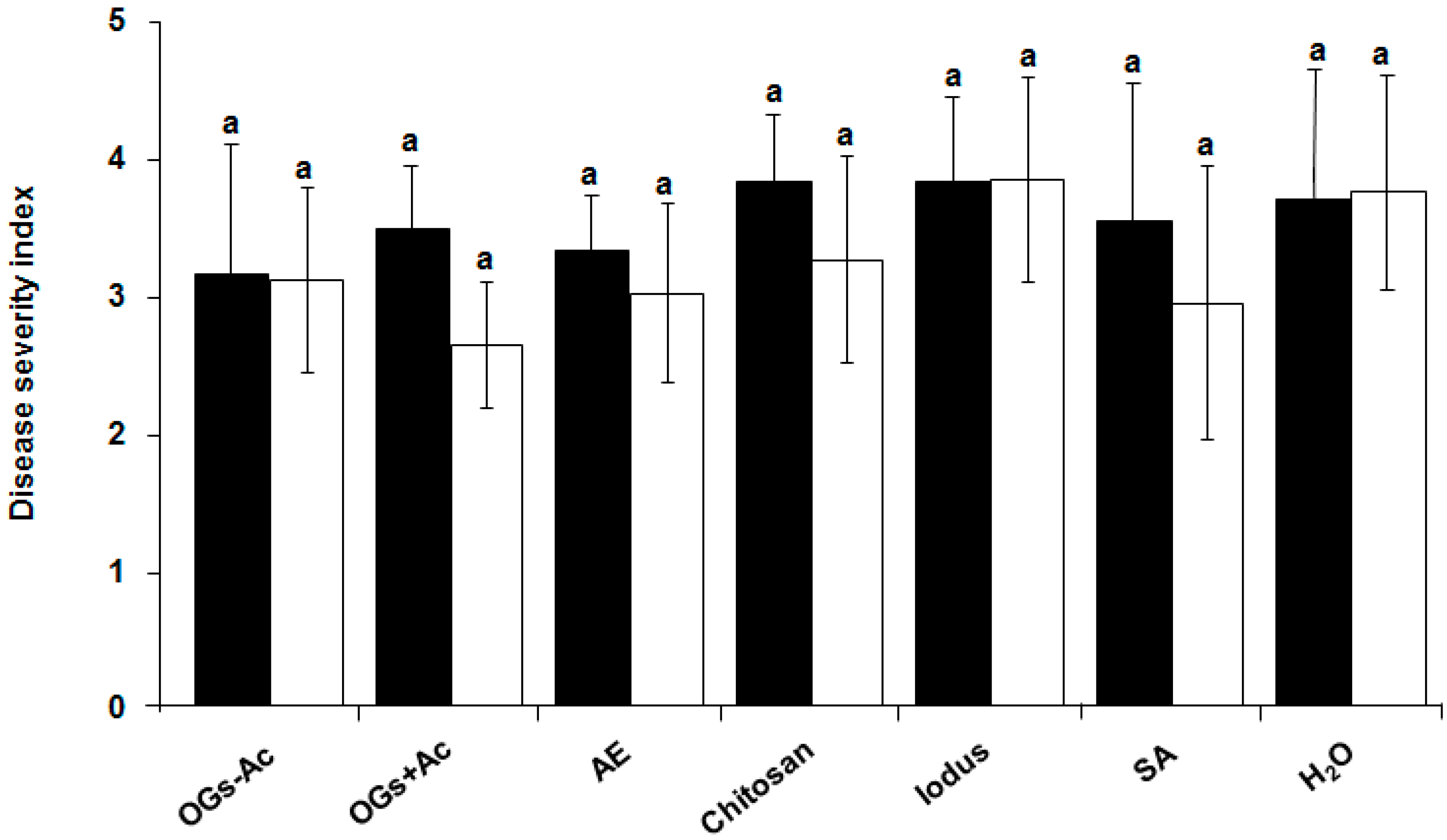
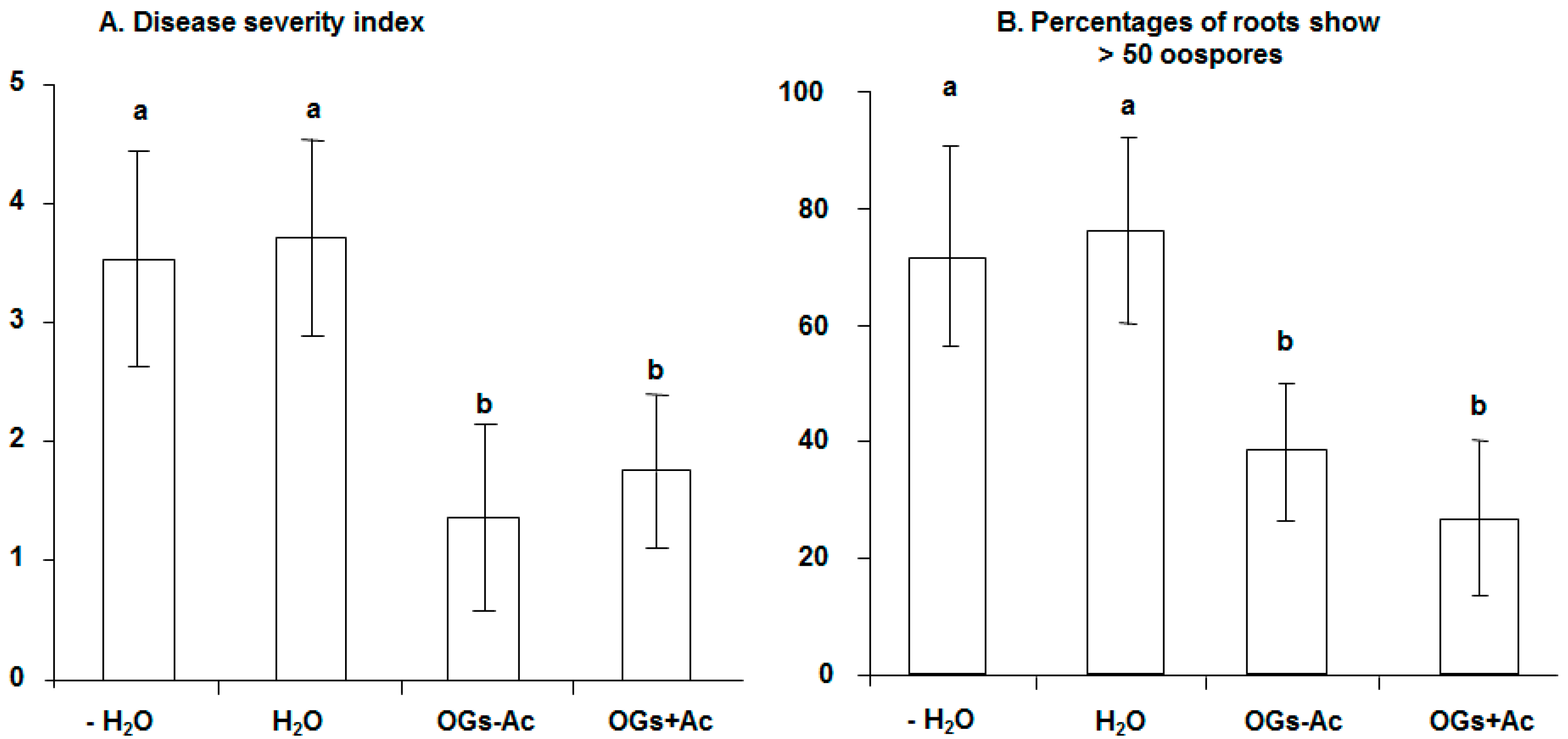
2.1. Elicitor Effect of OG Compounds
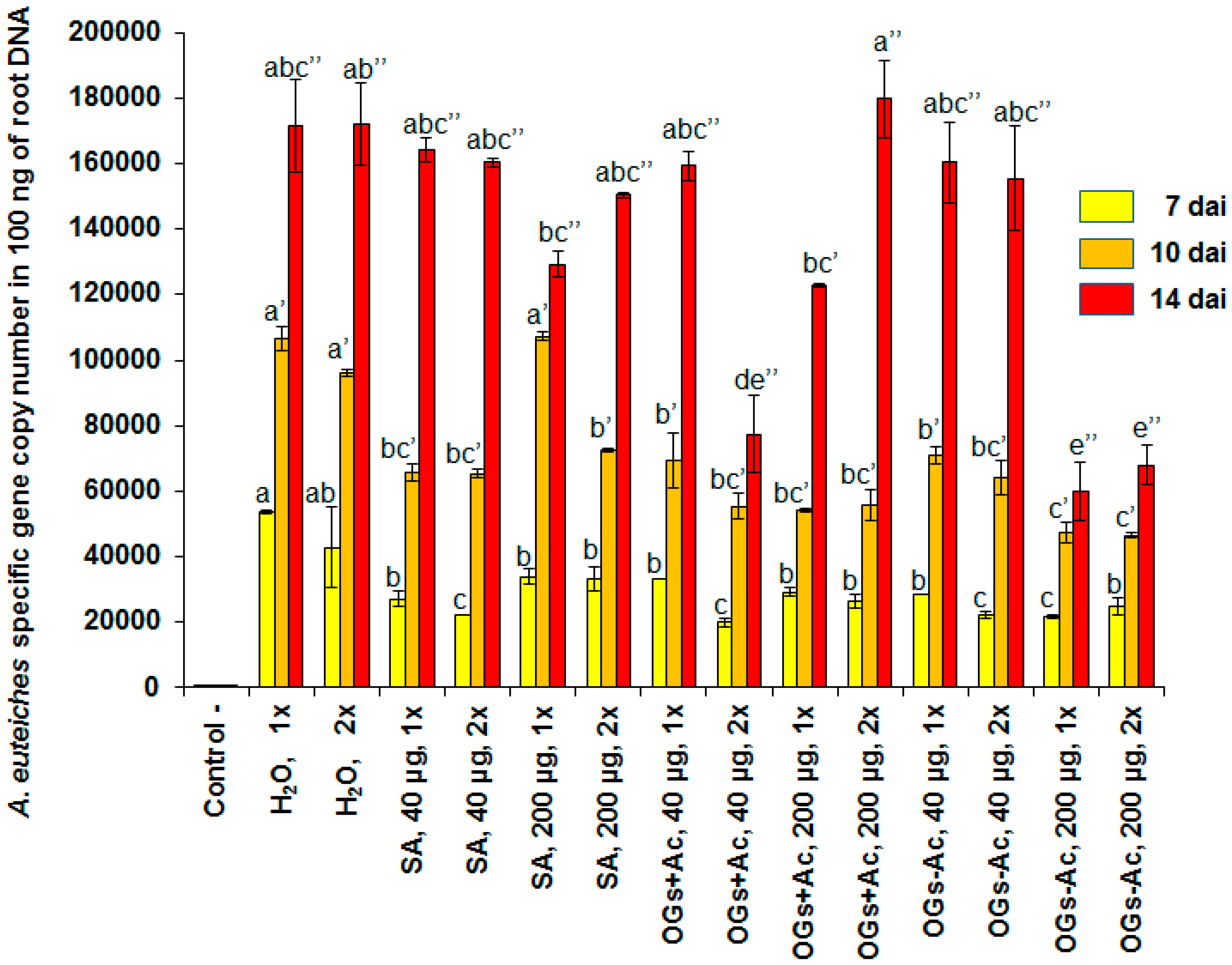
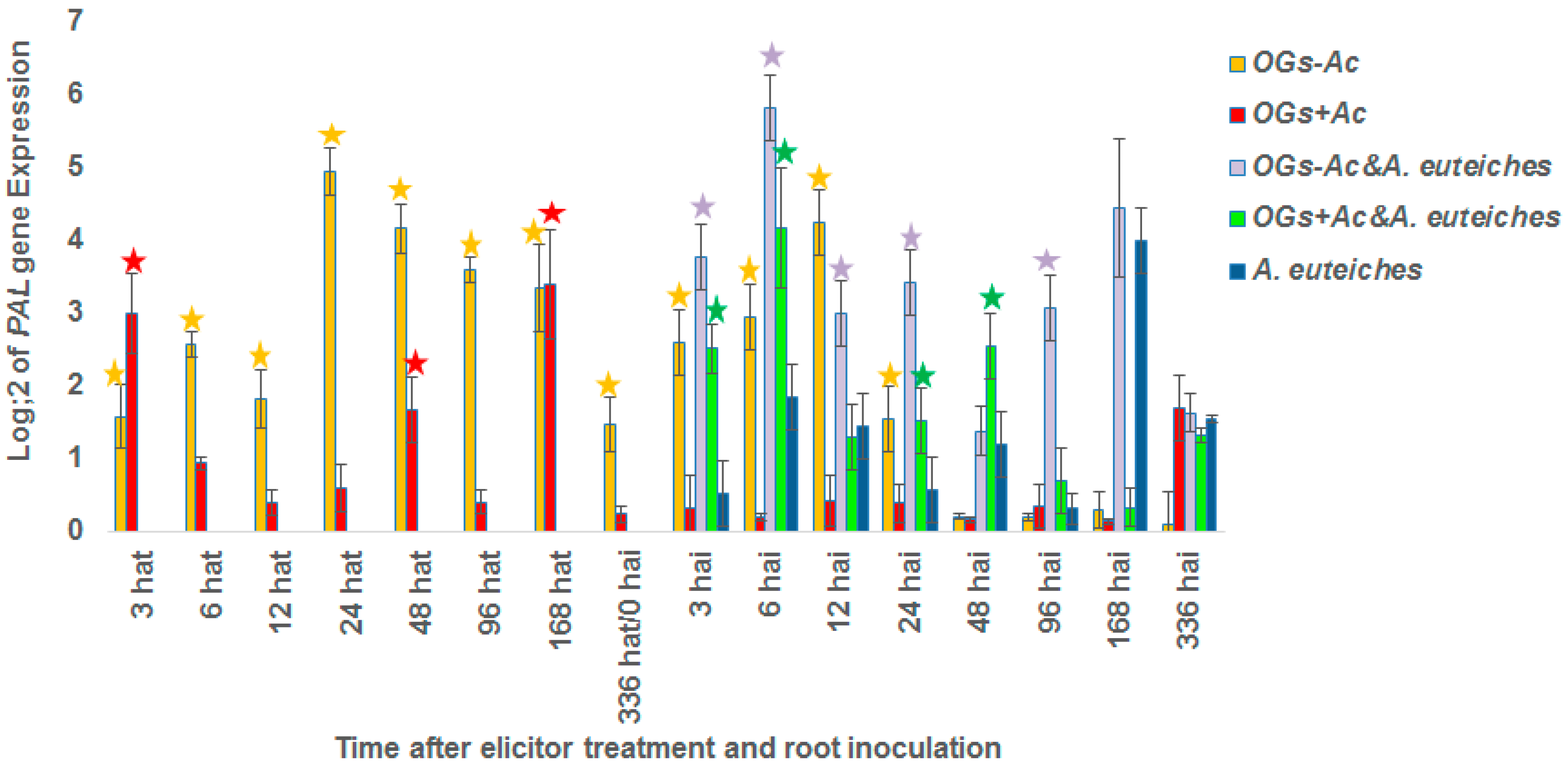
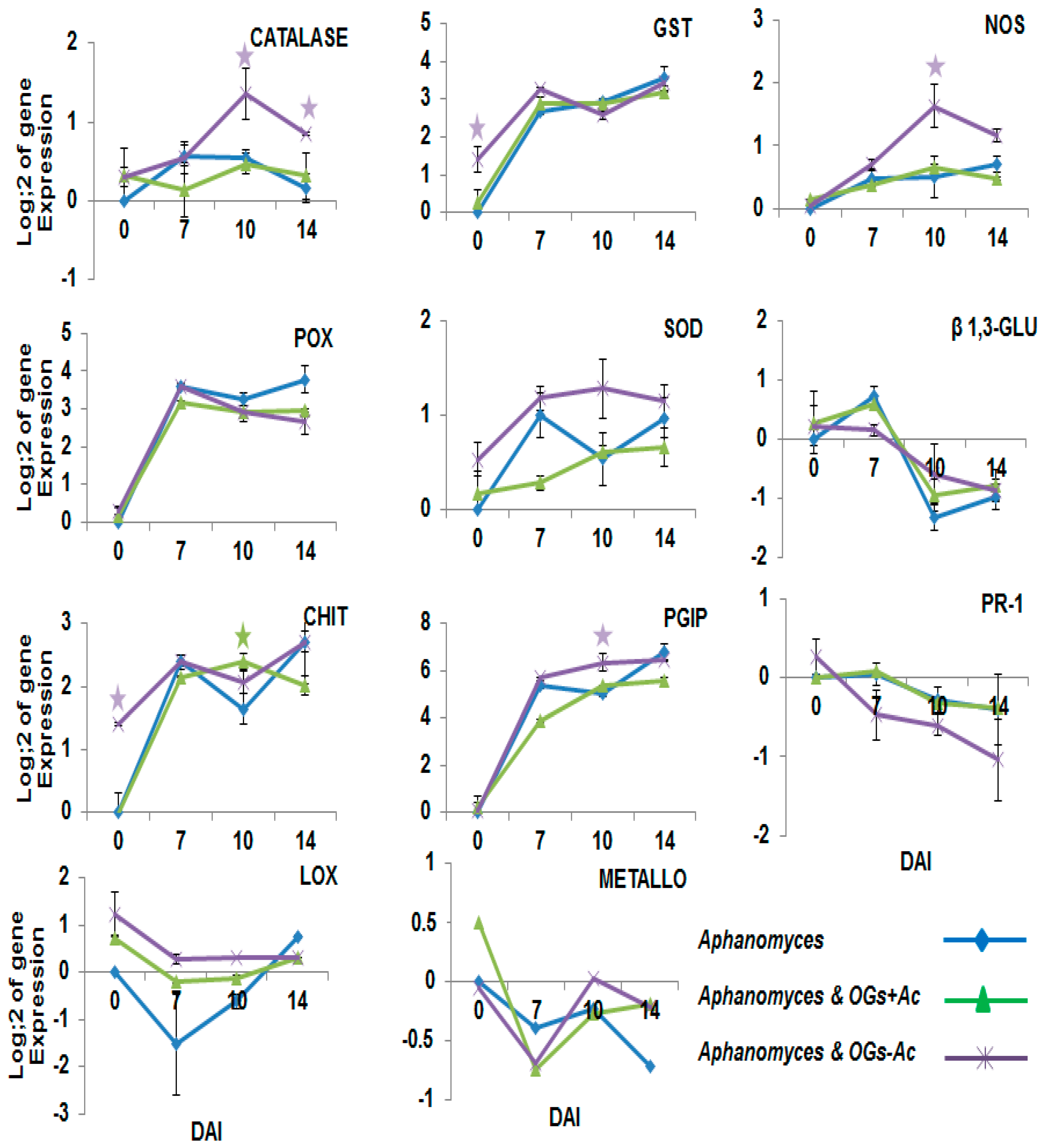
2.2. Gene Expression
3. Discussion
4. Materials and Methods
4.1. Elicitor Compounds
4.2. Plant Material and Growth Conditions
4.3. Inoculum Preparation and Inoculation
4.4. Protection Assay
4.5. DNA and RNA Extraction
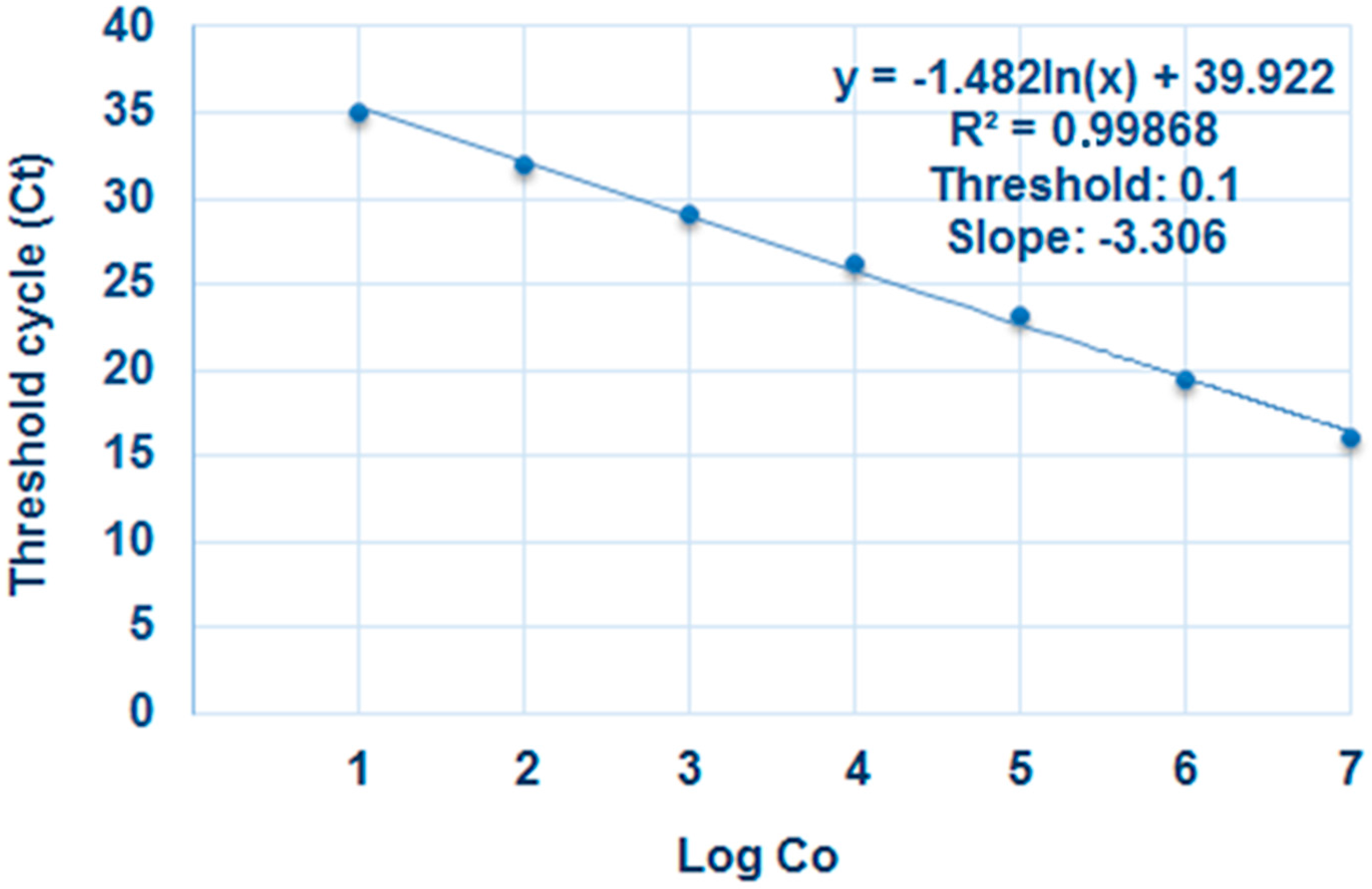
4.6. Real-Time PCR
4.6.1. Real-Time Quantitative PCR (qPCR)
4.6.2. Real-Time Reverse Transcription PCR (RT-PCR)
4.7. Statistical Analyses
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cervone, F.; Hahn, M.G.; DeLorenzo, G.; Darvill, A.; Albersheim, P. Host–pathogen interactions: XXXIII. A plant protein converts a fungal pathogenesis factor into an elicitor of plant defense responses. Plant Physiol. 1989, 90, 542–548. [Google Scholar] [CrossRef] [PubMed]
- Albersheim, P.; Darvill, A.; Augur, C.; Cheong, J.J.; Eberhard, S.; Hahn, M.G.; Marfa, V.; Mohnen, D.; O’Neill, M.A.; Spiro, M.D.; et al. Oligosaccharins: Oligosaccharide regulatory molecules. Acc. Chem. Res. 1992, 25, 77–83. [Google Scholar] [CrossRef]
- Côté, F.; Ham, K.S.; Hahn, M.; Bergmann, C.W.; Côté, F.; Ham, K.S.; Hahn, M.G.; Bergmann, C.W. Oligosaccharide elicitors in host-pathogen interactions. Generation, perception and signal transduction. Subcell. Biochem. 1998, 29, 385–432. [Google Scholar] [PubMed]
- Côté, F.; Hahn, M.G. Oligosaccharins: Structures and signal transduction. Plant Mol. Biol. 1994, 26, 1379–1411. [Google Scholar] [CrossRef] [PubMed]
- Braccini, I.; Perez, S. Molecular basis of Ca(2+)-induced gelation in alginates and pectins: The egg-box model revisited. Biomacromolecules 2001, 2, 1089–1096. [Google Scholar] [CrossRef] [PubMed]
- Cabrera, J.C.; Boland, A.; Messiaen, J.; Cambier, P.; Van Cutsem, P. Egg box conformation of oligogalacturonides: The time-dependent stabilization of the elicitor-active conformation increases its biological activity. Glycobiology 2008, 18, 473–482. [Google Scholar] [CrossRef] [PubMed]
- Farmer, E.E.; Ryan, C.A. Inter plant communication: Airborne methyl jasmonate induces expression of protease inhibitor genes in plant leaves. Proc. Natl. Acad. Sci. USA 1990, 87, 7713–7716. [Google Scholar] [CrossRef] [PubMed]
- Moloshok, T.; Pearce, G.; Ryan, C.A. Oligouronide signaling of proteinase inhibitor genes in plants: Structure-activity relationships of di- and trigalacturonic acids and their derivatives. Arch. Biochem. Biophys. 1992, 294, 731–734. [Google Scholar] [CrossRef]
- Moerschbacher, B.M.; Mierau, M.; Graeßner, B.; Noll, U.; Mor, A.J. Small oligomers of galacturonic acid are endogenous suppressors of disease resistance reactions in wheat leaves. J. Exp. Bot. 1999, 50, 605–612. [Google Scholar] [CrossRef]
- Wydra, K.; Beri, H. Structural changes of homogalacturonan, rhamnogalacturonan I and arabinogalactan protein in xylem cell walls of tomato genotypes in reaction to Ralstonia solanacearum. Physiol. Mol. Plant Pathol. 2006, 68, 41–50. [Google Scholar] [CrossRef]
- Wydra, K.; Beri, H. Immunohistochemical changes in methyl-ester distribution of homogalacturonan and side chain composition of rhamnogalacturonan I as possible components of basal resistance in tomato inoculated with Ralstonia solanacearum. Physiol. Mol. Plant Pathol. 2007, 70, 13–24. [Google Scholar] [CrossRef]
- Diogo, R.; Wydra, K. Silicon-induced basal resistance in tomato against Ralstonia solanacearum is related to modification of pectic cell wall polysaccharide structure. Physiol. Mol. Plant Pathol. 2007, 70, 120–129. [Google Scholar] [CrossRef]
- Osorio, S.; Castillejo, C.; Quesada, M.A.; Medina-Escobar, N.; Brownsey, G.J.; Suau, R.; Heredia, A.; Botella, M.A.; Valpuesta, V. Partial demethylation of oligogalacturonides by pectin methyl esterase 1 is required for eliciting defence responses in wild strawberry (Fragaria vesca). Plant J. 2008, 54, 43–55. [Google Scholar] [CrossRef] [PubMed]
- Wiethölter, N.; Graessener, B.; Mierau, M.; Mort, A.J.; Moerschbacher, B.M. Differences in the methyl ester distribution of homogalacturonans from near-isogenic wheat lines resistant and susceptible to the wheat stem rust fungus. Mol. Plant. Microbe Interact. 2003, 16, 945–952. [Google Scholar] [CrossRef] [PubMed]
- Randoux, B.; Renard-Merlier, D.; Mulard, G.; Rossard, S.; Duyme, F.; Sanssené, J.; Courtois, J.; Durand, R.; Reignault, P. Distinct defenses induced in wheat against powdery mildew by acetylated and nonacetylated oligogalacturonides. Phytopathology 2010, 100, 1352–1363. [Google Scholar] [CrossRef] [PubMed]
- Boller, T.; Felix, G. A renaissance of elicitors: Perception of microbe-associated molecular patterns and danger signals by pattern-recognition receptors. Annu. Rev. Plant Biol. 2009, 60, 379–406. [Google Scholar] [CrossRef] [PubMed]
- De Lorenzo, G.; Brutus, A.; Savatin, D.; Sicilia, F.; Gervone, F. Engineering plant resistance by constructing chimeric receptors that recognize damage-associated molecular patterns (DAMPs). FEBS Lett. 2011, 585, 1521–1528. [Google Scholar] [CrossRef] [PubMed]
- Ranf, S.; Eschen-Lippold, L.; Pecher, P.; Lee, J.; Scheel, D. Interplay between calcium signalling and early signalling elements during defence responses to microbe- or damage-associated molecular patterns. Plant J. 2011, 68, 100–113. [Google Scholar] [CrossRef] [PubMed]
- Wydra, K.; Beri, H.; Schacht, H. Polygalacturonase-inhibiting protein (PGIP) and structure and composition of cell wall polysaccharides of tomato in relation to resistance to Ralstonia solanacearum. In Emerging Trends in Plant-Microbe Interactions; Gananamanickam, S., Balasubramanian, R., Anand, N., Eds.; Centre for Advanced Studies in Botany: Chennai, India, 2005; pp. 217–223. [Google Scholar]
- Schacht, T.; Unger, C.; Pich, A.; Wydra, K. Endo- and exopolygalacturonases of Ralstonia solanacearum are inhibited by polygalacturonase-inhibiting protein (PGIP) activity in tomato stem extracts. Plant Physiol. Biochem. 2011, 49, 377–387. [Google Scholar] [CrossRef] [PubMed]
- Davis, K.R.; Darvill, A.G.; Alber- sheim, P.; Dell, A. Host–pathogen interactions: XXIX. Oligogalacturonides released from sodium polypectate by endopolygalacturonic acid lyase are elicitors of phytoalexins in soybean. Plant Physiol. 1986, 80, 568–577. [Google Scholar] [CrossRef] [PubMed]
- Davis, K.R.; Hahlbrock, K. Induction of defense responses in cultured parsley cells by plant cell wall fragments. Plant Physiol. 1987, 1286–1290. [Google Scholar] [CrossRef]
- Broekaert, W.F.; Pneumas, W.J. Pectic polysaccharides elicit chitinase accumulation in tobacco. Plant Physiol. 1988, 74, 740–744. [Google Scholar] [CrossRef]
- Rasul, S.; Dubreuil-Maurizi, C.; Lamotte, O.; Koen, E.; Poinssot, B.; Alcaraz, G.; Wendehenne, D.; Jeandroz, S. Nitric oxide production mediates oligogalacturonide-triggered immunity and resistance to Botrytis cinerea in Arabidopsis thaliana. Plant Cell Environ. 2012, 35, 1483–1499. [Google Scholar] [CrossRef] [PubMed]
- Cannesan, M.A.; Gangneux, C.; Lanoue, A.; Giron, D.; Laval, K.; Hawes, M.; Driouich, A.; Vicre-Gibouin, M. Association between border cell responses and localized root infection by pathogenic Aphanomyces euteiches. Ann. Bot. 2011, 108, 459–469. [Google Scholar] [CrossRef] [PubMed]
- Shang, H.; Grau, C.R.; Peters, R.D. Oospore germination of Aphanomyces euteiches in root exudates and on the rhizoplanes of crop plants. Plant Dis. 2000, 84, 994–998. [Google Scholar] [CrossRef]
- Baldauf, S.L.; Roger, A.J.; Wenk-Siefert, J.; Doolittle, W.F. A kingdom level phylogeny of eukaryotes based on combined protein data. Science 2000, 290, 972–977. [Google Scholar] [CrossRef] [PubMed]
- Limberg, G.; Korner, R.; Buchholt, H.C.; Christensen, T.M.; Roepstorff, P.; Mikkelsen, J.D. Analysis of different de-esterification mechanisms for pectin by enzymatic fingerprinting using endopectin lyase and endopolygalacturonase II from A. niger. Carbohydr. Res. 2000, 327, 293–307. [Google Scholar] [CrossRef]
- Ferrari, S. Oligogalacturonides: Plant damage-associated molecular patterns and regulators of growth and development. Front. Plant Sci. 2013, 4, 49. [Google Scholar] [CrossRef] [PubMed]
- Hadwiger, L.A. Pea-Fusarium solani interactions contributions of a system toward understanding disease resistance. Phytopathology 2008, 98, 372–379. [Google Scholar] [CrossRef] [PubMed]
- Rennenberg, H. Glutathione metabolism and possible biological roles in the higher plant. Phytochemistry 1982, 21, 2771–2781. [Google Scholar] [CrossRef]
- Smith, I.K.; Polle, A.; Rennemerg, H. Glutathione. In Stress Responses in Plants: Adaptation and Acclimation Mechanisms; Alscher, R.G., Cumming, J.R., Eds.; Wiley & Liss: New York, NY, USA, 1990; pp. 201–215. [Google Scholar]
- Liu, N.; Zhang, X.; Sun, Y.; Wang, P.; Li, X.; Pei, Y.; Li, F.; Hou, Y. Molecular evidence for the involvement of a polygalacturonase-inhibiting protein, GhPGIP1, in enhanced resistance to Verticillium and Fusarium wilts in cotton. Sci. Rep. 2017, 7, 39840. [Google Scholar] [CrossRef] [PubMed]
- Ghareeb, H.; Bozsó, Z.; Ott, P.G.; Repenning, C.; Stahl, F.; Wydra, K. Transcriptome of silicon-induced resistance against Ralstonia solanacearum in the silicon non accumulator tomato implicates priming effect. Physiol. Mol. Plant Pathol. 2011, 75, 83–89. [Google Scholar] [CrossRef]
- Moiseyev, G.P.; Beintema, J.J.; Fedoreyeve, L.I.; Yakovlev, J.I. High sequence similarity between a ribonuclease from Ginseng calluses and fungus-elicited proteins from parsley indicates that intracellular pathogenesis-related proteins are ribonucleases. Planta 1994, 193, 470–472. [Google Scholar] [CrossRef] [PubMed]
- Riggleman, R.D.; Fristensky, B.; Hadwiger, L.A. The disease resistance response in pea is associated with increased levels of specific mRNAs. Plant Mol. Biol. 1985, 4, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Chang, M.M.; Chiang, C.C.; Martin, M.W.; Hadwiger, L.A. Expression of a pea disease resistance response gene in the potato cultivar shepody. Am. Potato J. 1993, 70, 635–647. [Google Scholar] [CrossRef]
- Breiteneder, H.; Pettenburger, K.; Bito, A.; Valenta, R.; Kraft, D.; Rumpold, H.; Scheiner, O.; Breitenbach, M. The gene coding for the major birch pollen allergen Betv 1 is highly homologous to a pea disease resistance response gene. EMBO J. 1989, 8, 1935–1938. [Google Scholar] [PubMed]
- Badreddine, I.; Lafitte, C.; Heux, L.; Skandalis, N.; Spanou, Z.; Martinez, Y.; Esquerre-Tugaye, M.-T.; Bulone, V.; Dumas, B.; Bottin, A. Cell wall chitosaccharides are essential components and exposed patterns of the phytopathogenic oomycete Aphanomyces euteiches. Eukaryot. Cell 2008, 7, 1980–1993. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Nowak, G.; Culley, D.; Hadwiger, L.A.; Fristensky, B. Constitutive expression of pea defense gene DRR206 confers resistance to blackleg (Leptosphaeria maculans) disease in transgenic canola (Brassica napus). Mol. Plant. Microbe Interact. 1999, 12, 410–418. [Google Scholar] [CrossRef]
- Seneviratne, H.K.; Dalisay, D.S.; Kim, K.-W.; Moinuddin, S.G.A.; Yang, H.; Hartshorn, C.M.; Davin, L.B.; Lewis, N.G. Non-host disease resistance response in pea (Pisum sativum) pods: Biochemical function of DRR206 and phytoalexin pathway localization. Phytochemistry 2015, 113, 140–148. [Google Scholar] [CrossRef] [PubMed]
- Chu, H.D.; Le, Q.N.; Nguyen, H.Q.; Le, D.T. Genome-wide analysis of genes encoding methionine-rich proteins in Arabidopsis and Soybean suggesting their roles in the adaptation of plants to abiotic stress. Int. J. Genomics 2016, 2016, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Vassão, D.G.; Kim, K.W.; Davin, L.B.; Lewis, N.G. Lignans (neolignans) and allyl/propenyl phenols: Biogenesis, structural biology, and biological/human health considerations. In Comprehensive Natural Products Chemistry II, Structural Diversity I; Townsend, C., Ebizuka, Y., Eds.; Elsevier: Oxford, UK, 2010; Volume 1, pp. 815–928. [Google Scholar]
- Chiang, C.C.; Hadwiger, L.A. The Fusarium solani-induced expression of a pea gene family encoding high cysteine content proteins. Mol. Plant. Microbe Interact. 1991, 4, 324–331. [Google Scholar] [CrossRef] [PubMed]
- Fristensky, B.; Riggleman, C.R.; Wagoner, W.; Hadwiger, L.A. Gene expression in susceptible and disease resistant interactions of peas induced with Fusarium solani pathogens and chitosan. Physiol. Plant Pathol. 1985, 27, 15–28. [Google Scholar] [CrossRef]
- Janisiewicz, W.J.; Pereira, I.B.; Almeida, M.S.; Roberts, D.P.; Wisniewski, M.E.; Kurtenbach, E. Biocontrol activity of recombinant Pichia pastoris constitutively expressing pea defensin against blue mold of apple. (Abstr.). Phytopathology 2006, 96, S53. [Google Scholar]
- Selitrennikoff, C.P. Antifungal Proteins. Appl. Environ. Microbiol. 2001, 63, 834–839. [Google Scholar] [CrossRef] [PubMed]
- Lay, F.T.; Anderson, M.A. Defensins–Components of the innate immune system in plants. Curr. Protein Pept. Sci. 2005, 5, 85–101. [Google Scholar] [CrossRef]
- Selim, S.; Negrel, J.; Wendehenne, D.; Ochatt, S.; Gianinazzi, S.; van Tuinen, D. Stimulation of defense reactions in Medicago truncatula by antagonistic lipopeptides from Paenibacillus sp. strain B2. Appl. Environ. Microbiol. 2010, 76, 7420–7428. [Google Scholar] [CrossRef] [PubMed]
- Lamb, C.; Dixon, R.A. The oxidative burst in plant disease resistance. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1997, 48, 251–275. [Google Scholar] [CrossRef] [PubMed]
- Jeandet, P.; Clément, C.; Courot, E.; Cordelier, S. Modulation of phytoalexin biosynthesis in engineered plants for disease resistance. Int. J. Mol. Sci. 2013, 14, 14136–14170. [Google Scholar] [CrossRef] [PubMed]
- Jeandet, P.; Hébrard, C.; Deville, M.A.; Crouzet, J. Deciphering the role of phytoalexins in plant-microorganism interactions and human health. Molecules 2014, 19, 18033–18056. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Van Etten, H.D. Introduction of plant and fungal genes into pea (Pisum sativum L.) hairy roots reduces their ability to produce pisatin and affects their response to a fungal pathogen. Mol. Plant. Microbe Interact. 2004, 17, 798–804. [Google Scholar] [CrossRef] [PubMed]
- Maloney, A.P.; Van Etten, H.D. A gene from the fungal pathogen Nectria haematococca that encodes the phytoalexin-detoxifying enzyme pisatin demethylase defines a new cytochrome P450 family. Mol. Genet. Genomics 1994, 243, 506–514. [Google Scholar] [CrossRef]
- Delserone, L.M.; McCluskey, K.D.; Mathews, E.; Van Etten, H.D. Pisatin demethylation by fungal pathogens and nonpathogens of pea: Association with pisatin tolerance and virulence. Physiol. Mol. Plant Pathol. 1999, 55, 317–326. [Google Scholar] [CrossRef]
- Mackintosh, S.F.; Matthews, D.E.; Van Etten, H.D. Two additional genes for pisatin demethylation and their relationship to the pathogenicity of Nectria haematococca on pea. Mol. Plant. Microbe Interact. 1989, 2, 354–362. [Google Scholar] [CrossRef]
- Courtois, J.; Seguin, J.P.; Roblot, C.; Heyraud, A.; Gey, C.; Dantas, L.; Barbotin, J.N.; Courtois, B. Exopolysaccharide production by the Rhizobium meliloti M5N1CS strain. Location and quantification of the sites of O-acetylation. Carbohydr. Polym. 1994, 25, 7–12. [Google Scholar] [CrossRef]
- Carman, L.M.; Lockwood, J.L. Factors affecting zoospore production by Aphanomyces euteiches. Phytopathology 1959, 49, 535. [Google Scholar]
- Vandemark, G.J.; Kraft, J.M.; Larsen, R.C.; Gritsenko, M.A.; Boge, W.L. A PCR-based assay by sequence-characterized DNA markers for the identification and detection of Aphanomyces euteiches. Phytopathology 2000, 90, 1137–1144. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |


| Gene Name | GenBank | Forward, Reverse Primers and Probes (5′–3′) | Tm (°C) | Amplicon Length |
|---|---|---|---|---|
| Accession N° | ||||
| Aphanomyces euteiches | AF228037 | TTTTGGAACACCCAAACGTACTG | 58 | 61 |
| AGTCCAAGAGGCATTCGACAA | 58 | |||
| ACGCTGAGCTTGAC | 68 | |||
| Housekeeping genes | ||||
| GAPDH (Mtgap1) 1 | X73150 | GTCTTTGCACACAGGAACCCA | 59 | 123 |
| GGCACCACCCTTCAAATGAG | 59 | |||
| CCCATGGGCCAGCAC | 70 | |||
| Defense and cell rescue | ||||
| Pathogenesis protein 1 (PR1) | AJ586324 | CCTTCCCCTCATGGCTATCC | 59 | 69 |
| TGTGGTGAGTTTTGAGCATATGAGA | 59 | |||
| AGTACTATCCACATCAACAC | 68 | |||
| Proline-rich protein (PRP) | AJ233399 | TGGCTTCCTTAACCTTCCTACTGT | 58 | 64 |
| TTGGCAAACCCTTGAGGAAT | 58 | |||
| ACTCCTTCTTGCTCTTAT | 68 | |||
| Mitogen-activated protein kinase (MAPK) | X70703 | CATTCCGCGAATGTTTTGC | 58 | 59 |
| TTGGCGTTCAGGAGAAGGTT | 58 | |||
| AGGGACTTAAAACCC | 70 | |||
| Reactive oxygen species (ROS) | ||||
| Superoxide dismutase (SOD) | AB087845 | CCATCATAGGAAGGGCTGTTGT | 59 | 63 |
| CGTGACCACCTTTCCCAAGA | 59 | |||
| CCATGCCGATCCTGAT | 70 | |||
| Peroxidase (POX) | AB193816 | ATGCAAGAACAGCAAGCCAAA | 59 | 69 |
| GGGTTGCAAGGTCAGATGATG | 59 | |||
| AACAGTCAAATCCC | 70 | |||
| Nitric oxide synthase (NOS) | AY672712 | GGCGGTGGTCAGGGTCTT | 59 | 63 |
| CCCTTTGGGACACGCTTTT | 59 | |||
| TGGAAAGAATGGATCTATT | 68 | |||
| Glutathione S-transferase (GST) | AB087837 | GAGAATGCCCTTGGTAAATTTGA | 58 | 70 |
| ACGCAATATCCACCAAACTGAAT | 58 | |||
| CCCCTTCCTTCTTGGTC | 69 | |||
| Catalase (Cat) | X60169 | CCAAGTGGTCTCACCACAACAAT | 59 | 69 |
| TGACCTCCTCATCCCTGTGAA | 59 | |||
| CCATGAGGGTTTCATG | 69 | |||
| Metallothionein (Metalo) | AB176564 | TCCGGCGAAGATCCAGTTT | 59 | 69 |
| CCACACTTGCAGCCACCAT | 59 | |||
| TGGTGCTGAAATGAGTG | 69 | |||
| Cell wall proteins & Basal defense | ||||
| Chitinase (Chit) | L37876 | CCTTCAAGACCGCTTTATGGTT | 58 | 64 |
| ACGTCGTGGCAGGATGGTT | 60 | |||
| ACGCCTCAGTCACCT | 68 | |||
| Beta-1,3-glucanase (β 1,3-Glu) | S51479 | TGGAATTGGTTGGGTGAATGT | 58 | 65 |
| TTGCAGAGCCTCCATCTGAA | 58 | |||
| TTGTTTCTGAGAGTGGTTG | 68 | |||
| Polygalacturonase inhibiting protein (PGIP) | AB087839 | CAGTGCTTTTCGGGAGCAA | 59 | 66 |
| CAAACGACAGCAAGTTCCTTGA | 59 | |||
| AAAGGACACAGATACTTGAT | 69 | |||
| JA signaling pathways | ||||
| Lipoxygenase (LOX) | X17061 | TGATCCGCGGTCTTCAAGAG | 59 | 60 |
| CACCGTATTCTGCGGGATCT | 59 | |||
| TTCCTCCGAAAAGC | 69 | |||
| Phenylpropanoid & Phytoalexin pathway | ||||
| Phenylalanine ammonia lyase (PAL) | D10001 | GCACTTAGAACTTCACCGCAATG | 60 | |
| GAAAGTTTCCACCATGCAAAGC | 60 | |||
| CCCTTTGATTGATGTTTC | 69 | |||
| Cinnamate-4-hydroxylase (C4H) | U29243 | GCCATAACCGCCATCACAAT | 59 | 61 |
| GGGCCAGGAGGGAGTTTGAA | 59 | |||
| AACTCCGCGGCAAA | 68 | |||
| Chalcone isomerase (CHI) | U03433 | GCTGCAGCATCCTCCATCA | 58 | 56 |
| CACCGCTGGGAACTCATGT | 58 | |||
| CGCAATCCACGTCGAG | 67 | |||
| Chalcone synthase (CHS) | D10662 | GACATGGTGGTCGTCGAGGTA | 58 | 70 |
| GCCCCCATTCTTTTATAGCTTTC | 58 | |||
| AGACTAGGGAAAGAGGCT | 70 | |||
| Isoflavone reductase (CHR) | S72472 | CTTTTGGCGTTGTACCATTCG | 59 | 68 |
| TCTTTGGCAGGGTCAATCTCA | 59 | |||
| AACAAATAAAGGGAGATGCAG | 70 | |||
| Disease resistance response (DRR) | ||||
| DRR230 | AJ308155 | TTGCAGGAACAACGAGCACTT | 60 | 61 |
| GCACCAGCAGCGAAAATCAT | 60 | |||
| CTCAGTGGGAGGTGCA | 69 | |||
| DRR276 | M18249 | TGCTGACACTCTTACTCCAAAGGT | 58 | 66 |
| CCGTTTCCTTCAACAATTTCG | 58 | |||
| TTGATGCCATCAAAAGTA | 69 | |||
| Disease resistance response protein (DRR) | ||||
| DRR49 | X13383 | GGTGATGCTGCTCCTAGTGAAGA | 58 | 66 |
| CTTGAAAAGACCATCCCCCTTA | 58 | |||
| CAACTCAAGACTGACAAAG | 68 | |||
| DRR206 | M18250 | GCTGGAGCTGACCCAATTGT | 59 | 68 |
| AAGAAATCTCCAGTACCACCAGTGA | 59 | |||
| CCAAAACTAGAGATATTTCT | 69 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Selim, S.; Sanssené, J.; Rossard, S.; Courtois, J. Systemic Induction of the Defensin and Phytoalexin Pisatin Pathways in Pea (Pisum sativum) against Aphanomyces euteiches by Acetylated and Nonacetylated Oligogalacturonides. Molecules 2017, 22, 1017. https://doi.org/10.3390/molecules22061017
Selim S, Sanssené J, Rossard S, Courtois J. Systemic Induction of the Defensin and Phytoalexin Pisatin Pathways in Pea (Pisum sativum) against Aphanomyces euteiches by Acetylated and Nonacetylated Oligogalacturonides. Molecules. 2017; 22(6):1017. https://doi.org/10.3390/molecules22061017
Chicago/Turabian StyleSelim, Sameh, Jean Sanssené, Stéphanie Rossard, and Josiane Courtois. 2017. "Systemic Induction of the Defensin and Phytoalexin Pisatin Pathways in Pea (Pisum sativum) against Aphanomyces euteiches by Acetylated and Nonacetylated Oligogalacturonides" Molecules 22, no. 6: 1017. https://doi.org/10.3390/molecules22061017
APA StyleSelim, S., Sanssené, J., Rossard, S., & Courtois, J. (2017). Systemic Induction of the Defensin and Phytoalexin Pisatin Pathways in Pea (Pisum sativum) against Aphanomyces euteiches by Acetylated and Nonacetylated Oligogalacturonides. Molecules, 22(6), 1017. https://doi.org/10.3390/molecules22061017





