Evaluation of the Mycobactericidal Effect of Thio-functionalized Carbohydrate Derivatives
Abstract
:1. Introduction
2. Results
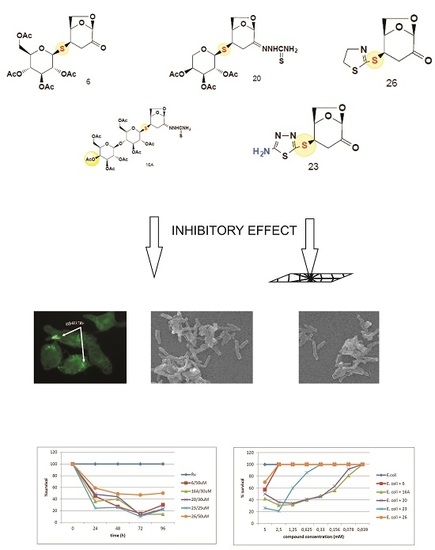
2.1. The Bactericidal Activity of Carbohydrates
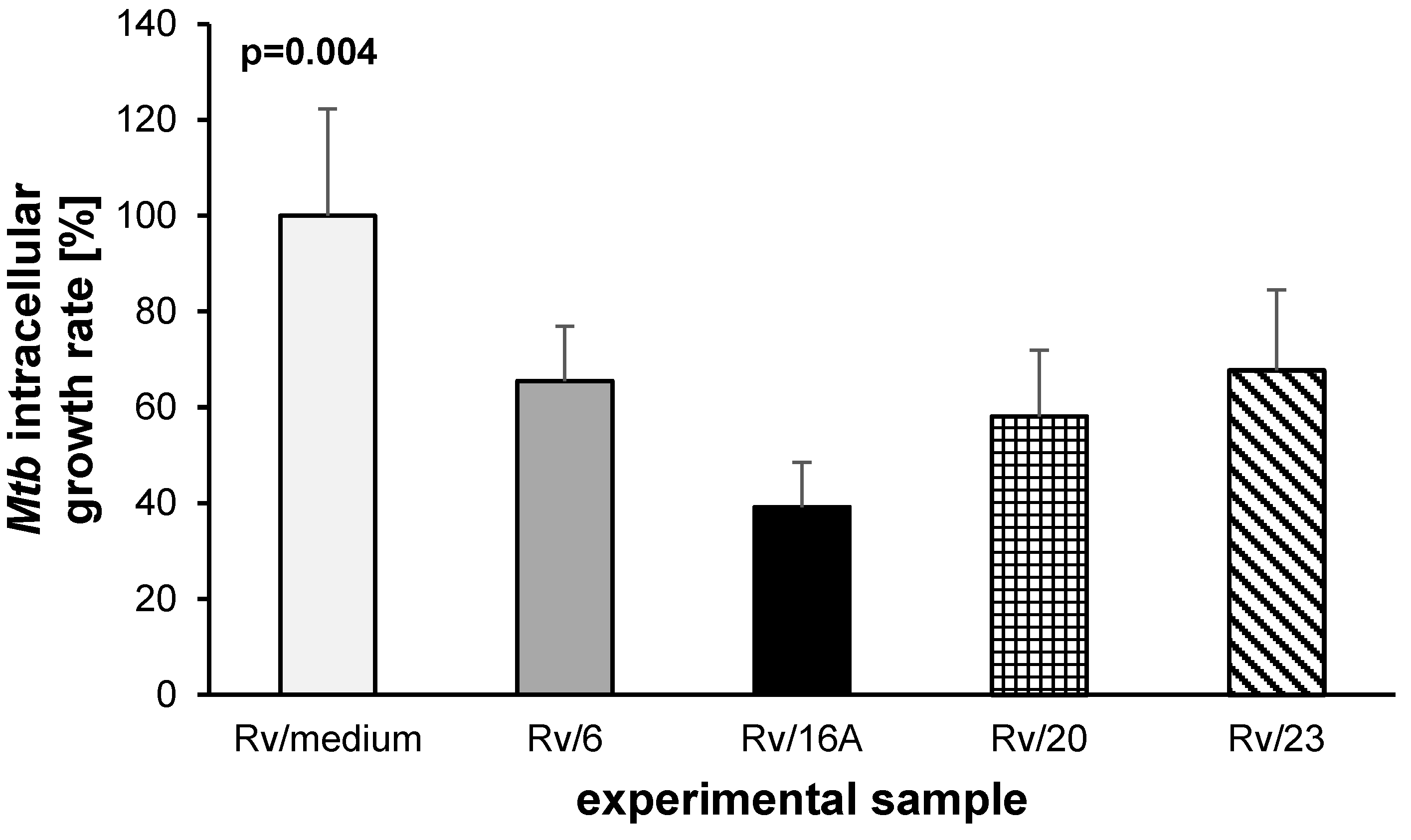
2.2. The Bactericidal Effect of Carbohydrates against Intracellularly Growing Tubercle Bacilli
2.3. The Carbohydrates’ Cytotoxicity
3. Discussion
4. Materials and Methods
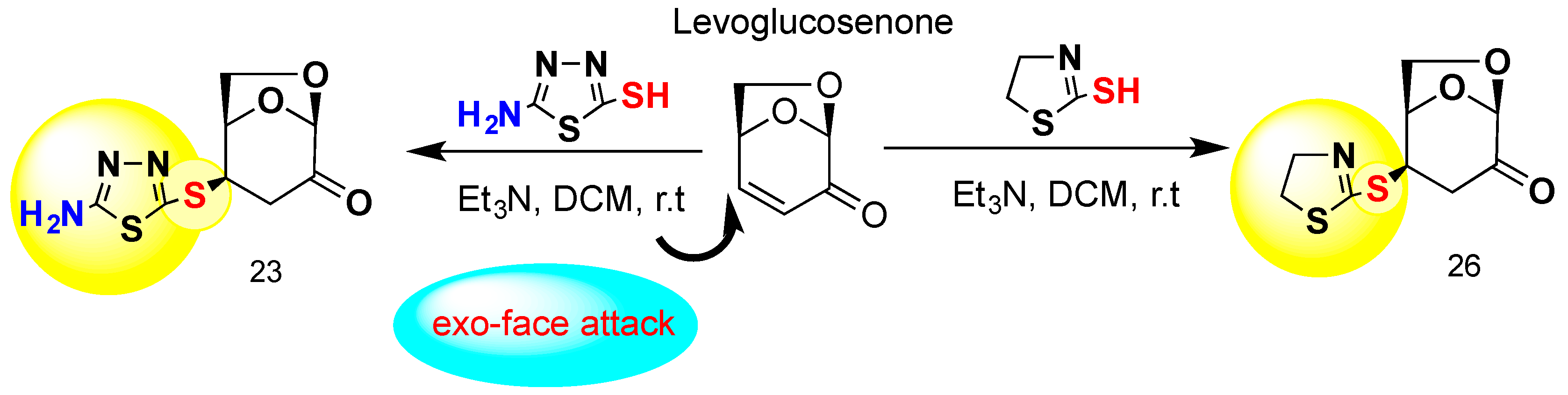
4.1. Chemistry
4.2. The Synthesis and Analysis of Carbohydrates
4.3. General Information
General Procedure for the Preparation of 23 and 26
4.4. Bacterial Growth Conditions and Susceptibility Tests
4.5. In Vitro Cytotoxicity Assay
4.6. Preparation of Human Monocyte-Derived Macrophages (MDMs)
4.7. Infection of MDMs with M. tuberculosis
4.8. Evaluation of the Bactericidal Effect of Carbohydrates against Intracellularly Growing Tubercle Bacilli
4.9. Statistical Analysis
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Witczak, Z.J. Thio sugars: Biological relevance as potential new therapeutics. Curr. Med. Chem. 1999, 6, 165–178. [Google Scholar] [PubMed]
- Witczak, Z.J.; Lorchak, D.; Nguyen, N. A click chemistry approach to glycomimetics: Michael addition of 2,3,4,6-tetra-o-acetyl-1-thio-beta-d-glucopyranose to 4-deoxy-1,2-o-isopropylidene-l-glycero-pent-4-enopyranos-3-ulose—A convenient route to novel 4-deoxy-(1→55)-5-c-thiodisaccharides. Carbohydr. Res. 2007, 342, 1929–1933. [Google Scholar] [CrossRef] [PubMed]
- Barchi, J.J. Emerging roles of carbohydrates and glycomimetics in anticancer drug design. Curr. Pharm. Des. 2000, 6, 485–501. [Google Scholar] [CrossRef] [PubMed]
- Berdy, J.A.A.; Bostian, M.; Mcnitt, K. Carbohydrates Antibiotics in Handbook of Antibiotic Compounds; CRC Press: Boca Raton, FL, USA, 1980; Volume 1. [Google Scholar]
- Witczak, Z.J. Carbohydrates as drugs and potential therapeuticals. Curr. Med. Chem. 1995, 1, 392–405. [Google Scholar]
- Oishi, H.; Noto, T.; Sasaki, H.; Suzuki, K.; Hayashi, T.; Okazaki, H.; Ando, K.; Sawada, M. Thiolactomycin, a new antibiotic. I. Taxonomy of the producing organism, fermentation and biological properties. J. Antibiot. (Tokyo) 1982, 35, 391–395. [Google Scholar] [CrossRef] [PubMed]
- Steinberg, T.H.; Mathews, D.E.; Durbin, R.D.; Burgess, R.R. Tagetitoxin: A new inhibitor of eukaryotic transcription by rna polymerase III. J. Biol. Chem. 1990, 265, 499–505. [Google Scholar] [PubMed]
- Udwadia, Z.F.; Amale, R.A.; Ajbani, K.K.; Rodrigues, C. Totally drug-resistant tuberculosis in India. Clin. Infect. Dis. 2012, 54, 579–581. [Google Scholar] [CrossRef] [PubMed]
- Jindani, A.; Dore, C.J.; Mitchison, D.A. Bactericidal and sterilizing activities of antituberculosis drugs during the first 14 days. Am. J. Respir. Crit. Care Med. 2003, 167, 1348–1354. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO Global Tuberculosis Report 2014; World Health Organization: Geneve, Switzerland, 2015. [Google Scholar]
- World Health Organization. WHO Drug-resistant Tb-surveillance and Response; World Health Organization: Geneve, Switrerland, 2015. [Google Scholar]
- Hoagland, D.T.; Liu, J.; Lee, R.B.; Lee, R.E. New agents for the treatment of drug-resistant mycobacterium tuberculosis. Adv. Drug Deliv. Rev. 2016, 102, 55–72. [Google Scholar] [CrossRef] [PubMed]
- Cheepsattayakorn, A.; Cheepsattayakorn, R. Novel compounds and drugs and recent patents in treating multidrug-resistant and extensively drug-resistant tuberculosis. Recent Pat. Antiinfect. Drug Discov. 2012, 7, 141–156. [Google Scholar] [CrossRef] [PubMed]
- Zumla, A.; Nahid, P.; Cole, S.T. Advances in the development of new tuberculosis drugs and treatment regimens. Nat. Rev. Drug Discov. 2013, 12, 388–404. [Google Scholar] [CrossRef] [PubMed]
- Zumla, A.I.; Gillespie, S.H.; Hoelscher, M.; Philips, P.P.; Cole, S.T.; Abubakar, I.; McHugh, T.D.; Schito, M.; Maeurer, M.; Nunn, A.J. New antituberculosis drugs, regimens, and adjunct therapies: Needs, advances, and future prospects. Lancet Infect. Dis. 2014, 14, 327–340. [Google Scholar] [CrossRef]
- Plocinska, R.; Korycka-Machala, M.; Plocinski, P.; Dziadek, J. Mycobacterial DNA replication as a target for antituberculosis drug discovery. Curr. Top. Med. Chem. 2017. [Google Scholar] [CrossRef]
- Wallis, R.S.; Maeurer, M.; Mwaba, P.; Chakaya, J.; Rustomjee, R.; Migliori, G.B.; Marais, B.; Schito, M.; Churchyard, G.; Swaminathan, S.; et al. Tuberculosis-advances in development of new drugs, treatment regimens, host-directed therapies, and biomarkers. Lancet Infect. Dis. 2016, 16, E34–E46. [Google Scholar] [CrossRef]
- Witczak, Z.J.; Kaplon, P.; Dey, P.M. Thio-sugars vii. Effect of 3-deoxy-4-s-(beta-d-gluco- and beta-d-galactopyranosyl)-4-thiodisaccharides and their sulfoxides and sulfones on the viability and growth of selected murine and human tumor cell lines. Carbohydr. Res. 2003, 338, 11–18. [Google Scholar] [CrossRef]
- Witczak, Z.J.; Poplawski, T.; Czubatka, A.; Sarnik, J.; Tokarz, P.; VanWert, A.L.; Bielski, R. A potential carb-pharmacophore for antineoplastic activity: Part 1. Bioorg. Med. Chem. Lett. 2014, 24, 1752–1757. [Google Scholar] [CrossRef] [PubMed]
- Czubatka, A.; Sarnik, J.; Lucent, D.; Blasiak, J.; Witczak, Z.J.; Poplawski, T. A novel carbohydrate derived compound fcp5 causes DNA strand breaks and oxidative modifications of DNA bases in cancer cells. Chem. Biol. Interact. 2015, 227, 77–88. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.L.; Chen, C.; Xiong, Y.; Zhang, L.H.; Ye, J.; Ye, X.S. Synthesis of n-substituted iminosugar derivatives and their immunosuppressive activities. Carbohydr. Res. 2010, 345, 780–786. [Google Scholar] [CrossRef] [PubMed]
- Compain, P.M.; Martin, O.R. Iminosugars: From Synthesis to Therapeutic Applications; Wiley: Chichester, UK, 2007. [Google Scholar]
- Strus, M.; Mikolajczyk, D.; Machul, A.; Heczko, P.B.; Chronowska, A.; Stochel, G.; Gallienne, E.; Nicolas, C.; Martin, O.R.; Kyziol, A. Effects of the selected iminosugar derivatives on Pseudomonas aeruginosa biofilm formation. Microb. Drug Resist. 2016, 22, 638–645. [Google Scholar] [CrossRef] [PubMed]
- Lillelund, V.H.; Jensen, H.H.; Liang, X.; Bols, M. Recent developments of transition-state analogue glycosidase inhibitors of non-natural product origin. Chem. Rev. 2002, 102, 515–553. [Google Scholar] [CrossRef] [PubMed]
- Richards, M.R.; Lowary, T.L. Chemistry and biology of galactofuranose-containing polysaccharides. ChemBioChem 2009, 10, 1920–1938. [Google Scholar] [CrossRef] [PubMed]
- Cren, S.; Gurcha, S.S.; Blake, A.J.; Besra, G.S.; Thomas, N.R. Synthesis and biological evaluation of new inhibitors of udp-galf transferase—A key enzyme in M. tuberculosis cell wall biosynthesis. Org. Biomol. Chem. 2004, 2, 2418–2420. [Google Scholar] [CrossRef] [PubMed]
- Peltier, P.; Euzen, R.; Daniellou, R.; Nugier-Chauvin, C.; Ferrieres, V. Recent knowledge and innovations related to hexofuranosides: Structure, synthesis and applications. Carbohydr. Res. 2008, 343, 1897–1923. [Google Scholar] [CrossRef] [PubMed]
- Gorska, S.; Jachymek, W.; Rybka, J.; Strus, M.; Heczko, P.B.; Gamian, A. Structural and immunochemical studies of neutral exopolysaccharide produced by Lactobacillus johnsonii 142. Carbohydr. Res. 2010, 345, 108–114. [Google Scholar] [CrossRef] [PubMed]
- Lipinski, T.; Jones, C.; Lemercinier, X.; Korzeniowska-Kowal, A.; Strus, M.; Rybka, J.; Gamian, A.; Heczko, P.B. Structural analysis of the Lactobacillus rhamnosus strain kl37c exopolysaccharide. Carbohydr. Res. 2003, 338, 605–609. [Google Scholar] [CrossRef]
- Ayers, B.; Long, H.; Sim, E.; Smellie, I.A.; Wilkinson, B.L.; Fairbanks, A.J. Stereoselective synthesis of beta-arabino glycosyl sulfones as potential inhibitors of mycobacterial cell wall biosynthesis. Carbohydr. Res. 2009, 344, 739–746. [Google Scholar] [CrossRef] [PubMed]
- Lo Conte, M.; Marra, A.; Chambery, A.; Gurcha, S.S.; Besra, G.S.; Dondoni, A. Modular approach to triazole-linked 1,6-alpha-d-oligomannosides to the discovery of inhibitors of mycobacterium tuberculosis cell wall synthetase. J. Org. Chem. 2010, 75, 6326–6336. [Google Scholar] [CrossRef] [PubMed]
- Benz, G.; Schroder, T.; Kurz, J.; Wunsche, C.; Karl, W.; Steffens, G.; Pfitzner, J.; Schmidt, D. Constitution of the deferriform of the albomycins delta-1, delta-2, and epsilon. Angew. Chem. Int. Ed. 1982, 21, 527–528. [Google Scholar] [CrossRef]
- Stefanska, A.L.; Fulston, M.; Houge-Frydrych, C.S.; Jones, J.J.; Warr, S.R. A potent seryl trna synthetase inhibitor sb-217452 isolated from a streptomyces species. J. Antibiot. (Tokyo) 2000, 53, 1346–1353. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, A.; Fiedler, H.P.; Braun, V. Uptake and conversion of the antibiotic albomycin by Escherichia-coli k-12. Eur. J. Biochem. 1979, 99, 517–524. [Google Scholar] [CrossRef] [PubMed]
- Pramanik, A.; Braun, V. Albomycin uptake via a ferric hydroxamate transport system of Streptococcus pneumoniae r6. J. Bact. 2006, 188, 3878–3886. [Google Scholar] [CrossRef] [PubMed]
- Kremer, L.; Douglas, J.D.; Baulard, A.R.; Morehouse, C.; Guy, M.R.; Alland, D.; Dover, L.G.; Lakey, J.H.; Jacobs, W.R.; Brennan, P.J.; et al. Thiolactomycin and related analogues as novel anti-mycobacterial agents targeting kasa and kasb condensing enzymes in mycobacterium tuberculosis. J. Biol. Chem. 2000, 275, 16857–16864. [Google Scholar] [CrossRef] [PubMed]
- Wilson, D.M.; Putzrath, R.M.; Wilson, T.H. Inhibition of growth of escherichia coli by lactose and other galactosides. Biochim. Biophys. Acta 1981, 649, 377–384. [Google Scholar] [CrossRef]
- Jankute, M.; Cox, J.A.; Harrison, J.; Besra, G.S. Assembly of the mycobacterial cell wall. Annu. Rev. Microbiol. 2015, 69, 405–423. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Kumar, N. Synthesis and biological activity of acetylacetone thiosemicarbazone and their matallic complexes. Int. Curr. Pharm. J. 2013, 2, 88–91. [Google Scholar] [CrossRef]
- Opletalova, V.; Dolezel, J. Thiosemicarbazones and their antimycobacterial effects. Ces. Slov. Farm. 2013, 62, 78–83. [Google Scholar]
- Singh, R.; Mishra, P.S.; Mishra, R. Synthesis and evaluation of biological activities of thiosemicarbazones derivatives. Int. J. PharmTech Res. 2011, 3, 1625–1629. [Google Scholar]
- Witczak, Z.J.; Chhabra, R.; Chen, H.; Xie, X.Q. Thiosugars 2 A novel approach to thiodisaccharides-the synthesis of 3-deoxy-4-thiocellobiose from levoglucosenone. Carbohydr. Res. 1997, 301, 167–175. [Google Scholar] [CrossRef]
- Witczak, Z.J.; Sarnik, J.; Czubatka, A.; Forma, E.; Poplawski, T. Thio-sugar motif of functional carb-pharmacophore for antineoplastic activity. Part 2. Bioorg. Med. Chem. Lett. 2014, 24, 5606–5611. [Google Scholar] [CrossRef] [PubMed]
- Broxterman, H.J.G.; Vandermarel, G.A.; Vanboom, J.H. Synthesis of 2-acetamido-2-deoxy-d-mannose analogs as potential inhibitors of sialic-acid biosynthesis. J. Carbohydr. Chem. 1991, 10, 215–237. [Google Scholar] [CrossRef]
- Creighton, A.M.; Owen, L.N. Some carbohydrate episulphides. J. Chem. Soc. 1960, 1024–1029. [Google Scholar] [CrossRef]
- Feather, M.S.; Whistler, R.L. Derivatives of 5-deoxy-5-mercapto-d-glucose. Tetrahedron Lett. 1962, 667–668. [Google Scholar] [CrossRef]
- Franz, A.H.; Gross, P.H.; Samoshin, V.V. Syntheses of small cluster oligosaccharide mimetics. Arkivoc 2008, 271–308. [Google Scholar]
- Czubatka-Bienkowska, A.; Sarnik, J.; Macieja, A.; Galita, G.; Witczak, Z.J.; Poplawski, T. Thio-functionalized carbohydrate thiosemicarbazides and their evaluation of anticancer activity. Bioorg. Med. Chem. Lett. 2017. [Google Scholar] [CrossRef]
- Czubatka-Bienkowska, A.; Sarnik, J.; Macieja, A.; Witczak, Z.J.; Poplawski, T. The synthesis and cytotoxic effect of novel glycosylated thiosemicarbazides and their analogs as anticancer agents. Proceedings of 252nd ACS National Meeting, Philadelphia, PA, USA, 21–25 August 2016; pp. 20–24. [Google Scholar]
- Czubatka-Bienkowska, A.; Sarnik, J.; Macieja, A.; Witczak, Z.J.; Poplawski, T. The synthesis characterization, molecular modeling and potential anticancer activities of novel 1,3,4-thiadiazole derivative. Proceedings of 252th ACS National Meeting, Philadelphia, PA, USA, 21–25 August 2016; pp. 20–24. [Google Scholar]
- Castro, A.; Castano, T.; Encinas, A.; Porcal, W.; Gil, C. Advances in the synthesis and recent therapeutic applications of 1,2,4-thiadiazole heterocycles. Bioorg. Med. Chem. 2006, 14, 1644–1652. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Li, C.Y.; Wang, X.M.; Yang, Y.H.; Zhu, H.L. 1,3,4-Thiadiazole: Synthesis, reactions, and applications in medicinal, agricultural, and materials chemistry. Chem. Rev. 2014, 114, 5572–5610. [Google Scholar] [CrossRef] [PubMed]
- Saeed, A.; Tehseen, Y.; Rafique, H.; Furtmannn, N.; Bajorath, J.; Florke, U.; Iqbal, J. Benzothiazoyl substituted iminothiazolidinones and benzamido-oxothiazolidines as potent and partly selective aldose reductase inhibitors. Med. Chem. Commun. 2014, 5, 1371–1380. [Google Scholar] [CrossRef]
- Clinical Laboratory Standards and Institute. Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria that Grow Aerobically, 8th ed.; Wayne, PA, USA, 2009; Volume 29. [Google Scholar]
- Danciger, J.S.; Lutz, M.; Hama, S.; Cruz, D.; Castrillo, A.; Lazaro, J.; Phillips, R.; Premack, B.; Berliner, J. Method for large scale isolation, culture and cryopreservation of human monocytes suitable for chemotaxis, cellular adhesion assays, macrophage and dendritic cell differentiation. J. Immunol. Methods 2004, 288, 123–134. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |

| Compounds | Tested Anti-Mtb | Tested Anti-E. coli, S. aureus |
|---|---|---|
| 5-thio-d-glucose pentaacetate (1) | MIC50 > 1 mM/OD | ND |
| 6-thio-β-d-fructopyranose (2) | MIC50 > 1 mM/OD | MIC50 > 2 mM/E. coli MIC50 > 2 mM/S. aureus |
| 1,6-anhydro-5-C-hydroxymethyl-α-l-altro-pyranose (3) | MIC50 > 1 mM/OD | ND |
| 2-C-methylene-myo-inositol oxide (4) | MIC50 > 1 mM/OD | ND |
| 1,5-anhydro-6-deoxy-6-methanesulfamido-d-glucitol (5) | MIC50 > 1 mM/OD | MIC50 > 2 mM/E. coli MIC50 > 2 mM/S. aureus |
| 1,6-anhydro-3-deoxy-4-S-(2,3,4,6-tetra-O-acetyl-β-d-glucopyranosyl-d-glycero-hexopyranos-2-ulose (6) | MIC50 = 0.05 mM/CFU | MIC50 = 5 mM/E. coli MIC50 = 2.5 mM/S. aureus |
| 1,6-anhydro-3-deoxy-4-S-β-d-glucopyranosyl-d-glycero-hexopyranos-2-ulose (7) | MIC50 = 0.5 mM/CFU | MIC50 > 10 mM/E. coli MIC50 > 10 mM/S. aureus |
| 1,6-anhydro-3-deoxy-4-S-β-salicylyl-d-glycero-hexopyranos-2-ulose (8) | MIC50 = 0.2 mM/CFU | MIC50 > 10 mM/E. coli MIC50 > 10 mM/S. aureus |
| 1,6-anhydro-3-deoxy-4-S-(2,3,4,6-tetra-O-acetyl-β-d-galactopyranosyl-d-glycero-hexopyranos-2-ulose (9) | MIC50 > 0.15 mM/OD | ND |
| 2-acetamido-2-dexy-1,5-anhydro-d-mannitol (10) | MIC50 > 1 mM/OD | MIC50 > 10 mM/E. coli MIC50 > 10 mM/S. aureus |
| 1,2-O-isopropylidene-5,6-anhydro-d-glucofuranose (11) | MIC50 = 0.75 mM/OD | ND |
| 1,2-O-isopropylidene-3,5-di-O-acetyl-6-methoxy-d-glucofuranose (12) | MIC50 > 1 mM/OD | MIC50 > 5 mM/E. coli MIC50 > 10 mM/S. aureus |
| 1-C-nitromethyl-d-gluco-pyranose (13) | MIC50 > 1 mM/OD | MIC50 > 10 mM/E. coli MIC50 > 10 mM/S. aureus |
| 1,6-anhydro-3,4-dideoxy-α-d-glycero-hex-3-enopyranos-2-thiosemicarbazone (14) | MIC50 = 0.35 mM/OD | MIC50 > 5 mM/E. coli MIC50 > 5 mM/S. aureus |
| 1,6-anhydro-3-deoxy-4-S-(octa-O-acetyl-β-d-lactosyl-d-glycero-hexopyranos-2-ulose (16) | MIC50 = 0.5 mM/OD | MIC50 > 5 mM/E. coli MIC50 > 5 mM/S. aureus |
| octa-O-acetyl-β-d-lactosyl-S-1-adamantane (19) | MIC50 = 0.5 mM/OD | ND |
| 1,6-anhydro-3,4-dideoxy-α-d-glycero-hexopyranos-2-thiosemicarbazone (14A) | MIC50 > 1 mM/OD | MIC50 > 2.5 mM/E. coli MIC50 > 1.25 mM/S. aureus |
| 1,6-anhydro-3-deoxy-4-S-(octa-O-acetyl-β-d-lactosyl-d-glycero-hexopyranos-2-thiosemicarbazone (16A) | MIC50 = 0.03 mM/CFU | MIC50 > 150 µM/E. coli MIC50 > 1.25 mM/S. aureus |
| 1,6-anhydro-3-deoxy-4-S-(2,3,4-tri-O-acetyl-β-l-arabino)-d-glycero-hexopyranos-2-ulose (20) | MIC50 = 0.03 mM/CFU | MIC50 > 300 µM/E. coli MIC50 > 1.25 mM/S. aureus |
| 1,6-anhydro-3-deoxy-4-S-(5-amino-1,3,4-thiadiazol-2-yl)-d-glycero-hexopyranos-2-ulose (23) | MIC50 = 0.025 mM/CFU | MIC50 > 1.25 mM/E. coli MIC50 > 0.625 mM/S. aureus |
| 1,6-anhydro-3-deoxy-4-S-(4,5-dihydrothiazol-2-yl-d-glycero-hexopyranos-2-ulose (26) | MIC50 = 0.05 mM/CFU | MIC50 > 2.5 mM/E. coli MIC50 > 2.5 mM/S. aureus |
| Experimental Sample | OD[570nm] ± SD | Cell Viability [%] |
|---|---|---|
| MDMs/medium | 2.381 ± 0.038 | n/a |
| MDMs/6 | 2.163 ± 0.054 | 91 |
| MDMs/16A | 2.345 ± 0.043 | 98 |
| MDMs/20 | 2.269 ± 0.048 | 95 |
| MDMs/23 | 2.428 ± 0.031 | 100 |
| Compounds | IC50 [µM] |
|---|---|
| 1 | >1000 |
| 2 | >1000 |
| 6 | 45 |
| 7 | 504 |
| 8 | 515 |
| 9 | 217 |
| 14 | 329 |
| 14a | >1000 |
| 16 | 250 |
| 16a | 240 |
| 19 | 227 |
| 20 | 500 |
| 23 | 4 |
| 26 | 11 |
| Compound | LogP/cLog | References |
|---|---|---|
| 5-thio-D-glucose pentaacetate (1) | LogP: −0.51 cLog: 0.72825 | [19] |
| 6-thio-β-d-fructopyranose (2) | LogP: −1.36 cLog: −1.9984 | [19] |
| 1,6-anhydro-5-C-hydroxymethyl-α-l-altro-pyranose (3) | LogP: −2.38 cLog: −1.5973 | [19] |
| 2-C-methylene-myo-inositol oxide (4) | LogP: −3.22 cLog: −11214 | [19] |
| 1,5-anhydro-6-deoxy-6-methanesulfamido-d-glucitol (5) | LogP: −3.6 cLog: −1.1515 | [19] |
| 1,6-anhydro-3-deoxy-4-S-(2,3,4,6-tetra-O-acetyl-β-d-glucopyranosyl-d-glycero-hexopyranos-2-ulose (6) | LogP: −1.13 cLog: 0.4407 | [18,42] |
| 1,6-anhydro-3-deoxy-4-S-β-d-glucopyranosyl-d-glycero-hexopyranos-2-ulose (7) | LogP: −2.05 cLog: −1.69975 | [18,42] |
| 1,6-anhydro-3-deoxy-4-S-β-salicylyl-d-glycero-hexopyranos-2-ulose (8) | LogP: 1.28 cLog: 0.8949 | [43] |
| 1,6-anhydro-3-deoxy-4-S-(2,3,4,6-tetra-O-acetyl-β-d-galactopyranosyl-d-glycero-hexopyranos-2-ulose (9) | LogP: −1.13 cLog: 0.4407 | [18,42] |
| 2-acetamido-2-dexy-1,5-anhydro-d-mannitol (10) | Log P: −2.75 cLog: 0.993001 | [44] |
| 1,2-O-isopropylidene-5,6-anhydro-3-hydroxy-d-glucofuranose (11) | LogP: −0.39 cLog: 0.64599 | [45] |
| 1,2-O-isopropylidene-3,5-di-O-acetyl-6-methoxy-d-glucofuranose (12) | LogP: −0.2 cLog: 0.9672 | [46] |
| 1-C-nitromethyl-d-gluco-pyranose (13) | LogP: −4.22 cLog: −1.9428 | [47] |
| 1,6-anhydro-3,4-dideoxy-α-d-glycero-hex-3-enopyranos-2-thiosemicarbazone (14) | LogP:−0.18 cLog:0.372 | [48,49] |
| 1,6-anhydro-3-deoxy-4-S-(octa-O-acetyl-β-d-lactosyl-d-glycero-hexopyranos-2-ulose (16) | LogP:−2.18 cLog:0.854498 | [48,49] |
| 1,6-anhydro-3,4-dideoxy-α-d-glycero-hexopyranos-2-thiosemicarbazone (14A) | LogP: −0.29 cLog: 0.204 | [48,49] |
| 1,6-anhydro-3-deoxy-4-S-(octa-O-acetyl-β-d-lactosyl-d-glycero-hexopyranos-2-thiosemicarbazone (16A) | LogP: NA cLog: 1.26 | [48,49] |
| Octa-O-acetyl-β-d-lactosyl-S-1-adamantane (19) | cLog: 4.0538 | [49] |
| 1,6-anhydro-3-deoxy-4-S-(2,3,4-tri-O-acetyl-β-l-arabino)-d-glycero-hexopyranos-2-ulose (20) | LogP: −0.9 cLog: 0.632201 | [49] |
| 1,6-anhydro-3-deoxy-4-S-(5-amino-1,3,4-thiadiazol-2-yl)-d-glycero-hexopyranos-2-ulose (23) | LogP: 1.37 cLog: −0.535599 | [50] & this work |
| 1,6-anhydro-3-deoxy-4-S-(4,5-dihydrothiazol-2-yl-d-glycero-hexopyranos-2-ulose (26) | LogP: 1.13 cLog: 0.0390999 | [50] & this work |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Korycka-Machała, M.; Brzostek, A.; Dziadek, B.; Kawka, M.; Popławski, T.; Witczak, Z.J.; Dziadek, J. Evaluation of the Mycobactericidal Effect of Thio-functionalized Carbohydrate Derivatives. Molecules 2017, 22, 812. https://doi.org/10.3390/molecules22050812
Korycka-Machała M, Brzostek A, Dziadek B, Kawka M, Popławski T, Witczak ZJ, Dziadek J. Evaluation of the Mycobactericidal Effect of Thio-functionalized Carbohydrate Derivatives. Molecules. 2017; 22(5):812. https://doi.org/10.3390/molecules22050812
Chicago/Turabian StyleKorycka-Machała, Małgorzata, Anna Brzostek, Bożena Dziadek, Malwina Kawka, Tomasz Popławski, Zbigniew J. Witczak, and Jarosław Dziadek. 2017. "Evaluation of the Mycobactericidal Effect of Thio-functionalized Carbohydrate Derivatives" Molecules 22, no. 5: 812. https://doi.org/10.3390/molecules22050812
APA StyleKorycka-Machała, M., Brzostek, A., Dziadek, B., Kawka, M., Popławski, T., Witczak, Z. J., & Dziadek, J. (2017). Evaluation of the Mycobactericidal Effect of Thio-functionalized Carbohydrate Derivatives. Molecules, 22(5), 812. https://doi.org/10.3390/molecules22050812