Impact of a Microbial Cocktail Used as a Starter Culture on Cocoa Fermentation and Chocolate Flavor
Abstract
:1. Introduction
2. Results
2.1. Culture-Independent Analysis with PCR–DGGE
2.2. Chemical Changes During Fermentation
2.2.1. Sugar Consumption and Metabolite Production
2.2.2. Volatile Compounds
2.3. Sensorial Analyses of Chocolate
3. Discussion
4. Materials and Methods
4.1. Fermentation Experiments, Inoculation, and Sampling
4.2. Culture-Independent Microbiological Analysis
4.2.1. DNA Extraction and Polymerase Chain Reaction
4.2.2. PCR–DGGE Analysis
4.3. Chromatographic Analysis
4.3.1. Sugars, Alcohols, and Organic Acid Extraction and HPLC Analyses
4.3.2. Characterization of Volatile Compounds by Headspace-Solid Phase Microextraction Gas Chromatography-Mass Spectrometry
4.4. Sensory Analysis
4.5. Statistical Analyses
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pereira, J.L.; Ram, A.; Figueiredo, J.M.; Almeida, L.C.C. The first occurrence of “witches’ broom” disease in the principal cocoa growing region of Brazil. Trop. Agric. 1990, 67, 188–189. [Google Scholar]
- Lopes, U.V.; Monteiro, W.R.; Pires, J.L.; Clement, D.; Yamada, M.M.; Gramacho, K.P. Cacao breeding in Bahia, Brazil: Strategies and results. Crop Breed. Appl. Biotechnol. 2011, 1, 73–81. [Google Scholar] [CrossRef]
- Moreira, I.M.V.; Miguel, M.G.C.P.; Duarte, W.F.; Dias, D.R.; Schwan, R.F. Microbial succession and the dynamics of metabolites and sugars during the fermentation of three different cocoa (Theobroma cacao L.) hybrids. Food Res. Int. 2013, 54, 9–17. [Google Scholar] [CrossRef]
- Ramos, C.L.; Dias, D.R.; Miguel, M.G.C.P.; Schwan, R.F. Impact of different cocoa hybrids (Theobroma cacao L.) and S. cerevisiae UFLA CA11 inoculation on microbial communities and volatile compounds of cocoa fermentation. Food Res. Int. 2014, 64, 908–918. [Google Scholar] [CrossRef]
- Clapperton, J.F.; Lockwood, R.; Yow, S.T.K.; Lim, D.H.K. Effects of planting materials on flavour. Cocoa Grow. Bull. 1994, 48, 47–63. [Google Scholar]
- Efraim, P.; Pires, J.L.; Garcia, A.O.; Grimaldi, R.; Luccas, V.; Pezoa-Garcia, N.H. Characteristics of cocoa butter and chocolates obtained from cocoa varieties grown in Bahia, Brazil. Eur. Food Res. Technol. 2013, 237, 419–428. [Google Scholar] [CrossRef]
- Menezes, A.G.T.; Batista, N.N.; Ramos, C.L.; Silva, A.R.A.; Efraim, P.; Pinheiro, A.C.M.; Schwan, R.F. Investigation of chocolate produced from four different Brazilian varieties of cocoa (Theobroma cacao L.) inoculated with Saccharomyces cerevisiae. Food Res. Int. 2016, 81, 83–90. [Google Scholar] [CrossRef]
- Oliveira, B.F.; Silva, S.D.V.M.; Damaceno, V.O.; Filho, L.P.S. Identificação de fontes de resistência a Ceratocystis cacaofunesta em mudas de cacaueiro. Agrotópica 2009, 21, 83–88. [Google Scholar]
- Pires, J.L.; Rosa, E.S.; Macêdo, M.M. Avaliação de clones de cacaueiro na Bahia, Brasil. Agrotópica 2012, 24, 79–84. [Google Scholar]
- Silva, S.D.V.M.; Pinto, L.R.M.; Oliveira, B.F.; Damaceno, V.O.; Pires, J.L.; Dias, C.T.S. Resistência de progênies de cacaueiro à murcha-de-Ceratocystis. Trop. Plant Pathol. 2012, 37, 191–195. [Google Scholar] [CrossRef]
- Biehl, B.; Meyer, B.; Crone, G.; Pollmann, L.; Said, M.B. Chemical and physical changes in the pulp during ripening and post-harvest storage of cocoa pods. J. Sci. Food Agric. 1989, 48, 189–208. [Google Scholar] [CrossRef]
- Schwan, R.F. Cocoa fermentations conducted with a defined microbial cocktail inoculum. Appl. Environ. Microbiol. 1998, 64, 1477–1483. [Google Scholar] [PubMed]
- Afoakwa, E.O.; Paterson, A.; Fowler, M.; Ryan, A. Flavor formation and character in cocoa and chocolate: A critical review. Crit. Rev. Food Sci. 2008, 48, 840–857. [Google Scholar] [CrossRef] [PubMed]
- Pereira, G.V.M.; Miguel, M.G.C.P.; Ramos, C.L.; Schwan, R.F. Microbiological and physicochemical characterization of small-scale cocoa fermentations and screening of yeast and bacteria strains to develop a defined starter culture. Appl. Environ. Microb. 2012, 78, 5395–5405. [Google Scholar] [CrossRef] [PubMed]
- Schwan, R.F.; Pereira, G.V.M.; Fleet, G.H. Microbial activities during cocoa fermentation. In Cocoa and Coffee Fermentations; Schwan, R.F., Fleet, G.H., Eds.; CRC Press: Boca Raton, FL, USA, 2014; pp. 129–190. [Google Scholar]
- Ardhana, M.; Fleet, G. The microbial ecology of cocoa bean fermentations in Indonesia. Int. J. Food Microbiol. 2003, 86, 87–99. [Google Scholar] [CrossRef]
- Schwan, R.F.; Wheals, A.E. The microbiology of cocoa fermentation and its role in chocolate quality. Crit. Rev. Food Sci. 2004, 44, 205–221. [Google Scholar] [CrossRef] [PubMed]
- Jespersen, L.; Nielsen, D.S.; Hønholt, S.; Jakobsen, M. Occurrence and diversity of yeasts involved in fermentation of West African cocoa beans. FEMS Yeast Res. 2005, 5, 441–453. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, D.S.; Teniola, O.D.; Ban-Koffi, L.; Owusu, M.; Andersson, T.S.; Holzapfel, W.H. The microbiology of Ghanaian cocoa fermentations analysed using culture dependent and culture independent methods. Int. J. Food Microbiol. 2007, 114, 168–186. [Google Scholar] [CrossRef] [PubMed]
- Pereira, G.V.M.; Magalhães, K.T.; Almeida, E.G.; Coelho, I.S.C.; Schwan, R.F. Spontaneous cocoa bean fermentation carried out in a novel-design stainless steel tank: Influence on the dynamics of microbial populations and physical-chemical properties. Int. J. Food Microbiol. 2013, 161, 121–133. [Google Scholar] [CrossRef] [PubMed]
- Camu, N.; De Winter, T.; Addo, S.K.; Takarama, J.S.; Bernaert, H.; De Vuyst, L. Fermentation of cocoa beans: Influence of microbial activities and polyphenol concentrations on the flavour of chocolate. J. Sci. Food Agric. 2008, 88, 2288–2297. [Google Scholar] [CrossRef]
- Papalexandratou, Z.; Vranckena, G.; De Bruyneb, K.; Vandammeb, P.; De Vuys, L. Spontaneous organic cocoa bean box fermentations in Brazil are characterized by a restricted species diversity of lactic acid bacteria and acetic acid bacteria. Food Microbiol. 2011, 28, 1326–1338. [Google Scholar] [CrossRef] [PubMed]
- Schwan, R.F.; Rose, A.H.; Board, R.G. Microbial fermentation of cocoa beans, with emphasis on enzymatic degradation of the pulp. J. Appl. Bacteriol. 1995, 79, 96S–107S. [Google Scholar]
- Rodriguez-Campos, J.; Escalona-buendía, H.B.; Orozco-Avila, I.; Lugo-Cervantes, E.; Jaramillo-Flores, M.E. Dynamics of volatile and non-volatile compounds in cocoa (Theobroma cocoa L.) during fermentation and drying process using principal components analysis. Food Res. Int. 2011, 44, 250–258. [Google Scholar] [CrossRef]
- Ho, V.T.T.; Zhao, J.; Fleet, G. Yeasts are essential for cocoa bean fermentation. Int. J. Food Microbiol. 2014, 174, 72–87. [Google Scholar] [CrossRef] [PubMed]
- Albertini, B.; Schoubben, A.; Guarnaccia, D.; Pinelli, F.; Vecchia, M.D.; Ricci, M.; Renzo, G.C.D.; Blasi, P. Effect of Fermentation and Drying on Cocoa Polyphenols. J. Agric. Food Chem. 2015, 63, 9948–9953. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Campos, J.; Escalona-Buendía, H.; Contreras-Ramos, S.; Orozco-Avila, I.; Jaramillo-Flores, E.; Lugo-Cervantes, E. Effect of fermentation time and drying temperature on volatile compounds in cocoa. Food Chem. 2012, 132, 277–288. [Google Scholar] [CrossRef] [PubMed]
- Serra-Bonvehí, J. Investigation of aromatic compounds in roasted cocoa powder. Eur. Food Res. Technol. 2005, 221, 19–29. [Google Scholar] [CrossRef]
- Aculey, P.; Snitkjaer, P.; Owusu, M.; Bassompiere, M.; Takrama, J.; Nørgaard, L. Ghanaian cocoa bean fermentation characterized by spectroscopic and chromatographic methods and chemometrics. J. Food Sci. 2010, 75, S300–S307. [Google Scholar] [CrossRef] [PubMed]
- Frauendorfer, F.; Schieberle, P. Changes in key aroma compounds of Criollo cocoa beans during roasting. J. Agric. Food Chem. 2008, 56, 10244–10251. [Google Scholar] [CrossRef] [PubMed]
- Luna, F.; Crouzillat, D.; Cirou, L.; Bucheli, P. Chemical Composition and Flavor of Ecuadorian Cocoa Liquor. J. Agric. Food Chem. 2002, 50, 3527–3532. [Google Scholar] [CrossRef] [PubMed]
- Batista, N.N.; Ramos, C.L.; Ribeiro, D.D.; Pinheiro, A.C.M.; Schwan, R.F. Dynamic behavior of Saccharomyces cerevisiae, Pichia kluyveri and Hanseniaspora uvarum during spontaneous and inoculated cocoa fermentations and their effect on sensory characteristics of chocolate. LWT Food Sci. Technol. 2015, 63, 221–227. [Google Scholar] [CrossRef]
- Moskowitz, H.R. Product Testing and Sensory Evaluation of Foods: Marketing and R &D Approaches; Food & Nutrition Press: Westport, CT, USA, 1983. [Google Scholar]
- Pineau, N.; Schich, P.; Cordelle, S.; Mathonnièrea, C.; Issanchouc, S.; Imbertd, A.; Köster, E. Temporal dominance of Sensations: Construction of the TDS curves and comparison with time intensity. Food Qual. Pref. 2009, 20, 450–455. [Google Scholar] [CrossRef]
- Nunes, C.A.; Pinheiro, A.C.M. SensoMaker Software Version 1.8; Universidade Federal de Lavras: Lavras, Brazil, 2002. [Google Scholar]
Sample Availability: Samples of the compounds are not available from the authors. |
 ), fructose (
), fructose (  ), and citric acid (
), and citric acid (  ) during fermentation of PH15 NI (A) and PH15 I (B).
) during fermentation of PH15 NI (A) and PH15 I (B).
 ), fructose (
), fructose (  ), and citric acid (
), and citric acid (  ) during fermentation of PH15 NI (A) and PH15 I (B).
) during fermentation of PH15 NI (A) and PH15 I (B).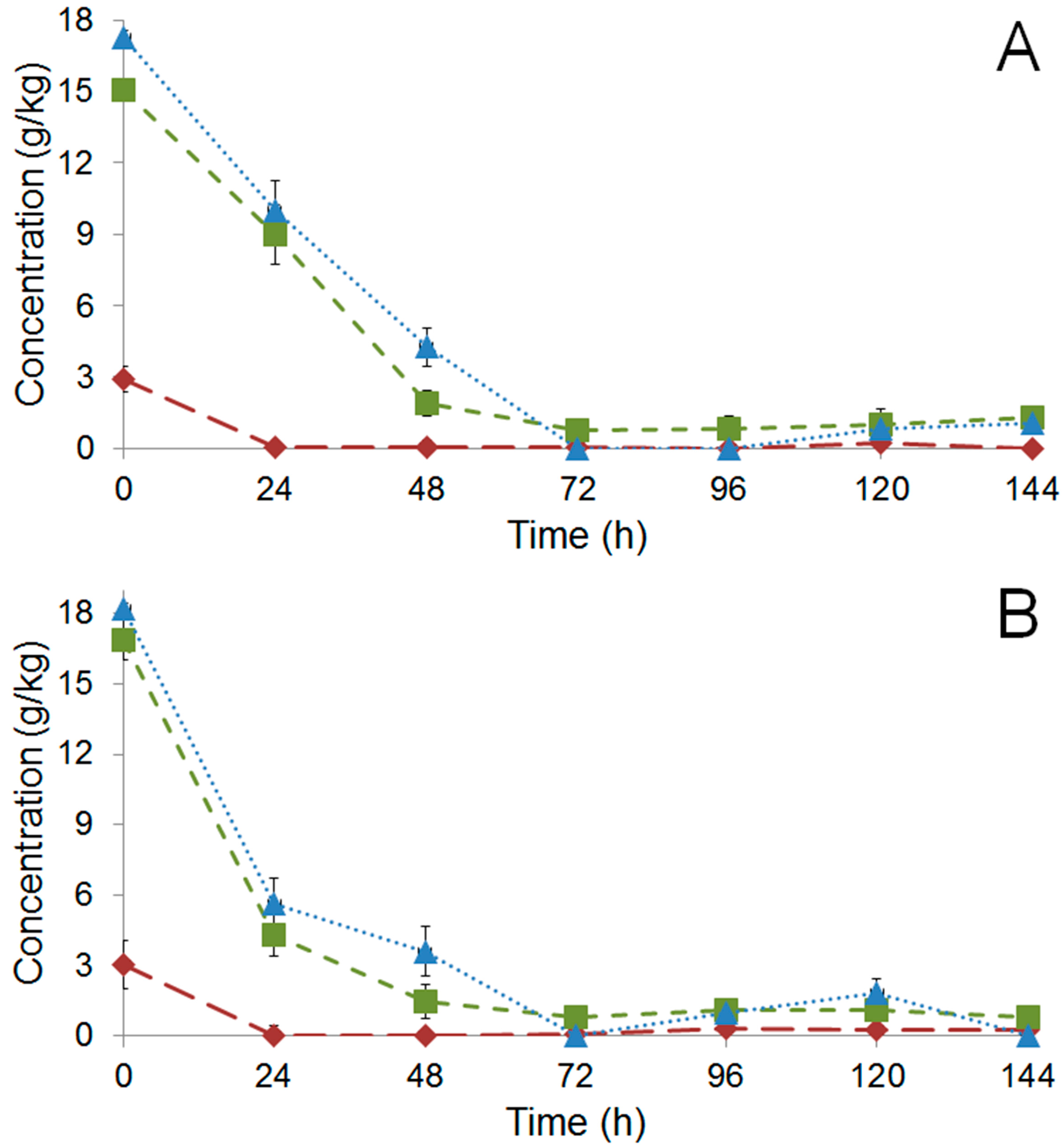
 ) and PH15 I (
) and PH15 I (  ); full symbols correspond to metabolites detected in the pulp while open symbols to those detected inside the beans.
); full symbols correspond to metabolites detected in the pulp while open symbols to those detected inside the beans.
 ) and PH15 I (
) and PH15 I (  ); full symbols correspond to metabolites detected in the pulp while open symbols to those detected inside the beans.
); full symbols correspond to metabolites detected in the pulp while open symbols to those detected inside the beans.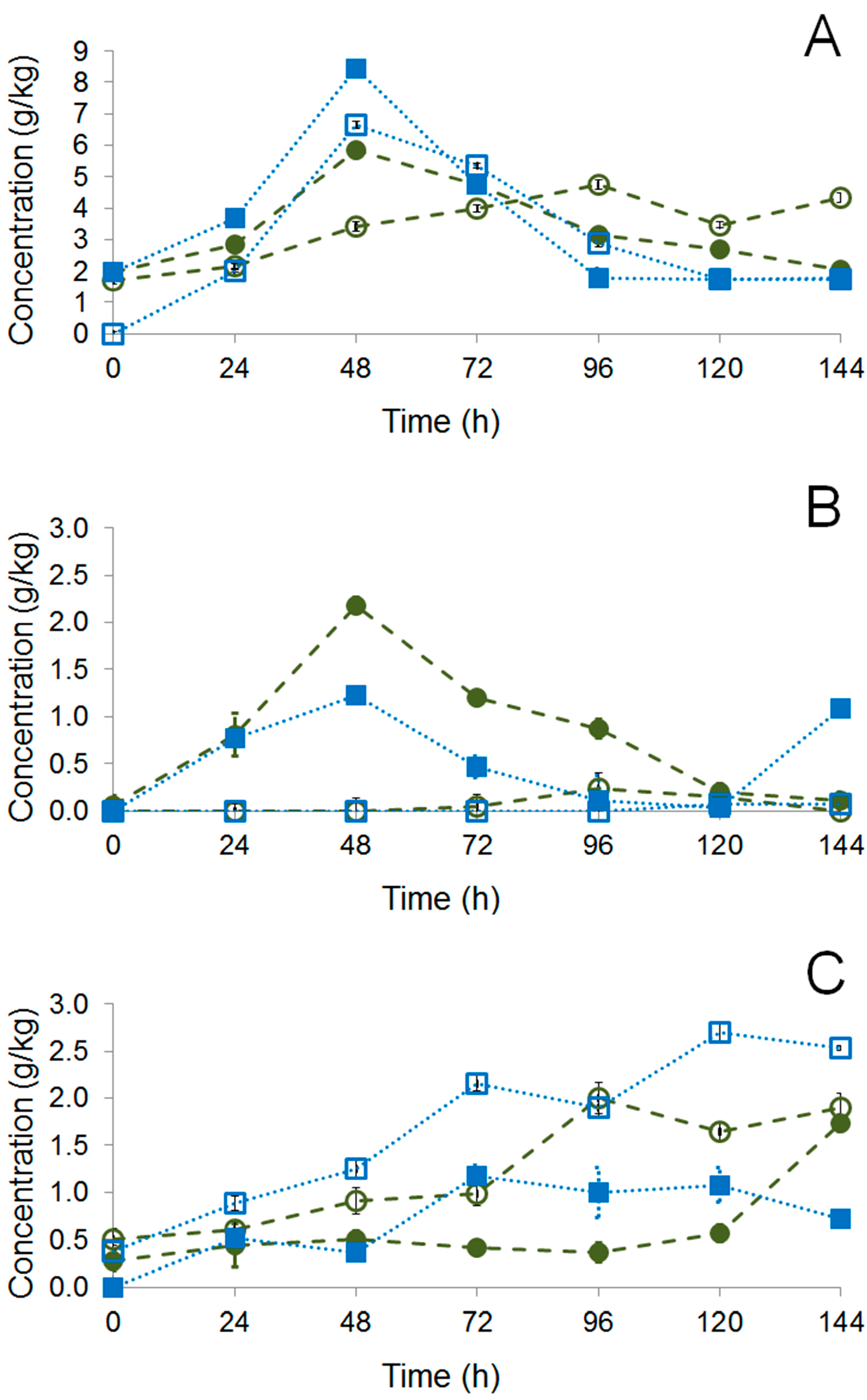
 PH15 NI), inoculated fermentation (
PH15 NI), inoculated fermentation (  PH15 I), and in the chocolate samples. Fermentation times: 0 h (A) and 144 h (B). Chocolate samples (C). Total amount of compounds: PH15 SI 0 h (37), PH15 SI 144 h (38), PH15 SI Ch (58), PH15 I 0 h (37), PH15 I 144 h (34), and PH15 I Ch (54).
PH15 I), and in the chocolate samples. Fermentation times: 0 h (A) and 144 h (B). Chocolate samples (C). Total amount of compounds: PH15 SI 0 h (37), PH15 SI 144 h (38), PH15 SI Ch (58), PH15 I 0 h (37), PH15 I 144 h (34), and PH15 I Ch (54).
 PH15 NI), inoculated fermentation (
PH15 NI), inoculated fermentation (  PH15 I), and in the chocolate samples. Fermentation times: 0 h (A) and 144 h (B). Chocolate samples (C). Total amount of compounds: PH15 SI 0 h (37), PH15 SI 144 h (38), PH15 SI Ch (58), PH15 I 0 h (37), PH15 I 144 h (34), and PH15 I Ch (54).
PH15 I), and in the chocolate samples. Fermentation times: 0 h (A) and 144 h (B). Chocolate samples (C). Total amount of compounds: PH15 SI 0 h (37), PH15 SI 144 h (38), PH15 SI Ch (58), PH15 I 0 h (37), PH15 I 144 h (34), and PH15 I Ch (54).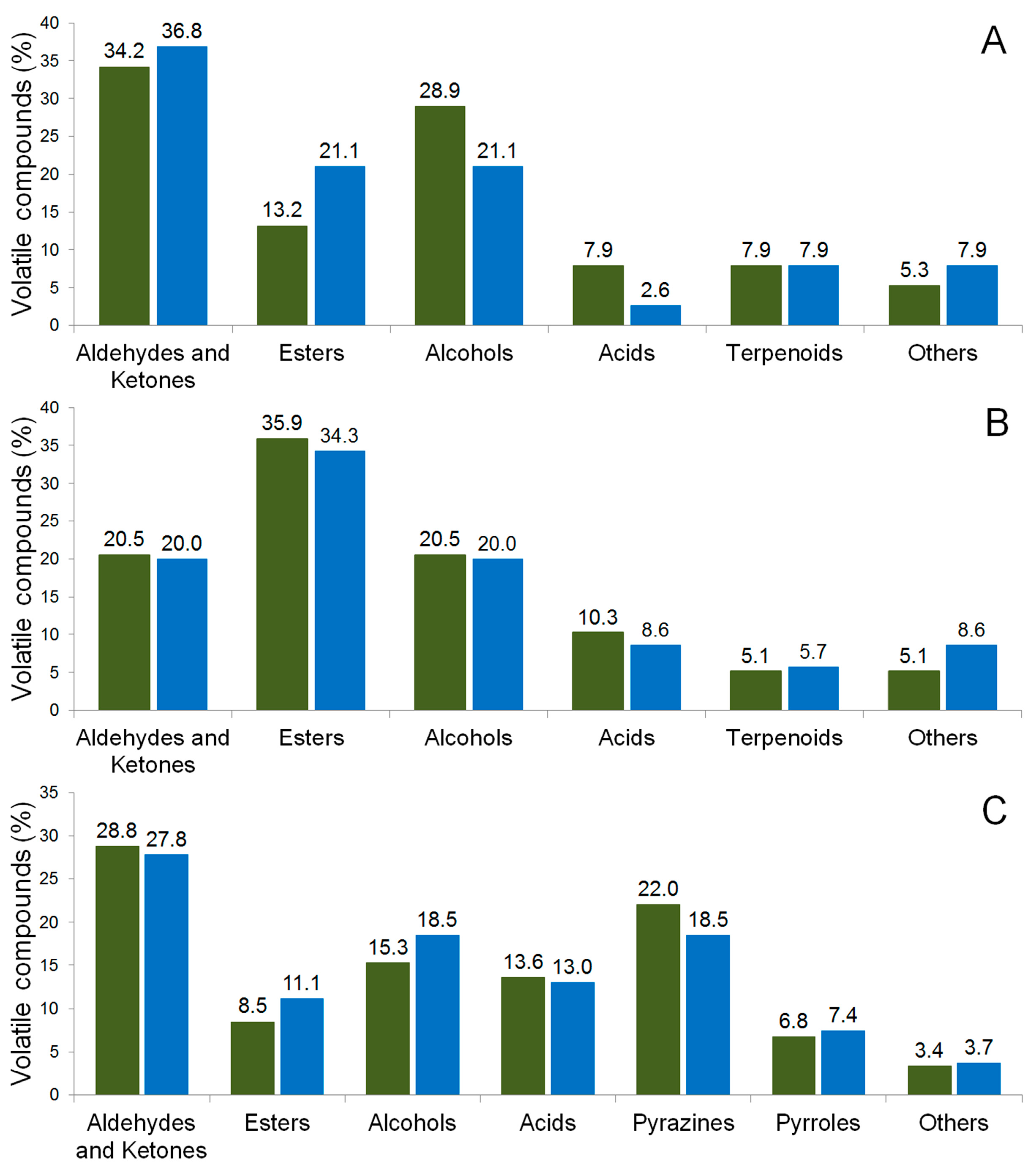
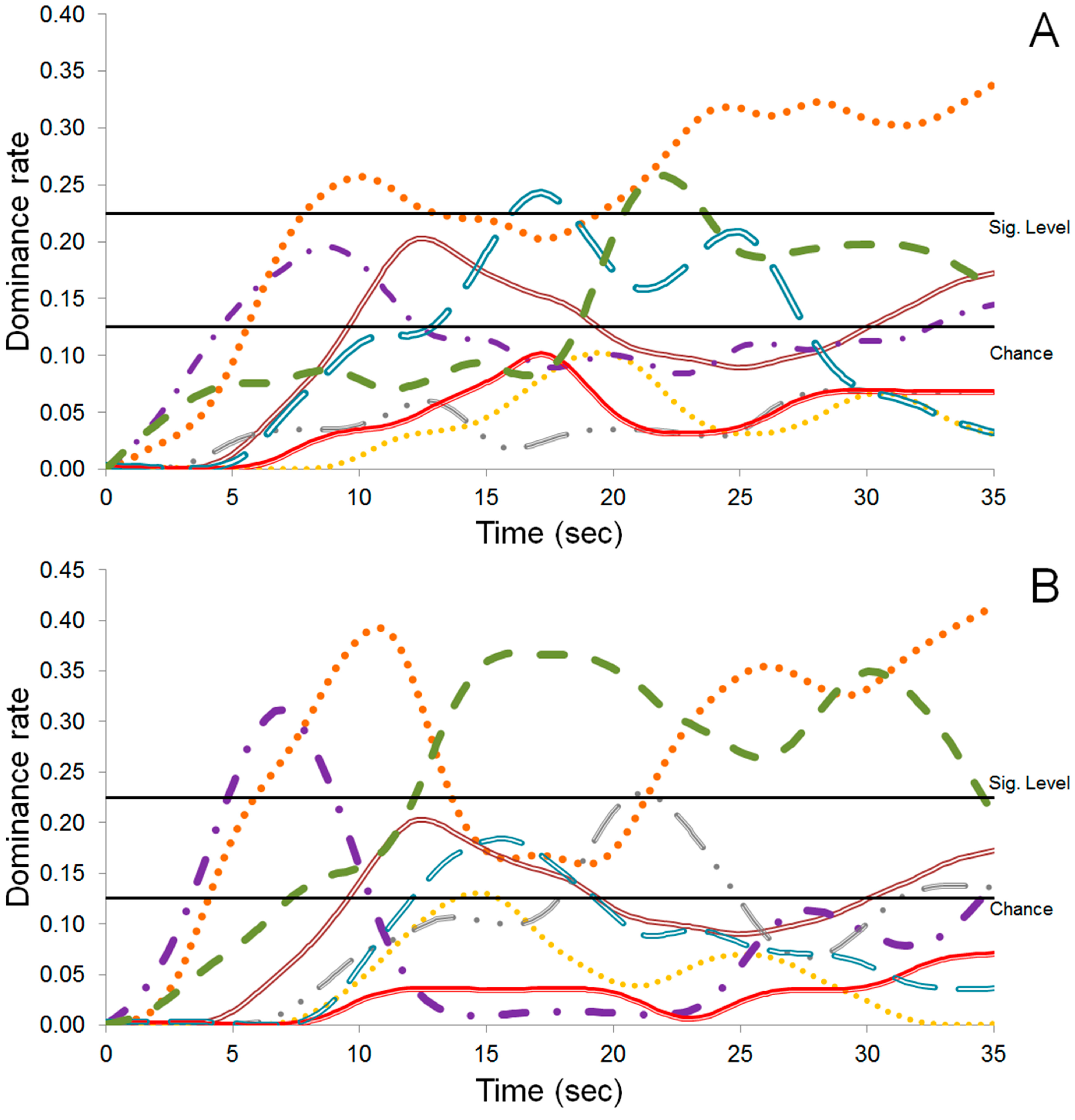
 ), Bitterness (
), Bitterness (  ), Nutty (
), Nutty (  ), Sweetness (
), Sweetness (  ), Astringent (
), Astringent (  ), Coffee (
), Coffee (  ), Fruity (
), Fruity (  ), and Cocoa (
), and Cocoa (  ).
).
 ), Bitterness (
), Bitterness (  ), Nutty (
), Nutty (  ), Sweetness (
), Sweetness (  ), Astringent (
), Astringent (  ), Coffee (
), Coffee (  ), Fruity (
), Fruity (  ), and Cocoa (
), and Cocoa (  ).
).
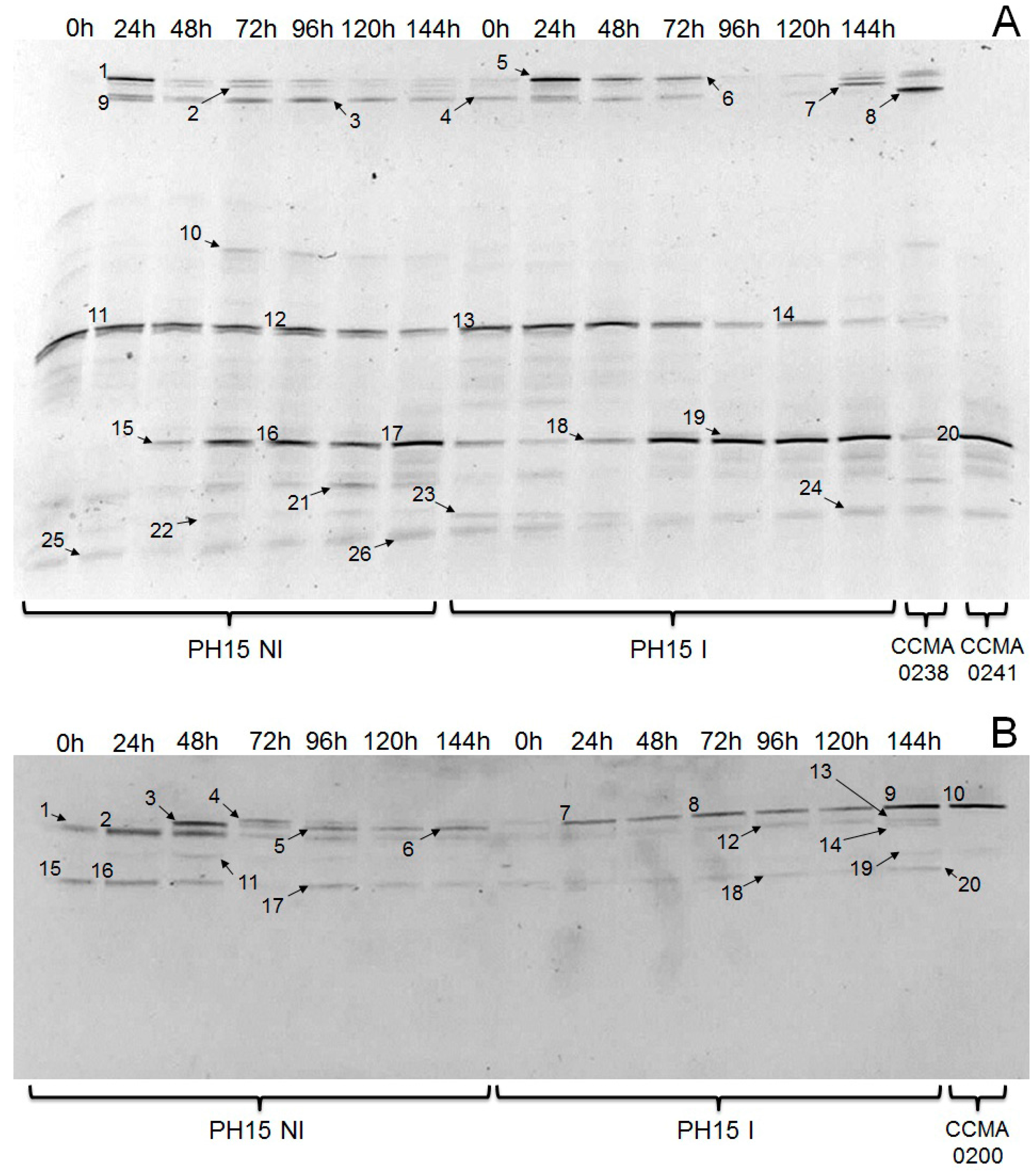
| Identification | Bands a | Similarity (%) | Accession Number | Sample Found |
|---|---|---|---|---|
| Prokaryotes | ||||
| Fructobacillus pseudoficulneus | 1, 5, 6 | 98 | AB498052.1 | PH15NI, PH15I |
| Leuconostoc sp. | 2 | 97 | DQ523491.1 | PH15NI |
| Lactobacillus plantarum | 3, 4, 8, 9 | 99 | KT327866.1 | PH15NI, PH15I |
| Lactobacillus helveticus | 7 | 99 | KP764179.1 | PH15I |
| Zymomonas mobilis | 10 | 100 | CP003715.1 | PH15NI |
| Uncultured bacterium | 11, 12, 13, 14 | 99 | LN875309.1 | PH15NI, PH15I |
| Acetobacter pasteurianus | 15, 16, 17, 18, 19, 20 | 100 | KM983001.1 | PH15NI, PH15I |
| Acetobacter sp. | 21 | 98 | KC796695.1 | PH15NI |
| Gluconobacter oxydans | 22, 23, 24 | 99 | CP003926 | PH15NI, PH15I |
| Bacillus sp. | 25, 26 | 99 | JF309224 | PH15NI |
| Eukaryotes | ||||
| Hanseniaspora uvarum | 1, 2 | 99 | KC544511 | PH15NI |
| Saccharomyces cerevisiae | 3, 5, 6, 7, 8, 9, 10 | 99 | KT229544.1 | PH15NI, PH15I |
| Saccharomyces sp. | 4 | 98 | KU350335.1 | PH15NI |
| Theobroma cacao | 11 | 97 | JQ228377.1 | PH15NI |
| Rhodotorula mucilaginosa | 12, 13 | 100 | HM588765 | PH15I |
| Lentinula edodes | 14 | 98 | KM015456.1 | PH15I |
| Pichia kluyveri | 15, 16, 17, 18, 20 | 99 | FM864201 | PH15NI, PH15I |
| Trichosporon asahii | 19 | 97 | JQ425402 | PH15I |
| Compounds | Odor Description a | Sample Found |
|---|---|---|
| Acids | ||
| 4-Hydroxybutanoic acid | PH15I Ch | |
| 4-Hydroxybutyric acid | PH15NI Ch | |
| Acetic acid | Sour, astringent | PH15NI 144 h |
| Benzeneacetic acid | PH15NI Ch, PH15I Ch | |
| Butanoic acid | Rancid, butter, cheese | PH15NI Ch, PH15I Ch |
| Hexanoic acid | Sweat, pungent | All |
| Isovaleric acid | Sweat, rancid | All |
| Octanoic acid | Sweaty, fatty | PH15NI (0 h and 144 h), PH15I 144 h, PH15NI Ch |
| Pentanoic acid | PH15NI Ch, PH15I Ch | |
| Valeric acid | Sweat, acid, rancid | PH15NI Ch, PH15I Ch |
| Alcohols | ||
| 2-Ethyl-1-hexanol | PH15NI Ch | |
| 1-Hexanol | Fruity, green | PH15NI (0 h and 144 h), PH15I 0 h |
| 1-Methoxy-2-butanol | PH15NI Ch, PH15I Ch | |
| 1-Nonanol | PH15NI Ch, PH15I Ch | |
| 1-Octanol | Fatty, waxy | All |
| 1-Penten-3-ol | PH15NI 0 h | |
| 2,3-Butanediol | Cocoa butter | PH15NI (0 h and 144 h), PH15I 144 h, PH15I Ch |
| 2,4-Pentanediol | PH15NI 0 h, PH15I 0 h, PH15NI Ch, PH15I Ch | |
| 2-Furanmethanol | PH15NI Ch | |
| 2-Heptanol | Sweet, citrusy | PH15NI (0 h and 144 h), PH15I (0 h and 144 h) |
| 2-Hexanol | Fruity, green | PH15NI 0 h, PH15I 0 h |
| 2-Pentanol | Green, mild green | PH15NI Ch, PH15I Ch |
| 3-Methyl-1-butanol | Malty, chocolate | PH15NI (0 h and 144 h), PH15I (0 h and 144 h) |
| Benzyl alcohol | Sweet, flower | All |
| Furfuryl alcohol | PH15I Ch | |
| Guaiacol | Smoke, sweet | PH15NI 144 h, PH15I 144 h |
| Phenylethyl Alcohol | Honey, rose, caramel | All |
| Aldehydes and Ketones | ||
| 3-methylpentanal | PH15NI 0 h, PH15I 0 h, PH15NI Ch, PH15I Ch | |
| (E)-2-Heptenal | PH15NI 0 h, PH15I 0 h | |
| (E)-2-Nonenal | Tallowy green | PH15NI 0 h, PH15I 0 h |
| (E)-2-Octanal | Fatty, waxy | PH15NI 0 h, PH15I 0 h |
| (E)-2-Undecenal | PH15NI Ch, PH15I Ch | |
| (E,E)-2,4-heptadienal | PH15NI 0 h, PH15I 0 h, PH15NI Ch, PH15I Ch | |
| 1-(2-hydroxyphenyl)ethanone | PH15NI 0 h, PH15I 0 h | |
| 2(5H)-Furanone | Caramel-like | PH15NI Ch, PH15I Ch |
| 2-Heptanone | PH15NI (0 h and 144 h), PH15I (0 h and 144 h) | |
| 2-Hydroxyphenyl methyl ketone | PH15NI 144 h | |
| 2-Nonanone | PH15NI 144 h, PH15I (0 h and 144 h) | |
| 2-Phenyl-2-butenal | Sweet, roasted | PH15NI 144 h, PH15I 144 h, PH15NI Ch, PH15I Ch |
| 2-Undecenal | PH15NI (0 h and 144 h), PH15I (0 h and 144 h) | |
| 3-Methyl-1,2-cyclopentanedione | PH15NI Ch, PH15I Ch | |
| 3-Penten-2-one | PH15NI Ch | |
| 4-hydroxy-3-methylbutanal | PH15NI 0 h, PH15I 0 h | |
| 4-Methylhexanal | PH15NI Ch, PH15I Ch | |
| 5-Methyl-2-furaldehyde | PH15NI Ch, PH15I Ch | |
| 5-Methyl-2-phenyl-2-hexenal | PH15NI Ch, PH15I Ch | |
| 3-methylpentanal | PH15NI 0 h, PH15I 0 h, PH15NI Ch, PH15I Ch | |
| Acetophenone | Floral | PH15NI (0 h and 144 h), PH15I (0 h and 144 h) |
| Benzaldehyde | Bitter | All |
| Benzeneacetaldehyde | PH15NI Ch, PH15I Ch | |
| Nonanal | PH15NI 0 h, PH15I 0 h, PH15NI Ch, PH15I Ch | |
| Octanal | PH15NI 0 h, PH15I 0 h, PH15NI Ch, PH15I Ch | |
| Pyranone | PH15NI Ch, PH15I Ch | |
| Acetoin | Butter, cream | PH15NI 144 h, PH15I 144 h, PH15NI Ch, PH15I Ch |
| Esters | ||
| 1-methylbutyl benzoate | PH15NI 144 h, PH15I 144 h | |
| 1-Methylhexyl acetate | PH15NI 144 h | |
| 2-Ethyl-1-hexyl acetate | PH15I Ch | |
| 2-Pentanyl benzoate | PH15NI 0 h, PH15I 0 h, PH15NI Ch, PH15I Ch | |
| 2-Phenethyl acetate | Fruity | PH15NI 144 h, PH15I 144 h |
| 3-methylbutyl formate | PH15NI 0 h | |
| Amyl acetate | Fruity, banana | PH15NI 144 h, PH15I 144 h |
| Dibutyl phthalate | PH15NI 144 h, PH15I (0 h and 144 h) | |
| Diisobutyl phthalate | PH15NI Ch, PH15I Ch | |
| Ethyl 2-hydroxypropanoate | PH15NI 144 h, PH15I 144 h | |
| Ethyl benzeneacetate | PH15NI 144 h | |
| Ethyl caprate | Pear, grape | PH15NI 144 h, PH15I 144 h |
| Ethyl caprylate | Fruity, flowery | PH15I (0 h and 144 h), PH15I Ch |
| Ethyl laurate | Fruity, floral | PH15NI 144 h, PH15I 144 h, PH15NI Ch, PH15I Ch |
| Ethyl myristate | Waxy, soapy | PH15NI 144 h, PH15I 144 h |
| Ethyl palmitate | Waxy, green | PH15NI (0 h and 144 h), PH15I (0 h and 144 h) |
| Ethyl phenylacetate | PH15NI 0 h | |
| Hexyl acetate | PH15NI 144 h | |
| Isoamylformate | PH15I 0 h | |
| Isobutyl phthalate | PH15NI (0 h and 144 h), PH15I (0 h and 144 h) | |
| Isopropyl palmitate | PH15I 0 h | |
| Methyl Palmitate | PH15NI 144 h, PH15I (0 h and 144 h) | |
| Phenylethyl acetate | fruity, sweet | PH15NI Ch, PH15I Ch |
| Pyrazines | ||
| 2,3,5,6-Tetramethylpyrazine | Chocolate, coffee | PH15NI Ch, PH15I Ch |
| 2,3,5-Trimethyl-6-isopentylpyrazine | PH15NI Ch, PH15I Ch | |
| 2,3,5-Trimethylpyrazine | Cocoa, rusted nuts | PH15NI Ch, PH15I Ch |
| 2,3-Dimethylpyrazine | Caramel, cocoa | PH15I Ch |
| 2,5-Dimethyl-3-isoamylpyrazine | PH15NI Ch, PH15I Ch | |
| 2,5-Dimethylpyrazine | Cocoa, rusted nuts | PH15NI Ch, PH15I Ch |
| 2,6-Dimethylpyrazine | Nutty, coffee, green | PH15I Ch |
| 2-Acetyl-3,5-dimethylpyrazine | PH15NI Ch, PH15I Ch | |
| 2-Ethyl-3,6-dimethylpyrazine | Roasted, smoky | PH15NI Ch, PH15I Ch |
| 2-Ethyl-6-methylpyrazine | PH15NI Ch, PH15I Ch | |
| 2-Ethylpyrazine | Peanut butter, nutty | PH15NI Ch |
| 2-Methyl-3,5-diethylpyrazine | PH15NI Ch | |
| 2-Methyl-6-vinylpyrazine | PH15NI Ch | |
| 2-Methylpyrazine | Chocolate, cocoa, nuts | PH15NI Ch, PH15I Ch |
| Pyrroles | ||
| 1,3-Dimethyl-5-pyrazolinone | PH15NI Ch, PH15I Ch | |
| 2-Acetylpyrrole | Chocolate, hazelnut | PH15NI Ch, PH15I Ch |
| 2-Pyrrolidinone | PH15NI Ch, PH15I Ch | |
| Pyrrole-2-carboxaldehyde | PH15NI Ch, PH15I Ch | |
| Terpenoids | ||
| (E)-Linalool oxide | Floral, green | PH15NI (0 h and 144 h), PH15I (0 h and 144 h) |
| (Z)-Linalool oxide | Floral | PH15NI 0 h, PH15I 0 h |
| Linalool | Flower, lavender | PH15NI (0 h and 144 h), PH15I (0 h and 144 h) |
| Others b | ||
| 1-methoxy-2-methylpropane | PH15NI (0 h and 144 h), PH15I 144 h | |
| 2-Butyltetrahydrofuran | PH15NI Ch, PH15I Ch | |
| 2-Pentylfuran | All | |
| 7-methyl pentadecane | PH15I (0 h and 144 h) | |
| Hexadecane | PH15I (0 h and 144 h) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Magalhães da Veiga Moreira, I.; De Figueiredo Vilela, L.; Da Cruz Pedroso Miguel, M.G.; Santos, C.; Lima, N.; Freitas Schwan, R. Impact of a Microbial Cocktail Used as a Starter Culture on Cocoa Fermentation and Chocolate Flavor. Molecules 2017, 22, 766. https://doi.org/10.3390/molecules22050766
Magalhães da Veiga Moreira I, De Figueiredo Vilela L, Da Cruz Pedroso Miguel MG, Santos C, Lima N, Freitas Schwan R. Impact of a Microbial Cocktail Used as a Starter Culture on Cocoa Fermentation and Chocolate Flavor. Molecules. 2017; 22(5):766. https://doi.org/10.3390/molecules22050766
Chicago/Turabian StyleMagalhães da Veiga Moreira, Igor, Leonardo De Figueiredo Vilela, Maria Gabriela Da Cruz Pedroso Miguel, Cledir Santos, Nelson Lima, and Rosane Freitas Schwan. 2017. "Impact of a Microbial Cocktail Used as a Starter Culture on Cocoa Fermentation and Chocolate Flavor" Molecules 22, no. 5: 766. https://doi.org/10.3390/molecules22050766
APA StyleMagalhães da Veiga Moreira, I., De Figueiredo Vilela, L., Da Cruz Pedroso Miguel, M. G., Santos, C., Lima, N., & Freitas Schwan, R. (2017). Impact of a Microbial Cocktail Used as a Starter Culture on Cocoa Fermentation and Chocolate Flavor. Molecules, 22(5), 766. https://doi.org/10.3390/molecules22050766








