Gold Nanoparticles Deposited on Surface Modified Carbon Xerogels as Reusable Catalysts for Cyclohexane C-H Activation in the Presence of CO and Water
Abstract
:1. Introduction
2. Results and Discussion
2.1. Characterisation of Xerogel Supports
2.2. Characterisation of Gold Catalysts
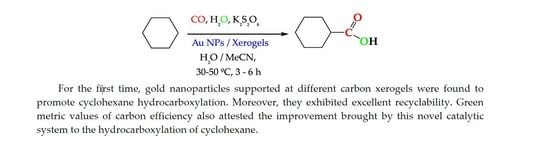
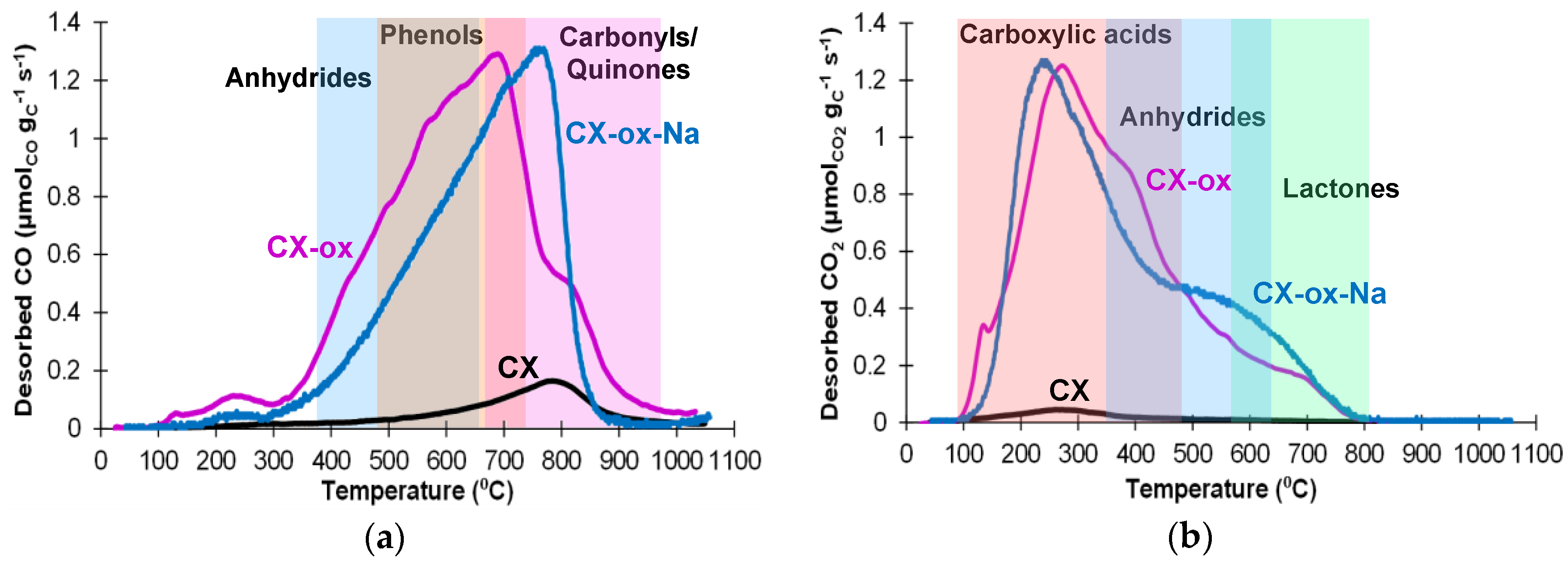
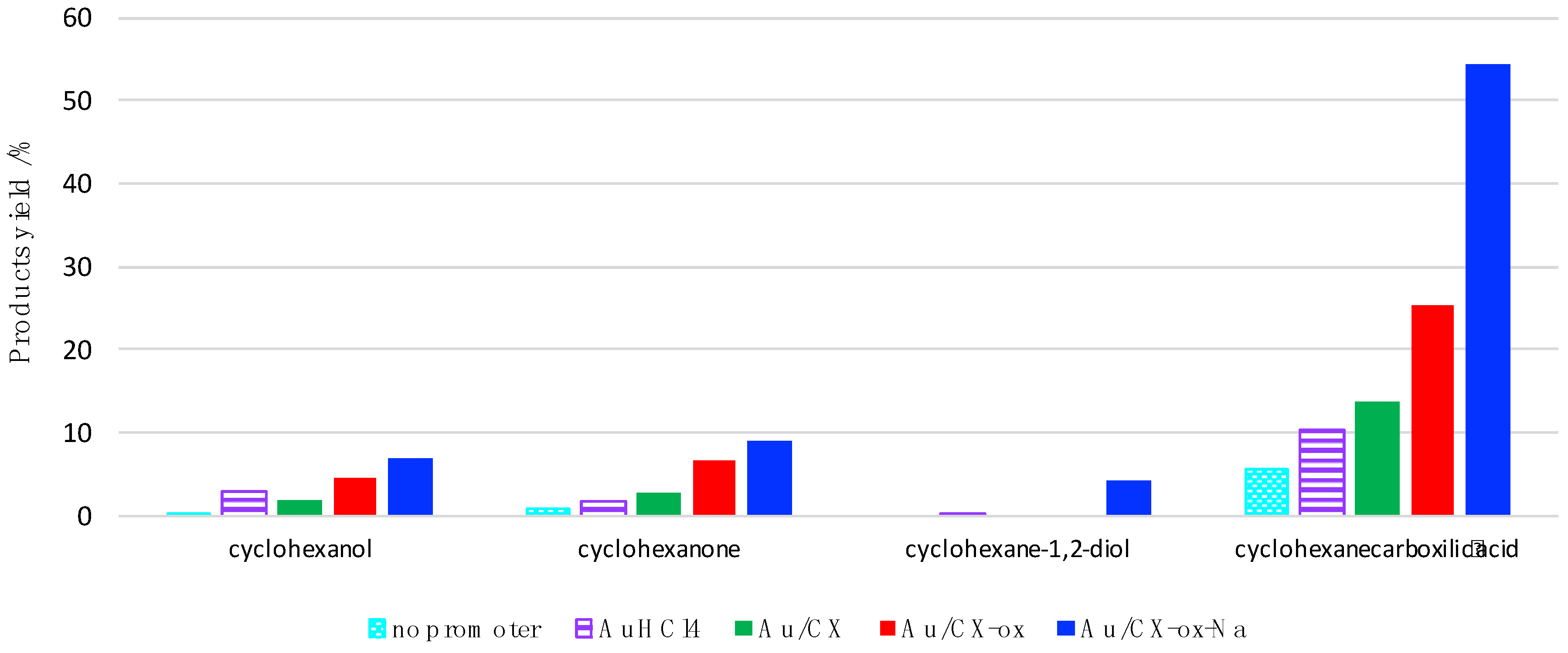
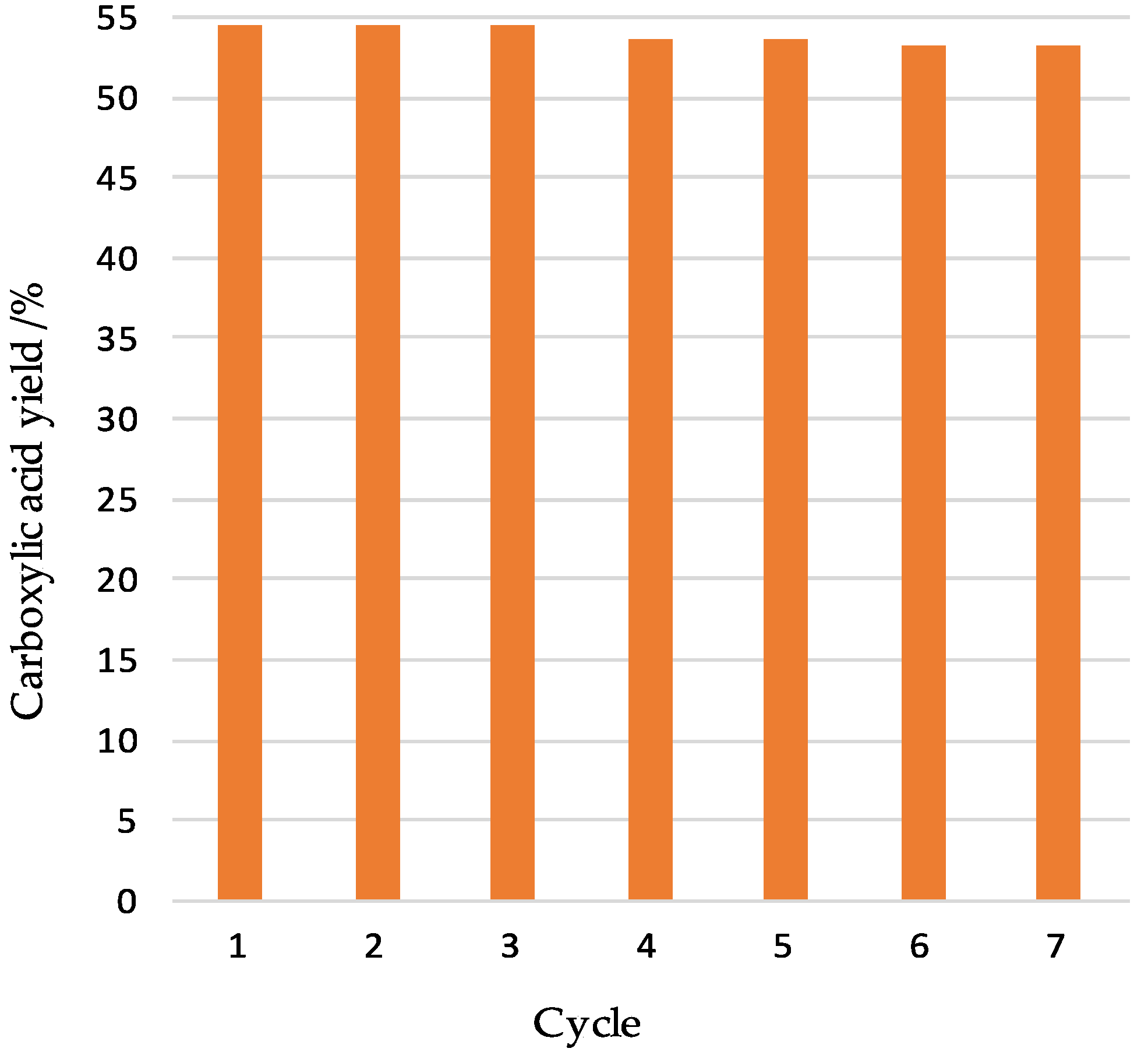
2.3. Catalytic Results
3. Materials and Methods
3.1. Reagents
3.2. Carbon Materials Preparation
3.3. Carbon Materials Characterisation
3.4. Gold Loading
3.5. Gold Catalysts Characterisation
3.6. Catalytic Tests
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Nishiguchi, T.; Nakata, K.; Takaki, K.; Fujiwara, Y. Transition Metal Catalyzed Acetic Acid Synthesis from Methane and CO. Chem. Lett. 1992, 21, 1141–1142. [Google Scholar] [CrossRef]
- Nakata, K.; Yamaoka, Y.; Miyata, T.; Taniguchi, Y.; Takaki, K.; Fujiwara, Y. Palladium(II) and/or copper(II)-catalyzed carboxylation of small alkanes such as methane and ethane with carbon monoxide. J. Organomet. Chem. 1994, 473, 329–334. [Google Scholar] [CrossRef]
- Derouane, E.D.; Haber, J.; Lemos, F.; Ramôa Ribeiro, F.; Guinet, M.E. Catalytic Activation and Functionalisation of Light Alkanes; NATO ASI Series; Kluwer Academic Publ: Dordrecht, The Netherlands, 1998; Volume 44. [Google Scholar]
- Jia, C.; Kitamura, T.; Fujiwara, Y. Catalytic Functionalization of Arenes and Alkanes via C-H Bond Activation. Acc. Chem. Res. 2001, 34, 633–639. [Google Scholar] [CrossRef] [PubMed]
- Pombeiro, A.J.L.; Fraústo da Silva, J.J.R.; Fujiwara, Y.; Silva, J.A.L.; Reis, P.M.; Palavra, A.F. Catalysts and Process for Direct Conversion of Methane into Acetic Acid. WO 2004/037416 A3, 6 May 2004. [Google Scholar]
- Reis, P.M.; Silva, J.A.L.; Palavra, A.F.; Fraústo da Silva, J.J.R.; Kitamura, T.; Fujiwara, Y.; Pombeiro, A.J.L. Single-Pot Conversion of Methane into Acetic Acid in the Absence of CO and with Vanadium Catalysts Such as Amavadine. Angew. Chem. Int. Ed. 2003, 42, 821–823. [Google Scholar] [CrossRef] [PubMed]
- Reis, P.M.; Silva, J.A.L.; Palavra, A.F.; Fraústo da Silva, J.J.R.; Pombeiro, A.J.L. Vanadium-catalyzed carboxylation of linear and cyclic C5 and C6 alkanes. J. Catal. 2005, 235, 333–340. [Google Scholar] [CrossRef]
- Kirillova, M.V.; Kuznetsov, M.L.; Reis, P.M.; da Silva, J.A.L.; Fraústo da Silva, J.J.R.; Pombeiro, A.J.L. Direct and Remarkably Efficient Conversion of Methane into Acetic Acid Catalyzed by Amavadine and Related Vanadium Complexes. A Synthetic and a Theoretical DFT Mechanistic Study. J. Am. Chem. Soc. 2007, 129, 10531–10545. [Google Scholar] [CrossRef] [PubMed]
- Kirillova, M.V.; Kuznetsov, M.L.; da Silva, J.A.L.; Guedes da Silva, M.F.C.; Fraústo da Silva, J.J.R.; Pombeiro, A.J.L. Amavadin and Other Vanadium Complexes as Remarkably Efficient Catalysts for One-Pot Conversion of Ethane to Propionic and Acetic Acids. Chem. Eur. J. 2008, 14, 1828–1842. [Google Scholar] [CrossRef] [PubMed]
- Kirillova, M.V.; Kirillov, A.M.; Kuznetsov, M.L.; Silva, J.A.L.; Frausto da Silva, J.J.R.; Pombeiro, A.J.L. Alkanes to carboxylic acids in aqueous medium: Metal-free and metal-promoted highly efficient and mild conversions. Chem. Commun. 2009, 17, 2353–2355. [Google Scholar] [CrossRef] [PubMed]
- Kirillova, M.V.; Kirillov, A.M.; Pombeiro, A.J.L. Mild, Single-Pot Hydrocarboxylation of Gaseous Alkanes to Carboxylic Acids in Metal-Free and Copper-Promoted Aqueous Systems. Chem. Eur. J. 2010, 16, 9485–9493. [Google Scholar] [CrossRef] [PubMed]
- Silva, T.F.S.; Luzyanin, K.V.; Kirillova, M.V.; da Silva, M.F.G.; Martins, L.M.D.R.S.; Pombeiro, A.J.L. Novel Scorpionate and Pyrazole Dioxovanadium Complexes, Catalysts for Carboxylation and Peroxidative Oxidation of Alkanes. Adv. Synth. Catal. 2010, 352, 171–187. [Google Scholar] [CrossRef]
- Phan, A.; Czaja, A.U.; Gándara, F.; Knobler, C.B.; Yaghi, O.M. Metal–Organic Frameworks of Vanadium as Catalysts for Conversion of Methane to Acetic Acid. Inorg. Chem. 2011, 50, 7388–7390. [Google Scholar] [CrossRef] [PubMed]
- Martins, L.M.D.R.S.; Pombeiro, A.J.L. Tris(pyrazol-1-yl)methane metal complexes for catalytic mild oxidative functionalizations of alkanes, alkenes and ketones. Coord. Chem. Rev. 2014, 265, 74–88. [Google Scholar] [CrossRef]
- Pombeiro, A.J.L. Toward Functionalization of Alkanes under Environmentally Benign Conditions. In Advances in Organometallic Chemistry and Catalysis, The Silver/Gold Jubilee ICOMC Celebratory Book; Pombeiro, A.J.L., Ed.; J. Wiley & Sons: New York, NY, USA, 2014; pp. 15–25. [Google Scholar]
- Martins, L.M.D.R.S.; Pombeiro, A.J.L. Water-Soluble C-Scorpionate Complexes—Catalytic and Biological Applications. Eur. J. Inorg. Chem. 2016, 2016, 2236–2252. [Google Scholar] [CrossRef]
- Shul’pin, G. New Trends in Oxidative Functionalization of Carbon-Hydrogen Bonds: A Review. Catalysts 2016, 6, 50. [Google Scholar] [CrossRef]
- Weissermel, W.; Horpe, H.J. Industrial Organic Chemistry, 2nd ed.; VCH Press: Weinheim, Germany, 1993. [Google Scholar]
- Seidel, A.; Bickford, M. Encyclopedia of Chemical Technology, 5th ed.; Wiley: New York, NY, USA, 2004. [Google Scholar]
- Fritz, U. Ullmann’s Encyclopedia of Industrial Chemistry, 6th ed.; Wiley-VCH: Weinheim, Germany, 2002. [Google Scholar]
- Seidel, A.B.M. (Ed.) Kirk-Othmer Encyclopedia of Chemical Technology; J. Wiley & Sons: New York, NY, USA, 2014. [Google Scholar]
- Kirillova, M.V.; Kirillov, A.M.; Pombeiro, A.J.L. Metal-Free and Copper-Promoted Single-Pot Hydrocarboxylation of Cycloalkanes to Carboxylic Acids in Aqueous Medium. Adv. Synth. Catal. 2009, 351, 2936–2948. [Google Scholar] [CrossRef]
- Hutchings, G.J.; Haruta, M. A golden age of catalysis: A perspective. Appl. Catal. A Gen. 2005, 291, 2–5. [Google Scholar] [CrossRef]
- Bond, G.C.; Louis, C.; Thompson, D.T. Catalysis by Gold; Imperial College Press: London, UK, 2006; Volume 6. [Google Scholar]
- Carrettin, S.; Blanco, M.C.; Corma, A.; Hashmi, A.S.K. Heterogeneous Gold-Catalysed Synthesis of Phenols. Adv. Synth. Catal. 2006, 348, 1283–1288. [Google Scholar] [CrossRef]
- Carabineiro, S.A.C.; Thompson, D. Catalytic Applications for Gold Nanotechnology. In Nanocatalysis; Heiz, U., Landman, U., Eds.; Springer: Berlin/Heidelberg, Germany, 2007; pp. 377–489. [Google Scholar]
- Carabineiro, S.A.C.; Thompson, D. Gold Catalysis. In Gold: Science and Applications; Corti, C., Holliday, R., Eds.; CRC Press, Taylor & Francis Group: Boca Raton, FL, USA; London, UK; New York, NY, USA, 2010; pp. 89–122. [Google Scholar]
- Wu, P.; Loh, K.P.; Zhao, X.S. Supported Gold Catalysts for Selective Oxidation of Organics. Sci. Adv. Mater. 2011, 3, 970–983. [Google Scholar] [CrossRef]
- De Almeida, M.P.; Martins, L.M.D.R.S.; Carabineiro, S.A.C.; Lauterbach, T.; Rominger, F.; Hashmi, A.S.K.; Pombeiro, A.J.L.; Figueiredo, J.L. Homogeneous and heterogenised new gold C-scorpionate complexes as catalysts for cyclohexane oxidation. Catal. Sci. Technol. 2013, 3, 3056–3069. [Google Scholar] [CrossRef]
- Carabineiro, S.A.C.; Martins, L.M.D.R.S.; Avalos-Borja, M.; Buijnsters, J.G.; Pombeiro, A.J.L.; Figueiredo, J.L. Gold nanoparticles supported on carbon materials for cyclohexane oxidation with hydrogen peroxide. Appl. Catal. A Gen. 2013, 467, 279–290. [Google Scholar] [CrossRef]
- Patil, N.T.; Konala, A. Mechanistic Dichotomy with Alkynes in the Formal Hydrohydrazination/Fischer Indolization Tandem Reaction Catalyzed by a Ph3PAuNTf2/pTSA Binary System. Eur. J. Org. Chem. 2010, 2010, 6831–6839. [Google Scholar] [CrossRef]
- Dupuy, S.; Gasperini, D.; Nolan, S.P. Highly Efficient Gold(I)-Catalyzed Regio- and Stereoselective Hydrocarboxylation of Internal Alkynes. ACS Catal. 2015, 5, 6918–6921. [Google Scholar] [CrossRef]
- Protzmann, G.; Luft, G. A new immobilized Rh(I)-carbonylation catalyst. Appl. Catal. A Gen. 1998, 172, 159–163. [Google Scholar] [CrossRef]
- Carabineiro, S.A.C.; Thavorn-amornsri, T.; Pereira, M.F.R.; Serp, P.; Figueiredo, J.L. Comparison between activated carbon, carbon xerogel and carbon nanotubes for the adsorption of the antibiotic ciprofloxacin. Catal. Today 2012, 186, 29–34. [Google Scholar] [CrossRef]
- Martins, L.M.D.R.S.; Peixoto de Almeida, M.; Carabineiro, S.A.C.; Figueiredo, J.L.; Pombeiro, A.J.L. Heterogenisation of a C-Scorpionate Fe-II Complex on Carbon Materials for Cyclohexane Oxidation with Hydrogen Peroxide. ChemCatChem 2013, 5, 3847–3856. [Google Scholar] [CrossRef]
- Sutradhar, M.; Martins, L.M.D.R.S.; Carabineiro, S.A.C.; Guedes da Silva, M.F.C.; Buijnsters, J.G.; Figueiredo, J.L.; Pombeiro, A.J.L. Oxidovanadium(V) Complexes Anchored on Carbon Materials as Catalysts for the Oxidation of 1-Phenylethanol. ChemCatChem 2016, 8, 2254–2266. [Google Scholar] [CrossRef]
- Carabineiro, S.A.C.; Thavorn-Amornsri, T.; Pereira, M.F.R.; Figueiredo, J.L. Adsorption of ciprofloxacin on surface-modified carbon materials. Water Res. 2011, 45, 4583–4591. [Google Scholar] [CrossRef] [PubMed]
- Figueiredo, J.L.; Pereira, M.F.R.; Freitas, M.M.A.; Órfão, J.J.M. Modification of the surface chemistry of activated carbons. Carbon 1999, 37, 1379–1389. [Google Scholar] [CrossRef]
- Figueiredo, J.L.; Pereira, M.F.R.; Freitas, M.M.A.; Órfão, J.J.M. Characterization of Active Sites on Carbon Catalysts. Ind. Eng. Chem. Res. 2007, 46, 4110–4115. [Google Scholar] [CrossRef]
- Figueiredo, J.L.; Pereira, M.F.R. The role of surface chemistry in catalysis with carbons. Catal. Today 2010, 150, 2–7. [Google Scholar] [CrossRef]
- Carabineiro, S.A.C.; Pereira, M.F.R.; Órfão, J.J.M.; Figueiredo, J.L. Surface Chemistry of Activated Carbons. In Activated Carbon: Classifications, Properties and Applications; Kwiatkowski, J.F., Ed.; Nova Science Pub Inc.: New York, NY, USA, 2011; pp. 125–168. [Google Scholar]
- Maia, F.; Mahata, N.; Jarrais, B.; Silva, A.R.; Pereira, M.F.R.; Freire, C.; Figueiredo, J.L. Jacobsen catalyst anchored onto modified carbon xerogel as enantioselective heterogeneous catalyst for alkene epoxidation. J. Mol. Catal. A Chem. 2009, 305, 135–141. [Google Scholar] [CrossRef]
- Alegre, C.; Gálvez, M.E.; Baquedano, E.; Moliner, R.; Pastor, E.; Lázaro, M.J. Oxygen-Functionalized Highly Mesoporous Carbon Xerogel Based Catalysts for Direct Methanol Fuel Cell Anodes. J. Phys. Chem. C 2013, 117, 13045–13058. [Google Scholar] [CrossRef]
- Alegre, C.; Gálvez, M.E.; Baquedano, E.; Pastor, E.; Moliner, R.; Lázaro, M.J. Influence of support’s oxygen functionalization on the activity of Pt/carbon xerogels catalysts for methanol electro-oxidation. Int. J. Hydrogen Energy 2012, 37, 7180–7191. [Google Scholar] [CrossRef]
- Önal, Y.; Schimpf, S.; Claus, P. Structure sensitivity and kinetics of d-glucose oxidation to d-gluconic acid over carbon-supported gold catalysts. J. Catal. 2004, 223, 122–133. [Google Scholar] [CrossRef]
- Rodrigues, E.G.; Carabineiro, S.A.C.; Chen, X.; Delgado, J.J.; Figueiredo, J.L.; Pereira, M.F.R.; Órfão, J.J.M. Selective Oxidation of Glycerol Catalyzed by Rh/Activated Carbon: Importance of Support Surface Chemistry. Catal. Lett. 2011, 141, 420–431. [Google Scholar] [CrossRef]
- Rodrigues, E.G.; Carabineiro, S.A.C.; Delgado, J.J.; Chen, X.; Pereira, M.F.R.; Órfão, J.J.M. Gold supported on carbon nanotubes for the selective oxidation of glycerol. J. Catal. 2012, 285, 83–91. [Google Scholar] [CrossRef]
- Afrooz, A.R.M.N.; Sivalapalan, S.T.; Murphy, C.J.; Hussain, S.M.; Schlager, J.J.; Saleh, N.B. Spheres vs. rods: The shape of gold nanoparticles influences aggregation and deposition behavior. Chemosphere 2013, 91, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Xu, M. Metal Nanoparticles of Various Shapes. In Advantages of Metal Nanoparticles, ECE-580 Mid-Term Paper; 2007; Available online: http://www.slideserve.com/nero/metal-nanoparticles-of-various-shapes (accessed on 7 April 2017).
- Huang, J.; Wang, Y.; Zheng, J.; Dai, W.-L.; Fan, K. Influence of support surface basicity and gold particle size on catalytic activity of Au/γ-AlOOH and Au/γ-Al2O3 catalyst in aerobic oxidation of α,ω-diols to lactones. Appl. Catal. B Environ. 2011, 103, 343–350. [Google Scholar] [CrossRef]
- Nepak, D.; Srinivas, D. Effect of alkali and alkaline earth metal ions on benzyl alcohol oxidation activity of titanate nanotube-supported Au catalysts. RSC Adv. 2015, 5, 47740–47748. [Google Scholar] [CrossRef]
- Shigenobu, H.; Mitsuyuki, S.; Tamotsu, K.; Takaharu, O.; Kenzi, T. Catalytic Activity of Silver and Gold Metals Doped with Alkali Metals. Bull. Chem. Soc. Jpn. 1977, 50, 842–846. [Google Scholar]
Sample Availability: Samples of the compounds are not available from the authors. |
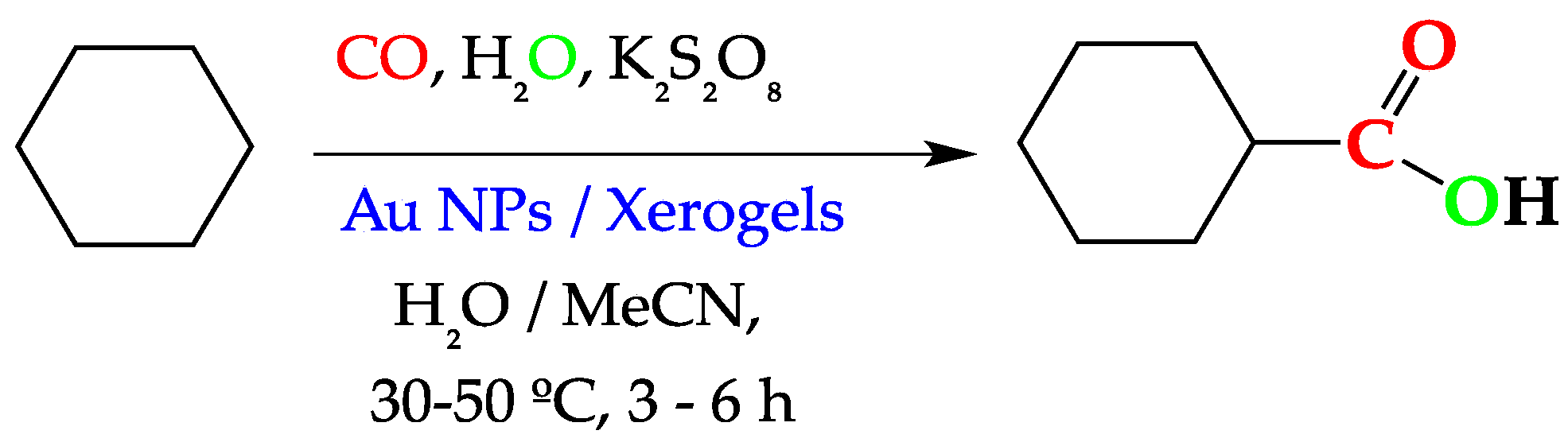

| Sample | SBET (m2/g) | Vp (cm3/g) | L (nm) | Vmicro (cm3/g) | Sexternal (m2/g) | CO (µmol/g) | CO2 (µmol/g) |
|---|---|---|---|---|---|---|---|
| CX | 604 | 0.91 | 13.7 | ~0 | 604 | 492 | 135 |
| CX-ox | 570 | 0.80 | 18.8 | 0.038 | 512 | 4609 | 3774 |
| CX-ox-Na | 560 | 0.75 | 17.6 | 0.036 | 496 | 3720 | 3793 |
| Sample | Au Average Size (nm) | Metal Dispersion (%) | Gold Loading (%) |
|---|---|---|---|
| Au/CX | 16.6 * | 6.9 * | 2.8 |
| Au/CX-ox | 14.2 | 8.1 | 1.4 |
| Au/CX-ox-Na | 13.7 | 8.4 | 0.5 |
| Entry | Au/μmol | P(CO)/atm | Temperature/°C | Total TON b | Yield/% c CyCOOH Cy-H=O CyOH Cy-H(OH)2 | |||
|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 2 | 50 | 375 | 54.5 | 9.1 | 7.1 | 4.3 |
| 2 | 2 | 20 | 50 | 311 | 28.3 | 19.4 | 14.5 | 3.9 |
| 3 | 2 | 2 | 30 | 111 | 12.0 | 5.3 | 4.9 | 0.8 |
| 4 | 2 | 2 | 80 | 245 | 24.1 | 14.4 | 9.5 | 0.9 |
| 5 | 20 | 2 | 50 | 23 | 19.9 | 12.3 | 12.5 | 0.5 |
| Entry | Metal Promoter | CyCOOH Yield (%) b/μmol of M Promotor | Carbon Efficiency/% c | Atom Economy/% d |
|---|---|---|---|---|
| 1 | HAuCl4·3H2O e | 5.2 | 7.7 | 33.5 |
| 2 | Au/CX e | 6.9 | 9.3 | |
| 3 | Au/CX-ox e | 12.7 | 18.4 | |
| 4 | Au/CX-ox-Na e | 27.3 | 37.9 | |
| 5 | Cr(OH)3·2.5H2O [10] | 0.4 | 0.6 | 33.5 |
| 6 | K2Cr2O7 [10] | 1.0 | 3.9 | |
| 7 | MoO3 [10] | 0.0 | 0.0 | |
| 8 | H4[PMo11VO40]·34H2O [10] | 0.4 | 1.1 | |
| 9 | MnO2 [10] | 0.5 | 1.4 | |
| 10 | Fe(OH)3·0.5H2O [10] | 1.0 | 1.6 | |
| 11 | Co(acac)3 [10] | 0.6 | 0.9 | |
| 12 | Zn(NO3)2 [10] | 0.5 | 0.8 | |
| 13 | Cu(NO3)2·2.5H2O [10] | 1.0 | 3.3 | |
| 14 | [Cu(H2tea)(N3)] [10] | 2.0 | 3.3 | |
| 15 | [Cu2(H2tea)2(tpa)]n·2nH2O [10] | 4.9 | 3.8 | |
| 16 | [OCu4{N(CH2CH2O)3}4(BOH)4][BF4]2 [10] | 18.1 | 7.1 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ribeiro, A.P.d.C.; Martins, L.M.D.R.d.S.; Carabineiro, S.A.C.; Figueiredo, J.L.; Pombeiro, A.J.L. Gold Nanoparticles Deposited on Surface Modified Carbon Xerogels as Reusable Catalysts for Cyclohexane C-H Activation in the Presence of CO and Water. Molecules 2017, 22, 603. https://doi.org/10.3390/molecules22040603
Ribeiro APdC, Martins LMDRdS, Carabineiro SAC, Figueiredo JL, Pombeiro AJL. Gold Nanoparticles Deposited on Surface Modified Carbon Xerogels as Reusable Catalysts for Cyclohexane C-H Activation in the Presence of CO and Water. Molecules. 2017; 22(4):603. https://doi.org/10.3390/molecules22040603
Chicago/Turabian StyleRibeiro, Ana Paula da Costa, Luísa Margarida Dias Ribeiro de Sousa Martins, Sónia Alexandra Correia Carabineiro, José Luís Figueiredo, and Armando José Latourrette Pombeiro. 2017. "Gold Nanoparticles Deposited on Surface Modified Carbon Xerogels as Reusable Catalysts for Cyclohexane C-H Activation in the Presence of CO and Water" Molecules 22, no. 4: 603. https://doi.org/10.3390/molecules22040603
APA StyleRibeiro, A. P. d. C., Martins, L. M. D. R. d. S., Carabineiro, S. A. C., Figueiredo, J. L., & Pombeiro, A. J. L. (2017). Gold Nanoparticles Deposited on Surface Modified Carbon Xerogels as Reusable Catalysts for Cyclohexane C-H Activation in the Presence of CO and Water. Molecules, 22(4), 603. https://doi.org/10.3390/molecules22040603