A Focused Multiple Reaction Monitoring (MRM) Quantitative Method for Bioactive Grapevine Stilbenes by Ultra-High-Performance Liquid Chromatography Coupled to Triple-Quadrupole Mass Spectrometry (UHPLC-QqQ)
Abstract
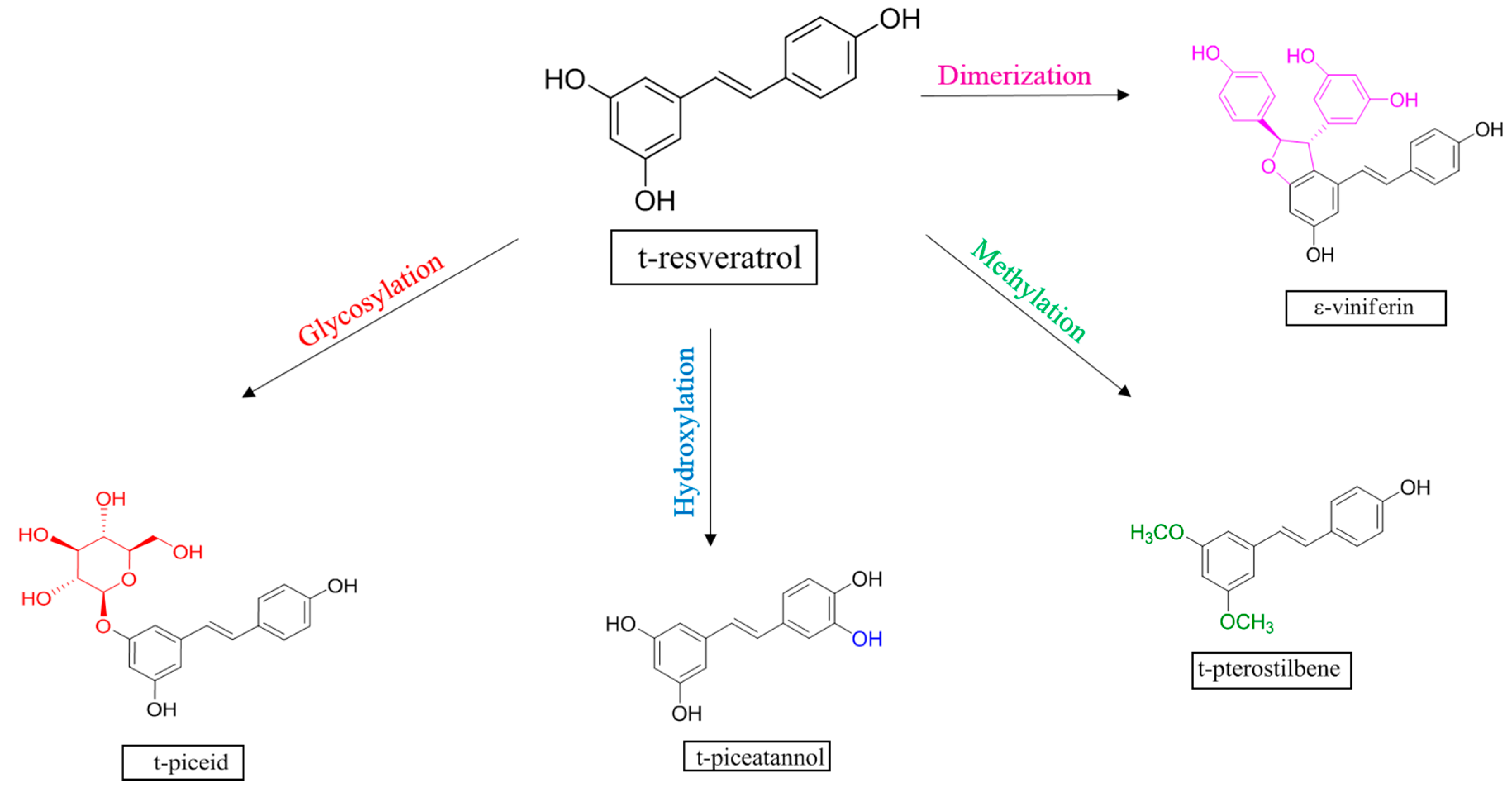
:1. Introduction
2. Results
2.1. Optimization of Ion Source Parameters
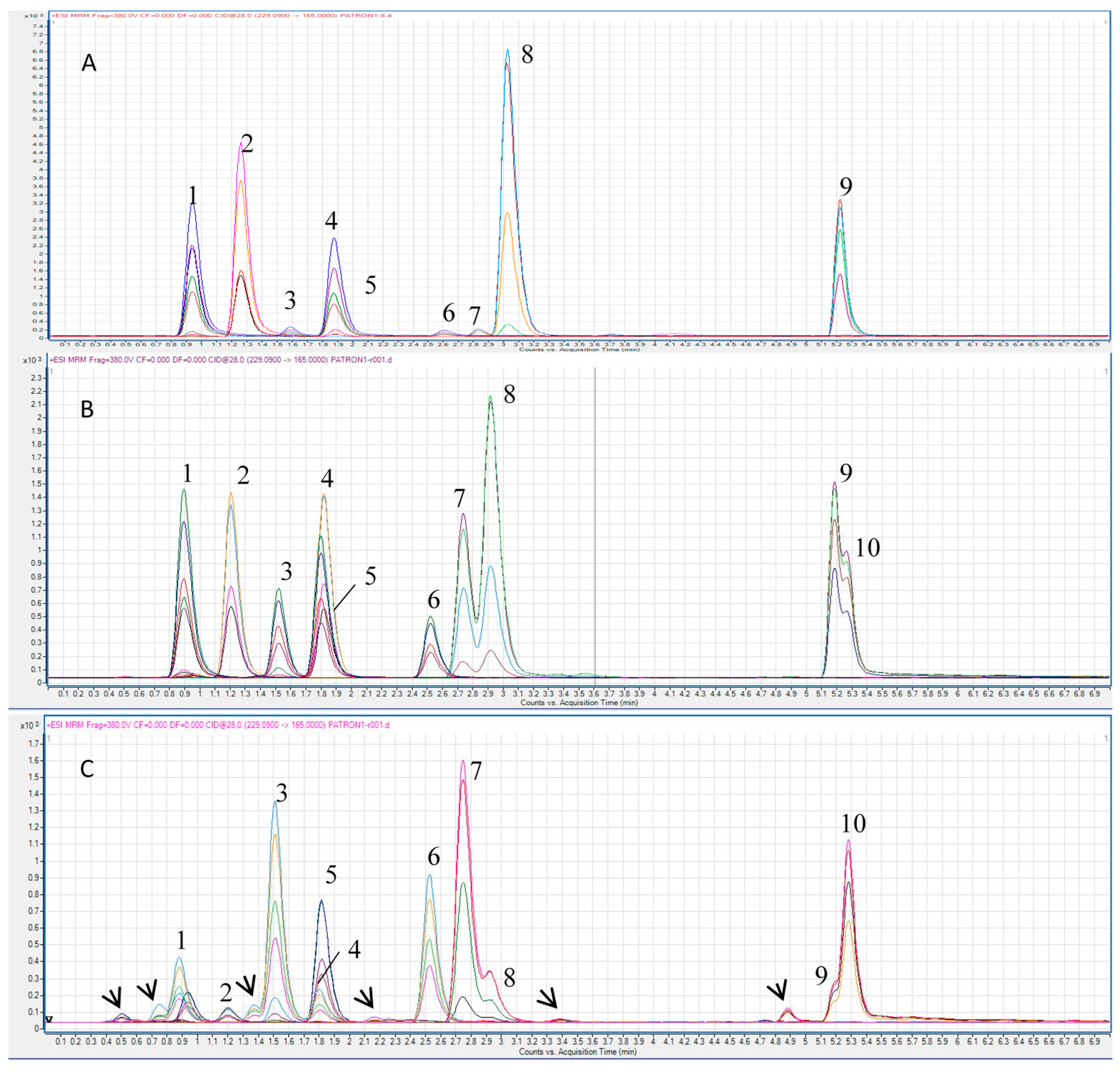
2.2. Selection of MRM Transitions and Optimization of Chromatographic Separation
2.3. MRM Quantitation of Stilbenes in a Standard Mixture
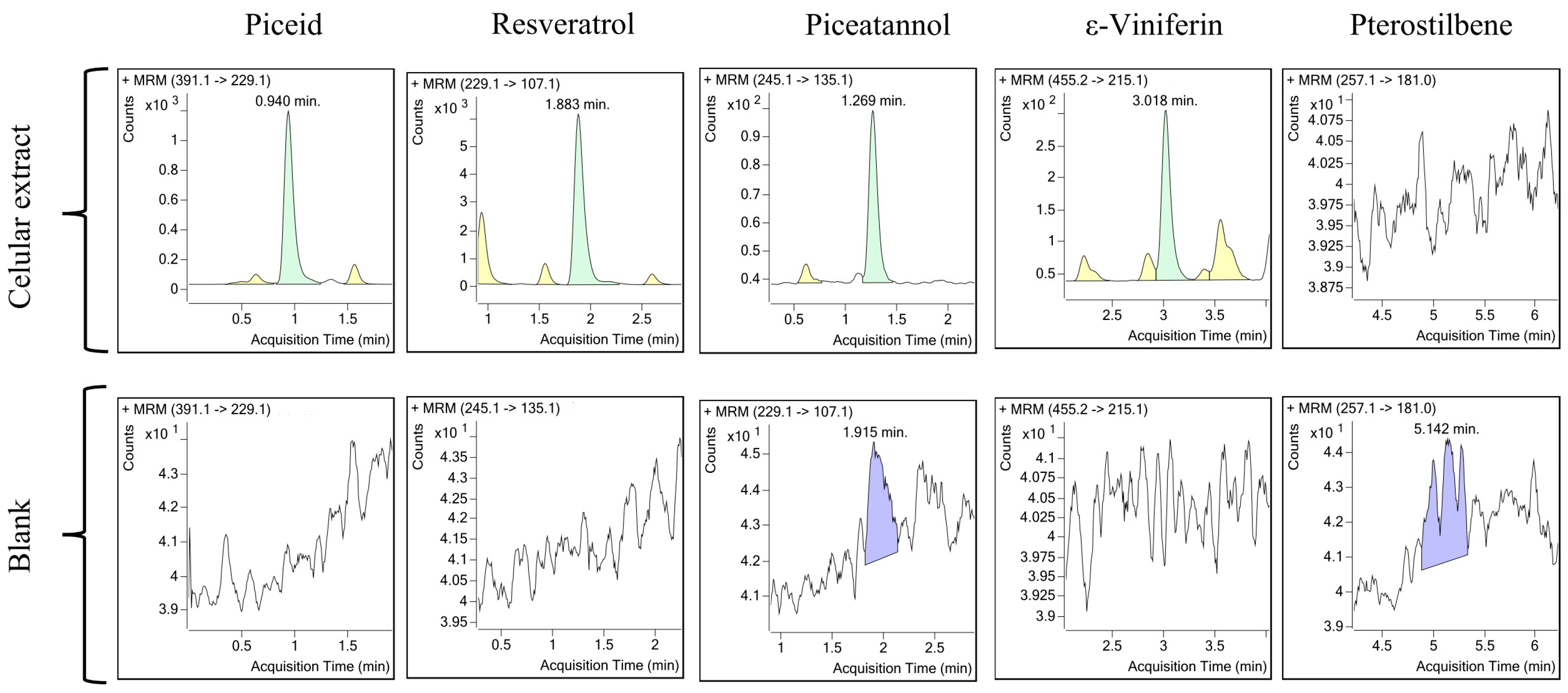
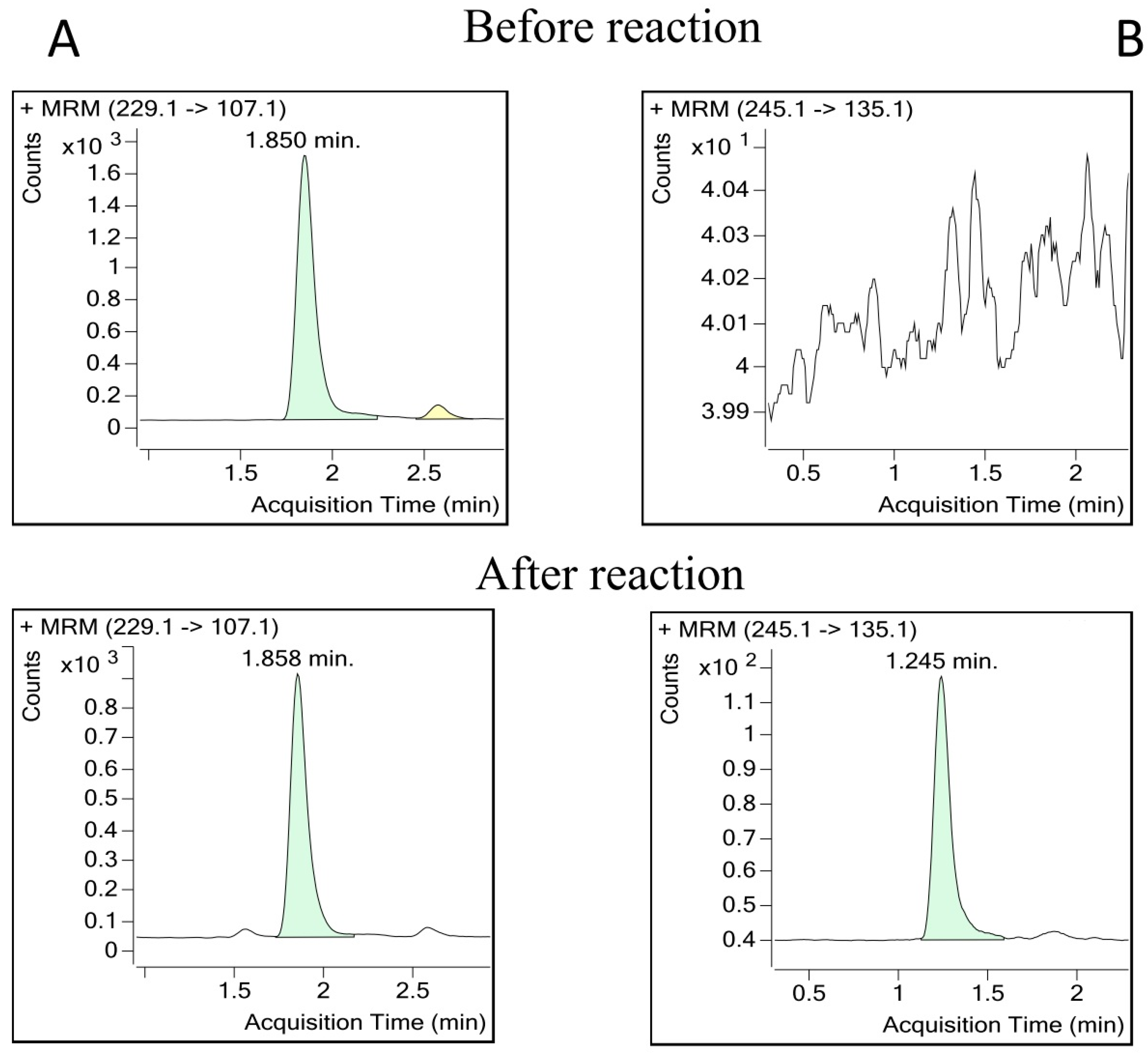
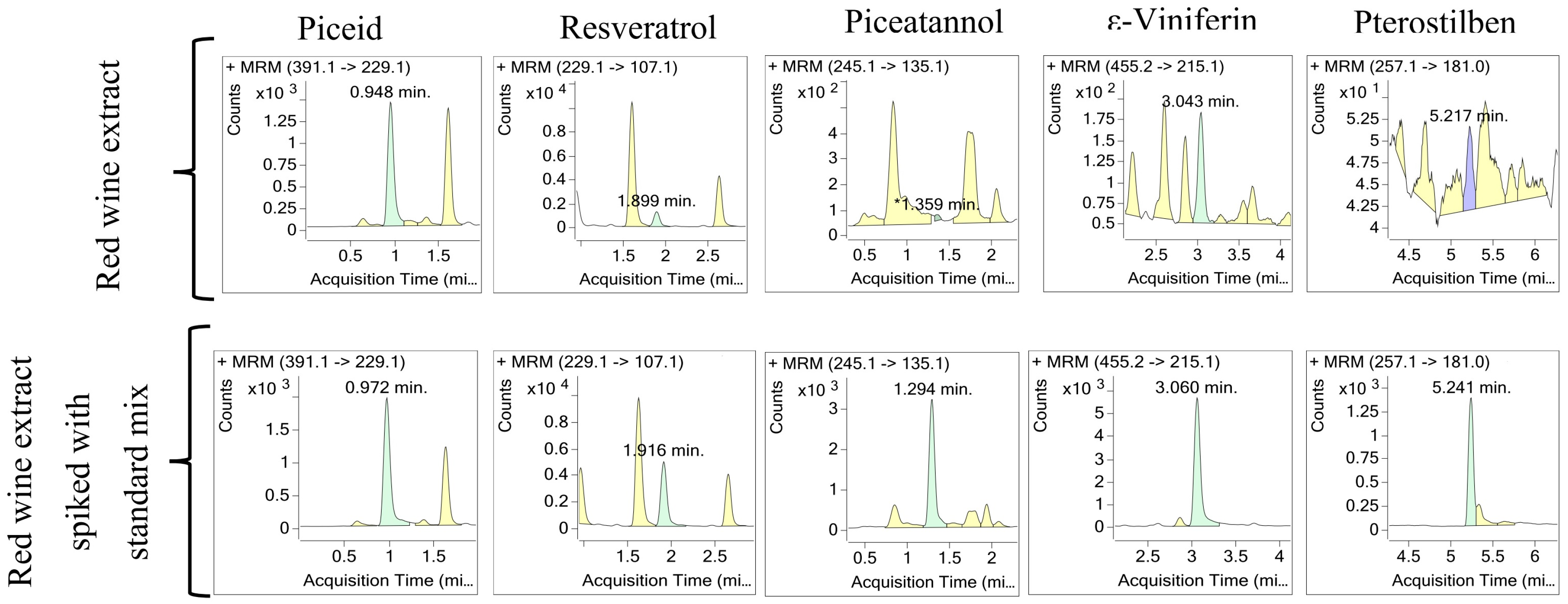
2.4. Examples of Applications of the Optimized Method
3. Discussion
4. Materials and Methods
4.1. MRM Method Optimization
4.1.1. Chemicals and Reagents
4.1.2. Preparation of Standard Solutions
4.1.3. Chromatography and Mass Conditions
4.1.4. Calibration Curves and Lower Limit of Quantitation
4.2. Method Applications
4.2.1. Analysis and Quantitation of Stilbenes Produced by V. vinifera Cell Culture Upon Elicitation
4.2.2. Biotransformation Assays
4.2.3. Analysis and Quantitation of Stilbenes in Red Wine
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pezet, R.; Gindro, K.; Viret, O.; Richter, H. Effects of resveratrol and pterostilbene on Plasmopara viticola zoospore mobility and disease development. Vitis 2004, 43, 145–148. [Google Scholar]
- Bru, R.; Selles, S.; Casado-Vela, J.; Belchi-Navarro, S.; Pedreño, M.A. Modified cyclodextrins are chemically defined glucan inducers of defense response in grapevine cell cultures. J. Agric. Food Chem. 2006, 54, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Adrian, M.; Jeandet, P. Effects of resveratrol on the ultrastructure of Botrytis cinerea conidia and biological significance in plant/pathogen interactions. Fitoterapia 2012, 83, 1345–1350. [Google Scholar] [CrossRef] [PubMed]
- Cantos, E.; Espin, J.C.; Fernandez, M.J.; Oliva, J.; Tomas-Barberan, F.A. Post-harvest UV-C irradiated grapes as potential source for producing stilbene-enriched red wines. J. Agric. Food Chem. 2003, 51, 1208–1241. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Esteso, M.J.; Selles, S.; Vera-Urbina, J.C.; Pedreño, M.A.; Bru, R. Changes of defense proteins in the extracelular proteome of grapevine (Vitis vinífera cv. Gamay) cell cultures in response to elicitors. J. Proteom. 2009, 73, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Esteso, M.J.; Selles, S.; Vera-Urbina, J.C.; Pedreño, M.A.; Bru, R. DIGE analysis of proteome changes accompanying large resveratrol production by grapevine (Vitis vinífera cv. Gamay) cell cultures in response to methyl-β-cyclodextrin and methyl jasmonate elicitors. J. Proteom. 2011, 74, 1421–1436. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Tang, K.; Yang, H.R.; Wen, P.F.; Zhang, P.; Wang, H.L.; Huang, W.D. Distribution of resveratrol and stilbene synthase in yong grape plants (Vitis vinifera L. cv. Cabernet sauvignon) and the effect of UV-C on its accumulation. Plant Physiol. Biochem. 2010, 48, 142–152. [Google Scholar] [CrossRef] [PubMed]
- Jeandet, P.; Clément, C.; Courot, E.; Cordelier, S. Modulation of phytoalexin biosynthesis in engineered plants for disease resistance. Int. J. Mol. Sci. 2013, 14, 14136–14170. [Google Scholar] [CrossRef] [PubMed]
- Jeandet, P.; Hébrard, C.; Deville, M.A.; Cordelier, S.; Dorey, S.; Aziz, A.; Crouzet, J. Deciphering the role of phytoalexins in plant-microorganism interactions and human health. Molecules 2014, 19, 18033–18056. [Google Scholar] [CrossRef] [PubMed]
- Tsai, H.-Y.; Ho, C.-T.; Chen, Y.-K. Biological actions and molecular effects of resveratrol, pterostilbene and 3′-hydroxypterostilbene. J. Food Drug Anal. 2016, 25, 134–147. [Google Scholar] [CrossRef]
- Bradamante, S.; Barenghi, L.; Villa, A. Cardiovascular protective effects of resveratrol. Cardiovasc. Drug Rev. 2014, 22, 169–188. [Google Scholar] [CrossRef]
- Le Corre, L.; Chalabi, N.; Delort, L.; Bignon, Y.J.; Bernard-Gallon, D.J. Resveratrol and breast cancer chemoprevention: Molecular mechanism. Mol. Nutr. Food Res. 2005, 49, 462–471. [Google Scholar] [CrossRef] [PubMed]
- Wolter, F.; Ulrich, S.; Stein, J. Molecular mechanism of the chemopreventive effects of resveratrol and its analogs in colorectal cancer: Key role of polyamines. J. Nutr. 2004, 134, 3219–3222. [Google Scholar] [PubMed]
- Ratan, H.L.; Steward, W.P.; Gescher, A.J.; Mellon, J.K. Resveratrol—A prostate cancer chemopreventive agent? Urol. Oncol. 2002, 7, 223–227. [Google Scholar] [CrossRef]
- Fernández-Pérez, F.; Belchí-Navarro, S.; Almagro, L.; Bru, R.; Pedreño, M.A.; Gómez-Ros, L.V. Cytotoxic effect of natural trans-Resveratrol obtained from elicted Vitis vinifera cell cultures on three cancer lines. Plant Foods Hum. Nutr. 2012, 67, 422–429. [Google Scholar] [CrossRef] [PubMed]
- De la Lastra, C.A.; Villegas, I. Resveratrol as an anti-inflamatory and antiaging agent: Mechanism and clinical implications. Mol. Nutr. Food Res. 2005, 49, 405–430. [Google Scholar] [CrossRef] [PubMed]
- De la Lastra, C.A.; Villegas, I. Resveratrol as an antioxidant and pro-oxidant agent: Mechanism and clinical implications. Biochem. Soc. Trans. 2007, 35, 1156–1160. [Google Scholar] [CrossRef] [PubMed]
- Okawara, M.; Katshuki, H.; Kurimoto, E.; Chibata, H.; Kume, T.; Akaike, K. Resveratrol protects dopaminergics neurons in midbrains slice culture from multiple insults. Biochem. Pharmacol. 2007, 73, 550–560. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baur, J.A.; Sinclair, D.A. Therapeutic potential of resveratrol: The in vivo evidence. Nat. Rev. Drug Discov. 2006, 5, 493–506. [Google Scholar] [CrossRef] [PubMed]
- Reinisalo, M.; Kårlund, A.; Koskela, A.; Kaarniranta, K.; Karjalainen, R.O. Polyphenol Stilbenes: Molecular Mechanisms of Defence against Oxidative Stress and Aging-Related Diseases. Oxid. Med. Cell. Longev. 2015. [Google Scholar] [CrossRef] [PubMed]
- Szekeres, T.; Saiko, P.; Fritzer-Szekeres, M.; Djavan, B.; Jäger, W. Chemopreventive effects of resveratrol and resveratrol derivatives. Ann. N. Y. Acad. Sci. 2011, 1215, 89–98. [Google Scholar] [CrossRef] [PubMed]
- Langcake, P.; Pryce, R.J. A new class of phytoalexins from grapevines. Experientia 1977, 33, 151–152. [Google Scholar] [CrossRef] [PubMed]
- Siemann, E.H.; Creasy, L.L. Concentration of the phytoalexin resveratrol in wine. Am. J. Enol. Vitic. 1992, 43, 49–52. [Google Scholar]
- Sanders, T.H.; McMichael, R.W., Jr.; Hendrix, K.W. Occurrence of resveratrol in edible peanuts. J. Agric. Food Chem. 2000, 48, 1243–1246. [Google Scholar] [CrossRef] [PubMed]
- Rimando, A.M.; Kalt, W.; Magee, J.B.; Dewey, J.; Ballington, J.R. Resveratrol, pterostilbene, and piceatannol in vaccinium berries. J. Agric. Food Chem. 2004, 52, 4713–4719. [Google Scholar] [CrossRef] [PubMed]
- Hurst, W.J.; Glinski, J.A.; Miller, K.B.; Apgar, J.; Davey, M.H.; Stuart, D.A. Survey of the trans-R and trans-piceid content of cocoa-containing and chocolate products. J. Agric. Food Chem. 2008, 56, 8374–8378. [Google Scholar] [CrossRef] [PubMed]
- Guilford, J.M.; Pezzuto, J.M. Wine and health: A review. Am. J. Enol. Vitic. 2011, 62, 471–486. [Google Scholar] [CrossRef]
- Jeandet, P.; Breuil, A.C.; Adrian, M.; Weston, L.A.; Debord, S.; Meunier, P.; Maume, B.; Bessis, R. HPLC analysis of grapevine phytoalexins coupling photodiode array detection and fluorometry. Anal. Chem. 1997, 69, 5172–5177. [Google Scholar] [CrossRef]
- Kallithraka, S.; Tsoutsouras, R.; Tzourou, E.; Lanaridis, P. Principal phenolic compounds in Greek red wines. Food Chem. 2006, 99, 784–793. [Google Scholar] [CrossRef]
- Prosen, H.; Strilic, M.; Kocar, D.; Rusjan, D. In vino veritas: LC-MS in wine analysis. LC-GC Eur. 2007, 20, 617–621. [Google Scholar]
- Püssa, T.; Janar, F.; Paul, K.; Ain, R. Survey of grapevine Vitis vinifera stem polyphenols by liquid chromatography-diode array detection-tamdem mass spectrometry. J. Agric. Food Chem. 2006, 54, 7488–7494. [Google Scholar] [CrossRef] [PubMed]
- Sun, B.; Leandro, M.C.; de Freitas, V.; Spranger, M.I. Fractionation of red wine polyphenols by solid-phase extraction and liquid chromatography. J. Chromatogr. A 2006, 1128, 27–38. [Google Scholar] [CrossRef] [PubMed]
- Mulinacci, N.; Innocenti, M.; Santamaria, A.R.; la Marca, G.; Pasqua, G. High-performance liquid chromatography/electrospray ionization tandem mass spectrometric investigation of stilbenoids in cell cultures of Vitis vinifera L., cv. Malvasia. Rapid Commun. Mass Spectrom. 2010, 24, 2065–2073. [Google Scholar] [CrossRef] [PubMed]
- Jaitz, L.; Siegl, K.; Eder, R.; Rak, G.; Abranko, L.; Koellensperger, G.; Hann, S. LC-MS/MS analysis of phenols for classification of red wine according to geographic origin, grape variety and vintage. Food Chem. 2010, 122, 366–372. [Google Scholar] [CrossRef]
- Ortega, N.; Romero, M.P.; Macià, A.; Reguant, J.; Anglès, N.; Morelló, J.R.; Motilva, M.J. Obtention and characterization of phenolic extracts from differents cocoa sources. J. Agric. Food Chem. 2008, 65, 9621–9627. [Google Scholar] [CrossRef] [PubMed]
- Ortega, N.; Macià, A.; Romero, M.P.; Trullols, E.; Morelló, J.R.; Anglès, N.; Motilva, M.J. Rapid determination of phenolic compound and alkaloids of carob flour by improved liquid chromatography tamdem mass spectrometry. J. Agric. Food Chem. 2009, 57, 7239–7244. [Google Scholar] [CrossRef] [PubMed]
- Lambert, M.; Meudec, E.; Verbaere, A.; Mazerolles, G.; Wirth, J.; Masson, G.; Cheynier, V.; Sommerer, N. A High-Troughput UHPLC-QqQ-Ms Method for polyphenol profiling in Rosé wines. Molecules 2015, 20, 7890–7914. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Márquez, A.; Morante-Carriel, J.A.; Ramírez-Estrada, K.; Cusidó, R.M.; Palazon, J.; Bru-Martínez, R. Production of highly bioactive resveratrol analogues pterosilbene and piceatannol in metabolically engineered grapevine cell cultures. Plant Biotech. J. 2016, 14, 1813–1825. [Google Scholar] [CrossRef] [PubMed]
- Piver, B.; Fer, M.; Vitrac, X.; Merillon, J.M.; Dreano, Y.; Berthou, F.; Lucas, D. Involvement of cytochrome P450 1A2 in the biotransformation of trans-resveratrol in human liver microsomes. Biochem. Pharmacol. 2004, 68, 773–782. [Google Scholar] [CrossRef] [PubMed]
- Kallithraka, S.; Arvanitoyannis, I.; El-Zajouli, A.; Kelafas, P. The application of an improved method for trans-R to determine the origin of Greek red wines. Food Chem. 2001, 75, 355–363. [Google Scholar] [CrossRef]
- Rotches-Ribalta, M.; Urpi-Sarda, M.; Llorach, R.; Boto-Ordoñez, M.; Jauregui, O.; Chiva-Blanch, G.; Perez-Garcia, L.; Jaeger, W.; Guillen, M.; Corella, D.; et al. Gut and microbial resveratrol metabolite profiling after moderate long-term consumption of red wine versus dealcoholized red wine in humans by an optimized ultra-high-pressure liquid chromatography tandem mass spectrometry method. J. Chromatogr. A 2012, 1265, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Vrhovsek, U.; Masuero, D.; Gasperotti, M.; Franceschi, P.; Caputi, L.; Viola, R.; Mattivi, F. A versatile targeted metabolomics method for the rapid quantifaction of multiple class of phenolics in fruits and beverages. J. Agric. Food Chem. 2012, 60, 8831–8840. [Google Scholar] [CrossRef] [PubMed]
- Yang, I.; Kim, E.; Kang, J.; Han, H.; Sul, S.; Park, S.B.; Kim, S.K. Photochemical generation of a new, highly fluorescent compound from non-fluorescent resveratrol. Chem. Commun. 2012, 48, 3839–3841. [Google Scholar] [CrossRef] [PubMed]
- Morales, M.; Ros Barcelo, A.; Pedreno, M.A.; Hemantaranja, A. Plant stilbenes: Recent advances in their chemistry and biology. Adv. Plant Physiol. 2000, 3, 39–70. [Google Scholar]
- Abrankó, L.; García-Reyes, J.F.; Molina-Diaz, A. In-source fragmentation and accurate mass analysis of multiclass flavonoid conjugates by electrospray ionization time-of-flight mass spectrometry. J. Mass Spectrom. 2011, 46, 478–488. [Google Scholar] [CrossRef] [PubMed]
- Stervbo, U.; Vang, O.; Bonnesen, C. A review of the content of the putative chemopreventive phytoalexin resveratrol in red wine. Food Chem. 2007, 101, 449–457. [Google Scholar] [CrossRef]
- Jemal, M.; Hawthrone, D.J. Effect of High Performance Liquid Chromatography mobile phase (Methanol versus Acetonitrile) on the positive and negative ion Electrospray Response of a compound that contains both an unsaturated lactone and a methyl sulfone group. Rapid Commun. Mass Spectrom. 1999, 13, 61–66. [Google Scholar] [CrossRef]
- Omar, J.M.; Yang, H.; Li, S.; Marquardt, R.R.; Jones Peter, J.H. Development of an Improved Reverse-Phase High-Performance Liquid Chromatogrpahy Method for the simultaneous Analyses of trans-/cis-Resveratrol, Quercitin, and Emodin in commercial Reveratrol supplements. J. Agric. Food Chem. 2014, 62, 5812–5817. [Google Scholar] [CrossRef] [PubMed]
- Pinasseau, L.; Verbaere, A.; Roques, M.; Meudec, E.; Vallverdu-Queralt, A.; Terrier, N.; Boulet, J.C.; Cheyner, V.; Sommerer, N. A Fast and robus UHPLC-MRM-Ms method to characterize and Quantify Grape Skin Tannins after Chemical Depolymerization. Molecules 2016, 21, 1409. [Google Scholar] [CrossRef] [PubMed]
- Buiarelli, F.; Coccioli, F.; Jasionowska, R.; Merolle, M.; Terracciano, A. Analysis of some stilbenes in Italian wines by liquid chromatography/tándem mass spectrometry. Rapid Commun. Mass Spectrom. 2007, 21, 2955–2964. [Google Scholar] [CrossRef] [PubMed]
- Schmidlin, L.; Poutaraud, A.; Claudel, P.; Mestre, P.; Prado, E.; Santos-Rosa, M.; Wiedemann-Merdinoglu, S.; Karst, F.; Merdinoglu, D.; Hugueney, P. A stress-inducible resveratrol O-methyltransferase involved in the biosynthesis of pterostilbene in grapevine. Plant Physiol. 2008, 148, 1630–1639. [Google Scholar] [CrossRef] [PubMed]
- Richard, T.; Poupard, P.; Nassra, M.; Papastamoulis, Y.; Iglesias, M.L.; Krisa, S.; Waffo-Teguo, P.; Mérillon, J.M.; Monti, J.P. Protective effect of ε-viniferin on β-amyloid peptide aggregation investigated by electrospray ionization mass spectrometry. Bioorganic Med. Chem. 2011, 19, 3152–3155. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.H.; Ahn, T.; Jung, H.C.; Pan, J.G.; Yun, C.H. Generation of the human metabolite piceatannol from the anticancer preventive agent resveratrol by bacterial cytochrome P450 BM3. Drug Metab. Dispos. 2009, 37, 932–936. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Not Available.
| Compound | Formula | Mass (Da) | Precursor Ion (m/z) | Product Ion (m/z) | Collision Energy (eV) | % Intensity | Retention Time (Min–Max) trans/cis | LOD (mg/L) | LOQ (mg/L) | Linearity b |
|---|---|---|---|---|---|---|---|---|---|---|
| Piceid | C20H22O8 | 390.13 | 391.1 | 114.8 | 20 | 86.5 | 0.93–0.99/1.52–1.63 | 0.04 | 0.07 | 640 |
| 308.8 | 8 | 67.5 | ||||||||
| 349.9 | 0 | 100 | ||||||||
| 229.1 a | 8 | 24.4 | ||||||||
| Resveratrol | C14H12O3 | 228.08 | 229.09 | 107.1 a | 24 | 100 | 1.85–1.99/2.55–2.65 | 0.12 | 0.22 | 800 |
| 91 a | 24 | 45.7 | ||||||||
| 135 a | 8 | 80.9 | ||||||||
| 165 a | 28 | 35.6 | ||||||||
| Piceatannol | C14H12O4 | 244.07 | 245.08 | 107.1 | 20 | 95.5 | 1.25–1.35/1.90–2.00 | 0.12 | 0.15 | 1600 |
| 135.1 | 12 | 100 | ||||||||
| 152 | 36 | 11.6 | ||||||||
| 181.1 | 24 | 38.7 | ||||||||
| ε-Viniferin | C28H22O6 | 454.14 | 455.15 | 107.1 | 32 | 100 | 3.00–3.17/2.80–2.90 | 0.07 | 0.09 | 1777 |
| 215.1 | 20 | 75.0 | ||||||||
| 349.1 | 16 | 38.5 | ||||||||
| 199.1 | 24 | 32.0 | ||||||||
| Pteroestilbene | C16H16O3 | 256,11 | 257.12 | 181 | 40 | 90.1 | 5.18–5.28/5.26–5.35 | 0.06 | 0.08 | 1000 |
| 133.1 | 12 | 100 | ||||||||
| 91 | 28 | 58.5 | ||||||||
| 165.1 | 40 | 72.2 |
| Compound | Concentration (mg/L) in | ||
|---|---|---|---|
| Extract #1 | Extract #2 | Extract #3 | |
| Piceid | 10.92 | 14.07 | 16.35 |
| Resveratrol | 46.93 | 62.24 | 50.82 |
| Piceatannol | 1.44 | 1.46 | 1.38 |
| ε-Viniferin | 1.48 | 0.93 | 0.11 |
| Pteroestilbene | n.d. | n.d. | n.d. |
| t-Piceid (mg/L) | t-Resveratrol (mg/L) | t-Piceatannol (mg/L) | ε-Viniferin (mg/L) | t-Pterostilbene (mg/L) | |
|---|---|---|---|---|---|
| wine | 0.826 | 0.281 | n.d. | 0.014 | n.d. |
| S/N | 140.24 | 60.86 | 0.93 | 15.6 | 1.67 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hurtado-Gaitán, E.; Sellés-Marchart, S.; Martínez-Márquez, A.; Samper-Herrero, A.; Bru-Martínez, R. A Focused Multiple Reaction Monitoring (MRM) Quantitative Method for Bioactive Grapevine Stilbenes by Ultra-High-Performance Liquid Chromatography Coupled to Triple-Quadrupole Mass Spectrometry (UHPLC-QqQ). Molecules 2017, 22, 418. https://doi.org/10.3390/molecules22030418
Hurtado-Gaitán E, Sellés-Marchart S, Martínez-Márquez A, Samper-Herrero A, Bru-Martínez R. A Focused Multiple Reaction Monitoring (MRM) Quantitative Method for Bioactive Grapevine Stilbenes by Ultra-High-Performance Liquid Chromatography Coupled to Triple-Quadrupole Mass Spectrometry (UHPLC-QqQ). Molecules. 2017; 22(3):418. https://doi.org/10.3390/molecules22030418
Chicago/Turabian StyleHurtado-Gaitán, Elías, Susana Sellés-Marchart, Ascensión Martínez-Márquez, Antonio Samper-Herrero, and Roque Bru-Martínez. 2017. "A Focused Multiple Reaction Monitoring (MRM) Quantitative Method for Bioactive Grapevine Stilbenes by Ultra-High-Performance Liquid Chromatography Coupled to Triple-Quadrupole Mass Spectrometry (UHPLC-QqQ)" Molecules 22, no. 3: 418. https://doi.org/10.3390/molecules22030418
APA StyleHurtado-Gaitán, E., Sellés-Marchart, S., Martínez-Márquez, A., Samper-Herrero, A., & Bru-Martínez, R. (2017). A Focused Multiple Reaction Monitoring (MRM) Quantitative Method for Bioactive Grapevine Stilbenes by Ultra-High-Performance Liquid Chromatography Coupled to Triple-Quadrupole Mass Spectrometry (UHPLC-QqQ). Molecules, 22(3), 418. https://doi.org/10.3390/molecules22030418







