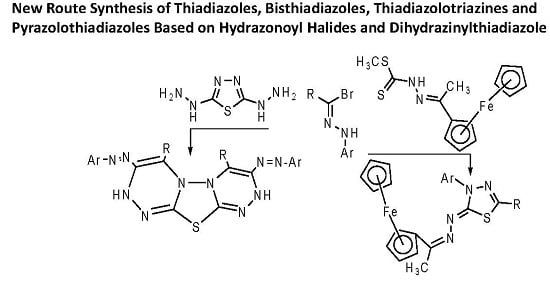
New Route Synthesis of Thiadiazoles, Bisthiadiazoles, Thiadiazolotriazines, and Pyrazolothiadiazoles Based on Hydrazonoyl Halides and Dihydrazinylthiadiazole
Abstract
:1. Introduction
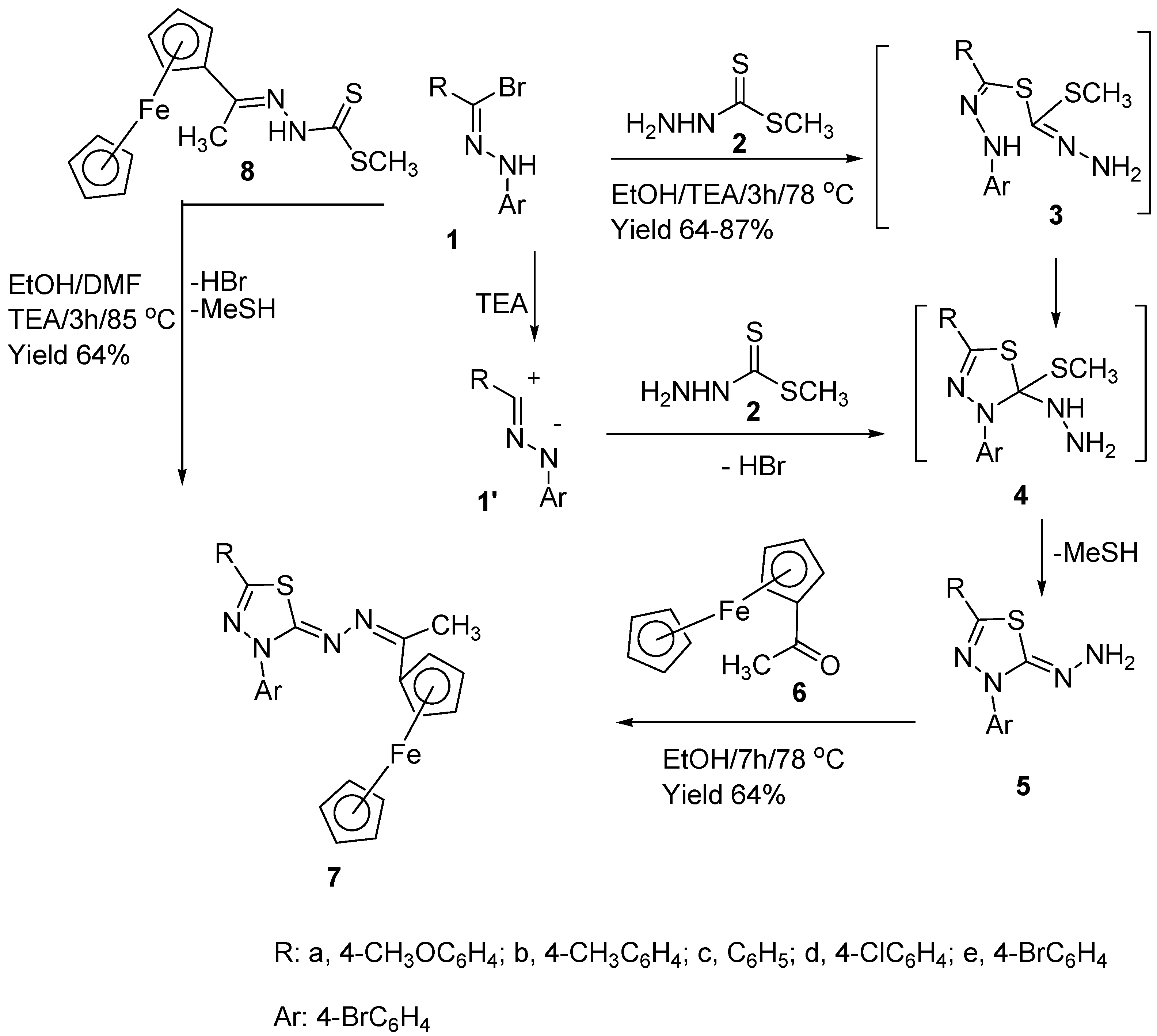
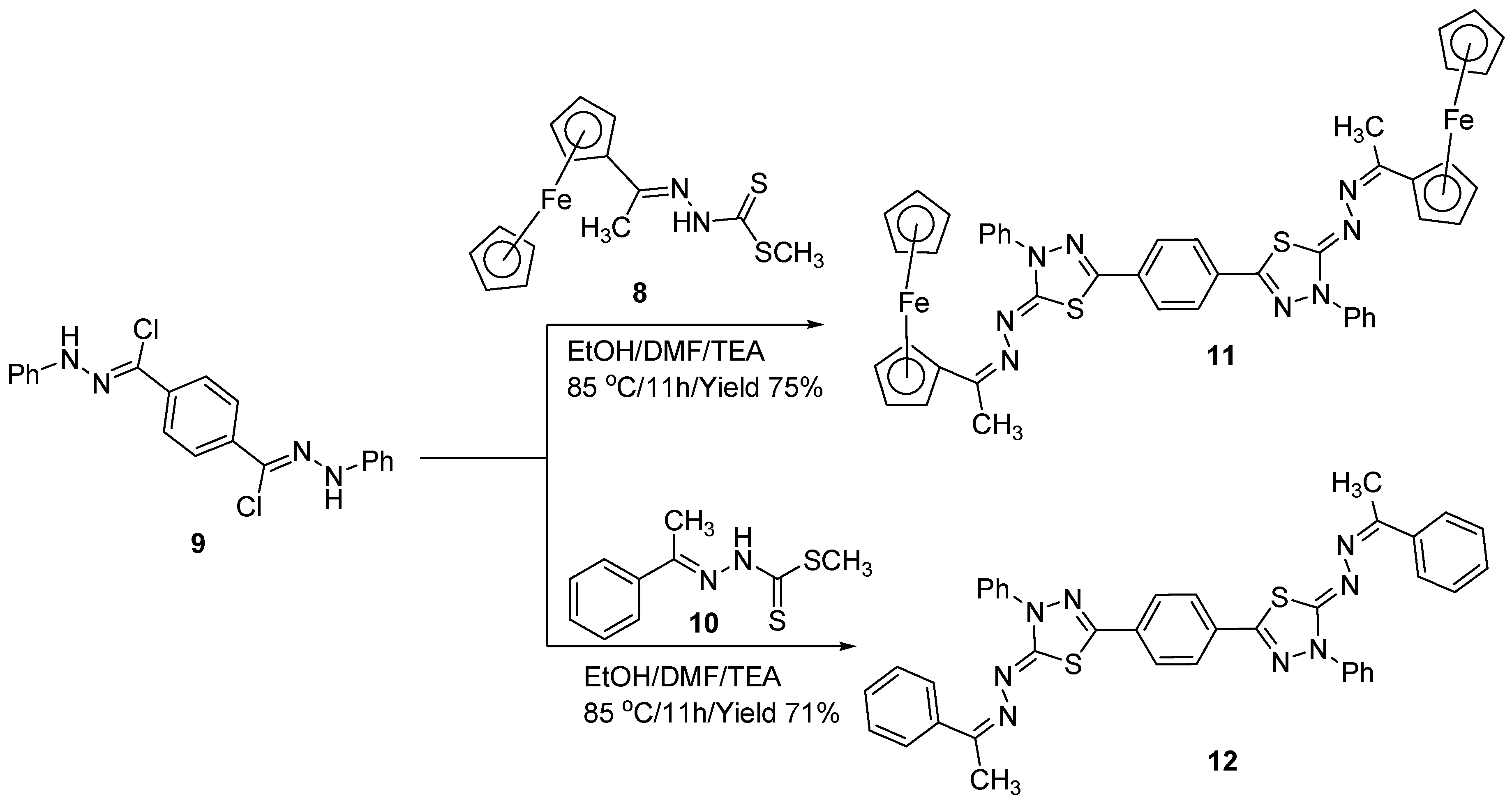
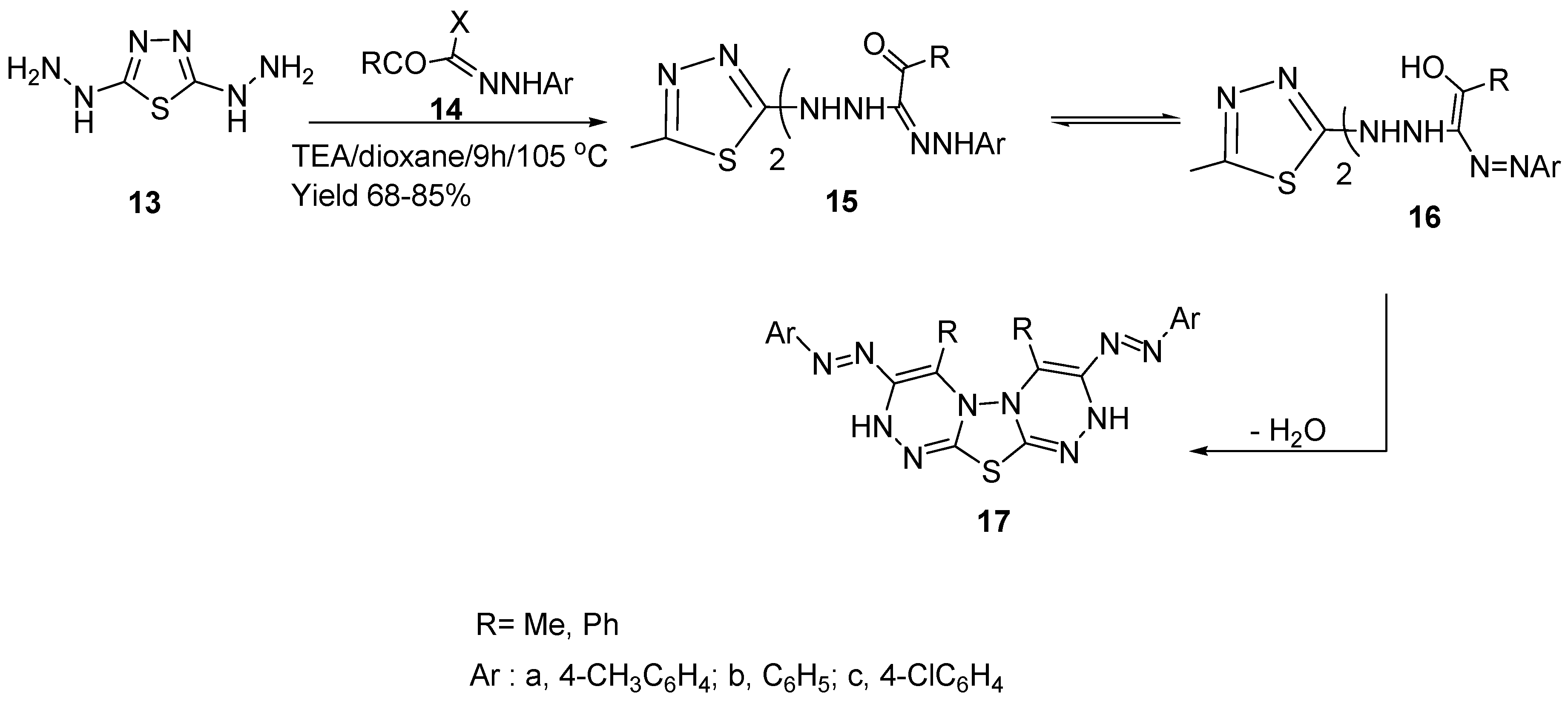
2. Results and Discussion
3. Experimental Section
3.1. Synthesis of Thiadiazoles (5a–e)
3.2. Synthesis of 2-(1-Ferrocenylethylidenehydrazono-3-(4-bromophenyl)-5-methoxyphenyl)-2,3-dihydro-1,3,4-thiadiazole (7)
3.3. Synthesis of Bisthiadiazoles (11) and (12)
3.4. Synthesis of [1,3,4]Thiadiazolo[2,3-c:5,4-c']bis([1,2,4]triazines) (17a–c)
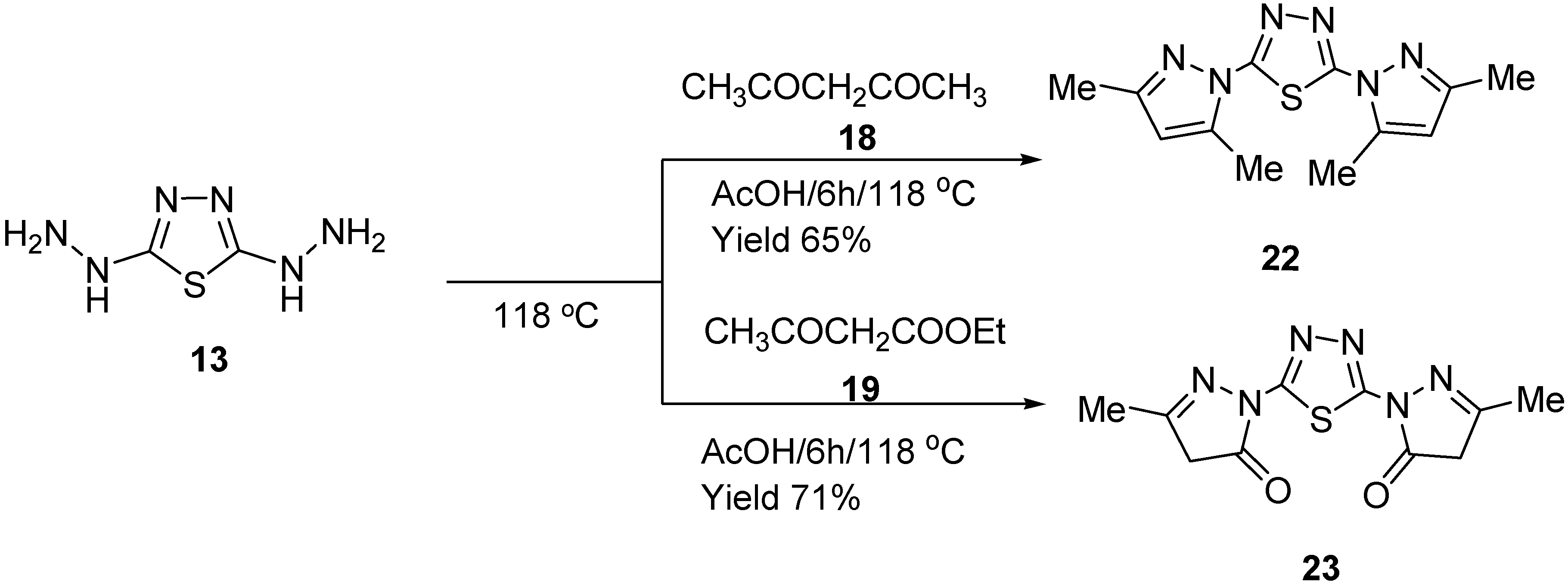
3.5. Synthesis of Pyrazolothiadiazole (22) and (23)
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Shawali, A.S.; Sayed, A.R.; Zayed, M.M. A convenient method for synthesis of bis-2,2′-(1,3,4-thiadiazole) and bis-3,3′-(1,2,4-triazole) derivatives. J. Sulfur Chem. 2011, 32, 311–314. [Google Scholar] [CrossRef]
- Sayed, A.R. Synthesis of 1,3,4-thiadiazines, bis-1,3,4-thiadiazoles, [1,2,4]triazino[3,4-b][1,3,4]thiadiazine, thiazolines from carbonothioic dihydrazide. Tetrahedron 2012, 68, 2784–2789. [Google Scholar] [CrossRef]
- Sayed, A.R. Synthesis of novel bis-thiadiazoles, bis-triazoles and polypyrazole derivatives based on hydrazonoyl halides. Tetrahedron 2013, 69, 5293–5298. [Google Scholar] [CrossRef]
- Shawali, A.S. 1,3,4-Thiadiazoles of pharmacological interest: Recent trends in their synthesis via tandem 1,3-dipolar cycloaddition: Review. J. Adv. Res. 2014, 5, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Sayed, A.; Zaki, Y.; Aish, E. A convenient route for the synthesis of new thiadiazoles. Turk. J. Chem. 2016, 40, 184–191. [Google Scholar] [CrossRef]
- Mann, J.; Baron, A.; Opoku-Boahen, Y.E.; Johansson, G.; Parkinson, L.R.; Kelland, S.; Neidle, A. A New Class of Symmetric Bisbenzimidazole-Based DNA Minor Groove-Binding Agents Showing Antitumor Activity. J. Med. Chem. 2001, 44, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Ujjinamatada, R.K.; Baier, A.; Borowski, P.; Hosmane, R.S. An analogue of AICAR with dual inhibitory activity against WNV and HCV NTP ase/helicase: Synthesis and in vitro screening of 4-carbamoyl-5-(4,6-diamino-2,5-dihydro-1,3,5-triazin-2-yl)imidazole-1-β-d-ribofuranoside. Bioorg. Med. Chem. Lett. 2007, 17, 2285–2288. [Google Scholar] [CrossRef] [PubMed]
- Rostom, S.A.F.; Ashour, H.M.A.; El Razik, H.A.; El Fattah, A.F.; El-Din, N.N. Azole antimicrobial pharmacophore-based tetrazoles: Synthesis and biological evaluation as potential antimicrobial and anticonvulsant agent. Bioorg. Med. Chem. 2009, 17, 2410–2422. [Google Scholar] [CrossRef] [PubMed]
- Adams, J.L.; Boehm, J.C.; Gallagher, T.F.S.; Kassis, E.F.; Webb, R.; Hall, M.; Sorenson, R.; Garigipati, G.D.E.; Lee, J.C. Pyrimidinylimidazole Inhibitors of p38: Cyclic N-1 Imidazole Substituents Enhance p38 Kinase Inhibition and Oral Activity. Bioorg. Med. Chem. Lett. 2001, 11, 2867–2870. [Google Scholar] [CrossRef]
- Steel, P.J. Aromatic biheterocycles: Syntheses, structures, and properties. Adv. Heterocycl. Chem. 1997, 67, 1–117. [Google Scholar]
- Weinstock, L.M.; Shankai, I. Comprehensive Heterocyclic Chemistry; Potts, K.T., Ed.; Pergmann: Oxford, UK, 1984; Volume 6. [Google Scholar]
- Glossman, M.D. Local and nonlocal density functional calculations of the molecular structure of isomeric thiadiazoles. J. Mol. Struct.: THEOCHEM 1997, 390, 67–78. [Google Scholar] [CrossRef]
- Mitnik, D.G.; Lucero, A.M. HF and DFT calculations of the molecular structure of isomeric thiadiazole dioxides. J. Mol. Struct.: THEOCHEM 2001, 549, 285–288. [Google Scholar] [CrossRef]
- Krygowski, T.M. Crystallographic studies of inter- and intramolecular interactions reflected in aromatic character of π-electron systems. J. Chem. Inf. Comput. Sci. 1993, 33, 70–73. [Google Scholar] [CrossRef]
- Arakelian, A.N.; Davis, K.E. 2,5-Dimercapto-1,3,4-thiadiazole derivatives and lubricants containing them. U.S. Patent 42 46126, 1981, 20 January 1981. [Google Scholar]
- Abdelhamid, A.O.; Sallam, M.M.M.; Amer, S.A. Reaction with hydrazonoyl halides. Part 32 [1]: Reaction of C-acyl-N-(3-phenyl-5-pyrazolyl)hydrazonoyl chlorides with potassium thiocyanate and synthesis of some new 2,3-dihydro-1,3,4-thiadiazoles and slenadiazoles. Heteroat. Chem. 2001, 12, 468–474. [Google Scholar] [CrossRef]
- Antolini, L.; Benedetti, A.; Fabretti, A.C.; Giusti, A.; Menziani, M.C. Crystal and molecular structure of bis(2-amino-5-methyl-1,3,4-thiadiazole-N3)dibromomercury(II). A spectroscopic study and INDO calculations. J. Chem. Soc. Dalton Trans. 1988, 1075–1077. [Google Scholar] [CrossRef]
- Dogan, H.N.; Duran, A.; Rollas, S.; Sener, G.; Uysal, M.K.; Gulen, D. Synthesis of new 2,5-Disubstituted-1,3,4-thiadiazoles and preliminary evaluation of anticonvulsant and antimicrobial activities. Bioorg. Med. Chem. 2002, 10, 2893–2898. [Google Scholar] [CrossRef]
- Palaska, E.; Sahin, G.; Kelicen, P.; Turlu, N.T.; Altinok, G. Synthesis and anti-inflammatory activity of 1-acylthiosemicarbazides, 1,3,4-oxadiazoles, 1,3,4-thiadiazoles and 1,2,4-triazole-3-thione. Il Farmaco 2002, 57, 101–107. [Google Scholar] [CrossRef]
- Karakus, S.; Rollas, S. Synthesis and antituberculosis activity of new N-phenyl-N′-[4-(5-alkyl/arylamino-1,3,4-thiadiazole-2-yl)phenyl]thioureas. Il Farmaco 2002, 57, 577–581. [Google Scholar] [CrossRef]
- Terzioglu, N.; Gursoy, A. Synthesis and anticancer evaluation of some new hydrazone derivatives of 2,6-dimethylimidazo[2,1-b][1,3,4]thiadiazole-5-carbohydrazide. Eur. J. Med. Chem. 2003, 38, 781–786. [Google Scholar] [CrossRef]
- Bassam, A.; Khalili, F.I.; Hamadneh, I.; Al-Dujaili, A.H. Synthesis and characterization of new polyamides containing symmetrical and unsymmetrical thiadiazole rings. Fibers Polym. 2016, 17, 166–173. [Google Scholar]
- Dawood, K.M.; Gomha, S.M. Synthesis and Anti-cancer Activity of 1,3,4-Thiadiazole and 1,3-Thiazole Derivatives Having 1,3,4-Oxadiazole Moiety. J. Heterocycl. Chem. 2015, 52, 1400–1405. [Google Scholar] [CrossRef]
- Mukund, P.S.; Levi, M.S.; Takahiro, S. Enantioselective 1,3-Dipolar Cycloaddition of Nitrile Imines to α-Substituted and α,β-Disubstituted α,β-Unsaturated Carbonyl Substrates: A Method for Synthesizing Dihydropyrazoles Bearing a Chiral Quaternary Center. Adv. Synth. Catal. 2006, 348, 2371–2375. [Google Scholar]
- Audrieth, L.F.; Scott, E.S.; Kippur, P.S. Hydrazine derivatives of the carbonoic and thiocarbonic acids. I. The preparation and properties of thiocarbohydrazide. J. Org. Chem. 1954, 19, 733–741. [Google Scholar] [CrossRef]
- Ismail, K.Z. Synthesis and physicochemical studies of metal complexes of ferrocene Schiff base derivatives. Transit. Met. Chem. 1997, 22, 565–569. [Google Scholar] [CrossRef]
- Stille, I.J.K.; Harris, F.W.; Bedford, M.A. Phenyl substituted dipyrazoles: 1,3-dipole addition reactions of sydnones and nitrilimines. J. Heterocycl. Chem. 1966, 3, 155–157. [Google Scholar] [CrossRef]
- Lin, A.J.; Klayman, D.L.J. A facile synthesis of unsymmetrical heterocyclic azines by cyclodesulfurization: Reaction of methyl arylalkylidenehydrazinecarbodithioates with diamines. J. Heterocycl. Chem. 1985, 22, 1–4. [Google Scholar] [CrossRef]
- Jackman, D.E.; Jackman, D.E. Structure and synthesis of the metribuzin blue dye. Synthesis and reactions of 2,5-dihydrazino-1,3,4-thiadiazole and its derivatives. J. Heterocycl. Chem. 1990, 27, 1053–1057. [Google Scholar] [CrossRef]
- Dubenko, R.G.; Gorbenko, E.F. Substituted glyoxalic acids. XI. Synthesis of arylhydrazones of acid chlorides of substituted glyoxalic acid and reactions with amines and hydrazine. Zh. Org. Khim. 1968, 4, 634–638. [Google Scholar]
- Sample Availability: Samples of the compounds 12, 17a–c, 22 and 23 are available from the authors.
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sayed, A.R.; Al-Shihry, S.S. New Route Synthesis of Thiadiazoles, Bisthiadiazoles, Thiadiazolotriazines, and Pyrazolothiadiazoles Based on Hydrazonoyl Halides and Dihydrazinylthiadiazole. Molecules 2017, 22, 336. https://doi.org/10.3390/molecules22020336
Sayed AR, Al-Shihry SS. New Route Synthesis of Thiadiazoles, Bisthiadiazoles, Thiadiazolotriazines, and Pyrazolothiadiazoles Based on Hydrazonoyl Halides and Dihydrazinylthiadiazole. Molecules. 2017; 22(2):336. https://doi.org/10.3390/molecules22020336
Chicago/Turabian StyleSayed, Abdelwahed R., and Shar Saad Al-Shihry. 2017. "New Route Synthesis of Thiadiazoles, Bisthiadiazoles, Thiadiazolotriazines, and Pyrazolothiadiazoles Based on Hydrazonoyl Halides and Dihydrazinylthiadiazole" Molecules 22, no. 2: 336. https://doi.org/10.3390/molecules22020336
APA StyleSayed, A. R., & Al-Shihry, S. S. (2017). New Route Synthesis of Thiadiazoles, Bisthiadiazoles, Thiadiazolotriazines, and Pyrazolothiadiazoles Based on Hydrazonoyl Halides and Dihydrazinylthiadiazole. Molecules, 22(2), 336. https://doi.org/10.3390/molecules22020336