An Efficient Synthesis of Novel Bioactive Thiazolyl-Phthalazinediones under Ultrasound Irradiation
Abstract
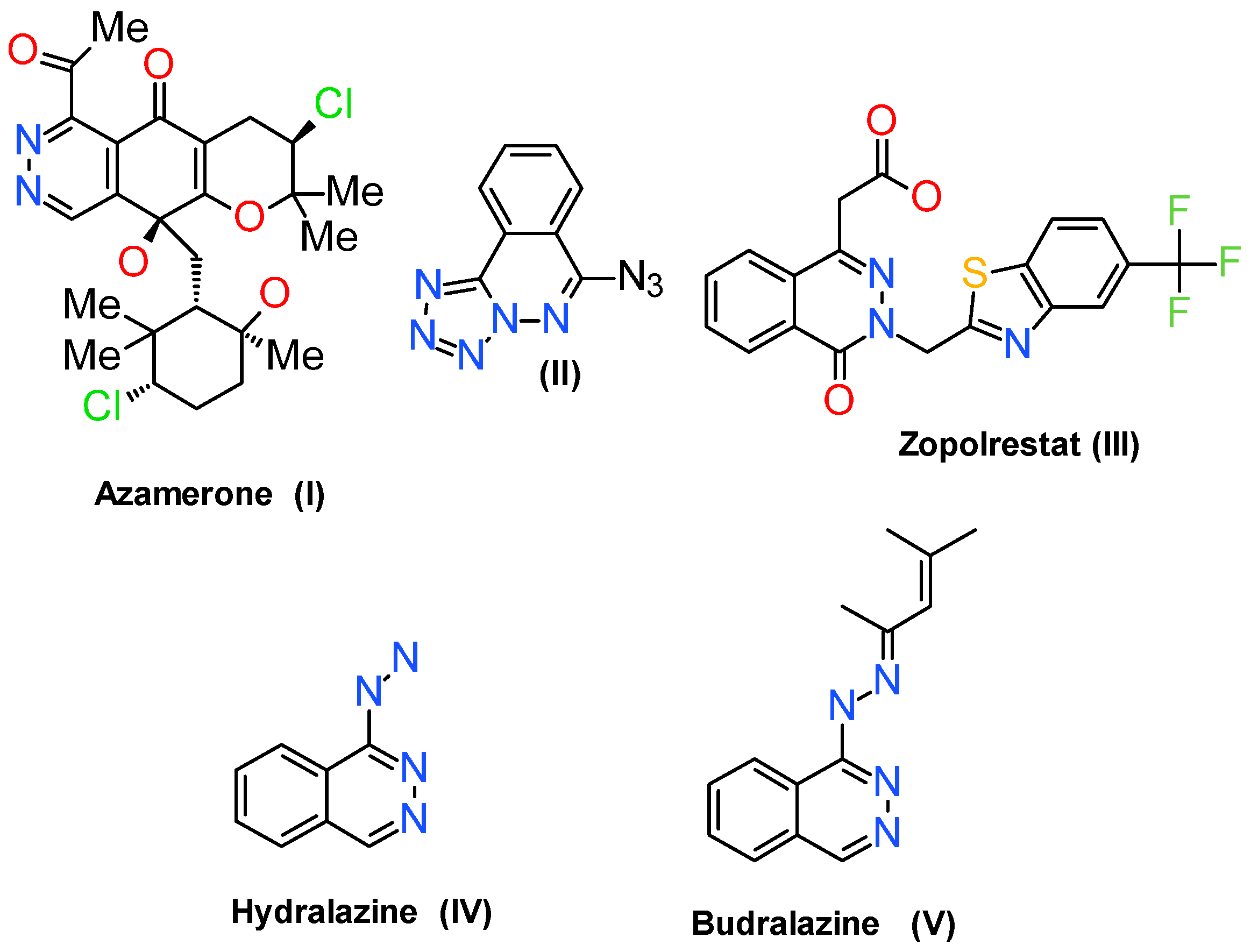
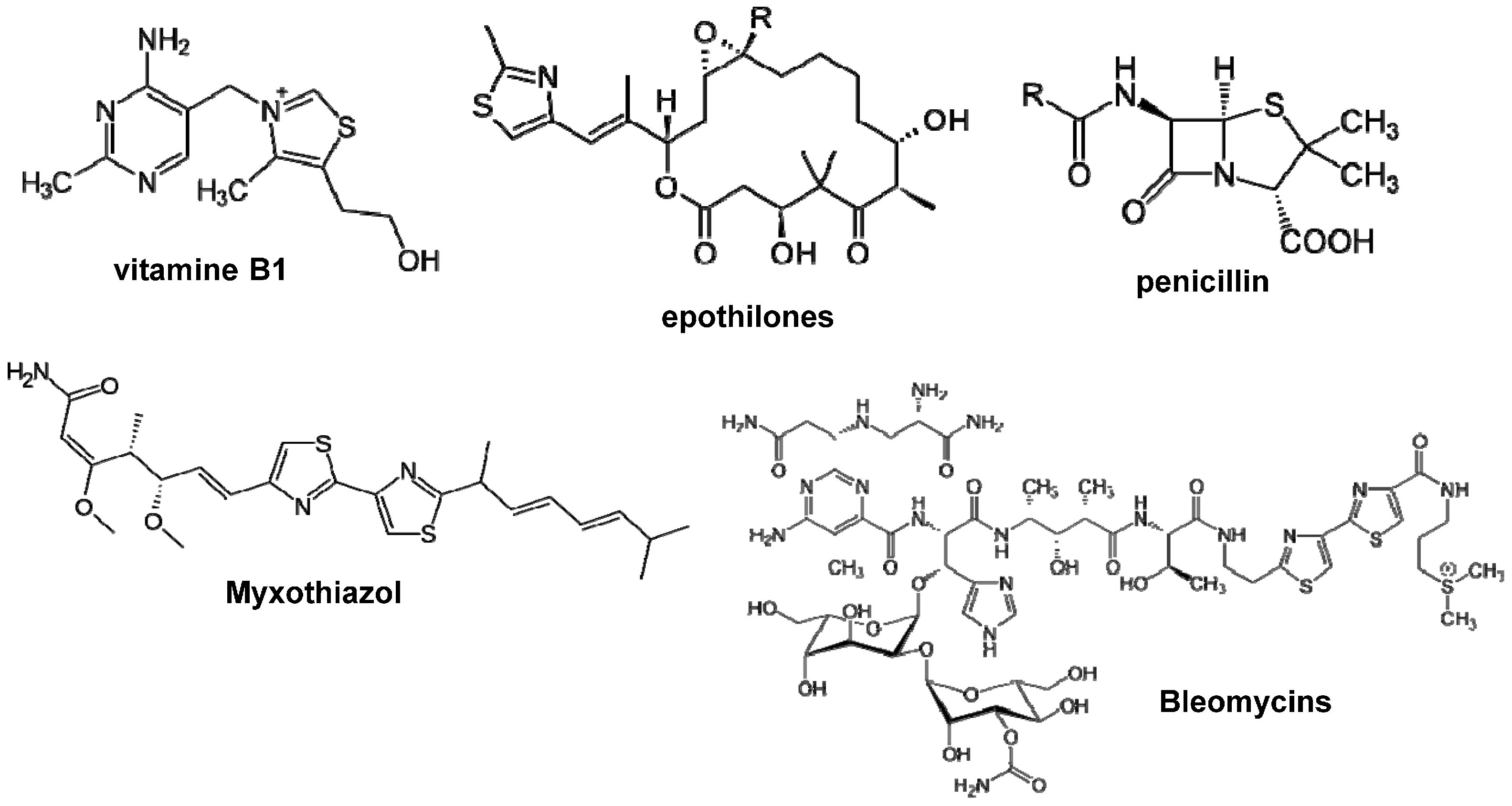
:1. Introduction
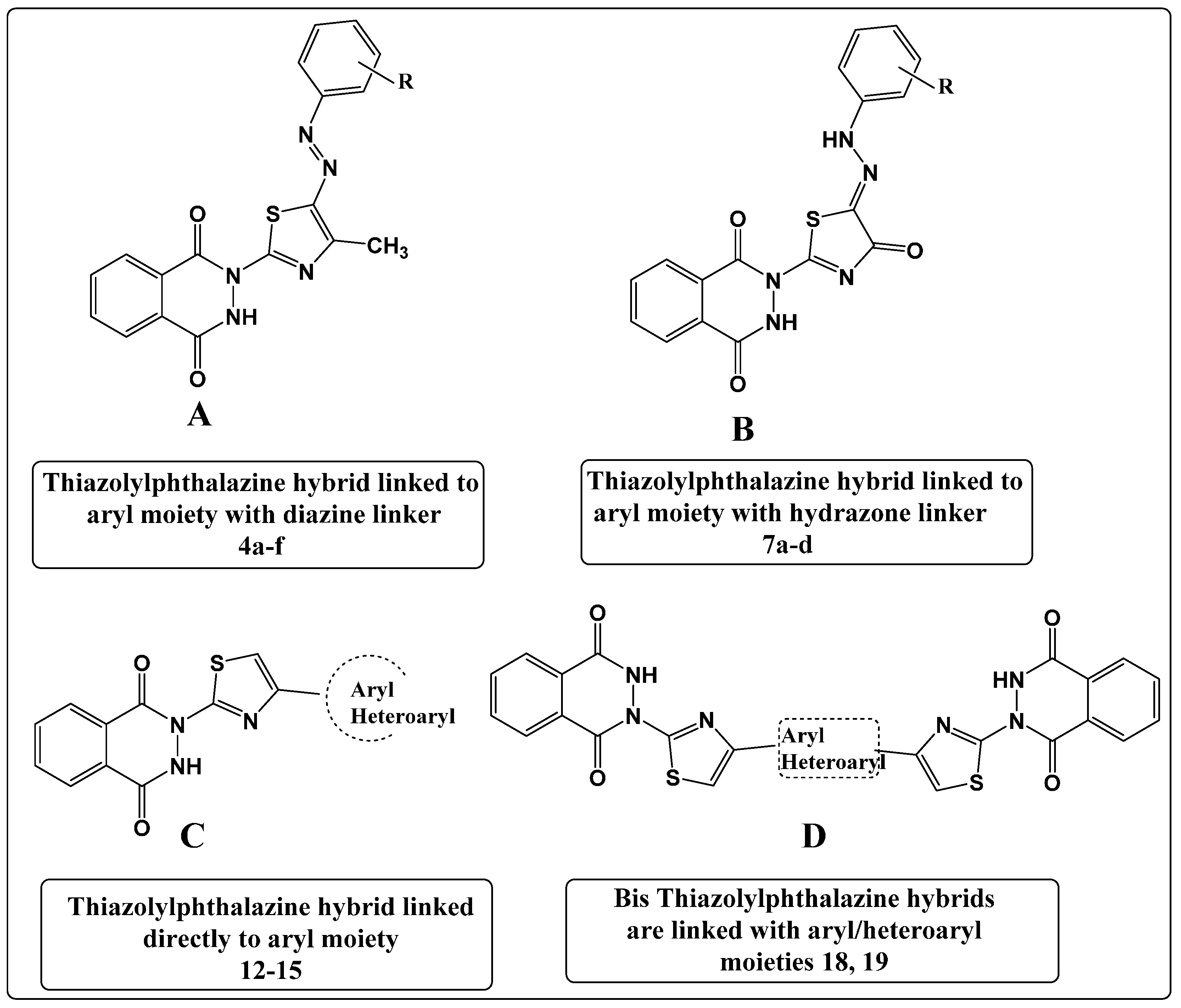
2. Results and Discussion
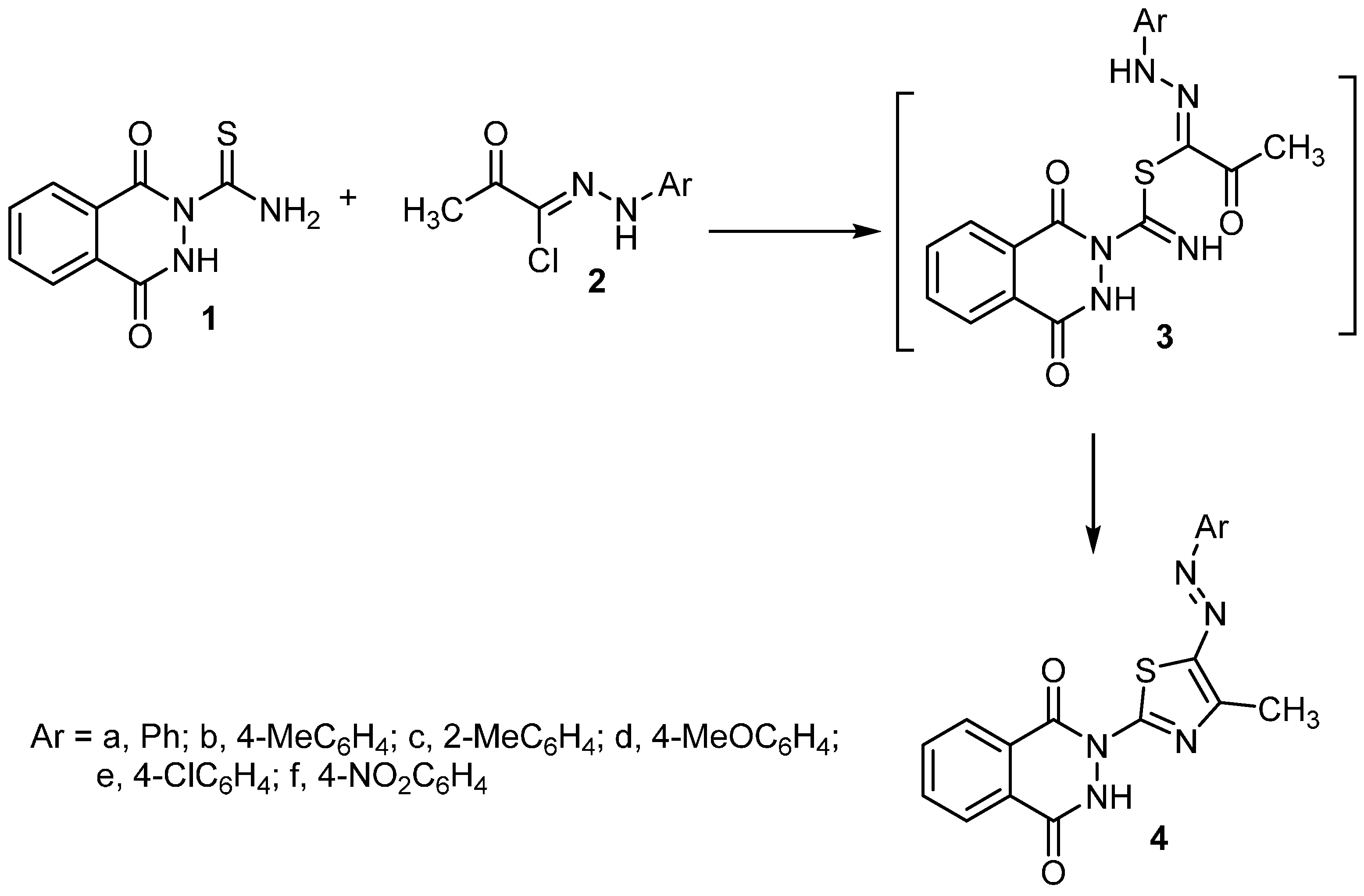
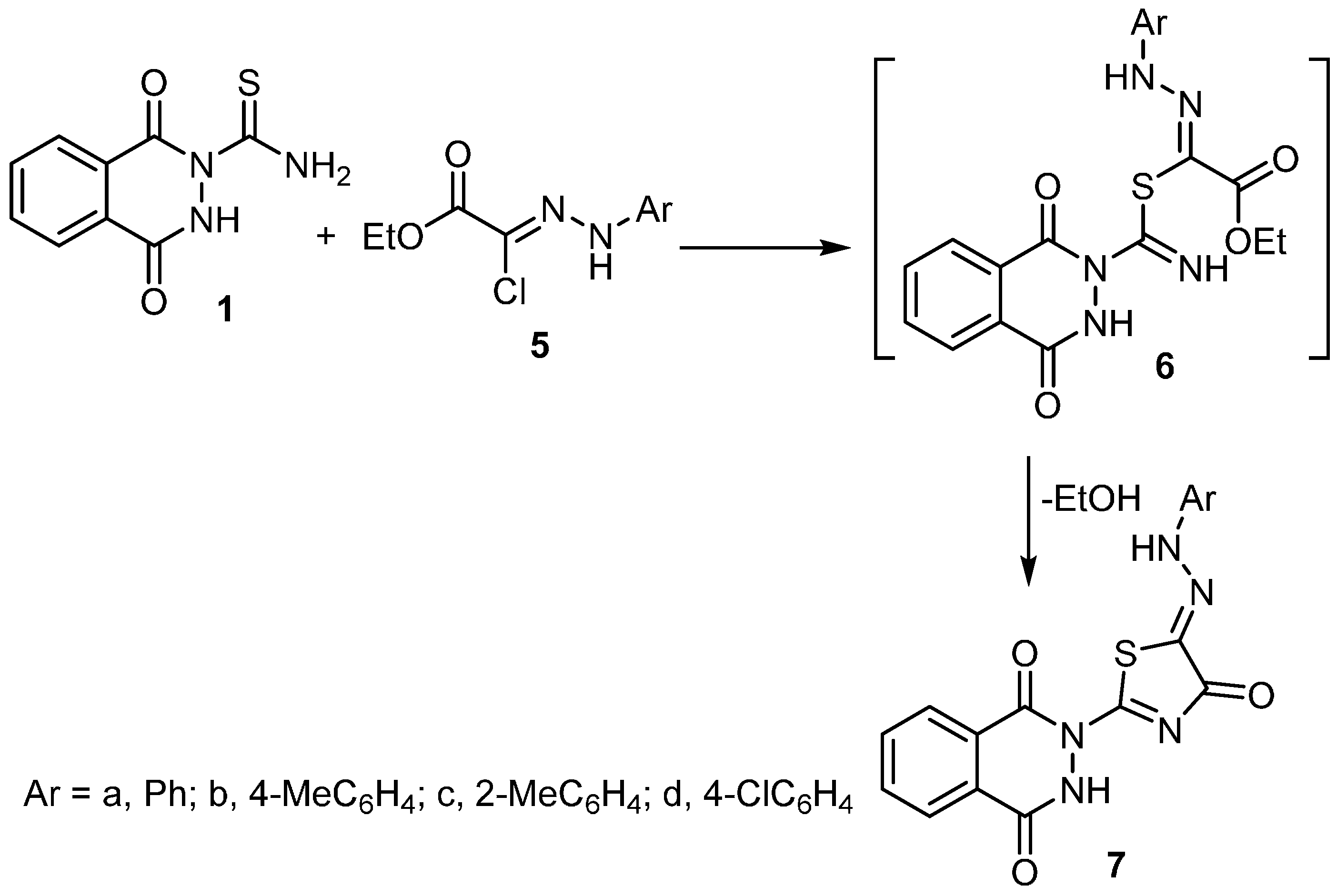
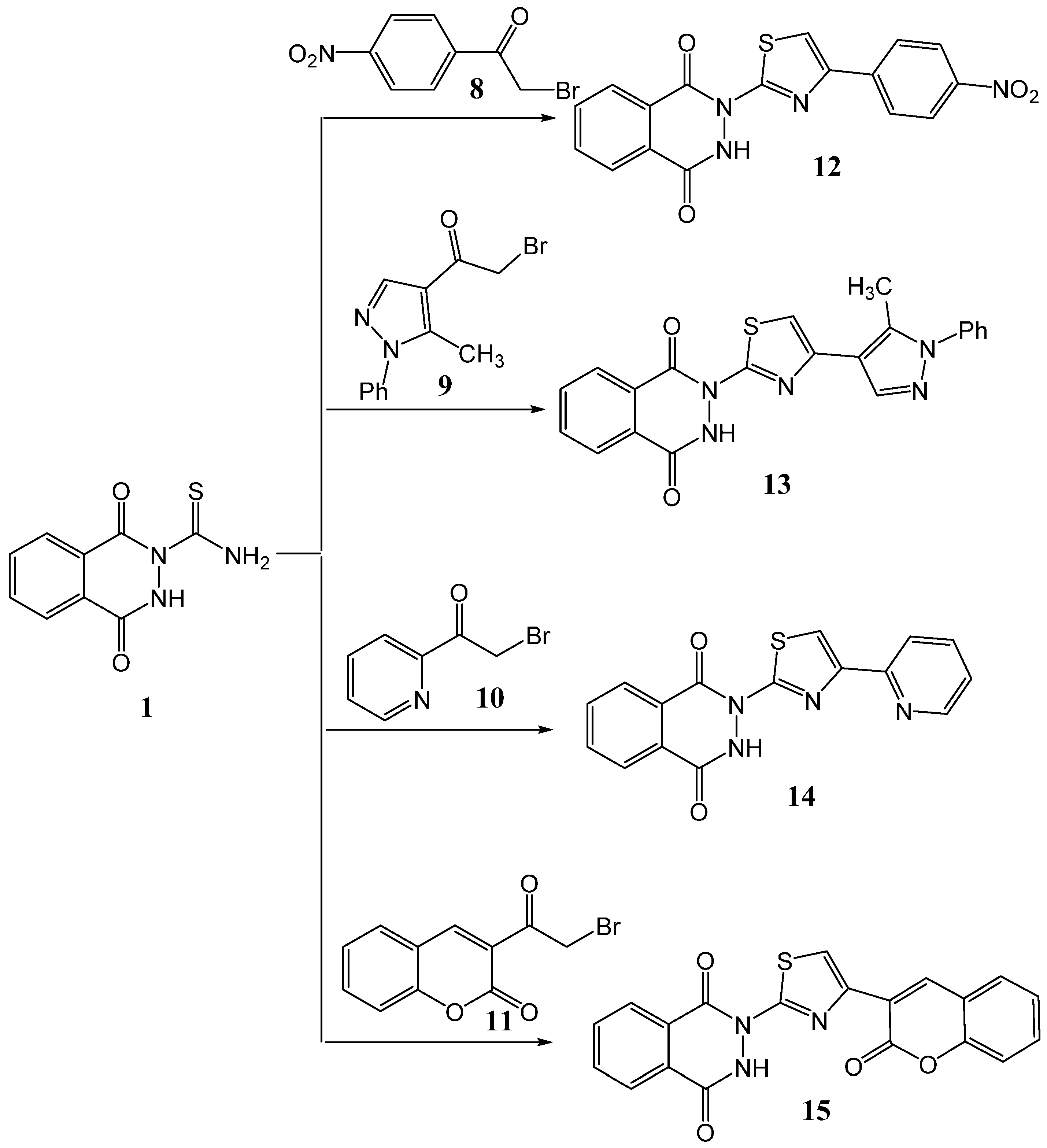
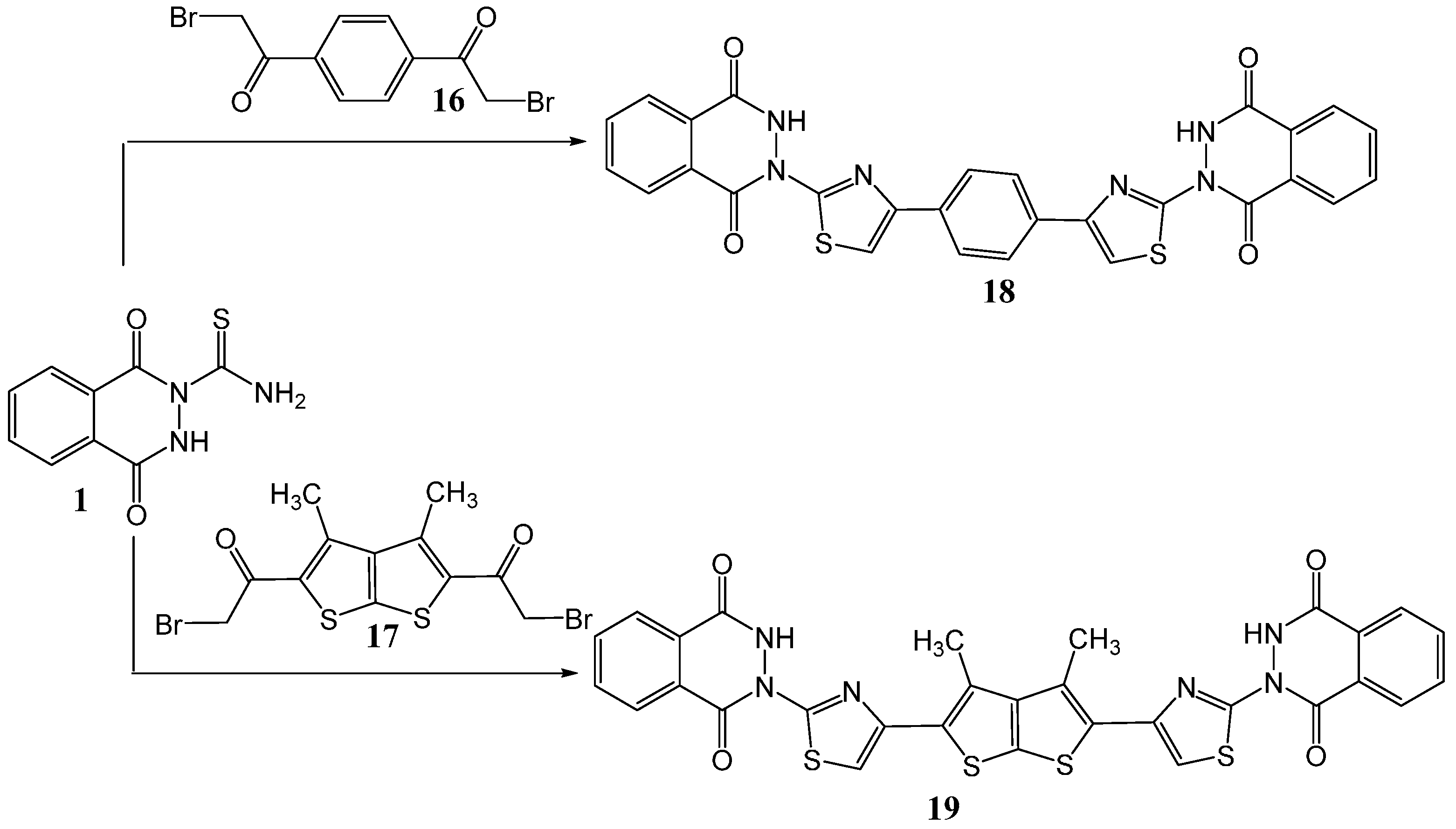
2.1. Synthesis
2.2. Antimicrobial Activity
2.2.1. Antibacterial Activity
2.2.2. Antifungal Activity
2.2.3. Structure-Activity Relationships (SAR)
3. Materials and Methods
3.1. General Experimental Procedures
3.2. Synthesis of Thiazole Derivatives 4a–f and 7a–d
3.2.1. Method A
3.2.2. Method B
3.3. Synthesis of Thiazole Derivatives 12–15
3.3.1. Method A
3.3.2. Method B
3.4. Synthesis of Bis-thiazole Derivatives 18 and 19
3.4.1. Method A
3.4.2. Method B
3.5. Agar Diffusion Medium Biological Assays
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cho, J.Y.; Kwon, H.C.; Williams, P.G.; Jensen, P.R.; Fenical, W. Azamerone, a Terpenoid Phthalazinone from a Marine-Derived Bacterium Related to the Genus Streptomyces (Actinomycetales). Org. Lett. 2006, 8, 2471–2474. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.B.; vander Helm, D. Structure of 6-azido-tetrazolo[5,1-a]phthalazine, C8H4N8, isolated from the toxic dinoflaggelate Gymnodiniumbreve. Acta Crystallogr. C 1985, 41, 1199–1202. [Google Scholar] [CrossRef]
- Quan, Z.S.; Sun, X.Y.; Guan, L.P. Synthesis and anticonvulsant activity of 6-alkoxy-[1,2,4] triazolo[3,4-a]phthalazine. Chem. Biol. Drug Des. 2009, 73, 313–319. [Google Scholar]
- Gong, P.; Jing-Xion, X.; Yan-Fang, Z.; Juan, L. Synthesis and anticancer activity of novel 1,4-disubstitutedphthalazines. Molecules 2006, 11, 574–582. [Google Scholar]
- Gong, P.; Zhang, Y.B.; He, L.; Zhai, X. Synthesis and invitro cytotoxicity of novel 1,4-disubstituted phthalazines. Chin. Chem. Lett. 2008, 19, 29–32. [Google Scholar]
- Cardia, M.C.; Distinto, S.; Maccioni, E.; Plumitallo, A.; Sanna, L.; Sanna, M.L.; Vigo, S. Synthesis and characterization of new phthalhydrazothiazole derivatives: A preliminary investigation on their activity against hepatocellular carcinoma. J. Heterocycl. Chem. 2009, 46, 674–679. [Google Scholar] [CrossRef]
- Mosaad, S.M.; Goudah, A.; Abotaleb, N.A. Synthesis and anti-inflammatory evaluation of some condensed [4-(3,4-dimethylphenyl)-1(2H)-oxo-phthalazin-2-yl]aceticacidhydrazide. Eur. J. Med. Chem. 2010, 45, 1267–1277. [Google Scholar]
- Awadallah, F.M.; Saleh, D.O.; El-Eraky, W. Synthesis vasorelaxant activity, and molecular modeling study of some new phthalazine derivatives. Eur. J. Med. Chem. 2012, 52, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, M.; Kamei, K.; Koga, T.; Akima, M.; Kuroki, T.; Ohi, N. Novel antiasthmatic agents with dual activities of thromboxane A2 synthetase inhibition and bronchodilation. 1.2-[2-(1-imidazolyl)alkyl]-1(2H)-phthalazinones. J. Med. Chem. 1993, 36, 4052–4060. [Google Scholar] [CrossRef] [PubMed]
- Eguchi, Y.; Sasaki, F.; Takashima, Y.; Nakajima, M.; Ishikawa, M. Studies on Antiatherosclerotic Agents. Synthesis of 7-Ethoxycarbonyl-4-formyl-6,8-dimethyl-1(2H)-phthalazinone Derivatives and Related Compounds. Chem. Pharm. Bull. 1991, 39, 795–797. [Google Scholar] [CrossRef] [PubMed]
- Cherkez, S.; Herzig, J.; Yellin, H. Synthesis, saludiuretic, and antihypertensive activity of 6,7-disubstituted-1(2H)-and3,4-dihydro-1(2H)-phthalazinones. J. Med. Chem. 1986, 29, 947–959. [Google Scholar] [CrossRef] [PubMed]
- Salvi, V.K.; Bhambi, D.; Jat, J.L.; Talesara, G.L. Synthesis and antimicrobial activity of some 2-[1-(4-oxo-3,4-dihydrophthalazine-1-yl)alkyl]-1H-isoindole-1,3(2H)-dione and their imidoxy derivatives. Arkivoc 2006, 14, 133–140. [Google Scholar]
- Derita, M.; del Olmo, E.; Barboza, B.; García-Cadenas, A.E.; López-Pérez, J.L.; Andújar, S.; Enriz, D.; Zacchino, S.; Feliciano, A.S. Synthesis, Bioevaluation and Structural Study of Substituted Phthalazin-1(2H)-ones Acting as Antifungal Agents. Molecules 2013, 18, 3479–3501. [Google Scholar] [CrossRef]
- AbdEl-Wahab, A.H.F.; Mohamed, H.M.; El-Agrody, A.M.; El-Nassag, M.A.; Bedair, A.H. Synthesis and Biological Screening of 4-Benzyl-2H-phthalazine Derivatives. Pharmaceuticals 2011, 4, 1158–1170. [Google Scholar] [CrossRef]
- El-Hashash, M.A.; Guirguis, D.B.; AbdEl-Wahed, N.A.M.; Kadhim, M.A. Synthesis of Novel Series of Phthalazine Derivatives with Antibacterial and Antifungal Evaluation. J. Chem. Eng. Process Technol. 2014, 5, 195–200. [Google Scholar]
- Shirai, A.; Fumoto, Y.; Shouno, T.; Maseda, H.; Omasa, T. Synthesis and biological activity of thiazolyl-acetic acid derivatives as possible antimicrobial agents. Biocontrol Sci. 2013, 18, 59–73. [Google Scholar] [CrossRef] [PubMed]
- Bretanha, L.C.; Teixeira, V.E.; Ritter, M.; Siqueira, G.M.; Cunico, W.; Pereira, C.M.P.; Freitag, R.A. Ultrasound-promoted synthesis of 3-trichloromethyl-5-alkyl(aryl)-1,2,4-oxadiazoles. Ultrason. Sonochem. 2011, 18, 704–707. [Google Scholar] [CrossRef] [PubMed]
- Naeimi, H.; Didar, A. Efficient sonochemical green reaction of aldehyde, thiobarbituric acid and ammonium acetate using magnetically recyclable nanocatalyst in water. Ultrason. Sonochem. 2017, 34, 881–888. [Google Scholar] [CrossRef] [PubMed]
- Xavier, D.M.; Goldani, B.S.; Seus, N.; Jacob, R.G.; Barcellos, T.; Paixão, M.W.; Luque, R.; Alves, D. Sonochemistry in organocatalytic enamine-azide[3+2]cycloadditions: A rapid alternative for the synthesis of 1,2,3-triazoylcarboxamides. Ultrason. Sonochem. 2017, 34, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Abbas, E.M.H.; Gomha, S.M.; Farghaly, T.A.; Abdalla, M.M. Synthesis of new thiazole derivatives as antitumor agents. Curr. Org. Synth. 2016, 13, 456–465. [Google Scholar] [CrossRef]
- Farghaly, T.A.; Gomha, S.M.; Sayed, A.R.; Khedr, M.A. Hydrazonoyl Halidesas Precursors for Synthesis of Bioactive Thiazole and Thiadiazole Derivatives: Synthesis, Molecular Docking and Pharmacological Study. Curr. Org. Synth. 2016, 13, 445–455. [Google Scholar] [CrossRef]
- Gomha, S.M.; Riyadh, S.M.; Abdalla, M.M. Solvent-drop grinding method: efficient synthesis, DPPH radical scavenging and anti-diabetic activities of chalcones, bis-chalcones, azolines, and bis-azolines. Curr. Org. Synth. 2015, 12, 220–228. [Google Scholar] [CrossRef]
- Gomha, S.M.; Salah, T.A.; Abdelhamid, A.O. Synthesis, characterization and pharmacological evaluation of some novel thiadiazoles and thiazoles incorporating pyrazole moiety as potent anticancer agents. Monatsh. Chem. 2015, 146, 149–158. [Google Scholar] [CrossRef]
- Dawood, D.H.; Batran, R.Z.; Farghaly, T.A.; Khedr, M.A.; Abdulla, M.M. New Coumarin Derivatives as Potent Selective COX-2 Inhibitors; Synthesis, Anti-inflammatory, QSAR and Molecular Modeling Studies. Arch. Pharm. Chem. Life Sci. 2015, 875–888. [Google Scholar] [CrossRef] [PubMed]
- Farghaly, T.A.; Abdallah, M.A.; Masaret, G.S.; Muhammad, Z.A. New and efficient approach for synthesis of novel bioactive[1,3,4]thiadiazoles incorporated with 1,3-thiazole moiety. Eur. J. Med. Chem. 2015, 320–333. [Google Scholar] [CrossRef] [PubMed]
- Gomha, S.M.; Edrees, M.M.; Altalbawy, F.M.A. Synthesis and Characterization of Some New Bis-Pyrazolyl-Thiazoles Incorporating the Thiophene Moiety as Potent Anti-Tumor Agents. Int. J. Mol. Sci. 2016, 17, 1499–1509. [Google Scholar] [CrossRef] [PubMed]
- Högberg, L.D.; Heddini, A.; Cars, O. The global need for effective antibiotics: Challenges and recent advances. Trends Pharmacol. Sci. 2010, 31, 509–515. [Google Scholar] [CrossRef] [PubMed]
- Farghaly, T.A.; Hassaneen, H.M.E.; Elzahabi, H.S.A. Eco-friendly synthesis and 2D-QSAR study of novel pyrazolines as potential anticolon cancer agents. Med. Chem. Res. 2015, 24, 652–668. [Google Scholar] [CrossRef]
- Abbas, E.M.H.; Gomha, S.M.; Farghaly, T.A. Multicomponent Reactions for Synthesis of Bioactive polyheterocyclic Ring Systems under controlled Microwave irradiation. Arabian J. Chem. 2014, 7, 623–629. [Google Scholar] [CrossRef]
- Gomha, S.M.; Khalil, K.D. A convenient ultrasound-promoted synthesis and cytotoxic activity of some new thiazole derivatives bearing a coumarin nucleus. Molecules 2012, 17, 9335–9347. [Google Scholar] [CrossRef] [PubMed]
- Naock, A.; Schroder, A.; Hartmann, H. Synthesis and spectral characterisation of a series of new heterocyclic triphenyl methane analogues. Dye Pigments 2002, 57, 131–147. [Google Scholar] [CrossRef]
- Sravanthi, T.V.; Pottem, M.R.; Manju, S.L. Design and Synthesis of Bis-Thiazol-2-ylidenes. Asian J. Chem. 2013, 25, 5370–5372. [Google Scholar]
- Martin-Cantalejo, Y.; Saez, B.; Soto, J.; Villa, M.J.; Brana, M.F. A New Convergent Synthesis of 4,4′-Bispyridyl-5,5′-Disubstituted-2,2-Bisoxazolesand–Bisthiazoles. Synthesis 2003, 2211–2215. [Google Scholar] [CrossRef]
- Cruickshank, R.; Duguid, J.P.; Marion, B.P.; Swain, R.H.A. Medicinal Microbiology, 12th ed.; Churchill Livingstone: London, UK, 1975; Volume II, pp. 196–202. [Google Scholar]
- Aggarwal, R.; Kumar, S.; Kaushik, P.; Kaushik, D.; Gupta, G.K. Synthesis and pharmacological evaluation of some novel 2-(5-hydroxy-5-trifluoromethyl-4,5-dihydropyrazol-1-yl)-4-(coumarin-3-yl)thiazoles. Eur. J. Med. Chem. 2013, 62, 508–514. [Google Scholar] [CrossRef] [PubMed]
- Cardia, M.C.; Distinto, S.; Maccioni, E.; Bonsignore, L.; DeLogu, A. Synthesis and biological activity evaluation of differently substituted 1,4-dioxo-3,4-dihydrophthalazine-2(1H)-carboxamides and -carbothioamides. J. Heterocycl. Chem. 2003, 40, 1011–1015. [Google Scholar] [CrossRef]
- Eweiss, N.F.; Osman, A. Synthesis of heterocycles. Part II new routes to acetyl thiadiazolines and alkylazothiazoles. J. Heterocycl. Chem. 1980, 17, 1713–1717. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 4, 7, 12–15, 18 and 19 are available from the authors.
| Entry | Solvent | Temperature (°C) | Time (min) | Yield (%) |
|---|---|---|---|---|
| 1 | EtOH | r.t. | 40 | 90 |
| 2 | EtOH | 50 | 35 | 92 |
| 3 | Dioxane | r.t. | 60 | 82 |
| 4 | Dioxane | 50 | 45 | 87 |
| 5 | DMF | r.t. | 40 | 85 |
| 6 | DMF | 50 | 35 | 85 |
| 7 | Benzene | r.t. | 45 | 89 |
| 8 | Benzene | 50 | 40 | 91 |
| 9 | Toluene | r.t. | 60 | 87 |
| 10 | Toluene | 50 | 50 | 90 |
| Product No. | Conventional | Sonication | ||
|---|---|---|---|---|
| Time (h) | Yield (%) | Time (min) | Yield (%) | |
| 4a | 4 | 68 | 35 | 92 |
| 4b | 4 | 69 | 50 | 84 |
| 4c | 6 | 70 | 50 | 88 |
| 4d | 4 | 71 | 40 | 86 |
| 4e | 4 | 74 | 55 | 88 |
| 4f | 3 | 67 | 45 | 87 |
| 7a | 6 | 65 | 60 | 85 |
| 7b | 5 | 67 | 40 | 83 |
| 7c | 5 | 68 | 50 | 85 |
| 7d | 4 | 69 | 55 | 83 |
| Product No. | Conventional | Sonication | ||
|---|---|---|---|---|
| Time (h) | Yield (%) | Time (min) | Yield (%) | |
| 12 | 5 | 70 | 35 | 84 |
| 13 | 4 | 67 | 50 | 82 |
| 14 | 4 | 72 | 60 | 88 |
| 15 | 6 | 69 | 40 | 84 |
| 18 | 5 | 70 | 55 | 85 |
| 19 | 7 | 68 | 45 | 83 |
| Compd. No. a | Inhibition Zone Diameter in mm and (%) Value | |||||
|---|---|---|---|---|---|---|
| Gram-Positive Bacteria | Gram-Negative Bacteria | Fungi | ||||
| SA | BS | S SP. | EC | AF | CA | |
| 1 | NA | 12 (37%) | 16 (94%) | NA | 8 (34%) | 13 (52%) |
| 4a | 8 (34%) | 11 (34%) | 15 (88%) | 11 (57%) | NA | 11 (44%) |
| 4b | 16 (69%) | 16 (50%) | 15 (88%) | 16 (84%) | 10 (43%) | NA |
| 4c | 11 (47%) | 13 (40%) | 14 (82%) | 13 (68%) | 12 (52%) | 15 (60%) |
| 4d | 10 (43%) | 13 (40%) | 20 (117%) | 10 (52%) | NA | 13 (52%) |
| 4e | 12 (52%) | NA | 9 (52%) | NA | NA | 12 (48%) |
| 4f | 14 (60%) | 12 (37%) | 15 (88%) | 15 (78%) | 12 (52%) | 15 (60%) |
| 7a | NA | 10 (31%) | NA | NA | NA | 10 (40%) |
| 7b | NA | 9 (28%) | 20 (117%) | 9 (47%) | 8 (34%) | 12 (48%) |
| 7c | NA | 12 (37%) | 18 (105%) | 10 (52%) | 9 (39%) | 14 (56%) |
| 7d | NA | NA | NA | NA | NA | NA |
| 12 | NA | 11 (34%) | NA | NA | NA | 11 (44%) |
| 13 | NA | 12 (37%) | NA | NA | NA | NA |
| 14 | 12 (52%) | NA | 12 (70%) | 16 (84%) | 14 (60%) | NA |
| 15 | 14 (60%) | 12 (37%) | 9 (52%) | 9 (47%) | 16 (69%) | NA |
| 18 | 15 (65%) | 15 (46%) | 15 (88%) | 15 (78%) | 15 (65%) | 21 (84%) |
| 19 | 11 (47%) | 11 (34%) | 10 (58%) | NA | 8 (34%) | 15 (60%) |
| AmphotericinB | - | - | - | - | 23 (100%) | 25 (100%) |
| Ampicillin | 23 (100%) | 32 (100%) | - | - | - | - |
| Gentamycin | - | - | 17 (100%) | 19 (100%) | - | - |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elsharabasy, F.S.; Gomha, S.M.; Farghaly, T.A.; Elzahabi, H.S.A. An Efficient Synthesis of Novel Bioactive Thiazolyl-Phthalazinediones under Ultrasound Irradiation. Molecules 2017, 22, 319. https://doi.org/10.3390/molecules22020319
Elsharabasy FS, Gomha SM, Farghaly TA, Elzahabi HSA. An Efficient Synthesis of Novel Bioactive Thiazolyl-Phthalazinediones under Ultrasound Irradiation. Molecules. 2017; 22(2):319. https://doi.org/10.3390/molecules22020319
Chicago/Turabian StyleElsharabasy, Fatma S., Sobhi M. Gomha, Thoraya A. Farghaly, and Heba S. A. Elzahabi. 2017. "An Efficient Synthesis of Novel Bioactive Thiazolyl-Phthalazinediones under Ultrasound Irradiation" Molecules 22, no. 2: 319. https://doi.org/10.3390/molecules22020319
APA StyleElsharabasy, F. S., Gomha, S. M., Farghaly, T. A., & Elzahabi, H. S. A. (2017). An Efficient Synthesis of Novel Bioactive Thiazolyl-Phthalazinediones under Ultrasound Irradiation. Molecules, 22(2), 319. https://doi.org/10.3390/molecules22020319







