A Study on the Electro-Optical Properties of Thiol-Ene Polymer Dispersed Cholesteric Liquid Crystal (PDChLC) Films
Abstract
:1. Introduction
2. Results and Discussions
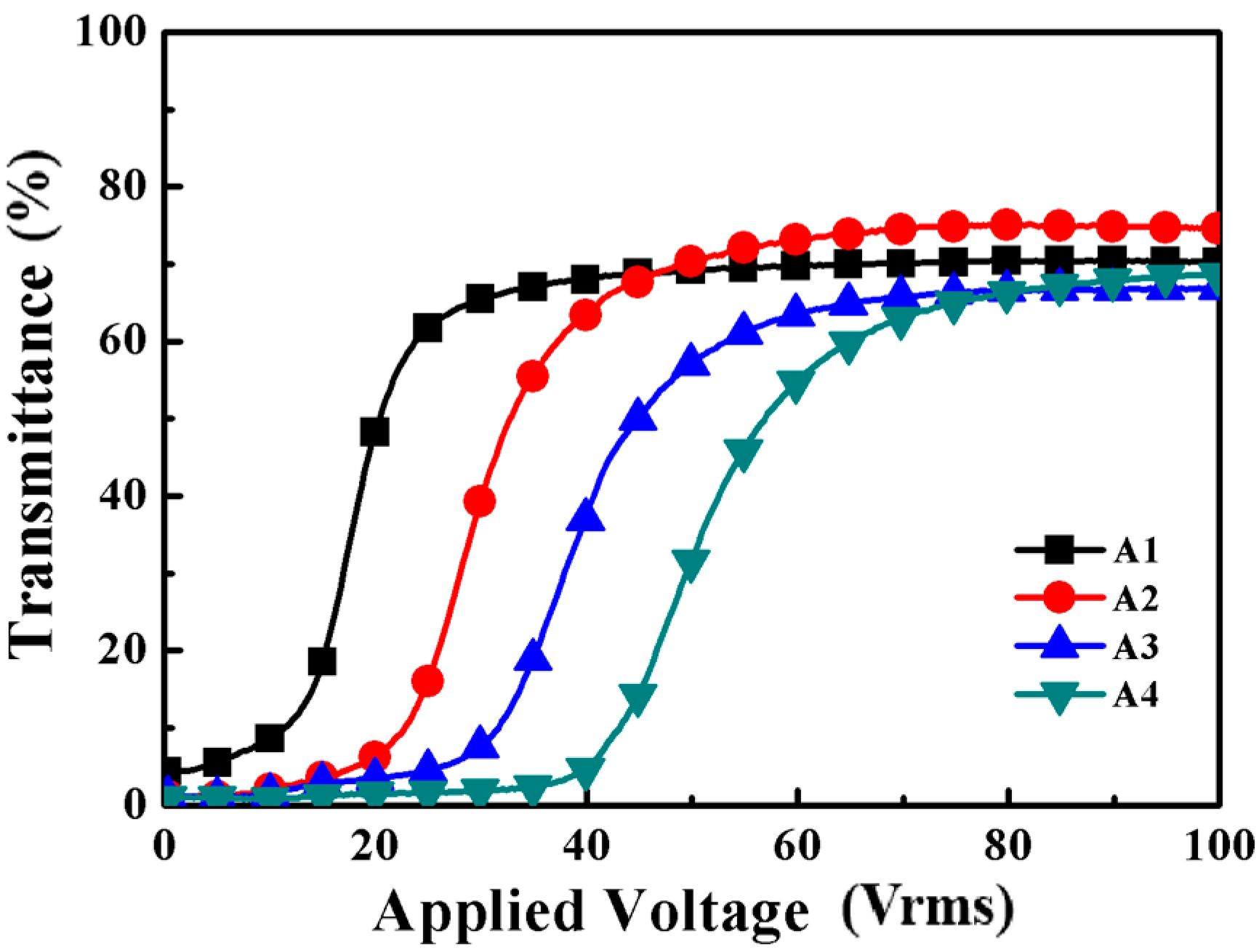
2.1. The Effects of Different Amounts of Chiral Dopant on the PDChLC Films
2.1.1. Morphology of the Polymer Network in PDChLC Films
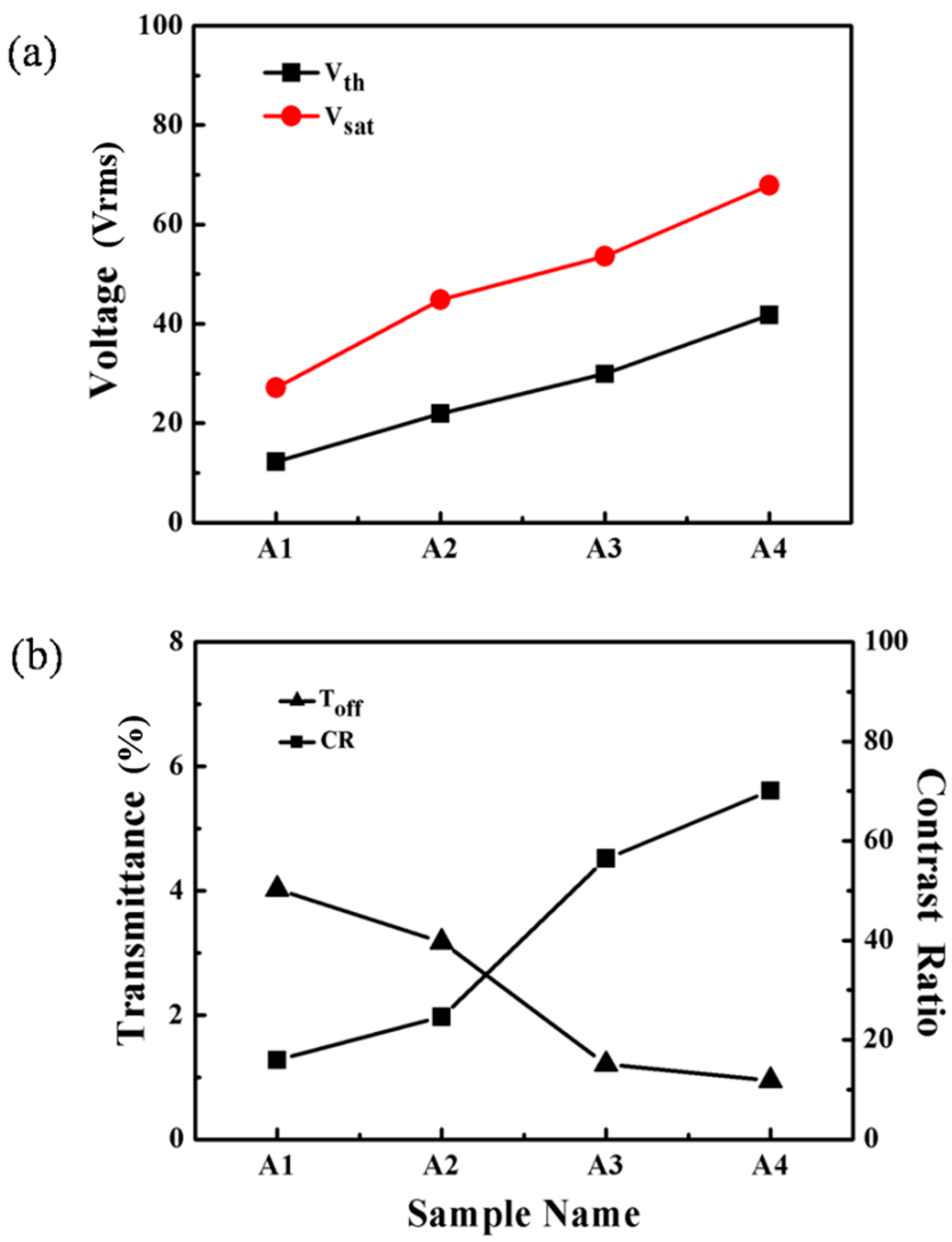
2.1.2. Electro-Optical Properties of PDChLC Films
2.2. The Thiol Functionality Effects in PDChLC Films
2.2.1. Morphology of the Polymer Network in PDChLC Films
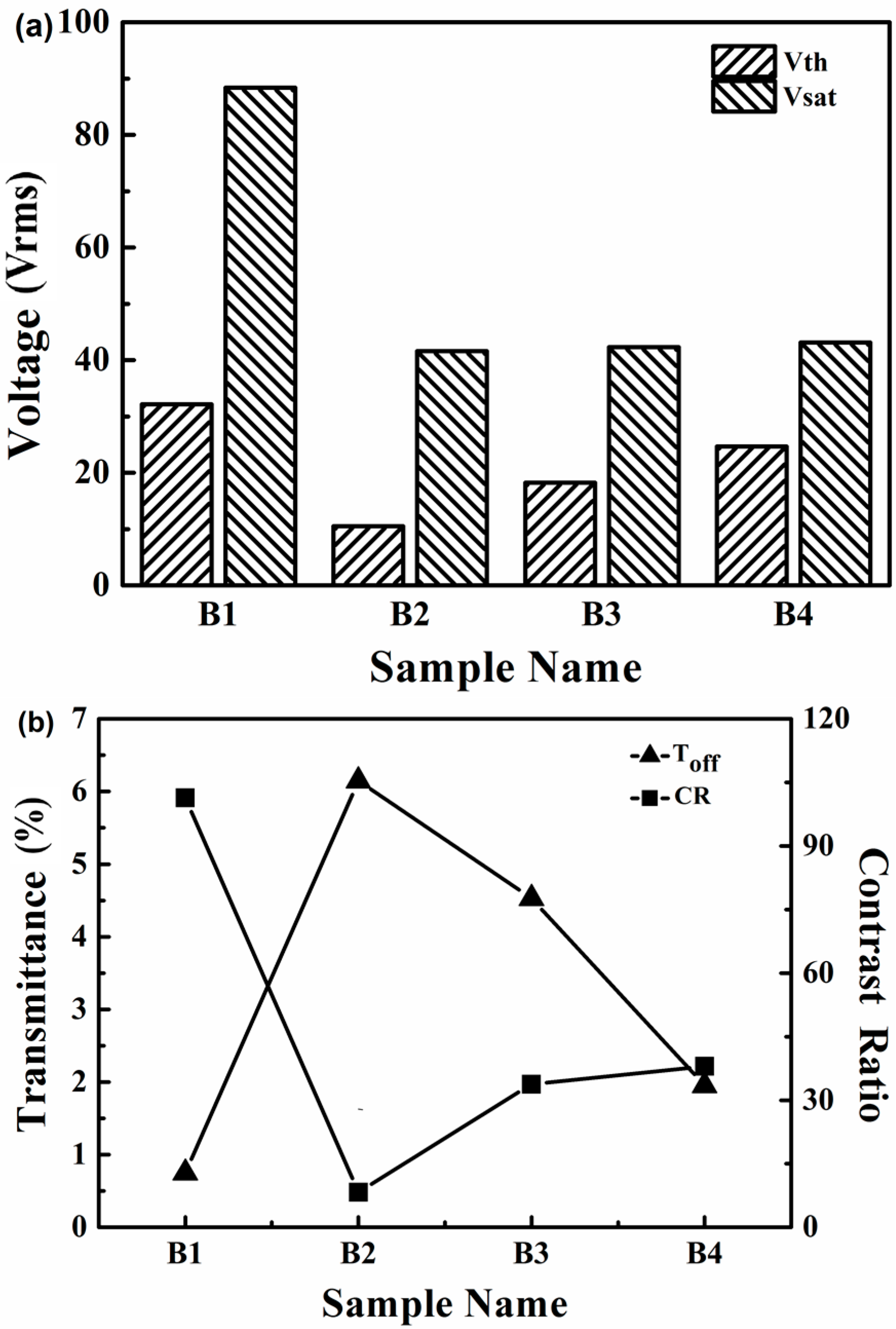
2.2.2. Electro-Optical Properties of PDChLC Films
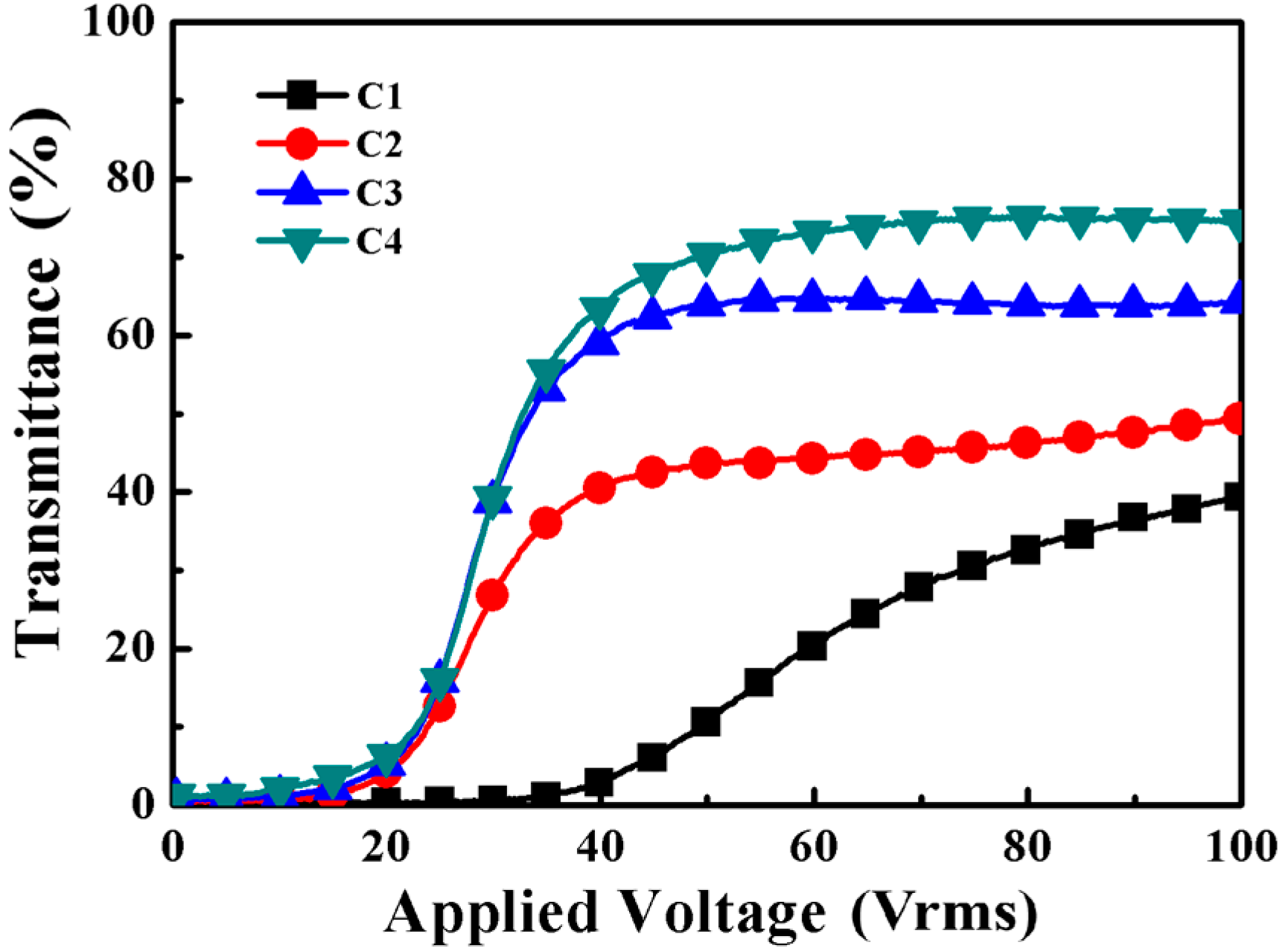
2.3. The Effects of Different Amounts of Thiol on the PDChLC Films
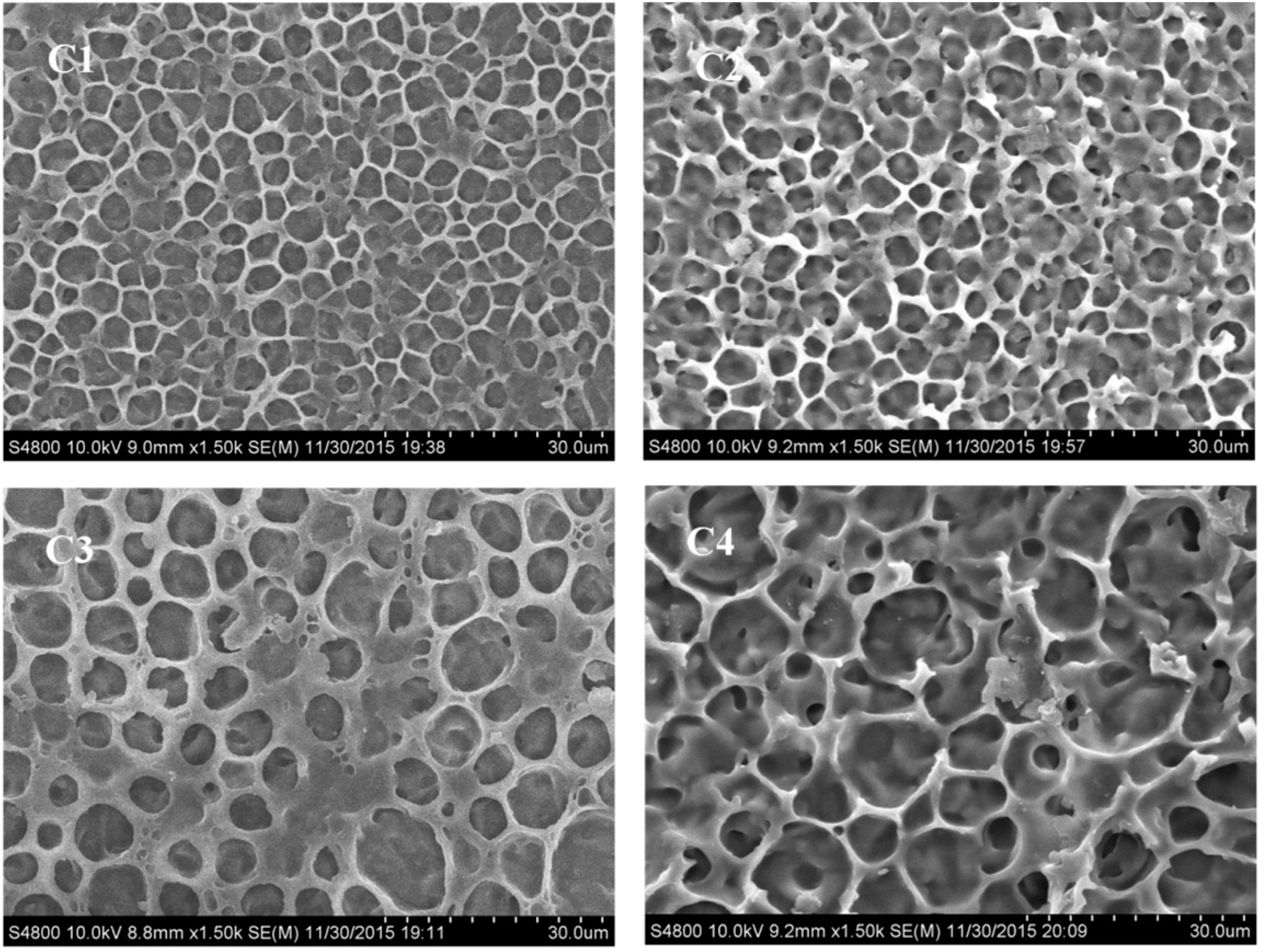
2.3.1. Morphology of the Polymer Network in PDChLC Films
2.3.2. Electro-Optical Properties of PDChLC Films
3. Materials and Methods
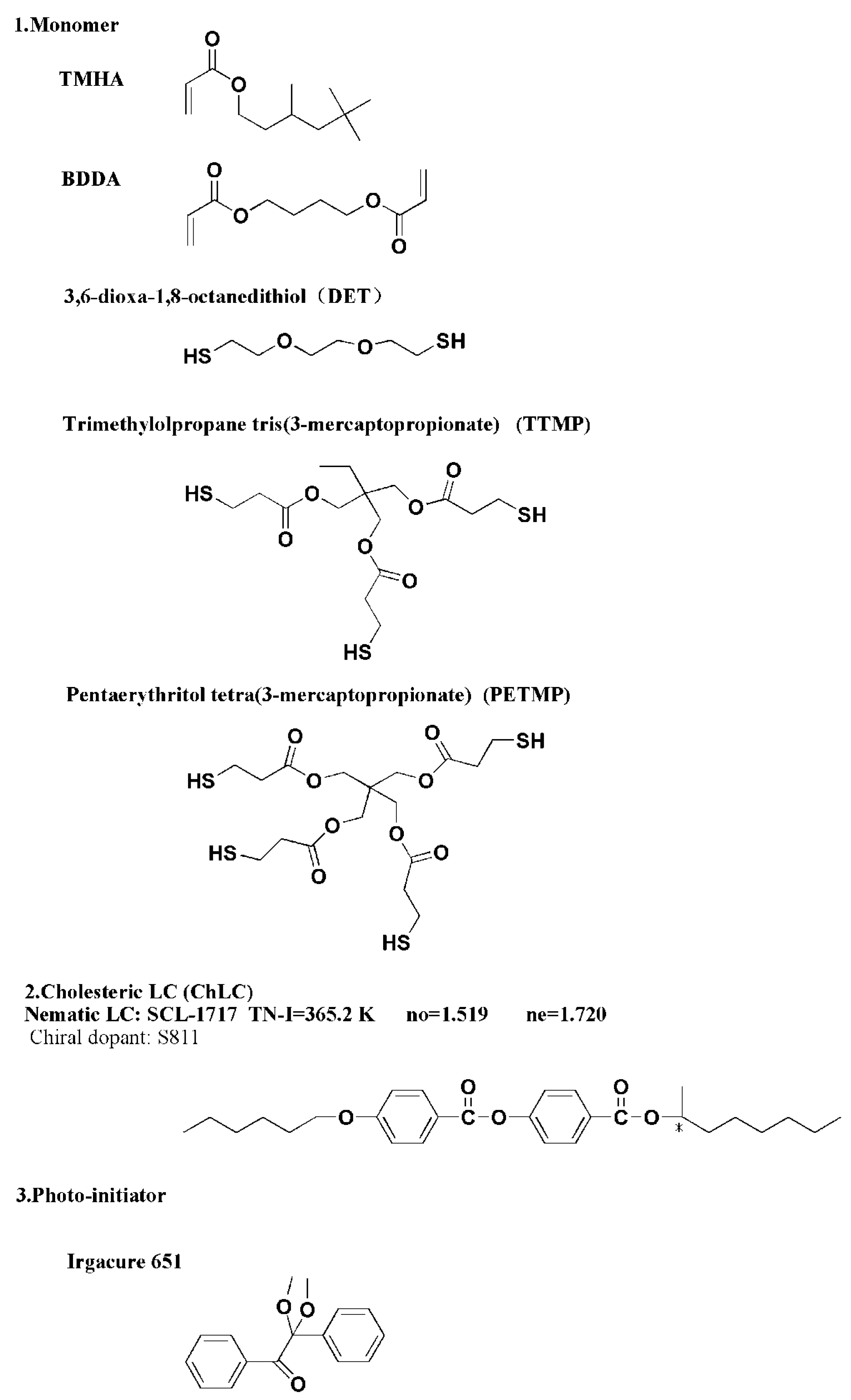
3.1. Materials
3.2. Sample Preparation
3.3. Measurements
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Masutani, A.; Roberts, T.; Schüller, B.; Hollfelder, N.; Kilickiran, P. Improvement of dichroic polymer dispersed liquid crystal performance using lift-off technique. Appl. Phys. Lett. 2006, 89, 183514. [Google Scholar] [CrossRef]
- Li, W.B.; Yu, L.L.; He, W.L.; Yuan, X.; Zhao, D.; Huang, W.; Cao, H.; Yang, Z.; Yang, H. Effect of a Photopolymerizable Monomer Containing a Hydrogen Bond on Near-Infrared Radiation Transmittance of Nematic Liquid Crystal/Monomers Composites. J. Phys. Chem. C 2008, 112, 13739–13743. [Google Scholar] [CrossRef]
- Doane, J.W.; Golemme, A.; West, J.; Whitehead, J.B., Jr. Polymer dispersed liquid crystals for display application. Mol. Cryst. Liq. Cryst. 1988, 165, 511–532. [Google Scholar] [CrossRef]
- Jain, S.C.; Thakur, R.S. Thermo-electro-optic switch based on polymer dispersed liquid crystal composite. Appl. Phys. Lett. 1992, 61, 1641–1642. [Google Scholar] [CrossRef]
- Drzaic, P.S. Polymer dispersed nematic liquid crystal for large area displays and light valves. J. Appl. Phys. 1986, 60, 2142–2148. [Google Scholar] [CrossRef]
- Kurihara, S.; Masumoto, K.; Nonaka, T. Optical shutter driven photochemically from anisotropic polymer network containing liquid crystalline and azobenzene molecules. Appl. Phys. Lett. 1998, 73, 160–162. [Google Scholar] [CrossRef]
- Doane, J.W.; Vaz, N.A.; Wu, B.G.; Žumer, S. Field controlled light scattering from nematic microdroplets. Appl. Phys. Lett. 1986, 48, 269–271. [Google Scholar] [CrossRef]
- Cheng, Z.; Wang, T.; Li, X.; Zhang, Y.; Yu, H.F. NIR-Vis-UV Light-Responsive Actuator Films of Polymer-Dispersed Liquid Crystal/Graphene Oxide Nanocomposites. ACS Appl. Mater. Interfaces 2015, 7, 27494–27501. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Cheng, Z.; Dong, Z.; Zhang, Y.; Yu, H.F. Photomechanical Response of Polymer-Dispersed Liquid Crystals/Graphene Oxide Nanocomposites. J. Mater. Chem. C 2014, 2, 8501–8506. [Google Scholar] [CrossRef]
- Ren, H.; Chen, D.; Shi, Y.; Yu, H.F.; Fu, Z. Multi-Responsive Fluorescence of Amphiphilic Diblock Copolymer Containing Carboxylate Azobenzene and N-isopropylacrylamide. Polymer 2016, 97, 533–542. [Google Scholar] [CrossRef]
- Yu, L.; Yu, H.F. Light-Powered Tumbler Movement of Graphene Oxide/Polymer Nanocomposites. ACS Appl. Mater. Interfaces 2015, 7, 3834–3839. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.F.; Liu, H.; Kobayashi, T. Fabrication and Photoresponse of Supramolecular Liquid-Crystalline Microparticles. ACS Appl. Mater. Interfaces 2011, 3, 1333–1340. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Heo, C.P.; Park, K.S.; Kim, B.K. Effect of prepolymer structure on the electro-optic performance of polymer dispersed liquid crystals. Polym. Int. 1998, 46, 143–149. [Google Scholar] [CrossRef]
- Malik, P.; Raina, K.K. Droplet orientation and optical properties of polymer dispersed liquid crystal composite films. Opt. Mater. 2004, 27, 613–617. [Google Scholar] [CrossRef]
- Tao, H.J.; Zhang, J.; Wang, X.L.; Gao, J.L. Phase separation and polymer crystallization in a poly(4-methyl-1-pentene)-dioctylsebacate-dimethylphthalate system via thermally induced phase separation. J. Polym. Sci. B Polym. Phys. 2007, 45, 153–161. [Google Scholar] [CrossRef]
- Liu, F.; Cao, H.; Mao, Q.; Song, P.; Yang, H. Effects of monomer structure on the morphology of polymer networks and the electro-optical properties of polymer-dispersed liquid crystal films. Liq. Cryst. 2012, 39, 419–424. [Google Scholar] [CrossRef]
- Schulte, M.D.; Clarson, S.J.; Natarajan, L.V.; Tomlin, D.W.; Bunning, T.J. The effect of fluorine-substituted acrylate monomers on the electro-optical and morphological properties of polymer dispersed liquid crystals. Liq. Cryst. 2000, 27, 467–475. [Google Scholar] [CrossRef]
- Senyurt, A.F.; Warren, G.; Whitehead, J.B.; Hoyle, C.E. Matrix physical structure effect on the electro-optic characteristics of thiol-ene based H-PDLC films. Polymer 2006, 47, 2741–2749. [Google Scholar] [CrossRef]
- White, T.J.; Natarajan, L.V.; Tondiglia, V.P.; Bunning, T.J.; Guymon, C.A. Polymerization Kinetics and Monomer Functionality Effects in Thiol-Ene Polymer Dispersed Liquid Crystals. Macromolecules 2007, 40, 1112–1120. [Google Scholar] [CrossRef]
- Hoyle, C.E.; Lee, T.Y.; Roper, T. Thiol–enes: Chemistry of the past with promise for the future. J. Polym. Sci. A Polym. Chem. 2004, 42, 5301–5338. [Google Scholar] [CrossRef]
- Morgan, C.R.; Magnotta, F.; Ketley, A.D. Thiol/ene photo-curable polymers. J. Polym. Sci. A Polym. Chem. 1977, 15, 627–645. [Google Scholar] [CrossRef]
- Cramer, N.B.; Scott, J.P.; Bowman, C.N. Photopolymerizations of thiol-ene polymers without photoinitiators. Macromolecules 2002, 35, 5361–5365. [Google Scholar] [CrossRef]
- Szczepanski, C.R.; Pfeifer, C.S.; Stansbury, J.W. A new approach to network heterogeneity: Polymerization induced phase separation in photo-initiated, free-radical methacrylic systems. Polymer 2012, 53, 4694–4701. [Google Scholar] [CrossRef] [PubMed]
- Kade, M.J.; Burke, D.J.; Hawker, C.J. The power of thiol-ene chemistry. J. Polym. Sci. A Polym. Chem. 2010, 48, 743–750. [Google Scholar] [CrossRef]
- Northrop, B.H.; Coffey, R.N. Thiol–ene cick chemistry: Computational and kinetic analysis of the influence of alkene functionality. J. Am. Chem. Soc. 2012, 134, 13804–13817. [Google Scholar] [CrossRef] [PubMed]
- Seung, W.O.; Jong, M.B.; Joon, H.; Tae, H.Y. Dye-doped cholesteric liquid crystal light shutter with a polymer dispersed liquid crystal film. Dyes Pigments 2016, 134, 36–40. [Google Scholar]
- Liu, J.; Ozga, K.; Liu, X.; Zhen, Z.; Plucinski, K.J.; Szota, M.; Kityk, I.V. The Relationship between the Monomer Chain Length and the Electro-Optical Properties of Polymer Dispersed Liquid Crystals. Arch. Metall. Mater. 2016, 61, 457–460. [Google Scholar] [CrossRef]
- Liu, J.L.; Liu, X.H.; Zhen, Z. Effects of chiral additives on the electro-optical properties of polymer dispersed liquid crystal. Mater. Lett. 2016, 163, 142–145. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, Y.; Cao, M.; Song, X.; Che, Y.; Zhang, H.; Zhang, H.; Yao, J. Platinum-scatterer-based random lasers from dye-doped polymer-dispersed liquid crystals in capillary tubes. Appl. Opt. 2016, 55, 5702–5706. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.G.; Erdmann, J.H.; Doane, J.W. Response times and voltages for PDLC light shutters. Liq. Cryst. 1989, 5, 1453–1465. [Google Scholar] [CrossRef]
- Drzaic, P.S. Reorientation dynamics of polymer dispersed nematic liquid crystal films. Liq. Cryst. 1988, 3, 1543–1558. [Google Scholar] [CrossRef]
- Maschke, U.; Coquerent, X.; Benmouna, M. Electro-Optical Properties of Polymer–Dispersed Liquid Crystals. Macromol. Rapid Commun. 2002, 23, 159–170. [Google Scholar] [CrossRef]
- Jain, S.C.; Rout, D.K. Electro–optic response of polymer dispersed liquid crystal films. J. Appl. Phys. 1991, 70, 6988–6992. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds are not available from the authors.





| Sample | Monomers | SLC1717/S811 (wt %) | |
|---|---|---|---|
| Composition of Monomers | Weight Ratio | ||
| Group A | |||
| A1 | TMHA/BDDA/PETMP | 10.4/2.6/7.0 | 78.0/2.0 |
| A2 | TMHA/BDDA/PETMP | 10.4/2.6/7.0 | 77.0/3.0 |
| A3 | TMHA/BDDA/PETMP | 10.4/2.6/7.0 | 76.0/4.0 |
| A4 | TMHA/BDDA/PETMP | 10.4/2.6/7.0 | 75.0/5.0 |
| Group B | |||
| B1 | TMHA/BDDA | 16.0/4.0 | 77.0/3.0 |
| B2 | TMHA/BDDA/DET | 12.0/3.0/5.0 | 77.0/3/0 |
| B3 | TMHA/BDDA/TTMP | 12.0/3.0/5.0 | 77.0/3.0 |
| B4 | TMHA/BDDA/PETMP | 12.0/3.0/5.0 | 77.0/3.0 |
| Group C | |||
| C1 | TMHA/BDDA/PETMP | 15.2/3.8/1.0 | 77.0/3.0 |
| C2 | TMHA/BDDA/PETMP | 13.6/3.4/3.0 | 77.0/3.0 |
| C3 | TMHA/BDDA/PETMP | 12.0/3.0/5.0 | 77.0/3.0 |
| C4 | TMHA/BDDA/PETMP | 10.4/2.6/7.0 | 77.0/3.0 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, Y.; Gao, Y.; Zhou, L.; Huang, J.; Fang, H.; Ma, H.; Zhang, Y.; Yang, J.; Song, P.; Zhang, C.; et al. A Study on the Electro-Optical Properties of Thiol-Ene Polymer Dispersed Cholesteric Liquid Crystal (PDChLC) Films. Molecules 2017, 22, 317. https://doi.org/10.3390/molecules22020317
Sun Y, Gao Y, Zhou L, Huang J, Fang H, Ma H, Zhang Y, Yang J, Song P, Zhang C, et al. A Study on the Electro-Optical Properties of Thiol-Ene Polymer Dispersed Cholesteric Liquid Crystal (PDChLC) Films. Molecules. 2017; 22(2):317. https://doi.org/10.3390/molecules22020317
Chicago/Turabian StyleSun, Yujian, Yanzi Gao, Le Zhou, Jianhua Huang, Hua Fang, Haipeng Ma, Yi Zhang, Jie Yang, Ping Song, Cuihong Zhang, and et al. 2017. "A Study on the Electro-Optical Properties of Thiol-Ene Polymer Dispersed Cholesteric Liquid Crystal (PDChLC) Films" Molecules 22, no. 2: 317. https://doi.org/10.3390/molecules22020317
APA StyleSun, Y., Gao, Y., Zhou, L., Huang, J., Fang, H., Ma, H., Zhang, Y., Yang, J., Song, P., Zhang, C., Zhang, L., Li, F., Zhao, Y., & Li, K. (2017). A Study on the Electro-Optical Properties of Thiol-Ene Polymer Dispersed Cholesteric Liquid Crystal (PDChLC) Films. Molecules, 22(2), 317. https://doi.org/10.3390/molecules22020317








