Actions of Quercetin, a Polyphenol, on Blood Pressure
Abstract
:1. Introduction
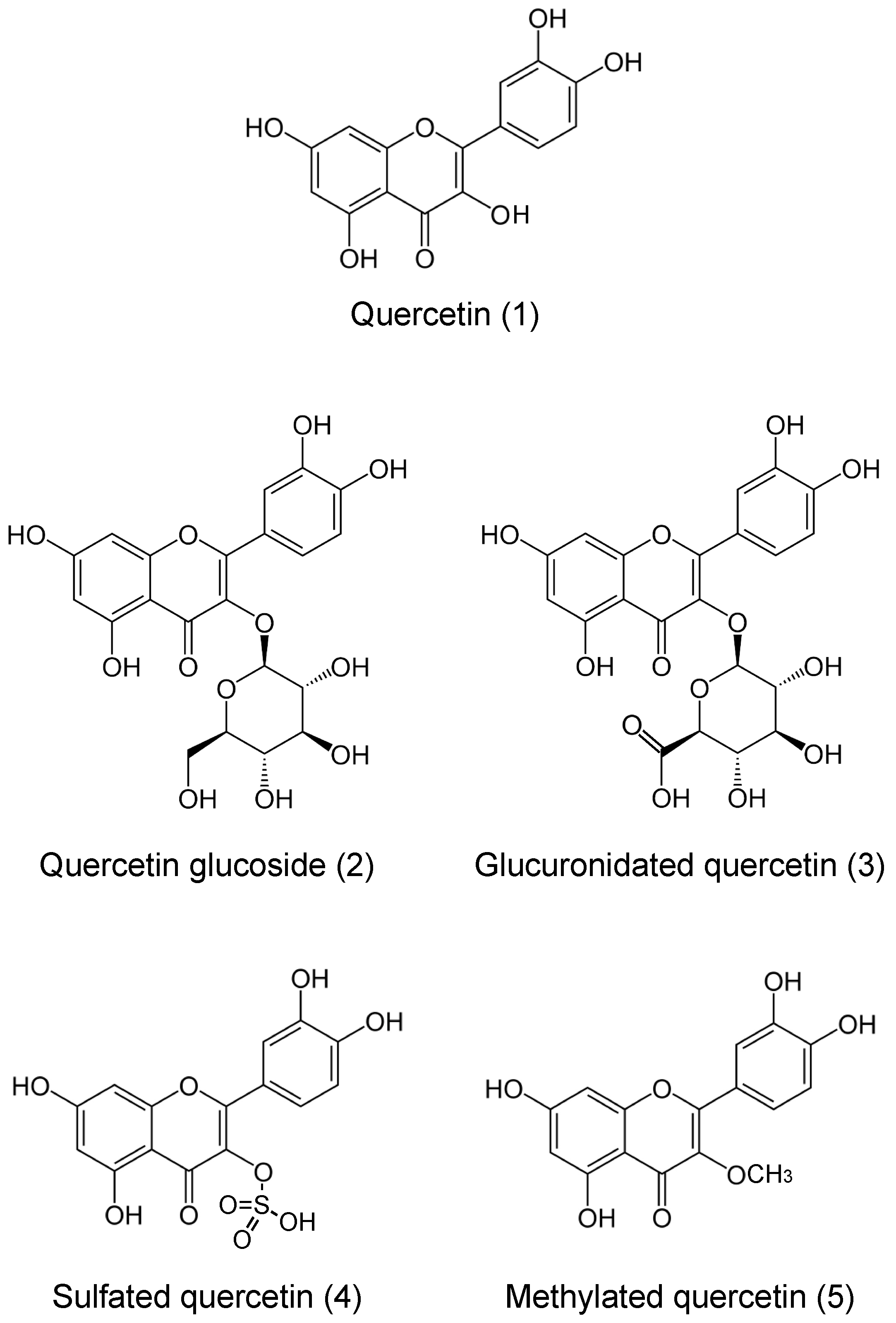
2. How Do We Take Up and Absorb Flavonoids into the Inside of the Body?
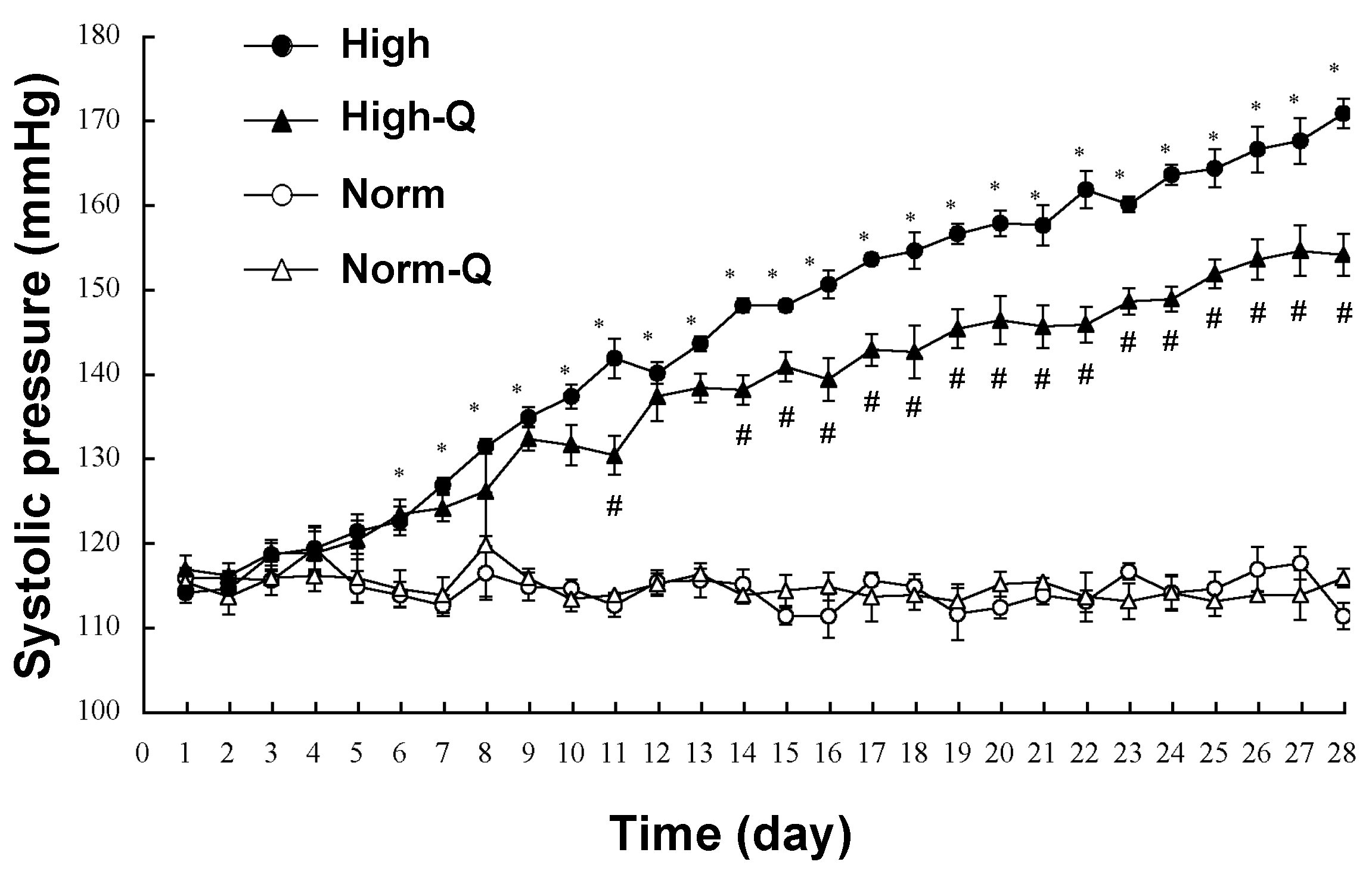
3. Actions of Quercetin on Blood Pressure in Human and Model Animals
4. Anti-Inflammatory and Anti-Oxidant Actions of Quercetin and Its Influence on Blood Pressure
5. Roles of ENaC in Control of Blood Pressure
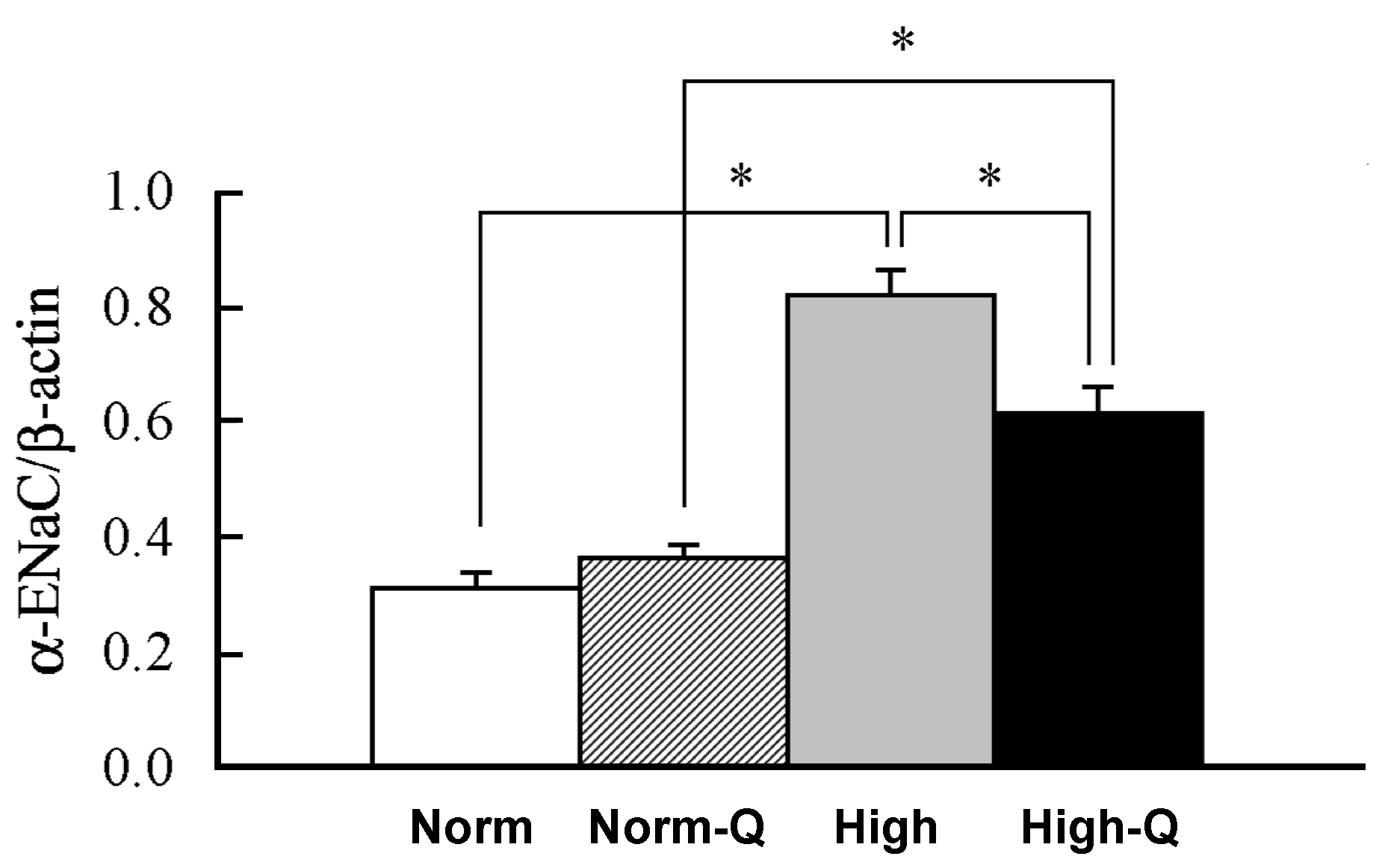
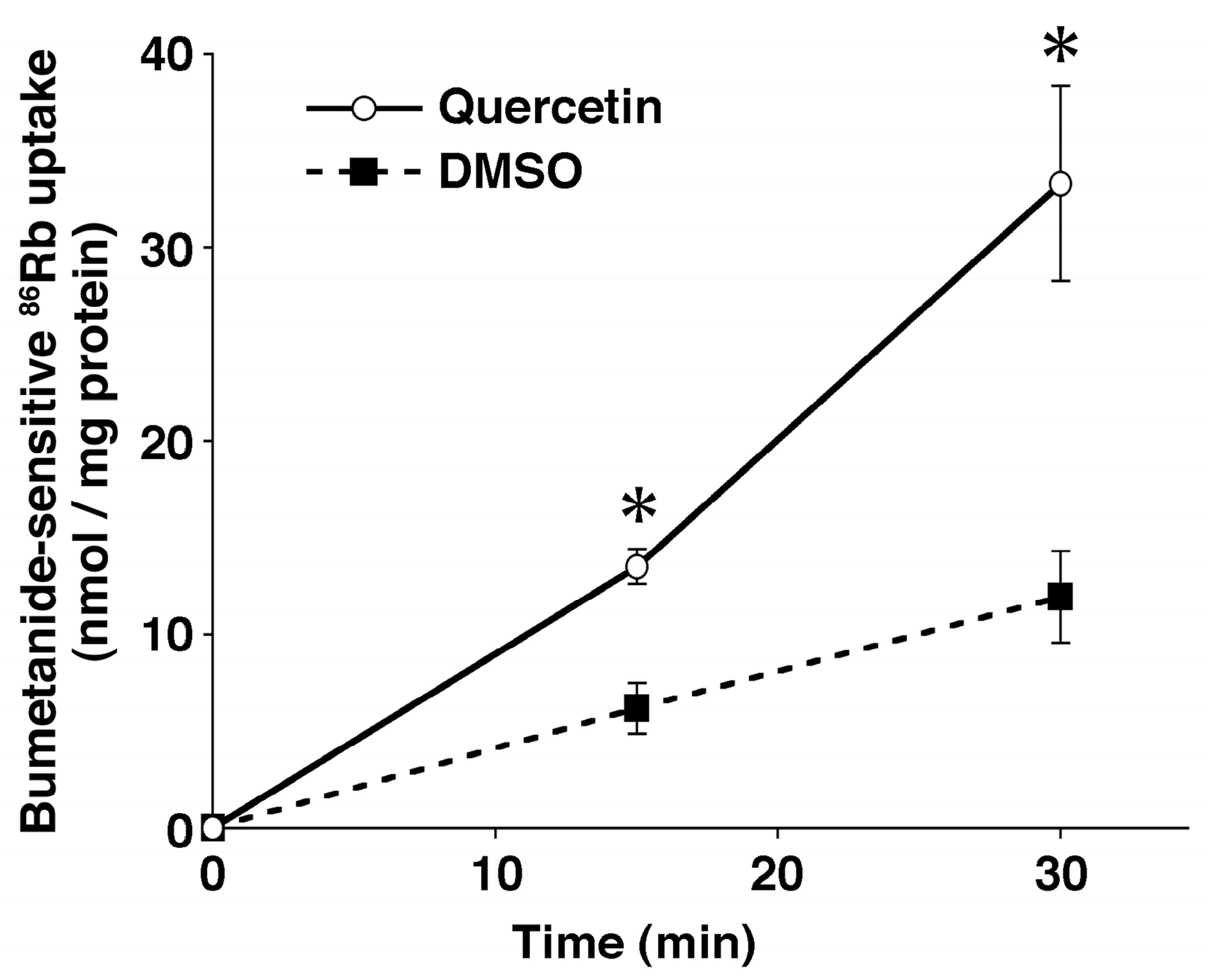
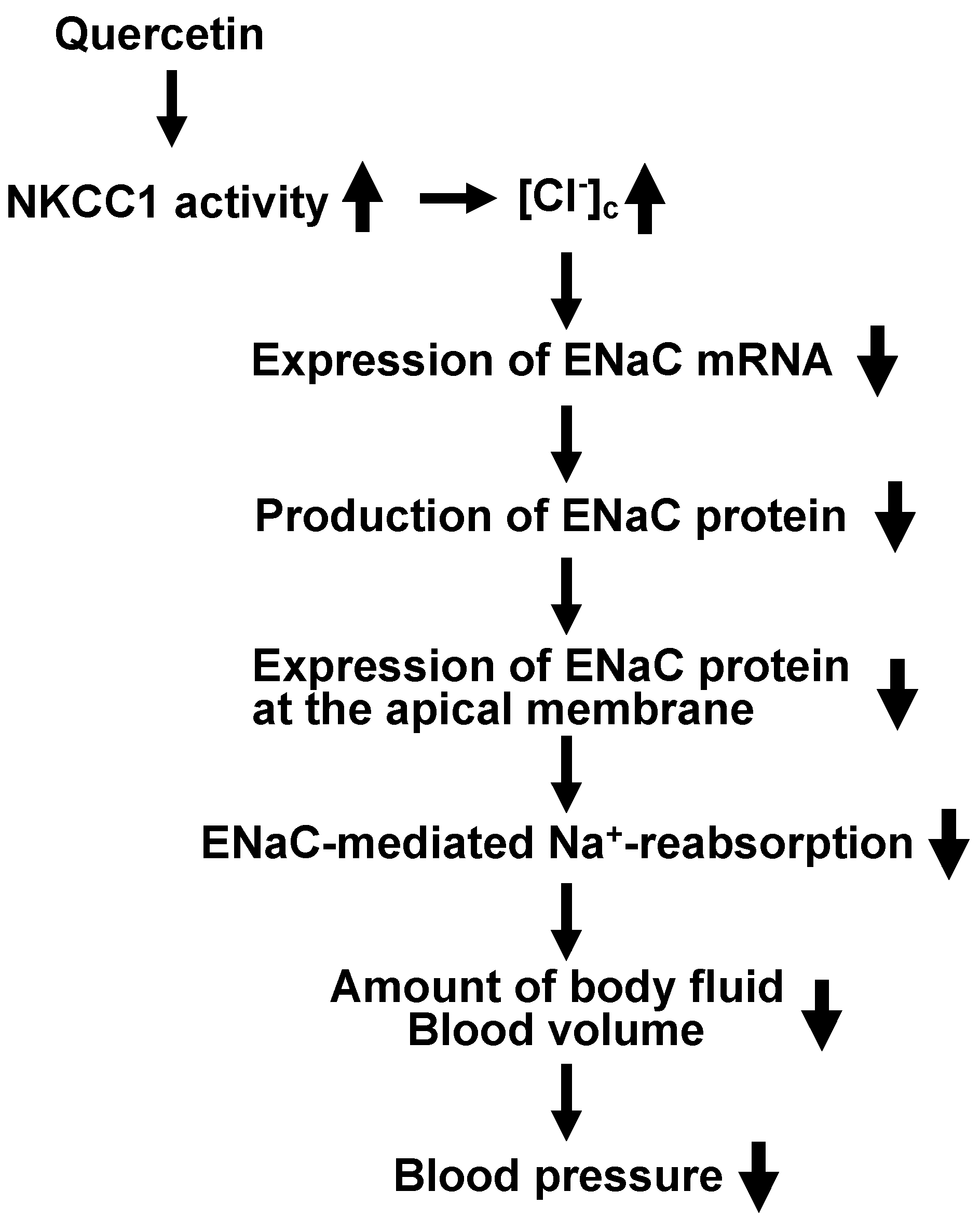
6. Anti-Hypertensive Action of Quercetin via Down-Regulation of ENaC Expression Dependent on Elevation of [Cl−]c Due to Its Stimulatory Action on NKCC-1
7. Conclusions
Acknowledgments
Conflicts of Interest
Abbreviations
| ACE | angiotensin-converting enzyme |
| CD | cluster differentiation |
| [Cl−]c | cytosolic Cl− concentration |
| DOCA | deoxycorticosterone acetate |
| ENaC | epithelial Na+ channel |
| HO-1 | heme oxygenase-1 |
| IL | interleukin |
| LPH | lactase phlorizin hydrolase |
| NKCC1 | Na+-K+-2Cl− cotransporter 1 |
| NPPB | (5-nitro-2-(3-phenylpropylamino)benzoic acid |
| PARP-1 | poly (ADP-ribose) polymerase-1 |
| SGLT-1 | sodium glucose transporter-1 |
| VCAM-1 | vascular cell adhesion molecule-1 |
References
- Endale, M.; Park, S.C.; Kim, S.; Kim, S.H.; Yang, Y.; Cho, J.Y.; Rhee, M.H. Quercetin disrupts tyrosine-phosphorylated phosphatidylinositol 3-kinase and myeloid differentiation factor-88 association, and inhibits MAPK/AP-1 and IKK/NF-κB-induced inflammatory mediators production in RAW 264.7 cells. Immunobiology 2013, 218, 1452–1467. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.M.; Hwang, M.K.; Lee, D.E.; Lee, K.W.; Lee, H.J. Protective effect of quercetin against arsenite-induced COX-2 expression by targeting PI3K in rat liver epithelial cells. J. Agric. Food Chem. 2010, 58, 5815–5820. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.P.; Mani, I.; Iversen, L.; Ziboh, V.A. Effects of naturally-occurring flavonoids and biflavonoids on epidermal cyclooxygenase and lipoxygenase from guinea-pigs. Prostaglandins Leukot. Essent. Fat. Acids 1998, 58, 17–24. [Google Scholar] [CrossRef]
- Mlcek, J.; Jurikova, T.; Skrovankova, S.; Sochor, J. Quercetin and its anti-allergic immune response. Molecules 2016, 21. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, M.F.; Hassan, N.A.; El Bassossy, H.M.; Fahmy, A. Quercetin protects against diabetes-induced exaggerated vasoconstriction in rats: Effect on low grade inflammation. PLoS ONE 2013, 8, e63784. [Google Scholar] [CrossRef] [PubMed]
- Manjeet, K.R.; Ghosh, B. Quercetin inhibits LPS-induced nitric oxide and tumor necrosis factor-α production in murine macrophages. Int. J. Immunopharmacol. 1999, 21, 435–443. [Google Scholar]
- Bureau, G.; Longpre, F.; Martinoli, M.G. Resveratrol and quercetin, two natural polyphenols, reduce apoptotic neuronal cell death induced by neuroinflammation. J. Neurosci. Res. 2008, 86, 403–410. [Google Scholar] [CrossRef] [PubMed]
- Kempuraj, D.; Madhappan, B.; Christodoulou, S.; Boucher, W.; Cao, J.; Papadopoulou, N.; Cetrulo, C.L.; Theoharides, T.C. Flavonols inhibit proinflammatory mediator release, intracellular calcium ion levels and protein kinase C theta phosphorylation in human mast cells. Br. J. Pharmacol. 2005, 145, 934–944. [Google Scholar] [CrossRef] [PubMed]
- Chow, J.M.; Shen, S.C.; Huan, S.K.; Lin, H.Y.; Chen, Y.C. Quercetin, but not rutin and quercitrin, prevention of H2O2-induced apoptosis via anti-oxidant activity and heme oxygenase 1 gene expression in macrophages. Biochem. Pharmacol. 2005, 69, 1839–1851. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Xia, B.; Xie, W.; Zhou, Y.; Xie, J.; Li, H.; Liao, D.; Lin, L.; Li, C. Phytochemistry and pharmacological activities of the genus Prunella. Food Chem. 2016, 204, 483–496. [Google Scholar] [CrossRef] [PubMed]
- Tipoe, G.L.; Leung, T.M.; Hung, M.W.; Fung, M.L. Green tea polyphenols as an anti-oxidant and anti-inflammatory agent for cardiovascular protection. Cardiovasc. Hematol. Disord. Drug Targets 2007, 7, 135–144. [Google Scholar] [CrossRef] [PubMed]
- Ishii, T.; Ishikawa, M.; Miyoshi, N.; Yasunaga, M.; Akagawa, M.; Uchida, K.; Nakamura, Y. Catechol type polyphenol is a potential modifier of protein sulfhydryls: development and application of a new probe for understanding the dietary polyphenol actions. Chem. Res. Toxicol. 2009, 22, 1689–1698. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Niisato, N.; Inui, T.; Marunaka, Y. Insulin is involved in transcriptional regulation of NKCC and the CFTR Cl- channel through PI3K activation and ERK inactivation in renal epithelial cells. J. Physiol. Sci. 2014, 64, 433–443. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Niisato, N.; Nishio, K.; Hamilton, K.L.; Marunaka, Y. Distinct action of flavonoids, myricetin and quercetin, on epithelial Cl- secretion: useful tools as regulators of Cl- secretion. BioMed Res. Int. 2014, 2014, 902735. [Google Scholar] [CrossRef] [PubMed]
- Aoi, W.; Niisato, N.; Miyazaki, H.; Marunaka, Y. Flavonoid-induced reduction of ENaC expression in the kidney of Dahl salt-sensitive hypertensive rat. Biochem. Biophys. Res. Commun. 2004, 315, 892–896. [Google Scholar] [CrossRef] [PubMed]
- Marunaka, Y. Characteristics and pharmacological regulation of epithelial Na+ channel (ENaC) and epithelial Na+ transport. J. Pharmacol. Sci. 2014, 126, 21–36. [Google Scholar] [CrossRef] [PubMed]
- Aoi, W.; Hosogi, S.; Niisato, N.; Yokoyama, N.; Hayata, H.; Miyazaki, H.; Kusuzaki, K.; Fukuda, T.; Fukui, M.; Nakamura, N.; et al. Improvement of insulin resistance, blood pressure and interstitial pH in early developmental stage of insulin resistance in OLETF rats by intake of propolis extracts. Biochem. Biophys. Res. Commun. 2013, 432, 650–653. [Google Scholar] [CrossRef] [PubMed]
- Hayata, H.; Miyazaki, H.; Niisato, N.; Yokoyama, N.; Marunaka, Y. Lowered extracellular pH is involved in the pathogenesis of skeletal muscle insulin resistance. Biochem. Biophys. Res. Commun. 2014, 445, 170–174. [Google Scholar] [CrossRef] [PubMed]
- Marunaka, Y.; Yoshimoto, K.; Aoi, W.; Hosogi, S.; Ikegaya, H. Low pH of interstitial fluid around hippocampus of the brain in diabetic OLETF rats. Mol. Cell Therapies 2014, 2, 6. [Google Scholar] [CrossRef] [PubMed]
- Aoi, W.; Marunaka, Y. The importance of regulation of body fluid pH in the development and progression of metabolic diseases. In Advances in Medicine and Biology; Berhardt, L.V., Ed.; Nova Publishers: Hauppauge, NY, USA, 2014; Volume 77, pp. 177–189. [Google Scholar]
- Aoi, W.; Marunaka, Y. Importance of pH homeostasis in metabolic health and diseases: crucial role of membrane proton transport. BioMed Res. Int. 2014, 2014, 598986. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, K.; Niisato, N.; Marunaka, Y. Quercetin stimulates NGF-induced neurite outgrowth in PC12 cells via activation of Na+/K+/2Cl− cotransporter. Cell. Physiol. Biochem. 2011, 28, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Niisato, N.; Eaton, D.C.; Marunaka, Y. Involvement of cytosolic Cl- in osmoregulation of α-ENaC gene expression. Am. J. Physiol. Renal Physiol. 2004, 287, F932–F939. [Google Scholar] [CrossRef] [PubMed]
- Jeon, S.B.; Kim, G.; Kim, J.I.; Seok, Y.M.; Kim, S.H.; Suk, K.; Shin, H.M.; Lee, Y.H.; Kim, I.K. Flavone inhibits vascular contraction by decreasing phosphorylation of the myosin phosphatase target subunit. Clin. Exp. Pharmacol. Physiol. 2007, 34, 1116–1120. [Google Scholar] [CrossRef] [PubMed]
- Fujimoto, S.; Niisato, N.; Sugimoto, T.; Marunaka, Y. Quercetin and NPPB-induced diminution of aldosterone action on Na+ absorption and ENaC expression in renal epithelium. Biochem. Biophys. Res. Commun. 2005, 336, 401–407. [Google Scholar] [CrossRef] [PubMed]
- Mezesova, L.; Bartekova, M.; Javorkova, V.; Vlkovicova, J.; Breier, A.; Vrbjar, N. Effect of quercetin on kinetic properties of renal Na,K-ATPase in normotensive and hypertensive rats. J. Physiol. Pharmacol. 2010, 61, 593–598. [Google Scholar] [PubMed]
- D’Andrea, G. Quercetin: A flavonol with multifaceted therapeutic applications? Fitoterapia 2015, 106, 256–271. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Hara, H. Role of flavonoids in intestinal tight junction regulation. J. Nutr. Biochem. 2011, 22, 401–408. [Google Scholar] [CrossRef] [PubMed]
- Day, A.J.; Canada, F.J.; Diaz, J.C.; Kroon, P.A.; McLauchlan, R.; Faulds, C.B.; Plumb, G.W.; Morgan, M.R.; Williamson, G. Dietary flavonoid and isoflavone glycosides are hydrolysed by the lactase site of lactase phlorizin hydrolase. FEBS Lett. 2000, 468, 166–170. [Google Scholar] [CrossRef]
- Wolffram, S.; Block, M.; Ader, P. Quercetin-3-glucoside is transported by the glucose carrier SGLT1 across the brush border membrane of rat small intestine. J. Nutr. 2002, 132, 630–635. [Google Scholar] [PubMed]
- Walgren, R.A.; Lin, J.T.; Kinne, R.K.; Walle, T. Cellular uptake of dietary flavonoid quercetin 4′-β-glucoside by sodium-dependent glucose transporter SGLT1. J. Pharmacol. Exp. Ther. 2000, 294, 837–843. [Google Scholar] [PubMed]
- Nemeth, K.; Plumb, G.W.; Berrin, J.G.; Juge, N.; Jacob, R.; Naim, H.Y.; Williamson, G.; Swallow, D.M.; Kroon, P.A. Deglycosylation by small intestinal epithelial cell β-glucosidases is a critical step in the absorption and metabolism of dietary flavonoid glycosides in humans. Eur. J. Nutr. 2003, 42, 29–42. [Google Scholar] [CrossRef] [PubMed]
- Crespy, V.; Morand, C.; Manach, C.; Besson, C.; Demigne, C.; Remesy, C. Part of quercetin absorbed in the small intestine is conjugated and further secreted in the intestinal lumen. Am. J. Physiol. Gastrointest. Liver Physiol. 1999, 277, G120–G126. [Google Scholar]
- Boonpawa, R.; Moradi, N.; Spenkelink, A.; Rietjens, I.M.; Punt, A. Use of physiologically based kinetic (PBK) modeling to study interindividual human variation and species differences in plasma concentrations of quercetin and its metabolites. Biochem. Pharmacol. 2015, 98, 690–702. [Google Scholar] [CrossRef] [PubMed]
- Edwards, R.L.; Lyon, T.; Litwin, S.E.; Rabovsky, A.; Symons, J.D.; Jalili, T. Quercetin reduces blood pressure in hypertensive subjects. J. Nutr. 2007, 137, 2405–2411. [Google Scholar] [PubMed]
- Egert, S.; Bosy-Westphal, A.; Seiberl, J.; Kurbitz, C.; Settler, U.; Plachta-Danielzik, S.; Wagner, A.E.; Frank, J.; Schrezenmeir, J.; Rimbach, G.; et al. Quercetin reduces systolic blood pressure and plasma oxidised low-density lipoprotein concentrations in overweight subjects with a high-cardiovascular disease risk phenotype: A double-blinded, placebo-controlled cross-over study. Br. J. Nutr. 2009, 102, 1065–1074. [Google Scholar] [CrossRef] [PubMed]
- Zahedi, M.; Ghiasvand, R.; Feizi, A.; Asgari, G.; Darvish, L. Does quercetin improve cardiovascular risk factors and inflammatory biomarkers in women with type 2 diabetes: A double-blind randomized controlled clinical trial. Int. J. Prev. Med. 2013, 4, 777–785. [Google Scholar] [PubMed]
- Serban, M.C.; Sahebkar, A.; Zanchetti, A.; Mikhailidis, D.P.; Howard, G.; Antal, D.; Andrica, F.; Ahmed, A.; Aronow, W.S.; Muntner, P.; et al. Effects of quercetin on blood pressure: A systematic review and meta-analysis of randomized controlled trials. J. Am. Heart Assoc. 2016, 5, e002713. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.; Ryu, K.H.; Park, S.H.; Jun, J.Y.; Shin, B.C.; Chung, J.H.; Yeum, C.H. Direct vascular actions of quercetin in aorta from renal hypertensive rats. Kidney Res. Clin. Pract. 2016, 35, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Galisteo, M.; Garcia-Saura, M.F.; Jimenez, R.; Villar, I.C.; Wangensteen, R.; Zarzuelo, A.; Vargas, F.; Duarte, J. Effects of quercetin treatment on vascular function in deoxycorticosterone acetate-salt hypertensive rats. Comparative study with verapamil. Planta Med. 2004, 70, 334–341. [Google Scholar] [PubMed]
- Merendino, N.; Molinari, R.; Costantini, L.; Mazzucato, A.; Pucci, A.; Bonafaccia, F.; Esti, M.; Ceccantoni, B.; Papeschi, C.; Bonafaccia, G. A new “functional” pasta containing tartary buckwheat sprouts as an ingredient improves the oxidative status and normalizes some blood pressure parameters in spontaneously hypertensive rats. Food Funct. 2014, 5, 1017–1026. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, M.M.; Franca-Silva, M.S.; Alves, N.F.; Porpino, S.K.; Braga, V.A. Quercetin improves baroreflex sensitivity in spontaneously hypertensive rats. Molecules 2012, 17, 12997–13008. [Google Scholar] [CrossRef] [PubMed]
- Ibarra, M.; Moreno, L.; Vera, R.; Cogolludo, A.; Duarte, J.; Tamargo, J.; Perez-Vizcaino, F. Effects of the flavonoid quercetin and its methylated metabolite isorhamnetin in isolated arteries from spontaneously hypertensive rats. Planta Med. 2003, 69, 995–1000. [Google Scholar] [PubMed]
- Parichatikanond, W.; Pinthong, D.; Mangmool, S. Blockade of the renin-angiotensin system with delphinidin, cyanin, and quercetin. Planta Med. 2012, 78, 1626–1632. [Google Scholar] [CrossRef] [PubMed]
- Olaleye, M.T.; Crown, O.O.; Akinmoladun, A.C.; Akindahunsi, A.A. Rutin and quercetin show greater efficacy than nifedipin in ameliorating hemodynamic, redox, and metabolite imbalances in sodium chloride-induced hypertensive rats. Hum. Exp. Toxicol. 2014, 33, 602–608. [Google Scholar] [CrossRef] [PubMed]
- Geraets, L.; Moonen, H.J.; Brauers, K.; Wouters, E.F.; Bast, A.; Hageman, G.J. Dietary flavones and flavonoles are inhibitors of poly(ADP-ribose)polymerase-1 in pulmonary epithelial cells. J. Nutr. 2007, 137, 2190–2195. [Google Scholar] [PubMed]
- Yang, D.; Liu, X.; Liu, M.; Chi, H.; Liu, J.; Han, H. Protective effects of quercetin and taraxasterol against H2O2-induced human umbilical vein endothelial cell injury in vitro. Exp. Ther. Med. 2015, 10, 1253–1260. [Google Scholar] [CrossRef] [PubMed]
- Krol, W.; Czuba, Z.; Scheller, S.; Gabrys, J.; Grabiec, S.; Shani, J. Anti-oxidant property of ethanolic extract of propolis (EEP) as evaluated by inhibiting the chemiluminescence oxidation of luminol. Biochem. Int. 1990, 21, 593–597. [Google Scholar] [PubMed]
- Javadi, F.; Eghtesadi, S.; Ahmadzadeh, A.; Aryaeian, N.; Zabihiyeganeh, M.; Foroushani, A.R.; Jazayeri, S. The effect of quercetin on plasma oxidative status, C-reactive protein and blood pressure in women with rheumatoid arthritis. Int. J. Prev. Med. 2014, 5, 293–301. [Google Scholar] [PubMed]
- Pfeuffer, M.; Auinger, A.; Bley, U.; Kraus-Stojanowic, I.; Laue, C.; Winkler, P.; Rufer, C.E.; Frank, J.; Bosch-Saadatmandi, C.; Rimbach, G.; et al. Effect of quercetin on traits of the metabolic syndrome, endothelial function and inflammation in men with different APOE isoforms. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 403–409. [Google Scholar] [CrossRef] [PubMed]
- Erdogan, A.; Most, A.K.; Wienecke, B.; Fehsecke, A.; Leckband, C.; Voss, R.; Grebe, M.T.; Tillmanns, H.; Schaefer, C.A.; Kuhlmann, C.R. Apigenin-induced nitric oxide production involves calcium-activated potassium channels and is responsible for antiangiogenic effects. J. Thromb. Haemost. 2007, 5, 1774–1781. [Google Scholar] [CrossRef] [PubMed]
- Dayoub, O.; Andriantsitohaina, R.; Clere, N. Pleiotropic beneficial effects of epigallocatechin gallate, quercetin and delphinidin on cardiovascular diseases associated with endothelial dysfunction. Cardiovasc. Hematol. Agents Med. Chem. 2013, 11, 249–264. [Google Scholar] [CrossRef] [PubMed]
- Galleano, M.; Pechanova, O.; Fraga, C.G. Hypertension, nitric oxide, oxidants, and dietary plant polyphenols. Curr. Pharm. Biotechnol. 2010, 11, 837–848. [Google Scholar] [CrossRef] [PubMed]
- Formica, J.V.; Regelson, W. Review of the biology of Quercetin and related bioflavonoids. Food Chem. Toxicol. 1995, 33, 1061–1080. [Google Scholar] [CrossRef]
- Bischoff, S.C. Quercetin: potentials in the prevention and therapy of disease. Curr. Opin. Clin. Nutr. Metab. Care 2008, 11, 733–740. [Google Scholar] [CrossRef] [PubMed]
- Perez-Vizcaino, F.; Duarte, J. Flavonols and cardiovascular disease. Mol. Aspects Med. 2010, 31, 478–494. [Google Scholar] [CrossRef] [PubMed]
- Seebaluck, R.; Gurib-Fakim, A.; Mahomoodally, F. Medicinal plants from the genus Acalypha (Euphorbiaceae)--a review of their ethnopharmacology and phytochemistry. J. Ethnopharmacol. 2015, 159, 137–157. [Google Scholar] [CrossRef] [PubMed]
- Aoi, W.; Niisato, N.; Sawabe, Y.; Miyazaki, H.; Marunaka, Y. Aldosterone-induced abnormal regulation of ENaC and SGK1 in Dahl salt-sensitive rat. Biochem. Biophys. Res. Commun. 2006, 341, 376–381. [Google Scholar] [CrossRef] [PubMed]
- Aoi, W.; Niisato, N.; Sawabe, Y.; Miyazaki, H.; Tokuda, S.; Nishio, K.; Yoshikawa, T.; Marunaka, Y. Abnormal expression of ENaC and SGK1 mRNA induced by dietary sodium in Dahl salt-sensitively hypertensive rats. Cell Biol. Int. 2007, 31, 1288–1291. [Google Scholar] [CrossRef] [PubMed]
- Marunaka, Y.; Niisato, N.; Taruno, A.; Ohta, M.; Miyazaki, H.; Hosogi, S.; Nakajima, K.; Kusuzaki, K.; Ashihara, E.; Nishio, K.; et al. Regulation of epithelial sodium transport via epithelial Na+ channel. J. Biomed. Biotechnol. 2011, 2011, 978196. [Google Scholar] [CrossRef] [PubMed]
- Schweda, F. Salt feedback on the renin-angiotensin-aldosterone system. Pflugers Arch. 2015, 467, 565–576. [Google Scholar] [CrossRef] [PubMed]
- Bollag, W.B. Regulation of aldosterone synthesis and secretion. Compr. Physiol. 2014, 4, 1017–1055. [Google Scholar] [PubMed]
- Warnock, D.G.; Kusche-Vihrog, K.; Tarjus, A.; Sheng, S.; Oberleithner, H.; Kleyman, T.R.; Jaisser, F. Blood pressure and amiloride-sensitive sodium channels in vascular and renal cells. Nat. Rev. Nephrol. 2014, 10, 146–157. [Google Scholar] [CrossRef] [PubMed]
- Canessa, C.M.; Horisberger, J.D.; Rossier, B.C. Epithelial sodium channel related to proteins involved in neurodegeneration. Nature 1993, 361, 467–470. [Google Scholar] [CrossRef] [PubMed]
- Canessa, C.M.; Schild, L.; Buell, G.; Thorens, B.; Gautschi, I.; Horisberger, J.D.; Rossier, B.C. Amiloride-sensitive epithelial Na+ channel is made of three homologous subunits. Nature 1994, 367, 463–467. [Google Scholar] [CrossRef] [PubMed]
- Ji, H.L.; Zhao, R.Z.; Chen, Z.X.; Shetty, S.; Idell, S.; Matalon, S. delta ENaC: A novel divergent amiloride-inhibitable sodium channel. Am. J. Physiol. Lung Cell. Mol. Physiol. 2012, 303, L1013–L1026. [Google Scholar] [CrossRef] [PubMed]
- Shimkets, R.A.; Warnock, D.G.; Bositis, C.M.; Nelson-Williams, C.; Hansson, J.H.; Schambelan, M.; Gill, J.R., Jr.; Ulick, S.; Milora, R.V.; Findling, J.W.; et al. Liddle’s syndrome: heritable human hypertension caused by mutations in the β subunit of the epithelial sodium channel. Cell 1994, 79, 407–414. [Google Scholar] [CrossRef]
- Snyder, P.M.; Price, M.P.; McDonald, F.J.; Adams, C.M.; Volk, K.A.; Zeiher, B.G.; Stokes, J.B.; Welsh, M.J. Mechanism by which Liddle’s syndrome mutations increase activity of a human epithelial Na+ channel. Cell 1995, 83, 969–978. [Google Scholar] [CrossRef]
- Firsov, D.; Schild, L.; Gautschi, I.; Merillat, A.M.; Schneeberger, E.; Rossier, B.C. Cell surface expression of the epithelial Na channel and a mutant causing Liddle syndrome: A quantitative approach. Proc. Natl. Acad. Sci. USA 1996, 93, 15370–15375. [Google Scholar] [CrossRef] [PubMed]
- Kamynina, E.; Debonneville, C.; Hirt, R.P.; Staub, O. Liddle’s syndrome: A novel mouse Nedd4 isoform regulates the activity of the epithelial Na+ channel. Kidney Int. 2001, 60, 466–471. [Google Scholar] [CrossRef] [PubMed]
- Hanukoglu, I.; Hanukoglu, A. Epithelial sodium channel (ENaC) family: Phylogeny, structure-function, tissue distribution, and associated inherited diseases. Gene 2016, 579, 95–132. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, F.; Staub, O. NEDD4-2 and salt-sensitive hypertension. Curr. Opin. Nephrol. Hypertens. 2015, 24, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Ronzaud, C.; Staub, O. Ubiquitylation and control of renal Na+ balance and blood pressure. Physiology (Bethesda) 2014, 29, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.Q.; Xiao, Y.; Tian, T.; Gao, L.G.; Zhou, X.L. Molecular genetics of Liddle’s syndrome. Clin. Chim. Acta 2014, 436, 202–206. [Google Scholar] [CrossRef] [PubMed]
- Bubien, J.K. Epithelial Na+ channel (ENaC), hormones, and hypertension. J. Biol. Chem. 2010, 285, 23527–23531. [Google Scholar] [CrossRef] [PubMed]
- Marunaka, Y.; Marunaka, R.; Sun, H.; Yamamoto, T.; Kanamura, N.; Taruno, A. Na+ homeostasis by epithelial Na+ channel (ENaC) and Nax channel (Nax): Cooperation of ENaC and Nax. Ann. Transl. Med. 2016, 4, S11. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, K.I.; Marunaka, Y. Intracellular chloride ion concentration in differentiating neuronal cell and its role in growing neurite. Biochem. Biophys. Res. Commun. 2016, 479, 338–342. [Google Scholar] [CrossRef] [PubMed]
- Cermak, R.; Follmer, U.; Wolffram, S. Dietary flavonol quercetin induces chloride secretion in rat colon. Am. J. Physiol. Gastrointest. Liver Physiol. 1998, 275, G1166–G1172. [Google Scholar]
- Cermak, R.; Vujicic, Z.; Scharrer, E.; Wolfram, S. The impact of different flavonoid classes on colonic Cl- secretion in rats. Biochem. Pharmacol. 2001, 62, 1145–1151. [Google Scholar] [CrossRef]
- Asano, J.; Niisato, N.; Nakajima, K.; Miyazaki, H.; Yasuda, M.; Iwasaki, Y.; Hama, T.; Dejima, K.; Hisa, Y.; Marunaka, Y. Quercetin stimulates Na+/K+/2Cl- cotransport via PTK-dependent mechanisms in human airway epithelium. Am. J. Respir. Cell Mol. Biol. 2009, 41, 688–695. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Niisato, N.; Marunaka, Y. Quercetin diminishes the cAMP-stimulated Cl− secretion by blocking Na+,K+-ATPase in epithelial cells. J. Physiol. Sci. 2014, 64, S116. [Google Scholar]
- Nguyen, T.D.; Canada, A.T.; Heintz, G.G.; Gettys, T.W.; Cohn, J.A. Stimulation of secretion by the T84 colonic epithelial cell line with dietary flavonols. Biochem. Pharmacol. 1991, 41, 1879–1886. [Google Scholar] [CrossRef]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marunaka, Y.; Marunaka, R.; Sun, H.; Yamamoto, T.; Kanamura, N.; Inui, T.; Taruno, A. Actions of Quercetin, a Polyphenol, on Blood Pressure. Molecules 2017, 22, 209. https://doi.org/10.3390/molecules22020209
Marunaka Y, Marunaka R, Sun H, Yamamoto T, Kanamura N, Inui T, Taruno A. Actions of Quercetin, a Polyphenol, on Blood Pressure. Molecules. 2017; 22(2):209. https://doi.org/10.3390/molecules22020209
Chicago/Turabian StyleMarunaka, Yoshinori, Rie Marunaka, Hongxin Sun, Toshiro Yamamoto, Narisato Kanamura, Toshio Inui, and Akiyuki Taruno. 2017. "Actions of Quercetin, a Polyphenol, on Blood Pressure" Molecules 22, no. 2: 209. https://doi.org/10.3390/molecules22020209
APA StyleMarunaka, Y., Marunaka, R., Sun, H., Yamamoto, T., Kanamura, N., Inui, T., & Taruno, A. (2017). Actions of Quercetin, a Polyphenol, on Blood Pressure. Molecules, 22(2), 209. https://doi.org/10.3390/molecules22020209






