Zinc Ion Removal on Hybrid Pectin-Based Beads Containing Modified Poly(Methyl Methacrylate) Waste
Abstract
:1. Introduction
2. Results and Discussion
2.1. Sorbent Preparation and Characterization
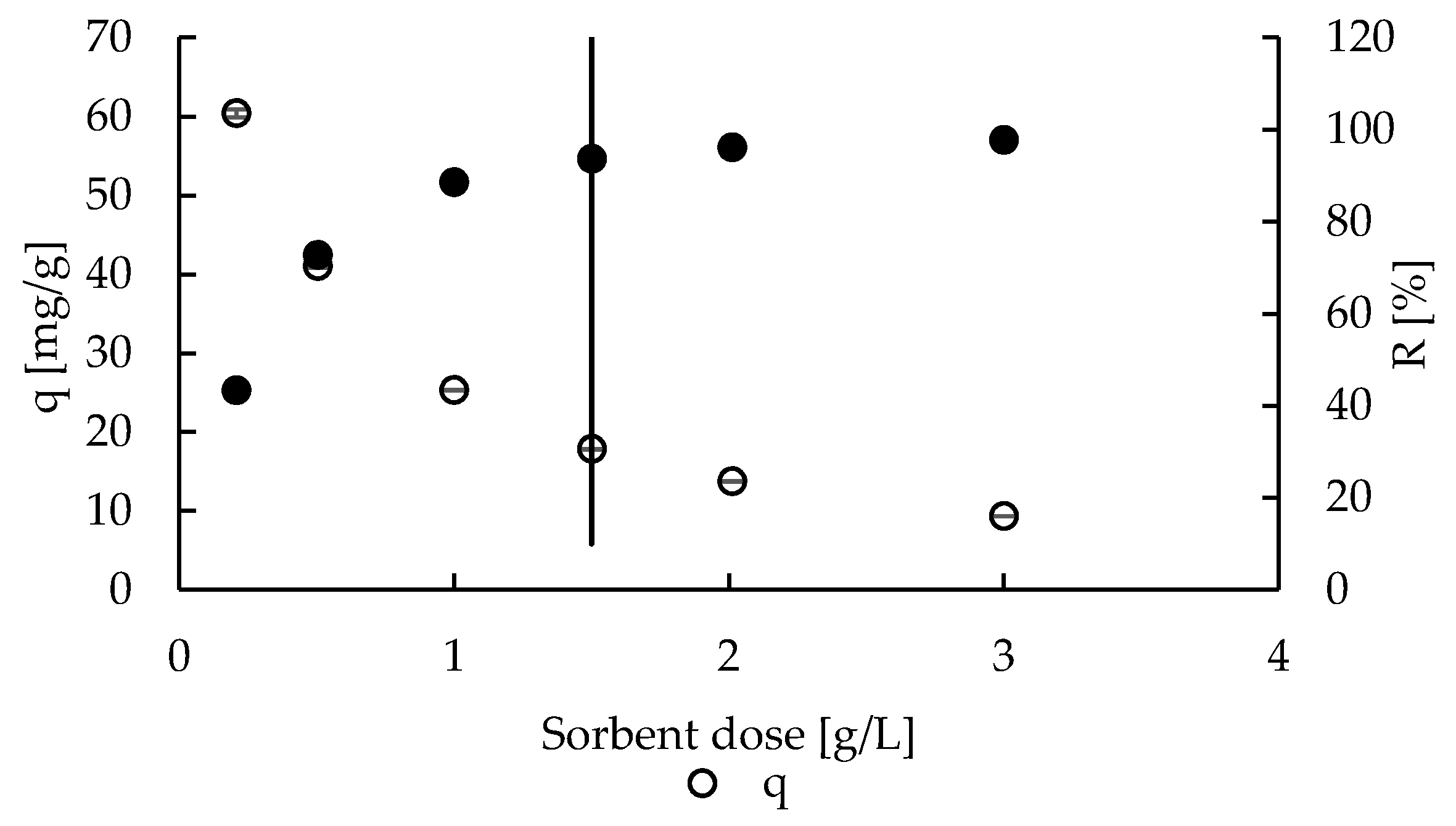
2.2. Sorption—Effect of Sorbent Dose
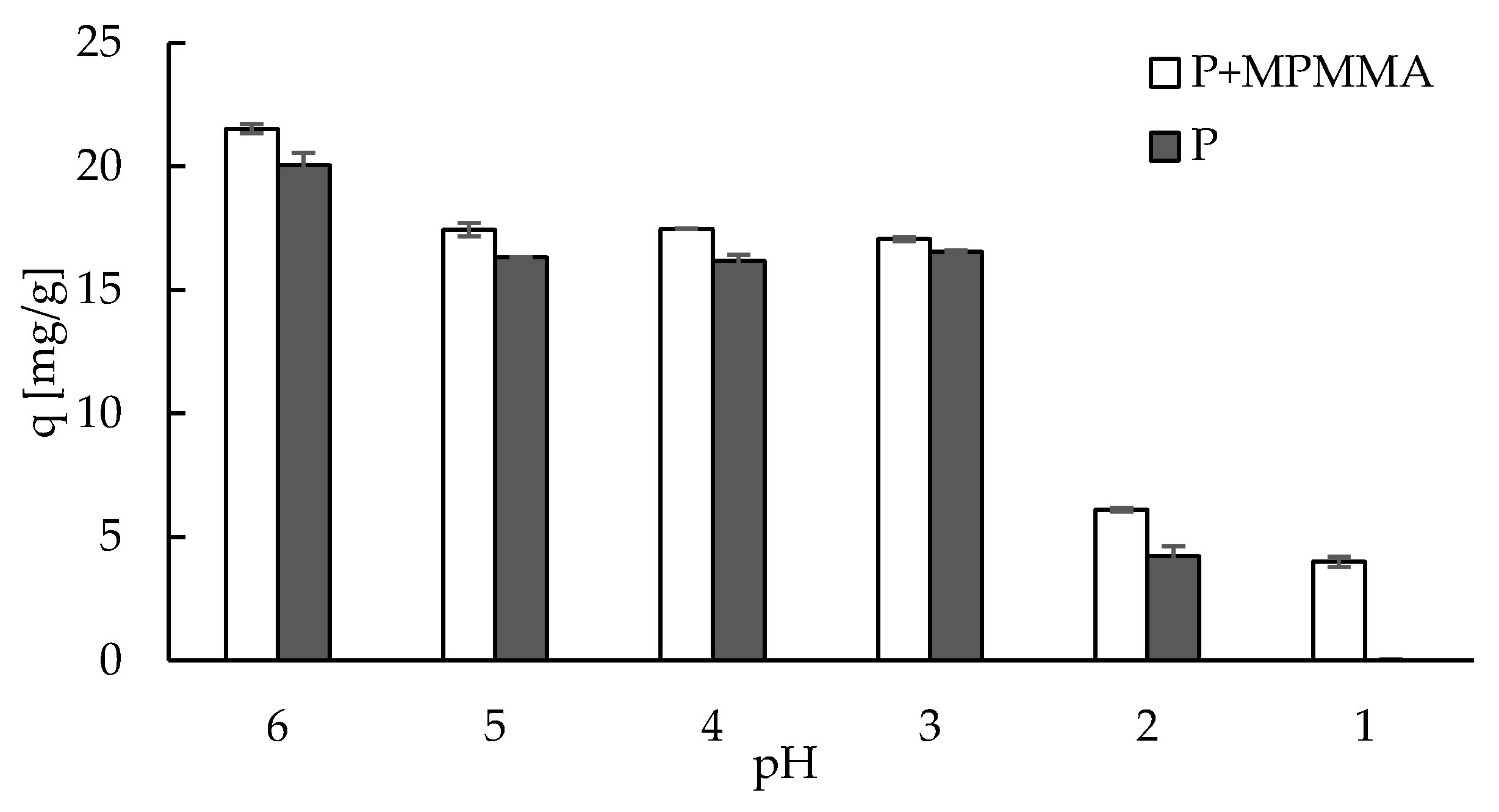
2.3. Sorption—Effect of pH
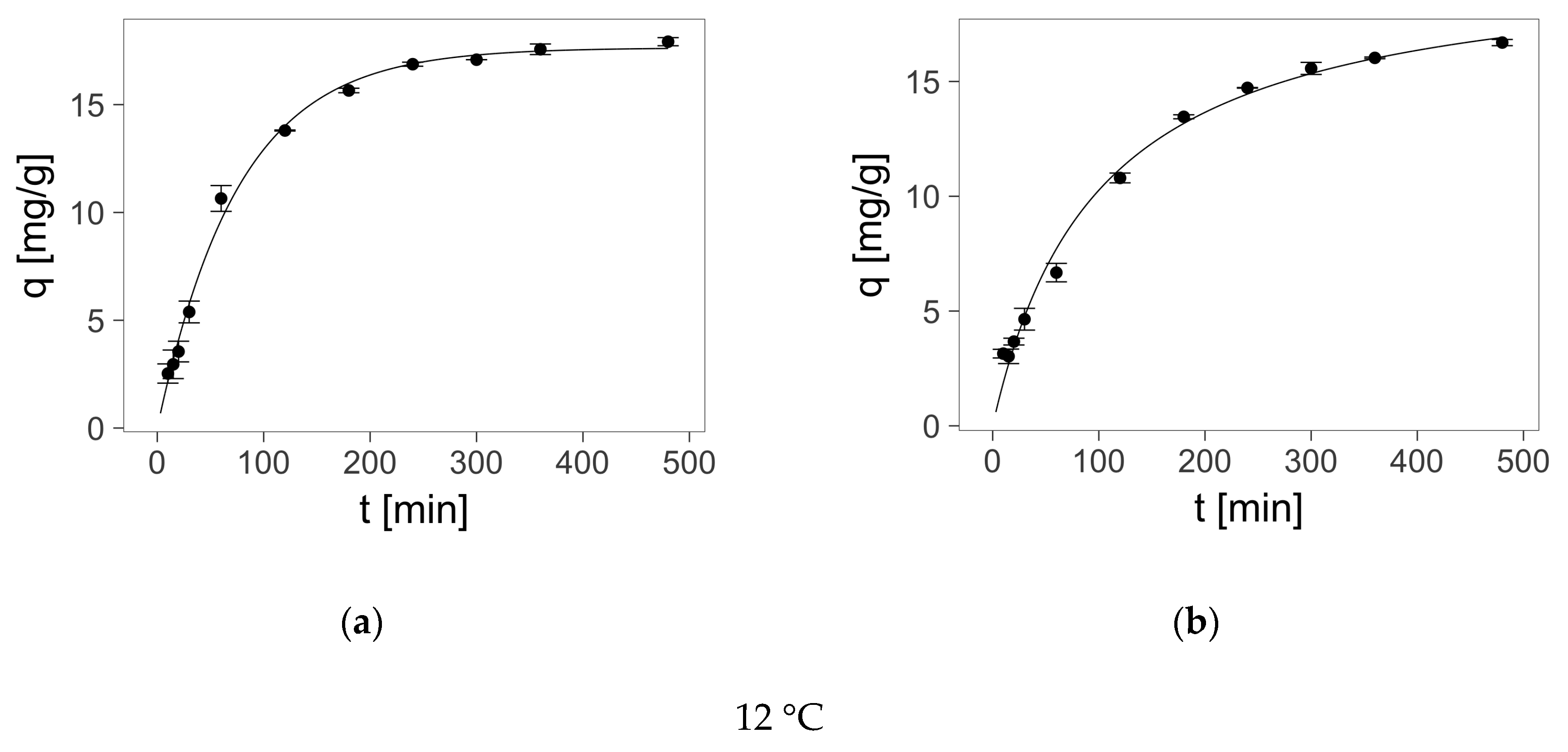
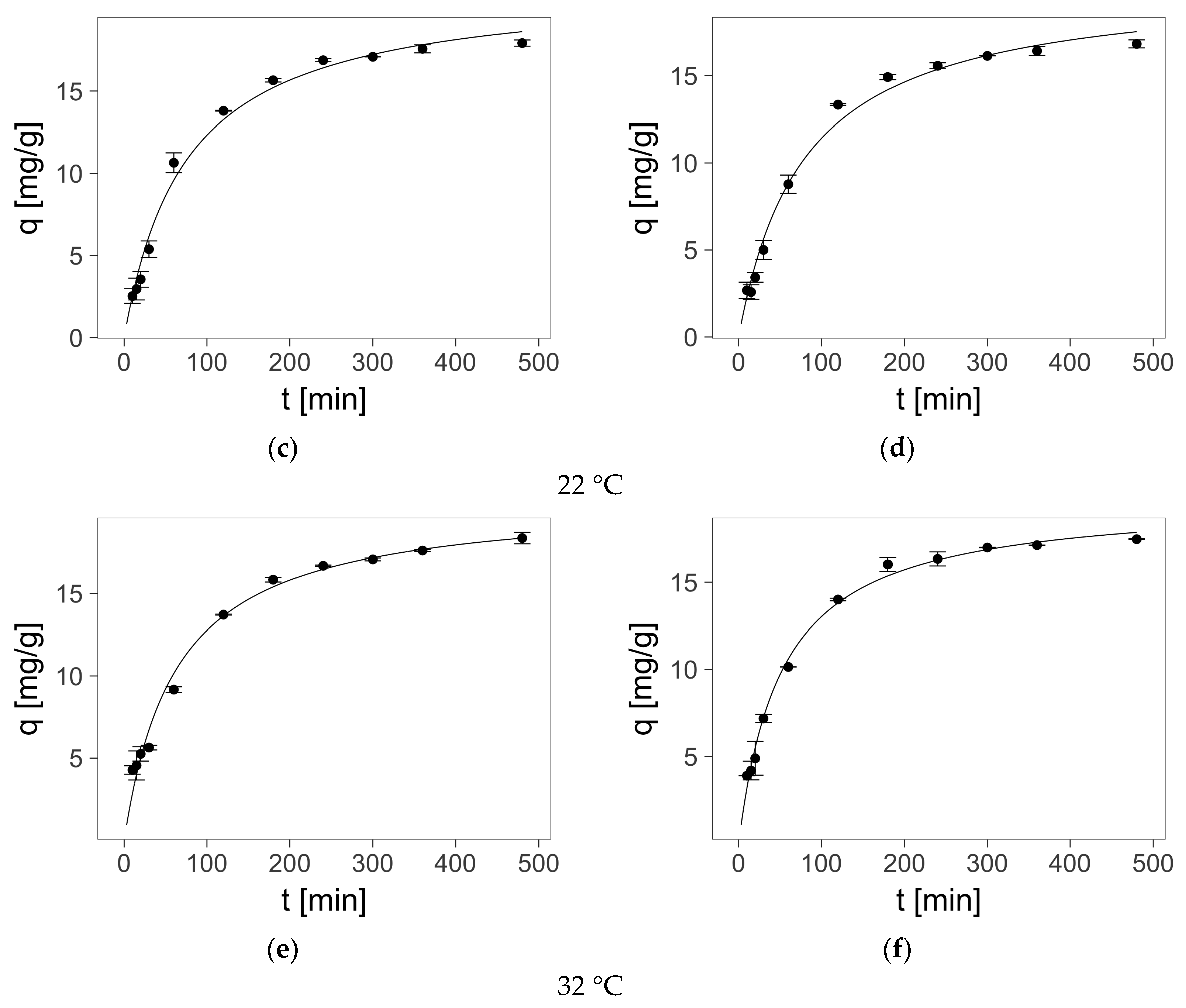
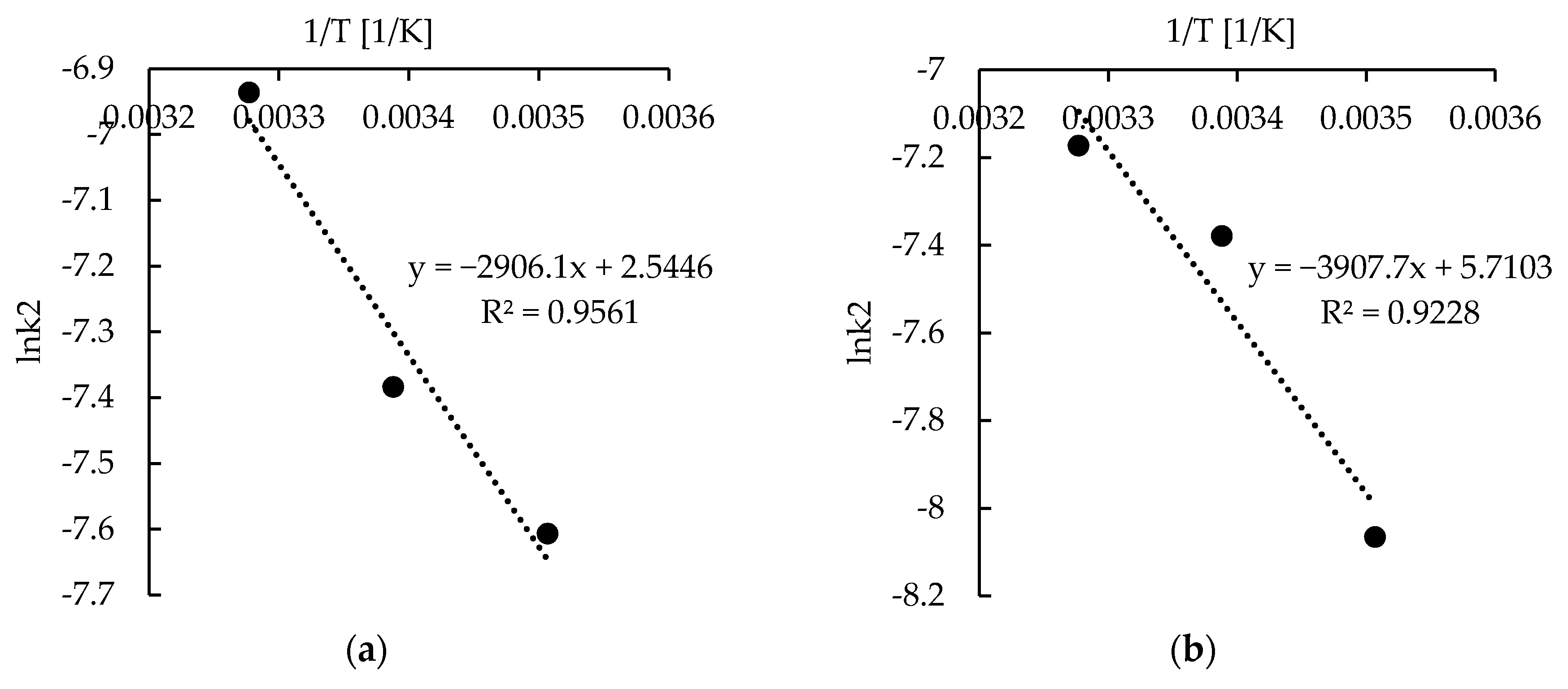
2.4. Sorption—Kinetics
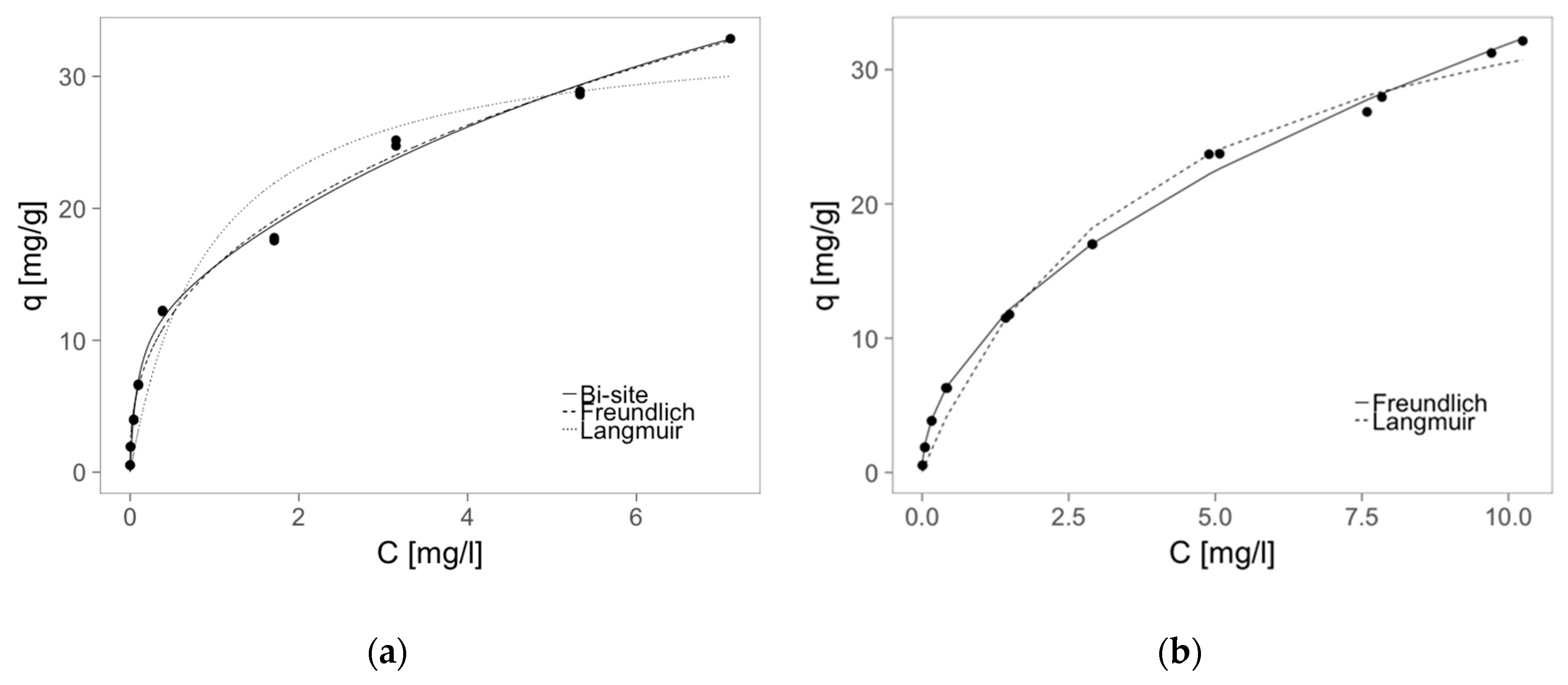
2.5. Sorption—Isotherms
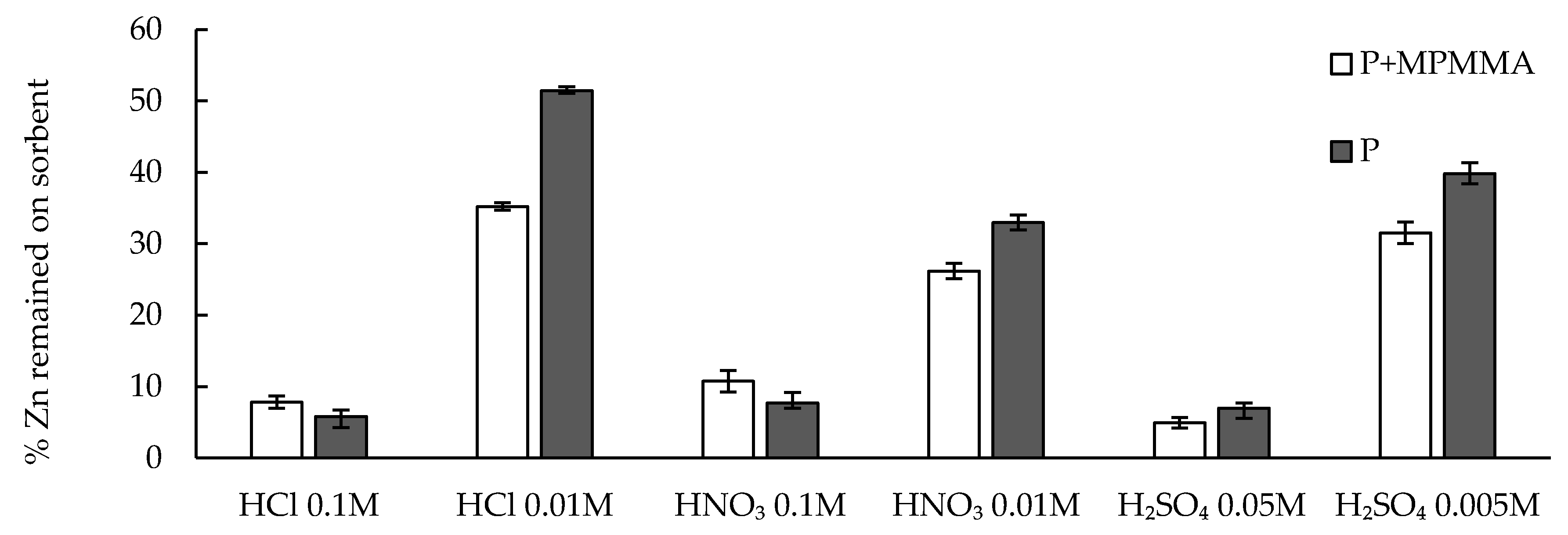
2.6. Desorption—Equilibrium
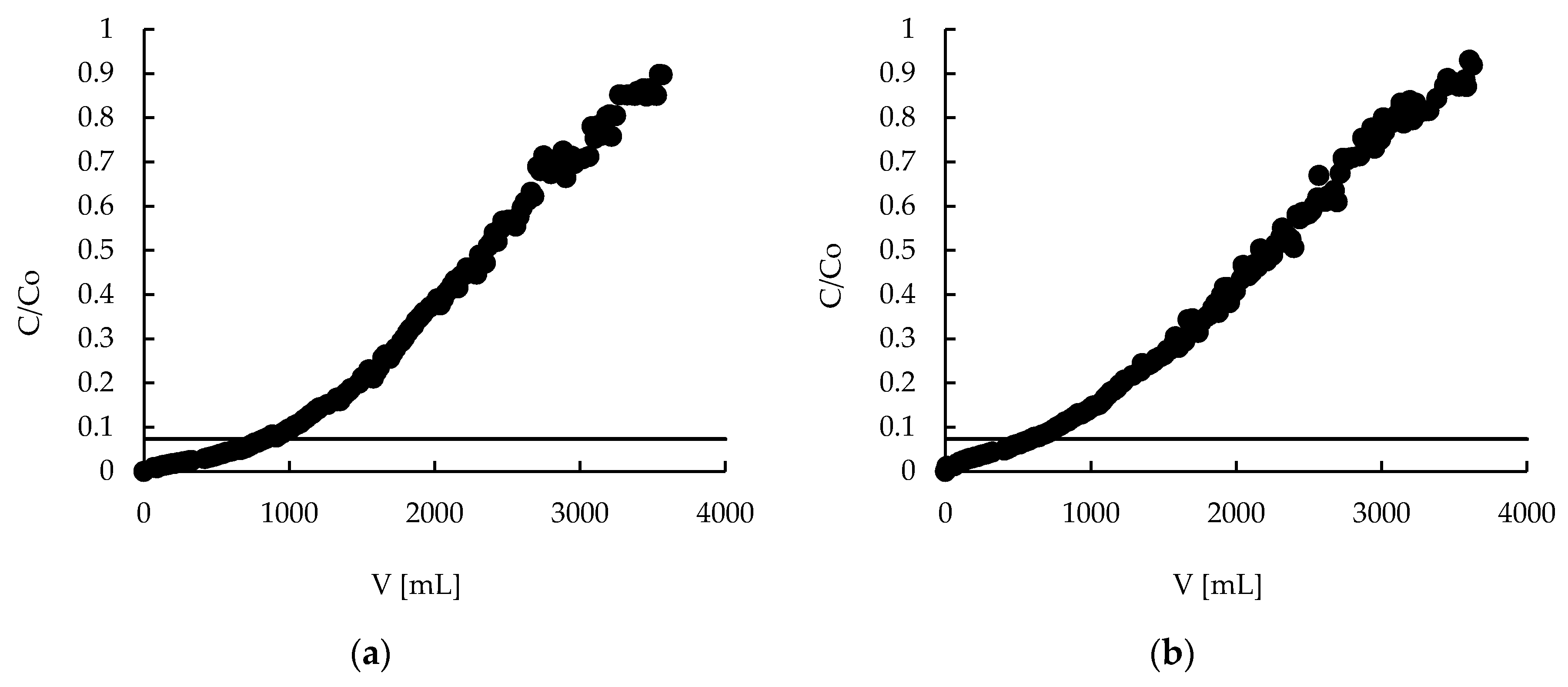
2.7. Sorption—Breakthrough Curves
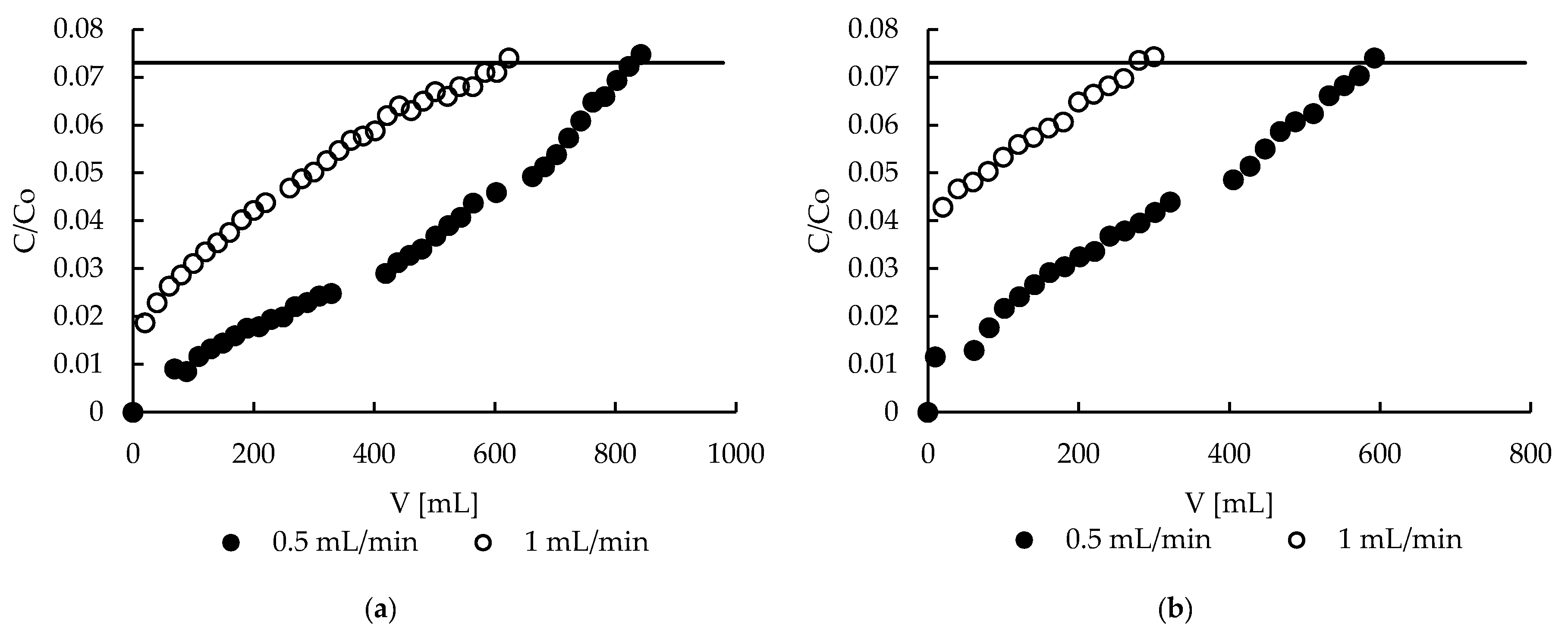
2.8. Sorption—Effect of Flow Rate
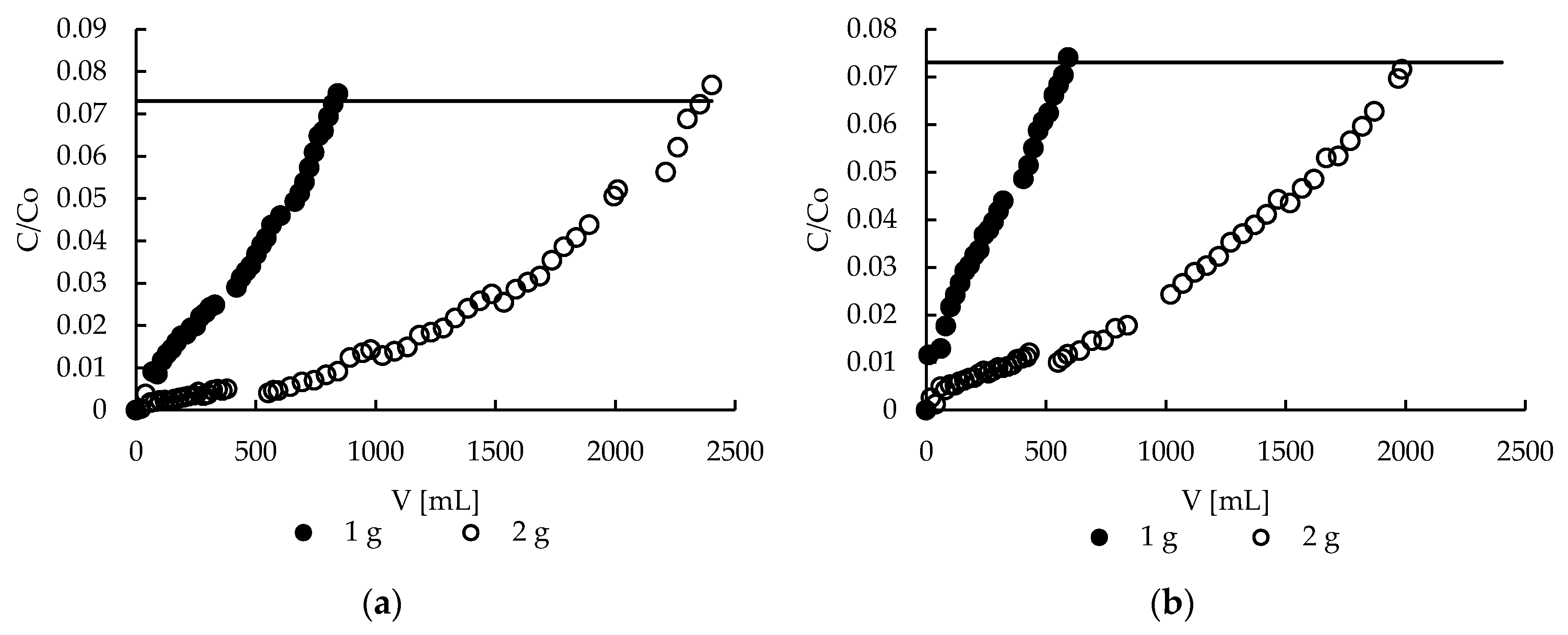
2.9. Sorption—Effect of Bed Height
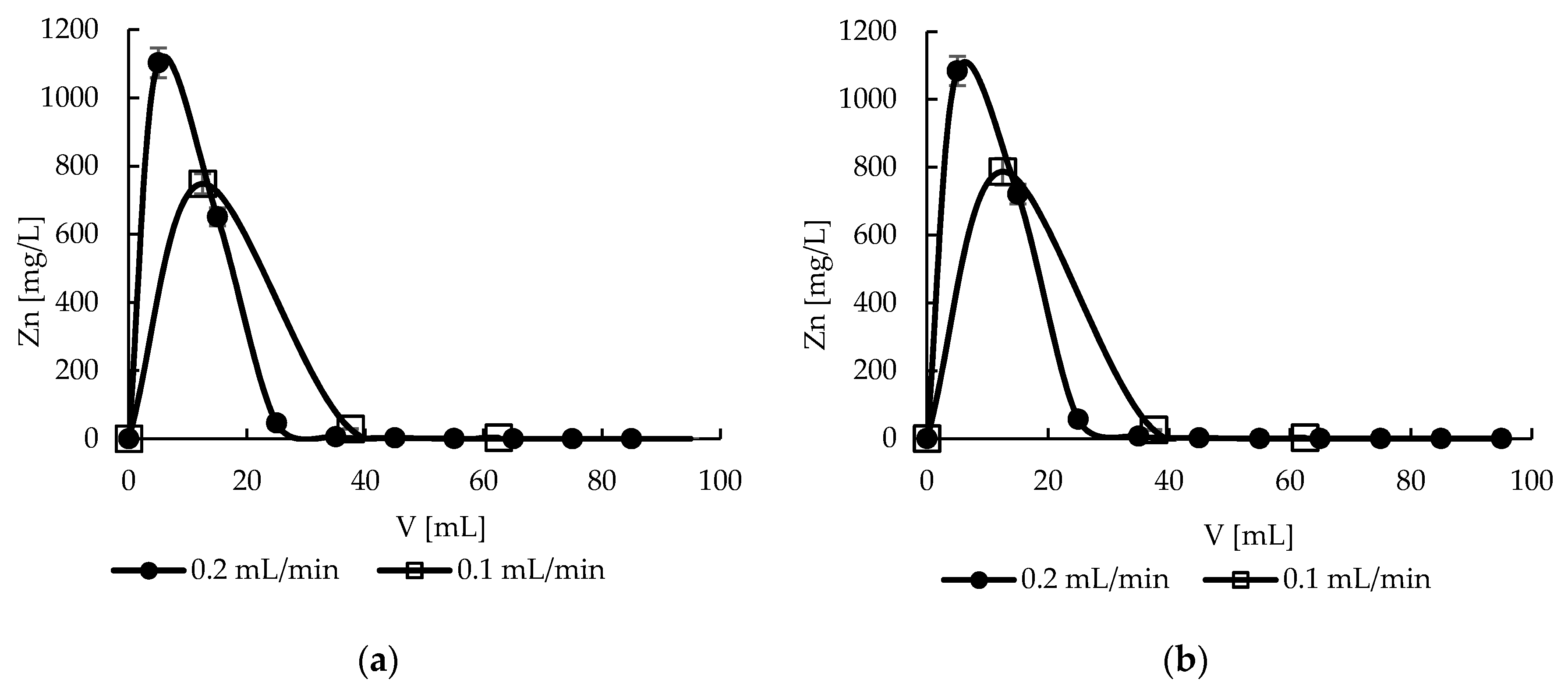
2.10. Desorption—Effect of Flow Rate
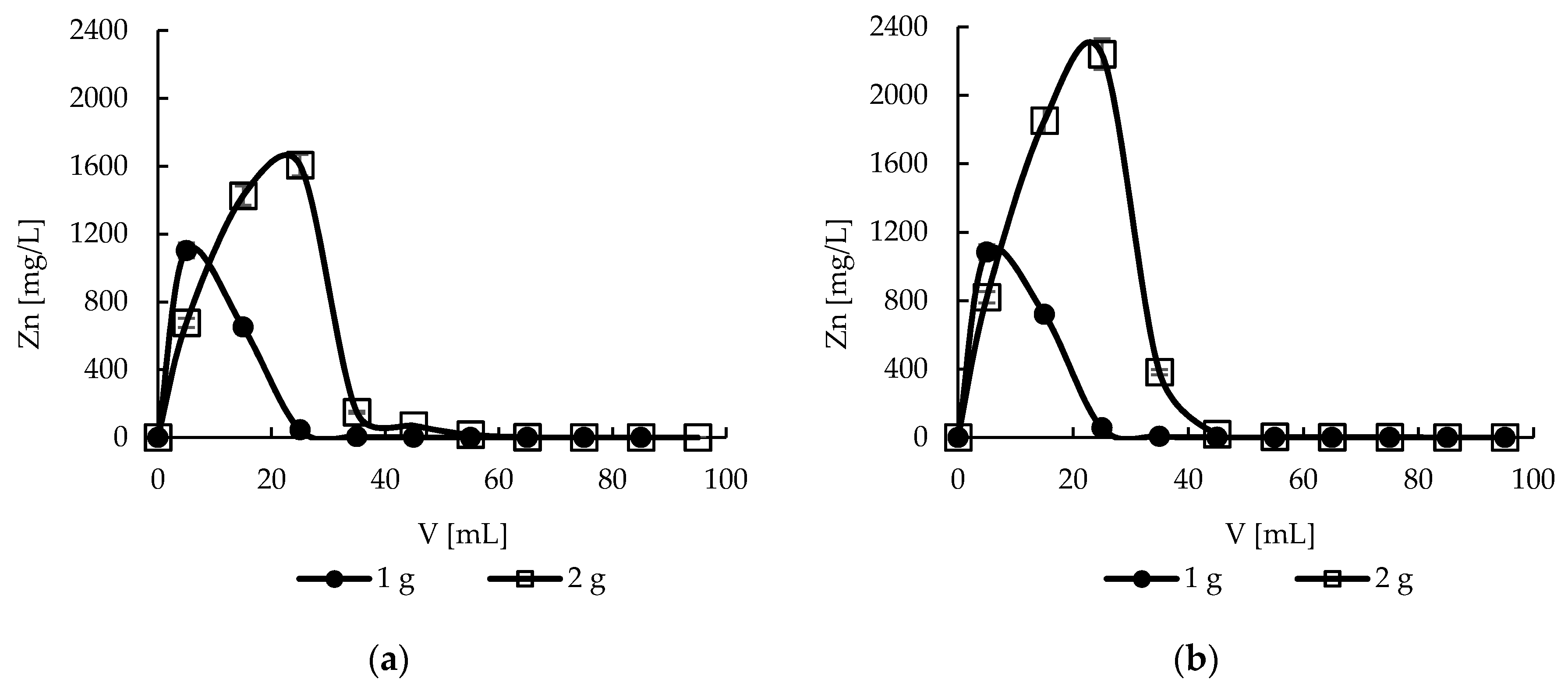
2.11. Desorption—Effect of Bed Height
2.12. Sorption—Desorption—Mass Balance of the Whole Process
3. Materials and Methods
3.1. Materials
3.2. Analytical Method
3.3. Sorption—General Procedure for Batch Studies
3.4. Sorption—Effect of Sorbent Dose
3.5. Sorption—Effect of pH
3.6. Sorption—Kinetics
3.7. Sorption—Isotherms
3.8. Desorption—Equilibrium
3.9. Sorption—General Procedure for Column Studies
3.10. Desorption—General Procedure for Column Studies
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hao, X.; Mohamad, O.A.; Xie, P.; Rensing, C.; Wei, G. Removal of Zinc from Aqueous Solution by Metal Resistant Symbiotic Bacterium Mesorhizobium amorphae. Sep. Sci. Technol. 2014, 49, 376–387. [Google Scholar] [CrossRef]
- Plum, L.M.; Rink, L.; Haase, H. The essential toxin: Impact of zinc on human health. Int. J. Environ. Res. Public Health 2010, 7, 1342–1365. [Google Scholar] [CrossRef] [PubMed]
- Wyszkowska, J.; Kucharski, J.; Tomkiel, M.; Wyszkowska, J.; Baćmaga, M.; Borowik, A.; Boros-Lajszner, E. Changes in the microbiological and biochemical properties of soil contaminated with zinc. J. Elem. 2012, 22, 437–451. [Google Scholar] [CrossRef]
- Directive, E.C. Directive 2010/75/EU of the European Parliament and of the Council. Off. J. Eur. Union L 2010, 334, 17–119. [Google Scholar]
- Polish Ministry of Environment (MŚ). Rozporządzenie Ministra Środowiska w sprawie warunków, jakie należy spełnić przy wprowadzaniu ścieków do wód lub do ziemi, oraz w sprawie substancji szczególnie szkodliwych dla środowiska wodnego. Dz. U. 2014, r. Poz., 1800. [Google Scholar]
- Agency for Toxic Substances and Disease Registry (ATSDR). Regulations and Guidelines Applicable to Zinc and Zinc Compounds; Agency for Toxic Substances and Disease Registry: Atlanta, GA, USA, 2003.
- Castro, L.; Blázquez, M.L.; González, F.; Muñoz, J.A.; Ballester, A. Biosorption of Zn(II) from industrial effluents using sugar beet pulp and F. vesiculosus: From laboratory tests to a pilot approach. Sci. Total Environ. 2017, 598, 856–866. [Google Scholar] [CrossRef] [PubMed]
- Dermentzis, K.K.; Christoforidis, A.; Valsamidou, E. Removal of nickel, copper, zinc and chromium from synthetic and industrial wastewater by electrocoagulation. Int. J. Environ. Sci. 2011, 1, 697–710. [Google Scholar] [CrossRef]
- Kočanová, V.; Cuhorka, J.; Dušek, L.; Mikulášek, P. Application of nanofiltration for removal of zinc from industrial wastewater. Desalin. Water Treat. 2017, 75, 342–347. [Google Scholar] [CrossRef]
- Foroutan, R.; Esmaeili, H.; Rishehri, S.D.; Sadeghzadeh, F.; Mirahmadi, S.; Kosarifard, M.; Ramavandi, B. Zinc, nickel, and cobalt ions removal from aqueous solution and plating plant wastewater by modified Aspergillus flavus biomass: A dataset. Data Brief 2017, 12, 485–492. [Google Scholar] [CrossRef] [PubMed]
- Girczys, J.; Kupich, I. Chemical composition of mine water discharged from Zn-Pb ore mines in view of its management. J. Pol. Miner. Eng. Soc. 2010, 11, 1–8. [Google Scholar]
- Wulan, D.R.; Cahyaningsih, S. Djaenudin Influence of voltage input to heavy metal removal from electroplating wastewater using electrocoagulation process. In Proceedings of the 1st International Symposium on Green Technology for Value Chains, BSD City, Indonesia, 2–3 October 2016; pp. 1–7. [Google Scholar]
- Malkoc, S.; Kaynak, E.; Guven, K. Biosorption of zinc(II) on dead and living biomass of Variovorax paradoxus and Arthrobacter viscosus. Desalin. Water Treat. 2016, 57, 15445–15454. [Google Scholar] [CrossRef]
- Zhou, C.; Gong, X.; Han, J.; Guo, R. Removal of Pb(II) and Zn(II) from Aqueous Solutions by Raw Crab Shell: A Comparative Study. Water Environ. Res. 2016, 88, 374–383. [Google Scholar] [CrossRef] [PubMed]
- Bulgariu, L.; Hlihor, R.M.; Bulgariu, D.; Gavrilescu, M. Sorptive removal of cadmium(II) ions from aqueous solution by mustard biomass. Environ. Eng. Manag. J. 2012, 11, 1969–1976. [Google Scholar]
- Ng, Z.-G.; Lim, J.-W.; Isa, M.H.; Pasupuleti, V.R.; Yunus, N.M.; Lee, K.-C. Adsorptive removal of hexavalent chromium using sawdust: Enhancement of biosorption and bioreduction. Sep. Sci. Technol. 2017, 52, 1707–1716. [Google Scholar] [CrossRef]
- Petrović, M.; Šoštarić, T.; Stojanović, M.; Petrović, J.; Mihajlović, M.; Ćosović, A.; Stanković, S. Mechanism of adsorption of Cu2+ and Zn2+ on the corn silk (Zea mays L.). Ecol. Eng. 2017, 99, 83–90. [Google Scholar] [CrossRef]
- Afroze, S.; Sen, T.K.; Ang, H.M. Adsorption removal of zinc (II) from aqueous phase by raw and base modified Eucalyptus sheathiana bark: Kinetics, mechanism and equilibrium study. Process Saf. Environ. Prot. 2016, 102, 336–352. [Google Scholar] [CrossRef]
- Rodrigues, A.C.D.; do Amaral Sobrinho, N.M.B.; dos Santos, F.S.; dos Santos, A.M.; Pereira, A.C.C.; Lima, E.S.A. Biosorption of Toxic Metals by Water Lettuce (Pistia stratiotes) Biomass. Water. Air. Soil Pollut. 2017, 228, 156. [Google Scholar] [CrossRef]
- Matouq, M.; Jildeh, N.; Qtaishat, M.; Hindiyeh, M.; Al Syouf, M.Q. The adsorption kinetics and modeling for heavy metals removal from wastewater by Moringa pods. J. Environ. Chem. Eng. 2015, 3, 775–784. [Google Scholar] [CrossRef]
- Amer, M.W.; Ahmad, R.A.; Awwad, A.M. Biosorption of Cu(II), Ni(II), Zn(II) and Pb(II) ions from aqueous solution by Sophora japonica pods powder. Int. J. Ind. Chem. 2015, 6, 67–75. [Google Scholar] [CrossRef]
- Ebrahimzadeh Rajaei, G.; Aghaie, H.; Zare, K.; Aghaie, M. Adsorption of Cu(II) and Zn(II) ions from aqueous solutions onto fine powder of Typha latifolia L. root: Kinetics and isotherm studies. Res. Chem. Intermed. 2013, 39, 3579–3594. [Google Scholar] [CrossRef]
- Chaidir, Z.; Zein, R.; Munaf, E. Biosorption of zinc(II) ions from aqueous solution by Androgaphis paniculata leaves powder on batch method. J. Chem. Pharm. Res. 2015, 7, 28–38. [Google Scholar]
- Cutillas-Barreiro, L.; Paradelo, R.; Igrexas-Soto, A.; Núñez-Delgado, A.; Fernández-Sanjurjo, M.J.; Álvarez-Rodriguez, E.; Garrote, G.; Nóvoa-Muñoz, J.C.; Arias-Estévez, M. Valorization of biosorbent obtained from a forestry waste: Competitive adsorption, desorption and transport of Cd, Cu, Ni, Pb and Zn. Ecotoxicol. Environ. Saf. 2016, 131, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Ahluwalia, S.S.; Goyal, D. Microbial and plant derived biomass for removal of heavy metals from wastewater. Bioresour. Technol. 2007, 98, 2243–2257. [Google Scholar] [CrossRef] [PubMed]
- Annadural, G.; Juang, R.S.; Lee, D.J. Adsorption of heavy metals from water using banana and orange peels. Water Sci. Technol. 2003, 47, 185–190. [Google Scholar] [PubMed]
- Balaria, A.; Schiewer, S. Assessment of biosorption mechanism for Pb binding by citrus pectin. Sep. Purif. Technol. 2008, 63, 577–581. [Google Scholar] [CrossRef]
- De Carvalho, R.P.; Chong, K.H.; Volesky, B. Evaluation of the Cd, Cu, and Zn Biosorption in Two-Metal Systems Using an Algal Biosorbent. Biotechnol. Prog. 1995, 11, 39–44. [Google Scholar] [CrossRef]
- Liang, S.; Guo, X.Y.; Feng, N.C.; Tian, Q.H. Effective removal of heavy metals from aqueous solutions by orange peel xanthate. Trans. Nonferr. Met. Soc. China 2010, 20, 187–191. [Google Scholar] [CrossRef]
- Machado, M.D.; Santos, M.S.F.; Gouveia, C.; Soares, H.M.V.M.; Soares, E.V. Removal of heavy metals using a brewer’s yeast strain of Saccharomyces cerevisiae: The flocculation as a separation process. Bioresour. Technol. 2008, 99, 2107–2115. [Google Scholar] [CrossRef] [PubMed]
- Matheickal, J.T.; Yu, Q. Biosorption of lead(II) and copper(II) from aqueous solutions by pre-treated biomass of Australian marine algae. Bioresour. Technol. 1999, 69, 223–229. [Google Scholar] [CrossRef]
- Mishra, V.; Tadepalli, S. Biosorption of Toxic Heavy Metals on Sawdust. CLEAN—Soil Air Water 2015, 43, 360–367. [Google Scholar] [CrossRef]
- Pavan, F.A.; Mazzocato, A.C.; Jacques, R.A.; Dias, S.L.P. Ponkan peel: A potential biosorbent for removal of Pb(II) ions from aqueous solution. Biochem. Eng. J. 2008, 40, 357–362. [Google Scholar] [CrossRef]
- Pehlivan, E.; Altun, T. Biosorption of chromium(VI) ion from aqueous solutions using walnut, hazelnut and almond shell. J. Hazard. Mater. 2008, 155, 378–384. [Google Scholar] [CrossRef] [PubMed]
- Schiewer, S.; Balaria, A. Biosorption of Pb2+ by original and protonated citrus peels: Equilibrium, kinetics, and mechanism. Chem. Eng. J. 2009, 146, 211–219. [Google Scholar] [CrossRef]
- Schiewer, S.; Iqbal, M. The role of pectin in Cd binding by orange peel biosorbents: A comparison of peels, depectinated peels and pectic acid. J. Hazard. Mater. 2010, 177, 899–907. [Google Scholar] [CrossRef] [PubMed]
- Schiewer, S.; Patil, S.B. Modeling the effect of pH on biosorption of heavy metals by citrus peels. J. Hazard. Mater. 2008, 157, 8–17. [Google Scholar] [CrossRef] [PubMed]
- Schiewer, S.; Patil, S.B. Pectin-rich fruit wastes as biosorbents for heavy metal removal: Equilibrium and kinetics. Bioresour. Technol. 2008, 99, 1896–1903. [Google Scholar] [CrossRef] [PubMed]
- Wasewar, K.L. Adsorption of metals onto tea factory waste: A review. Int. J. Res. Rev. Appl. Sci. 2010, 3, 303–322. [Google Scholar]
- Wong, K.K.; Lee, C.K.; Low, K.S.; Haron, M.J. Removal of Cu and Pb from electroplating wastewater using tartaric acid modified rice husk. Process Biochem. 2003, 39, 437–445. [Google Scholar] [CrossRef]
- Jakóbik-Kolon, A.; Milewski, A.; Zdybal, D.; Mitko, K.; Laskowska, E.; Mielańczyk, A.; Bok-Badura, J. Zinc sorption on modified waste poly(methyl methacrylate). Materials 2017, 10, 755. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Lin, X.; Wu, P.; Luo, X. Pectin/Al2O3–ZrO2 core/shell bead sorbent for fluoride removal from aqueous solution. RSC Adv. 2016, 6, 27738–27749. [Google Scholar] [CrossRef]
- Albarelli, J.Q.; Santos, D.T.; Murphy, S.; Oelgemo, M. Use of Ca–alginate as a novel support for TiO2 immobilization in methylene blue decolorisation. Water Sci. Technol. 2009, 60, 1081–1087. [Google Scholar] [CrossRef] [PubMed]
- Hammouda, S.B.; Adhoum, N.; Monser, L. Synthesis of magnetic alginate beads based on Fe3O4 nanoparticles for the removal of 3-methylindole from aqueous solution using Fenton process. J. Hazard. Mater. 2015, 294, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Algothmi, W.M.; Bandaru, N.M.; Yu, Y.; Shapter, J.G.; Ellis, A.V. Alginate-graphene oxide hybrid gel beads: An efficient copper adsorbent material. J. Colloid Interface Sci. 2013, 397, 32–38. [Google Scholar] [CrossRef] [PubMed]
- Pereira, F.A.R.; Sousa, K.S.; Cavalcanti, G.R.S.; França, D.B.; Queiroga, L.N.F.; Santos, I.M.G.; Fonseca, M.G.; Jaber, M. Green biosorbents based on chitosan-montmorillonite beads for anionic dye removal. J. Environ. Chem. Eng. 2017, 5, 3309–3318. [Google Scholar] [CrossRef]
- Munagapati, V.S.; Yarramuthi, V.; Kim, D.S. Methyl orange removal from aqueous solution using goethite, chitosan beads and goethite impregnated with chitosan beads. J. Mol. Liq. 2017, 240, 329–339. [Google Scholar] [CrossRef]
- Kluczka, J.; Gnus, M.; Dudek, G.; Turczyn, R. Removal of Boron from Aqueous Solution by Composite Chitosan Beads. Sep. Sci. Technol. 2017, 52, 1559–1571. [Google Scholar] [CrossRef]
- Dursun, A.Y. A comparative study on determination of the equilibrium, kinetic and thermodynamic parameters of biosorption of copper(II) and lead(II) ions onto pretreated Aspergillus niger. Biochem. Eng. J. 2006, 28, 187–195. [Google Scholar] [CrossRef]
- Jakóbik-Kolon, A.; Mitko, K.; Bok-Badura, J. Zinc sorption studies on pectin-based biosorbents. Materials 2017, 10, 844. [Google Scholar] [CrossRef] [PubMed]
- Butewicz, A.; Campos Gavilan, K.; Pestov, A.V.; Yatluk, Y.; Trochimczuk, A.W.; Guibal, E. Palladium and platinum sorption on a thiocarbamoyl-derivative of Chitosan. J. Appl. Polym. Sci. 2010, 116, 3318–3330. [Google Scholar] [CrossRef]
- Jakóbik-Kolon, A.; Milewski, A.K.; Mitko, K.; Lis, A. Preparation of Pectin-Based Biosorbents for Cadmium and Lead Ions Removal. Sep. Sci. Technol. 2014, 49, 1679–1688. [Google Scholar] [CrossRef]
- Lagergren, S. Zur Theorie der Sogenannten Adsorption Gelöster Stoffe, Kungliga Svenska Vetenskapsakademiens. Handlingar 1898, 24, 1–39. [Google Scholar]
- Ho, Y.S. Citation Review of Lagergren Kinetic Rate Equation on Adsorption Reactions. Scientometrics 2004, 59, 171–177. [Google Scholar]
- Zheng, J.C.; Feng, H.M.; Lam, M.H.W.; Lam, P.K.S.; Ding, Y.W.; Yu, H.Q. Removal of Cu(II) in aqueous media by biosorption using water hyacinth roots as a biosorbent material. J. Hazard. Mater. 2009, 171, 780–785. [Google Scholar] [CrossRef] [PubMed]
- Duong, D.D. Adsorption Analysis: Equilibria and Kinetics; Imperial College Press: London, UK, 1998; Volume 2, ISBN 1860941303. [Google Scholar]
- Moré, J.J. The Levenberg-Marquardt algorithm: Implementation and theory. In Lecture Notes in Mathematics 630: Numerical Analysis; Watson, G.A., Ed.; Springer: Berlin, Germany, 1978; pp. 105–116. [Google Scholar]
- Elzhov, T.V.; Mullen, K.M.; Spiess, A.-N.; Bolker, B. Minpack.lm: R Interface to the Levenberg-Marquardt Nonlinear Least-Squares Algorithm Found in MINPACK, Plus Support for Bounds. Available online: https://cran.r-project.org/package=minpack.lm (accessed on 27 November 2017).
Sample Availability: Samples of the compounds (sole pectin biosorbent, hybrid pectin biosorbent with modified PMMA) are available from the authors. |

| Sorbent | Swelling Index in Water | Swelling Index in Zinc Solution |
|---|---|---|
| P + MPMMA | 2.50 ± 0.03 | 2.43 ± 0.03 |
| P | 2.30 ± 0.01 | 2.50 ± 0.01 |
| Pseudo-First-Order Kinetics for P + MPMMA Sorbent | Pseudo-First-Order Kinetics for P Sorbent | |||||
| 12 °C | 22 °C | 32 °C | 12 °C | 22 °C | 32 °C | |
| R2 | 0.992 | 0.996 | 0.985 | 0.991 | 0.998 | 0.992 |
| qm (mg/g) | 17.60 ± 0.80 | 17.63 ± 0.24 | 17.51 ± 0.58 | 16.43 ± 0.61 | 16.65 ± 0.18 | 16.94 ± 0.37 |
| k1·103 (1/min) | 7.53 ± 0.94 | 13.19 ± 0.65 | 14.22 ± 1.71 | 9.86 ± 1.16 | 12.55 ± 0.48 | 16.98 ± 1.41 |
| Pseudo-Second-Order Kinetics for P + MPMMA Sorbent | Pseudo-Second-Order Kinetics for P Sorbent | |||||
| 12 °C | 22 °C | 32 °C | 12 °C | 22 °C | 32 °C | |
| R2 | 0.990 | 0.992 | 0.986 | 0.989 | 0.992 | 0.992 |
| qm (mg/g) | 22.69 ± 1.35 | 21.50 ± 0.68 | 20.77 ± 0.78 | 20.39 ± 0.96 | 20.41 ± 0.64 | 19.80 ± 0.48 |
| k2·104 (g/mg∙min) | 3.14 ± 0.67 | 6.24 ± 0.83 | 7.67 ± 1.27 | 4.97 ± 0.92 | 6.21 ± 0.82 | 9.72 ± 1.07 |
| Calculated Parameters of Langmuir Isotherm | Calculated Parameters of Freundlich Isotherm | ||||
| P + MPMMA | P | P + MPMMA | P | ||
| qm (mg/g) | 35.03 ± 2.21 | 42.17 ± 2.43 | Kf ((mg/g)(L/mg)1/n) | 15.46 ± 0.27 | 9.92 ± 0.20 |
| B (L/mg) | 0.93 ± 0.22 | 0.26 ± 0.04 | n | 2.61 ± 0.07 | 1.97 ± 0.04 |
| R2 | 0.973 | 0.992 | R2 | 0.996 | 0.98 |
| Calculated Parameters of Bi-Site Langmuir Isotherm | |||||
| P + MPMMA | |||||
| qm1 (mg/g) | 50.2 ± 11.5 | ||||
| qm2 (mg/g) | 11.8 ± 1.7 | ||||
| B1 (L/mg) | 0.10 ± 0.05 | ||||
| B2 (L/mg) | 12.1 ± 3.9 | ||||
| R2 | 0.996 | ||||
| Sorbent | (1) Zn Introduced to the Column (mg) | (2) Zn in Effluent (Sorption Process) (mg) | (3) Zn in Effluent (Desorption Process) (mg) | (4) the Sum of (2) and (3) (mg) | Recovery (%) |
|---|---|---|---|---|---|
| P + MPMMA | 126.58 | 45.92 | 82.68 | 128.61 | 101.60 |
| P | 128.67 | 52.07 | 78.08 | 130.15 | 101.15 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jakóbik-Kolon, A.; Szybaj, A.; Mitko, K.; Bok-Badura, J. Zinc Ion Removal on Hybrid Pectin-Based Beads Containing Modified Poly(Methyl Methacrylate) Waste. Molecules 2017, 22, 2274. https://doi.org/10.3390/molecules22122274
Jakóbik-Kolon A, Szybaj A, Mitko K, Bok-Badura J. Zinc Ion Removal on Hybrid Pectin-Based Beads Containing Modified Poly(Methyl Methacrylate) Waste. Molecules. 2017; 22(12):2274. https://doi.org/10.3390/molecules22122274
Chicago/Turabian StyleJakóbik-Kolon, Agata, Adrianna Szybaj, Krzysztof Mitko, and Joanna Bok-Badura. 2017. "Zinc Ion Removal on Hybrid Pectin-Based Beads Containing Modified Poly(Methyl Methacrylate) Waste" Molecules 22, no. 12: 2274. https://doi.org/10.3390/molecules22122274
APA StyleJakóbik-Kolon, A., Szybaj, A., Mitko, K., & Bok-Badura, J. (2017). Zinc Ion Removal on Hybrid Pectin-Based Beads Containing Modified Poly(Methyl Methacrylate) Waste. Molecules, 22(12), 2274. https://doi.org/10.3390/molecules22122274







