Composition and Antibacterial Activity of the Essential Oils of Orthosiphon stamineus Benth and Ficus deltoidea Jack against Pathogenic Oral Bacteria
Abstract
:1. Introduction
2. Results
2.1. Analysis of the Components of the Essential Oils
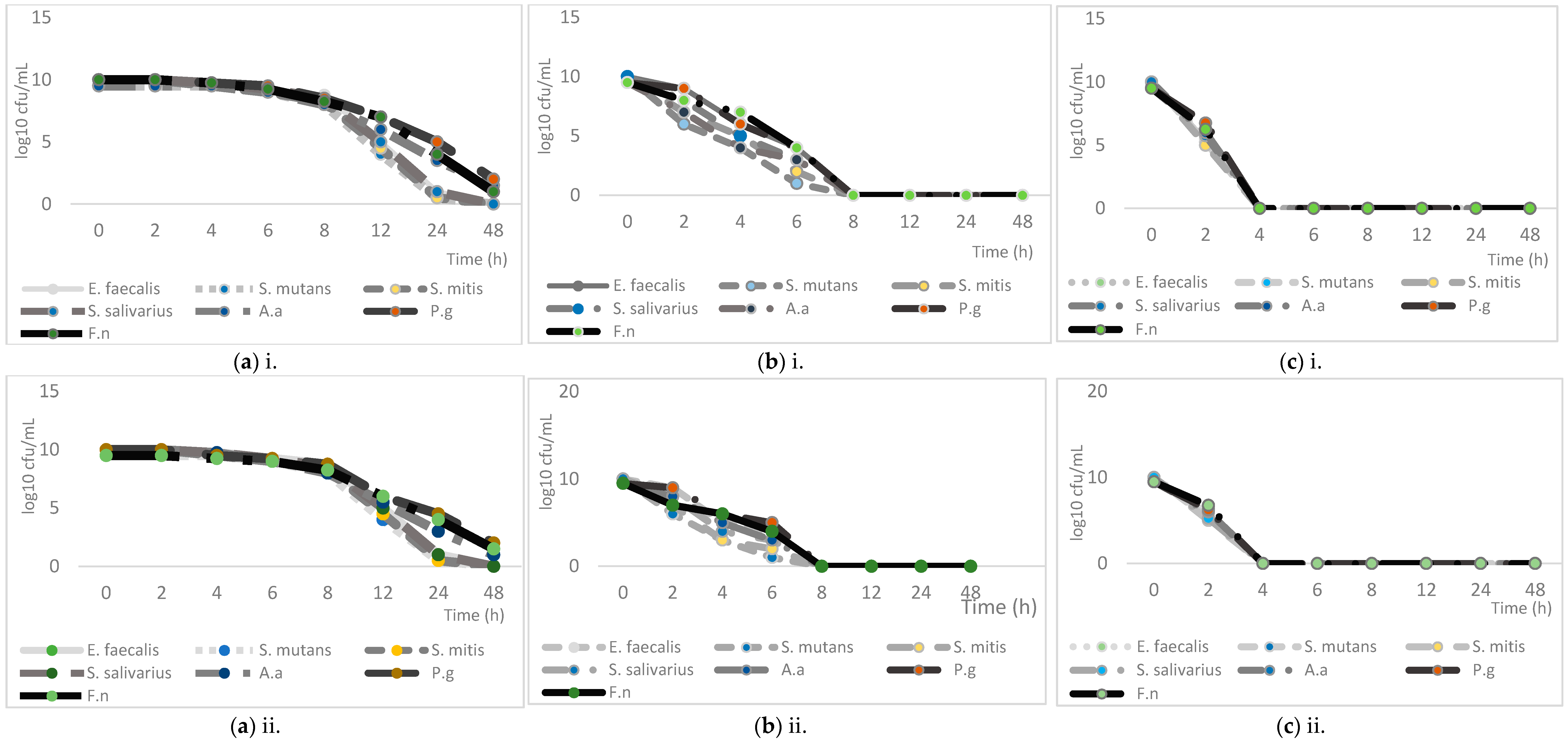
2.2. Antibacterial Activity of the Essential Oils and Their Major Compounds
2.3. Morphological Changes of the Bacterial Cells
2.4. Herbal Oils and Amoxicillin Synergistic Effect
3. Discussion
4. Materials and Methods
4.1. Chemicals and Reagents
4.2. Plant Materials and Preparation of Oils
4.3. Gas Chromatographic (GC) Analyses of the Essential Oils
4.4. Bacterial Culture and Maintenance of Growth
4.5. Determination of Minimum Inhibitory Concentration (MIC) and Minimum Bactericidal Concentration (MBC)
4.6. Checkerboard Assay and Time-Kill Assay
- (i)
- The FIC of oil (FICA) = MIC combination/MIC alone.
- (ii)
- The FIC of antibiotic (FICB) = MIC combination/MIC alone.
- (iii)
- FIC index = FICA + FICB (FIC index of the combination in each well is the sum of the FIC for each drug (test sample and antibiotic) present in the well).
4.7. Anti-Adhesion and Anti-Biofilm Assays
4.8. Observation under the Field Emission Scanning Electron Microscope (FESEM)
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Curtis, M.A.; Zenobia, C.; Darveau, R.P. The relationship of the oral microbiotia to periodontal health and disease. Cell Host Microbe 2011, 10, 302–306. [Google Scholar] [CrossRef] [PubMed]
- Aas, J.A.; Griffen, A.L.; Dardis, S.R.; Lee, A.M.; Olsen, I.; Dewhirst, F.E.; Leys, E.J.; Paster, B.J. Bacteria of dental caries in primary and permanent teeth in children and young adults. J. Clin. Microbiol. 2008, 46, 1407–1417. [Google Scholar] [CrossRef] [PubMed]
- Saito, D.; Leonardo Rde, T.; Rodrigues, J.L.; Tsai, S.M.; Höfling, J.F.; Gonçalves, R.B. Identification of bacteria in endodontic infections by sequence analysis of 16S rDNA clone libraries. J. Med. Microbiol. 2006, 55, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Narayanan, L.L.; Vaishnavi, C. Endodontic microbiology. J. Conserv. Dent. 2010, 13, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Petersen, P.E.; Bourgeois, D.; Ogawa, H.; Estupinan-Day, S.; Ndiaye, C. The global burden of oral diseases and risks to oral health. Bull. World Health Organ. 2005, 83, 661–669. [Google Scholar] [PubMed]
- Rautemaa, R.; Lauhio, A.; Cullinan, M.P.; Seymour, G.J. Oral infections and systemic disease—An emerging problem in medicine. Clin. Microbiol. Infect. 2007, 13, 1041–1047. [Google Scholar] [CrossRef] [PubMed]
- Kutsch, V.K. Dental caries: An updated medical model of risk assessment. J. Prosthet. Dent. 2014, 111, 280–285. [Google Scholar] [CrossRef] [PubMed]
- Edris, A.E. Pharmaceutical and therapeutic potentials of essential oils and their individual volatile constituents: A review. Phytother. Res. 2007, 21, 308–323. [Google Scholar] [CrossRef] [PubMed]
- Lahlou, M. Methods to study the phytochemistry and bioactivity of essential oils. Phytother. Res. 2004, 18, 435–448. [Google Scholar] [CrossRef] [PubMed]
- Palombo, E.A. Traditional medicinal plant extracts and natural products with activity against oral bacteria: Potential application in the prevention and treatment of oral diseases. Evid. Based Complement. Altern. Med. 2011. [Google Scholar] [CrossRef] [PubMed]
- Himani, B.; Seema, B.; Bhole, N.; Mayank, Y.; Vinod, S.; Mamta, S. Misai Kuching: A glimpse of maestro. Int. J. Pharm. Sci. Rev. Res. 2013, 22, 55–59. [Google Scholar]
- Amer, O.Z.; Salman, I.M.; Asmawi, M.Z.; Ibraheem, Z.O.; Yam, M.F. Orthosiphon stamineus: Traditional uses, phytochemistry, pharmacology, and toxicology: A review. J. Med. Food 2012, 15, 678–690. [Google Scholar] [CrossRef] [PubMed]
- Adam, Z.; Khamis, S.; Ismail, A.; Hamid, M. Ficus deltoidea: A potential alternative medicine for diabetes mellitus. Evid. Based Complement. Altern. Med. 2012, 2012, 632763. [Google Scholar] [CrossRef] [PubMed]
- Lansky, E.P.; Paavilainen, H.M.; Pawlus, A.D.; Newman, R.A. Ficus spp. (fig): Ethnobotany and potential as anticancer and anti-inflammatory agents. J. Ethnopharmacol. 2008, 119, 195–213. [Google Scholar] [CrossRef] [PubMed]
- Abdullah, Z.; Hussain, K.; Zhari1, I.; Rasadah, M.A.; Mazura, P.; Jamaludin, F.; Sahdan, R. Evaluation of extracts of leaf of three Ficus deltoidea varieties for antioxidant activities and secondary metabolites. Pharmacogn. Res. 2009, 1, 216–223. [Google Scholar]
- Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Testing; Nineteenth Informational Supplement M100–S19; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2009. [Google Scholar]
- Zengin, H.; Baysal, A.H. Antibacterial and antioxidant activity of essential oil terpenes against pathogenic and spoilage-forming bacteria and cell structure-activity relationships evaluated by SEM microscopy. Molecules 2014, 19, 17773–17798. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Hu, D.H.; Feng, Y. Antibacterial activity and mode of action of the Artemisia capillaris essential oil and its constituents against respiratory tract infection-causing pathogens. Mol. Med. Rep. 2015, 11, 2852–2860. [Google Scholar] [CrossRef] [PubMed]
- European Committee for Antimicrobial Susceptibility Testing (EUCAST) of the European Society of Clinical Microbiology and Infectious Diseases. Terminology Relating to Methods for the Determination of Susceptibility of Bacteria to Antimicrobial Agents; EUCAST Definitive Document E. Def 1.2; European Committee for Antimicrobial Susceptibility Testing (EUCAST) of the European Society of Clinical Microbiology and Infectious Diseases (ESCMID): Basel, Switzerland, 2000. [Google Scholar]
- Sakharkar, M.K.; Jayaraman, P.; Soe, W.M.; Chow, V.T.K.; Sing, L.C.; Sakharkar, K.R. In vitro combinations of antibiotics and phytochemicals against Pseudomonas aeruginosa. J. Microbiol. Immunol. Infect. 2009, 42, 364–370. [Google Scholar] [PubMed]
- Hossain, M.A.; Ismail, Z.; Rahman, A.; Kang, S.C. Chemical composition and anti-fungal properties of the essential oils and crude extracts of Orthosiphon stamineus Benth. Ind. Crop Prod. 2008, 27, 328–334. [Google Scholar] [CrossRef]
- Grison-Pigé, L.; Bessière, J.-M.; Hossaert-McKey, M. Specific attraction of fig-pollinating wasps: Role of volatile compounds released by tropical figs. J. Chem. Ecol. 2002, 28, 283–295. [Google Scholar] [CrossRef] [PubMed]
- Koay, Y.C.; Amir, F. A survey of the chemical constituents and biological activities of Orthosiphon stamineus. Sci. Int. 2012, 24, 133–138. [Google Scholar]
- Bush, K. β-lactam antibiotics: Penicillin, and other β-lactam antibiotics. In Antibiotic and Chemotherapy: Anti-Infective Agents and Their Use in Therapy, 8th ed.; Finch, R.G., Greenwood, D., Norrby, S.R., Whitley, R.J., Eds.; Churchill Livingstone: Philadelphia, PA, USA, 2003; pp. 224–278. [Google Scholar]
- Kaur, S.P.; Rao, R.; Nanda, S. Amoxicillin: A broad spectrum antibiotic. Int. J. Pharm. Pharm. Sci. 2011, 3, 30–37. [Google Scholar]
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods—A review. Int. J. Food Microbiol. 2004, 94, 223–253. [Google Scholar] [CrossRef] [PubMed]
- Schelz, Z.; Molnar, J.; Hohmann, J. Antimicrobial and antiplasmid activities of essential oils. Fitoterapia 2006, 77, 279–285. [Google Scholar] [CrossRef] [PubMed]
- Di Pasqua, R.; Betts, G.; Hoskins, N.; Edwards, M.; Ercolini, D.; Mauriello, G. Membrane toxicity of antimicrobial compounds from essential oils. J. Agric. Food Chem. 2007, 55, 4863–4870. [Google Scholar] [CrossRef] [PubMed]
- Carson, C.F.; Mee, B.J.; Riley, T.V. Mechanism of action of Melaleuca alternifolia (Tea tree) oil on Staphylococcus aureus determined by time-kill, lysis, leakege, and salt tolerance assays and electron microscopy. Antimicrob. Agent Chemother. 2002, 46, 1914–1920. [Google Scholar] [CrossRef]
- Budavari, S. (Ed.) The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals, 12th ed.; Merck & Co Inc.: Kenilworth, NJ, USA, 1996. [Google Scholar]
- Baser, K.H.; Demirci, B.; Iscan, G.; Hashimoto, T.; Demirci, F.; Noma, Y.; Asakawa, Y. The essential oil constituents and antimicrobial activity of Anthemis aciphylla BOISS. var. discoidea BOISS. Chem. Pharm. Bull. 2006, 54, 222–225. [Google Scholar] [CrossRef] [PubMed]
- Pichette, A.; Larouche, P.-L.; Lebrun, M.; Legault, J. Composition and antibacterial activity of Abies balsamea essential oil. Phytother. Res. 2006, 20, 371–373. [Google Scholar] [CrossRef] [PubMed]
- Sabulal, B.; Dan, M.; John, J.A.; Kurup, R.; Pradeep, N.S.; Valsamma, R.K.; George, V. Caryophyllene-rich rhizome oil of Zingiber nimmonii from South India: Chemical characterization and antimicrobial activity. Phytochemistry 2006, 67, 2469–2473. [Google Scholar] [CrossRef] [PubMed]
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological effects of essential oils. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef] [PubMed]
- Bassolé, I.H.N.; Lamien-Meda, A.; Bayala, B.; Tirogo, S.; Franz, C.; Novak, J.; Nebié, R.C.; Dicko, M.H. Composition and antimicrobial activities of Lippia multiflora Moldenke, Mentha x piperita L. and Ocimum basilicum L. essential oils and their major monoterpene alcohols alone and in combination. Molecules 2010, 15, 7825–7839. [Google Scholar] [CrossRef] [PubMed]
- Koutsoudaki, C.; Krsek, M.; Rodger, A. Chemical composition and antibacterial activity of the essential oil and the gum of Pistacia lentiscus Var. chia. J. Agric. Food Chem. 2005, 53, 7681–7685. [Google Scholar] [CrossRef] [PubMed]
- Sökmen, A.; Gulluce, M.; Akpulat, H.A.; Daferera, D.; Tepe, B.; Polissiou, M. The in vitro antimicrobial and antioxidant activities of the essential oils and methanol extracts of endemic Thymus spathulifolius. Food Control 2004, 15, 627–634. [Google Scholar] [CrossRef]
- Sokovic, M.; Van Griensven, L.J.L.D. Antimicrobial activity of essential oils and their components against the three major pathogens of the cultivated button mushroom, Agaricus bisporus. Eur. J. Plant Pathol. 2006, 116, 211–224. [Google Scholar] [CrossRef]
- Tabanca, N.; Kirimer, N.; Demirci, B.; Demirci, F.; Baser, K.H.C. Composition and antimicrobial activity of the essential oils of Micromeria cristata subsp. phyrgia and the enantiomeric distribution of borneol. J. Agric. Food Chem. 2001, 49, 4300–4303. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, A.C.R.; Lopes, P.M.; de Azevedo, M.M.B.; Costa, D.C.M.; Alviano, C.S.; Alviano, D.S. Biological activities of α-pinene and β-pinene enantiomers. Molecules 2012, 17, 6305–6316. [Google Scholar] [CrossRef] [PubMed]
- Oussalah, M.; Caillet, S.; Lacroix, M. Mechanism of action of Spanish oregano, Chinese cinnamon, and savory essential oils against cell membranes and walls of Escherichia coli O157:H7 and Listeria monocytogenes. J. Food Prot. 2006, 69, 1046–1055. [Google Scholar] [CrossRef] [PubMed]
- Bunawan, H.; Amin, N.M.; Bunawan, S.N.; Baharum, S.N.; Noor, N.M. Ficus deltoidea Jack: A review on its phytochemical and pharmacological importance. Evid. Based Complement. Altern. Med. 2014. [Google Scholar] [CrossRef] [PubMed]
- Rhayour, K.; Bouchikhi, T.; Tantaoui-Elaraki, A.; Sendide, K.; Remmal, A. The mechanism of bactericidal action of oregano and clove essential oils and of their phenolic major components on Escherichia coli and Bacillus subtilis. J. Essent. Oil Res. 2003, 5, 286–292. [Google Scholar] [CrossRef]
- Fine, D.H.; Furgang, D.; Sinatra, K.; Charles, C.; McGuire, A.; Kumar, L.D. In vivo antimicrobial effectiveness of an essential oil-containing mouth rinse 12 h after a single use and 14 days’ use. J. Clin. Periodontol. 2005, 32, 335–340. [Google Scholar] [CrossRef] [PubMed]
- Lauten, J.D.; Boyd, L.; Hanson, M.B.; Lillie, D.; Gullion, C.; Madden, T.E. A clinical study: Melaleuca, manuka, calendula and green tea mouth rinse. Phytother. Res. 2005, 19, 951–957. [Google Scholar] [CrossRef] [PubMed]
- Sharma, N.C.; Araujo, M.W.B.; Wu, M.M.; Qaqish, J.; Charles, C.H. Superiority of an essential oil mouthrinse when compared with a 0.05% cetylpyridinium chloride containing mouthrinse: A six-month study. Int. Dent. J. 2010, 60, 175–180. [Google Scholar] [PubMed]
- Fine, D.H.; Markowitz, K.; Furgang, D.; Goldsmith, D.; Ricci-Nittel, D.; Charles, C.H.; Peng, P.; Lynch, M.C. Effect of rinsing with an essential oil–containing mouthrinse on subgingival periodontopathogens. J. Periodontol. 2007, 78, 1935–1942. [Google Scholar] [CrossRef] [PubMed]
- Stoeken, J.E.; Paraskevas, S.; van der Weijden, G.A. The long-term effect of a mouthrinse containing essential oils on dental plaque and gingivitis: A systematic review. J. Periodontol. 2007, 78, 1218–1228. [Google Scholar] [CrossRef] [PubMed]
- Jantan, I.; Mohd Yassin, M.S.; Chen, B.C.; Lau, L.C.; Ng, L.S. Antifungal Activity of the Essential Oils of Nine Zingiberaceae Species. Pharm. Biol. 2003, 41, 392–397. [Google Scholar] [CrossRef]
- Clinical and Laboratory Standards Institute. M11-A6 Methods for Antimicrobial Susceptibility Testing of Anaerobic Bacteria, 6th ed.; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2004. [Google Scholar]
- Clinical and Laboratory Standards Institute. M07-A9 Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically, 9th ed.; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2012. [Google Scholar]
- Clinical and Laboratory Standards Institute. M100-S25 Performance Standards for Antimicrobial Susceptibility Testing, 25th ed.; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2015. [Google Scholar]
- Coyle, M.B. Manual of Antimicrobial Susceptibility Testing; American Society for Microbiology: Washington, DC, USA, 2005. [Google Scholar]
- Odds, F.C. Synergy, antagonism, and what the chequerboard puts between them. J. Antimicrob. Chemother. 2003, 52, 1. [Google Scholar] [CrossRef] [PubMed]
- Basri, D.F.; Chan, K.L.; Azmi, A.M.; Latip, J. Evaluation of the combined effects of stilbenoid from Shorea gibbosa and vancomycin against Methicillin-Resistant Staphylococcus aureus (MRSA). Pharmaceuticals 2012, 5, 1032–1043. [Google Scholar] [CrossRef] [PubMed]
- Kahraman, G.U.; Mervi, G.; Vedat, G.O.; Lutfu, C.; Eija, K.; Veli-Jukka, U. Anti-biofilm properties of Satureja hortensis L. essential oil against periodontal pathogens. Anaerobe 2009, 15, 164–167. [Google Scholar]
- Faraz, N.; Zia-Ul-Islam; Rehman, R.; Sehrish. Antibiofilm forming activity of naturally occurring compound. Biomedica 2012, 28, 171–175. [Google Scholar]
- Adukwu, E.C.; Allen, S.C.H.; Phillips, C.A. The anti-biofilm activity of lemongrass (Cymbopogon flexuosus) and grapefruit (Citrus paradisi) essential oils against five strains of Staphylococcus aureus. J. Appl. Microbiol. 2012, 113, 1217–1227. [Google Scholar] [CrossRef] [PubMed]
- Zainal-Abidin, Z.; Mohd-Said, S.; Abdul Majid, F.A.; Wan Mustapha, W.A.; Jantan, I. Anti-Bacterial Activity of Cinnamon Oil on Oral Pathogens. Open Conf. Proc. J. 2013, 4, 12–16. [Google Scholar] [CrossRef]
Sample Availability: Samples of the essential oils are available from the authors. |
| No. | Compound | O. stamineus | F. deltoidea | Kovat Index Value | Methods of Identification |
|---|---|---|---|---|---|
| Percentage Composition | Percentage Composition | ||||
| 1 | α-Pinene | 0.1 | - | 939 | a, b |
| 2 | Camphene | 0.05 | - | 954 | a, b |
| 3 | 1-Octen-3-ol | 0.35 | - | 979 | a, b |
| 4 | 1,3-Benzenediamine | 0.1 | - | 1169 | a, b |
| 5 | Naphthalene | 3.8 | - | 1181 | a, b |
| 6 | trans-Geraniol | 0.3 | - | 1253 | a, b |
| 7 | Anethole | 1.1 | - | 1253 | a, b |
| 8 | Cinnamaldehyde | 0.4 | - | 1270 | a, b |
| 9 | Bornyl acetate | 0.16 | - | 1292 | a, b |
| 10 | 1-Methylnaphthalene | 0.55 | - | 1289 | a, b |
| 11 | 2-Methylnaphthalene | 0.5 | - | 1312 | a, b |
| 12 | α-Cubebene | 0.98 | 1.55 | 1351 | a, b |
| 13 | Eugenol | 8.1 | - | 1359 | a, b |
| 14 | α-Copaene | 2.26 | 3.3 | 1377 | a, b |
| 15 | β-Bourbonene | 0.57 | 0.09 | 1388 | a, b |
| 16 | Methyl eugenol | 7.4 | - | 1392 | a, b |
| 17 | 7-Tetradecane | 0.34 | - | 1400 | a, b |
| 18 | β-Caryophyllene | 26.31 | 36.3 | 1409 | a, b |
| 19 | E-β-Damascenone | 0.6 | - | 1414 | a, b |
| 20 | γ-Elemene | 0.25 | - | 1437 | a, b |
| 21 | Aromadendrene | 0.9 | - | 1441 | a, b |
| 22 | α-Humulene | 5.06 | 5.5 | 1455 | a, b |
| 23 | γ-Muurolene | 0.2 | 0.39 | 1480 | a, b |
| 24 | Bicyclogermacrene | 7.68 | - | 1500 | a, b |
| 25 | β-Bisabolene | 0.4 | - | 1506 | a, b |
| 26 | cis-Z-α-Bisabolene epoxide | 0.4 | - | 1507 | a, b |
| 27 | trans-Calamenene | 0.5 | - | 1529 | a, b |
| 28 | α-Calacorene | 0.6 | - | 1546 | a, b |
| 29 | Dimethyl-ionone | 0.67 | - | 1567 | a, b |
| 30 | Caryophyllene oxide | 1.2 | 8.7 | 1583 | a, b |
| 31 | (Z)-β-Ocimene | - | 0.1 | 1037 | a, b |
| 32 | Linalool | - | 0.4 | 1097 | a, b |
| 33 | α-Ylangene | - | 0.14 | 1375 | a, b |
| 34 | β-Elemene | - | 0.1 | 1391 | a, b |
| 35 | α-Santalene | - | 0.1 | 1418 | a, b |
| 36 | β-Copaene | - | 0.3 | 1432 | a, b |
| 37 | α-trans-Bergamotene | - | 0.4 | 1435 | a, b |
| 38 | β-Farnesene | - | 0.5 | 1457 | a, b |
| 39 | Alloaromadandrene | - | 1.5 | 1460 | a, b |
| 40 | Germacrene D | - | 7.68 | 1485 | a, b |
| 41 | β-Selinene | - | 1.1 | 1490 | a, b |
| 42 | α-Selinene | - | 1.03 | 1498 | a, b |
| 43 | α-Muurolene | - | 0.5 | 1500 | a, b |
| 44 | α-Farnesene | - | 1.1 | 1506 | a, b |
| 45 | Germacrene A | - | 1.0 | 1509 | a, b |
| 46 | Myristicin | - | 1.7 | 1519 | a, b |
| 47 | δ-Cadinene | - | 1.3 | 1523 | a, b |
| 48 | α-Cadinene | - | 1.6 | 1539 | a, b |
| 49 | Germacrene B | - | 1.5 | 1561 | a, b |
| 50 | Dendrolasin | - | 0.7 | 1572 | a, b |
| 51 | Himachalene epoxide | - | 0.1 | 1580 | a, b |
| 52 | cis-β-Elemonene | - | 0.2 | 1590 | a, b |
| 53 | Humulene epoxide | - | 0.1 | 1608 | a, b |
| 54 | β-Himachalene oxide | - | 1.4 | 1616 | a, b |
| 55 | α-Muurolol | - | 0.3 | 1646 | a, b |
| 56 | α-Cadinol | - | 0.2 | 1654 | a, b |
| 57 | z-α-Santalol | - | 0.1 | 1675 | a, b |
| 58 | α-Bisabolol | - | 0.2 | 1686 | a, b |
| 59 | epi-β-Bisabolol | - | 0.3 | 1672 | a, b |
| 60 | z-epi-β-Santalol | - | 0.1 | 1703 | a, b |
| 61 | α-Vetivone | - | 0.1 | 1823 | a, b |
| 62 | n-Hexadecanol | - | 0.1 | 1876 | a, b |
| Samples | O. stamineus Essential Oil a | F. deltoidea Essential Oil a | trans-Caryophyllene a | α-Humulene a | Eugenol a | Germacrene D a | Amoxicillin b | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bacteria | MIC (mg/mL) | MBC (mg/mL) | Anti-Adhesion (mg/mL) | MIC (mg/mL) | MBC (mg/mL) | Anti-Adhesion (mg/mL) | MIC (mg/mL) | MBC (mg/mL) | Anti-Adhesion (mg/mL) | MIC (mg/mL) | MBC (mg/mL) | Anti-Adhesion (mg/mL) | MIC (mg/mL) | MBC (mg/mL) | Anti-Adhesion (mg/mL) | MIC (mg/mL) | MBC (mg/mL) | Anti-Adhesion (mg/mL) | MIC And MBC (mg/mL) | Anti-Adhesion (mg/mL) |
| E. faecalis | 1.25 | 2.5 | 2.5 | 1.25 | 2.5 | 2.5 | 1.25 | 2.5 | 2.5 | 1.25 | 2.5 | 2.5 | 1.25 | 2.5 | 2.5 | 1.25 | 2.5 | 2.5 | 0.05 | 0.5 |
| S. mutans | 1.25 | 2.5 | 2.5 | 1.25 | 2.5 | 2.5 | 1.25 | 2.5 | 2.5 | 1.25 | 2.5 | 2.5 | 1.25 | 2.5 | 2.5 | 1.25 | 2.5 | 2.5 | 0.05 | 0.5 |
| S. mitis | 1.25 | 2.5 | 2.5 | 1.25 | 2.5 | 2.5 | 1.25 | 2.5 | 2.5 | 1.25 | 2.5 | 2.5 | 1.25 | 2.5 | 2.5 | 1.25 | 2.5 | 2.5 | 0.05 | 0.5 |
| S. salivarius | 1.25 | 2.5 | 2.5 | 1.25 | 2.5 | 2.5 | 0.63 | 1.25 | 1.25 | 0.63 | 1.25 | 1.25 | 1.25 | 2.5 | 2.5 | 1.25 | 2.5 | 2.5 | 0.05 | 0.5 |
| A. actinomy. | 1.25 | 2.5 | 2.5 | 0.63 | 1.25 | 1.25 | 0.63 | 1.25 | 1.25 | 0.63 | 1.25 | 1.25 | 1.25 | 2.5 | 2.5 | 1.25 | 2.5 | 2.5 | 0.05 | 0.5 |
| P. gingivalis | 1.25 | 2.5 | 2.5 | 0.63 | 1.25 | 1.25 | 0.63 | 1.25 | 1.25 | 0.63 | 1.25 | 1.25 | 1.25 | 2.5 | 2.5 | 1.25 | 2.5 | 2.5 | 0.05 | 0.5 |
| F. nucleatum | 1.25 | 2.5 | 2.5 | 0.63 | 1.25 | 1.25 | 0.63 | 1.25 | 1.25 | 0.63 | 1.25 | 1.25 | 1.25 | 2.5 | 2.5 | 1.25 | 2.5 | 2.5 | 0.05 | 0.5 |
| Treatment | E. faecalis | S. mutans | S. mitis | S. salivarius |
|---|---|---|---|---|
| Untreated bacterial cell (normal) |  |  |  |  |
| Positive control (treated with Amoxicillin) a |  |  |  |  |
| Bacterial cell treated with O. stamineus oil b |  |  |  |  |
| Bacterial cell treated with F. deltoidea oil c |  |  |  |  |
| Treatment | A. actinomycetemcomitans | P. gingivalis | F. nucleatum |
|---|---|---|---|
| Untreated bacterial cell (normal) |  |  | 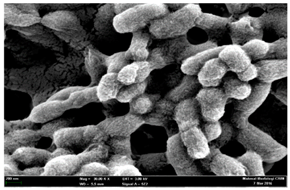 |
| Positive control (treated with Amoxicillin) a |  |  |  |
| Bacterial cell treated with O. stamineus oil b |  |  |  |
| Bacterial cell treated with F. deltoidea oil c |  |  |  |
| Bacteria | Essential Oils | MIC (mg/mL) | FIC Index e | Activity | |||
|---|---|---|---|---|---|---|---|
| Alone (Essential Oil) a | Alone (Amoxicillin) b | Combination (Essential Oil) c | Combination (EO + Amoxicillin) d | ||||
| E. faecalis | O. stamineus | 1.25 | 0.05 | 0.625 | 0.0125 | 0.75 | additive |
| F. deltoidea | 1.25 | 0.05 | 0.625 | 0.0125 | 0.75 | additive | |
| S. mutans | O. stamineus | 1.25 | 0.05 | 0.625 | 0.0125 | 0.75 | additive |
| F. deltoidea | 1.25 | 0.05 | 0.625 | 0.0125 | 0.75 | additive | |
| S. mitis | O. stamineus | 1.25 | 0.05 | 0.625 | 0.0125 | 0.75 | additive |
| F. deltoidea | 1.25 | 0.05 | 0.625 | 0.0125 | 0.75 | additive | |
| S. salivarius | O. stamineus | 1.25 | 0.05 | 0.625 | 0.0125 | 0.75 | additive |
| F. deltoidea | 0.63 | 0.05 | 0.625 | 0.0125 | 0.75 | additive | |
| A. actinomycetemcomitans | O. stamineus | 1.25 | 0.05 | 0.625 | 0.0125 | 0.75 | additive |
| F. deltoidea | 0.63 | 0.05 | 0.625 | 0.0125 | 0.75 | additive | |
| P. gingivalis | O. stamineus | 1.25 | 0.05 | 0.625 | 0.0125 | 0.75 | additive |
| F. deltoidea | 1.25 | 0.05 | 0.625 | 0.0125 | 0.75 | additive | |
| F. nucleatum | O. stamineus | 1.25 | 0.05 | 0.625 | 0.0125 | 0.75 | additive |
| F. deltoidea | 0.63 | 0.05 | 0.625 | 0.0125 | 0.75 | additive | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Azizan, N.; Mohd Said, S.; Zainal Abidin, Z.; Jantan, I. Composition and Antibacterial Activity of the Essential Oils of Orthosiphon stamineus Benth and Ficus deltoidea Jack against Pathogenic Oral Bacteria. Molecules 2017, 22, 2135. https://doi.org/10.3390/molecules22122135
Azizan N, Mohd Said S, Zainal Abidin Z, Jantan I. Composition and Antibacterial Activity of the Essential Oils of Orthosiphon stamineus Benth and Ficus deltoidea Jack against Pathogenic Oral Bacteria. Molecules. 2017; 22(12):2135. https://doi.org/10.3390/molecules22122135
Chicago/Turabian StyleAzizan, Nuramirah, Shahida Mohd Said, Zamirah Zainal Abidin, and Ibrahim Jantan. 2017. "Composition and Antibacterial Activity of the Essential Oils of Orthosiphon stamineus Benth and Ficus deltoidea Jack against Pathogenic Oral Bacteria" Molecules 22, no. 12: 2135. https://doi.org/10.3390/molecules22122135
APA StyleAzizan, N., Mohd Said, S., Zainal Abidin, Z., & Jantan, I. (2017). Composition and Antibacterial Activity of the Essential Oils of Orthosiphon stamineus Benth and Ficus deltoidea Jack against Pathogenic Oral Bacteria. Molecules, 22(12), 2135. https://doi.org/10.3390/molecules22122135





