Optimization of Bioactive Polyphenols Extraction from Picea Mariana Bark
Abstract
:1. Introduction
2. Results and Discussion
2.1. Effects of Extraction Parameters on Multiple Response Factors
2.1.1. Extractable Matter Yields
2.1.2. Phenolic, Proanthocyanidin Contents and Antioxidant Capacity
2.1.3. Other Phytochemicals
2.2. High Performance Liquid Chromatography Fingerprint and Chemometric Analysis of Black Spruce Bark Extract Low Molecular Weight Phenolic Compounds
2.3. Isolation, Identification and Quantification of Low Molecular Weight Phenolic Constituents of Black Spruce Bark Extract
3. Materials and Methods
3.1. Plant Material
3.2. Factorial Design for Multiple Extraction Experiment
3.3. Extraction Procedure for Optimization Experiment
3.4. Isolation and Characterization of the Hot Water Extract Constituents
3.5. Phytochemicals Assessments of the Extracts
3.5.1. Total Phenol Content
3.5.2. Proanthocyanidin Content
3.5.3. Total Sugar Content
3.5.4. DPPH Assay
3.6. HPLC-DAD Analysis
3.7. Chemometric Analysis
3.8. HRMS and NMR Analysis
3.9. Statistical Analysis
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Garcia-Perez, M.E.; Royer, M.; Herbette, G.; Desjardins, Y.; Pouliot, R.; Stevanovic, T. Picea mariana bark: A new source of trans-resveratrol and other bioactive polyphenols. Food Chem. 2012, 135, 1173–1182. [Google Scholar] [CrossRef] [PubMed]
- Francezon, N.; Stevanovic, T. Integrated process for the production of natural extracts from black spruce bark. Ind. Crops Prod. 2017, 108, 348–354. [Google Scholar] [CrossRef]
- Counet, C.; Callemien, D.; Collin, S. Chocolate and cocoa: New sources of trans-resveratrol and trans-piceid. Food Chem. 2006, 98, 649–657. [Google Scholar] [CrossRef]
- Wang, Y.; Catana, F.; Yang, Y.; Roderick, R.; Van Breemen, R.B. An LC-MS method for analyzing total resveratrol in grape juice, cranberry juice, and in wine. J. Agric. Food Chem. 2002, 50, 431–435. [Google Scholar] [CrossRef] [PubMed]
- Manners, G.D.; Swan, E.P. Stilbenes in the barks of five canadian Picea species. Phytochemistry 1971, 10, 607–610. [Google Scholar] [CrossRef]
- Roupe, K.A.; Remsberg, C.M.; Yáñez, J.A.; Davies, N.M. Pharmacometrics of stilbenes: Seguing towards the clinic. Curr. Clin. Pharmacol. 2006, 1, 81–101. [Google Scholar] [CrossRef] [PubMed]
- Frémont, L. Biological effects of resveratrol. Life Sci. 2000, 66, 663–673. [Google Scholar] [CrossRef]
- Clinical trial, C. A Single Dose Study of Epn-07 by Oral Gavage in Rats with a 14 Day Recovery Period (up and down Procedure), Confidential; Charles River Laboratories: Edinburgh, UK, 2012. [Google Scholar]
- Chemat, F.; Vian, M.A.; Cravotto, G. Green extraction of natural products: Concept and principles. Int. J. Mol. Sci. 2012, 13, 8615–8627. [Google Scholar] [CrossRef] [PubMed]
- Hartonen, K.; Riekkola, M.-L. Chapter 2—Water as the first choice green solvent. In The Application of Green Solvents in Separation Processes; Pena-Pereira, F., Tobiszewski, M., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 19–55. ISBN 9780128052976. [Google Scholar]
- Geoffroy, T.R.; Fortin, Y.; Stevanovic, T. Hot-water extraction optimization of sugar maple (Acer saccharum Marsh.) and red maple (Acer rubrum L.) bark applying principal component analysis. J. Wood Chem. Technol. 2017, 1–12. [Google Scholar] [CrossRef]
- Royer, M.; Prado, M.; García-Pérez, M.E.; Diouf, P.N.; Stevanovic, T. Study of nutraceutical, nutricosmetics and cosmeceutical potentials of polyphenolic bark extracts from canadian forest species. PharmaNutrition 2013, 1, 158–167. [Google Scholar] [CrossRef]
- Stevanovic, T.; Diouf, P.N.; Garcia-Perez, M.E. Bioactive polyphenols from healthy diets and forest biomass. Curr. Nutr. Food Sci. 2009, 5, 264–295. [Google Scholar] [CrossRef]
- Garcia-Perez, M.E.; Royer, M.; Duque-Fernandez, A.; Diouf, P.N.; Stevanovic, T.; Pouliot, R. Antioxidant, toxicological and antiproliferative properties of canadian polyphenolic extracts on normal and psoriatic keratinocytes. J. Ethnopharmacol. 2010, 132, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Schneider, M.J.; Montali, J.A.; Zen, D.; Stanton, C.E. Alkaloids of Picea. J. Nat. Prod. 1991, 54, 905–909. [Google Scholar] [CrossRef]
- Kemppainen, K.; Siika-Aho, M.; Pattathil, S.; Giovando, S.; Kruus, K. Spruce bark as an industrial source of condensed tannins and non-cellulosic sugars. Ind. Crops Prod. 2014, 52, 158–168. [Google Scholar] [CrossRef]
- Giannoccaro, E.; Wang, Y.J.; Chen, P. Effects of solvent, temperature, time, solvent-to-sample ratio, sample size, and defatting on the extraction of soluble sugars in soybean. J. Food Sci. 2006, 71. [Google Scholar] [CrossRef]
- Elleuch, M.; Bedigian, D.; Roiseux, O.; Besbes, S.; Blecker, C.; Attia, H. Dietary fibre and fibre-rich by-products of food processing: Characterisation, technological functionality and commercial applications: A review. Food Chem. 2011, 124, 411–421. [Google Scholar] [CrossRef]
- Xu, X.; Jiang, J.; Liang, Y.; Yi, L.; Cheng, J. Chemical fingerprint analysis for quality control of Fructus Aurantii Immaturus based on HPLC-DAD combined with chemometric methods. Anal. Methods 2010, 2, 2002–2010. [Google Scholar] [CrossRef]
- Meda, N.; Rott, M.; Suwal, S.; Poubelle, P.; Stevanovic, T. Chemometrics-based approach to analysis of phenolic fingerprints of red and sugar maple bud extracts. Austin Biochem. 2017, 2, 1009. [Google Scholar]
- Pan, H.; Lundgren, L.N. Phenolic extractives from root bark of Picea abies. Phytochemistry 1995, 39, 1423–1428. [Google Scholar] [CrossRef]
- Carnachan, S.M.; Harris, P.J. Ferulic acid is bound to the primary cell walls of all gymnosperm families. Biochem. Syst. Ecol. 2000, 28, 865–879. [Google Scholar] [CrossRef]
- Li, S.H.; Niu, X.M.; Zahn, S.; Gershenzon, J.; Weston, J.; Schneider, B. Diastereomeric stilbene glucoside dimers from the bark of norway spruce (Picea abies). Phytochemistry 2008, 69, 772–782. [Google Scholar] [CrossRef] [PubMed]
- Gabaston, J.; Richard, T.; Biais, B.; Waffo-Teguo, P.; Pedrot, E.; Jourdes, M.; Corio-Costet, M.-F.; Mérillon, J.-M. Stilbenes from common spruce (Picea abies) bark as natural antifungal agent against downy mildew (Plasmopara viticola). Ind. Crops Prod. 2017, 103, 267–273. [Google Scholar] [CrossRef]
- Waffo-Teguo, P.; Lee, D.; Cuendet, M.; Mérillon, J.-M.; Pezzuto, J.M.; Kinghorn, A.D. Two new stilbene dimer glucosides from grape (Vitis vinifera) cell cultures. J. Nat. Prod. 2001, 64, 136–138. [Google Scholar] [CrossRef] [PubMed]
- Huang, K.-S.; Wang, Y.-H.; Li, R.-L.; Lin, M. Five new stilbene dimers from the lianas of Gnetum hainanense. J. Nat. Prod. 2000, 63, 86–89. [Google Scholar] [CrossRef] [PubMed]
- Yao, C.-S.; Lin, M. Bioactive stilbene dimers from Gnetum cleistostachyum. Nat. Prod. Res. 2005, 19, 443–448. [Google Scholar] [CrossRef] [PubMed]
- Rivière, C.; Pawlus, A.D.; Mérillon, J.-M. Natural stilbenoids: Distribution in the plant kingdom and chemotaxonomic interest in Vitaceae. Nat. Prod. Rep. 2012, 29, 1317–1333. [Google Scholar] [CrossRef] [PubMed]
- Mannila, E.; Talvitie, A. Stilbenes from Picea abies bark. Phytochemistry 1992, 31, 3288–3289. [Google Scholar] [CrossRef]
- Yao, C.-S.; Lin, M.; Liu, X.; Wang, Y. Stilbenes from Gnetum cleistostachyum. Acta Chim. Sin. 2003, 61, 1331–1334. [Google Scholar]
- Liang, Y.; Zhu, J.; Huang, H.; Xiang, D.; Li, Y.; Zhang, D.; Li, J.; Wang, Y.; Jin, H.; Jiang, G. SESN2/sestrin 2 induction-mediated autophagy and inhibitory effect of isorhapontigenin (ISO) on human bladder cancers. Autophagy 2016, 12, 1229–1239. [Google Scholar] [CrossRef] [PubMed]
- Li, H.-L.; Wang, A.-B.; Huang, Y.; Liu, D.-P.; Wei, C.; Williams, G.M.; Zhang, C.-N.; Liu, G.; Liu, Y.-Q.; Hao, D.-L. Isorhapontigenin, a new resveratrol analog, attenuates cardiac hypertrophy via blocking signaling transduction pathways. Free Radic. Biol. Med. 2005, 38, 243–257. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.N.; Liu, G.T. Effect of isorhapontigenin on respiratory burst of rat neutrophils. Phytomedicine 2002, 9, 734–738. [Google Scholar] [CrossRef] [PubMed]
- Diouf, P.N.; Tibirna, C.M.; García-Pérez, M.-E.; Royer, M.; Dubé, P.; Stevanovic, T. Structural elucidation of condensed tannin from Picea mariana bark. J. Biomater. Nanobiotechnol. 2013, 4, 1–8. [Google Scholar] [CrossRef]
- Waffo-Téguo, P.; Hawthorne, M.E.; Cuendet, M.; Mérillon, J.-M.; Kinghorn, A.D.; Pezzuto, J.M.; Mehta, R.G. Potential cancer-chemopreventive activities of wine stilbenoids and flavans extracted from grape (Vitis vinifera) cell cultures. Nutr. Cancer 2001, 40, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Piotrowska, H.; Kucinska, M.; Murias, M. Biological activity of piceatannol: Leaving the shadow of resveratrol. Mutat. Res. Rev. Mutat. Res. 2012, 750, 60–82. [Google Scholar] [CrossRef] [PubMed]
- Du, Q.-H.; Peng, C.; Zhang, H. Polydatin: A review of pharmacology and pharmacokinetics. Pharm. Biol. 2013, 51, 1347–1354. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zhang, D.-Q.; Liao, Z.; Wang, B.; Gong, S.; Wang, C.; Zhang, M.-Z.; Wang, G.-H.; Cai, H.; Liao, F.-F. Anti-oxidant polydatin (piceid) protects against substantia nigral motor degeneration in multiple rodent models of Parkinson’s disease. Mol. Neurodegener. 2015, 10, 4. [Google Scholar] [CrossRef] [PubMed]
- Diouf, P.N.; Stevanovic, T.; Cloutier, A. Study on chemical composition, antioxidant and anti-inflammatory activities of hot water extract from picea mariana bark and its proanthocyanidin-rich fractions. Food Chem. 2009, 113, 897–902. [Google Scholar] [CrossRef]
- St-Pierre, F.; Achim, A.; Stevanovic, T. Composition of ethanolic extracts of wood and bark from Acer saccharum and Betula alleghaniensis trees of different vigor classes. Ind. Crops Prod. 2013, 41, 179–187. [Google Scholar] [CrossRef]
- Porter, L.J.; Hrstich, L.N.; Chan, B.G. The conversion of procyanidins and prodelphinidins to cyanidin and delphinidin. Phytochemistry 1985, 25, 223–230. [Google Scholar] [CrossRef]
- Albalasmeh, A.A.; Berhe, A.A.; Ghezzehei, T.A. A new method for rapid determination of carbohydrate and total carbon concentrations using UV spectrophotometry. Carbohydr. Polym. 2013, 97, 253–261. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Henry, G.E.; Seeram, N.P. Identification and bioactivities of resveratrol oligomers and flavonoids from Carex folliculata seeds. J. Agric. Food Chem. 2009, 57, 7282–7287. [Google Scholar] [CrossRef] [PubMed]
- Struijs, K.; Vincken, J.-P.; Verhoef, R.; van Oostveen-van Casteren, W.H.; Voragen, A.G.; Gruppen, H. The flavonoid herbacetin diglucoside as a constituent of the lignan macromolecule from flaxseed hulls. Phytochemistry 2007, 68, 1227–1235. [Google Scholar] [CrossRef] [PubMed]
- Mulat, D.G.; Latva-Mäenpää, H.; Koskela, H.; Saranpää, P.; Wähälä, K. Rapid chemical characterisation of stilbenes in the root bark of Norway spruce by off-line HPLC/DAD–NMR. Phytochem. Anal. 2014, 25, 529–536. [Google Scholar] [CrossRef] [PubMed]
- Commodari, F.; Khiat, A.; Ibrahimi, S.; Brizius, A.R.; Kalkstein, N. Comparison of the phytoestrogen trans-resveratrol (3,4′,5-trihydroxystilbene) structures from x-ray diffraction and solution NMR. Magn. Reson. Chem. 2005, 43, 567–572. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds 1–8 are available from the authors. |
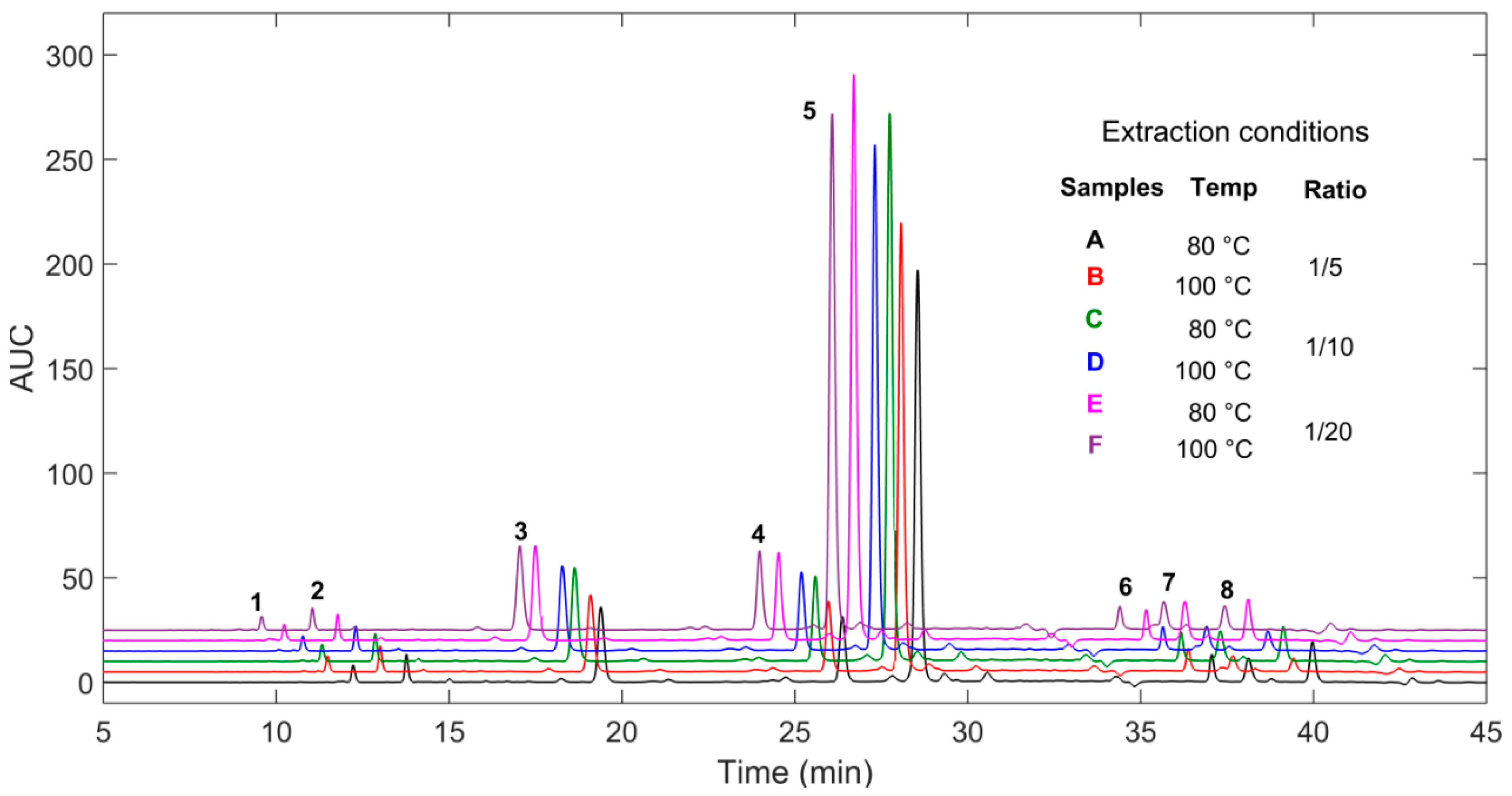

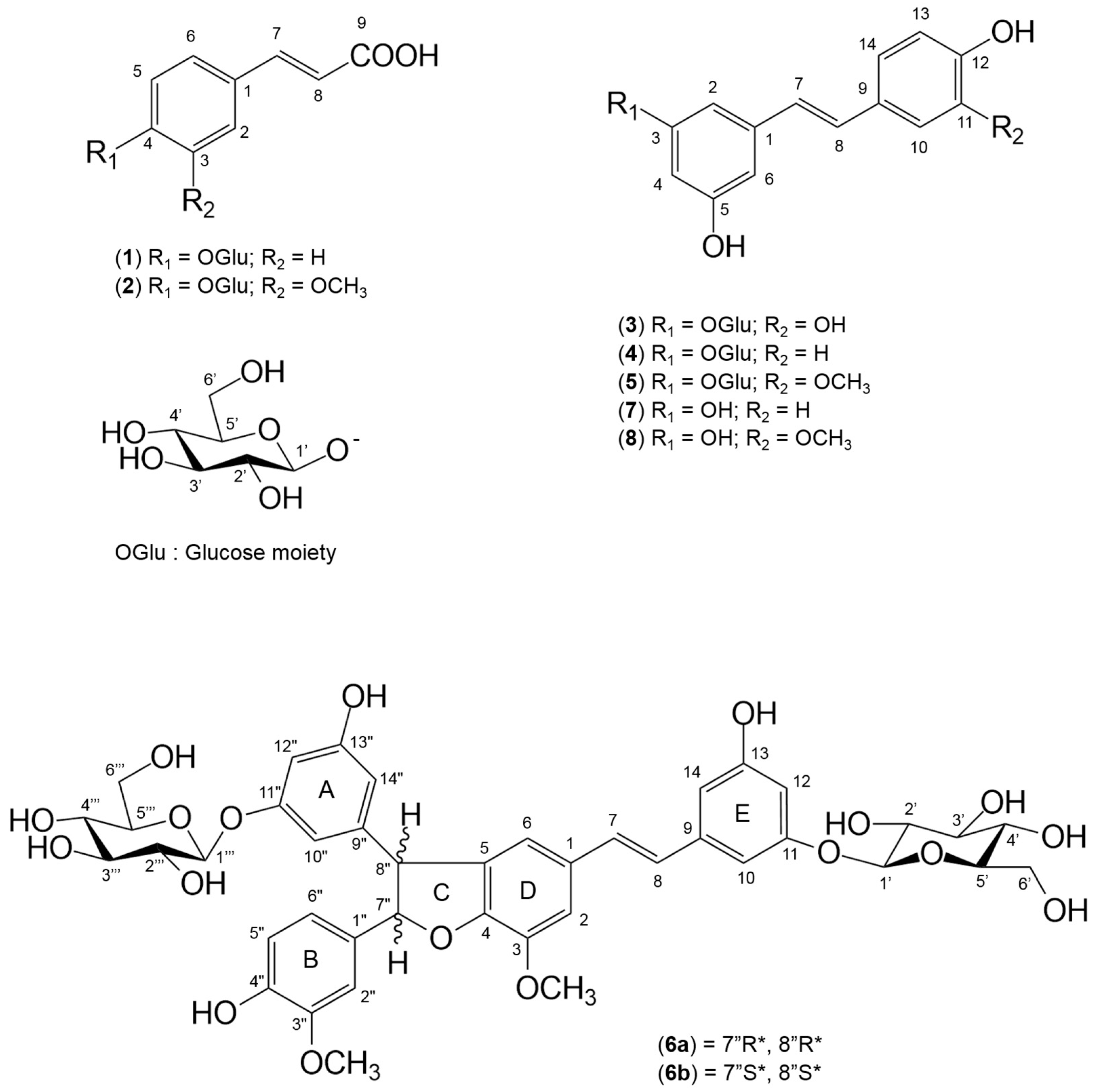
| Experimental Design | Response Factors | |||||||
|---|---|---|---|---|---|---|---|---|
| Extract Name | Time (min) | Temperature (°C) | Ratio (mg/mL) | Yield (%) | Phen (mg GAE/g) | PA (mg CyE/g) | Sugar (mg ARGF/g) | Antiox (µmol TE/g) |
| A | 60 | 80 | 200 | 11.3 ± 0.4 | 442 ± 25 | 224 ± 8 | 662 ± 77 | 1021 ± 54 |
| B | 60 | 100 | 200 | 14.3 ± 0.7 | 471 ± 6 | 252 ± 2 | 581 ± 3 | 1082 ± 29 |
| C | 60 | 80 | 100 | 14.5 ± 0.4 | 469 ± 5 | 236 ± 15 | 543 ± 49 | 1087 ± 19 |
| D | 60 | 100 | 100 | 17.7 ± 0.3 | 472 ± 27 | 254 ± 8 | 574 ± 7 | 1071 ± 14 |
| E | 60 | 80 | 50 | 15.6 ± 0.5 | 485 ± 4 | 252 ± 16 | 604 ± 24 | 1007 ± 101 |
| F | 60 | 100 | 50 | 19.3 ± 0.2 | 468 ± 2 | 264 ± 1 | 556 ± 21 | 1091 ± 6 |
| G | 90 | 80 | 200 | 11.5 ± 0.5 | 456 ± 5 | 245 ± 1 | 608 ± 71 | 1029 ± 52 |
| H | 90 | 100 | 200 | 14.6 ± 0.2 | 426 ± 15 | 232 ± 20 | 600 ± 13 | 1017 ± 64 |
| I | 90 | 80 | 100 | 14.5 ± 0.3 | 486 ± 19 | 249 ± 14 | 606 ± 9 | 1079 ± 9 |
| J | 90 | 100 | 100 | 18.1 ± 0.4 | 438 ± 3 | 232 ± 1 | 555 ± 25 | 1047 ± 30 |
| K | 90 | 80 | 50 | 16.1 ± 0.2 | 472 ± 3 | 246 ± 34 | 538 ± 24 | 1061 ± 11 |
| L | 90 | 100 | 50 | 19.4 ± 0.6 | 459 ± 10 | 243 ± 3 | 573 ± 37 | 991 ± 56 |
| M | 120 | 80 | 200 | 11.2 ± 0.2 | 464 ± 45 | 245 ± 3 | 571 ± 23 | 901 ± 174 |
| N | 120 | 100 | 200 | 14.7 ± 0.3 | 399 ± 9 | 220 ± 15 | 637 ± 4 | 1026 ± 62 |
| O | 120 | 80 | 100 | 15.1 ± 0.4 | 467 ± 35 | 235 ± 15 | 617 ± 21 | 962 ± 87 |
| P | 120 | 100 | 100 | 18.8 ± 0.4 | 447 ± 1 | 238 ± 4 | 673 ± 6 | 1049 ± 27 |
| Q | 120 | 80 | 50 | 16.5 ± 0.1 | 445 ± 5 | 244 ± 6 | 579 ± 7 | 1086 ± 17 |
| R | 120 | 100 | 50 | 20.1 ± 0.7 | 502 ± 56 | 236 ± 17 | 534 ± 11 | 1075 ± 20 |
| Identification | Quantification | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Comp. | Ret. Time (min) | λmax (nm) | Exact Mass | Formula | Suggested Compound | Regression Equation | R2 | Linear Range ug/mL | LOD | LOQ | Concentration Range mg/100 g Dry Extracts |
| 1 | 12.4 | 290 | 326.0994 | C15H18O8 | trans-p-coumaric acid β-d-glucopyranoside | y = 2140.2x + 8.2609 | 0.9997 | 5–500 | 0.1 | 0.4 | 160–265 |
| 2 | 13.9 | 290, 315 | 356.1093 | C16H20O9 | trans-ferulic acid β-d-glucopyranoside | y = 1486.4x − 60.765 | 0.9987 | 5–1000 | 0.4 | 1.3 | 894–1073 |
| 3 | 20.2 | 325, 305 | 406.1243 | C20H21O9 | trans-astringin | y = 2284x − 441.91 | 0.999 | 5–1000 | 3.8 | 11.4 | 2272–4613 |
| 4 | 26.8 | 305, 320 | 390.1303 | C20H22O8 | trans-piceid | y = 3384.1x − 528.13 | 0.9971 | 5–500 | 0.2 | 0.6 | 1805–3094 |
| 5 | 29.4 | 325, 303, 290 | 420.1416 | C21H24O9 | trans-isorhapontin | y = 2944.6x − 265.82 | 0.9979 | 1–1000 | 0.6 | 1.9 | 4256–11971 |
| 6 | 37.3 | 330, 310, 283 | 838.2684 | C42H46O18 | piceaside O and P | y = 1056.4x − 329.29 | 0.9933 | 1–1000 | 0.4 | 1.2 | 3508–4853 |
| 7 | 38.5 | 305, 320 | 228.0796 | C14H12O3 | trans-resveratrol | y = 7520.7x + 43.49 | 0.9914 | 1–500 | 0.2 | 0.6 | 53–302 |
| 8 | 40.3 | 325, 303, 290 | 258.0892 | C15H14O4 | trans-isorhapontigenin | y = 2062.4x − 415.08 | 0.9979 | 5–1000 | 0.3 | 0.9 | 2257–3654 |
| Position | δH (6a) a | δC (6a) b | δH (6b) a | δC (6b) b | HMBC (6a and 6b) |
|---|---|---|---|---|---|
| 1 | 139.7 (d) | 139.7 (d) | |||
| 2 | 7.11 (br s) | 110.5 (d) | 7.11 (br s) | 110.4 (d) | C-3, 7, 6, 4 |
| 3 | 144.3 (s) | 144.3 (s) | |||
| 4 | 148.0 (d) | 148.0 (d) | |||
| 5 | 131.5 (d) | 131.5 (d) | |||
| 6 | 6.83 (br s) | 115.6 (d) | 6.81 (br s) | 115.5 (d) | |
| 7 | 7.03 (d, 16.3) | 128.6 (d) | 7.03 (d, 16.3) | 128.6 (d) | C-8, 1, 2, 6 |
| 8 | 6.87 (d, 16) | 126.2 (d) | 6.87 (d, 16) | 126.2 (d) | C-7, 9, 1, 10, 14 |
| 9 | 131.9 (d) | 131.8 (d) | |||
| 10 | 6.78 (overlap) | 105.7 (d) | 6.78 (overlap) | 105.6 (d) | C-11, 8, 9, 12, 14 |
| 11 | 159.0 (s) | 159.0 (s) | |||
| 12 | 6.45 (t, 2.2) | 102.9 (s) | 6.45 (t, 2.2) | 102.9 (s) | C-10, 14, 13, 11 |
| 13 | 158.1 (s) | 158.1 (s) | |||
| 14 | 6.62 (t, 1.7) | 107.0 (s) | 6.62 (t, 1.7) | 107.0 (s) | C-13, 12, 8, 10 |
| 1′ | 4.87 (overlap) | 100.4 (s) | 4.87 (overlap) | 100.4 (s) | C-11 |
| 2′ | 3.37–3.51 (overlap) | 73.3 (d) | 3.37–3.51 (overlap) | 73.4 (d) | |
| 3′ | 3.37–3.51 (overlap) | 76.5 (s) | 3.37–3.51 (overlap) | 76.5 (s) | |
| 4′ | 3.37–3.51 (overlap) | 70.0 (d) | 3.37–3.51 (overlap) | 70.0 (d) | |
| 5′ | 3.37–3.51 (overlap) | 76.6 (d) | 3.37–3.51 (overlap) | 76.6 (d) | |
| 6′ | 3.79 (dd, 12.1, 2.3) | 60.9 (d) | 3.79 (dd, 12.1, 2.3) | 60.7 (d) | C-4′, 5′ |
| 1″ | 131.6 (d) | 131.6 (d) | |||
| 2″ | 6.93 (d, 6.9) | 109.4 (d) | 6.93 (d, 6.9) | 109.3 (d) | C-6″, 4″, 7″, 3″ 1″ |
| 3″ | 147.7 (s) | 147.7 (s) | |||
| 4″ | 146.5 (d) | 146.5 (d) | |||
| 5″ | 6.79 (overlap) | 114.8 (s) | 6.79 (overlap) | 114.8 (s) | C-1″, 3″ |
| 6″ | 6.80 (overlap) | 119.0 (d) | 6.80 (overlap) | 118.8 (d) | C-7″, 1″ |
| 7″ | 5.43 (d, 8.8) | 94.0 (d) | 5.44 (d, 8.6) | 94.0 (d) | C-8″, 2″, 6″, 1″, 3″, 9″ |
| 8″ | 4.51 (d, 8.8) | 57.6 (d) | 4.50 (d, 8.6) | 57.6 (d) | C-7″, 9″, 5, 10″, 14″ |
| 9″ | 143.8 (d) | 143.6 (d) | |||
| 10″ | 6.42 (ddd, 3.8, 2.2, 1.5) | 107.6 (d) | 6.42 (ddd, 3.8, 2.2, 1.5) | 107.4 (d) | C-11″, 12″, 8″, 14″,9″ |
| 11″ | 159.1 (s) | 159.1 (s) | |||
| 12″ | 6.50 (t, 2.2) | 102.5 (d) | 6.50 (t, 2.2) | 102.3 (d) | C-13″, 11″, 10″, 14″ |
| 13″ | 158.5 (d) | 158.5 (d) | |||
| 14″ | 6.34 (dd, 2.2, 1.4) | 109.0 (d) | 6.31 (dd, 2.2, 1.4) | 108.9 (d) | C-13″, 12″, 10″, 8″ |
| 1′′′ | 4.79 (d, 7,5) | 100.8 (d) | 4.90 (d, 7.6) | 100.9 (d) | C-11″ |
| 2′′′ | 3.37–3.51 (overlap) | 73.5 (s) | 3.37–3.51 (overlap) | 73.5 (s) | |
| 3′′′ | 3.37–3.51 (overlap) | 76.5 (d) | 3.37–3.51 (overlap) | 76.5 (d) | |
| 4′′′ | 3.37–3.51 (overlap) | 69.7 (d) | 3.37–3.51 (overlap) | 69.6 (d) | |
| 5′′′ | 3.37–3.51 (overlap) | 76.8 (d) | 3.37–3.51 (overlap) | 76.8 (d) | |
| 6′′′ | 3.93 (dd, 12.1, 2.1) | 61.2 (d) | 3.91 (dd, 12.1, 2.1) | 61.2 (d) | C-4″, 5″ |
| OCH3 | 3.95 (s) | 55.4 (s) | 3.95 (s) | 55.4 (s) | C-3 |
| OCH3″ | 3.82 (s) | 55.0 (d) | 3.81 (s) | 55.0 (d) | C-3″ |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Francezon, N.; Meda, N.-S.-B.R.; Stevanovic, T. Optimization of Bioactive Polyphenols Extraction from Picea Mariana Bark. Molecules 2017, 22, 2118. https://doi.org/10.3390/molecules22122118
Francezon N, Meda N-S-BR, Stevanovic T. Optimization of Bioactive Polyphenols Extraction from Picea Mariana Bark. Molecules. 2017; 22(12):2118. https://doi.org/10.3390/molecules22122118
Chicago/Turabian StyleFrancezon, Nellie, Naamwin-So-Bâwfu Romaric Meda, and Tatjana Stevanovic. 2017. "Optimization of Bioactive Polyphenols Extraction from Picea Mariana Bark" Molecules 22, no. 12: 2118. https://doi.org/10.3390/molecules22122118
APA StyleFrancezon, N., Meda, N.-S.-B. R., & Stevanovic, T. (2017). Optimization of Bioactive Polyphenols Extraction from Picea Mariana Bark. Molecules, 22(12), 2118. https://doi.org/10.3390/molecules22122118






